Submitted:
12 January 2026
Posted:
14 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
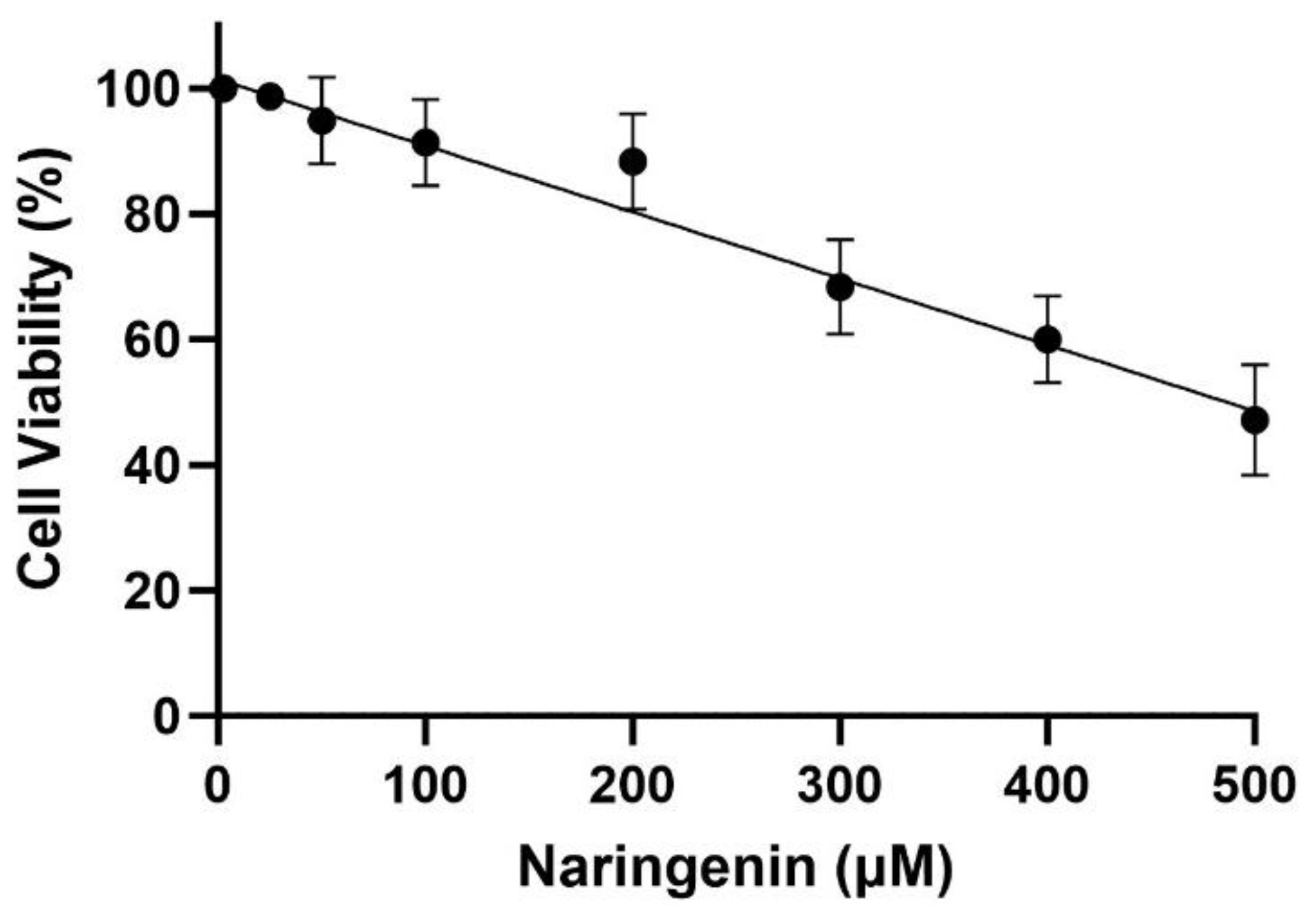
3.1. Cytotoxicity Effect of Naringenin and Morin on HMC3 Cells
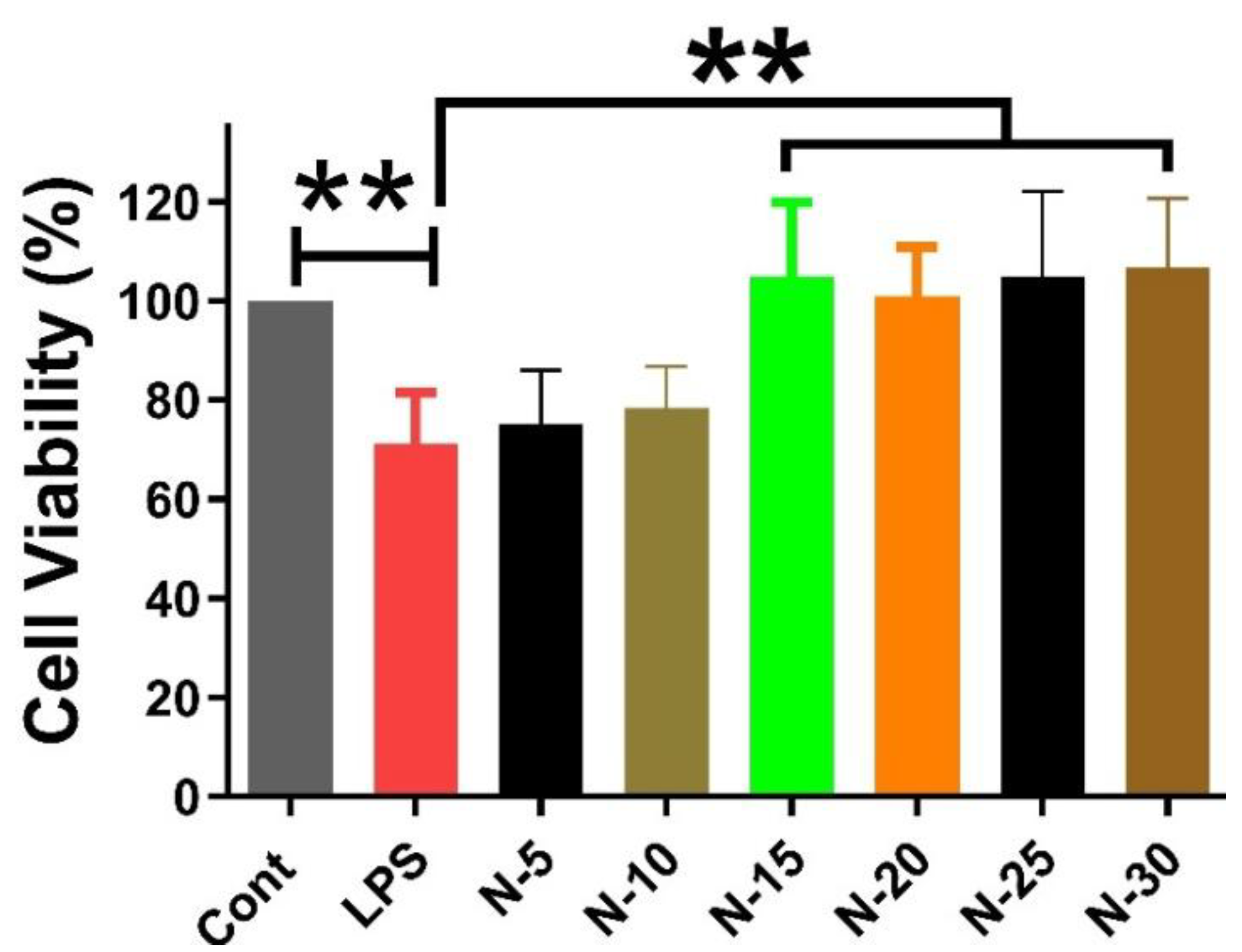
3.2. Cell Viability Assay
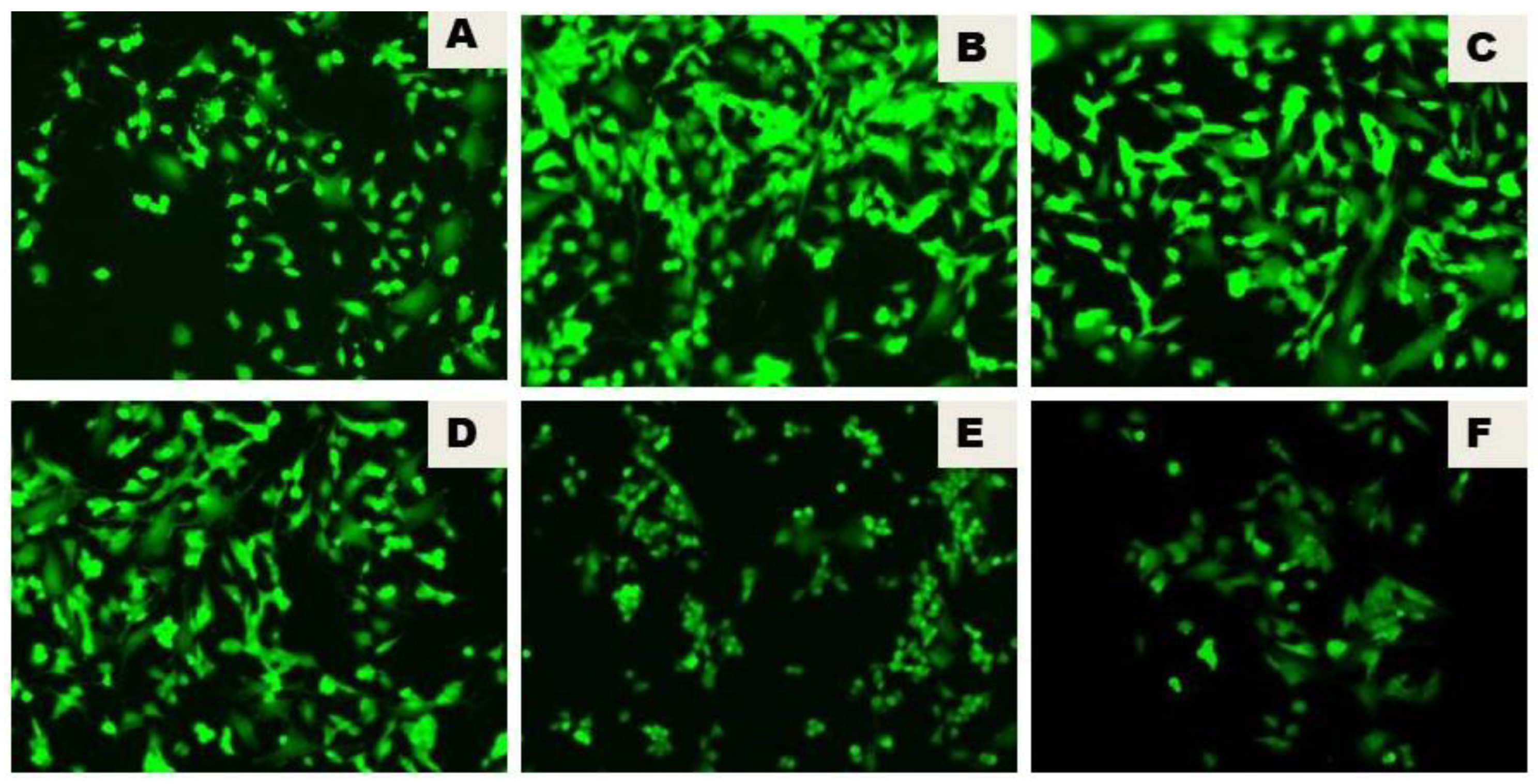
3.3. Naringenin Reduced the LPS-Initiated ROS Production
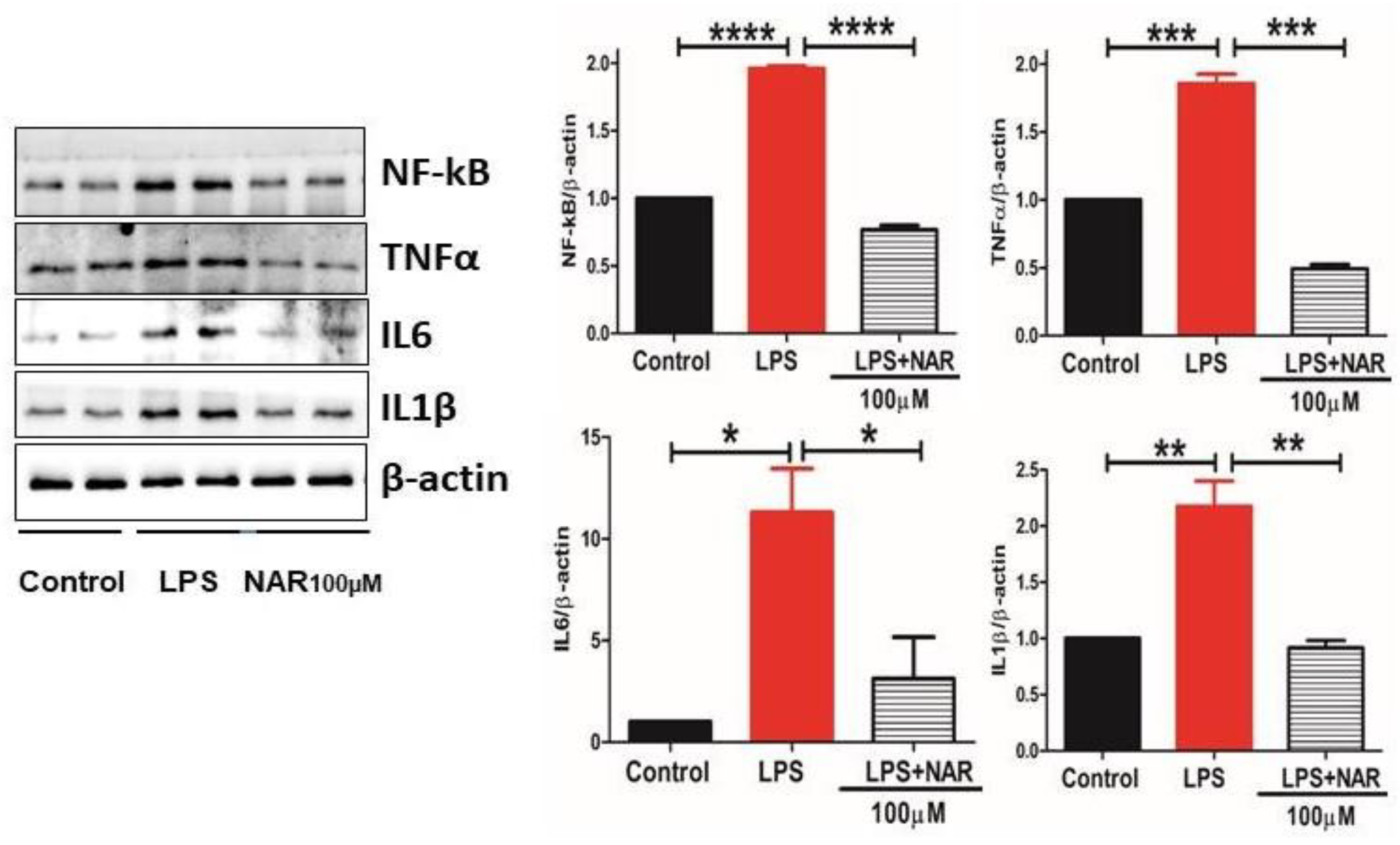
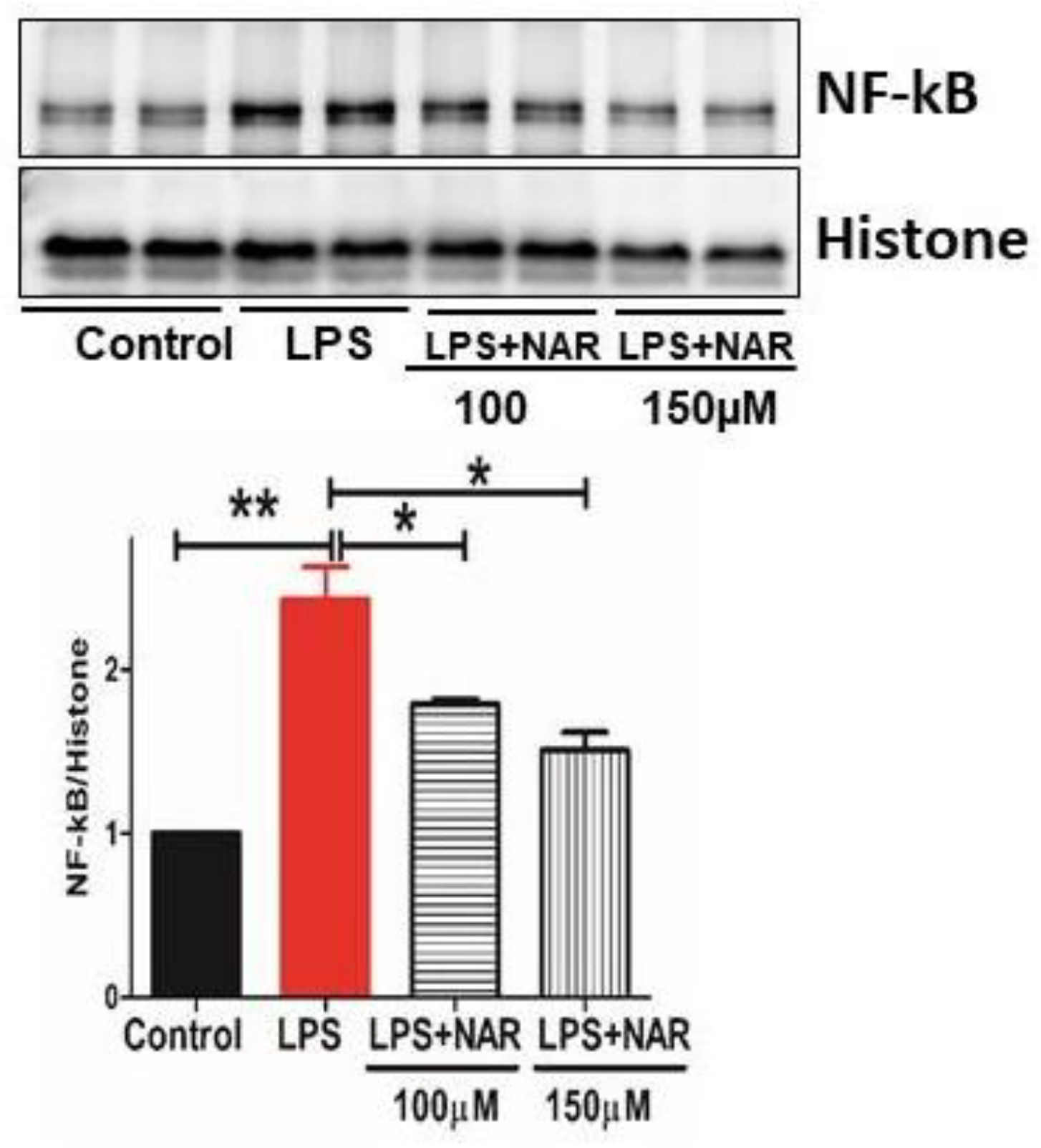
3.4. Effect of Naringenin on the Level of Proinflammatory Cytokines and NF-κB Activity
3.5. Effect of Naringenin on Nuclear Fraction of NF-κB Activity
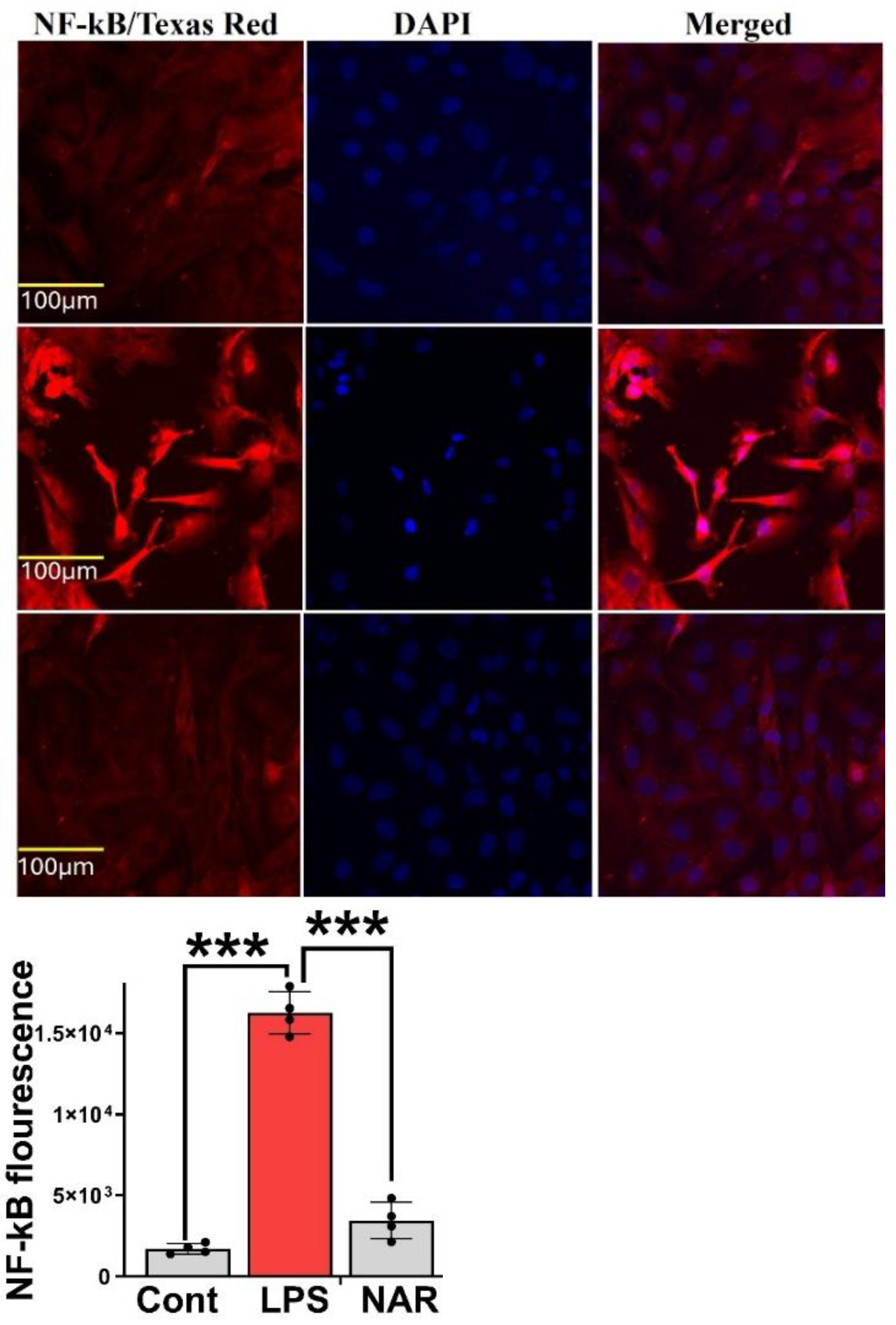
3.6. Immunocytochemistry Revealed Inhibition of LPS-Induced NF-κB Overexpression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Almarghalani, D.A.; Shah, Z.A. Progress on siRNA-based gene therapy targeting secondary injury after intracerebral hemorrhage. Gene Ther. 2021, 30, 1–7. [Google Scholar] [CrossRef]
- Magid-Bernstein, J.; Girard, R.; Polster, S.; Srinath, A.; Romanos, S.; Awad, I.A.; Sansing, L.H. Cerebral hemorrhage: Pathophysiology, treatment, and future directions. Circ. Res. 2022, 130, 1204–1229. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, W.; Liu, J.; Zhang, B.; Zheng, L.; Zou, W. The dual role of microglia in intracerebral hemorrhage. Behav. Brain Res. 2024, 473, 115198. [Google Scholar] [CrossRef]
- Gong, Y. Microglial Mechanisms and Therapeutic Potential in Brain Injury Post-Intracerebral Hemorrhage. Journal of Inflammation Research 2025, 2955–2973. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Xu, Z.; Liu, Y.; Yang, J.; Bai, L.; Li, X.; Sun, Q.; Li, H.; Chen, G. Unveiling microglia heterogeneity in intracerebral hemorrhage. Neuroscience 2024, 555, 167–177. [Google Scholar] [CrossRef]
- Ostrowska, Z.; Robaszkiewicz, K.; Moraczewska, J. Regulation of actin filament turnover by cofilin-1 and cytoplasmic tropomyosin isoforms. Biochim. et Biophys. Acta (BBA) - Proteins Proteom. 2017, 1865, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Shehjar, F.; Almarghalani, D.A.; Mahajan, R.; Hasan, S.A.-M.; Shah, Z.A. The multifaceted role of cofilin in neurodegeneration and stroke: Insights into pathogenesis and targeting as a therapy. Cells 2024, 13, 188. [Google Scholar] [CrossRef] [PubMed]
- Uruk, G.; Mocanu, E.; Shaw, A.E.; Bamburg, J.R.; Swanson, R.A. Cofilactin rod formation mediates inflammation-induced neurite degeneration. Cell Rep. 2024, 43, 113914–113914. [Google Scholar] [CrossRef]
- Xing, J.; Wang, Y.; Peng, A.; Li, J.; Niu, X.; Zhang, K. The role of actin cytoskeleton CFL1 and ADF/cofilin superfamily in inflammatory response. Front. Mol. Biosci. 2024, 11, 1408287. [Google Scholar] [CrossRef]
- Almarghalani, D.A.; Sha, X.; Mrak, R.E.; Shah, Z.A. Spatiotemporal Cofilin Signaling, Microglial Activation, Neuroinflammation, and Cognitive Impairment Following Hemorrhagic Brain Injury. Cells 2023, 12, 1153. [Google Scholar] [CrossRef]
- Alhadidi, Q.; Shah, Z.A. Cofilin mediates LPS-induced microglial cell activation and associated neurotoxicity through activation of NF-κB and JAK–STAT pathway. Mol. Neurobiol. 2017, 55, 1676–1691. [Google Scholar] [CrossRef]
- Alhadidi, Q.; Nash, K.M.; Alaqel, S.; Bin Sayeed, M.S.; Shah, Z.A. Cofilin knockdown attenuates hemorrhagic brain injury-induced oxidative stress and microglial activation in mice. Neuroscience 2018, 383, 33–45. [Google Scholar] [CrossRef]
- Almarghalani, D.A.; Bahader, G.A.; Ali, M.; Tillekeratne, L.M.V.; Shah, Z.A. Cofilin inhibitor improves neurological and cognitive functions after Intracerebral Hemorrhage by suppressing endoplasmic reticulum stress related-neuroinflammation. Pharmaceuticals 2024, 17, 114. [Google Scholar] [CrossRef]
- Rust, M.B.; Gurniak, C.B.; Renner, M.; Vara, H.; Morando, L.; Görlich, A.; Sassoè-Pognetto, M.; Al Banchaabouchi, M.; Giustetto, M.; Triller, A.; et al. Learning, AMPA receptor mobility and synaptic plasticity depend on n-cofilin-mediated actin dynamics. EMBO J. 2010, 29, 1889–1902. [Google Scholar] [CrossRef] [PubMed]
- Goodson, M.; Rust, M.B.; Witke, W.; Bannerman, D.; Mott, R.; Ponting, C.P.; Flint, J. Cofilin-1: A modulator of anxiety in mice. PLOS Genet. 2012, 8, e1002970. [Google Scholar] [CrossRef]
- Wolf, M.; Zimmermann, A.-M.; Görlich, A.; Gurniak, C.B.; Sassoè-Pognetto, M.; Friauf, E.; Witke, W.; Rust, M.B. ADF/Cofilin Controls Synaptic Actin Dynamics and Regulates Synaptic Vesicle Mobilization and Exocytosis. Cereb. Cortex 2014, 25, 2863–2875. [Google Scholar] [CrossRef] [PubMed]
- Rust, M.B. Novel functions for ADF/cofilin in excitatory synapses - lessons from gene-targeted mice. Commun. Integr. Biol. 2015, 8, e1114194–e1114194. [Google Scholar] [CrossRef]
- Nakamura, T.; Xi, G.; Hua, Y.; Schallert, T.; Hoff, J.T.; Keep, R.F. Intracerebral hemorrhage in mice: Model characterization and application for genetically modified mice. J. Cereb. Blood Flow Metab. 2004, 24, 487–494. [Google Scholar] [CrossRef]
- Xie, Y.; Li, Y.-J.; Lei, B.; Kernagis, D.; Liu, W.-W.; Bennett, E.R.; Venkatraman, T.; Lascola, C.D.; Laskowitz, D.T.; Warner, D.S.; et al. Sex differences in gene and protein expression after intracerebral hemorrhage in mice. Transl. Stroke Res. 2018, 10, 231–239. [Google Scholar] [CrossRef]
- Chen, Z.; Xi, G.; Mao, Y.; Keep, R.F.; Hua, Y. Effects of progesterone and testosterone on ICH-induced brain injury in rats, in Intracerebral Hemorrhage Research: From Bench to Bedside; Springer, 2011; pp. 289–293. [Google Scholar]
- Chen, T.-Y.; Tsai, K.-L.; Lee, T.-Y.; Chiueh, C.C.; Lee, W.-S.; Hsu, C. Sex-specific role of thioredoxin in neuroprotection against iron-induced brain injury conferred by estradiol. Stroke 2010, 41, 160–165. [Google Scholar] [CrossRef] [PubMed]
- Sterenstein, A.; Garg, R. The impact of sex on epidemiology, management, and outcome of spontaneous intracerebral hemorrhage (sICH). J. Stroke Cerebrovasc. Dis. 2024, 33, 107755–107755. [Google Scholar] [CrossRef] [PubMed]
- Nash, K.M.; Schiefer, I.T.; Shah, Z.A. Development of a reactive oxygen species-sensitive nitric oxide synthase inhibitor for the treatment of ischemic stroke. Free. Radic. Biol. Med. 2018, 115, 395–404. [Google Scholar] [CrossRef]
- García-Campos, P.; Báez-Matus, X.; Jara-Gutiérrez, C.; Paz-Araos, M.; Astorga, C.; Cea, L.A.; Rodríguez, V.; Bevilacqua, J.A.; Caviedes, P.; Cárdenas, A.M. N-Acetylcysteine Reduces Skeletal Muscles Oxidative Stress and Improves Grip Strength in Dysferlin-Deficient Bla/J Mice. Int. J. Mol. Sci. 2020, 21, 4293. [Google Scholar] [CrossRef]
- Clark, W.; Gunion-Rinker, L.; Lessov, N.; Hazel, K. Citicoline treatment for experimental Intracerebral Hemorrhage in mice. Stroke 1998, 29, 2136–2140. [Google Scholar] [CrossRef]
- Bahader, G.A.; Nash, K.M.; Almarghalani, D.A.; Alhadidi, Q.; McInerney, M.F.; Shah, Z.A. Type-I diabetes aggravates post-hemorrhagic stroke cognitive impairment by augmenting oxidative stress and neuroinflammation in mice. Neurochem. Int. 2021, 149, 105151. [Google Scholar] [CrossRef]
- Deacon, R.M.J.; Rawlins, J.N.P. T-maze alternation in the rodent. Nat. Protoc. 2006, 1, 7–12. [Google Scholar] [CrossRef]
- A Bevins, R.; Besheer, J. Object recognition in rats and mice: a one-trial non-matching-to-sample learning task to study 'recognition memory'. Nat. Protoc. 2006, 1, 1306–1311. [Google Scholar] [CrossRef]
- Denninger, J.K.; Smith, B.M.; Kirby, E.D. Novel object recognition and object location behavioral testing in mice on a budget. JoVE (Journal of Visualized Experiments) 2018, e58593. [Google Scholar]
- Li, Z.; Li, M.; Shi, S.X.; Yao, N.; Cheng, X.; Guo, A.; Zhu, Z.; Zhang, X.; Liu, Q. Brain transforms natural killer cells that exacerbate brain edema after intracerebral hemorrhage. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Young, K.; Morrison, H. Quantifying microglia morphology from photomicrographs of immunohistochemistry prepared tissue using ImageJ. Journal of visualized experiments: JoVE 2018, 57648. [Google Scholar]
- Wang, C.; Xu, Y. Mechanisms for sex differences in energy homeostasis. J. Mol. Endocrinol. 2019, 62, R129–R143. [Google Scholar] [CrossRef]
- Benz, V.; Bloch, M.; Wardat, S.; Böhm, C.; Maurer, L.; Mahmoodzadeh, S.; Wiedmer, P.; Spranger, J.; Foryst-Ludwig, A.; Kintscher, U. Sexual dimorphic regulation of body weight dynamics and adipose tissue lipolysis. PLOS ONE 2012, 7, e37794. [Google Scholar] [CrossRef]
- Rathod, Y.D.; Di Fulvio, M. The feeding microstructure of male and female mice. PLoS ONE 2021, 16, e0246569. [Google Scholar] [CrossRef] [PubMed]
- Satoh, J.; Kino, Y.; Asahina, N.; Takitani, M.; Miyoshi, J.; Ishida, T.; Saito, Y. TMEM119 marks a subset of microglia in the human brain. Neuropathology 2015, 36, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Bennett, M.L.; Bennett, F.C.; Liddelow, S.A.; Ajami, B.; Zamanian, J.L.; Fernhoff, N.B.; Mulinyawe, S.B.; Bohlen, C.J.; Adil, A.; Tucker, A.; et al. New tools for studying microglia in the mouse and human CNS. Proc. Natl. Acad. Sci. USA 2016, 113, E1738–E1746. [Google Scholar] [CrossRef]
- Ma, W.; Oswald, J.; Angulo, A.R.; Chen, Q. Tmem119 expression is downregulated in a subset of brain metastasis-associated microglia. BMC Neurosci. 2024, 25, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ruan, C.; Elyaman, W. A new understanding of TMEM119 as a marker of microglia. Front. Cell. Neurosci. 2022, 16, 902372. [Google Scholar] [CrossRef]
- Shi, X.; Bai, H.; Wang, J.; Wang, J.; Huang, L.; He, M.; Zheng, X.; Duan, Z.; Chen, D.; Zhang, J.; et al. Behavioral assessment of sensory, motor, emotion, and cognition in rodent models of intracerebral hemorrhage. Front. Neurol. 2021, 12, 667511. [Google Scholar] [CrossRef]
- Bamburg, J.R.; Minamide, L.S.; Wiggan, O.; Tahtamouni, L.H.; Kuhn, T.B. Cofilin and actin dynamics: multiple modes of regulation and their impacts in neuronal development and degeneration. Cells 2021, 10, 2726. [Google Scholar] [CrossRef]
- Liu, J. Microglial TMEM119 binds to amyloid-β to promote its clearance in an Aβ-depositing mouse model of Alzheimer’s disease. Immunity 2025. [Google Scholar] [CrossRef]
- Bedolla, A.; Wegman, E.; Weed, M.; Stevens, M.K.; Ware, K.; Paranjpe, A.; Alkhimovitch, A.; Ifergan, I.; Taranov, A.; Peter, J.D.; et al. Adult microglial TGFβ1 is required for microglia homeostasis via an autocrine mechanism to maintain cognitive function in mice. Nat. Commun. 2024, 15, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Mercurio, D.; Fumagalli, S.; Schafer, M.K.-H.; Pedragosa, J.; Ngassam, L.D.C.; Wilhelmi, V.; Winterberg, S.; Planas, A.M.; Weihe, E.; De Simoni, M.-G. Protein expression of the microglial marker tmem119 decreases in association with morphological changes and location in a mouse model of traumatic brain injury. Front. Cell. Neurosci. 2022, 16, 820127. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Vegeto, E.; Poletti, A.; Maggi, A. Estrogens, neuroinflammation, and neurodegeneration. Endocr. Rev. 2016, 37, 372–402. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



