Submitted:
12 January 2026
Posted:
14 January 2026
You are already at the latest version
Abstract
Nickel-supported over SiO₂-CeO₂ mixed oxides were investigated as catalysts for syngas production via the dry reforming of methane. The SiO₂-CeO₂ supports were optimized, playing on the preparation method and ceria loading with the aim of stabilizing nickel nanoparticles, enhancing the catalytic performance, and improving the resistance to coke formation under high-temperature reforming conditions. To investigate the effect of support composition, SiO₂-CeO₂ mixed oxides with ceria contents ranging from 5 to 30 wt% were prepared using two synthesis routes: sol-gel and wetness impregnation methods. A nickel loading of 5 wt% was deposited on the resulting supports. The catalysts were characterized by XRD, N₂ physisorption, temperature-programmed reduction, and Raman spectroscopy. Catalytic activity tests were conducted over reduced catalysts in an H₂-He stream at 750 °C, using a feed mixture containing 15 vol% CH₄ and 15 vol% CO₂ in He. The effect of temperature on catalytic performance was evaluated in the range of 450–800 °C. Thermogravimetric, XRD and Raman analyses of spent catalysts were used to assess carbon deposition and the nature of crystalline phases. The results highlight the role of CeO₂ content and preparation method in determining nickel dispersion, reducibility, catalytic performance in DRM, and coke resistance.
Keywords:
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Synthesis Procedures of Si-SG, SiCe(5-30)-SG, SiCe(5-30)-WI and the Corresponding Ni-Based Powders
3.2. Characterization of the Materials
3.3. DRM Catalytic Tests
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gholami, Z.; Gholami, F.; Šimek, J.; Svobodová, K.; Vakili, M. Hydrogen production for a decarbonized future: a review of production technologies. Journal of Industrial and Engineering Chemistry 2026, 153, 240–278. [Google Scholar] [CrossRef]
- Wittich, K.; Krämer, M.; Bottke, N.; Schunk, S.A. Catalytic Dry Reforming of Methane: Insights from Model Systems. ChemCatChem 2020, 12, 2130–2147. [Google Scholar] [CrossRef]
- Agún, B.; Abánades, A. Comprehensive review on dry reforming of methane: Challenges and potential for greenhouse gas mitigation. International Journal of Hydrogen Energy 2025, 103, 395–414. [Google Scholar] [CrossRef]
- Nguyen, D.L.T.; Vy Tran, A.; Vo, D.-V.N.; Tran Nguyen, H.; Rajamohan, N.; Trinh, T.H.; Nguyen, T.L.; Le, Q.V.; Nguyen, T.M. Methane dry reforming: A catalyst challenge awaits. Journal of Industrial and Engineering Chemistry 2024, 140, 169–189. [Google Scholar] [CrossRef]
- Osazuwa, O.U.; Ng, K.H. An overview on the carbon deposited during dry reforming of methane (DRM): Its formation, deposition, identification, and quantification. Results in Engineering 2025, 25, 104328. [Google Scholar] [CrossRef]
- Wang, Z.; Mei, Z.; Wang, L.; Wu, Q.; Xia, C.; Li, S.; Wang, T.; Liu, C. Insight into the activity of Ni-based thermal catalysts for dry reforming of methane. Journal of Materials Chemistry A 2024, 12, 24802–24838. [Google Scholar] [CrossRef]
- Xu, Z.; Park, E.D. Recent Advances in Coke Management for Dry Reforming of Methane over Ni-Based Catalysts. Catalysts 2024, 14, 176. [Google Scholar] [CrossRef]
- Zhu, H.; Chen, H.; Zhang, M.; Liang, C.; Duan, L. Recent advances in promoting dry reforming of methane using nickel-based catalysts. Catalysis Science & Technology 2024, 14, 1712–1729. [Google Scholar] [CrossRef]
- Shi, Y.; Tian, X.; Deng, Z.; Shi, W.; Fan, W.; Wang, F. Review and Outlook of Confined Ni Catalysts for the Dry Reforming of Methane Reaction. Energy & Fuels 2024, 38, 1633–1656. [Google Scholar] [CrossRef]
- Chen, C.; Wei, J.; Lu, Y.; Duyar, M.S.; Huang, Y.; Lin, L.; Ye, R. Confinement effects over Ni-based catalysts for methane dry reforming. Catalysis Science & Technology 2023, 13, 6089–6101. [Google Scholar] [CrossRef]
- Montini, T.; Melchionna, M.; Monai, M.; Fornasiero, P. Fundamentals and Catalytic Applications of CeO2-Based Materials. Chemical Reviews 2016, 116, 5987–6041. [Google Scholar] [CrossRef]
- Teh, L.P.; Setiabudi, H.D.; Timmiati, S.N.; Aziz, M.A.A.; Annuar, N.H.R.; Ruslan, N.N. Recent progress in ceria-based catalysts for the dry reforming of methane: A review. Chemical Engineering Science 2021, 242, 116606. [Google Scholar] [CrossRef]
- Niu, Y.; Zheng, X.; Guo, B.; Liu, H.; Jin, Y.; Niu, J. Particle size tuning of Ni/CeO2 catalysts and their performance in methane dry reforming reaction. Gas Science and Engineering 2026, 145, 205790. [Google Scholar] [CrossRef]
- Janlon, J.; Chatsuwan, N.; Kongparakul, S.; Ratthiwal, J.; Fong, Y.Y.; Reubroycharoen, P.; Chanlek, N.; Tran, T.T.V.; Samart, C. Highly stable Ni-based catalysts supported on silica nanofibers for the dry reforming of methane with CO2-rich feedstock. Journal of the Energy Institute 2025, 123, 102313. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.; Zhang, G.; Zhang, X.; Wang, Y.; Zhao, Y.; Li, G. Ni/MSS@CeO2 sandwich catalysts for methane dry reforming: the role of reduction on oxygen vacancies. Journal of Colloid and Interface Science 2025, 694, 137712. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Z.; Wang, N.; Zha, Y.; Zheng, K.; Xu, Y.; Liu, B.; Liu, X. Strong activity-based volcano-type relationship for dry reforming of methane through modulating Ni-CeO2 interaction over Ni/CeO2-SiO2 catalysts. Chem Catalysis 2025, 5, 101189. [Google Scholar] [CrossRef]
- Cherevotan, A.; Ray, B.; Churipard, S.R.; Kaur, K.; Gautam, U.K.; Vinod, C.P.; Peter, S.C. Influence of support textural property on CO2 to methane activity of Ni/SiO2 catalysts. Applied Catalysis B: Environmental 2022, 317, 121692. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, G.; Liu, J.; Li, T.; Wang, Y.; Zhao, Y.; Li, G.; Zhang, Y. Dry reforming of methane over Ni/SiO2 catalysts: Role of support structure properties. Fuel 2023, 340, 127490. [Google Scholar] [CrossRef]
- Gao, S.; Li, Y.; Guo, W.; Ding, X.; Zheng, L.; Wu, L.; Yan, H.; Wang, Y. Morphology effect of ceria support with hierarchical structure on the catalytic performance for nickel-based catalysts in dry reforming of methane. Molecular Catalysis 2022, 533, 112766. [Google Scholar] [CrossRef]
- Han, K.; Xu, S.; Wang, Y.; Wang, S.; Zhao, L.; Kambonde, J.; Yu, H.; Shi, W.; Wang, F. Confining Ni and ceria in silica shell as synergistic multifunctional catalyst for methane dry reforming reaction. Journal of Power Sources 2021, 506, 230232. [Google Scholar] [CrossRef]
- Liu, Z.; Grinter, D.C.; Lustemberg, P.G.; Nguyen-Phan, T.-D.; Zhou, Y.; Luo, S.; Waluyo, I.; Crumlin, E.J.; Stacchiola, D.J.; Zhou, J.; et al. Dry Reforming of Methane on a Highly-Active Ni-CeO2 Catalyst: Effects of Metal-Support Interactions on C−H Bond Breaking. Angewandte Chemie International Edition 2016, 55, 7455–7459. [Google Scholar] [CrossRef]
- Li, B.; Yuan, X.; Li, B.; Wang, X. Ceria-modified nickel supported on porous silica as highly active and stable catalyst for dry reforming of methane. Fuel 2021, 301, 121027. [Google Scholar] [CrossRef]
- Zhou, Q.; Fu, X.; Hui Lim, K.; Li, Z.; Liao, M.; Lu, J.; Liu, F.; Kawi, S. Complete confinement of Ce/Ni within SiO2 nanotube with high oxygen vacancy concentration for CO2 methane reforming. Fuel 2022, 325, 124819. [Google Scholar] [CrossRef]
- Grabchenko, M.V.; Dorofeeva, N.V.; Svetlichnyi, V.A.; Larichev, Y.V.; La Parola, V.; Liotta, L.F.; Kulinich, S.A.; Vodyankina, O.V. Ni-Based SBA-15 Catalysts Modified with CeMnOx for CO2 Valorization via Dry Reforming of Methane: Effect of Composition on Modulating Activity and H2/CO Ratio. Nanomaterials 2023, 13, 2641. [Google Scholar] [CrossRef] [PubMed]
- La Parola, V.; Liotta, L.F.; Pantaleo, G.; Testa, M.L.; Venezia, A.M. CO2 reforming of CH4 over Ni supported on SiO2 modified by TiO2 and ZrO2: Effect of the support synthesis procedure. Applied Catalysis A: General 2022, 642, 118704. [Google Scholar] [CrossRef]
- Ali, S.; Khader, M.M.; Almarri, M.J.; Abdelmoneim, A.G. Ni-based nano-catalysts for the dry reforming of methane. Catalysis Today 2020, 343, 26–37. [Google Scholar] [CrossRef]
- Taufiq-Yap, Y.H.; Sudarno; Rashid, U.; Zainal, Z. CeO2–SiO2 supported nickel catalysts for dry reforming of methane toward syngas production. Applied Catalysis A: General 2013, 468, 359–369. [Google Scholar] [CrossRef]
- Sudarno; Razali, S.M.; Asikin-Mijan, N.; Sivasangar, S.; Taufiq-Yap, Y.H. Enhanced dry reforming toward hydrogen production over Ni/CeO2SiO2 via different catalyst synthesis routes. International Journal of Hydrogen Energy 2019, 44, 20738–20750. [Google Scholar] [CrossRef]
- Peszke, J.; Dulski, M.; Nowak, A.; Balin, K.; Zubko, M.; Sułowicz, S.; Nowak, B.; Piotrowska-Seget, Z.; Talik, E.; Wojtyniak, M.; et al. Unique properties of silver and copper silica-based nanocomposites as antimicrobial agents. RSC Advances 2017, 7, 28092–28104. [Google Scholar] [CrossRef]
- Kosacki, I.; Suzuki, T.; Anderson, H.U.; Colomban, P. Raman scattering and lattice defects in nanocrystalline CeO2 thin films. Solid State Ionics 2002, 149, 99–105. [Google Scholar] [CrossRef]
- Grabchenko, M.V.; Mamontov, G.V.; Zaikovskii, V.I.; La Parola, V.; Liotta, L.F.; Vodyankina, O.V. Design of Ag-CeO2/SiO2 catalyst for oxidative dehydrogenation of ethanol: Control of Ag–CeO2 interfacial interaction. Catalysis Today 2019, 333, 2–9. [Google Scholar] [CrossRef]
- Dong, F.; Meng, Y.; Han, W.; Zhao, H.; Tang, Z. Morphology effects on surface chemical properties and lattice defects of Cu/CeO2 catalysts applied for low-temperature CO oxidation. Scientific Reports 2019, 9, 12056. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.C.; Hartlaub, S.; Petrovic, I.; Yilmaz, B. Raman Spectroscopy Characterization of Amorphous Coke Generated in Industrial Processes. ACS Omega 2022, 7, 2565–2570. [Google Scholar] [CrossRef]
- Szybowicz, M.; Nowicka, A.B.; Dychalska, A. Chapter 1 - Characterization of Carbon Nanomaterials by Raman Spectroscopy. In Characterization of Nanomaterials; Mohan Bhagyaraj, S., Oluwafemi, O.S., Kalarikkal, N., Thomas, S., Eds.; Woodhead Publishing, 2018; pp. 1–36. [Google Scholar]
- Gil, S.; Garcia-Vargas, J.M.; Liotta, L.F.; Pantaleo, G.; Ousmane, M.; Retailleau, L.; Giroir-Fendler, A. Catalytic Oxidation of Propene over Pd Catalysts Supported on CeO2, TiO2, Al2O3 and M/Al2O3 Oxides (M = Ce, Ti, Fe, Mn). Catalysts 2015, 5, 671–689. [Google Scholar] [CrossRef]
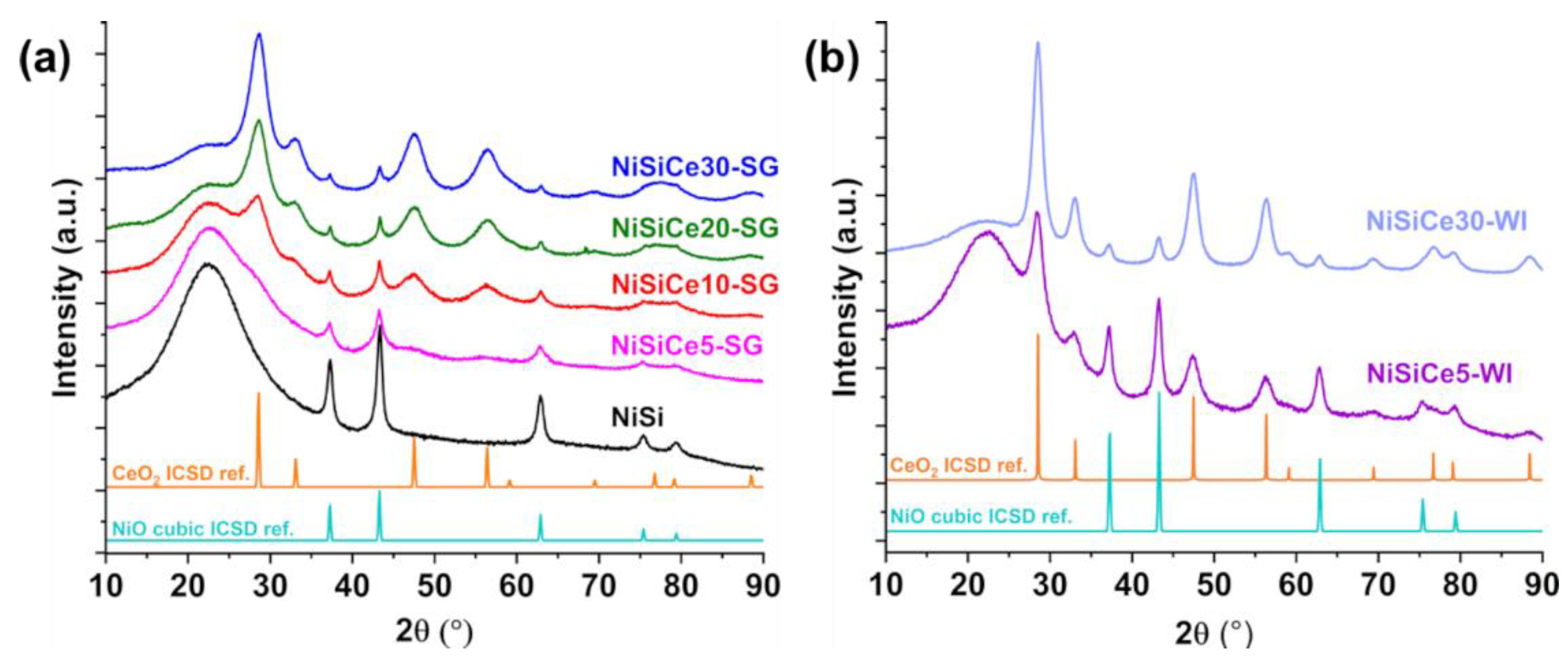
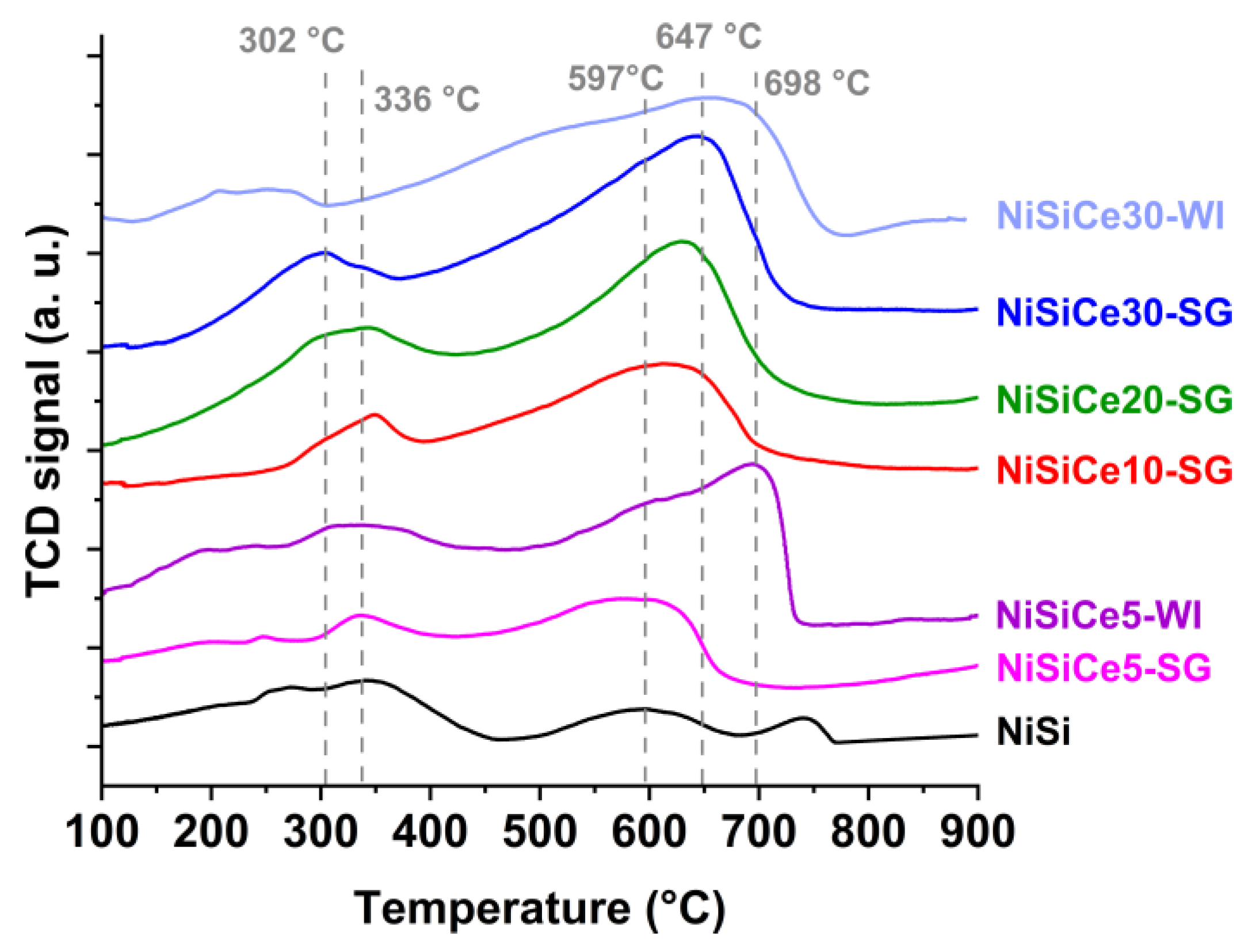
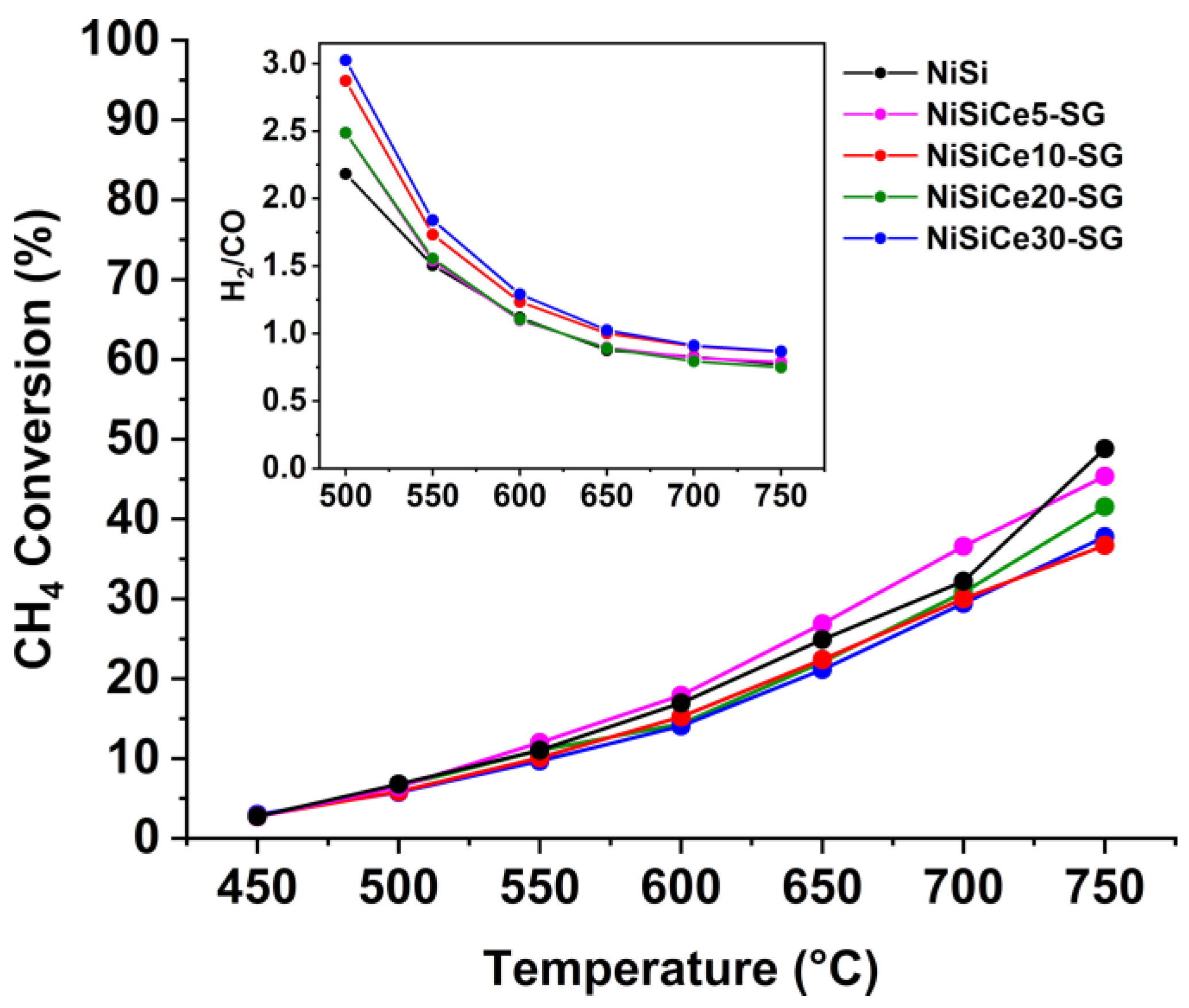
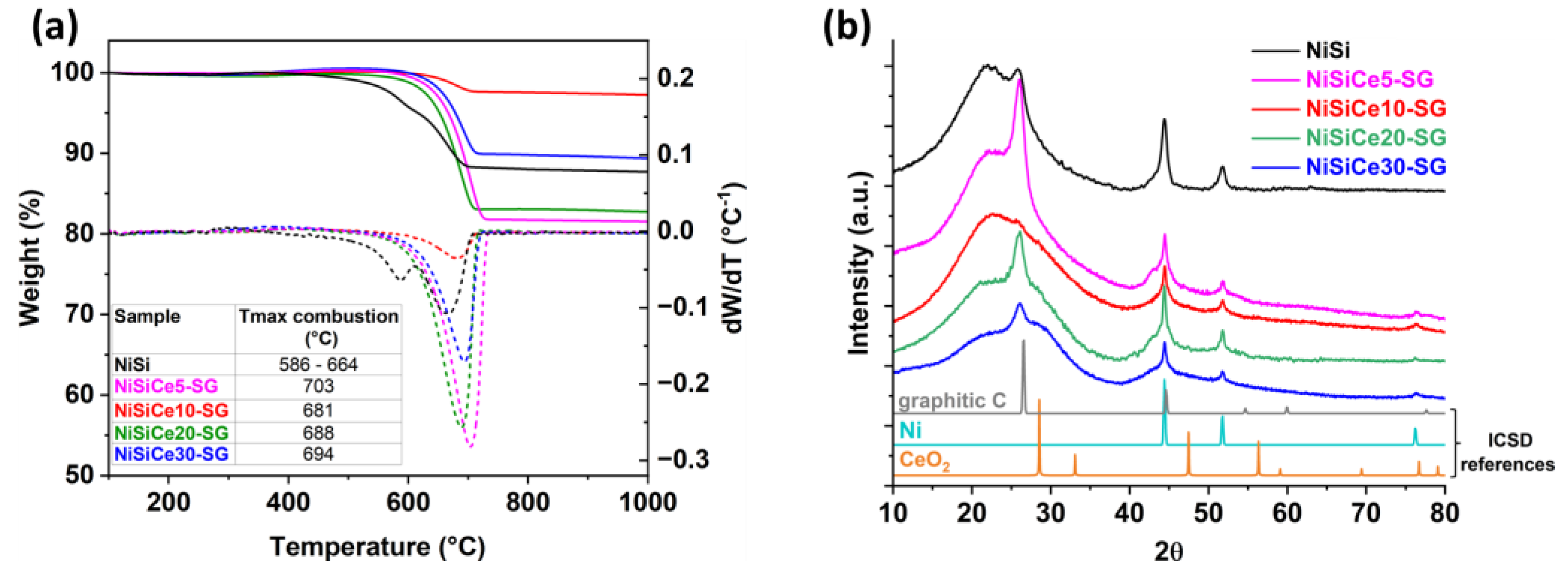
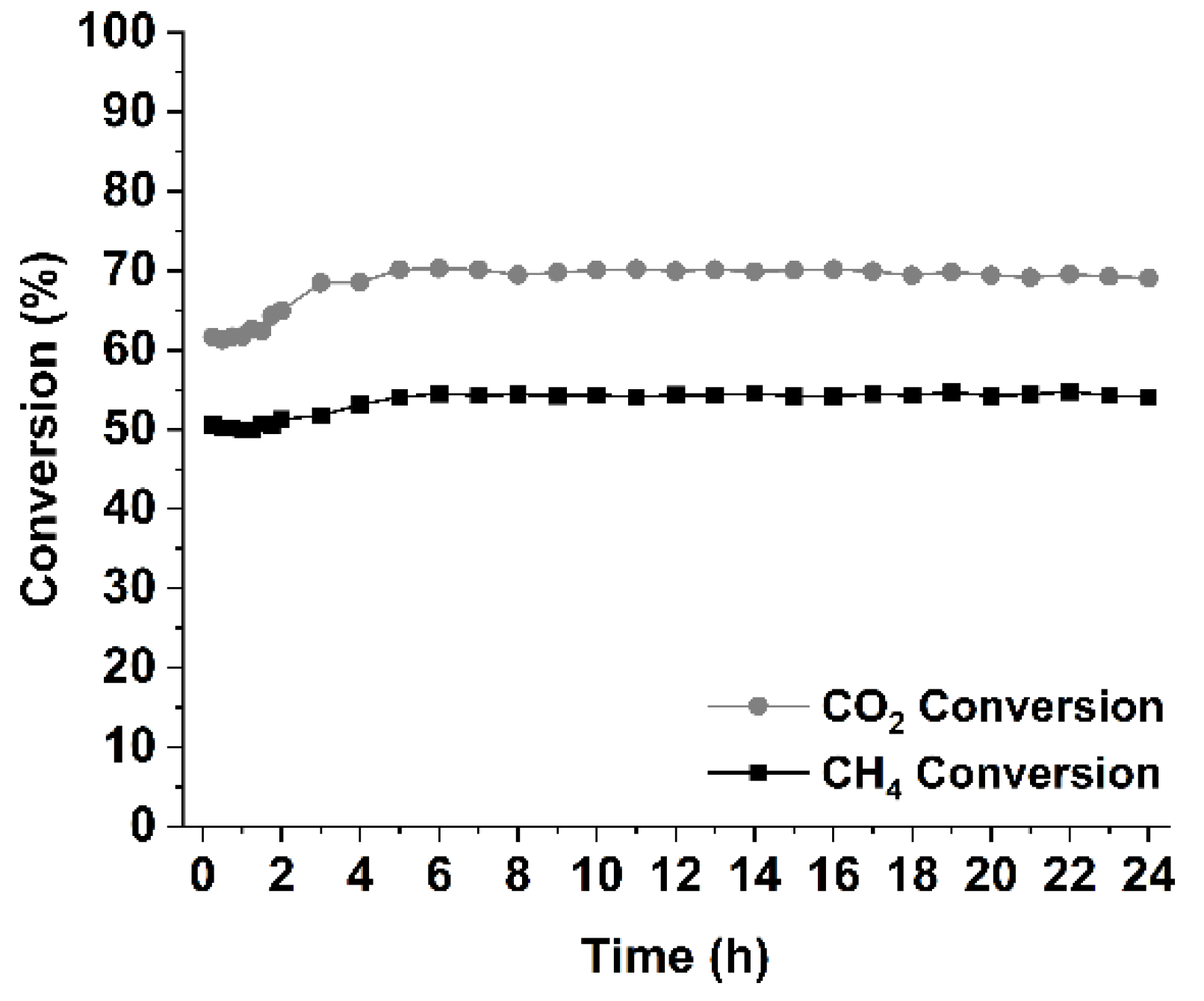
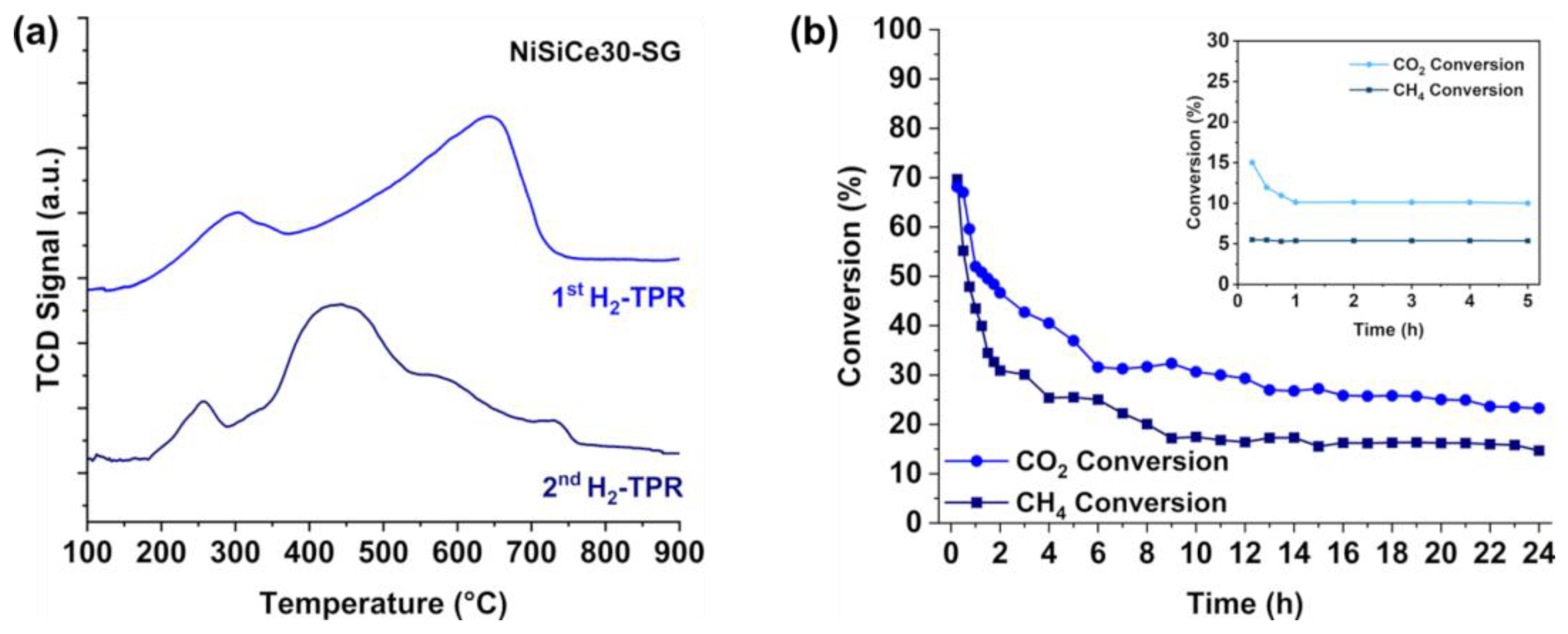
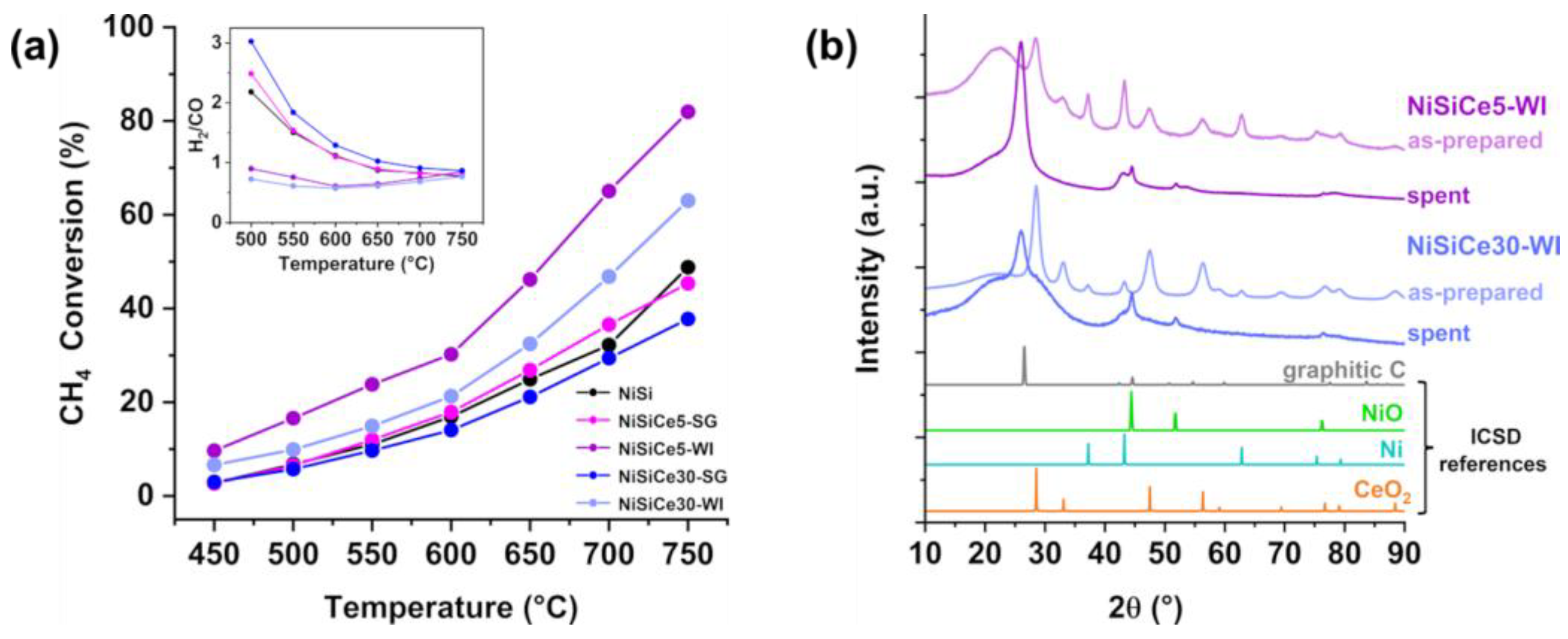
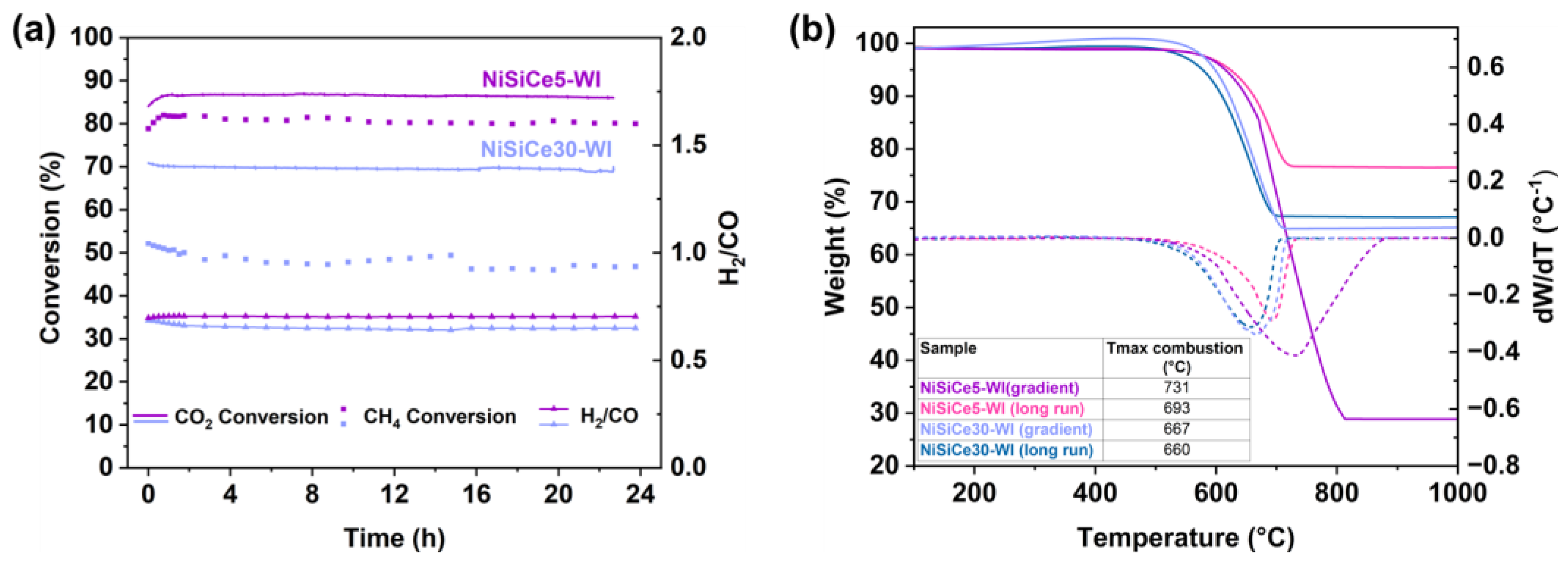
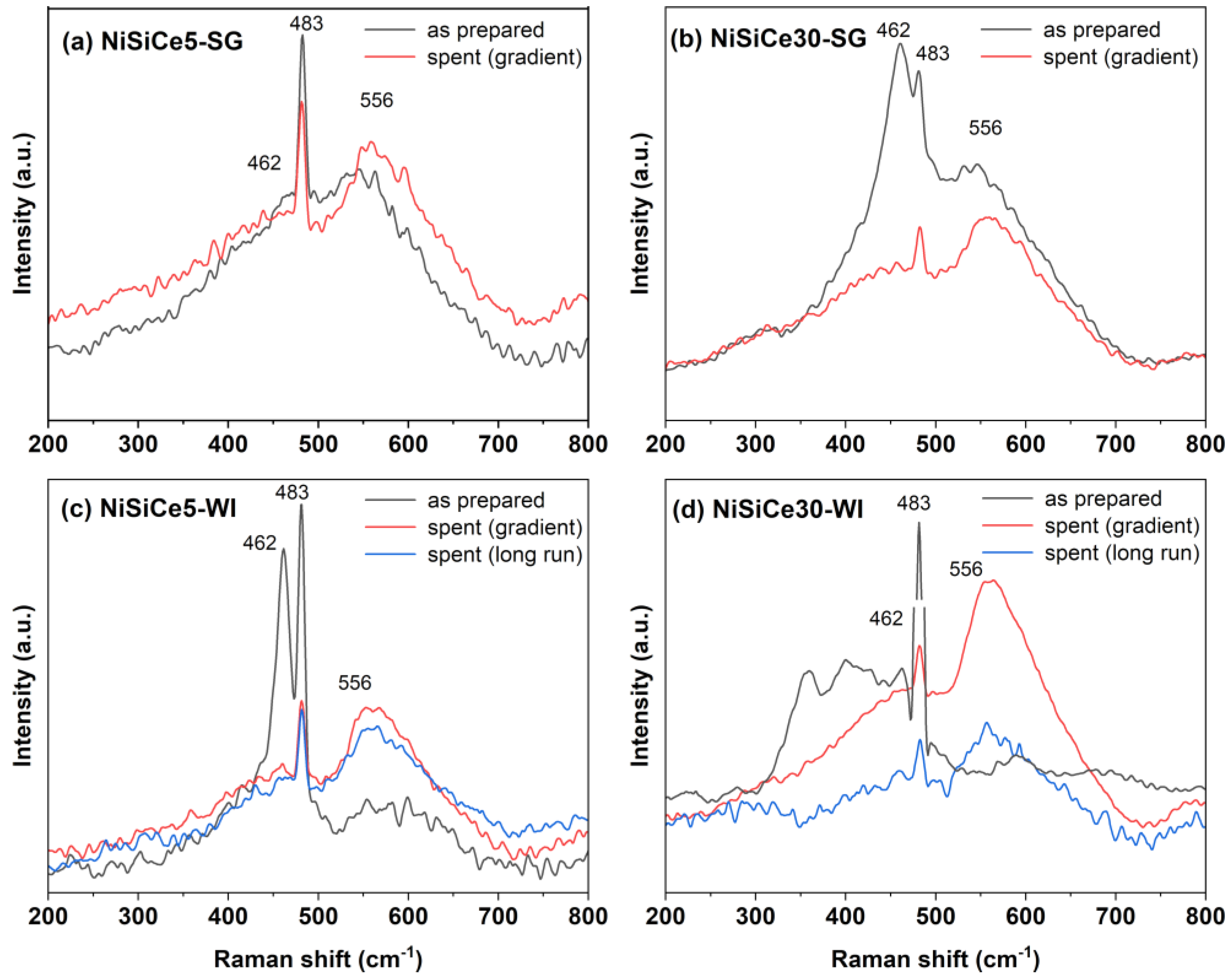
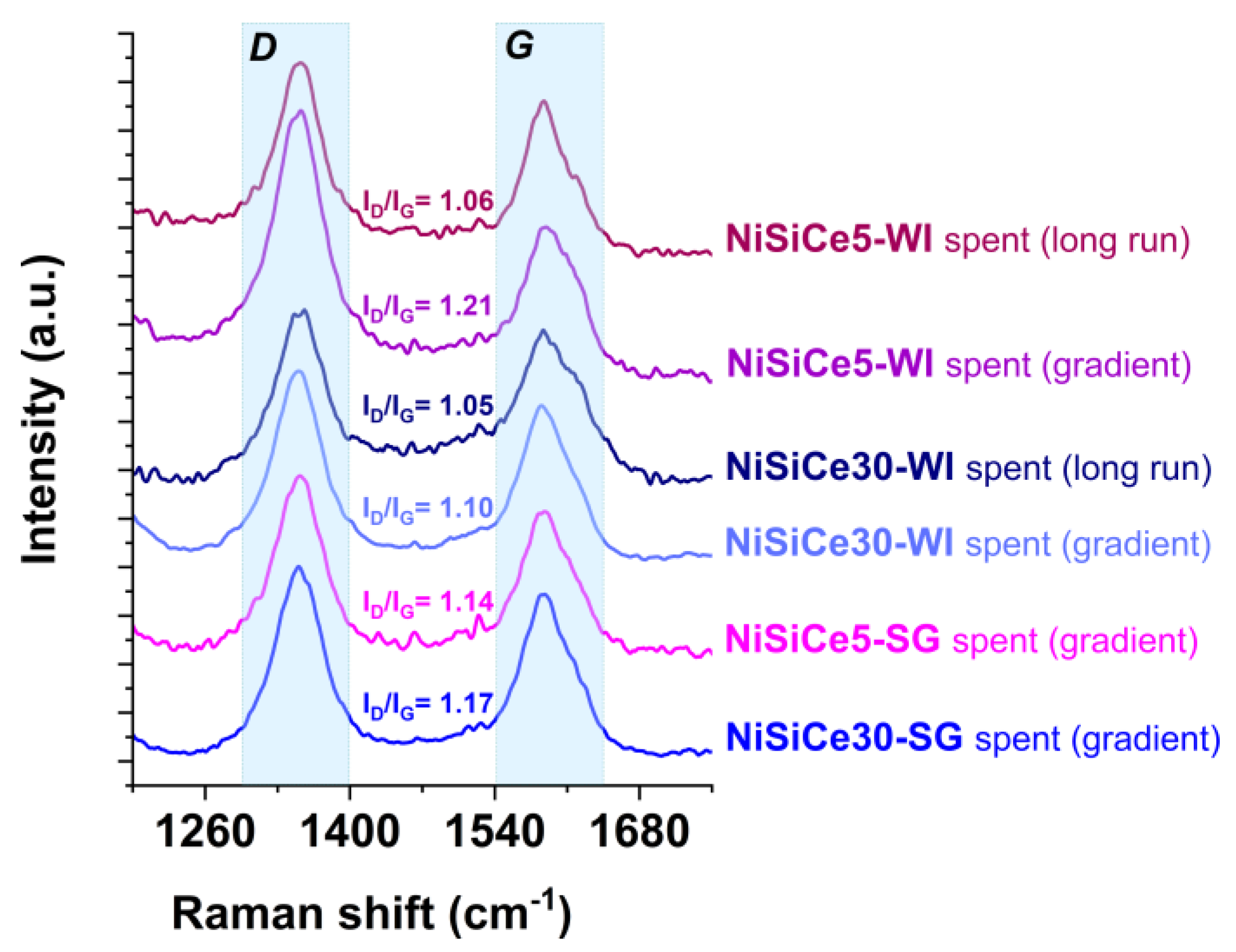
| Sample | SSA (m2/g) | Pore Volume (cm3g-1) | BJH mean pore size (nm) | NiO crystal size 1 (nm) |
|---|---|---|---|---|
| NiSi | 242 | 0.50 | 16.4 | 22.7 |
| NiSiCe5-SG | 310 | 0.16 | 2.73 | 17.0 |
| NiSiCe10-SG | 230 | 0.12 | 2.85 | 16.6 |
| NiSiCe20-SG | 149 | 0.10 | 3.12 | 15.7 |
| NiSiCe30-SG | 163 | 0.12 | 4.18 | 11.8 |
| NiSiCe5-WI | 275 | 0.48 | 17.7 | 19.0 |
| NiSiCe30-WI | 228 | 0.52 | 14.9 | 11.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




