Submitted:
15 January 2026
Posted:
16 January 2026
You are already at the latest version
Abstract
Keywords:
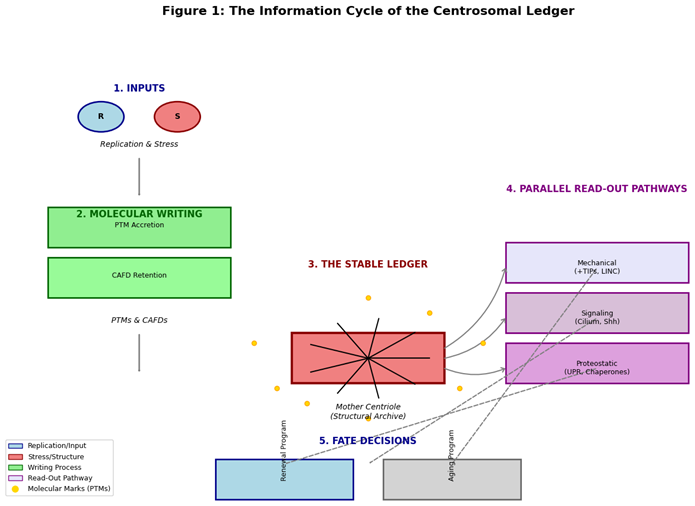
1. Introduction: Beyond the Microtubule Organizer
2. The Molecular Syntax of the Ledger: Encoding Time and Experience
2.1. Post-Translational Modifications as Chronological and Damage Marks
- Neutral PTMs (Replicative Tally): Enzymatic modifications like polyglutamylation accumulate predictably with each cell cycle on the mother centriole. They act as a maturity index, marking functional readiness for roles like basal body formation (Bré et al., 2021). The level of polyglutamylation (PolyG) can be conceptualized as a function of division number (n):where ΔG represents the addition per cycle.PolyG(n) ≈ PolyG(n-1) + ΔG
- Pathological PTMs (Stress Log): Stochastic, damage-associated modifications create a damage index. This includes oxidation of scaffold proteins (e.g., SAS-6), glycation of long-lived proteins (e.g., CEP135), and ubiquitination marking misfolded clients (Wang et al., 2021). These correlate with functional decline.
| Modification Type | Example | Proposed Information Encoded | Functional Consequence |
|---|---|---|---|
| Neutral PTM | Polyglutamylation | Number of successful divisions (Maturity) | Stabilization; recruitment of specific effectors (e.g., ciliary assembly proteins) |
| Neutral PTM | Acetylation (K40) | Age of microtubule structure | Reduced dynamics, increased mechanical resistance |
| Pathological PTM | Cysteine Oxidation | History of oxidative stress | Weakened structural integrity, errors in duplication |
| Pathological PTM | Advanced Glycation End-products (AGEs) | Cumulative metabolic stress | Loss of protein function, aggregation propensity |
| Protein Retention | CAFDs (e.g., YAP/TAZ) | Past signaling context (e.g., Hippo pathway activity) | Altered transcriptional preparedness upon release |
2.2. Proteomic Composition as a Snapshot of State
3. Decoding the Ledger: Cellular Read-Out Mechanisms
3.1. Mechanical Read-Out
3.2. Signaling Read-Out
3.3. Proteostatic Read-Out
4. The Ledger in the Hierarchy of Biological Clocks
- Level 1: Fast Oscillators. Circadian clock proteins (e.g., PER2) localize to centrosomes, suggesting the ledger integrates daily metabolic rhythms with long-term age data (Wils et al., 2021).
- Level 2: Replicative Clocks. The ledger interacts with telomeric and epigenetic clocks. Centriole dysfunction induces genomic instability, accelerating telomere attrition. Conversely, epigenetic changes may lock in a “senescent” interpretation of ledger data.
- Level 3: Systemic Aging. In stem cells, asymmetric inheritance of a “young” vs. “old” ledger determines self-renewal vs. differentiation balance. Age-related decline in this process depletes regenerative niches, driving tissue aging (Geiger et al., 2013).
5. Comparative Cell Biology: Ledger Adaptations
- Germline: Employs strict asymmetric inheritance and stringent quality control to reset the ledger, minimizing intergenerational damage transfer (Fishman et al., 2018).
- Somatic Stem Cells: Exhibits “leaky” asymmetry, where imperfect segregation gradually increases damage load in the stem pool, contributing to niche aging.
- Differentiated Cells: The ledger is “paused”; the centriole becomes a static basal body, with age-related damage manifesting as ciliopathies.
- Cancer Cells: The ledger is “hijacked”—amplification and structural distortion disrupt normal timekeeping, enabling unchecked proliferation at the cost of genomic instability (Marteil et al., 2018).
| Cell Type | Ledger State | Inheritance Mode | Primary Time-Related Output |
|---|---|---|---|
| Germ Cell | Reset, Immortal | Strictly Asymmetric | Fertility, generational continuity |
| Somatic Stem Cell | Aging, Leaky | Asymmetric (with error) | Tissue homeostasis, niche depletion |
| Differentiated Cell | Paused, Static | Symmetric / Non-dividing | Sensory function, age-related ciliopathies |
| Cancer Cell | Corrupted, Amplified | Aberrant, Symmetric | Uncontrolled proliferation, genomic instability |
6. Evolutionary Origins: Why the Centriole?
7. Experimental Framework and Falsifiability
- Prediction: Targeted reversal of centriolar PTMs (e.g., deacetylation, redox repair) in aged stem cells will rejuvenate function.
- Prediction: Microinjection of in vitro “aged” (glycated/oxidized) centrioles into young cells will induce senescence markers.
- Prediction: Comparative biology will show that centriole-less organisms (plants) rely on fundamentally different primary drivers for stem cell aging.
8. Philosophical and Practical Implications
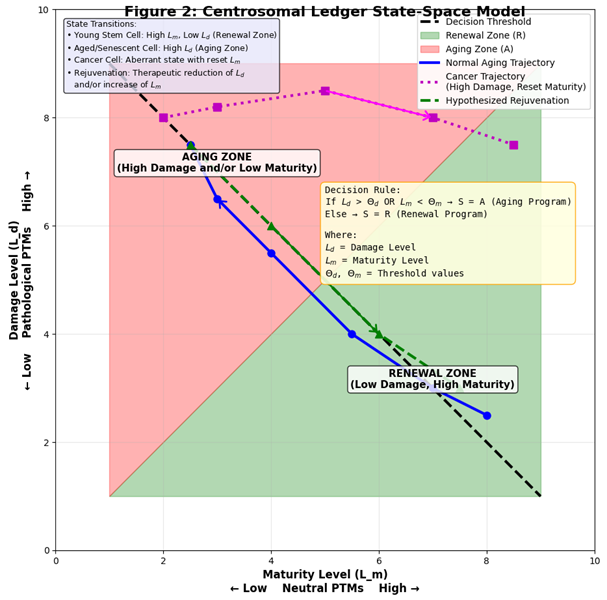
9. The Integrative Centrosomal Ledger Model: A Formal Summary
- L_m = Maturity Level (from neutral PTMs)
- L_d = Damage Level (from pathological PTMs)
- If L_d > Θ_d or L_m < Θ_m --> S = A (Engage Senescence/Apoptosis)
- Else --> S = R (Promote Proliferation/Differentiation)
Conclusions
References
- Bornens, M. The centrosome in cells and organisms. Science 2012, 335(6067), 422–426. [Google Scholar] [CrossRef] [PubMed]
- Bré, M. H.; Redeker, V.; Levilliers, N. Post-translational modifications of tubulin and microtubule stability in cilia and flagella. In The Cytoskeleton: Diverse Roles in Structure and Mechanism; Springer: Cham, 2021; pp. 47–70. [Google Scholar] [CrossRef]
- Breslow, D. K.; Holland, A. J. Mechanism and regulation of centriole and cilium biogenesis. Annual Review of Biochemistry 2019, 88, 691–724. [Google Scholar] [CrossRef] [PubMed]
- Conroy, P. C.; Saladino, C.; Dantas, T. J.; Lalor, P.; Dockery, P.; Morrison, C. G. Centrosome adaptation to DNA damage by 53BP1 recruitment facilitates error-free mitosis. Nature Communications 2022, 13(1), 620. [Google Scholar] [CrossRef]
- Carvalho-Santos, Z.; Azimzadeh, J.; Pereira-Leal, J. B.; Bettencourt-Dias, M. Evolution: Tracing the origins of centrioles, cilia, and flagella. The Journal of Cell Biology 2011, 194(2), 165–175. [Google Scholar] [CrossRef] [PubMed]
- Fishman, E. L.; Jo, K.; Nguyen, Q. P. H.; Kong, D.; Royfman, R.; Cekic, A. R.; …; Avidor-Reiss, T. A novel atypical sperm centriole is functional during human fertilization. Nature Communications 2018, 9(1), 2210. [Google Scholar] [CrossRef] [PubMed]
- Geiger, H.; de Haan, G.; Florian, M. C. The ageing haematopoietic stem cell compartment. Nature Reviews Immunology 2013, 13(5), 376–389. [Google Scholar] [CrossRef] [PubMed]
- Jaba, T. Dasatinib and quercetin: short-term simultaneous administration yields senolytic effect in humans. Issues and Developments in Medicine and Medical Research 2022, Vol. 2, 22–31. [Google Scholar]
- Jakobsen, L.; Vanselow, K.; Skogs, M.; Toyoda, Y.; Lundberg, E.; Poser, I.; …; Andersen, J. S. Novel asymmetrically localizing components of human centrosomes identified by complementary proteomics methods. The EMBO Journal 2011, 30(8), 1520–1535. [Google Scholar] [CrossRef] [PubMed]
- Lomakin, A. J.; Cattin, C. J.; Cuvelier, D.; Alraies, Z.; Molina, M.; Nader, G. P. F.; …; Piel, M. The nucleus acts as a ruler tailoring cell responses to spatial constraints. Science 2015, 348(6232), 560–563. [Google Scholar] [CrossRef] [PubMed]
- Marteil, G.; Guerrero, A.; Vieira, A. F.; de Almeida, B. P.; Machado, P.; Mendonça, S.; …; Bettencourt-Dias, M. Over-elongation of centrioles in cancer promotes centriole amplification and mitotic defects. The EMBO Journal 2018, 37(22), e100303. [Google Scholar] [CrossRef]
- Venkei, Z. G.; Yamashita, Y. M. Emerging mechanisms of asymmetric stem cell division. The Journal of Cell Biology 2018, 217(11), 3785–3795. [Google Scholar] [CrossRef] [PubMed]
- Wang, W. J.; Acehan, D.; Kao, C. H.; Jane, W. N.; Uryu, K.; Tsou, M. F. B. The deubiquitinase USP33 regulates centrosome biogenesis via the SAS-6 protein. Journal of Cell Science 2021, 134(2), jcs247155. [Google Scholar] [CrossRef]
- Wils, L.; Rombauts, F.; van der Horst, G. T. J. The circadian clock and centrioles: Insights into the regulation of cell division. Seminars in Cell & Developmental Biology 2021, 126, 78–84. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




