Submitted:
27 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Mathematical Framework
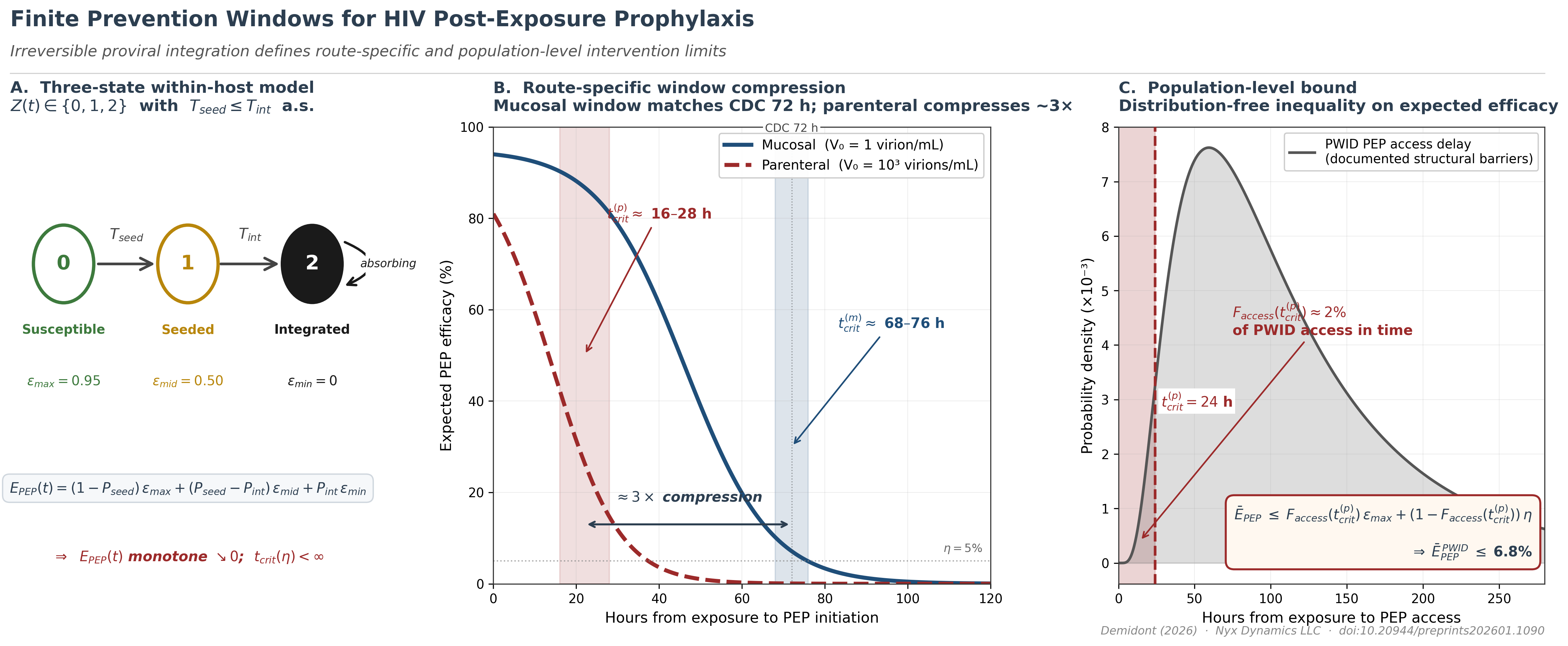
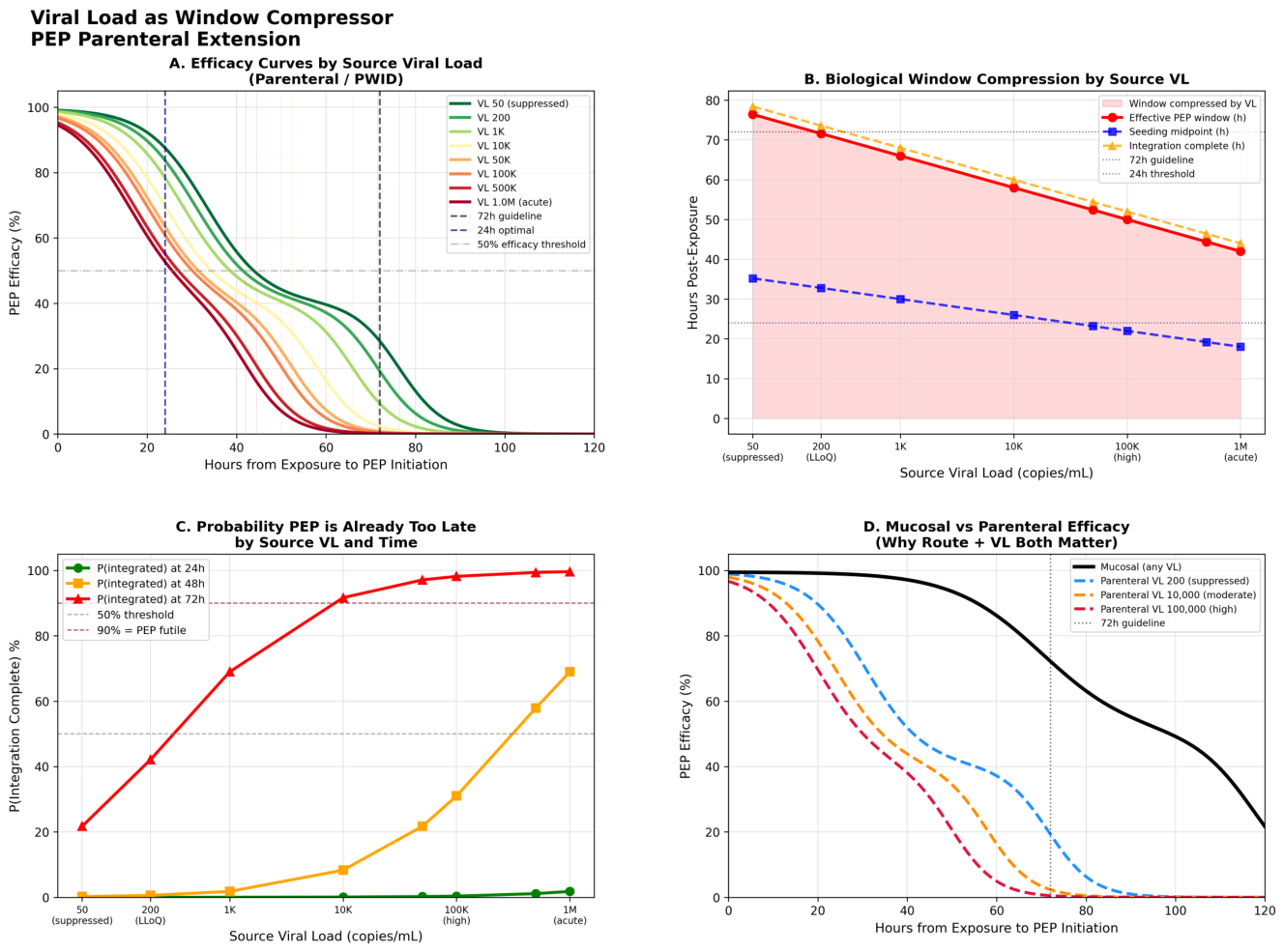
Route-Dependent Window Compression
Parameterization and Simulation
Results: Route-Specific Windows
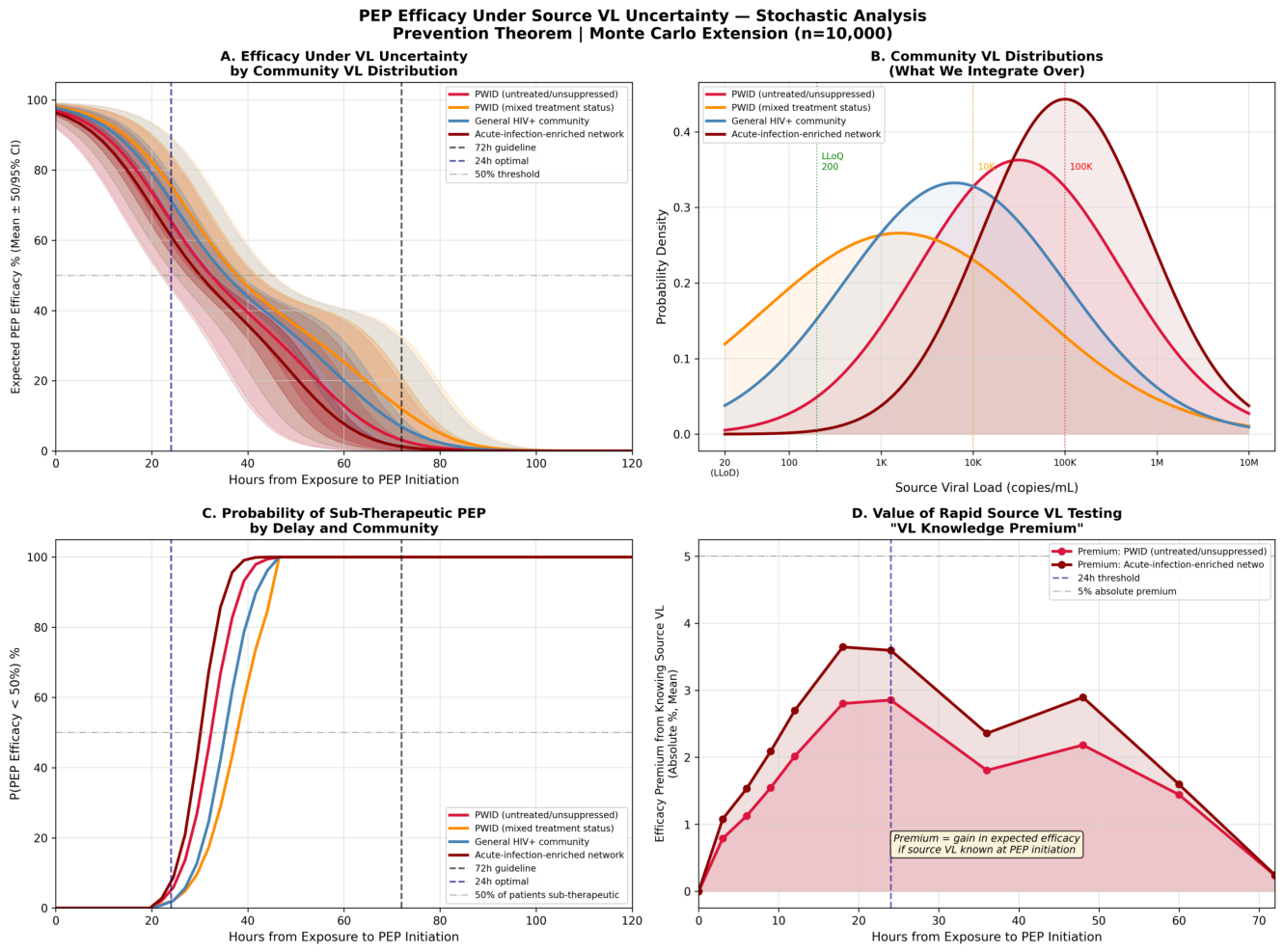
Stochastic Efficacy Analysis
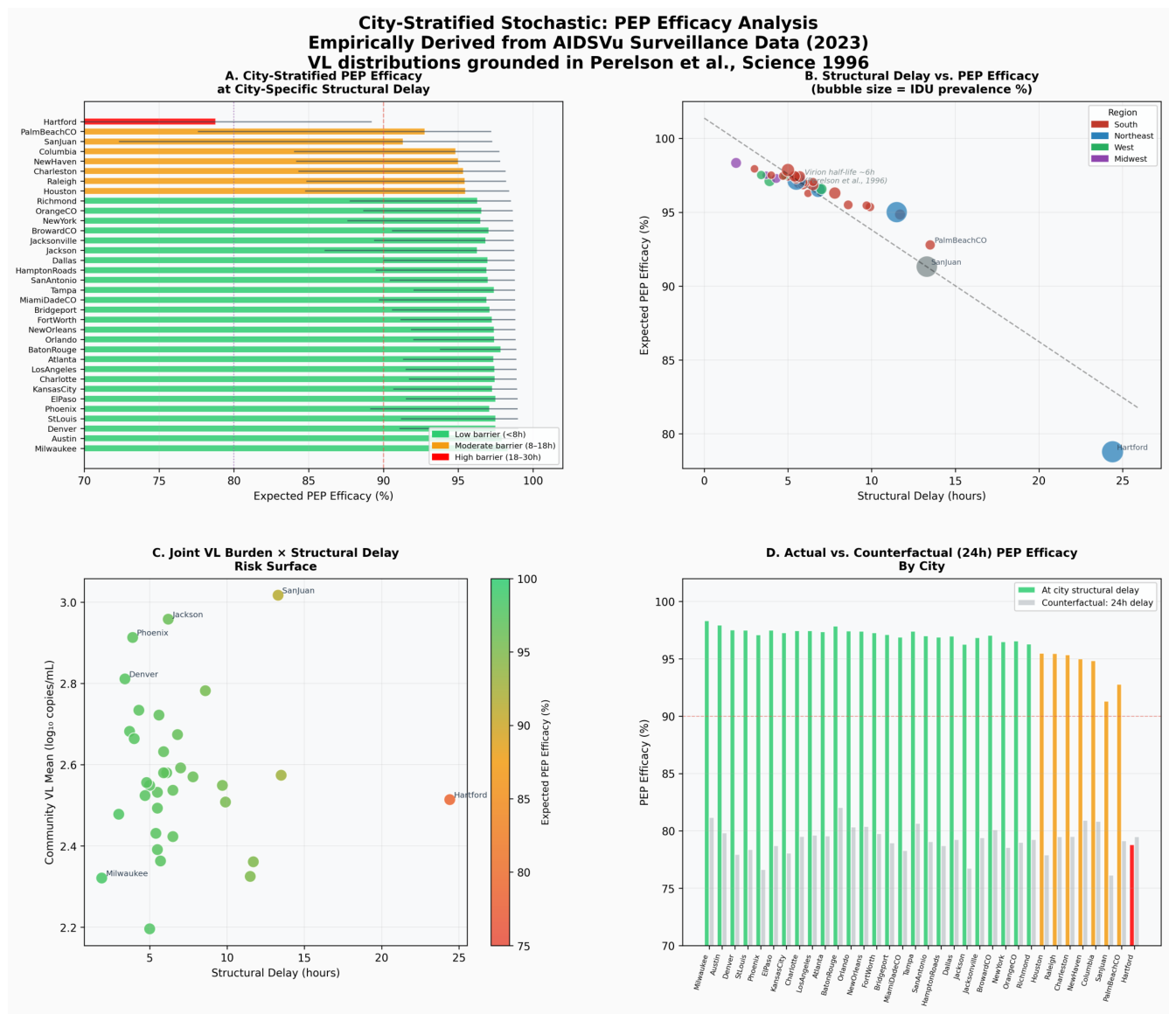
City-Stratified Analysis
Population-Level Efficacy Bound
Stochastic VL Uncertainty and the Critical Window
Implications
Conclusion
Materials and Methods
Mathematical Proofs
Numerical Simulation
City-Stratified Parameterization
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Use of Artificial Intelligence
References
- Tanner, M.R.; et al. Antiretroviral postexposure prophylaxis after sexual, injection drug use, or other nonoccupational exposure to HIV — CDC recommendations, United States, 2025. MMWR Recomm. Rep. 2025. [Google Scholar] [CrossRef]
- Wahl, A.; Al-Harthi, L. HIV infection of non-classical cells in the brain. Retrovirology 2023, 20, 1. [Google Scholar] [CrossRef]
- Siliciano, J.D.; Siliciano, R.F. Enhanced culture assay for detection and quantitation of latently infected, resting CD4+ T-cells. Methods Mol. Biol. 2015, 1354, 3–15. [Google Scholar]
- Perelson, A.S. , Decay characteristics of HIV-1-infected compartments during combination therapy. Nature 1997, 387, 188–191. [Google Scholar] [CrossRef]
- Perelson, A.S.; Neumann, A.U.; Markowitz, M.; Leonard, J.M.; Ho, D.D. HIV-1 dynamics in vivo: virion clearance rate, infected cell life-span, and viral generation time. Science 1996, 271, 1582–1586. [Google Scholar] [CrossRef]
- McMichael, A.J.; Rowland-Jones, S.L. The immune response to HIV. Immunity 2010, 33, 431–441. [Google Scholar]
- Tsai, C.C. , Prevention of SIV infection in macaques by (R)-9-(2-phosphonylmethoxypropyl)adenine. Science Timing data: J. Virol. 72, 4265–4273 (1998). 1995, 270, 1197–1199. [Google Scholar] [CrossRef] [PubMed]
- Otten, R.A. , Efficacy of postexposure prophylaxis after intravaginal exposure of pig-tailed macaques to a human-derived retrovirus (HIV type 2). J. Infect. Dis. 2000, 182, 1820–1825. [Google Scholar]
- Taylor, D. , Time to PEP initiation and access barriers for injection drug users. J. Acquir. Immune Defic. Syndr. 2019, 80, e56–e59. [Google Scholar]
- Baugher, K.A. , HIV preexposure prophylaxis use among persons who inject drugs — United States, 2023. MMWR Morb. Mortal. Wkly. Rep. 2025. [Google Scholar]
- Grant, R.M. , Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N. Engl. J. Med. 2010, 363, 2587–2599. [Google Scholar] [CrossRef]
- Anderson, P.L. , Emtricitabine–tenofovir concentrations and pre-exposure prophylaxis efficacy. Sci. Transl. Med. 2012, 4, 151ra125. [Google Scholar] [CrossRef] [PubMed]
- Swanstrom, A.E. , Lenacapavir protects macaques against intravenous SHIV challenge. Sci. Transl. Med. 2023. [Google Scholar]
- Vidal, S.J. , Long-acting antiretrovirals prevent SHIV infection in NHP mucosal challenge. J. Infect. Dis. 2022, 226, 2051–2059. [Google Scholar]
- Demidont, A.C. accepted. Clinical decision support algorithm for LAI-PrEP bridge period navigation at UNAIDS global target scale. Viruses 2026. [Google Scholar] [CrossRef]
- Emau, P. , Post-exposure prophylaxis for SIV revisited: animal model for HIV prevention. AIDS Res. Ther. 3 2006, 8. [Google Scholar]
| Route | Study | PEP delay | Duration | Protection | Model prediction |
|---|---|---|---|---|---|
| Parenteral (IV) | Tsai 1998[7] | 24 h | 28 days | 100% | Concordant |
| Tsai 1998 | 24 h | 10 days | 50% | Concordant | |
| Tsai 1998 | 24 h | 3 days | 0% | Concordant | |
| Tsai 1998 | 48 h | 28 days | 50% | Concordant | |
| Emau 2006 [16] | 24 h | 28 days | 100% | Concordant | |
| Mucosal (vaginal) | Otten 2000[8] | 12 h | 28 days | 100% | Concordant |
| Otten 2000 | 36 h | 28 days | 100% | Concordant | |
| Otten 2000 | 72 h | 28 days | 50% | Concordant |
| Distribution | 24 h mean | 36 h mean | 48 h | 48 h P(futile) | 72 h P(futile) | Peak CI (time) |
|---|---|---|---|---|---|---|
| PWID untreated | 65.5% | 44.6% | 100% | 9.1% | 91.8% | 39.7 pp (49 h) |
| PWID mixed Tx | 75.4% | 52.8% | 100% | 3.1% | 56.5% | 40.1 pp (54 h) |
| General community | 71.2% | 49.2% | 100% | 3.5% | 76.0% | 39.8 pp (54 h) |
| Acute-enriched | 61.2% | 41.3% | 100% | 13.8% | 98.8% | 37.4 pp (49 h) |
| Interpretation | upper bound | |
|---|---|---|
| 1% | Severe structural barriers | |
| 2% | Base case (log-normal, median 72 h) | |
| 5% | Moderate barrier reduction | |
| 10% | Substantial barrier reduction | |
| 20% | Aggressive rapid-access programs |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




