Submitted:
07 January 2026
Posted:
08 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Immune Development in the Placenta
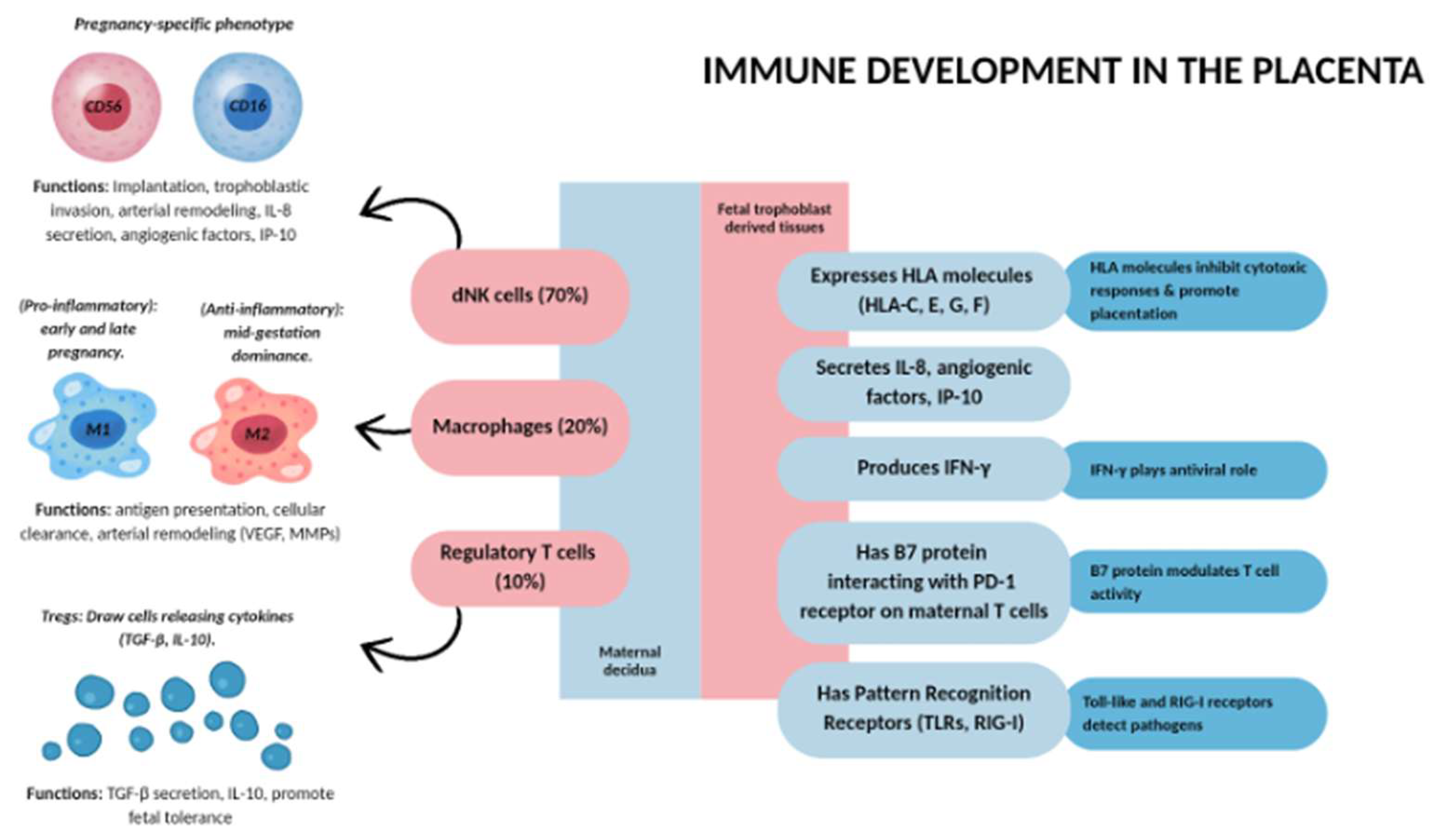
2.1. Immune Cells at the Maternal–Fetal Interface
2.1.1. Natural Killer Cells
2.1.2. Decidual Macrophages
2.1.3. Regulatory T Cells
2.2. Mechanisms of Immune Adaptation and Tolerance
- Human Leukocyte Antigen (HLA): Extravillous trophoblasts express non-classical HLA molecules (HLA-C, HLA-E, HLA-G, HLA-F) that inhibit cytotoxic immune responses and facilitate placentation. HLA-G promotes NK-cell secretion of fetal growth factors [55].
- B7 Protein Family: High expression of costimulatory molecules such as B7-H1 on trophoblasts interacts with PD-1 receptors on maternal T cells, suppressing Th17 responses and enhancing Treg activity [56].
- TNF Superfamily Members: Placental Fas ligand (FasL) and TNF-related apoptosis-inducing ligand (TRAIL) induce apoptosis in activated maternal T cells [55], preventing immune-mediated rejection.
- Pattern Recognition Receptors: Toll-like receptors (TLRs) and RIG-I like receptors on trophoblasts detect pathogen-associated molecular patterns, initiating controlled inflammatory cascades that preserve defense without disrupting tolerance [57].
- Maternal–Fetal IgG Transfer: Immunoglobulin G is actively transported across the placenta during the second and third trimesters, conferring passive immunity to the neonate and infant [58].
- Interferon-γ (IFN-γ): Produced by syncytiotrophoblast, IFN-γ plays a crucial role in antiviral protection and local immune signaling [59].
- Other Immunomodulators: Galectin-1, sex hormones such as progesterone and estrogen, and complement regulatory proteins (CD46, CD55, CD59) collectively maintain immune equilibrium and protect the fetus from complement-mediated injury [19].
3. The Maternal Microbiome: A Hidden Regulator of Pregnancy
3.1. Placental Microbiome
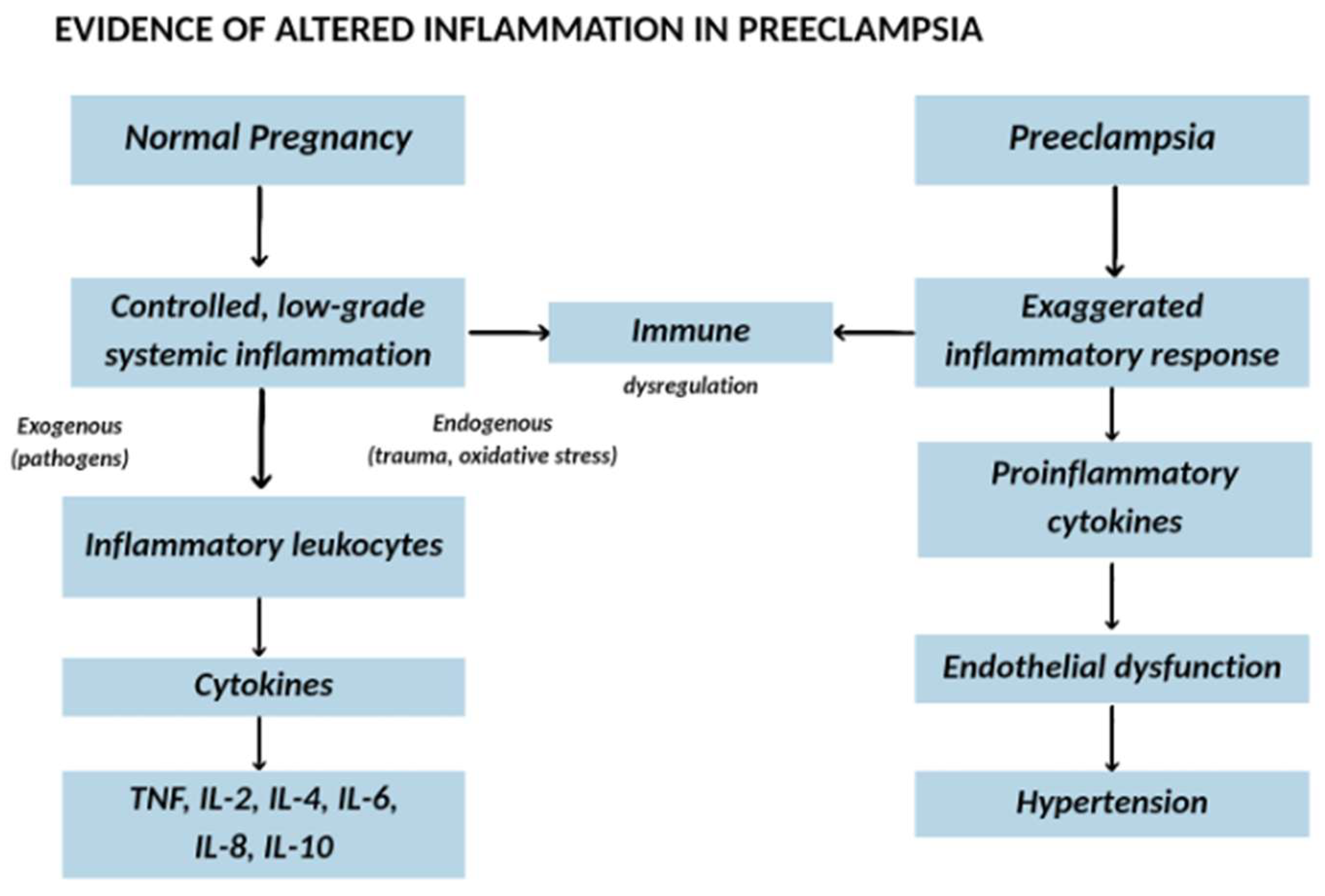
4. Evidence of Altered Inflammation in Preeclampsia
4.1. Immune Dysregulation and Cytokine Imbalance
4.2. Cytokines and Endothelial Dysfunction in Preeclampsia
- TNF-α and IL-6: Promote endothelial adhesion molecule expression, increase vascular permeability, and impair endothelial nitric oxide synthase (eNOS) activity, reducing nitric oxide (NO) bioavailability and vasodilation [91,92]. Experimental models show that a two-fold increase in TNF-α elevates mean arterial pressure (MAP) and reduces renal plasma flow and glomerular filtration rate (GFR) [92,93]. IL-6 exerts similar effects and has been linked to increased plasma renin activity [92,94].
- IL-17: In animal models, IL-17 increases MAP, induces placental oxidative stress, and stimulates B cells to produce angiotensin II type 1 receptor autoantibodies (AT1-AA), exacerbating hypertension and vascular injury [95].
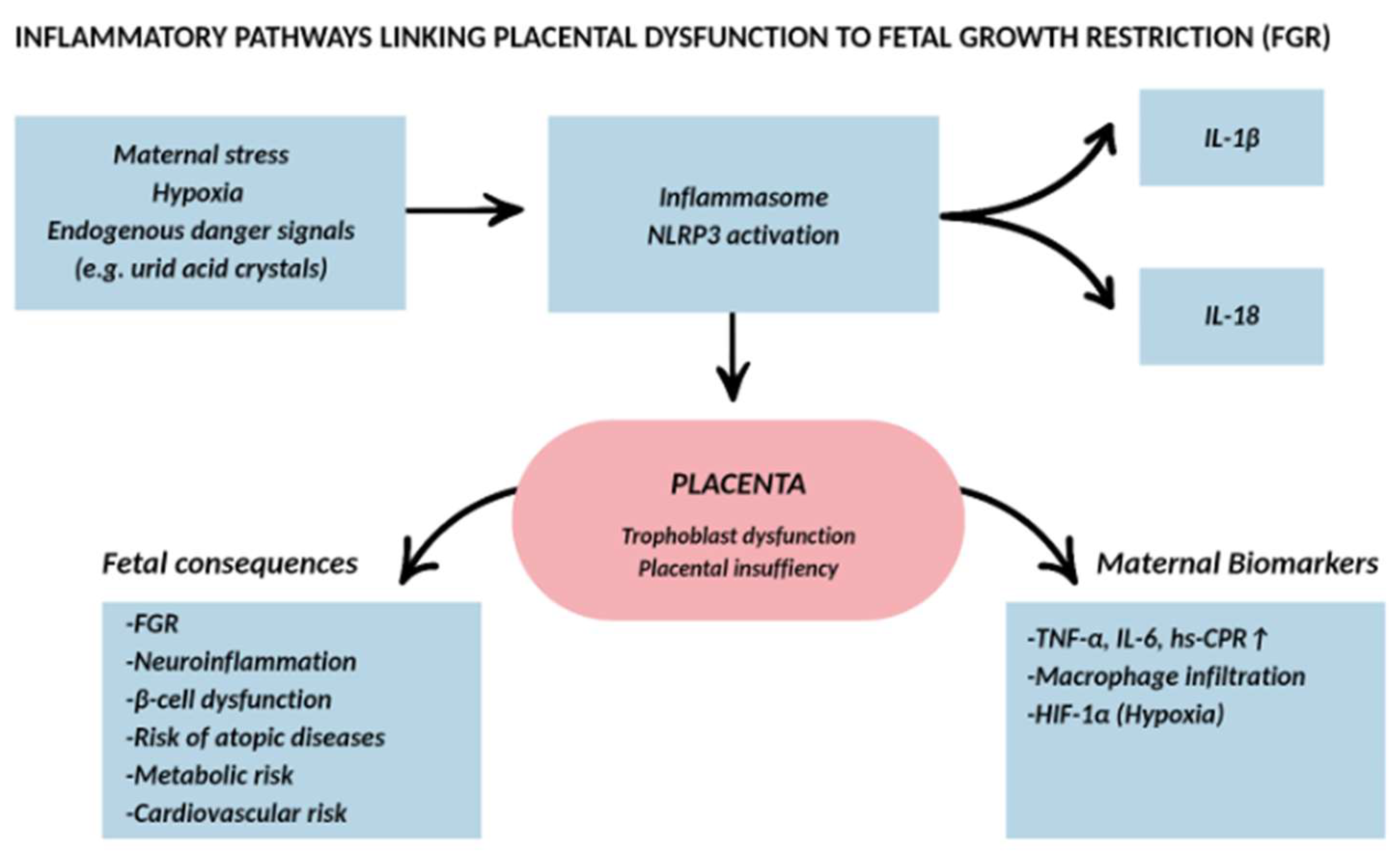
5. Inflammation and Fetal Growth Restriction
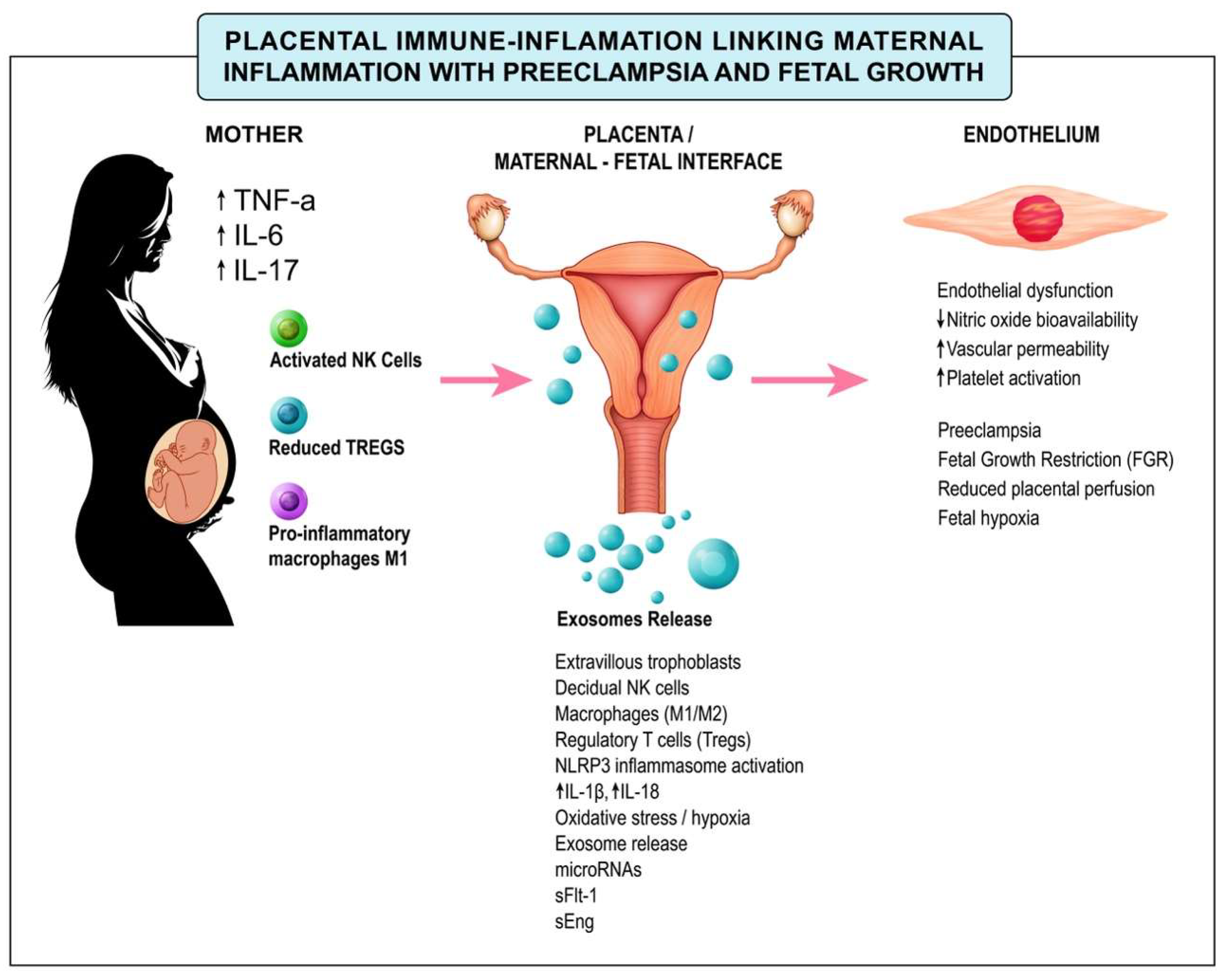
6. Immune Dysregulation in Placental Insufficiency: Clinical Implications and Translational Perspectives
7. Therapy Development
7.1. Established Therapies
7.1.1. Low-Dose Aspirin
7.1.2. Anticoagulants
7.1.3. Corticosteroids
7.2. Adjunctive and Supportive Interventions
7.2.1. Antioxidants and Nutritional Modulation
7.2.3. Lifestyle and Microbiome Modulation
7.3. Emerging and Experimental Therapies
7.3.1. Cytokine-Targeted Biologics
7.3.2. Inflammasome Inhibitors
7.3.3. Cell-Based Therapies
7.3.4. Safety Considerations
7.4. Future Therapies
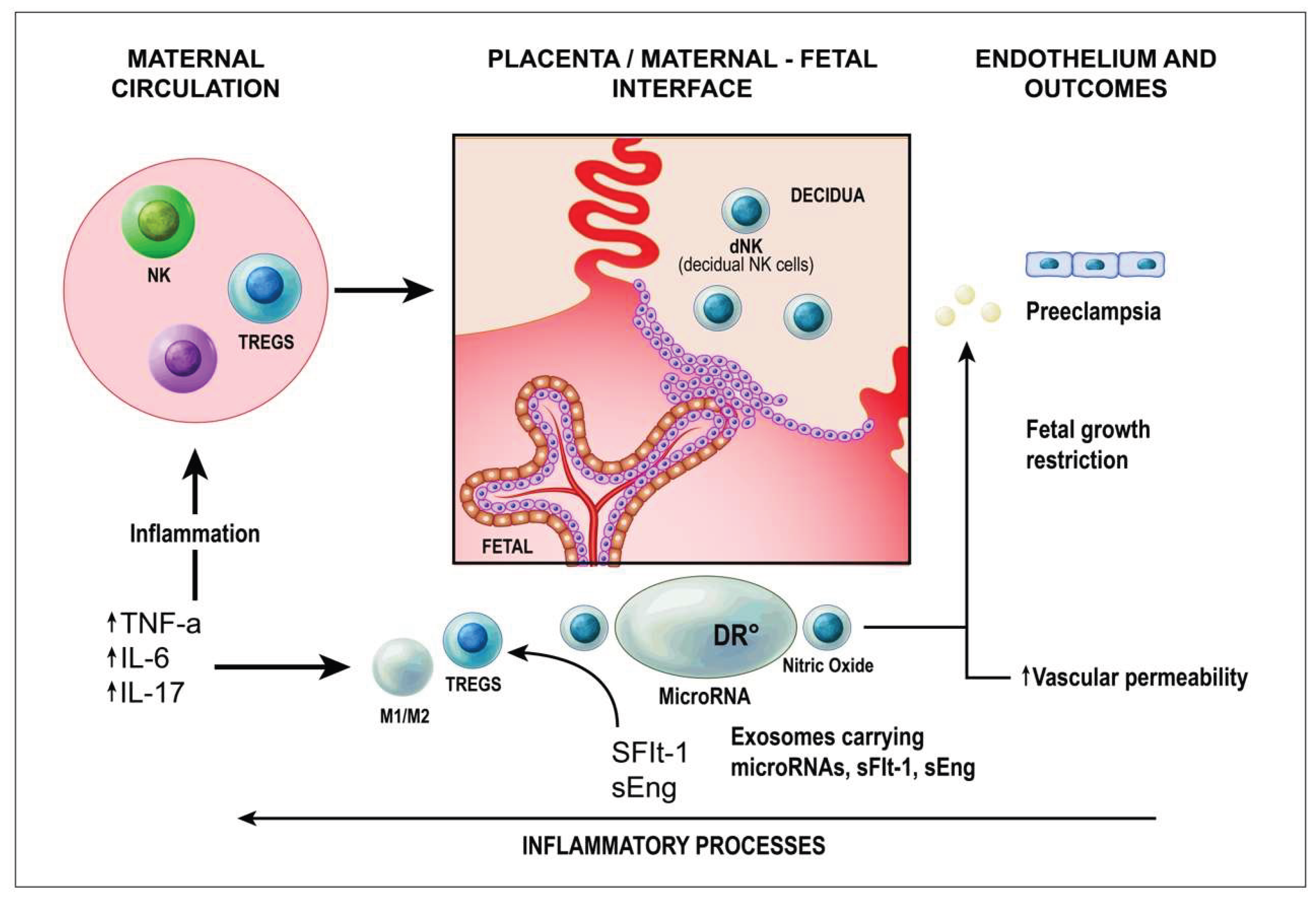
8. Exosomal Signaling at the Maternal–Fetal Interface: Orchestrating Immune and Vascular Adaptation
9. Future Perspectives
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Abbreviations
| FGR | Fetal growth restriction |
| Th1 | T-helper 1 cells |
| Th2 | T-helper 2 cells |
| HDP | Hypertensive disorders of pregnancy |
| uNK | Uterine natural killer cells |
| HLA-G | Human Leukocyte Antigen-G |
| PD-L1 | Programmed Death-Ligand 1 |
| sFlt-1 | soluble fms-like tyrosine kinase-1 |
| NLRP3 | NOD-like receptor family, pyrin domain containing 3 |
| IL | Interleukin |
| TNF-α | Tumor necrosis factor-α |
| dNK | Decidual natural killer cells |
| CD56 | Cluster of Differentiation 56 |
| IP-10 | Interferon-inducible protein-10 |
| VEGF | Vascular endothelial growth factor |
| Tregs | Regulatory T cells |
| TGF-β | Transforming Growth Factor-Beta |
| FasL | Placental Fas ligand |
| TLRs | Toll-like receptors |
| PTB | Preterm birth |
| LBW | Low birth weight |
| MCP-1 | Monocyte chemotactic protein-1 |
| PlGF | Placental growth factor |
| sEng | Soluble endoglin |
| MHC | Histocompatibility complex |
| eNOS | Endothelial nitric oxide synthase |
| NO | Nitric oxide |
| MAP | Mean arterial pressure |
| HIF-1α | Hypoxia-inducible factor 1-alpha |
| NF-κB | Nuclear factor kappa B |
| LDA | Low-dose aspirin |
| COX-1 | Cyclooxygenase-1 |
| LMWH | Low molecular weight heparin |
| MSC | Mesenchymal stem cell |
| miRNAs | microRNAs |
References
- Dekel, N.; Gnainsky, Y.; Granot, I.; Racicot, K.; Mor, G. The role of inflammation for a successful implantation. Am J Reprod Immunol N Y N 1989 2014, 72, 141–7. [Google Scholar] [CrossRef]
- Dimitriadis, E.; Rolnik, D.L.; Zhou, W.; Estrada-Gutierrez, G.; Koga, K.; Francisco, R.P.V.; et al. Pre-eclampsia. Nat Rev Dis Primer 2023, 9, 8. [Google Scholar] [CrossRef]
- Hanna, J; Goldman-Wohl, D; Hamani, Y; Avraham, I; Greenfield, C; Natanson-Yaron, S; et al. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med. 2006, 12, 1065–74. [Google Scholar] [CrossRef]
- Mor, G; Aldo, P; Alvero, AB. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol. 2017, 17, 469–82. [Google Scholar] [CrossRef]
- Redman, CWG; Sargent, IL. Placental Stress and Pre-eclampsia: A Revised View. Placenta 2009, 30, 38–42. [Google Scholar] [CrossRef]
- Burton, GJ; Jauniaux, E. Pathophysiology of placental-derived fetal growth restriction. Am J Obstet Gynecol. 2018, 218, S745–61. [Google Scholar] [CrossRef]
- Staff, AC. The two-stage placental model of preeclampsia: An update. J Reprod Immunol 2019, 134-135, 1–10. [Google Scholar] [CrossRef]
- UNFPA WBG; WHO; UNICEF. Trends in Maternal Mortality: 1990 to 2015; WHO Geneva, 2015. [Google Scholar]
- Abalos, E; Cuesta, C; Grosso, AL; Chou, D; Say, L. Global and regional estimates of preeclampsia and eclampsia: A systematic review. Eur J Obstet Gynecol Reprod Biol. 2013, 170, 1–7. [Google Scholar] [CrossRef]
- World Health Organization. Maternal mortality: key facts [Internet]; Geneva, Switzerland, 2023. [Google Scholar]
- World Health Organization (WHO). United Nations Children’s Fund (UNICEF). Maternal and perinatal death surveillance and response: materials to support implementation [Internet]; Who/Unicef, 2021. [Google Scholar]
- Burstein, R; Henry, NJ; Collison, ML; Marczak, LB; Sligar, A; Watson, S; et al. Mapping 123 million neonatal, infant and child deaths between 2000 and 2017. Nature 2019, 574, 353–8. [Google Scholar] [CrossRef]
- Roberts, JM; Hubel, CA. The two stage model of preeclampsia: variations on the theme. Placenta 2009, 30 Suppl A(Suppl A), S32–37. [Google Scholar] [CrossRef]
- Carr, H; Cnattingius, S; Granath, F; Ludvigsson, JF; Edstedt Bonamy, AK. Preterm Birth and Risk of Heart Failure Up to Early Adulthood. J Am Coll Cardiol. 2017, 69, 2634–42. [Google Scholar] [CrossRef]
- Crispi, F; Dominguez, C; Llurba, E; Martin-Gallan, P; Cabero, L; Gratacos, E. Placental angiogenic growth factors and uterine artery Doppler findings for characterization of different subsets in preeclampsia and in isolated intrauterine growth restriction. Am J Obstet Gynecol. 2006, 195, 201–7. [Google Scholar] [CrossRef]
- Karumanchi, SA. Angiogenic Factors in Preeclampsia: From Diagnosis to Therapy. Hypertension 2016, 67, 1072–9. [Google Scholar] [CrossRef]
- Llurba, E; Crispi, F; Verlohren, S. Update on the pathophysiological implications and clinical role of angiogenic factors in pregnancy. Fetal Diagn Ther. 2015, 37, 81–92. [Google Scholar] [CrossRef]
- Vento-Tormo, R; Efremova, M; Botting, RA; Turco, MY; Vento-Tormo, M; Meyer, KB; et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature 2018, 563, 347–53. [Google Scholar] [CrossRef]
- Blois, SM; Ilarregui, JM; Tometten, M; Garcia, M; Orsal, AS; Cordo-Russo, R; et al. A pivotal role for galectin-1 in fetomaternal tolerance. Nat Med. 2007, 13, 1450–7. [Google Scholar] [CrossRef]
- Redman, CWG; Tannetta, DS; Dragovic, RA; Gardiner, C; Southcombe, JH; Collett, GP; et al. Review: Does size matter? Placental debris and the pathophysiology of pre-eclampsia. Placenta 2012. [Google Scholar] [CrossRef]
- Deer, E; Herrock, O; Campbell, N; Cornelius, D; Fitzgerald, S; Amaral, LM; et al. The role of immune cells and mediators in preeclampsia. Nat Rev Nephrol. 2023, 19, 257–70. [Google Scholar] [CrossRef]
- Hausvater, A; Giannone, T; Sandoval, YHG; Doonan, RJ; Antonopoulos, CN; Matsoukis, IL; et al. The association between preeclampsia and arterial stiffness. J Hypertens 2012. [Google Scholar] [CrossRef]
- Herraiz, I; Droge, LA; Gomez-Montes, E; Henrich, W; Galindo, A; Verlohren, S. Characterization of the soluble fms-like tyrosine kinase-1 to placental growth factor ratio in pregnancies complicated by fetal growth restriction. Obstet Gynecol. 2014, 124, 265–73. [Google Scholar] [CrossRef]
- Maynard, SE; Min, JY; Merchan, J; Lim, KH; Li, J; Mondal, S; et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003, 111, 649–58. [Google Scholar] [CrossRef]
- Shibata, E; Rajakumar, A; Powers, RW; Larkin, RW; Gilmour, C; Bodnar, LM; et al. Soluble fms-like tyrosine kinase 1 is increased in preeclampsia but not in normotensive pregnancies with small-for-gestational-age neonates: relationship to circulating placental growth factor. J Clin Endocrinol Metab. 2005, 90, 4895–903. [Google Scholar] [CrossRef]
- Levine, RJ; Maynard, SE; Qian, C; Lim, KH; England, LJ; Yu, KF; et al. Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med. 2004, 350, 672–83. [Google Scholar] [CrossRef]
- Moss, CG; Dilworth, MR; Harris, LK; Freeman, S; Heazell, AEP. Understanding a Potential Role for the NLRP3 Inflammasome in Placenta-Mediated Pregnancy Complications. Am J Reprod Immunol N Y N 1989 2025, 93, e70077. [Google Scholar] [CrossRef]
- Alfian, I; Chakraborty, A; Yong, HEJ; Saini, S; Lau, RWK; Kalionis, B; et al. The Placental NLRP3 Inflammasome and Its Downstream Targets, Caspase-1 and Interleukin-6, Are Increased in Human Fetal Growth Restriction: Implications for Aberrant Inflammation-Induced Trophoblast Dysfunction. Cells 2022, 11(9). [Google Scholar] [CrossRef]
- Garcia-Puente, LM; Fraile-Martinez, O; García-Montero, C; Bujan, J; De León-Luis, JA; Bravo, C; et al. Placentas from Women with Late-Onset Preeclampsia Exhibit Increased Expression of the NLRP3 Inflammasome Machinery. Biomolecules 2023, 13(11). [Google Scholar] [CrossRef]
- Menkhorst, E; Santos, LL; Zhou, W; Yang, G; Winship, AL; Rainczuk, KE; et al. IL11 activates the placental inflammasome to drive preeclampsia. Front Immunol. 2023, 14, 1175926. [Google Scholar] [CrossRef]
- Szarka, A; Rigó, JJ; Lázár, L; Beko, G; Molvarec, A. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol. 2010, 11, 59. [Google Scholar] [CrossRef]
- Pinheiro, MB; Martins-Filho, OA; Mota, APL; Alpoim, PN; Godoi, LC; Silveira, ACO; et al. Severe preeclampsia goes along with a cytokine network disturbance towards a systemic inflammatory state. Cytokine 2013, 62, 165–73. [Google Scholar] [CrossRef]
- Tosun, M; Celik, H; Avci, B; Yavuz, E; Alper, T; Malatyalioğlu, E. Maternal and umbilical serum levels of interleukin-6, interleukin-8, and tumor necrosis factor-alpha in normal pregnancies and in pregnancies complicated by preeclampsia. J Matern-Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet 2010, 23, 880–6. [Google Scholar]
- Zheng, S; Xu, P; Shen, H; Shu, C. Immune dysregulation in preeclampsia: integrative analysis of peripheral transcriptomes and placental single-cell land-scapes. Front Immunol. 2025, 16, 1638603. [Google Scholar] [CrossRef]
- Rolnik, DL; Wright, D; Poon, LC; O’Gorman, N; Syngelaki, A; de Paco Matallana, C; et al. Aspirin versus Placebo in Pregnancies at High Risk for Preterm Preeclampsia. N Engl J Med. 2017, 377, 613–22. [Google Scholar] [CrossRef]
- García-Montero, C; Fraile-Martinez, O; De Leon-Oliva, D; Boaru, DL; Garcia-Puente, LM; De León-Luis, JA; et al. Exploring the Role of Mediterranean and Westernized Diets and Their Main Nutrients in the Modulation of Oxidative Stress in the Placenta: A Narrative Review. Antioxid Basel Switz 2023, 12(11). [Google Scholar] [CrossRef]
- Fernandes, KA; Lim, AI. Maternal-driven immune education in offspring. Immunol Rev. 2024, 323(1), 288–302. [Google Scholar] [CrossRef]
- Delaroque, C; Chassaing, B. Microbiome in heritage: how maternal microbiome transmission impacts next generation health. Microbiome 2025, 13(1), 196. [Google Scholar] [CrossRef]
- Andonotopo, W; Bachnas, MA; Dewantiningrum, J; Adi Pramono, MB; Sulistyowati, S; Hariyasa Sanjaya, IN; et al. Nutriepigenomics in perinatal medicine: maternal nutrition as a modulator of fetal gene expression and long-term health. J Perinat Med 2025. [Google Scholar] [CrossRef]
- Mangan, MSJ; Olhava, EJ; Roush, WR; Seidel, HM; Glick, GD; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov. 2018, 17, 688. [Google Scholar] [CrossRef]
- Mor, G; Cardenas, I. The immune system in pregnancy: a unique complexity. Am J Reprod Immunol N Y N 1989 2010, 63, 425–33. [Google Scholar]
- Racicot, K; Kwon, JY; Aldo, P; Silasi, M; Mor, G. Understanding the complexity of the immune system during pregnancy. Am J Reprod Immunol N Y N 1989 2014, 72, 107–16. [Google Scholar] [CrossRef]
- Riley, JK. Trophoblast immune receptors in maternal-fetal tolerance. Immunol Invest. 2008, 37(5), 395–426. [Google Scholar] [CrossRef]
- Huppertz, B; Ghosh, D; Sengupta, J. An integrative view on the physiology of human early placental villi. Prog Biophys Mol Biol. 2014, 114, 33–48. [Google Scholar] [CrossRef]
- Burton, GJ; Woods, AW; Jauniaux, E; Kingdom, JCP. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta 2009, 30, 473–82. [Google Scholar] [CrossRef]
- Iwasaki, A; Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat Immunol. 2015, 16(4), 343–53. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–35. [Google Scholar] [CrossRef]
- Levenson, D; Romero, R; Miller, D; Galaz, J; Garcia-Flores, V; Neshek, B; et al. The maternal-fetal interface at single-cell resolution: uncovering the cellular anatomy of the placenta and decidua. Am J Obstet Gynecol. 2025, 232, S55–79. [Google Scholar] [CrossRef]
- Moffett, A; Colucci, F. Uterine NK cells: active regulators at the maternal-fetal interface. J Clin Invest. 2014, 124, 1872–9. [Google Scholar] [CrossRef]
- Alippe, Y; Hatterschide, J; Coyne, CB; Diamond, MS. Innate immune responses to pathogens at the maternal-fetal interface. Nat Rev Immunol. 2025, 25, 869–84. [Google Scholar] [CrossRef]
- Svensson-Arvelund, J; Ernerudh, J. The Role of Macrophages in Promoting and Maintaining Homeostasis at the Fetal-Maternal Interface. Am J Reprod Immunol N Y N 1989 2015, 74, 100–9. [Google Scholar] [CrossRef]
- Houser, BL. Decidual macrophages and their roles at the maternal-fetal interface. Yale J Biol Med. 2012, 85, 105–18. [Google Scholar]
- Aluvihare, VR; Kallikourdis, M; Betz, AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol. 2004, 5, 266–71. [Google Scholar] [CrossRef]
- Robertson, SA; Care, AS; Moldenhauer, LM. Regulatory T cells in embryo implantation and the immune response to pregnancy. J Clin Invest. 2018, 128, 4224–35. [Google Scholar] [CrossRef]
- Tilburgs, T; Scherjon, SA; van der Mast, BJ; Haasnoot, GW; Versteeg-V D Voort-Maarschalk, M; Roelen, DL; et al. Fetal-maternal HLA-C mismatch is associated with decidual T cell activation and induction of functional T regulatory cells. J Reprod Immunol. 2009, 82, 148–57. [Google Scholar] [CrossRef]
- Saito, S; Nakashima, A. Review: The role of autophagy in extravillous trophoblast function under hypoxia. Placenta 2013, 34 Suppl, S79–84. [Google Scholar] [CrossRef]
- Abrahams, VM; Visintin, I; Aldo, PB; Guller, S; Romero, R; Mor, G. A role for TLRs in the regulation of immune cell migration by first trimester trophoblast cells. J Immunol Baltim Md 1950 2005, 175, 8096–104. [Google Scholar] [CrossRef]
- Palmeira, P; Quinello, C; Silveira-Lessa, AL; Zago, CA; Carneiro-Sampaio, M. IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol. 2012, 2012, 985646. [Google Scholar] [CrossRef]
- Gomez-Lopez, N; Romero, R; Xu, Y; Miller, D; Leng, Y; Panaitescu, B; et al. The immunophenotype of amniotic fluid leukocytes in normal and complicated pregnancies. Am J Reprod Immunol. 2018, 79, e12827. [Google Scholar] [CrossRef]
- Frishman, S; Nuriel-Ohayon, M; Turjeman, S; Pinto, Y; Yariv, O; Tenenbaum-Gavish, K; et al. Positive effects of diet-induced microbiome modification on GDM in mice following human faecal transfer. Gut 2024, 73, e17. [Google Scholar] [CrossRef]
- Koren, O; Konnikova, L; Brodin, P; Mysorekar, IU; Collado, MC. The maternal gut microbiome in pregnancy: implications for the developing immune system. Nat Rev Gastroenterol Hepatol 2024, 21, 35–45. [Google Scholar] [CrossRef]
- Fasano, A; Chassaing, B; Haller, D; Flores Ventura, E; Carmen-Collado, M; Pastor, N; et al. Microbiota during pregnancy and early life: role in maternal-neonatal outcomes based on human evidence. Gut Microbes 2024, 16, 2392009. [Google Scholar] [CrossRef]
- Pinto, Y; Frishman, S; Turjeman, S; Eshel, A; Nuriel-Ohayon, M; Shrossel, O; et al. Gestational diabetes is driven by microbiota-induced inflammation months before diagnosis. Gut 2023, 72, 918–28. [Google Scholar] [CrossRef]
- Huang, L; Cai, M; Li, L; Zhang, X; Xu, Y; Xiao, J; et al. Gut microbiota changes in preeclampsia, abnormal placental growth and healthy pregnant women. BMC Microbiol. 2021, 21, 265. [Google Scholar] [CrossRef]
- Aagaard, K; Ma, J; Antony, KM; Ganu, R; Petrosino, J; Versalovic, J. The placenta harbors a unique microbiome. Sci Transl Med. 2014, 6, 237ra65. [Google Scholar] [CrossRef]
- Panzer, JJ; Romero, R; Greenberg, JM; Winters, AD; Galaz, J; Gomez-Lopez, N; et al. Is there a placental microbiota? A critical review and re-analysis of published placental microbiota datasets. BMC Microbiol 2023, 23, 76. [Google Scholar] [CrossRef]
- Saadaoui, M; Djekidel, MN; Murugesan, S; Kumar, M; Elhag, D; Singh, P; et al. Exploring the composition of placental microbiome and its potential origin in preterm birth. Front Cell Infect Microbiol 2024, 14, 1486409. [Google Scholar] [CrossRef]
- de Goffau, MC; Lager, S; Sovio, U; Gaccioli, F; Cook, E; Peacock, SJ; et al. Human placenta has no microbiome but can contain potential pathogens. Nature 2019, 572, 329–34. [Google Scholar] [CrossRef]
- Arora, N; Sadovsky, Y; Dermody, TS; Coyne, CB. Microbial Vertical Transmission during Human Pregnancy. Cell Host Microbe 2017, 21, 561–7. [Google Scholar] [CrossRef]
- Romero, R; Hassan, SS; Gajer, P; Tarca, AL; Fadrosh, DW; Bieda, J; et al. The vaginal microbiota of pregnant women who subsequently have spontaneous preterm labor and delivery and those with a normal delivery at term. Microbiome 2014, 2, 18. [Google Scholar] [CrossRef]
- Zakis, DR; Paulissen, E; Kornete, L; Kaan, AMM; Nicu, EA; Zaura, E. The evidence for placental microbiome and its composition in healthy pregnancies: A systematic review. J Reprod Immunol. 2022, 149, 103455. [Google Scholar] [CrossRef]
- Reijnders, IF; Mulders, AGMGJ; Koster, MPH. Placental development and function in women with a history of placenta-related complications: a systematic review. Acta Obstet Gynecol Scand. 2018, 97(3), 248–57. [Google Scholar] [CrossRef]
- Sargent, IL; Borzychowski, AM; Redman, CWG. Immunoregulation in normal pregnancy and pre-eclampsia: an overview. Reprod Biomed Online 2006, 13, 680–6. [Google Scholar] [CrossRef]
- Tilburgs, T; Roelen, DL; van der Mast, BJ; van Schip, JJ; Kleijburg, C; de Groot-Swings, GM; et al. Differential distribution of CD4(+)CD25(bright) and CD8(+)CD28(-) T-cells in decidua and maternal blood during human pregnancy. Placenta 2006, 27, S47–53. [Google Scholar] [CrossRef]
- Redman, CWG; Sargent, IL. Preeclampsia and the systemic inflammatory response. Semin Nephrol. 2004, 24, 565–70. [Google Scholar] [CrossRef]
- Redman, CW; Sargent, IL; Staff, AC. IFPA Senior Award Lecture: making sense of pre-eclampsia - two placental causes of preeclampsia? Placenta 2014, 35, S20–25. [Google Scholar] [CrossRef]
- Boulanger, H; Bounan, S; Mahdhi, A; Drouin, D; Ahriz-Saksi, S; Guimiot, F; et al. Immunologic aspects of preeclampsia. AJOG Glob Rep. 2024, 4, 100321. [Google Scholar] [CrossRef]
- Deer, E; Vaka, VR; McMaster, KM; Wallace, K; Cornelius, DC; Amaral, LM; et al. Vascular endothelial mitochondrial oxidative stress in response to preeclampsia: a role for angiotension II type 1 autoantibodies. Am J Obstet Gynecol. 2021, 3(1). [Google Scholar] [CrossRef]
- Brien, ME; Duval, C; Palacios, J; Boufaied, I; Hudon-Thibeault, AA; Nadeau-Vallée, M; et al. Uric Acid Crystals Induce Placental Inflammation and Alter Trophoblast Function via an IL-1-Dependent Pathway: Implications for Fetal Growth Restriction. J Immunol Baltim Md 1950 2017, 198, 443–51. [Google Scholar] [CrossRef]
- Matzinger, P. The danger model: a renewed sense of self. Science 2002, 296, 301–5. [Google Scholar] [CrossRef]
- Sacks, GP; Studena, K; Sargent, K; Redman, CW. Normal pregnancy and preeclampsia both produce inflammatory changes in peripheral blood leukocytes akin to those of sepsis. Am J Obstet Gynecol. 1998, 179, 80–6. [Google Scholar] [CrossRef]
- Salomon, C; Yee, SW; Mitchell, MD; Rice, GE. The Possible Role of Extravillous Trophoblast-Derived Exosomes on the Uterine Spiral Arterial Remodeling under Both Normal and Pathological Conditions. BioMed Res Int. 2014, 2014. [Google Scholar] [CrossRef]
- Romero, R; Nien, JK; Espinoza, J; Todem, D; Fu, W; Chung, H; et al. A longitudinal study of angiogenic (placental growth factor) and anti-angiogenic (soluble endoglin and soluble vascular endothelial growth factor receptor-1) factors in normal pregnancy and patients destined to develop preeclampsia and deliver a small fo. J Matern-Fetal Neonatal Med. 2008, 21, 9–23. [Google Scholar] [CrossRef]
- Levine, RJ; Lam, C; Qian, C; Yu, KF; Maynard, SE; Sachs, BP; et al. Soluble endoglin and other circulating antiangiogenic factors in preeclampsia. N Engl J Med. 2006, 355, 992–1005. [Google Scholar] [CrossRef]
- American College of Obstetricians Gynecologists’ Committee. Gestational Hypertension and Preeclampsia. Obstet Gynecol. 2020, 135, e237-60. [Google Scholar]
- La Rocca, C; Carbone, F; Longobardi, S; Matarese, G. The immunology of pregnancy: Regulatory T cells control maternal immune tolerance toward the fetus. Immunol Lett. 2014, 162, 41–8. [Google Scholar] [CrossRef]
- Dhillion, P; Wallace, K; Herse, F; Scott, J; Wallukat, G; Heath, J; et al. IL-17-mediated oxidative stress is an important stimulator of AT1-AA and hypertension during pregnancy. Am J Physiol Regul Integr Comp Physiol. 2012, 303, R353–358. [Google Scholar] [CrossRef]
- Geldenhuys, J; Rossouw, TM; Lombaard, HA; Ehlers, MM; Kock, MM. Disruption in the Regulation of Immune Responses in the Placental Subtype of Preeclampsia. Front Immunol. 2018, 9, 1659. [Google Scholar] [CrossRef]
- Kharfi, A; Giguère, Y; Sapin, V; Massé, J; Dastugue, B; Forest, JC. Trophoblastic remodeling in normal and preeclamptic pregnancies: implication of cytokines. Clin Biochem. 2003, 36, 323–31. [Google Scholar] [CrossRef]
- Lyall, F; Robson, SC; Bulmer, JN. Spiral artery remodeling and trophoblast invasion in preeclampsia and fetal growth restriction: relationship to clinical outcome. Hypertens. 2013, 62, 1046–54. [Google Scholar] [CrossRef]
- Madazli, R; Aydin, S; Uludag, S; Vildan, O; Tolun, N. Maternal plasma levels of cytokines in normal and preeclamptic pregnancies and their relationship with diastolic blood pressure and fibronectin levels. Acta Obstet Gynecol Scand. 2003, 82, 797–802. [Google Scholar] [CrossRef]
- Aggarwal, R; Jain, AK; Mittal, P; Kohli, M; Jawanjal, P; Rath, G. Association of pro- and anti-inflammatory cytokines in preeclampsia. J Clin Lab Anal. 2019, 33, e22834. [Google Scholar] [CrossRef]
- LaMarca, B; Speed, J; Fournier, L; Babcock, SA; Berry, H; Cockrell, K; et al. Hypertension in Response to Chronic Reductions in Uterine Perfusion in Pregnant Rats. Hypertension 2008, 52, 1161–7. [Google Scholar] [CrossRef]
- Gadonski, G; LaMarca, BBD; Sullivan, E; Bennett, W; Chandler, D; Granger, JP. Hypertension Produced by Reductions in Uterine Perfusion in the Pregnant Rat. Hypertension 2006, 48, 711–6. [Google Scholar] [CrossRef]
- Dhillion, P; Wallace, K; Herse, F; Scott, J; Wallukat, G; Heath, J; et al. IL-17-mediated oxidative stress is an important stimulator of AT1-AA and hypertension during pregnancy. Am J Physiol-Regul Integr Comp Physiol. 2012, 303, R353–8. [Google Scholar] [CrossRef]
- Crispi, F; Crovetto, F; Gratacos, E. Intrauterine growth restriction and later cardiovascular function. Early Hum Dev. 2018, 126, 23–7. [Google Scholar] [CrossRef]
- Figueras, F; Gratacos, E. Stage-based approach to the management of fetal growth restriction. Prenat Diagn 2014, 34, 655–9. [Google Scholar] [CrossRef]
- Crispi, F; Figueras, F; Cruz-Lemini, M; Bartrons, J; Bijnens, B; Gratacos, E. Cardiovascular programming in children born small for gestational age and relationship with prenatal signs of severity. Am J Obstet Gynecol. 2012, 207, e1-9. [Google Scholar] [CrossRef]
- Crispi, F; Bijnens, B; Figueras, F; Bartrons, J; Eixarch, E; Le Noble, F; et al. Fetal growth restriction results in remodeled and less efficient hearts in children. Circulation 2010, 121, 2427–36. [Google Scholar] [CrossRef]
- Crispi, F; Miranda, J; Gratacós, E. Long-term cardiovascular consequences of fetal growth restriction: biology, clinical implications, and opportunities for prevention of adult disease. Am J Obstet Gynecol. 2018, 218, S869–79. [Google Scholar] [CrossRef]
- Lees, CC; Stampalija, T; Baschat, AA; da Silva Costa, F; Ferrazzi, E; Figueras, F; et al. ISUOG Practice Guidelines: diagnosis and management of small-for-gestational-age fetus and fetal growth restriction. Ultrasound Obstet Gynecol. 2020, 56, 298–312. [Google Scholar] [CrossRef]
- Miranda, J; Paules, C; Noell, G; Youssef, L; Paternina-Caicedo, A; Crovetto, F; et al. Similarity network fusion to identify phenotypes of small-for-gestational-age fetuses. iScience 2023, 26, 107620. [Google Scholar] [CrossRef]
- Zarate, MA; De Dios, RK; Balasubramaniyan, D; Zheng, L; Sherlock, LG; Rozance, PJ; et al. The Acute Hepatic NF-κB-Mediated Proinflammatory Response to Endotoxemia Is Attenuated in Intrauterine Growth-Restricted Newborn Mice. Front Immunol. 2021, 12, 706774. [Google Scholar] [CrossRef]
- Arcangeli, T; Thilaganathan, B; Hooper, R; Khan, KS; Bhide, A. Neurodevelopmental delay in small babies at term: a systematic review. Ultrasound Obstet Gynecol. 2012, 40, 267–75. [Google Scholar] [CrossRef]
- Baschat, AA. Neurodevelopment after fetal growth restriction. Fetal Diagn Ther. 2014, 36, 136–42. [Google Scholar] [CrossRef]
- Eixarch, E; Meler, E; Iraola, A; Illa, M; Crispi, F; Hernandez-Andrade, E; et al. Neurodevelopmental outcome in 2-year-old infants who were small-for-gestational age term fetuses with cerebral blood flow redistribution. Ultrasound Obstet Gynecol. 2008, 32, 894–9. [Google Scholar] [CrossRef]
- Azizieh, F; Dingle, K; Raghupathy, R; Johnson, K; VanderPlas, J; Ansari, A. Multivariate analysis of cytokine profiles in pregnancy complications. Am J Reprod Immunol. 2018, 79, e12818. [Google Scholar] [CrossRef]
- Salafia, CM; Minior, VK; Pezzullo, JC; Popek, EJ; Rosenkrantz, TS; Vintzileos, AM. Intrauterine growth restriction in infants of less than thirty-two weeks’ gestation: Associated placental pathologic features. Am J Obstet Gynecol. 1995, 173, 1049–57. [Google Scholar] [CrossRef]
- Salafia, CM; Rukat, C; Dygulska, B; Miller, RK; Misra, DP. Placental chronic inflammatory histopathology and fetal growth in a cohort with universal placental examination. Placenta 2024, 154, 193–200. [Google Scholar] [CrossRef]
- Tan, CX; Yeoh, HX; Tazilan, NA; Tan, JW; Alfian, N; Zakaria, H; et al. HIF-1A Expression in Placenta of Pregnancies Complicated with Preeclampsia and Fetal Growth Restriction. Diagnostics 2025, 15, 1843. [Google Scholar] [CrossRef]
- Ordóñez-Díaz, MD; Gil-Campos, M; Flores-Rojas, K; Muñoz-Villanueva, MC; Aguilera-García, CM; Torre-Aguilar, MJ; et al. Plasma Adipokines Profile in Prepubertal Children with a History of Prematurity or Extrauterine Growth Restriction. Nutrients 2020, 12, 1201. [Google Scholar] [CrossRef]
- Zarate, MA; De Dios, RK; Balasubramaniyan, D; Zheng, L; Sherlock, LG; Rozance, PJ; et al. The Acute Hepatic NF-κB-Mediated Proinflammatory Response to Endotoxemia Is Attenuated in Intrauterine Growth-Restricted Newborn Mice. Front Immunol. 2021, 12, 706774. [Google Scholar] [CrossRef]
- Sasaki, Y; Darmochwal-Kolarz, D; Suzuki, D; Sakai, M; Ito, M; Shima, T; et al. Proportion of peripheral blood and decidual CD4+ CD25bright regulatory T cells in pre-eclampsia. Clin Exp Immunol. 2007, 149, 139–45. [Google Scholar] [CrossRef]
- ting, Xu T; Zhou, F; C yan, Deng; G qiong, Huang; ke, Li J; dong, Wang X. Low-Dose Aspirin for Preventing Preeclampsia and Its Complications: A Meta-Analysis. J Clin Hypertens. 2015, (20), 1–7. [Google Scholar]
- Chen, J; Huai, J; Yang, H. Low-molecular-weight heparin for the prevention of preeclampsia in high-risk pregnancies without thrombophilia: a systematic review and meta-analysis. BMC Pregnancy Childbirth 2024, 24, 68. [Google Scholar] [CrossRef]
- Martins, JG; Biggio, JR; Abuhamad, A. Society for Maternal-Fetal Medicine Consult Series #52: Diagnosis and management of fetal growth restriction: (Replaces Clinical Guideline Number 3, April 2012). Am J Obstet Gynecol. 2020, 223, B2–17. [Google Scholar]
- WHO recommendations on: Antenatal corticosteroids for improving preterm birth outcomes. Geneva, 2022 .
- Liabsuetrakul, T; Yamamoto, Y; Kongkamol, C; Ota, E; Mori, R; Noma, H. Medications for preventing hypertensive disorders in high-risk pregnant women: a systematic review and network meta-analysis. Syst Rev. 2022, 11, 135. [Google Scholar] [CrossRef]
- Alves, PRMM; Fragoso, MBT; Tenório, MCS; Bueno, NB; Goulart, MOF; Oliveira, ACM. The role played by oral antioxidant therapies in preventing and treating preeclampsia: An updated meta-analysis. Nutr Metab Cardiovasc Dis NMCD 2023, 33, 1277–92. [Google Scholar] [CrossRef]
- Hobson, SR; Gurusinghe, S; Lim, R; Alers, NO; Miller, SL; Kingdom, JC; et al. Melatonin improves endothelial function in vitro and prolongs pregnancy in women with early-onset preeclampsia. J Pineal Res. 2018, 65, e12508. [Google Scholar] [CrossRef]
- Dou, Y; Lin, B; Cheng, H; Wang, C; Zhao, M; Zhang, J; et al. The reduction of melatonin levels is associated with the development of preeclampsia: a meta-analysis. Hypertens Pregnancy 2019, 38, 65–72. [Google Scholar] [CrossRef]
- Cluver, CA; Rohwer, C; Rohwer, AC. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev. 2025, 12, CD001059. [Google Scholar]
- Gregorio, BM; Souza-Mello, V; Mandarim-de-Lacerda, CA; Aguila, MB. Maternal fish oil supplementation benefits programmed offspring from rat dams fed low-protein diet. Am J Obstet Gynecol. 2008, 199, 82.e1–7. [Google Scholar] [CrossRef]
- Crovetto, F; Crispi, F; Borras, R; Paules, C; Casas, R; Martín-Asuero, A; et al. Mediterranean diet, Mindfulness-Based Stress Reduction and usual care during pregnancy for reducing fetal growth restriction and adverse perinatal outcomes: IMPACT BCN (Improving Mothers for a better PrenAtal Care Trial BarCeloNa): a study protocol for a randomized controlled trial. Trials 2021, 22, 362. [Google Scholar]
- Zhou, J; Yan, P; Ma, W; Li, J. Cytokine modulation and immunoregulation of uterine NK cells in pregnancy disorders. Cytokine Growth Factor Rev. 2025, 81, 40–53. [Google Scholar] [CrossRef] [PubMed]
- Santos, KN; Bizzotto, JQ; Bueno-Pereira, TO; Romao-Veiga, M; Ribeiro-Vasques, VR; Oliveira, LRC; et al. Preeclamptic plasma disrupts endothelial function and promotes inflammation in endothelial cells: beneficial effects of glibenclamide and MCC950 in this scenario. J Recept Signal Transduct Res. 2025, 45, 237–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, X; Travis, OK; Shields, CA; Tardo, GA; Giachelli, C; Nutter, CW; et al. NLRP3 inhibition improves maternal hypertension, inflammation, and vascular dysfunction in response to placental ischemia. Am J Physiol-Regul Integr Comp Physiol. 2023, 324, R556–67. [Google Scholar] [CrossRef] [PubMed]
- Grimes, S; Bombay, K; Lanes, A; Walker, M; Corsi, DJ. Potential biological therapies for severe preeclampsia: a systematic review and meta-analysis. BMC Pregnancy Childbirth 2019, 19, 163. [Google Scholar] [CrossRef]
- Magee, LA; Brown, MA; Hall, DR; Gupte, S; Hennessy, A; Karumanchi, SA; et al. The 2021 International Society for the Study of Hypertension in Pregnancy classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2022, 27, 148–69. [Google Scholar] [CrossRef]
- Magee, LA; Smith, GN; Bloch, C; Côté, AM; Jain, V; Nerenberg, K; et al. Guideline No. 426: Hypertensive Disorders of Pregnancy: Diagnosis, Prediction, Prevention, and Management. J Obstet Gynaecol Can JOGC J Obstet Gynecol Can JOGC 2022, 44, 547–571.e1. [Google Scholar] [CrossRef]
- Aryani, A; Denecke, B. Exosomes as a Nanodelivery System: a Key to the Future of Neuromedicine? Mol Neurobiol 2016, 53, 818–34. [Google Scholar] [CrossRef]
- An, T; Qin, S; Xu, Y; Tang, Y; Huang, Y; Situ, B; et al. Exosomes serve as tumour markers for personalized diagnostics owing to their important role in cancer metastasis. J Extracell Vesicles 2015, 4(1). [Google Scholar] [CrossRef]
- Atay, S; Gercel-Taylor, C; Kesimer, M; Taylor, DD. Morphologic and proteomic characterization of exosomes released by cultured extravillous trophoblast cells. Exp Cell Res. 2011, 317, 1192–202. [Google Scholar] [CrossRef]
- Colombo, M; Raposo, G; Théry, C. Biogenesis, Secretion, and Intercellular Interactions of Exosomes and Other Extracellular Vesicles. Annu Rev Cell Dev Biol. 2014, 30, 255–89. [Google Scholar] [CrossRef]
- Bullerdiek, J; Flor, I. Exosome-delivered microRNAs of "chromosome 19 microRNA cluster" as immunomodulators in pregnancy and tumorigenesis. Mol Cytogenet. 2012, 5, 27. [Google Scholar] [CrossRef]
- Holder, B; Jones, T; Sancho Shimizu, V; Rice, TF; Donaldson, B; Bouqueau, M; et al. Macrophage Exosomes Induce Placental Inflammatory Cytokines: A Novel Mode of Maternal-Placental Messaging. Traffic 2016, 17, 168–78. [Google Scholar] [CrossRef]
- Mincheva-Nilsson, L; Baranov, V. Placenta-derived exosomes and syncytiotrophoblast microparticles and their role in human reproduction: immune modulation for pregnancy success. Am J Reprod Immunol. 2014, 72, 440–57. [Google Scholar] [CrossRef]
- Salomon, C; Kobayashi, M; Ashman, K; Sobrevia, L; Mitchell, MD; Rice, GE. Hypoxia-induced changes in the bioactivity of cytotrophoblast-derived exosomes. PloS One 2013, 8, e79636. [Google Scholar] [CrossRef]
- Salomon, C; Guanzon, D; Scholz-Romero, K; Longo, S; Correa, P; Illanes, SE; et al. Placental Exosomes as Early Biomarker of Preeclampsia: Potential Role of Exosomal MicroRNAs Across Gestation. J Clin Endocrinol Metab. 2017, 102, 3182–94. [Google Scholar] [CrossRef]
- Lykoudi, A; Kolialexi, A; Lambrou, GI; Braoudaki, M; Siristatidis, C; Papaioanou, GK; et al. Dysregulated placental microRNAs in Early and Late onset Preeclampsia. Placenta 2018, 61, 24–32. [Google Scholar] [CrossRef]
- Miranda, J; Paules, C; Nair, S; Lai, A; Palma, C; Scholz-Romero, K; et al. Placental exosomes profile in maternal and fetal circulation in intrauterine growth restriction - Liquid biopsies to monitoring fetal growth. Placenta 2018, 64, 34–43. [Google Scholar] [CrossRef]
- Taglauer, ES; Fernandez-Gonzalez, A; Willis, GR; Reis, M; Yeung, V; Liu, X; et al. Mesenchymal stromal cell-derived extracellular vesicle therapy prevents preeclamptic physiology through intrauterine immunomodulation†. Biol Reprod. 2021, 104, 457–67. [Google Scholar] [CrossRef] [PubMed]
| Category | Type | Mechanism of Action | Evidence Level |
|---|---|---|---|
| Low-Dose Aspirin | Established | COX-1 inhibition → ↓ platelet aggregation & inflammation | High – Multiple RCTs in PE prevention |
| Low Molecular Weight Heparin | Established | Anticoagulation + modulation of complement & leukocyte trafficking | Moderate – Observational studies & some RCTs |
| Antenatal Corticosteroids | Established | Promote fetal lung maturation, transient anti-inflammatory effect | High – Standard of care in threatened preterm birth |
| Antioxidants & Nutritional Modulation | Adjunctive | Reduce oxidative stress, improve endothelial function | Low to Moderate – Inconsistent RCT results |
| Lifestyle & Microbiome Modulation | Adjunctive | Improve immune tolerance & reduce systemic inflammation | Low – Ongoing trials |
| Cytokine-Targeted Biologics | Emerging | Neutralize pro-inflammatory cytokines (e.g., TNF-α, IL-6) | Experimental – Limited pregnancy-specific data |
| Inflammasome Inhibitors | Emerging | Block NLRP3 inflammasome activation | Preclinical – Animal models |
| Cell-Based Therapies | Emerging | Promote immune tolerance & tissue repair at maternal–fetal interface | Preclinical – Early-phase studies |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




