4. DAMP-Driven Biomarkers and Pathways Relevant to Precision Radiotherapy
Importantly, the evaluation of DAMPs extends beyond single molecules. Composite signatures integrating HMGB1, CRT exposure, and interferon responses have been shown to align with hallmarks of immunogenic cell death across several tumor types, including glioma. In clinical contexts, such signatures have been associated with immune activation and, in selected settings, with improved treatment outcomes when radiotherapy is combined with immunomodulatory approaches. Although glioblastoma remains challenging, the consistency of these biological relationships underscores their relevance for biomarker development rather than isolated experimental phenomena.
Another translational area informed by DAMP biology is treatment monitoring. Serial sampling studies demonstrate that radiation-induced DAMP release occurs dynamically over the course of therapy (hours, days) and can be detected in liquid biopsy formats, including plasma and cerebrospinal fluid. At the same time, imaging techniques capable of capturing inflammation, perfusion changes, or microenvironmental remodeling offer complementary information on downstream immune effects. Integrating molecular and imaging biomarkers therefore provides a realistic pathway toward biologically guided adaptation of therapy, where biological signals reflect treatment effectiveness more directly than anatomical change alone [
12,
15,
21,
22,
23].
Nanotechnology contributes further precision by enabling controlled delivery and measurement within this biological framework. Several nanotheranostic platforms have been engineered to co-localize therapeutic effects with imaging components, allowing simultaneous intervention and monitoring. In preclinical glioma models, nanoscale radiosensitizers, immunomodulators, and targeted carriers have been used to enhance local radiation effects while permitting in-vivo visualization of distribution and response. When evaluated alongside DAMP-related markers, such systems provide complementary information: DAMPs characterize biological impact, whereas nanotheranostic probes inform localization and kinetics of therapy [
23,
25].
Radiation modality is another dimension with direct translational relevance. Comparative investigations indicate that distinct radiation qualities may differentially influence DAMP release, mitochondrial stress responses, and downstream immune activation. Studies of proton irradiation, in particular, have reported differences in DNA damage patterning and associated immune signalling relative to photon therapy. [
23] These observations do not imply superiority of one modality over another; rather, they indicate that radiation type contributes to the biological signature that can be captured and potentially used to guide clinical decision-making. In this context, the alignment of dosimetry, molecular biomarkers and imaging represents a coherent strategy to move toward biologically informed radiotherapy planning.
In
Table 1 we summarized published evidence comparing photon, proton, carbon-ion and BNCT irradiation with respect to the induction of canonical DAMPs. Across modalities, the same core DAMPs (ecto-calreticulin, HMGB1, ATP and extracellular DNA) are consistently observed, with differences mainly reflecting quantitative and kinetic variation rather than modality-specific DAMP species.
DAMP-related signatures and ICD hallmarks provide quantifiable indicators of radiation-induced immune engagement; imaging-enabled nanoplatforms allow localized therapy and simultaneous monitoring; and modality-specific biological effects create opportunities to match treatment delivery with tumor biology. The cumulative evidence indicates that integrating these components is feasible within prospective clinical research, offering a structured pathway toward precision radiotherapy that is grounded in measurable biological parameters rather than theoretical assumptions.
4.1. Do Different Radiation Modalities Induce Different DAMPs?
Across preclinical and early translational studies, photon, proton and high-LET radiations (carbon ions, BNCT) consistently induce the same canonical DAMPs associated with immunogenic cell death (ICD): surface-exposed calreticulin (ecto-CRT), extracellular ATP, HMGB1 release and the presence of nuclear or mitochondrial DNA in the extracellular space. These DAMPs signal through shared receptor systems – CD91 for calreticulin, P2RX7/P2RY2 for ATP, TLR4 and RAGE for HMGB1, and the cGAS–STING pathway for cytosolic DNA – driving the established ICD cascade. Comprehensive reviews by Vaes et al. and Zhu et al. emphasize that these molecules and pathways are common across different radiation regimens, rather than specific to a particular modality [
10].
For photons vs. protons, the most direct mechanistic comparison comes from Gameiro et al. In multiple human tumor cell lines, including radioresistant subsets, they showed that proton and photon irradiation at iso-effective doses induced a “common immunogenic modulation signature”: upregulation of MHC class I, ICAM-1, tumor antigens (CEA, MUC1) and calreticulin exposure on the cell surface, which in turn increased susceptibility to antigen-specific T-cell killing [
16]. The authors explicitly concluded that tumor cells surviving proton or photon radiation share the same immunogenic phenotype, with no evidence of modality-specific DAMPs [
16,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29]
Huang et al. extended this observation by directly comparing ecto-CRT induction after photon, proton and carbon-ion irradiation in several tumor cell lines, including U251 glioma. All three modalities increased ecto-CRT in a time- and dose-dependent manner. At iso-doses, proton and photon beams were essentially equivalent in inducing ecto-CRT, whereas carbon ions produced stronger CRT exposure at 2 and 4 Gy but not at higher doses. This confirms that the DAMP species are the same for low-LET photons and protons; high-LET heavy ions change the intensity, not the identity, of DAMP signalling. [
27,
28,
29]
The same pattern holds for HMGB1. Yoshimoto et al. examined HMGB1 release from multiple human cancer cell lines after X-ray versus carbon-ion irradiation and found that both modalities induced HMGB1 release, and when doses were normalised to iso-survival levels, X-rays and carbon ions were equally effective at inducing HMGB1 [
14]. Onishi et al. later showed that increasing LET within carbon-ion beams further enhances HMGB1 release, reinforcing that high-LET radiation can quantitatively boost HMGB1 while still utilising the same molecular mediator [
29].
For BNCT, Imamichi et al. demonstrated that both BNCT and γ-irradiation trigger extracellular HMGB1 release in vitro, but BNCT produce a more pronounced and sustained HMGB1 elevation in mouse plasma, peaking around day 3 after treatment. Again, the DAMP is HMGB1 in both conditions; BNCT modifies the kinetics and amplitude, not the DAMP repertoire. [
13]. The data support a conclusion:
photon, proton, carbon-ion and BNCT all induce the same canonical DAMPs associated with ICD. High-LET modalities can enhance the magnitude and timing of DAMP release, but no modality-specific DAMP species have been consistently identified. Differences are primarily quantitative and kinetic, not qualitative.
4.2. How can DAMPs Influence Radiotherapy Outcomes – and How Does RT Choice Interact with This Biology?
DAMPs influence radiotherapy outcomes by shaping the balance between effective antitumor immunity and tolerogenic or chronically inflamed microenvironments.
Mechanistically, the sequence is well defined. Early after irradiation, calreticulin translocates to the cell surface, acting as an “eat-me” signal that facilitates phagocytosis of dying tumor cells by dendritic cells via CD91. Extracellular ATP is released and engages P2RX7, promoting NLRP3 inflammasome activation and IL-1β secretion, which supports dendritic cell maturation. Subsequently, HMGB1 is released and binds TLR4 and RAGE on antigen-presenting cells, enhancing antigen processing and cross-presentation. In parallel, leakage of nuclear and mitochondrial DNA into the cytosol or extracellular space activates cGAS–STING, leading to type I interferon production and a transcriptional programme that recruits and primes CD8
+ T cells [
9].
Vaes et al. systematically reviewed clinical data where radiotherapy-induced ICD markers were assessed in patients and found that elevated HMGB1, ecto-CRT and activation of IFN-I–related signatures correlate with features of immune activation and, in selected settings, with improved outcomes in combination with immunotherapies [
9]. Zhu et al. summarised similar relationships across radiotherapy trials where ICD hallmarks were linked to enhanced infiltration of effector T cells and better responses to immune checkpoint blockade, particularly in tumors historically considered “cold” [
10].
Another clinical example is provided by Clasen et al., who performed weekly plasma sampling in head-and-neck squamous cell carcinoma patients receiving definitive radiochemotherapy with photons. Plasma HMGB1, measured by ELISA, increased dynamically over the course of treatment in most patients. Importantly, specific HMGB1 trajectories correlated with oncologic outcome: patients with a sustained increase in HMGB1 during radiochemotherapy had more favourable local control, whereas decreasing or flat profiles were associated with higher progression risk. HMGB1 dynamics also correlated with tumor volume, CRP and treatment-related toxicity [
27]. This study shows that DAMP levels are not merely a byproduct of damage but carry prognostic information relevant to treatment success.
In preclinical models, high-LET modalities (carbon ions, BNCT) amplify these same DAMP-mediated mechanisms. Yoshimoto and Onishi demonstrated that carbon ions induce HMGB1 release at levels comparable or higher than X-rays at iso-survival doses and can more strongly enhance downstream immune responses in combination with immunotherapy [
14,
29]. Authors showed that BNCT produces an early and sustained HMGB1 plasma peak, which they propose as a biomarker of BNCT effectiveness. High-LET radiations magnify the same DAMP-driven immunological cascade and can, under controlled conditions, translate into superior antitumor immunity – but the underlying DAMP biology remains shared.
For proton versus photon radiotherapy, the key message from Gameiro and Huang is that at the tumor-cell level, DAMP-related immunogenic modulation is very similar: both modalities induce ecto-CRT, upregulate antigen presentation machinery and enhance susceptibility to CTL killing. The clinically relevant difference arises more at the systemic immune level than at the level of DAMP species per se. Multiple clinical and dosimetric studies (in glioblastoma and other tumors) have demonstrated that proton plans substantially reduce low-dose exposure of circulating blood and lymphoid organs compared to photons, resulting in lower rates of severe radiation-induced lymphopenia. Because DAMP-driven ICD requires functional dendritic cells and T-cell populations to translate into effective immunity, preservation of the host immune compartment is likely to enhance the clinical impact of a given DAMP/ICD signal.
Thus, from a translational standpoint:
Different RT types do not fundamentally change which DAMPs are generated,
high-LET beams (C-ions, BNCT) can increase how much and how fast they are released,
protons may improve the conditions in which DAMP-induced immunity operates, by sparing blood and immune organs, even if the local DAMP pattern is similar to photons.
This implies that the choice of RT modality can influence outcome indirectly via DAMP biology: by modulating the intensity of ICD (high-LET) or preserving the immune system’s ability to respond (protons), rather than by switching to entirely different DAMP pathways.
4.3. Can DAMPs be Markers of Radiotherapy Success?
The accumulated evidence strongly supports the idea that DAMPs and ICD signatures are promising biomarkers of radiotherapy effectiveness, although they are not yet fully validated predictive or surrogate markers in glioblastoma.
In the clinical setting, Vaes et al. summarised multiple studies where ICD markers were measured during or after radiotherapy in different tumor types. HMGB1 increases after CRT for oesophageal, rectal and head-and-neck cancers; ecto-CRT in tumor biopsies correlates with dendritic cell infiltration and favourable prognosis in cervical cancer; and type I interferon–related gene signatures have been associated with better response to RT plus immunotherapy in several solid tumors. Clasen’s weekly HMGB1 study is a particularly clear demonstration that serial DAMP measurements during RT carry prognostic information and can be used to stratify patients by risk, fulfilling key criteria for biomarkers of treatment success.
Preclinical data provide mechanistic support: blocking HMGB1, CRT or ATP signalling attenuates the immune contribution to tumor control after RT, while enhancing these pathways (e.g. via STING agonists) augments T-cell responses and abscopal effects. These interventions modulate radiotherapy outcome in a manner tightly coupled to DAMP biology, strengthening the rationale to use DAMP signatures as biomarkers and potential therapeutic targets.
In glioblastoma specifically, systematic liquid-biopsy studies have shown that circulating DNA, exosomes and immune-related proteins can be monitored over time and correlate with disease burden and treatment response, even though DAMP-specific panels (HMGB1, ecto-CRT, mtDNA) are only beginning to be explored [
21]. The analytical tools – ELISA, qPCR/digital PCR, EV proteomics and, increasingly, DAMP-sensitive imaging probes – already exist and have been proven feasible in other tumor types.
DAMP-related markers fulfil the criteria of “promising biomarkers” of radiotherapy success: they can be repeatedly measured, show dynamic changes over the course of treatment, and correlate with immune activation and clinical outcome in several tumor entities. However, they have not yet reached the status of fully validated predictive or surrogate endpoints in glioblastoma, and their integration into clinical decision-making will require prospective, biomarker-driven trials.
In this framework, our own experience – that proton radiotherapy appears to produce a qualitatively different immune context than photon RT – fits very well with the available data: the DAMP biology at the tumor level is shared; the systemic environment in which these signals act is improved by proton plans that better preserve the immune system.
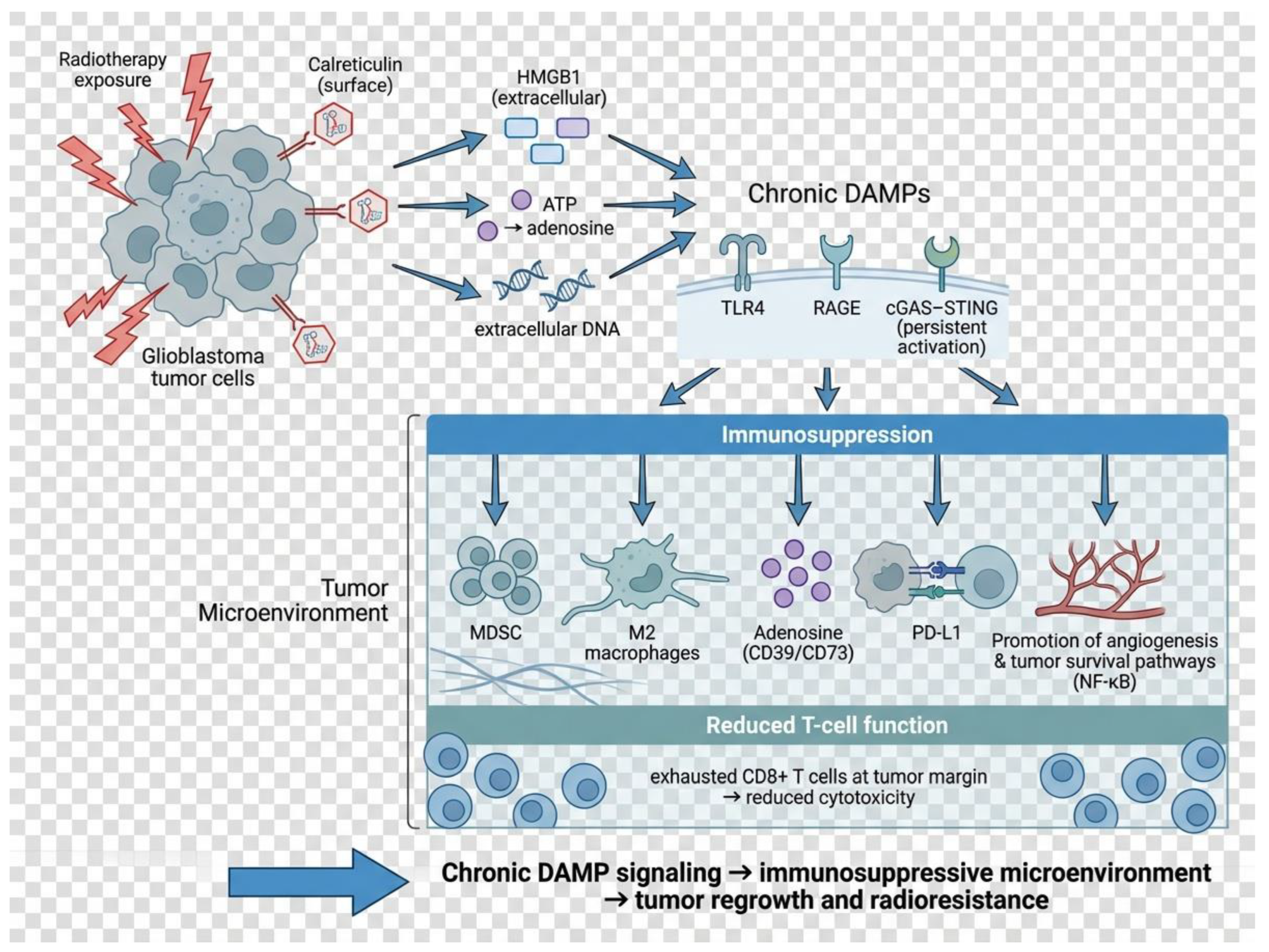
4.4. When DAMPs Promote Tolerance Rather than Immunity
DAMP signaling is fundamentally dualistic. Although acute, well-orchestrated release of calreticulin, ATP, HMGB1, and extracellular DNA promotes ICD, persistent or dysregulated DAMP release generates a biological milieu that paradoxically sustains tumor survival and treatment resistance. In the context of chronic tissue injury, repeated fractions of radiotherapy and persistent tumor necrosis, DAMP-driven activation of pattern-recognition receptors (PRRs)—including TLR4, RAGE, and cGAS–STING—can shift from initiating protective immunity toward reinforcing chronic, tumor-supportive inflammation [
1,
2,
3].
This tolerogenic reprogramming proceeds through several, partly overlapping mechanisms. First, chronic HMGB1 exposure enhances NF-κB signaling, angiogenesis, DNA repair capacity and autophagy, ultimately conferring radioresistance at the tumor-cell level [
4,
5,
6,
7]. Experimental models demonstrate that blocking extracellular HMGB1 reverses myeloid recruitment and restores radiosensitivity after irradiation, highlighting HMGB1 as a central node of therapy resistance [
8,
9,
10].
Second, ATP released as an early DAMP is rapidly catabolized by CD39 and CD73, generating adenosine, a potent immunosuppressive metabolite. Adenosine signaling via A2A and A2B receptors limits cytotoxic T- and NK-cell function, promotes regulatory T cells, and fosters vascular remodeling—thereby attenuating radiotherapy-induced antitumor immunity and reducing responses to immune checkpoint inhibitors. Therapeutic targeting of the CD39–CD73–A2AR axis restores sensitivity to radiotherapy in multiple preclinical models, illustrating how a DAMP that initially promotes ICD can evolve into a dominant suppressive pathway when chronically present [
11,
12,
13].
Third, persistent DAMP signaling drives the accumulation and functional polarization of myeloid-derived suppressor cells (MDSC) and tumor-associated macrophages (TAM), which inhibit antigen presentation, suppress T-cell infiltration, and upregulate immune checkpoints such as PD-L1. This phenomenon has been extensively documented across tumor types, where DAMP-dependent sterile inflammation correlates with immune escape, metastatic dissemination, and therapeutic failure [
14,
15,
16]. Recent genomic signatures enriched for DAMP-related genes further link these pathways with immunosuppressive tumor microenvironments and inferior clinical outcomes [
17].
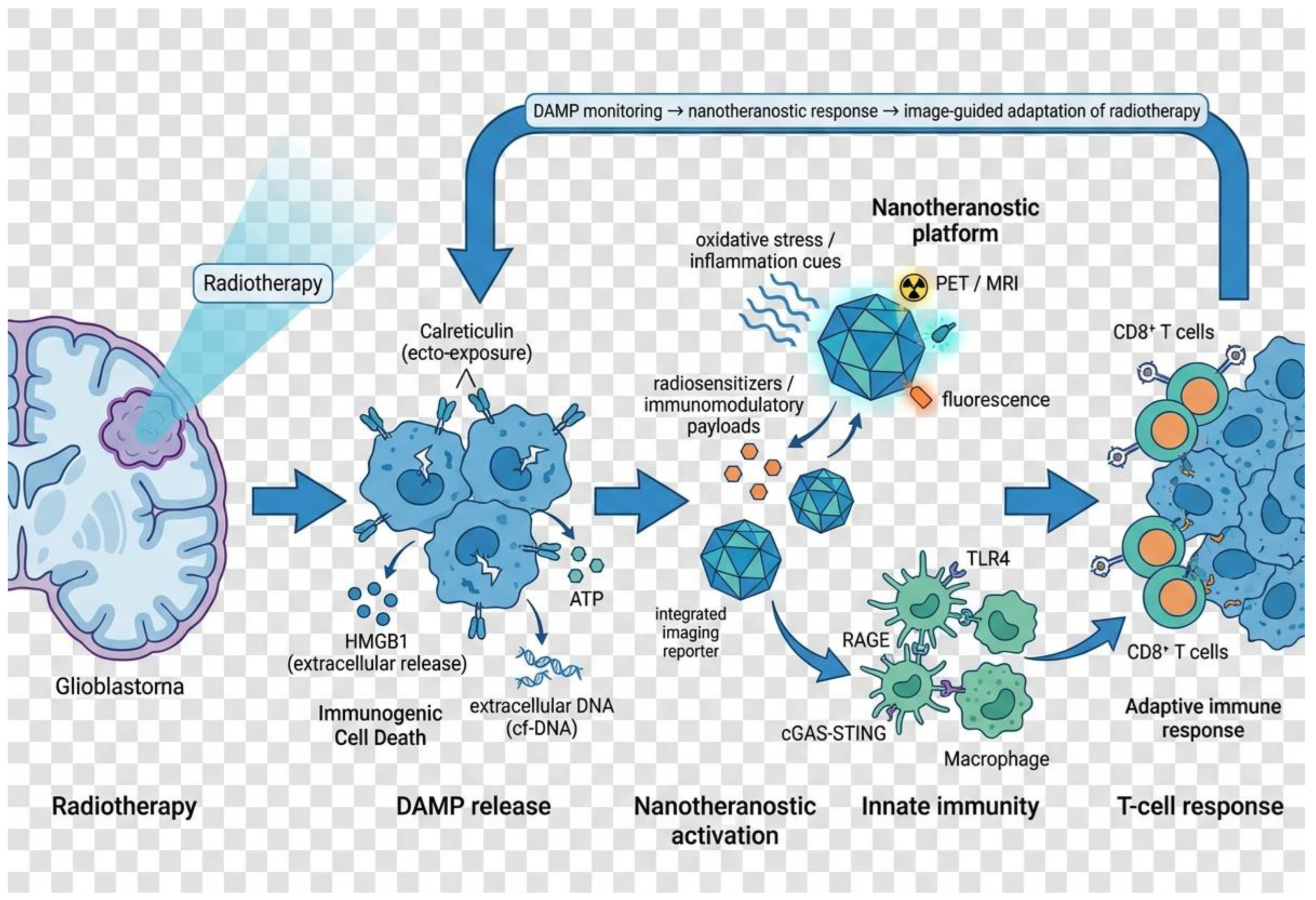
Figure 2.
DAMP-guided nanotheranostics enhance radiation-induced antitumor immunity. Radiotherapy induces immunogenic cell death and release of DAMPs (calreticulin, HMGB1, ATP, extracellular DNA). Nanotheranostic platforms sense radiation-induced stress, deliver radiosensitizers or immunomodulatory payloads, and provide imaging feedback (PET/MRI/fluorescence). DAMP signaling activates dendritic cells and macrophages via TLR4, RAGE and cGAS–STING, leading to T-cell priming and tumor infiltration. Integration of molecular monitoring with imaging enables adaptive, biology-guided radiotherapy.
Figure 2.
DAMP-guided nanotheranostics enhance radiation-induced antitumor immunity. Radiotherapy induces immunogenic cell death and release of DAMPs (calreticulin, HMGB1, ATP, extracellular DNA). Nanotheranostic platforms sense radiation-induced stress, deliver radiosensitizers or immunomodulatory payloads, and provide imaging feedback (PET/MRI/fluorescence). DAMP signaling activates dendritic cells and macrophages via TLR4, RAGE and cGAS–STING, leading to T-cell priming and tumor infiltration. Integration of molecular monitoring with imaging enables adaptive, biology-guided radiotherapy.
Glioblastoma exemplifies this biology. Necrotic tumor cores continuously release DAMPs—including HMGB1, heat-shock proteins, and other TLR ligands—driving polarization of glioma-associated macrophages toward an M2-like phenotype, suppression of MHC expression, and induction of checkpoint molecules. These effects create a TME that is highly resistant to radiotherapy and poorly responsive to immunotherapy, despite the presence of abundant inflammatory signals [
18,
19,
20]. Thus, in GBM, DAMP signaling frequently becomes tolerogenic rather than immunogenic, converting radiation-induced injury into a chronic, self-reinforcing program of immune suppression.
The published data argue that ICD should not be conceptualized as a binary switch but rather as a continuum. At one end, coordinated, transient DAMP release promotes effective dendritic-cell activation and cytotoxic T-cell recruitment; at the other, sustained or excessive DAMP signaling drives myeloid suppression, checkpoint induction, vascular remodeling, and radiotherapy resistance. Understanding this spectrum—and the conditions that shift DAMP signaling from immunity to tolerance—is essential for designing biomarker-guided strategies and rational combinations capable of converting radiation-induced damage into durable antitumor immunity.