Submitted:
12 January 2026
Posted:
13 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Traumatic Brain Injury (TBI): Definition, Prevalence, and Biomechanics
1.2. Classification of TBI (Severity, Type, Temporal Phases)
1.3. Alzheimer’s Disease (AD): Overview and Pathology
1.4. Epidemiological Link Between TBI and Neurodegenerative Diseases
1.5. Aim of the Review
2. Results
2.1. Physical and Neuropathological Consequences of TBI
2.1.1. Types and Severity of TBI
2.1.2. Acute Phase of TBI
2.1.3. Chronic Phase of TBI
2.1.4. Diffuse Axonal Injury (DAI)
2.1.5. Chronic Traumatic Encephalopathy (Neuropathological Features, Clinical Manifestation, Risk Factors, Diagnosis)
2.2. Molecular and Cellular Mechanisms Linking TBI to AD
2.2.1. Blood -Brain Barrier Dysfunction
2.2.2. Chronic Vascular Changes
2.2.3. Excitotoxicity
2.2.4. Oxidative Stress
2.2.5. Mitochondrial Dysfunction and Ferroptosis
2.2.6. Coagulopathy and Microvascular Hemorrhage
2.2.7. Cerebral Blood Flow Changes
2.2.8. Genetic Susceptibility Linking TBI and Alzheimer’s Disease
2.3. Clinical Presentation of TBI-Related Neurodegeneration
2.4. Risk Factors
2.4.1. Age
2.4.2. Sex and Gender
2.4.3. Vascular and Systemic Comorbidities
2.4.4. Lifestyle Exposures
2.4.5. Sleep and Neuroinflammation
2.4.6. Neurovascular Dysfunction
2.5. Biomarker Evidence
2.5.1. Fluid Biomarkers
2.5.2. Imaging Biomarkers
2.5.3. Comparison to Alzheimer’s Disease Biomarker Profiles
2.5.4. Neuroinflammation as a Mediator of Tau Pathology
2.5.5. Evidence from Animal Models Linking TBI to Neurodegeneration
2.6. Causal Relationship Between TBI and AD
2.6.1. Epidemiological Evidence Linking TBI to Dementia and Alzheimer’s Disease
2.6.2. Temporality of the Relationship
2.6.3. Biological Plausibility
| Post-TBI Neurodegeneration | Chronic Traumatic Encephalopathy (CTE) | Alzheimer’s Disease (AD) | |
|---|---|---|---|
| Initiating Events | Single or repetitive TBI across the severity spectrum (mild-severe), including blast exposure | Repetitive head impacts (RHI), often sub-concussive, associated with contact sports or military blast exposure | Multifactorial etiology involving advanced age, genetic susceptibility (APOε4, TREM2 variants) |
| Neuroanatomical Topography | Selective vulnerability of long association and white matter tracts, including corpus callosum, internal capsule with diffuse involvement of deep and periventricular white-matter networks | Predominant involvement of cortical sulcal depths, especially in frontal and temporal cortices, with a characteristic perivascular distribution and extends to limbic structures and brainstem in advanced stages. |
Early involvement of medial temporal love structures (hippocampus and entorhinal cortex), followed by spread to temporoparietal association cortices. |
| Predominant Pathology | Diffuse Axonal Injury and microvascular disruption. Chronic cases may demonstrate mixed proteinopathy and accumulation of tau and β- amyloid | Pathognomonic perivascular accumulation of hyperphosphorylated tau at sulcal depths, involving both neurons and astrocytes, accompanied by frequent TDP-43 co-pathology in the medial temporal lobe, diencephalon and progressive degeneration of white matter |
Accumulation of extracellular neuritic β-amyloid plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau, beginning in the entorhinal cortex and hippocampus and spreading to temporoparietal and frontal association |
| Imaging | White-matter atrophy, reduced fractional anisotropy on diffusion MRI and microbleeds on susceptibility-weighted imaging, patterns of cortical atrophy |
Regional cortical thinning and sulcal atrophy have been described, alongside tau PET signal in characteristic sulcal and perivascular regions, while standard MRI may be non-specific | Hippocampal and temporoparietal cortical atrophy on structural MRI, positive amyloid and tau PET, imaging consistent with established AD biomarker patterns |
2.7. Therapeutic Interventions
2.7.1. Stem Cell-Based Approaches for BBB and Neural Repair
2.7.2. Natural Compounds as Neuroprotective Agents
2.7.3. Targeting Mitochondrial Dysfunction
2.7.4. Transcriptional and Hormonal Modulation
2.7.5. Immunomodulation and Neuroinflammation
2.7.6. Pharmacological Interventions in Acute and Chronic TBI
2.7.7. Neurovascular-Linked Therapies
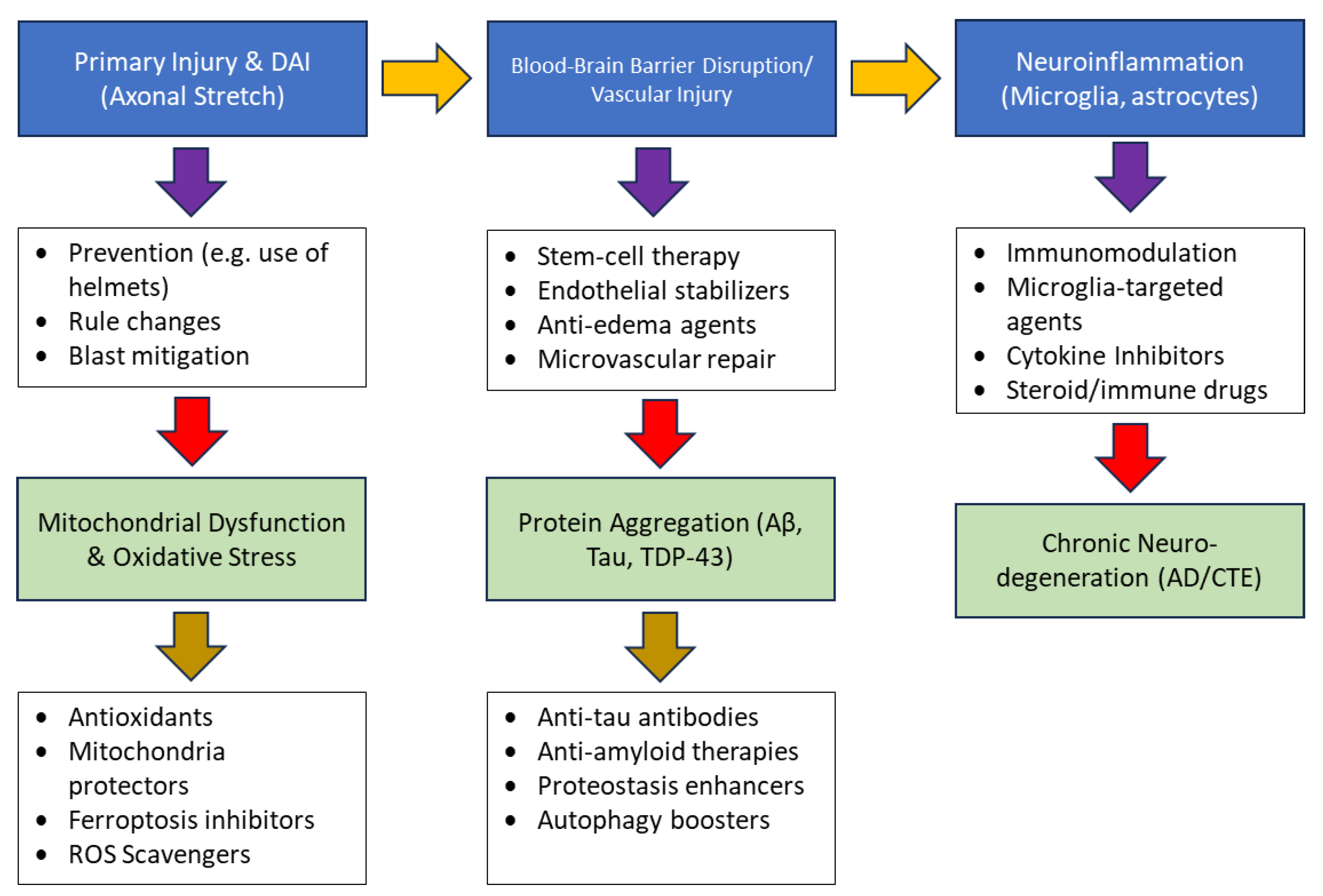
3. Discussion
4. Methods
4.1. Review Design and Rationale
4.2. Eligibility Criteria
4.3. Databases Assessed
4.4. Search Strategy
4.5. Screening and Selection Process
4.6. Data Extraction
4.7. Data Synthesis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TBI | Traumatic brain injury |
| CTE | Chronic traumatic encephalopathy |
| AD | Alzheimer’s disease |
| DAI | Diffuse axonal injury |
| BBB | Blood-brain barrier |
| ApoE | Apolipoprotein E |
| CSF | Cerebrospinal fluid |
| GCS | Glascow Coma Scale |
| CBI-M | Clinical, Biomarker, Imaging Modifiers |
| Aβ | Amyloid-beta |
| NFTs | Neurofibrillary tangles |
| ROS | Reactive oxygen species |
| CNS | Central nervous system |
| VaD | Vascular dementia |
| FTLD | Frontotemporal lobar degeneration |
| APP | Amyloid precursor protein |
| PD | Parkinson’s disease |
| MND | Motor neuron disease |
| MRI | Magnetic resonance imaging |
| BACE1 | β-secretase |
| FA | Fractional anisotropy |
| FLAIR | Fluid-attenuated inversion recovery |
| LOC | Loss of consciousness |
| NMDA | N-methyl-D-aspartate |
| AMPA | α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid |
| DTI | Diffusion tensor imaging |
| FA | Fractional anisotropy |
| RHI | Repetitive head impacts |
| TDP-43 | Transactive response DNA-binding protein 43 |
| TES | Traumatic encephalopathy syndrome |
| TMEM106B | Transmembrane protein 106B |
| sTREM 2 | Soluble triggering receptor expressed on myeloid cells 2 |
| ICAM | Intercellular adhesion molecule |
| VCAM-1 | Vascular Cell Adhesion Molecule-1 |
| VEGF | Vascular endothelial growth factor |
| Cav-1 | Caveolin-1 |
| LRP1 | Low-density lipoprotein receptor-related protein 1 |
| CypA | Cyclophilin A |
| MMP-9 | Matrix metalloproteinase |
| Mfsd2a | Major facilitator superfamily domain-containing protein 2A |
| Wnt | Wingless/Integrated |
| VSMCs | Vascular smooth muscle cells |
| α-SMA | Alpha-smooth muscle actin |
| β-CTF | Beta C-terminal fragment of APP |
| ADAM10 | A disintegrin and metalloproteinase domain-containing protein 10 |
| RNS | Reactive nitrogen species |
| OXPHOS | Oxidative phosphorylation |
| ATP | Adenosine triphosphate |
| CISD1 | CDGSH iron–sulfur domain-containing protein 1 |
| DHODH | Dihydroorotate dehydrogenase |
| CBF | Cerebral blood flow |
| TOMM40 | Translocase of outer mitochondrial membrane 40 |
| BAG3 | BCL2-associated athanogene 3 |
| GWAS | Genome-wide association studies |
| TREM2 | Triggering receptor expressed on myeloid cells 2 |
| TGF-β | Transforming growth factor beta |
| Shh | Sonic hedgehog |
| mTOR | Mechanistic (mammalian) target of rapamycin |
| TBI-ND | Traumatic brain injury-related neurodegeneration |
| RR | Risk ratio |
| Brain-PAG | Brain-predicted age gaps |
| IL | Interleukin |
| TNF | Tumor necrosis factor |
| NVU | Neurovascular unit |
| T-tau | Total tau |
| P-tau | Phosphorylated tau |
| NfL | Neurofilament light chain |
| GFAP | Glial fibrillary acidic protein |
| UCH-L1 | Ubiquitin carboxy-terminal hydrolase L1 |
| NSE | Neuron specific enolase |
| SRC | Sports-related concussion |
| PSEN1 | Presenilin-1 gene |
| CCL2 | C-C motif chemokine ligand 2 |
| PET | Positron emission tomography |
| bvFTD | Behavioral variant frontotemporal dementia |
| TBI | Traumatic brain injury |
| SNAP-25 | Synaptosomal-associated protein 25 |
| VILIP-1 | Visinin-like protein 1 |
| MAG | Myelin-associated glycoprotein |
| NLRP3 | NOD-, LRR-, and pyrin domain-containing protein 3 |
| NFκB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| Fli-1 | Friend leukemia integration 1 |
| TCR | T cell receptor |
| ZIPK | Zipper-interacting protein kinase |
| OTCs | Orthopedic trauma controls |
| OR | Odds ratio |
| NSCs | Neural stem cells |
| EPCs | Endothelial progenitor cells |
| HSCs | Hematopoietic stem cells |
| MSCs | Mesenchymal stem cells |
| PS1 | Presenilin-1 protein |
| HD | Huntington’s disease |
| ALS | Amyotrophic lateral sclerosis |
| T4 | Thyroxine |
| T3 | Triiodothyronine |
| BDNF | Brain-derived neurotrophic factor |
| GDNF | Glial cell line-derived neurotrophic factor |
| IFN-γ | Interferon-gamma |
| mAbs | Monoclonal antibodies |
| ICP | Intracranial pressure |
| FFP | Fresh frozen plasma |
| DOACs | Direct oral anticoagulants |
| SSRIs | Selective serotonin reuptake inhibitors |
| TCAs | Tricyclic antidepressants |
| 3K3A-APC | 3K3A-activated protein C |
| PCC | Population-Concept-Context |
| MeSH | Medical Subject Headings |
References
- National Academies of Sciences E; Division H and M; Services B on HC. Injury C on the R of the D of VAE for TB. Diagnosis and Assessment of Traumatic Brain Injury. National Academies Press (US), 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK542595/.
- Zhong, H.; Feng, Y.; Shen, J.; Rao, T.; Dai, H.; Zhong, W.; Zhao, G. Global Burden of Traumatic Brain Injury in 204 Countries and Territories From 1990 to 2021. Am. J. Prev. Med. 2025, 68, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.I.R.; Menon, D.K.; Manley, G.T.; Abrams, M.; Åkerlund, C.; Andelic, N.; Aries, M.; Bashford, T.; Bell, M.J.; Bodien, Y.G.; et al. Traumatic brain injury: Progress and challenges in prevention, clinical care, and research. Lancet Neurol. 2022, 21, 1004–1060. [Google Scholar] [CrossRef] [PubMed]
- James, S.L.; Theadom, A.; Ellenbogen, R.G.; Bannick, M.S.; Montjoy-Venning, W.; Lucchesi, L.R.; Abbasi, N.; Abdulkader, R.; Abraha, H.N.; Adsuar, J.C.; et al. Global, regional, and national burden of traumatic brain injury and spinal cord injury, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 56–87. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.C.; Robinson, M.E. Military-related traumatic brain injury and neurodegeneration. Alzheimer’s Dement. 2014, 10, S242–S253. [Google Scholar] [CrossRef]
- Leo, P.; McCrea, M. Epidemiology. In PubMed; 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK326730/.
- Jha, R.M.; Kochanek, P.M.; Simard, J.M. Pathophysiology and treatment of cerebral edema in traumatic brain injury. Neuropharmacology 2019, 145, 230–246. [Google Scholar] [CrossRef]
- Reddipaga, M.; Kannan, K. On the construction of a viscoelastic constitutive model for brain tissue maximizing tension–compression asymmetry. Int. J. Eng. Sci. 2025, 218. [Google Scholar] [CrossRef]
- Keating, C.E.; Cullen, D.K. Mechanosensation in traumatic brain injury. Neurobiol. Dis. 2020, 148, 105210. [Google Scholar] [CrossRef]
- Cullen, D.K.; Vernekar, V.N.; LaPlaca, M.C. Trauma-Induced Plasmalemma Disruptions in Three-Dimensional Neural Cultures Are Dependent on Strain Modality and Rate. J. Neurotrauma 2011, 28, 2219–2233. [Google Scholar] [CrossRef]
- Du, Z.; Wang, P.; Luo, P.; Fei, Z.; Zhuang, Z.; Liu, Z. Mechanical mechanism and indicator of diffuse axonal injury under blast-type acceleration. J. Biomech. 2023, 156, 111674. [Google Scholar] [CrossRef]
- Mesfin, F.B.; Taylor, R.S. Diffuse axonal injury (DAI). In Nih.gov; 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK448102/.
- Smith, D.H.; Meaney, D.F.; Shull, W.H. Diffuse Axonal Injury in Head Trauma. J. Head Trauma Rehabilitation 2003, 18, 307–316. [Google Scholar] [CrossRef]
- Johnson, V.E.; Stewart, W.; Smith, D.H. Axonal pathology in traumatic brain injury. Exp. Neurol. 2013, 246, 35–43. [Google Scholar] [CrossRef]
- Zima, L.; Moore, A.N.; Smolen, P.; Kobori, N.; Noble, B.; Robinson, D.; Hood, K.N.; Homma, R.; Al Mamun, A.; Redell, J.B.; et al. The evolving pathophysiology of TBI and the advantages of temporally-guided combination therapies. Neurochem. Int. 2024, 180, 105874. [Google Scholar] [CrossRef] [PubMed]
- Mena, J.H.; Sanchez, A.I.; Rubiano, A.M.; Peitzman, A.B.; Sperry, J.L.; Gutierrez, M.I.M.; Puyana, J.C. Effect of the Modified Glasgow Coma Scale Score Criteria for Mild Traumatic Brain Injury on Mortality Prediction: Comparing Classic and Modified Glasgow Coma Scale Score Model Scores of 13. J. Trauma Inj. Infect. Crit. Care 2011, 71, 1185–1193. [Google Scholar] [CrossRef]
- Manley, G.T.; Dams-O’cOnnor, K.; Alosco, M.L.; O Awwad, H.; Bazarian, J.J.; Bragge, P.; Corrigan, J.D.; Doperalski, A.; Ferguson, A.R.; Mac Donald, C.L.; et al. A new characterisation of acute traumatic brain injury: The NIH-NINDS TBI Classification and Nomenclature Initiative. Lancet Neurol. 2025, 24, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.Y.; Lee, A.Y.W. Traumatic Brain Injuries: Pathophysiology and Potential Therapeutic Targets. Front. Cell. Neurosci. 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Su, E.; Bell, M. Diffuse Axonal Injury. Nih.gov. 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK326722/.
- Palacios, E.M.; Owen, J.P.; Yuh, E.L.; Wang, M.B.; Vassar, M.J.; Ferguson, A.R.; Diaz-Arrastia, R.; Giacino, J.T.; Okonkwo, D.O.; Robertson, C.S.; et al. The evolution of white matter microstructural changes after mild traumatic brain injury: A longitudinal DTI and NODDI study. Sci. Adv. 2020, 6, eaaz6892. [Google Scholar] [CrossRef]
- Freire, M.A.M.; Rocha, G.S.; Bittencourt, L.O.; Falcao, D.; Lima, R.R.; Cavalcanti, J.R.L.P. Cellular and Molecular Pathophysiology of Traumatic Brain Injury: What Have We Learned So Far? Biology 2023, 12, 1139. [Google Scholar] [CrossRef]
- Ganti, L.; Bodhit, A.N.; Daneshvar, Y.; Hatchitt, K.; Kuchibhotla, S.; Pulvino, C.; Ayala, S.W.; Peters, K.R. Effectiveness of seatbelts in mitigating traumatic brain injury severity. World J. Emerg. Med. 2021, 12, 68–72. [Google Scholar] [CrossRef]
- Hinson, H.E.; Rowell, S.M.; Schreiber, M. Clinical evidence of inflammation driving secondary brain injury. J. Trauma Acute Care Surg. 2015, 78, 184–191. [Google Scholar] [CrossRef]
- Tesco, G.; Lomoio, S. Pathophysiology of neurodegenerative diseases: An interplay among axonal transport failure, oxidative stress, and inflammation? Semin. Immunol. 2022, 59, 101628. [Google Scholar] [CrossRef]
- Wilson, L.; Stewart, W.; Dams-O’COnnor, K.; Diaz-Arrastia, R.; Horton, L.; Menon, D.K.; Polinder, S. The chronic and evolving neurological consequences of traumatic brain injury. Lancet Neurol. 2017, 16, 813–825. [Google Scholar] [CrossRef]
- Devi, Y.; Khan, S.; Rana, P.; Deepak, D.; Dhandapani, M.; Ghai, S.; Gopichandran, L.; Dhandapani, S. Cognitive, Behavioral, and Functional Impairments among Traumatic Brain Injury Survivors: Impact on Caregiver Burden. J. Neurosci. Rural. Pr. 2020, 11, 629–635. [Google Scholar] [CrossRef]
- Aoun, R.; Rawal, H.; Attarian, H.; Sahni, A. Impact of traumatic brain injury on sleep: An overview. Nat. Sci. Sleep 2019, ume 11, 131–140. [Google Scholar] [CrossRef]
- Scheltens, P.; De Strooper, B.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s disease. Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef]
- Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2024, 20, 3708–3821. [CrossRef] [PubMed]
- Kamatham, P.T.; Shukla, R.; Khatri, D.K.; Vora, L.K. Pathogenesis, diagnostics, and therapeutics for Alzheimer’s disease: Breaking the memory barrier. Ageing Res. Rev. 2024, 101, 102481. [Google Scholar] [CrossRef] [PubMed]
- Iaccarino, L.; Burnham, S.; Dell’AGnello, G.; Dowsett, S.; Epelbaum, S. Diagnostic Biomarkers of Amyloid and Tau Pathology in Alzheimer’s Disease: An Overview of Tests for Clinical Practice in the United States and Europe. J. Prev. Alzheimer’s Dis. 2023, 10, 426–442. [Google Scholar] [CrossRef] [PubMed]
- Weerasinghe-Mudiyanselage, P.D.E.; Ang, M.J.; Kang, S.; Kim, J.-S.; Moon, C. Structural Plasticity of the Hippocampus in Neurodegenerative Diseases. Int. J. Mol. Sci. 2022, 23, 3349. [Google Scholar] [CrossRef]
- Barone, E.; Di Domenico, F.; Perluigi, M.; Butterfield, D.A. The interplay among oxidative stress, brain insulin resistance and AMPK dysfunction contribute to neurodegeneration in type 2 diabetes and Alzheimer disease. Free. Radic. Biol. Med. 2021, 176, 16–33. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhao, G.; Zhao, Y. Dysregulation of energy metabolism in Alzheimer’s disease. J. Neurol. 2024, 272, 1–14. [Google Scholar] [CrossRef]
- He, L.; Zhang, R.; Yang, M.; Lu, M. The role of astrocyte in neuroinflammation in traumatic brain injury. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2023, 1870, 166992. [Google Scholar] [CrossRef]
- Brett, B.L.; Gardner, R.C.; Godbout, J.; Dams-O’cOnnor, K.; Keene, C.D. Traumatic Brain Injury and Risk of Neurodegenerative Disorder. Biol. Psychiatry 2022, 91, 498–507. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.E.; Byers, A.L.; Gardner, R.C.; Seal, K.H.; Boscardin, W.J.; Yaffe, K. Association of Mild Traumatic Brain Injury With and Without Loss of Consciousness With Dementia in US Military Veterans. JAMA Neurol. 2018, 75, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.; Vasefi, M. Increased Risk of Dementia and its Subtypes following Various Forms of Acquired Brain Injury: A Meta-analysis and Systematic Review. Neuroscience 2025, 580, 332–350. [Google Scholar] [CrossRef] [PubMed]
- Werner, C.; Engelhard, K. Pathophysiology of traumatic brain injury. Br. J. Anaesth. 2007, 99, 4–9. [Google Scholar] [CrossRef]
- Acosta, S.A.; Tajiri, N.; Sanberg, P.R.; Kaneko, Y.; Borlongan, C.V. Increased Amyloid Precursor Protein and Tau Expression Manifests as Key Secondary Cell Death in Chronic Traumatic Brain Injury. J. Cell. Physiol. 2016, 232, 665–677. [Google Scholar] [CrossRef]
- Delic, V.; Beck, K.D.; Pang, K.C.H.; Citron, B.A. Biological links between traumatic brain injury and Parkinson’s disease. Acta Neuropathol. Commun. 2020, 8, 1–16. [Google Scholar] [CrossRef]
- Evans, T.; Jaramillo, C.; Sataranatarajan, K.; Watts, L.; Sabia, M.; Qi, W.; Van Remmen, H. The effect of mild traumatic brain injury on peripheral nervous system pathology in wild-type mice and the G93A mutant mouse model of motor neuron disease. Neuroscience 2015, 298, 410–423. [Google Scholar] [CrossRef]
- Siedler, D.G.; Chuah, M.I.; Kirkcaldie, M.T.K.; Vickers, J.C.; King, A.E. Diffuse axonal injury in brain trauma: Insights from alterations in neurofilaments. Front. Cell. Neurosci. 2014, 8, 429. [Google Scholar] [CrossRef]
- Graham, N.S.N.; Jolly, A.; Zimmerman, K.; Bourke, N.J.; Scott, G.; Cole, J.H.; Schott, J.M.; Sharp, D.J. Diffuse axonal injury predicts neurodegeneration after moderate–severe traumatic brain injury. Brain 2020, 143, 3685–3698. [Google Scholar] [CrossRef]
- Santurro, A.; De Simone, M.; Choucha, A.; Morena, D.; Consalvo, F.; Romano, D.; Terrasi, P.; Corrivetti, F.; Scrofani, R.; Narciso, N.; et al. Integrative Diagnostic and Prognostic Paradigms in Diffuse Axonal Injury: Insights from Clinical, Histopathological, Biomolecular, Radiological, and AI-Based Perspectives. Int. J. Mol. Sci. 2025, 26, 7808. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-H.; Chiang, C.-W.; Perez-Torres, C.J.; Sun, P.; Wallendorf, M.; Schmidt, R.E.; Cross, A.H.; Song, S.-K. Diffusion MRI quantifies early axonal loss in the presence of nerve swelling. J. Neuroinflammation 2017, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schallerer, C.; Neuschmid, S.; Ehrlich, B.E.; McGuone, D. Calpain in Traumatic Brain Injury: From Cinderella to Central Player. Cells 2025, 14, 1253. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Y.-W.; Wang, X.; Zhang, H.; You, X.; Liao, F.-F.; Xu, H. Intracellular Trafficking of Presenilin 1 Is Regulated by β-Amyloid Precursor Protein and Phospholipase D1. J. Biol. Chem. 2009, 284, 12145–12152. [Google Scholar] [CrossRef]
- Rodrigues, E.M.; Weissmiller, A.M.; Goldstein, L.S. Enhanced -secretase processing alters APP axonal transport and leads to axonal defects. Hum. Mol. Genet. 2012, 21, 4587–4601. [Google Scholar] [CrossRef]
- Albayram, O.; Angeli, P.; Bernstein, E.; et al. Targeting Prion-like Cis Phosphorylated Tau Pathology in Neurodegenerative Diseases. J. Alzheimer’s Dis. Park. 2018, 8, 443. [Google Scholar] [CrossRef]
- McKenzie, K.J.; McLellan, D.R.; Gentleman, S.M.; Maxwell, W.L.; Gennarelli, T.A.; Graham, D.I. Is β-APP a marker of axonal damage in short-surviving head injury? Acta Neuropathol. 1996, 92, 608–613. [Google Scholar] [CrossRef]
- Bruggeman, G.F.; Haitsma, I.K.; Dirven, C.M.F.; Volovici, V. Traumatic axonal injury (TAI): Definitions, pathophysiology and imaging—a narrative review. Acta Neurochir. 2020, 163, 31–44. [Google Scholar] [CrossRef]
- Johnson, V.E.; Stewart, J.E.; Begbie, F.D.; Trojanowski, J.Q.; Smith, D.H.; Stewart, W. Inflammation and white matter degeneration persist for years after a single traumatic brain injury. Brain 2013, 136, 28–42. [Google Scholar] [CrossRef]
- Graham, N.S.; Cole, J.H.; Bourke, N.J.; Schott, J.M.; Sharp, D.J. Distinct patterns of neurodegeneration after TBI and in Alzheimer’s disease. Alzheimer’s Dement. 2023, 19, 3065–3077. [Google Scholar] [CrossRef]
- Rutgers, D.; Toulgoat, F.; Cazejust, J.; Fillard, P.; Lasjaunias, P.; Ducreux, D. White Matter Abnormalities in Mild Traumatic Brain Injury: A Diffusion Tensor Imaging Study. Am. J. Neuroradiol. 2007, 29, 514–519. [Google Scholar] [CrossRef]
- Hayes, J.P.; Bigler, E.D.; Verfaellie, M. Traumatic Brain Injury as a Disorder of Brain Connectivity. J. Int. Neuropsychol. Soc. 2016, 22, 120–137. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.H.; Jolly, A.; De Simoni, S.; Bourke, N.; Patel, M.C.; Scott, G.; Sharp, D.J. Spatial patterns of progressive brain volume loss after moderate-severe traumatic brain injury. Brain 2018, 141, 822–836. [Google Scholar] [CrossRef] [PubMed]
- Poudel, G.R.; Dominguez, J.F.D.; Verhelst, H.; Linden, C.V.; Deblaere, K.; Jones, D.K.; Cerin, E.; Vingerhoets, G.; Caeyenberghs, K. Network diffusion modeling predicts neurodegeneration in traumatic brain injury. Ann. Clin. Transl. Neurol. 2020, 7, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Mckee, A.C.; Daneshvar, D.H. Chapter 4 - The neuropathology of traumatic brain injury. ScienceDirect. 1 January 2015. Available online: https://www.sciencedirect.com/science/article/abs/pii/B9780444528926000040.
- Permenter, C.M.; Sherman, A.L. Postconcussive syndrome. In PubMed; 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK534786/.
- Pellot, J.E.; De Jesus, O. Cerebral Contusion. In PubMed; 2020. Available online: https://www.ncbi.nlm.nih.gov/books/NBK562147/.
- Ginsburg, J.; Huff, J.S. Traumatic Brain Injury. In PubMed; 17 February 2025. Available online: https://www.ncbi.nlm.nih.gov/books/NBK557861/.
- Koerte, I.K.; Hufschmidt, J.; Muehlmann, M.; Lin, A.P.; Shenton, M.E. Advanced Neuroimaging of Mild Traumatic Brain Injury. In Nih.gov; 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK326714/.
- Dong, X.; Wang, Y.; Qin, Z. Molecular Mechanisms of Excitotoxicity and Their Relevance to Pathogenesis of Neurodegenerative Diseases. Acta Pharmacol Sin 2009, 30, 379–387. [Google Scholar] [CrossRef]
- Hardingham, G.E.; Fukunaga, Y.; Bading, H. Extrasynaptic NMDARs oppose synaptic NMDARs by triggering CREB shut-off and cell death pathways. Nat. Neurosci. 2002, 5, 405–414. [Google Scholar] [CrossRef]
- Liang, D.; Bhatta, S.; Gerzanich, V.; Simard, J.M. Cytotoxic edema: Mechanisms of pathological cell swelling. Neurosurg. Focus 2007, 22, 1–9. [Google Scholar] [CrossRef]
- Archie, S.R.; Al Shoyaib, A.; Cucullo, L. Blood-Brain Barrier Dysfunction in CNS Disorders and Putative Therapeutic Targets: An Overview. Pharmaceutics 2021, 13, 1779. [Google Scholar] [CrossRef]
- Yang, C.; Hawkins, K.E.; Doré, S.; Candelario-Jalil, E. Neuroinflammatory mechanisms of blood-brain barrier damage in ischemic stroke. Am. J. Physiol. Cell Physiol. 2019, 316, C135–C153. [Google Scholar] [CrossRef]
- Donat, C.K.; Scott, G.; Gentleman, S.M.; Sastre, M. Microglial Activation in Traumatic Brain Injury. Front. Aging Neurosci. 2017, 9, 208. [Google Scholar] [CrossRef]
- Seehaus, A.; Roebroeck, A.; Bastiani, M.; Fonseca, L.; Bratzke, H.; Lori, N.; Vilanova, A.; Goebel, R.; Galuske, R. Histological validation of high-resolution DTI in human post mortem tissue. Front. Neuroanat. 2015, 9, 98. [Google Scholar] [CrossRef]
- Tsitsopoulos, P.P.; Marklund, N. Amyloid-β Peptides and Tau Protein as Biomarkers in Cerebrospinal and Interstitial Fluid Following Traumatic Brain Injury: A Review of Experimental and Clinical Studies. Front. Neurol. 2013, 4, 79. [Google Scholar] [CrossRef] [PubMed]
- A Tagge, C.; Fisher, A.M.; Minaeva, O.V.; Gaudreau-Balderrama, A.; A Moncaster, J.; Zhang, X.-L.; Wojnarowicz, M.W.; Casey, N.; Lu, H.; Kokiko-Cochran, O.N.; et al. Concussion, microvascular injury, and early tauopathy in young athletes after impact head injury and an impact concussion mouse model. Brain 2018, 141, 422–458. [Google Scholar] [CrossRef]
- Kilinc, D.; Gallo, G.; Barbee, K.A. Mechanical membrane injury induces axonal beading through localized activation of calpain. Exp. Neurol. 2009, 219, 553–561. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.C.; Alosco, M.L.; Huber, B.R. Repetitive Head Impacts and Chronic Traumatic Encephalopathy. Neurosurg. Clin. North Am. 2016, 27, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Martland, H.S. PUNCH DRUNK. JAMA 1928, 91, 1103–1107. [Google Scholar] [CrossRef]
- Baugh, C.M.; Robbins, C.A.; Stern, R.A.; McKee, A.C. Current Understanding of Chronic Traumatic Encephalopathy. Curr. Treat. Options Neurol. 2014, 16, 1–13. [Google Scholar] [CrossRef]
- Yi, J.; Padalino, D.J.; Chin, L.S.; Montenegro, P.; Cantu, R.C. Chronic Traumatic Encephalopathy. Optom. Vis. Sci. 2013, 12, 28–32. [Google Scholar] [CrossRef]
- Castellani, R.J.; Perry, G. Dementia Pugilistica Revisited. J. Alzheimer’s Dis. 2017, 60, 1209–1221. [Google Scholar] [CrossRef]
- Corsellis, J.A.N.; Bruton, C.J.; Freeman-Browne, D. The aftermath of boxing. Psychol. Med. 1973, 3, 270–303. [Google Scholar] [CrossRef]
- McKee, A.C.; Cantu, R.C.; Nowinski, C.J.; Hedley-Whyte, E.T.; Gavett, B.E.; Budson, A.E.; Santini, V.E.; Lee, H.-S.; Kubilus, C.A.; Stern, R.A. Chronic Traumatic Encephalopathy in Athletes: Progressive Tauopathy After Repetitive Head Injury. J. Neuropathol. Exp. Neurol. 2009, 68, 709–735. [Google Scholar] [CrossRef]
- McKee, A.C.; Stein, T.D.; Nowinski, C.J.; Stern, R.A.; Daneshvar, D.H.; Alvarez, V.E.; Lee, H.-S.; Hall, G.; Wojtowicz, S.M.; Baugh, C.M.; et al. The spectrum of disease in chronic traumatic encephalopathy. Brain 2012, 136, 43–64. [Google Scholar] [CrossRef]
- McKee, A.C.; Stein, T.D.; Kiernan, P.T.; Alvarez, V.E. The Neuropathology of Chronic Traumatic Encephalopathy. Brain Pathol. 2015, 25, 350–364. [Google Scholar] [CrossRef]
- Osterman, C.; Hamlin, D.; Suter, C.M.; Affleck, A.J.; Gloss, B.S.; Turner, C.P.; Faull, R.L.M.; Stein, T.D.; McKee, A.; Buckland, M.E.; et al. Perivascular glial reactivity is a feature of phosphorylated tau lesions in chronic traumatic encephalopathy. Acta Neuropathol. 2025, 149, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Falcon, B.; Zivanov, J.; Zhang, W.; Murzin, A.G.; Garringer, H.J.; Vidal, R.; Crowther, R.A.; Newell, K.L.; Ghetti, B.; Goedert, M.; et al. Novel tau filament fold in chronic traumatic encephalopathy encloses hydrophobic molecules. Nature 2019, 568, 420–423. [Google Scholar] [CrossRef]
- Hay, J.; Johnson, V.E.; Smith, D.H.; Stewart, W. Chronic Traumatic Encephalopathy: The Neuropathological Legacy of Traumatic Brain Injury. Annu. Rev. Pathol. Mech. Dis. 2016, 11, 21–45. [Google Scholar] [CrossRef] [PubMed]
- Murray, H.C.; Osterman, C.; Bell, P.; Vinnell, L.; Curtis, M.A. Neuropathology in chronic traumatic encephalopathy: A systematic review of comparative post-mortem histology literature. Acta Neuropathol. Commun. 2022, 10, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Nicks, R.; Shah, A.; Stathas, S.A.; Kirsch, D.; Horowitz, S.M.; Saltiel, N.; Calderazzo, S.M.; Butler, M.L.M.D.; Cormier, K.A.; Aytan, N.; et al. Neurodegeneration in the cortical sulcus is a feature of chronic traumatic encephalopathy and associated with repetitive head impacts. Acta Neuropathol. 2024, 148, 1–16. [Google Scholar] [CrossRef]
- Fukuda, A.M.; Badaut, J. Aquaporin 4: A player in cerebral edema and neuroinflammation. J. Neuroinflammation 2012, 9, 279. [Google Scholar] [CrossRef]
- Gardner, A.; Iverson, G.L.; McCrory, P. Chronic traumatic encephalopathy in sport: A systematic review. Br. J. Sports Med. 2013, 48, 84–90. [Google Scholar] [CrossRef]
- Stern, R.A.; Daneshvar, D.H.; Baugh, C.M.; Seichepine, D.R.; Montenigro, P.H.; Riley, D.O.; Fritts, N.G.; Stamm, J.M.; Robbins, C.A.; McHale, L.; et al. Clinical presentation of chronic traumatic encephalopathy. Neurology 2013, 81, 1122–1129. [Google Scholar] [CrossRef] [PubMed]
- Fesharaki-Zadeh, A. Chronic Traumatic Encephalopathy: A Brief Overview. Front. Neurol. 2019, 10, 713. [Google Scholar] [CrossRef] [PubMed]
- Gavett, B.E.; Stern, R.A.; McKee, A.C. Chronic Traumatic Encephalopathy: A Potential Late Effect of Sport-Related Concussive and Subconcussive Head Trauma. Clin. Sports Med. 2011, 30, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Tharmaratnam, T.; Iskandar, M.A.; Tabobondung, T.C.; Tobbia, I.; Gopee-Ramanan, P.; Tabobondung, T.A. Chronic Traumatic Encephalopathy in Professional American Football Players: Where Are We Now? Front. Neurol. 2018, 9, 445. [Google Scholar] [CrossRef]
- Montenigro, P.H.; Baugh, C.M.; Daneshvar, D.H.; Mez, J.; E Budson, A.; Au, R.; I Katz, D.; Cantu, R.C.; A Stern, R. Clinical subtypes of chronic traumatic encephalopathy: Literature review and proposed research diagnostic criteria for traumatic encephalopathy syndrome. Alzheimer’s Res. Ther. 2014, 6, 68. [Google Scholar] [CrossRef]
- Mez, J.; Daneshvar, D.H.; Kiernan, P.T.; Abdolmohammadi, B.; Alvarez, V.E.; Huber, B.R.; Alosco, M.L.; Solomon, T.M.; Nowinski, C.J.; McHale, L.; et al. Clinicopathological Evaluation of Chronic Traumatic Encephalopathy in Players of American Football. In JAMA; 2017; 318, pp. 360–370. Available online: https://pubmed.ncbi.nlm.nih.gov/28742910/.
- Alosco, M.L.; Mez, J.; Tripodis, Y.; Kiernan, P.T.; Abdolmohammadi, B.; Murphy, L.; Kowall, N.W.; Stein, T.D.; Huber, B.R.; Goldstein, L.E.; et al. Age of first exposure to tackle football and chronic traumatic encephalopathy. Ann. Neurol. 2018, 83, 886–901. [Google Scholar] [CrossRef]
- Cherry, J.D.; Mez, J.; Crary, J.F.; Tripodis, Y.; Alvarez, V.E.; Mahar, I.; Huber, B.R.; Alosco, M.L.; Nicks, R.; Abdolmohammadi, B.; et al. Variation in TMEM106B in chronic traumatic encephalopathy. Acta Neuropathol. Commun. 2018, 6, 1–9. [Google Scholar] [CrossRef]
- Al-Rubaie, A. Traumatic Brain Injury and Dementia: Mechanisms, Risk Stratification, and Clinical Management. J. Clin. Neurol. 2025, 21, 265–276. [Google Scholar] [CrossRef]
- van Amerongen, S.; Pulukuri, S.V.; Tuz-Zahra, F.; Tripodis, Y.; Cherry, J.D.; Bernick, C.; Geda, Y.E.; Wethe, J.V.; Katz, D.I.; Alosco, M.L.; et al. Inflammatory biomarkers for neurobehavioral dysregulation in former American football players: Findings from the DIAGNOSE CTE Research Project. J. Neuroinflammation 2024, 21, 1–15. [Google Scholar] [CrossRef]
- Alosco, M.L.; Tripodis, Y.; Fritts, N.G.; Heslegrave, A.; Baugh, C.M.; Conneely, S.; Mariani, M.; Martin, B.M.; Frank, S.; Mez, J.; et al. Cerebrospinal fluid tau, Aβ, and sTREM2 in Former National Football League Players: Modeling the relationship between repetitive head impacts, microglial activation, and neurodegeneration. Alzheimer’s Dement. 2018, 14, 1159–1170. [Google Scholar] [CrossRef]
- Wolburg, H.; Noell, S.; Mack, A.; Wolburg-Buchholz, K.; Fallier-Becker, P. Brain endothelial cells and the glio-vascular complex. Cell Tissue Res. 2008, 335, 75–96. [Google Scholar] [CrossRef]
- Li, Z.; Li, M.; Fang, Z.; Wang, H. Immunological Mechanisms and Therapeutic Strategies in Cerebral Ischemia–Reperfusion Injury: From Inflammatory Response to Neurorepair. Int. J. Mol. Sci. 2025, 26, 8336. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.-A.; Yan, L.; Wen, J.; Satyanarayanan, S.K.; Yu, F.; Lu, J.; Liu, Y.U.; Su, H. Cellular and molecular mechanisms in vascular repair after traumatic brain injury: A narrative review. Burn. Trauma 2023, 11, tkad033. [Google Scholar] [CrossRef] [PubMed]
- Argaw, A.T.; Gurfein, B.T.; Zhang, Y.; Zameer, A.; John, G.R. VEGF-mediated disruption of endothelial CLN-5 promotes blood-brain barrier breakdown. Proc. Natl. Acad. Sci. 2009, 106, 1977–1982. [Google Scholar] [CrossRef] [PubMed]
- Winkler, E.A.; Sagare, A.P.; Zlokovic, B.V. The Pericyte: A Forgotten Cell Type with Important Implications for Alzheimer’s Disease? Brain Pathol. 2014, 24, 371–386. [Google Scholar] [CrossRef]
- Sun, B.; Li, L.; Harris, O.A.; Luo, J. Blood–brain barrier disruption: A pervasive driver and mechanistic link between traumatic brain injury and Alzheimer’s disease. Neurodegener. 2025, 14, 1–22. [Google Scholar] [CrossRef]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 2018, 14, 133–150. [Google Scholar] [CrossRef]
- Ikonomovic, M.D.; Mi, Z.; Abrahamson, E.E. Disordered APP metabolism and neurovasculature in trauma and aging: Combined risks for chronic neurodegenerative disorders. Ageing Res. Rev. 2017, 34, 51–63. [Google Scholar] [CrossRef]
- Nelson, A.R.; Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Neurovascular dysfunction and neurodegeneration in dementia and Alzheimer’s disease. Biochim. et Biophys. Acta (BBA) Mol. Basis Dis. 2016, 1862, 887–900. [Google Scholar] [CrossRef]
- Vázquez-Rosa, E.; Shin, M.-K.; Dhar, M.; Chaubey, K.; Cintrón-Pérez, C.J.; Tang, X.; Liao, X.; Miller, E.; Koh, Y.; Barker, S.; et al. P7C3-A20 treatment one year after TBI in mice repairs the blood–brain barrier, arrests chronic neurodegeneration, and restores cognition. Proc. Natl. Acad. Sci. 2020, 117, 27667–27675. [Google Scholar] [CrossRef]
- Zhao, T.; Pan, P.; Jia, Y.; Zhou, Y.; Zhang, X.; Guan, H.; Li, Q.; Zhou, Y. Crossing Pathological Boundaries: Multi-Target Restoration of the Neurovascular Unit in Alzheimer’s and Vascular Dementia—From Modern Therapeutics to Traditional Chinese Medicine. Aging Dis. 2025. [Google Scholar] [CrossRef]
- Manukjan, N.; Chau, S.; Caiment, F.; van Herwijnen, M.; Smeets, H.J.; Fulton, D.; Ahmed, Z.; Blankesteijn, W.M.; Foulquier, S. Wnt7a Decreases Brain Endothelial Barrier Function Via β-Catenin Activation. Mol. Neurobiol. 2023, 61, 4854–4867. [Google Scholar] [CrossRef] [PubMed]
- Özen, I.; Abu Hamdeh, S.; Ruscher, K.; Marklund, N. Traumatic brain injury causes early aggregation of beta-amyloid peptides and NOTCH3 reduction in vascular smooth muscle cells of leptomeningeal arteries. Acta Neuropathol. 2025, 149, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Hayes, G.; Pinto, J.; Sparks, S.N.; Wang, C.; Suri, S.; Bulte, D.P. Vascular smooth muscle cell dysfunction in neurodegeneration. Front. Neurosci. 2022, 16, 1010164. [Google Scholar] [CrossRef]
- Baracaldo-Santamaría, D.; Ariza-Salamanca, D.F.; Corrales-Hernández, M.G.; Pachón-Londoño, M.J.; Hernandez-Duarte, I.; Calderon-Ospina, C.-A. Revisiting Excitotoxicity in Traumatic Brain Injury: From Bench to Bedside. Pharmaceutics 2022, 14, 152. [Google Scholar] [CrossRef] [PubMed]
- Guerriero, R.M.; Giza, C.C.; Rotenberg, A. Glutamate and GABA Imbalance Following Traumatic Brain Injury. Curr. Neurol. Neurosci. Rep. 2015, 15, 1–11. [Google Scholar] [CrossRef]
- Kirdajova, D.B.; Kriska, J.; Tureckova, J.; Anderova, M. Ischemia-Triggered Glutamate Excitotoxicity From the Perspective of Glial Cells. Front. Cell. Neurosci. 2020, 14, 51. [Google Scholar] [CrossRef]
- Angeloni, C.; Prata, C.; Sega, F.V.D.; Piperno, R.; Hrelia, S. Traumatic Brain Injury and NADPH Oxidase: A Deep Relationship. Oxidative Med. Cell. Longev. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Toledano-Díaz, A.; Álvarez, M.I.; Toledano, A. The relationships between neuroglial alterations and neuronal changes in Alzheimer’s disease, and the related controversies I: Gliopathogenesis and glioprotection. J. Central Nerv. Syst. Dis. 2022, 14. [Google Scholar] [CrossRef]
- Feissner, R.F.; Skalska, J.; E Gaum, W.; Sheu, S.-S. Crosstalk signaling between mitochondrial Ca2+ and ROS. Front. Biosci. 2009, ume, 1197–1218. [Google Scholar] [CrossRef]
- Wei, Z.; Yu, H.; Zhao, H.; Wei, M.; Xing, H.; Pei, J.; Yang, Y.; Ren, K. Broadening horizons: Ferroptosis as a new target for traumatic brain injury. Burn. Trauma 2024, 12, tkad051. [Google Scholar] [CrossRef]
- Li, J.; Jia, B.; Xu, Y.; Zhao, Y.; Wang, S.; Yang, R.; Su, L.; Zeng, X.; Li, Q.; Luo, C. Inhibition of mitoNEET ameliorates traumatic brain injury-induced ferroptosis and cognitive dysfunction by stabilizing dihydroorotate dehydrogenase. Exp. Neurol. 2025, 389, 115235. [Google Scholar] [CrossRef]
- Mantash, S.; Aboulouard, S.; Dakik, H.; Zirem, Y.; Ziane-Chaouche, L.; Nehme, A.; Mallah, K.; El-Kurdi, M.; Ramadan, N.; Fournier, I.; et al. Uncovering injury-specific proteomic signatures and neurodegenerative risks in single and repetitive traumatic brain injury. Signal Transduct. Target. Ther. 2025, 10, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.; Li, L.; Schäfer, N.; Huang, Q.; Maegele, M.; Gu, Z. Endothelial glycocalyx in traumatic brain injury associated coagulopathy: Potential mechanisms and impact. J. Neuroinflammation 2021, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, F.; Dong, J.-F. Coagulopathy induced by traumatic brain injury: Systemic manifestation of a localized injury. Blood 2018, 131, 2001–2006. [Google Scholar] [CrossRef] [PubMed]
- Carminita, E.; Tourn, J.; Crescence, L.; Brouilly, N.; Merrill-Skoloff, G.; Mazharian, A.; Dubois, C.; Panicot-Dubois, L. A thrombus is formed by a gradient of platelet activation and procoagulant endothelium. Res. Pr. Thromb. Haemost. 2023, 7, 102209. [Google Scholar] [CrossRef]
- Barry, M.; Pati, S. Targeting repair of the vascular endothelium and glycocalyx after traumatic injury with plasma and platelet resuscitation. Matrix Biol. Plus 2022, 14, 100107. [Google Scholar] [CrossRef]
- Xu, L.; Ware, J.B.; Kim, J.J.; Shahim, P.; Silverman, E.; Magdamo, B.; Dabrowski, C.; Wesley, L.; Le, M.D.; Morrison, J.; et al. Arterial Spin Labeling Reveals Elevated Cerebral Blood Flow with Distinct Clusters of Hypo- and Hyperperfusion after Traumatic Brain Injury. J. Neurotrauma 2021, 38, 2538–2548. [Google Scholar] [CrossRef]
- Toth, P.; Szarka, N.; Farkas, E.; Ezer, E.; Czeiter, E.; Amrein, K.; Ungvari, Z.; Hartings, J.A.; Buki, A.; Koller, A. Traumatic brain injury-induced autoregulatory dysfunction and spreading depression-related neurovascular uncoupling: Pathomechanisms, perspectives, and therapeutic implications. Am. J. Physiol. Circ. Physiol. 2016, 311, H1118–H1131. [Google Scholar] [CrossRef]
- Hartmann, D.A.; Coelho-Santos, V.; Shih, A.Y. Pericyte Control of Blood Flow Across Microvascular Zones in the Central Nervous System. Annu. Rev. Physiol. 2022, 84, 331–354. [Google Scholar] [CrossRef]
- Grubb, S.; Lauritzen, M.; Aalkjær, C. Brain capillary pericytes and neurovascular coupling. Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol. 2021, 254, 110893. [Google Scholar] [CrossRef]
- Hamilton, N.B.; Attwell, D.; Hall, C.N. Pericyte-mediated regulation of capillary diameter: A component of neurovascular coupling in health and disease. Front. Neuroenergetics 2010, 2. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Klomparens, K. Updates on the association of brain injury and Alzheimer’s disease. Brain Circ. 2020, 6, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Bamel, K.; Panwar, A.; Kumar, M.; Kumar, S.; Sharma, V.; Sharma, A. Traumatic brain injury as a precursor to neurodegenerative diseases: Mechanisms linking TBI to Alzheimer’s disease. Brain Disord. 2025, 18. [Google Scholar] [CrossRef]
- Dong, G.; Ma, C.C.; Mao, S.; Brown, K.S.-M.; Naik, S.M.; McDonough, G.A.; Wijethunga, S.P.; Kim, J.; Kirkham, S.L.; Shao, D.D.; et al. Diverse somatic genomic alterations in single neurons in chronic traumatic encephalopathy. Science 2025, 390, eadu1351. [Google Scholar] [CrossRef]
- Liu, C.-C.; Kanekiyo, T.; Xu, H.; Bu, G. Apolipoprotein E and Alzheimer disease: Risk, mechanisms and therapy. Nat. Rev. Neurol. Correction in Nat. Rev. Neurol. 2013, 9, 184.. 2013, 9, 106–118. [Google Scholar] [CrossRef]
- Mayeux, R.; Ottman, R.; Maestre, G.; Ngai, C.; Tang, M.-X.; Ginsberg, H.; Chun, M.; Tycko, B.; Shelanski, M. Synergistic Effects of Traumatic Head Injury and Apolipoprotein-epsilon4 in Patients With Alzheimer’s Disease. Neurology 1995, 45, 555–557. [Google Scholar] [CrossRef]
- McFadyen, C.A.; Zeiler, F.A.; Newcombe, V.; Synnot, A.; Steyerberg, E.; Gruen, R.L.; Rosand, J.; Palotie, A.; Maas, A.I.; Menon, D.K. Apolipoprotein E4 Polymorphism and Outcomes from Traumatic Brain Injury: A Living Systematic Review and Meta-Analysis. J. Neurotrauma 2021, 38, 1124–1136. [Google Scholar] [CrossRef]
- Blumenfeld, J.; Yip, O.; Kim, M.J.; Huang, Y. Cell type-specific roles of APOE4 in Alzheimer disease. Nat. Rev. Neurosci. 2024, 25, 91–110. [Google Scholar] [CrossRef]
- LoBue, C.; Woon, F.L.; Rossetti, H.C.; Hynan, L.S.; Hart, J.; Cullum, C.M. Traumatic brain injury history and progression from mild cognitive impairment to Alzheimer disease. Neuropsychology 2018, 32, 401–409. [Google Scholar] [CrossRef]
- Xu, J.; Duan, J.; Cai, Z.; et al. TOMM40-APOE chimera linking Alzheimer’s highest risk genes: A new pathway for mitochondria regulation and APOE4 pathogenesis. bioRxiv (Cold Spring Harbor Laboratory). Published online. 10 October 2024. [CrossRef]
- Sweeney, N.; Kim, T.Y.; Morrison, C.T.; Li, L.; Acosta, D.; Liang, J.; Datla, N.V.; Fitzgerald, J.A.; Huang, H.; Liu, X.; et al. Neuronal BAG3 attenuates tau hyperphosphorylation, synaptic dysfunction, and cognitive deficits induced by traumatic brain injury via the regulation of autophagy-lysosome pathway. Acta Neuropathol. 2024, 148, 1–27. [Google Scholar] [CrossRef]
- Grimm, M.O.W.; Mett, J.; Stahlmann, C.P.; Haupenthal, V.J.; Zimmer, V.C.; Hartmann, T. Neprilysin and Aβ Clearance: Impact of the APP Intracellular Domain in NEP Regulation and Implications in Alzheimer’s Disease. Front. Aging Neurosci. 2013, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.K.; Ansari, S.; Maurya, V.K.; Kumar, S.; Sharma, D.; Malhotra, H.S.; Tiwari, S.; Srivastava, C.; Paweska, J.T.; Abdel-Moneim, A.S.; et al. Neprilysin-Mediated Amyloid Beta Clearance and Its Therapeutic Implications in Neurodegenerative Disorders. ACS Pharmacol. Transl. Sci. 2024, 7, 3645–3657. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, R.; Wojtas, A.; Bras, J.; Carrasquillo, M.; Rogaeva, E.; Majounie, E.; Cruchaga, C.; Sassi, C.; Kauwe, J.S.; Younkin, S.; et al. TREM2 Variants in Alzheimer’s Disease. N. Engl. J. Med. 2013, 368, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Luo, J. TGF-β as a Key Modulator of Astrocyte Reactivity: Disease Relevance and Therapeutic Implications. Biomedicines 2022, 10, 1206. [Google Scholar] [CrossRef]
- Macura, I.J.; Zivanovic, A.; Perovic, M.; Ciric, J.; Major, T.; Kanazir, S.; Ivkovic, S. The Expression of Major Facilitator Superfamily Domain-Containing Protein2a (Mfsd2a) and Aquaporin 4 Is Altered in the Retinas of a 5xFAD Mouse Model of Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 14092. [Google Scholar] [CrossRef]
- Ben-Zvi, A.; Lacoste, B.; Kur, E.; Andreone, B.J.; Mayshar, Y.; Yan, H.; Gu, C. Mfsd2a is critical for the formation and function of the blood–brain barrier. Nature 2014, 509, 507–511. [Google Scholar] [CrossRef]
- Salehi, A.; Jullienne, A.; Baghchechi, M.; Hamer, M.; Walsworth, M.; Donovan, V.; Tang, J.; Zhang, J.H.; Pearce, W.J.; Obenaus, A. Up-regulation of Wnt/β-catenin expression is accompanied with vascular repair after traumatic brain injury. J. Cereb. Blood Flow Metab. 2017, 38, 274–289. [Google Scholar] [CrossRef]
- Zou, D.; Hu, W.; Qin, J.; Wei, Z.; Wang, D.; Li, L. Rapid orderly migration of neutrophils after traumatic brain injury depends on MMP9/13. Biochem. Biophys. Res. Commun. 2021, 579, 161–167. [Google Scholar] [CrossRef]
- Wu, J.; He, J.; Tian, X.; Zhong, J.; Li, H.; Sun, X. Activation of the Hedgehog Pathway Promotes Recovery of Neurological Function After Traumatic Brain Injury by Protecting the Neurovascular Unit. Transl. Stroke Res. 2020, 11, 720–733. [Google Scholar] [CrossRef]
- Razak, L.H.A.; Denis, T.; Murugiah, Y.; Yoong, W.K.; Idris, Z.; Senik, M. The Effect of Traumatic Brain Injury on Memory. Malays. J. Med Sci. 2024, 31, 52–74. [Google Scholar] [CrossRef]
- Paterno, R.; Folweiler, K.A.; Cohen, A.S. Pathophysiology and Treatment of Memory Dysfunction After Traumatic Brain Injury. Curr. Neurol. Neurosci. Rep. 2017, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ozga, J.E.; Povroznik, J.M.; Engler-Chiurazzi, E.B.; Haar, C.V. Executive (dys)function after traumatic brain injury: Special considerations for behavioral pharmacology. Behav. Pharmacol. 2018, 29, 617–637. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, M.G.d.F.; Faleiro, R.M.; de Paula, J.J.; Kummer, A.; Caramelli, P.; Teixeira, A.L.; de Souza, L.C.; Miranda, A.S. Cognitive Impairment Following Acute Mild Traumatic Brain Injury. Front. Neurol. 2019, 10, 198. [Google Scholar] [CrossRef] [PubMed]
- Howlett, J.R.; Nelson, L.D.; Stein, M.B. Mental Health Consequences of Traumatic Brain Injury. Biol. Psychiatry 2021, 91, 413–420. [Google Scholar] [CrossRef]
- Chan, A.; Ouyang, J.; Nguyen, K.; Jones, A.; Basso, S.; Karasik, R.; Chan, \. Traumatic brain injuries: A neuropsychological review. Front. Behav. Neurosci. 2024, 18, 1326115. [Google Scholar] [CrossRef]
- Sundstrom, A.; Nilsson, L.-G.; Cruts, M.; Adolfsson, R.; Van Broeckhoven, C.; Nyberg, L. Increased risk of dementia following mild head injury for carriers but not for non-carriers of the APOE e4 allele. Int. Psychogeriatrics 2006, 19, 159–165. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Li, X.; Zhang, S.; Zhao, J.; Zhu, X.; Tian, G. Head Injury as a Risk Factor for Dementia and Alzheimer’s Disease: A Systematic Review and Meta-Analysis of 32 Observational Studies. PLOS ONE 2017, 12, e0169650. [Google Scholar] [CrossRef]
- Gardner, R.C.; Burke, J.F.; Nettiksimmons, J.; Kaup, A.; Barnes, D.E.; Yaffe, K. Dementia Risk After Traumatic Brain Injury vs Nonbrain Trauma: The Role of Age and Severity. JAMA Neurol. 2014, 71, 1490–1497. [Google Scholar] [CrossRef]
- Winter, L.; Moriarty, H.P.; Robinson, K.M.; Leiby, B.E.; Schmidt, K.; Whitehouse, C.R.P.; Swanson, R.L.D. Age Suppresses the Association Between Traumatic Brain Injury Severity and Functional Outcomes: A Study Using the NIDILRR TBIMS Dataset. J. Head Trauma Rehabilitation 2024, 39, E582–E590. [Google Scholar] [CrossRef]
- Zhang, X.; Pan, Y.; Wu, T.; Zhao, W.; Zhang, H.; Ding, J.; Ji, Q.; Jia, X.; Li, X.; Lee, Z.; et al. Brain age prediction using interpretable multi-feature-based convolutional neural network in mild traumatic brain injury. NeuroImage 2024, 297. [Google Scholar] [CrossRef] [PubMed]
- Gupte, R.P.; Brooks, W.M.; Vukas, R.R.; Pierce, J.D.; Harris, J.L. Sex Differences in Traumatic Brain Injury: What We Know and What We Should Know. J. Neurotrauma 2019, 36, 3063–3091. [Google Scholar] [CrossRef] [PubMed]
- Farace, E.; Alves, W.M. Do women fare worse? A metaanalysis of gender differences in outcome after traumatic brain injury. Neurosurg. Focus 2000, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Broshek, D.K.; Kaushik, T.; Freeman, J.R.; Erlanger, D.; Webbe, F.; Barth, J.T. Sex differences in outcome following sports-related concussion. J. Neurosurg. 2005, 102, 856–863. [Google Scholar] [CrossRef]
- Schneider, A.L.C.; Barber, J.; Temkin, N.; Gardner, R.C.; Manley, G.; Diaz-Arrastia, R.; Sandsmark, D. for the TRACK-TBI Study Investigators Associations of Preexisting Vascular Risk Factors With Outcomes After Traumatic Brain Injury: A TRACK-TBI Study. J. Head Trauma Rehabilitation 2022, 38, E88–E98. [Google Scholar] [CrossRef]
- Csiszar, A.; Tarantini, S.; Fülöp, G.A.; Kiss, T.; Valcarcel-Ares, M.N.; Galvan, V.; Ungvari, Z.; Yabluchanskiy, A. Hypertension impairs neurovascular coupling and promotes microvascular injury: Role in exacerbation of Alzheimer’s disease. GeroScience 2017, 39, 359–372. [Google Scholar] [CrossRef]
- Tatara, Y.; Shimada, R.; Kibayashi, K. Effects of Preexisting Diabetes Mellitus on the Severity of Traumatic Brain Injury. J. Neurotrauma 2021, 38, 886–902. [Google Scholar] [CrossRef]
- Wu, A.; Molteni, R.; Ying, Z.; Gomez-Pinilla, F. A saturated-fat diet aggravates the outcome of traumatic brain injury on hippocampal plasticity and cognitive function by reducing brain-derived neurotrophic factor. Neuroscience 2003, 119, 365–375. [Google Scholar] [CrossRef]
- Sivandzade, F.; Alqahtani, F.; Sifat, A.; Cucullo, L. The cerebrovascular and neurological impact of chronic smoking on post-traumatic brain injury outcome and recovery: An in vivo study. J. Neuroinflammation 2020, 17, 1–18. [Google Scholar] [CrossRef]
- Hu, H.; Fu, J.-T.; Bi, Y.-L.; Ma, Y.-H.; Huang, Y.-Y.; Wang, X.; Tan, L.; Yu, J.-T. Tau Pathologies Mediate the Association of Cigarette Smoking with Cognitive Impairment in Older Adults Without Dementia: The CABLE Study. J. Alzheimer’s Dis. 2022, 86, 1849–1859. [Google Scholar] [CrossRef]
- Brzecka, A.; Leszek, J.; Ashraf, G.M.; Ejma, M.; Ávila-Rodriguez, M.F.; Yarla, N.S.; Tarasov, V.V.; Chubarev, V.N.; Samsonova, A.N.; Barreto, G.E.; et al. Sleep Disorders Associated With Alzheimer’s Disease: A Perspective. Front. Neurosci. 2018, 12, 330. [Google Scholar] [CrossRef] [PubMed]
- Houle, S.; Tapp, Z.; Dobres, S.; Ahsan, S.; Reyes, Y.; Cotter, C.; Mitsch, J.; Zimomra, Z.; Peng, J.; Rowe, R.K.; et al. Sleep fragmentation after traumatic brain injury impairs behavior and conveys long-lasting impacts on neuroinflammation. Brain, Behav. Immun. - Heal. 2024, 38, 100797. [Google Scholar] [CrossRef] [PubMed]
- Green, T.R.F.; Ortiz, J.B.; Wonnacott, S.; Williams, R.J.; Rowe, R.K. The Bidirectional Relationship Between Sleep and Inflammation Links Traumatic Brain Injury and Alzheimer’s Disease. Front. Neurosci. 2020, 14. [Google Scholar] [CrossRef] [PubMed]
- Daigle, B.; Bevilacqua, L.M.; Noël, V.P.; Lebel, M.; Côté, D.; Lévesque, M.; Menard, C. Neurogliovascular alterations in brain pathologies across lifespan. Glial Heal. Res. 2024, 1. [Google Scholar] [CrossRef]
- Zetterberg, H.; Blennow, K. Fluid biomarkers for mild traumatic brain injury and related conditions. Nat. Rev. Neurol. 2016, 12, 563–574. [Google Scholar] [CrossRef]
- Singh, A.K.; Asif, S.; Pandey, D.K.; Chaudhary, A.; Kapoor, V.; Verma, P.K. Biomarkers in Acute Traumatic Brain Injury: A Systematic Review and Meta-Analysis. Cureus 2024, 16. [Google Scholar] [CrossRef]
- Huebschmann, N.A.; Luoto, T.M.; Karr, J.E.; Berghem, K.; Blennow, K.; Zetterberg, H.; Ashton, N.J.; Simrén, J.; Posti, J.P.; Gill, J.M.; et al. Comparing Glial Fibrillary Acidic Protein (GFAP) in Serum and Plasma Following Mild Traumatic Brain Injury in Older Adults. Front. Neurol. 2020, 11. [Google Scholar] [CrossRef]
- Seidenfaden, S.-C.; Kjerulff, J.L.; Juul, N.; Kirkegaard, H.; Møller, M.F.; Münster, A.-M.B.; Bøtker, M.T. Temporal Changes in Serum S100B Levels From Prehospital to Early In-Hospital Sampling in Patients Suffering Traumatic Brain Injury. Front. Neurol. 2022, 13, 800015. [Google Scholar] [CrossRef]
- Spaziani, G.; Rozzi, G.; Baroni, S.; Simeoni, B.; Racco, S.; Barone, F.; Fuorlo, M.; Franceschi, F.; Covino, M. From the Emergency Department to Follow-Up: Clinical Utility of Biomarkers in Mild Traumatic Brain Injury. Emerg. Care Med. 2025, 2, 45. [Google Scholar] [CrossRef]
- Whitehouse, D.P.; Monteiro, M.; Czeiter, E.; Vyvere, T.V.; Valerio, F.; Ye, Z.; Amrein, K.; Kamnitsas, K.; Xu, H.; Yang, Z.; et al. Relationship of admission blood proteomic biomarkers levels to lesion type and lesion burden in traumatic brain injury: A CENTER-TBI study. EBioMedicine 2021, 75, 103777. [Google Scholar] [CrossRef]
- Shahim, P.; Tegner, Y.; Marklund, N.; Blennow, K.; Zetterberg, H. Neurofilament light and tau as blood biomarkers for sports-related concussion. Neurology 2018, 90, e1780–e1788. [Google Scholar] [CrossRef]
- Li, L.M.; Kodosaki, E.; Heslegrave, A.; Zetterberg, H.; Graham, N.; Zimmerman, K.; Soreq, E.; Parker, T.; Garbero, E.; Moro, F.; et al. High-dimensional proteomic analysis for pathophysiological classification of traumatic brain injury. Brain 2024, 148, 1015–1030. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.S.N.; A Zimmerman, K.; Hain, J.; Rooney, E.; Lee, Y.; Del Giovane, M.; Parker, T.; Wilson, M.G.; Malhotra, P.; David, M.C.B.; et al. Biomarker evidence of neurodegeneration in mid-life former rugby players. Brain 2025, 148, 2684–2697. [Google Scholar] [CrossRef] [PubMed]
- Fantoni, E.; Collij, L.; Alves, I.L.; Buckley, C.; Farrar, G. The Spatial-Temporal Ordering of Amyloid Pathology and Opportunities for PET Imaging. J. Nucl. Med. 2019, 61, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R., Jr.; Knopman, D.S.; Jagust, W.J.; Petersen, R.C.; Weiner, M.W.; Aisen, P.S.; Shaw, L.M.; Vemuri, P.; Wiste, H.J.; Weigand, S.D.; et al. Tracking pathophysiological processes in Alzheimer’s disease: An updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013, 12, 207–216. [Google Scholar] [CrossRef]
- Thal, D.R.; Beach, T.G.; Zanette, M.; Heurling, K.; Chakrabarty, A.; Ismail, A.; Smith, A.P.; Buckley, C. [18F]flutemetamol amyloid positron emission tomography in preclinical and symptomatic Alzheimer’s disease: Specific detection of advanced phases of amyloid-β pathology. Alzheimer’s Dement. 2015, 11, 975–985. [Google Scholar] [CrossRef]
- Eagle, S.R.; Puccio, A.; Svirsky, S.; Mountz, J.; Laymon, C.; Borasso, A.; Henry, L.; Okonkwo, D.O. Identifying a Biological Signature of Trauma-Related Neurodegeneration Following Repeated Traumatic Brain Injuries Compared with Healthy Controls. Neurotrauma Rep. 2025, 6, 560–568. [Google Scholar] [CrossRef]
- Macdonald, C.; Dikranian, K.; Song, S.; Bayly, P.; Holtzman, D.; Brody, D. Detection of traumatic axonal injury with diffusion tensor imaging in a mouse model of traumatic brain injury. Exp. Neurol. 2007, 205, 116–131. [Google Scholar] [CrossRef]
- A Zimmerman, K.; Laverse, E.; Samra, R.; Lopez, M.Y.; E Jolly, A.; Bourke, N.J.; Graham, N.S.N.; Patel, M.C.; Hardy, J.; Kemp, S.; et al. White matter abnormalities in active elite adult rugby players. Brain Commun. 2021, 3, fcab133. [Google Scholar] [CrossRef]
- Wojtowicz, M.; Gardner, A.J.; Stanwell, P.; Zafonte, R.; Dickerson, B.C.; Iverson, G.L. Cortical thickness and subcortical brain volumes in professional rugby league players. NeuroImage: Clin. 2018, 18, 377–381. [Google Scholar] [CrossRef]
- A Zimmerman, K.; A Hain, J.; Graham, N.S.N.; Rooney, E.J.; Lee, Y.; Del-Giovane, M.; Parker, T.D.; Friedland, D.; Cross, M.J.; Kemp, S.; et al. Prospective cohort study of long-term neurological outcomes in retired elite athletes: The Advanced BiomaRker, Advanced Imaging and Neurocognitive (BRAIN) Health Study protocol. BMJ Open 2024, 14, e082902. [Google Scholar] [CrossRef] [PubMed]
- Raji, C.A.; Meysami, S.; Porter, V.R.; Merrill, D.A.; Mendez, M.F. Diagnostic utility of brain MRI volumetry in comparing traumatic brain injury, Alzheimer disease and behavioral variant frontotemporal dementia. BMC Neurol. 2024, 24, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Shetty, T.; Raince, A.; Manning, E.; Tsiouris, A.J. Imaging in Chronic Traumatic Encephalopathy and Traumatic Brain Injury. Sports Heal. A Multidiscip. Approach 2015, 8, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Legramante, J.M.; Minieri, M.; Belli, M.; Giovannelli, A.; Agnoli, A.; Bajo, D.; Bellincampi, L.; De Angelis, A.M.; Terrinoni, A.; Pieri, M.; et al. Evaluation of GFAP/UCH-L1 biomarkers for computed tomography exclusion in mild traumatic brain injury (mTBI). Int. J. Emerg. Med. 2024, 17, 1–7. [Google Scholar] [CrossRef]
- Park, Y.; Kc, N.; Paneque, A.; Cole, P.D. Tau, Glial Fibrillary Acidic Protein, and Neurofilament Light Chain as Brain Protein Biomarkers in Cerebrospinal Fluid and Blood for Diagnosis of Neurobiological Diseases. Int. J. Mol. Sci. 2024, 25, 6295. [Google Scholar] [CrossRef]
- Blennow, K.; Zetterberg, H. Biomarkers for Alzheimer’s disease: Current status and prospects for the future. J. Intern. Med. 2018, 284, 643–663. [Google Scholar] [CrossRef]
- Palmqvist, S.; Janelidze, S.; Stomrud, E.; Zetterberg, H.; Karl, J.; Zink, K.; Bittner, T.; Mattsson, N.; Eichenlaub, U.; Blennow, K.; et al. Performance of Fully Automated Plasma Assays as Screening Tests for Alzheimer Disease–Related β-Amyloid Status. JAMA Neurol. 2019, 76, 1060–1069. [Google Scholar] [CrossRef]
- Ashton, N.J.; Leuzy, A.; Karikari, T.K.; Mattsson-Carlgren, N.; Dodich, A.; Boccardi, M.; Corre, J.; Drzezga, A.; Nordberg, A.; Ossenkoppele, R.; et al. The validation status of blood biomarkers of amyloid and phospho-tau assessed with the 5-phase development framework for AD biomarkers. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2140–2156. [Google Scholar] [CrossRef]
- Yadikar, H.; Kobeissy, F.H.; Robertson, C.; Tsetsou, S.; Williamson, J.B.; Lamb, D.G.; Wagner, A.K.; Kibaugh, T.; Kao, S.-H.; Kou, Z.; et al. Early cerebrospinal fluid elevations of pTau-217 in severe traumatic brain injury subjects. Front. Neurol. 2025, 16, 1632679. [Google Scholar] [CrossRef]
- Shahim, P.; Politis, A.; van der Merwe, A.; Moore, B.; Ekanayake, V.; Lippa, S.M.; Chou, Y.-Y.; Pham, D.L.; Butman, J.A.; Diaz-Arrastia, R.; et al. Time course and diagnostic utility of NfL, tau, GFAP, and UCH-L1 in subacute and chronic TBI. Neurology 2020, 95, e623–e636. [Google Scholar] [CrossRef]
- Ramos-Cejudo, J.; Wisniewski, T.; Marmar, C.; Zetterberg, H.; Blennow, K.; de Leon, M.J.; Fossati, S. Traumatic Brain Injury and Alzheimer’s Disease: The Cerebrovascular Link. EBioMedicine 2018, 28, 21–30. [Google Scholar] [CrossRef]
- Johnson, V.E.; Stewart, W.; Smith, D.H. Traumatic brain injury and amyloid-β pathology: A link to Alzheimer’s disease? Nat. Rev. Neurosci. 2010, 11, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Friberg, S.; Lindblad, C.; Zeiler, F.A.; Zetterberg, H.; Granberg, T.; Svenningsson, P.; Piehl, F.; Thelin, E.P. Fluid biomarkers of chronic traumatic brain injury. Nat. Rev. Neurol. 2024, 20, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.S.; Blissitt, G.; Zimmerman, K.; Orton, L.; Friedland, D.; Coady, E.; Laban, R.; Veleva, E.; Heslegrave, A.J.; Zetterberg, H.; et al. Poor long-term outcomes and abnormal neurodegeneration biomarkers after military traumatic brain injury: The ADVANCE study. J. Neurol. Neurosurg. Psychiatry 2024, 96, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Asken, B.M.; Tanner, J.A.; VandeVrede, L.; Mantyh, W.G.; Casaletto, K.B.; Staffaroni, A.M.; La Joie, R.; Iaccarino, L.; Soleimani-Meigooni, D.; Rojas, J.C.; et al. Plasma P-tau181 and P-tau217 in Patients With Traumatic Encephalopathy Syndrome With and Without Evidence of Alzheimer Disease Pathology. Neurology 2022, 99, e594–e604. [Google Scholar] [CrossRef]
- Andersson, E.; Öst, M.; Dalla, K.; Zetterberg, H.; Blennow, K.; Nellgård, B. Acute-Phase Neurofilament Light and Glial Fibrillary Acidic Proteins in Cerebrospinal Fluid Predict Long-Term Outcome After Severe Traumatic Brain Injury. Neurocritical Care 2024, 41, 813–827. [Google Scholar] [CrossRef]
- Heuvel, F.O.; Li, Z.; Riedel, D.; Halbgebauer, S.; Oeckl, P.; Mayer, B.; Gotzman, N.; Shultz, S.; Semple, B.; Tumani, H.; et al. Dynamics of synaptic damage in severe traumatic brain injury revealed by cerebrospinal fluid SNAP-25 and VILIP-1. J. Neurol. Neurosurg. Psychiatry 2024, 95, 1158–1167. [Google Scholar] [CrossRef]
- Schindler, S.E.; Li, Y.; Todd, K.W.; Herries, E.M.; Henson, R.L.; Gray, J.D.; Wang, G.; Graham, D.L.; Shaw, L.M.; Trojanowski, J.Q.; et al. Emerging cerebrospinal fluid biomarkers in autosomal dominant Alzheimer’s disease. Alzheimer’s Dement. 2019, 15, 655–665. [Google Scholar] [CrossRef]
- Harting, M.T.; Jimenez, F.; Adams, S.D.; Mercer, D.W.; Cox, C.S., Jr. Acute, regional inflammatory response after traumatic brain injury: Implications for cellular therapy. Surgery 2008, 144, 803–813. [Google Scholar] [CrossRef]
- Needham, E.J.; Stoevesandt, O.; Thelin, E.P.; Zetterberg, H.; Zanier, E.R.; Al Nimer, F.; Ashton, N.J.; Outtrim, J.G.; Newcombe, V.F.J.; Mousa, H.S.; et al. Complex Autoantibody Responses Occur following Moderate to Severe Traumatic Brain Injury. J. Immunol. 2021, 207, 90–100. [Google Scholar] [CrossRef]
- Walker, A.; Chapin, B.; Abisambra, J.; DeKosky, S.T. Association between single moderate to severe traumatic brain injury and long-term tauopathy in humans and preclinical animal models: A systematic narrative review of the literature. Acta Neuropathol. Commun. 2022, 10, 1–20. [Google Scholar] [CrossRef]
- Zanier, E.R.; Bertani, I.; Sammali, E.; Pischiutta, F.; Chiaravalloti, M.A.; Vegliante, G.; Masone, A.; Corbelli, A.; Smith, D.H.; Menon, D.K.; et al. Induction of a transmissible tau pathology by traumatic brain injury. Brain 2018, 141, 2685–2699. [Google Scholar] [CrossRef] [PubMed]
- Johnson, V.E.; Stewart, W.; Smith, D.H. Widespread Tau and Amyloid-Beta Pathology Many Years After a Single Traumatic Brain Injury in Humans. Brain Pathol. 2011, 22, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Risling, M.; Smith, D.; Stein, T.D.; Thelin, E.P.; Zanier, E.R.; Ankarcrona, M.; Nilsson, P. Modelling human pathology of traumatic brain injury in animal models. J. Intern. Med. 2019, 285, 594–607. [Google Scholar] [CrossRef] [PubMed]
- Moloney, C.M.; Lowe, V.J.; Murray, M.E. Visualization of neurofibrillary tangle maturity in Alzheimer’s disease: A clinicopathologic perspective for biomarker research. Alzheimer’s Dement. 2021, 17, 1554–1574. [Google Scholar] [CrossRef]
- Collins-Praino, L.E.; Corrigan, F. Does neuroinflammation drive the relationship between tau hyperphosphorylation and dementia development following traumatic brain injury? Brain, Behav. Immun. 2017, 60, 369–382. [Google Scholar] [CrossRef]
- Griffin, W.T.; Sheng, J.G.; Gentleman, S.M.; Graham, D.I.; Mrak, R.E.; Roberts, G.W. Microglial interleukin-lα expression in human head injury: Correlations with neuronal and neuritic β-amyloid precursor protein expression. Neurosci. Lett. 1994, 176, 133–136. [Google Scholar] [CrossRef]
- Ising, C.; Venegas, C.; Zhang, S.; Scheiblich, H.; Schmidt, S.V.; Vieira-Saecker, A.; Schwartz, S.; Albasset, S.; McManus, R.M.; Tejera, D.; et al. NLRP3 inflammasome activation drives tau pathology. Nature 2019, 575, 669–673. [Google Scholar] [CrossRef]
- Cai, L.; Gong, Q.; Qi, L.; Xu, T.; Suo, Q.; Li, X.; Wang, W.; Jing, Y.; Yang, D.; Xu, Z.; et al. ACT001 attenuates microglia-mediated neuroinflammation after traumatic brain injury via inhibiting AKT/NFκB/NLRP3 pathway. Cell Commun. Signal. 2022, 20, 1–23. [Google Scholar] [CrossRef]
- Li, J.; Du, M.; Dong, G.; Hu, D.; Zhang, H.; Yu, Z.; Lu, Y.; Yang, Y. Early intervention with pericyte Fli-1 post-TBI attenuates hippocampal BBB disruption and subsequent neuroinflammation with neurological deficits. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2025, 1871, 167671. [Google Scholar] [CrossRef]
- Mouzon, B.C.; Bachmeier, C.; Ferro, A.; Ojo, J.; Crynen, G.; Acker, C.M.; Davies, P.; Mullan, M.; Stewart, W.; Crawford, F. Chronic neuropathological and neurobehavioral changes in a repetitive mild traumatic brain injury model. Ann. Neurol. 2013, 75, 241–254. [Google Scholar] [CrossRef]
- Ayerra, L.; Shumilov, K.; Ni, A.; Aymerich, M.S.; Friess, S.H.; Celorrio, M. Chronic traumatic brain injury induces neurodegeneration, neuroinflammation, and cognitive deficits in a T cell-dependent manner. Brain Res. 2025, 1850, 149446. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; She, F.; Zhang, L.; Kim, G.; Li, R.; Zheng, X.; Wang, Z.; Chen, R.; Wang, L.; Chen, D.; et al. Zipper-interacting protein kinase mediates neuronal cell death and cognitive dysfunction in traumatic brain injury via regulating DEDD. Cell Death Dis. 2025, 16, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lisi, I.; Moro, F.; Mazzone, E.; Marklund, N.; Pischiutta, F.; Kobeissy, F.; Mao, X.; Corrigan, F.; Helmy, A.; Nasrallah, F.; et al. Exploiting blood-based biomarkers to align preclinical models with human traumatic brain injury. Brain 2024, 148, 1062–1080. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Chen, X.-H.; Nonaka, M.; Trojanowski, J.Q.; Lee, V.-Y.; Saatman, K.E.; Leoni, M.J.; Xu, B.-N.; Wolf, J.A.; Meaney, D.F. Accumulation of Amyloid β and Tau and the Formation of Neurofilament Inclusions Following Diffuse Brain Injury in the Pig. J. Neuropathol. Exp. Neurol. 1999, 58, 982–992. [Google Scholar] [CrossRef]
- Liu, C.-C.; Zhao, N.; Fu, Y.; Wang, N.; Linares, C.; Tsai, C.-W.; Bu, G. ApoE4 Accelerates Early Seeding of Amyloid Pathology. Neuron 2017, 96, 1024–1032.e3. [Google Scholar] [CrossRef]
- Uryu, K.; Laurer, H.; McIntosh, T.; Praticò, D.; Martinez, D.; Leight, S.; Lee, V.M.-Y.; Trojanowski, J.Q. Repetitive Mild Brain Trauma Accelerates Aβ Deposition, Lipid Peroxidation, and Cognitive Impairment in a Transgenic Mouse Model of Alzheimer Amyloidosis. J. Neurosci. 2002, 22, 446–454. [Google Scholar] [CrossRef]
- Mielke, M.M.; Ransom, J.E.; Mandrekar, J.; Turcano, P.; Savica, R.; Brown, A.W. Traumatic Brain Injury and Risk of Alzheimer’s Disease and Related Dementias in the Population. J. Alzheimer’s Dis. 2022, 88, 1049–1059. [Google Scholar] [CrossRef]
- Russell, E.R.; Mackay, D.F.; Lyall, D.; Stewart, K.; A MacLean, J.; Robson, J.; Pell, J.P.; Stewart, W. Neurodegenerative disease risk among former international rugby union players. J. Neurol. Neurosurg. Psychiatry 2022, 93, 1262–1268. [Google Scholar] [CrossRef]
- Hume, P.A.; Theadom, A.; Lewis, G.N.; Quarrie, K.L.; Brown, S.R.; Hill, R.; Marshall, S.W. A Comparison of Cognitive Function in Former Rugby Union Players Compared with Former Non-Contact-Sport Players and the Impact of Concussion History. Sports Med. 2016, 47, 1209–1220. [Google Scholar] [CrossRef]
- Snowden, T.M.; Hinde, A.K.; Reid, H.M.; Christie, B.R. Does Mild Traumatic Brain Injury Increase the Risk for Dementia? A Systematic Review and Meta-Analysis. J. Alzheimer’s Dis. 2020, 78, 757–775. [Google Scholar] [CrossRef] [PubMed]
- Mavroudis, I.; Kazis, D.; Petridis, F.E.; Balmus, I.-M.; Papaliagkas, V.; Ciobica, A. The Association Between Traumatic Brain Injury and the Risk of Cognitive Decline: An Umbrella Systematic Review and Meta-Analysis. Brain Sci. 2024, 14, 1188. [Google Scholar] [CrossRef]
- Barnes, D.E.; Kaup, A.; Kirby, K.A.; Byers, A.L.; Diaz-Arrastia, R.; Yaffe, K. Traumatic brain injury and risk of dementia in older veterans. Neurology 2014, 83, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Saltiel, N.; Tripodis, Y.; Menzin, T.; Olaniyan, A.; Baucom, Z.; Yhang, E.; Palmisano, J.N.; Martin, B.; Uretsky, M.; Nair, E.; et al. Relative Contributions of Mixed Pathologies to Cognitive and Functional Symptoms in Brain Donors Exposed to Repetitive Head Impacts. Ann. Neurol. 2023, 95, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Brett, B.L.; Temkin, N.; Barber, J.; et al. Long-term Multi-domain Patterns of Change Following Traumatic Brain Injury: A TRACK-TBI LONG Study. Neurology Published online. 2023. [Google Scholar] [CrossRef]
- Hammond, F.M.; Hart, T.; Bushnik, T.; Corrigan, J.D.; Sasser, H. Change and Predictors of Change in Communication, Cognition, and Social Function Between 1 and 5 Years After Traumatic Brain Injury. J. Head Trauma Rehabilitation 2004, 19, 314–328. [Google Scholar] [CrossRef]
- Hanrahan, J.G.; Burford, C.; Nagappan, P.; Adegboyega, G.; Rajkumar, S.; Kolias, A.; Helmy, A.; Hutchinson, P.J. Is dementia more likely following traumatic brain injury? A systematic review. J. Neurol. 2023, 270, 3022–3051. [Google Scholar] [CrossRef]
- Roberts, G.; Gentleman, S.; Lynch, A.; Graham, D. βA4 amyloid protein deposition in brain after head trauma. Lancet 1991, 338, 1422–1423. [Google Scholar] [CrossRef]
- Dams-O’COnnor, K.; E Gibbons, L.; Bowen, J.D.; McCurry, S.M.; Larson, E.B.; Crane, P.K. Risk for late-life re-injury, dementia and death among individuals with traumatic brain injury: A population-based study. J. Neurol. Neurosurg. Psychiatry 2012, 84, 177–182. [Google Scholar] [CrossRef]
- Daneshvar, D.H.; Goldstein, L.E.; Kiernan, P.T.; Stein, T.D.; McKee, A.C. Post-traumatic neurodegeneration and chronic traumatic encephalopathy. Mol. Cell. Neurosci. 2015, 66, 81–90. [Google Scholar] [CrossRef]
- Guo, Z.; Cupples, L.; Kurz, A.; Auerbach, S.; Volicer, L.; Chui, H.; Green, R.; Sadovnick, A.; Duara, R.; DeCarli, C.; et al. Head injury and the risk of AD in the MIRAGE study. Neurology 2000, 54, 1316–1323. [Google Scholar] [CrossRef] [PubMed]
- Arciniega, H.; Baucom, Z.H.; Tuz-Zahra, F.; Tripodis, Y.; John, O.; Carrington, H.; Kim, N.; E Knyazhanskaya, E.; Jung, L.B.; Breedlove, K.; et al. Brain morphometry in former American football players: Findings from the DIAGNOSE CTE research project. Brain 2024, 147, 3596–3610. [Google Scholar] [CrossRef] [PubMed]
- Vaibhav, K.; Gulhane, M.; Ahluwalia, P.; Kumar, M.; Ahluwalia, M.; Rafiq, A.M.; Amble, V.; Zabala, M.G.; Miller, J.B.; Goldman, L.; et al. Single episode of moderate to severe traumatic brain injury leads to chronic neurological deficits and Alzheimer’s-like pathological dementia. GeroScience 2024, 46, 5439–5457. [Google Scholar] [CrossRef] [PubMed]
- Hicks, A.J.; Ponsford, J.L.; Spitz, G.; Dore, V.; Krishnadas, N.; Roberts, C.; Rowe, C.C. β-Amyloid and Tau Imaging in Chronic Traumatic Brain Injury. Neurology 2022, 99, E1131–E1141. [Google Scholar] [CrossRef]
- Fedak, K.M.; Bernal, A.; Capshaw, Z.A.; Gross, S. Applying the Bradford Hill criteria in the 21st century: How data integration has changed causal inference in molecular epidemiology. Emerg. Themes Epidemiology 2015, 12, 1–9. [Google Scholar] [CrossRef]
- Washington, P.M.; Villapol, S.; Burns, M.P. Polypathology and dementia after brain trauma: Does brain injury trigger distinct neurodegenerative diseases, or should they be classified together as traumatic encephalopathy? Exp. Neurol. 2016, 275, 381–388. [Google Scholar] [CrossRef]
- Emrani, S.; Koutures, A.; Tripodis, Y.; Uretsky, M.; Abdolmohammadi, B.; Nowinski, C.; Daneshvar, D.H.; Dwyer, B.; Katz, D.I.; Goldstein, L.E.; et al. Characterizing white matter and vascular pathologies in brain donors exposed to repetitive head impacts. Acta Neuropathol. 2025, 149, 1–16. [Google Scholar] [CrossRef]
- Chong, F.P.; Ng, K.Y.; Koh, R.Y.; Chye, S.M. Tau Proteins and Tauopathies in Alzheimer’s Disease. Cell. Mol. Neurobiol. 2018, 38, 965–980. [Google Scholar] [CrossRef]
- McGroarty, J.; Salinas, S.; Evans, H.; Jimenez, B.; Tran, V.; Kadavakollu, S.; Vashist, A.; Atluri, V. Inflammasome-Mediated Neuroinflammation: A Key Driver in Alzheimer’s Disease Pathogenesis. Biomolecules 2025, 15, 676. [Google Scholar] [CrossRef]
- Seplovich, G.; Bouchi, Y.; Vaccari, J.P.d.R.; Pareja, J.C.M.; Reisner, A.; Blackwell, L.; Mechref, Y.; Wang, K.K.; Tyndall, J.A.; Tharakan, B.; et al. Inflammasome links traumatic brain injury, chronic traumatic encephalopathy, and Alzheimer’s disease. Neural Regen. Res. 2024, 20, 1644–1664. [Google Scholar] [CrossRef]
- Powell, J.R.; Zong, X.; Weinstein, J.M.; DeLellis, S.M.; Kane, S.F.; Means, G.E.; Mihalik, J.P. Mild Traumatic Brain Injury and Career Stage Associate with Visible Perivascular Spaces in Special Operations Forces Soldiers. Ann. Biomed. Eng. 2024, 52, 2812–2817. [Google Scholar] [CrossRef] [PubMed]
- Tinney, E.M.; España-Irla, G.; Warren, A.E.; Whitehurst, L.N.; Stillman, A.M.; Hillman, C.H.; Morris, T.P.; Initiative, F.T.A.D.N. Axonal injury, sleep disturbances, and memory following traumatic brain injury. Ann. Clin. Transl. Neurol. 2024, 11, 2314–2326. [Google Scholar] [CrossRef] [PubMed]
- Kriukova, K.; Boswell, M.; Asifriyaz, T.; Gong, J.; Duncan, D.; Vespa, P. Changes in Hippocampal Volume after Traumatic Brain Injury (TBI). Research Square (Research Square). Published online. 4 December 2024. [CrossRef]
- Chia, Y.C.; Anjum, C.E.; Yee, H.R.; Kenisi, Y.; Chan, M.K.S.; Wong, M.B.F.; Pan, S.Y. Stem Cell Therapy for Neurodegenerative Diseases: How Do Stem Cells Bypass the Blood-Brain Barrier and Home to the Brain? Stem Cells Int. 2020, 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Zhou, W.; Li, D.; Peng, F.; Wang, C.; Yu, L.; Du, J.; Zheng, Y.; Sang, Y.; Zhang, Y.; et al. Engineering Neural Stem Cells with Micropatches for Improved Therapy of Traumatic Brain Injury. Angew. Chem. Int. Ed. Engl. 2025, 64, e202512804. [Google Scholar] [CrossRef]
- Lu, M.-H.; Ji, W.-L.; Chen, H.; Sun, Y.-Y.; Zhao, X.-Y.; Wang, F.; Shi, Y.; Hu, Y.-N.; Liu, B.-X.; Wu, J.-W.; et al. Intranasal Transplantation of Human Neural Stem Cells Ameliorates Alzheimer’s Disease-Like Pathology in a Mouse Model. Front. Aging Neurosci. 2021, 13. [Google Scholar] [CrossRef]
- Roncalli, J.G.; Tongers, J.; Renault, M.-A.; Losordo, D.W. Endothelial progenitor cells in regenerative medicine and cancer: A decade of research. Trends Biotechnol. 2008, 26, 276–283. [Google Scholar] [CrossRef]
- Huang, X.-T.; Zhang, Y.-Q.; Li, S.-J.; Li, S.H.; Tang, Q.; Wang, Z.-T.; Dong, J.-F.; Zhang, J.-N. Intracerebroventricular transplantation of ex vivo expanded endothelial colony-forming cells restores blood–brain Barrier integrity and promotes angiogenesis of mice with traumatic brain injury. J. Neurotrauma 2013, 30, 2080–2088. [Google Scholar] [CrossRef]
- Zhang, S.; Zhi, Y.; Li, F.; Huang, S.; Gao, H.; Han, Z.; Ge, X.; Li, D.; Chen, F.; Kong, X.; et al. Transplantation of in vitro cultured endothelial progenitor cells repairs the blood-brain barrier and improves cognitive function of APP/PS1 transgenic AD mice. J. Neurol. Sci. 2018, 387, 6–15. [Google Scholar] [CrossRef]
- de Vasconcelos, P.; Lacerda, J.F. Hematopoietic Stem Cell Transplantation for Neurological Disorders: A Focus on Inborn Errors of Metabolism. Front. Cell. Neurosci. 2022, 16, 895511. [Google Scholar] [CrossRef]
- Mishra, P.; Silva, A.; Sharma, J.; Nguyen, J.; Pizzo, D.P.; Hinz, D.; Sahoo, D.; Cherqui, S. Rescue of Alzheimer’s disease phenotype in a mouse model by transplantation of wild-type hematopoietic stem and progenitor cells. Cell Rep. 2023, 42, 112956. [Google Scholar] [CrossRef]
- Via, A.G.; Frizziero, A.; Oliva, F. Biological properties of mesenchymal Stem Cells from different sources. Muscles Ligaments Tendons J. 2012, 2, 154–162. [Google Scholar] [PubMed]
- Menge, T.; Zhao, Y.; Zhao, J.; Wataha, K.; Gerber, M.; Zhang, J.; Letourneau, P.; Redell, J.; Shen, L.; Wang, J.; et al. Mesenchymal Stem Cells Regulate Blood-Brain Barrier Integrity Through TIMP3 Release After Traumatic Brain Injury. Sci. Transl. Med. 2012, 4, 161ra150. [Google Scholar] [CrossRef] [PubMed]
- Eyvari-Brooshghalan, S.; Haddadi, R.; Shahidi, S.; Ghaderi, S.; Rashno, M.; Kalantari, A.; Salehi, I.; Komaki, A.; Sarihi, A. Acute Treatment with Fucoidan Ameliorates Traumatic Brain Injury-Induced Neurological Damages and Memory Deficits in Rats: Role of BBB Integrity, Microglial Activity, Neuroinflammation, and Oxidative Stress. Mol. Neurobiol. 2024, 62, 5990–6013. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Chao, H.; Li, Z.; Xu, X.; Liu, Y.; Hou, L.; Liu, N.; Ji, J. Melatonin attenuates traumatic brain injury-induced inflammation: A possible role for mitophagy. J. Pineal Res. 2016, 61, 177–186. [Google Scholar] [CrossRef]
- Ding, K.; Xu, J.; Wang, H.; Zhang, L.; Wu, Y.; Li, T. Melatonin protects the brain from apoptosis by enhancement of autophagy after traumatic brain injury in mice. Neurochem. Int. 2015, 91, 46–54. [Google Scholar] [CrossRef]
- Rehman, S.U.; Ikram, M.; Ullah, N.; Alam, S.I.; Park, H.Y.; Badshah, H.; Choe, K.; Kim, M.O. Neurological Enhancement Effects of Melatonin against Brain Injury-Induced Oxidative Stress, Neuroinflammation, and Neurodegeneration via AMPK/CREB Signaling. Cells 2019, 8, 760. [Google Scholar] [CrossRef]
- Gunara, S.O.; Akgun, M.Y.; Seker, U.; Ucar, E.A.; Ates, O.; Basar, I. Neuroprotection unveiled: Melatonin mitigates apoptotic pathways in traumatic brain injury. Front. Neurol. 2025, 16, 1551449. [Google Scholar] [CrossRef]
- Carteri, R.B. Mitochondria as a Therapeutic Target: Focusing on Traumatic Brain Injury. J. Integr. Neurosci. 2025, 24, 25292. [Google Scholar] [CrossRef]
- Riou, A.; Broeglin, A.; Grimm, A. Mitochondrial transplantation in brain disorders: Achievements, methods, and challenges. Neurosci. Biobehav. Rev. 2024, 169, 105971. [Google Scholar] [CrossRef]
- Alshial, E.E.; Abdulghaney, M.I.; Wadan, A.-H.S.; Abdellatif, M.A.; Ramadan, N.E.; Suleiman, A.M.; Waheed, N.; Abdellatif, M.; Mohammed, H.S. Mitochondrial dysfunction and neurological disorders: A narrative review and treatment overview. Life Sci. 2023, 334, 122257. [Google Scholar] [CrossRef]
- Bamshad, C.; Najafi-Ghalehlou, N.; Pourmohammadi-Bejarpasi, Z.; Tomita, K.; Kuwahara, Y.; Sato, T.; Feizkhah, A.; Roushnadeh, A.M.; Roudkenar, M.H. Mitochondria: How eminent in ageing and neurodegenerative disorders? Hum. Cell 2022, 36, 41–61. [Google Scholar] [CrossRef] [PubMed]
- Alkaslasi, M.R.; Lloyd, E.Y.H.; Gable, A.S.; Silberberg, H.; Yarur, H.E.; Tsai, V.S.; Sohn, M.; Margolin, G.; Tejeda, H.A.; Le Pichon, C.E. The transcriptional response of cortical neurons to concussion reveals divergent fates after injury. Nat. Commun. 2025, 16, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Diamante, G.; Ahn, I.S.; Palafox-Sanchez, V.; Cheng, J.; Cheng, M.; Ying, Z.; Wang, S.S.-M.; Abuhanna, K.D.; Phi, N.; et al. Thyroid hormone T4 mitigates traumatic brain injury in mice by dynamically remodeling cell type specific genes, pathways, and networks in hippocampus and frontal cortex. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2024, 1870, 167344. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J. Thyroid hormone in brain development. Endocr. Abstr. 2015. [Google Scholar] [CrossRef]
- Mele, C.; De Marchi, L.; Pitino, R.; Costantini, L.; Cavigiolo, B.; Caputo, M.; Marzullo, P.; Aimaretti, G. The interplay between thyrotropic axis, neurological complications, and rehabilitation outcomes in patients with traumatic brain injury. Best Pr. Res. Clin. Endocrinol. Metab. 2025, 39, 102001. [Google Scholar] [CrossRef]
- Crupi, R.; Paterniti, I.; Campolo, M.; Di Paola, R.; Cuzzocrea, S.; Esposito, E. Exogenous T3 administration provides neuroprotection in a murine model of traumatic brain injury. Pharmacol. Res. 2013, 70, 80–89. [Google Scholar] [CrossRef]
- Corps, K.N.; Roth, T.L.; McGavern, D.B. Inflammation and Neuroprotection in Traumatic Brain Injury. JAMA Neurol. 2015, 72, 355–362. [Google Scholar] [CrossRef]
- Needham, E.; Helmy, A.; Zanier, E.; Jones, J.; Coles, A.; Menon, D. The immunological response to traumatic brain injury. J. Neuroimmunol. 2019, 332, 112–125. [Google Scholar] [CrossRef]
- Czerpaniak, K.; Nascimento, L.F.D.; Guo, T.; Zhang, J.; Liu, X.; Sarzaeim, M.; Fine, Z.D.; Cao, Y. Low-Dose Interleukin-2 Reverses Traumatic Brain Injury-Induced Cognitive Deficit and Pain in a Murine Model. Ann. Neurol. 2024, 96, 508–525. [Google Scholar] [CrossRef]
- Wang, P.; Okada-Rising, S.; Scultetus, A.H.; Bailey, Z.S. The Relevance and Implications of Monoclonal Antibody Therapies on Traumatic Brain Injury Pathologies. Biomedicines 2024, 12, 2698. [Google Scholar] [CrossRef]
- Kondo, A.; Shahpasand, K.; Mannix, R.; Qiu, J.; Moncaster, J.; Chen, C.-H.; Yao, Y.; Lin, Y.-M.; Driver, J.A.; Sun, Y.; et al. Antibody against early driver of neurodegeneration cis P-tau blocks brain injury and tauopathy. Nature 2015, 523, 431–436. [Google Scholar] [CrossRef]
- Vella, M.A.; Crandall, M.L.; Patel, M.B. Acute Management of Traumatic Brain Injury. Surg. Clin. North Am. 2017, 97, 1015–1030. [Google Scholar] [CrossRef] [PubMed]
- Collaborators, C.T. Effects of tranexamic acid on death, disability, vascular occlusive events and other morbidities in patients with acute traumatic brain injury (CRASH-3): A randomised, placebo-controlled trial. Lancet 2019, 394, 1713–1723. [Google Scholar] [CrossRef]
- Alnemari, A.M.; Krafcik, B.M.; Mansour, T.R.; Gaudin, D. A Comparison of Pharmacologic Therapeutic Agents Used for the Reduction of Intracranial Pressure After Traumatic Brain Injury. World Neurosurg. 2017, 106, 509–528. [Google Scholar] [CrossRef] [PubMed]
- Roberts, I. Barbiturates for acute traumatic brain injury. Cochrane Database Systematic Rev. 2000, CD000033. [Google Scholar]
- Thompson, K.; Pohlmann-Eden, B.; A Campbell, L.; Abel, H. Pharmacological treatments for preventing epilepsy following traumatic head injury. Cochrane Database Systematic Rev. 2015, 2015, CD009900. [Google Scholar] [CrossRef] [PubMed]
- Tani, J.; Wen, Y.-T.; Hu, C.-J.; Sung, J.-Y. Current and Potential Pharmacologic Therapies for Traumatic Brain Injury. Pharmaceuticals 2022, 15, 838. [Google Scholar] [CrossRef]
- Edwards, P.; Arango, M.; Balica, L.; Cottingham, R.; El-Sayed, H.; Farrell, B.; Fernandes, J.; Gogichaisvili, T.; Golden, N.; Hartzenberg, B.; et al. Final results of MRC CRASH, a randomised placebo-controlled trial of intravenous corticosteroid in adults with head injury—outcomes at 6 months. Lancet 2005, 365, 1957–1959. [Google Scholar] [CrossRef]
- Zafonte, R.; Friedewald, W.T.; Lee, S.M.; Levin, B.; Diaz-Arrastia, R.; Ansel, B.; Eisenberg, H.; Timmons, S.D.; Temkin, N.; Novack, T.; et al. The Citicoline Brain Injury Treatment (COBRIT) Trial: Design and Methods. J. Neurotrauma 2009, 26, 2207–2216. [Google Scholar] [CrossRef]
- Espinoza, T.R.; Wright, D.W. The Role of Progesterone in Traumatic Brain Injury. J. Head Trauma Rehabilitation 2011, 26, 497–499. [Google Scholar] [CrossRef]
- Yue, J.K.; Burke, J.F.; Upadhyayula, P.S.; Winkler, E.A.; Deng, H.; Robinson, C.K.; Pirracchio, R.; Suen, C.G.; Sharma, S.; Ferguson, A.R.; et al. Selective Serotonin Reuptake Inhibitors for Treating Neurocognitive and Neuropsychiatric Disorders Following Traumatic Brain Injury: An Evaluation of Current Evidence. Brain Sci. 2017, 7, 93. [Google Scholar] [CrossRef]
- Duarte, D.C.; Duarte, J.C.; González, Á.A.O.; García, J.F.C. Psychiatric disorders in post-traumatic brain injury patients: A scoping review. Heliyon 2023, 9, e12905. [Google Scholar] [CrossRef]
- Cognacq, G.; E Attwood, J.; DeLuca, G.C. Traumatic Brain Injury and Alzheimer’s Disease: A Shared Neurovascular Hypothesis. Neurosci. Insights 2025, 20. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




