Submitted:
15 January 2026
Posted:
16 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Patients and Methods
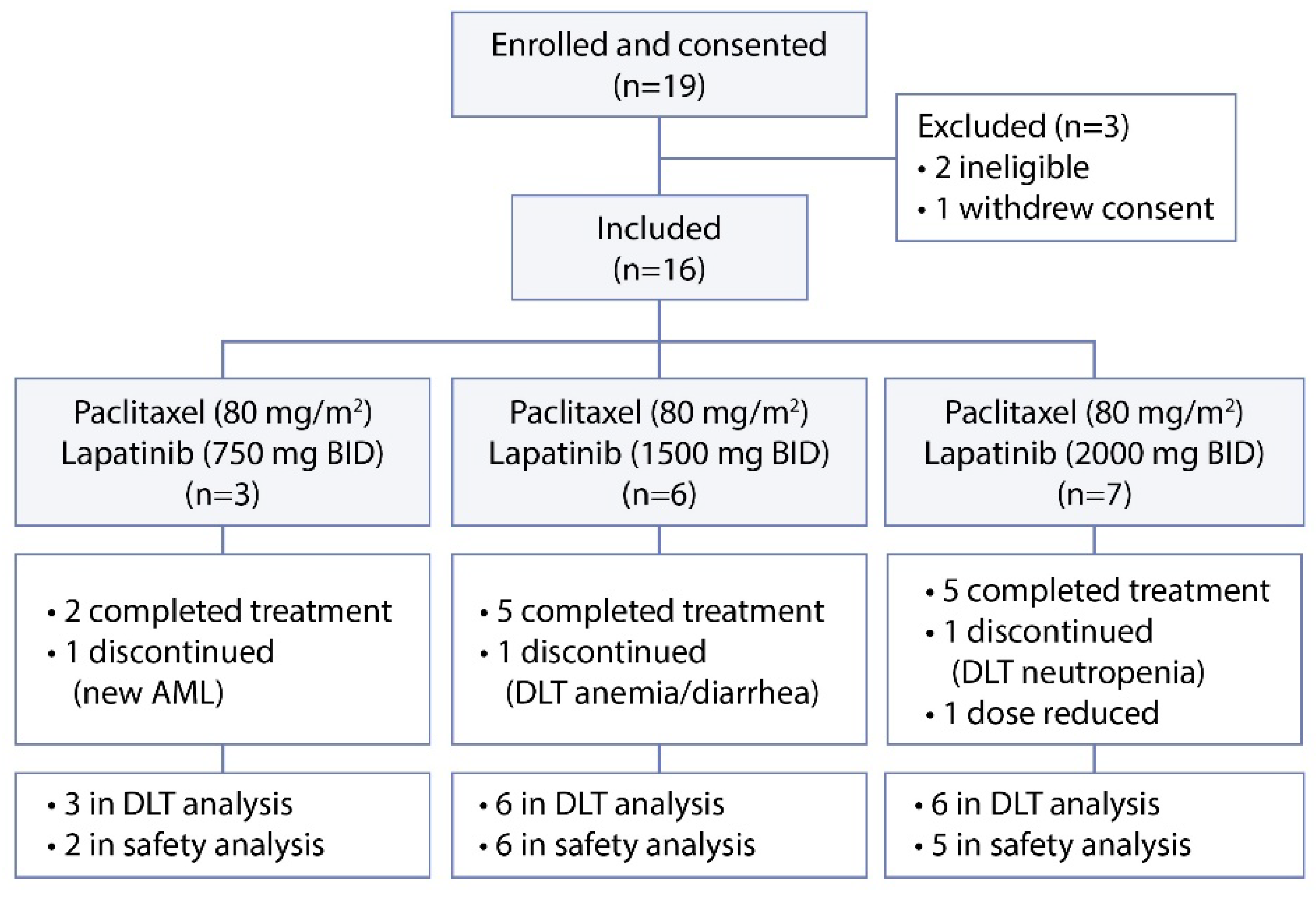
2.1. Study Design
2.2. Patient Population
2.3. Treatment Plan
2.4. Assessments
2.5. Statistical Methods
2.6. Plasma Drug Concentrations
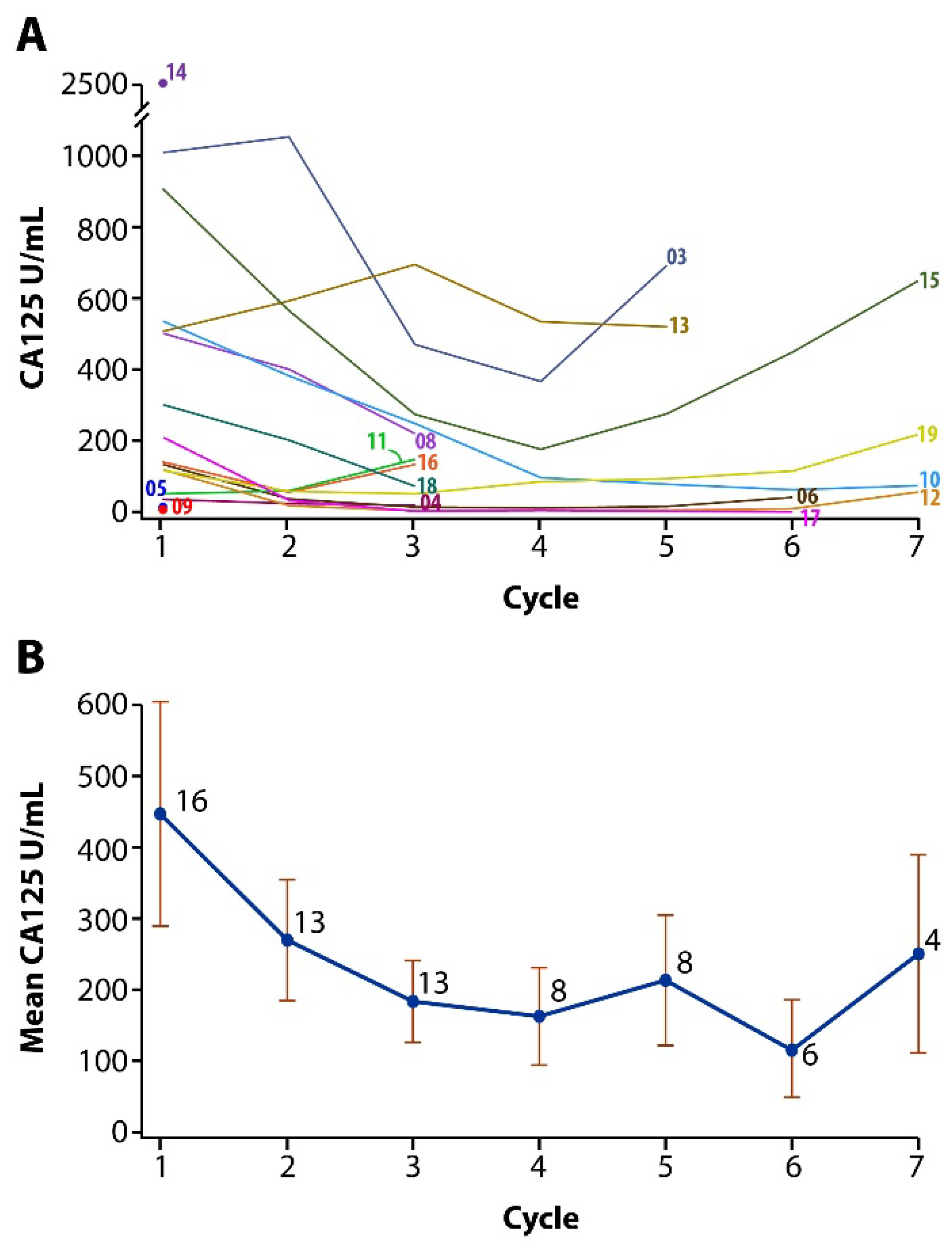
2.7. Plasma CA125 Concentrations
3. Results
3.1. Patient Characteristics
3.2. Safety and Tolerability
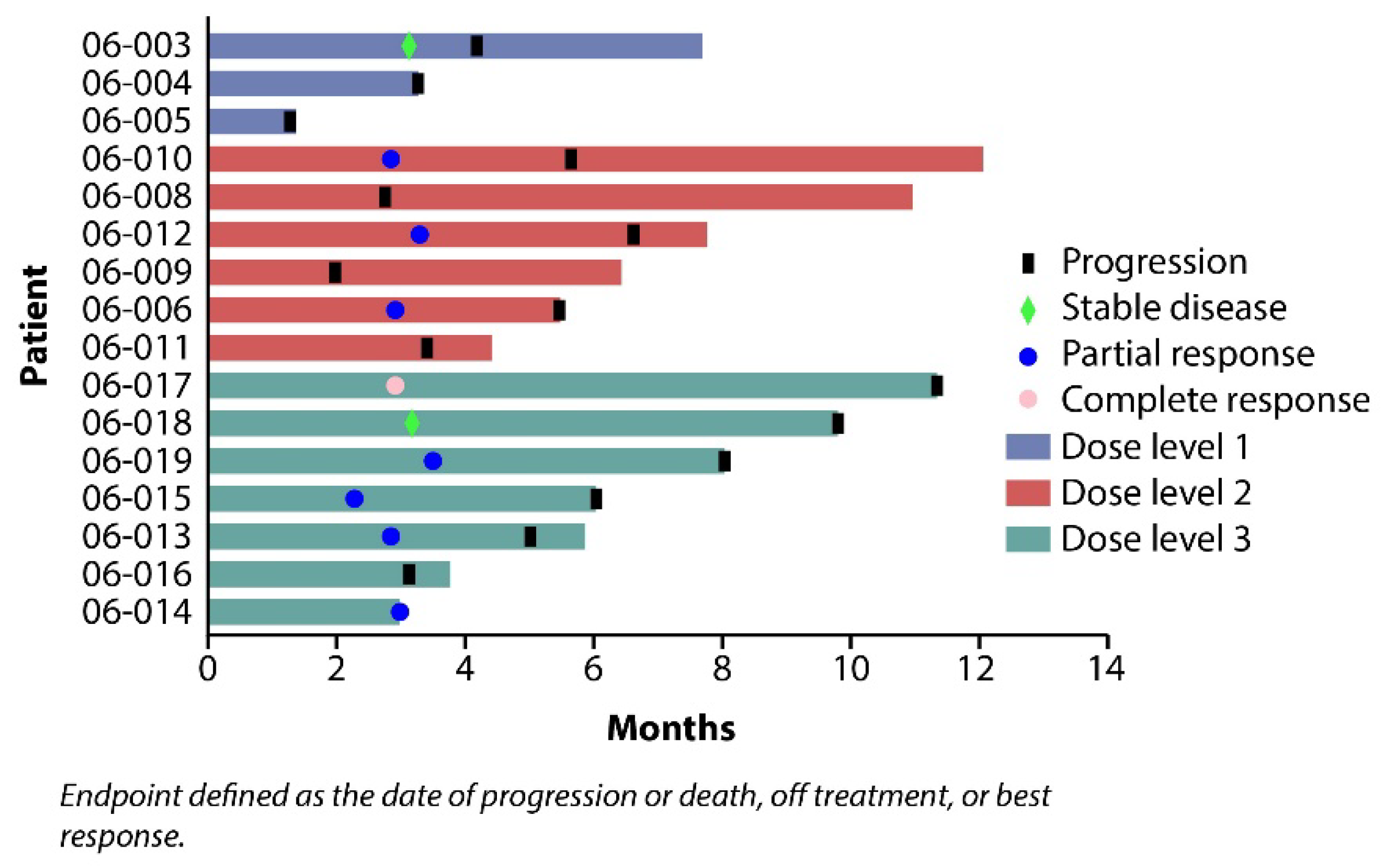
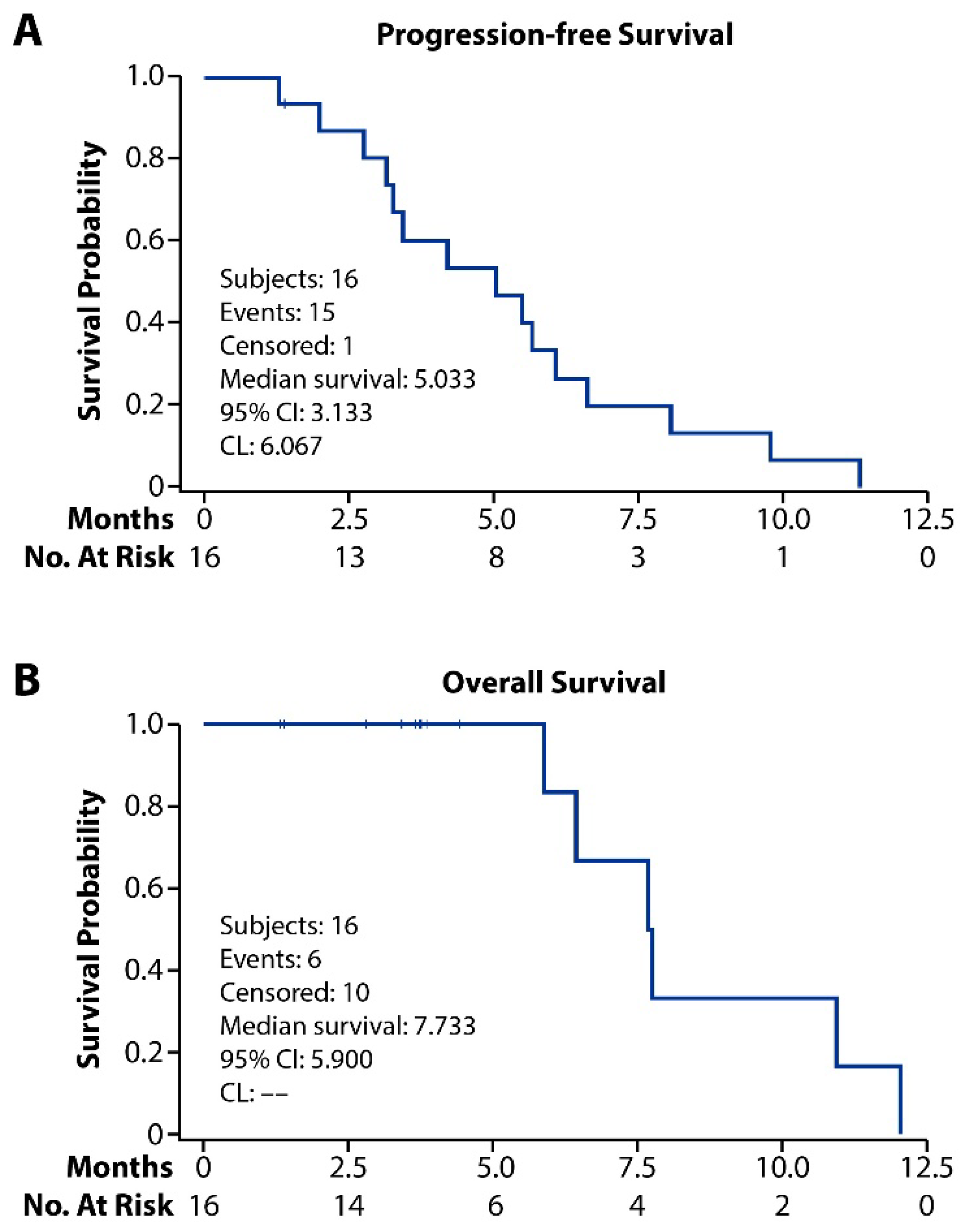
3.3. Efficacy
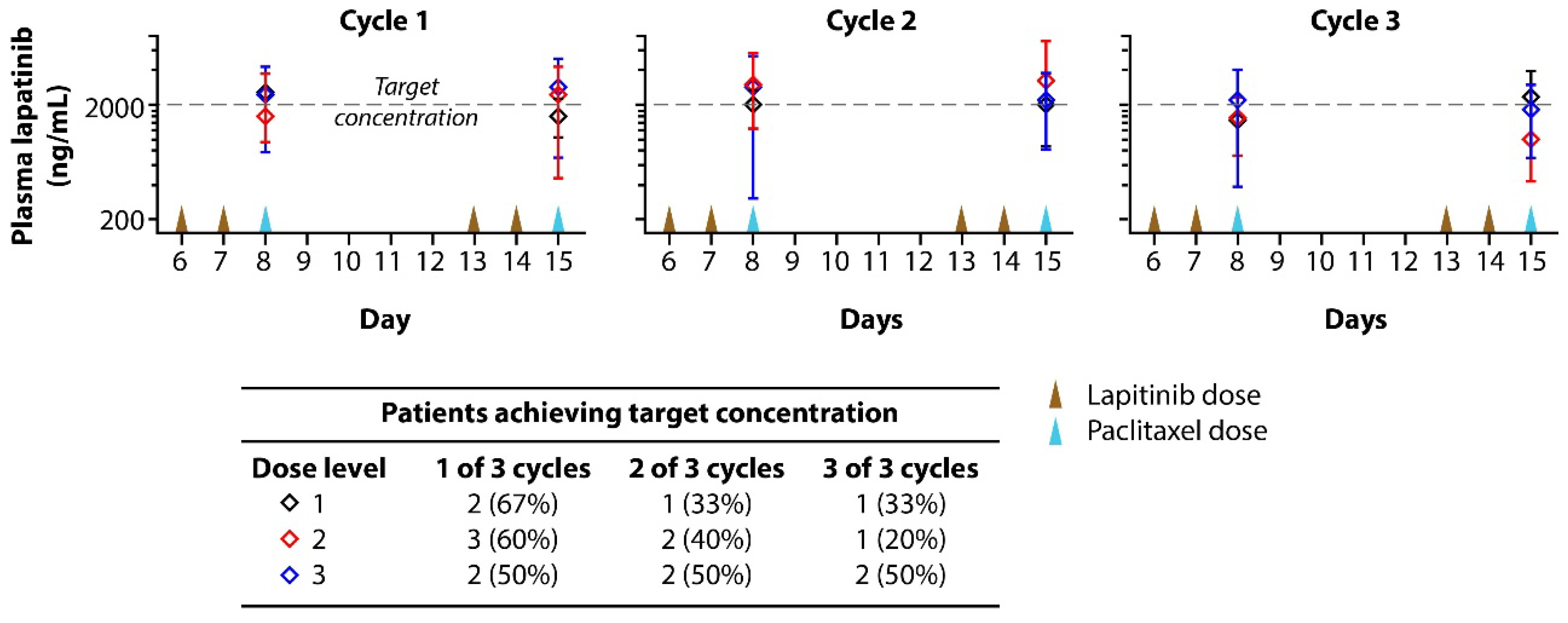
3.4. Plasma Lapatinib Concentrations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Kratzer, T.B.; Giaquinto, A.N.; Sung, H.; Jemal, A. Cancer statistics, 2025. CA: A Cancer J. Clin. 2025, 75, 10–45. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Giaquinto, A.N.; Jemal, A. Cancer statistics, 2024. CA: a cancer journal for clinicians 2024, 74, 12–49. [Google Scholar] [CrossRef]
- NCCN Clinical Practice Guideline in Oncology, Ovarian Cancer Including Fallopian Tube Cancer and Primary Peritoneal Cancer. Version 1.2020—March 11, 2020. Available online: https://www.nccn.org/professionals/physician_gls/pdf/ovarian.pdf (accessed on).
- Gaillard, S.; Oaknin, A.; Ray-Coquard, I.; Vergote, I.; Scambia, G.; Colombo, N.; Fernandez, C.; Alfaro, V.; Kahatt, C.; Nieto, A.; et al. Lurbinectedin versus pegylated liposomal doxorubicin or topotecan in patients with platinum-resistant ovarian cancer: A multicenter, randomized, controlled, open-label phase 3 study (CORAIL). Gynecol. Oncol. 2021, 163, 237–245. [Google Scholar] [CrossRef]
- Hamanishi, J.; Takeshima, N.; Katsumata, N.; Ushijima, K.; Kimura, T.; Takeuchi, S.; Matsumoto, K.; Ito, K.; Mandai, M.; Nakai, H.; et al. Nivolumab Versus Gemcitabine or Pegylated Liposomal Doxorubicin for Patients With Platinum-Resistant Ovarian Cancer: Open-Label, Randomized Trial in Japan (NINJA). J. Clin. Oncol. 2021, 39, 3671–3681. [Google Scholar] [CrossRef]
- Pujade-Lauraine, E.; Fujiwara, K.; A Ledermann, J.; Oza, A.M.; Kristeleit, R.; Ray-Coquard, I.-L.; E Richardson, G.; Sessa, C.; Yonemori, K.; Banerjee, S.; et al. Avelumab alone or in combination with chemotherapy versus chemotherapy alone in platinum-resistant or platinum-refractory ovarian cancer (JAVELIN Ovarian 200): an open-label, three-arm, randomised, phase 3 study. Lancet Oncol. 2021, 22, 1034–1046. [Google Scholar] [CrossRef] [PubMed]
- Pujade-Lauraine, E.; Hilpert, F.; Weber, B.; Reuss, A.; Poveda, A.; Kristensen, G.; Sorio, R.; Vergote, I.; Witteveen, P.; Bamias, A.; et al. Bevacizumab Combined With Chemotherapy for Platinum-Resistant Recurrent Ovarian Cancer: The AURELIA Open-Label Randomized Phase III Trial. J. Clin. Oncol. 2014, 32, 1302–1308. [Google Scholar] [CrossRef] [PubMed]
- Geyer, C.E.; Forster, J.; Lindquist, D.; Chan, S.; Romieu, C.G.; Pienkowski, T.; Jagiello-Gruszfeld, A.; Crown, J.; Chan, A.; Kaufman, B.; et al. Lapatinib plus Capecitabine for HER2-Positive Advanced Breast Cancer. N. Engl. J. Med. 2006, 355, 2733–2743. [Google Scholar] [CrossRef]
- Blackwell, K.L.; Burstein, H.J.; Storniolo, A.M.; Rugo, H.; Sledge, G.; Koehler, M.; Ellis, C.; Casey, M.; Vukelja, S.; Bischoff, J.; et al. Randomized Study of Lapatinib Alone or in Combination With Trastuzumab in Women With ErbB2-Positive, Trastuzumab-Refractory Metastatic Breast Cancer. J. Clin. Oncol. 2010, 28, 1124–1130. [Google Scholar] [CrossRef]
- Guan, Z.; Xu, B.; DeSilvio, M.L.; Shen, Z.; Arpornwirat, W.; Tong, Z.; Lorvidhaya, V.; Jiang, Z.; Yang, J.; Makhson, A.; et al. Randomized Trial of Lapatinib Versus Placebo Added to Paclitaxel in the Treatment of Human Epidermal Growth Factor Receptor 2–Overexpressing Metastatic Breast Cancer. J. Clin. Oncol. 2013, 31, 1947–1953. [Google Scholar] [CrossRef]
- Di Leo, A.; Gomez, H.L.; Aziz, Z.; Zvirbule, Z.; Bines, J.; Arbushites, M.C.; Guerrera, S.F.; Koehler, M.; Oliva, C.; Stein, S.H.; et al. Phase III, Double-Blind, Randomized Study Comparing Lapatinib Plus Paclitaxel With Placebo Plus Paclitaxel As First-Line Treatment for Metastatic Breast Cancer. J. Clin. Oncol. 2008, 26, 5544–5552. [Google Scholar] [CrossRef]
- Chien, A.J.; Illi, J.A.; Ko, A.H.; Korn, W.M.; Fong, L.; Chen, L.-M.; Kashani-Sabet, M.; Ryan, C.J.; Rosenberg, J.E.; Dubey, S.; et al. A Phase I Study of a 2-Day Lapatinib Chemosensitization Pulse Preceding Nanoparticle Albumin-Bound Paclitaxel for Advanced Solid Malignancies. Clin. Cancer Res. 2009, 15, 5569–5575. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, A.K.; Follit, C.A.; Chen, G.; Williams, N.S.; Vogel, P.D.; Wise, J.G. Targeted inhibitors of P-glycoprotein increase chemotherapeutic-induced mortality of multidrug resistant tumor cells. Sci. Rep. 2018, 8, 1–18. [Google Scholar] [CrossRef]
- Penson, R.T.; Oliva, E.; Skates, S.J.; Glyptis, T.; Fuller, A.F.; Goodman, A.; Seiden, M.V. Expression of multidrug resistance-1 protein inversely correlates with paclitaxel response and survival in ovarian cancer patients: a study in serial samples. Gynecol. Oncol. 2004, 93, 98–106. [Google Scholar] [CrossRef]
- Moasser, M.M.; Wilmes, L.J.; Wong, C.H.; Aliu, S.; Li, K.-L.; Wang, D.; Hom, Y.K.; Hann, B.; Hylton, N.M. Improved tumor vascular function following high-dose epidermal growth factor receptor tyrosine kinase inhibitor therapy. J. Magn. Reson. Imaging 2007, 26, 1618–1625. [Google Scholar] [CrossRef]
- McCorkle, J.R.; Gorski, J.W.; Liu, J.; Riggs, M.B.; McDowell, A.B.; Lin, N.; Wang, C.; Ueland, F.R.; Kolesar, J.M. Lapatinib and poziotinib overcome ABCB1-mediated paclitaxel resistance in ovarian cancer. PLOS ONE 2021, 16, e0254205. [Google Scholar] [CrossRef]
- Morikawa, A.; de Stanchina, E.; Pentsova, E.; Kemeny, M.M.; Li, B.T.; Tang, K.; Patil, S.; Fleisher, M.; Van Poznak, C.; Norton, L.; et al. Phase I Study of Intermittent High-Dose Lapatinib Alternating with Capecitabine for HER2-Positive Breast Cancer Patients with Central Nervous System Metastases. Clin. Cancer Res. 2019, 25, 3784–3792. [Google Scholar] [CrossRef]
- Yu, A.; Faiq, N.; Green, S.; Lai, A.; Green, R.; Hu, J.; Cloughesy, T.F.; Mellinghoff, I.; Nghiemphu, P.L. Report of safety of pulse dosing of lapatinib with temozolomide and radiation therapy for newly-diagnosed glioblastoma in a pilot phase II study. J. Neuro-Oncology 2017, 134, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Burris, H.A.; Hurwitz, H.I.; Dees, E.C.; Dowlati, A.; Blackwell, K.L.; O’NEil, B.; Marcom, P.K.; Ellis, M.J.; Overmoyer, B.; Jones, S.F.; et al. Phase I Safety, Pharmacokinetics, and Clinical Activity Study of Lapatinib (GW572016), a Reversible Dual Inhibitor of Epidermal Growth Factor Receptor Tyrosine Kinases, in Heavily Pretreated Patients With Metastatic Carcinomas. J. Clin. Oncol. 2005, 23, 5305–5313. [Google Scholar] [CrossRef]
- Markman, M.; Blessing, J.; Rubin, S.C.; Connor, J.; Hanjani, P.; Waggoner, S. Phase II trial of weekly paclitaxel (80 mg/m2) in platinum and paclitaxel-resistant ovarian and primary peritoneal cancers: A Gynecologic Oncology Group study. Gynecol. Oncol. 2006, 101, 436–440. [Google Scholar] [CrossRef]
- Markman, M.; Hall, J.; Spitz, D.; Weiner, S.; Carson, L.; Van Le, L.; Baker, M. Phase II Trial of Weekly Single-Agent Paclitaxel in Platinum/Paclitaxel-Refractory Ovarian Cancer. J. Clin. Oncol. 2002, 20, 2365–2369. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.A.; Sill, M.W.; Lankes, H.A.; Godwin, A.K.; Mannel, R.S.; Armstrong, D.K.; Carolla, R.L.; Liepman, M.K.; Spirtos, N.M.; Fischer, E.G.; et al. A phase II evaluation of lapatinib in the treatment of persistent or recurrent epithelial ovarian or primary peritoneal carcinoma: A gynecologic oncology group study. Gynecol. Oncol. 2012, 124, 569–574. [Google Scholar] [CrossRef]
- Lheureux, S.; Krieger, S.; Weber, B.; Pautier, P.; Fabbro, M.; Selle, F.; Bourgeois, H.; Petit, T.; Lortholary, A.; Plantade, A.; et al. Expected Benefits of Topotecan Combined With Lapatinib in Recurrent Ovarian Cancer According to Biological Profile. Int. J. Gynecol. Cancer 2012, 22, 1483–1488. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.N.; Angelergues, A.; Konecny, G.E.; García, Y.; Banerjee, S.; Lorusso, D.; Lee, J.-Y.; Moroney, J.W.; Colombo, N.; Roszak, A.; et al. Mirvetuximab Soravtansine in FRα-Positive, Platinum-Resistant Ovarian Cancer. New Engl. J. Med. 2023, 389, 2162–2174. [Google Scholar] [CrossRef] [PubMed]
- Matulonis, U.A.; Lorusso, D.; Oaknin, A.; Pignata, S.; Dean, A.; Denys, H.; Colombo, N.; Van Gorp, T.; Konner, J.A.; Marin, M.R.; et al. Efficacy and Safety of Mirvetuximab Soravtansine in Patients With Platinum-Resistant Ovarian Cancer With High Folate Receptor Alpha Expression: Results From the SORAYA Study. J. Clin. Oncol. 2023, 41, 2436–2445. [Google Scholar] [CrossRef] [PubMed]
| TOTAL N = 16 |
||
|---|---|---|
| Toxicity N (%) | GRADE 3 / 4 | ANY GRADE |
| Hematological toxicity | ||
| Anemia | 1 (6.3) | 8 (50.0) |
| Thrombocytopenia | 1 (6.3) | 3 (18.8) |
| Leukopenia | 1 (6.3) | 9 (56.3) |
| Non-hematological toxicity | ||
| Alanine aminotransferase increased | 0 (0) | 3 (18.8) |
| Aspartate aminotransferase increased | 0 (0) | 4 (25.0) |
| Creatinine increased | 2 (12.5) | 6 (37.5) |
| Abdominal pain | 0 (0) | 2 (12.5) |
| Constipation | 0 (0) | 2 (12.5) |
| Diarrhea | 3 (18.8) | 14 (87.5) |
| Nausea | 0 (0) | 4 (25.0) |
| Fatigue | 0 (0) | 3 (18.8) |
| Hyperkalemia | 0 (0) | 4 (25.0) |
| Hyperphosphatemia | 0 (0) | 2 (12.5) |
| Hypercalcemia | 0 (0) | 4 (25.0) |
| Hyponatremia | 1 (6.3) | 8 (50.0) |
| Hypokalemia | 2 (12.5) | 9 (56.3) |
| Hypophosphatemia | 0 (0.0) | 3 (18.8) |
| Hypocalcemia | 0 (0) | 8 (50.0) |
| Hypomagnesemia | 0 (0) | 7 (43.8) |
| Hypoalbuminemia | 0 (0) | 4 (25.0) |
| Back pain | 0 (0) | 3 (18.8) |
| Muscle cramp | 0 (0) | 2 (12.5) |
| Peripheral sensory neuropathy | 0 (0) | 2 (12.5) |
| Dyspnea | 0 (0) | 4 (25.0) |
| Alopecia | 0 (0) | 2 (12.5) |
| Rash maculo-papular | 0 (0) | 3 (18.8) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




