Submitted:
04 January 2026
Posted:
05 January 2026
You are already at the latest version
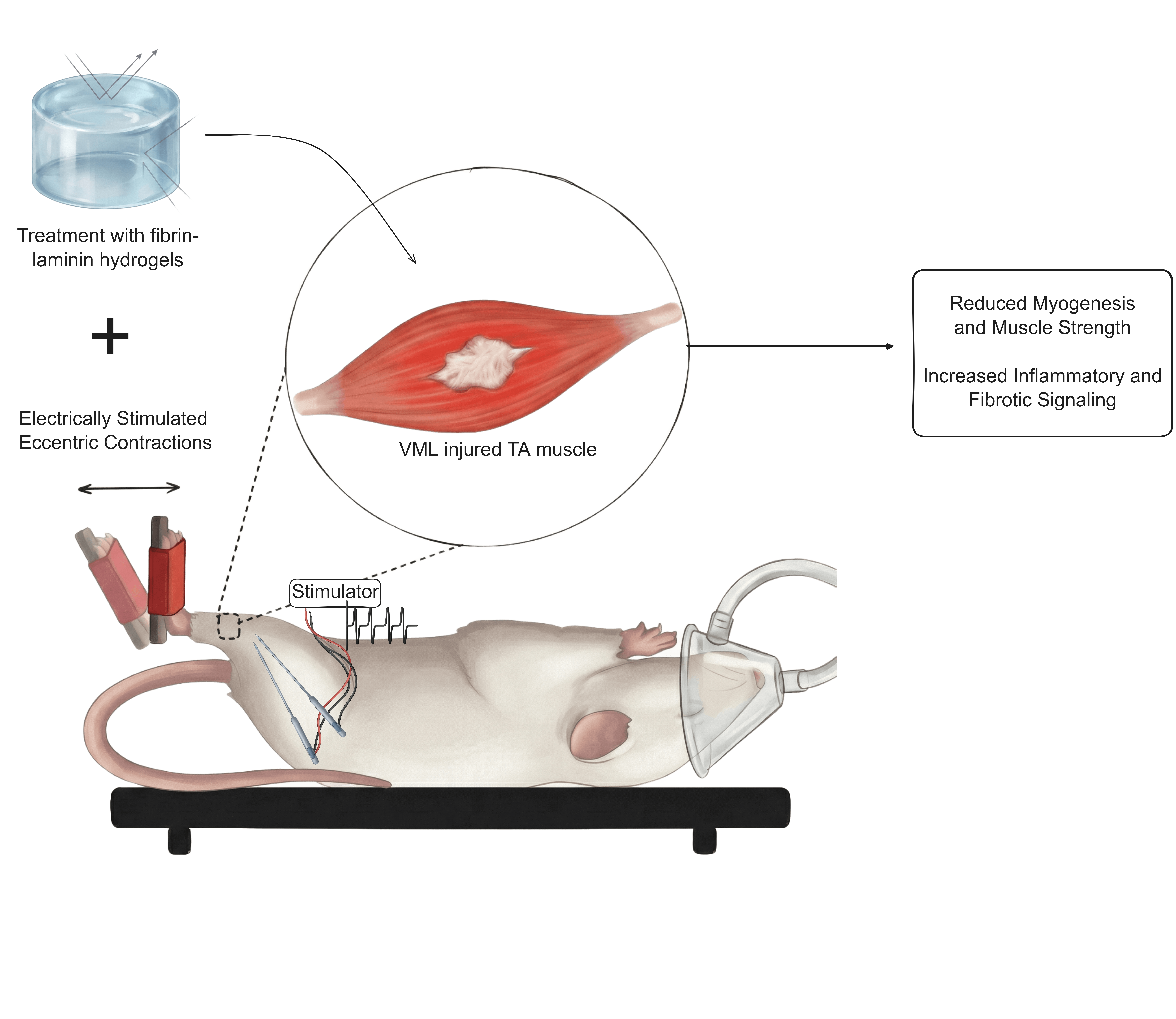
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Hydrogel Synthesis
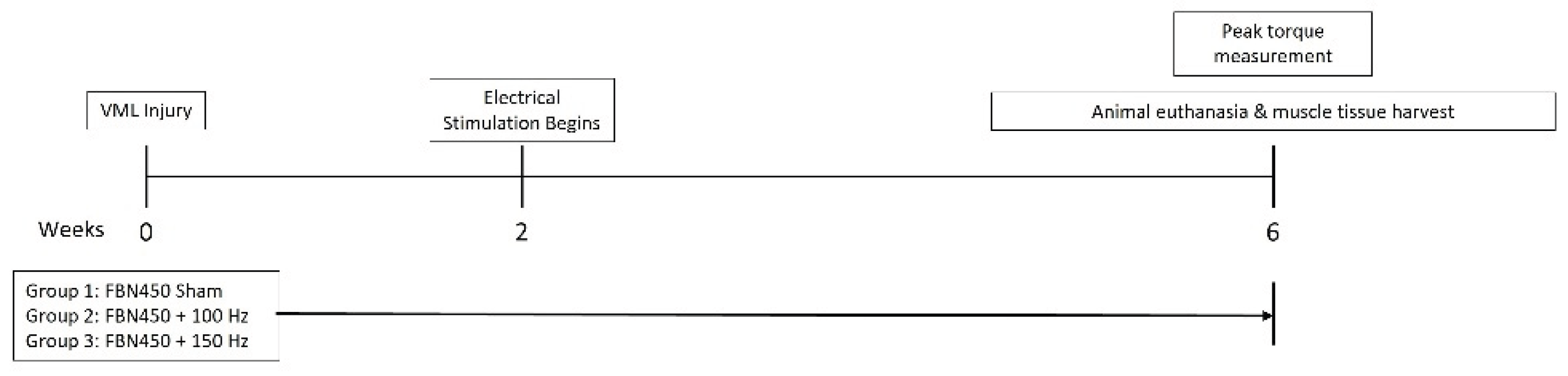
2.2. Rodent Model of VML
2.3. Electrical Stimulation Protocol
2.4. In Vivo Functional Testing
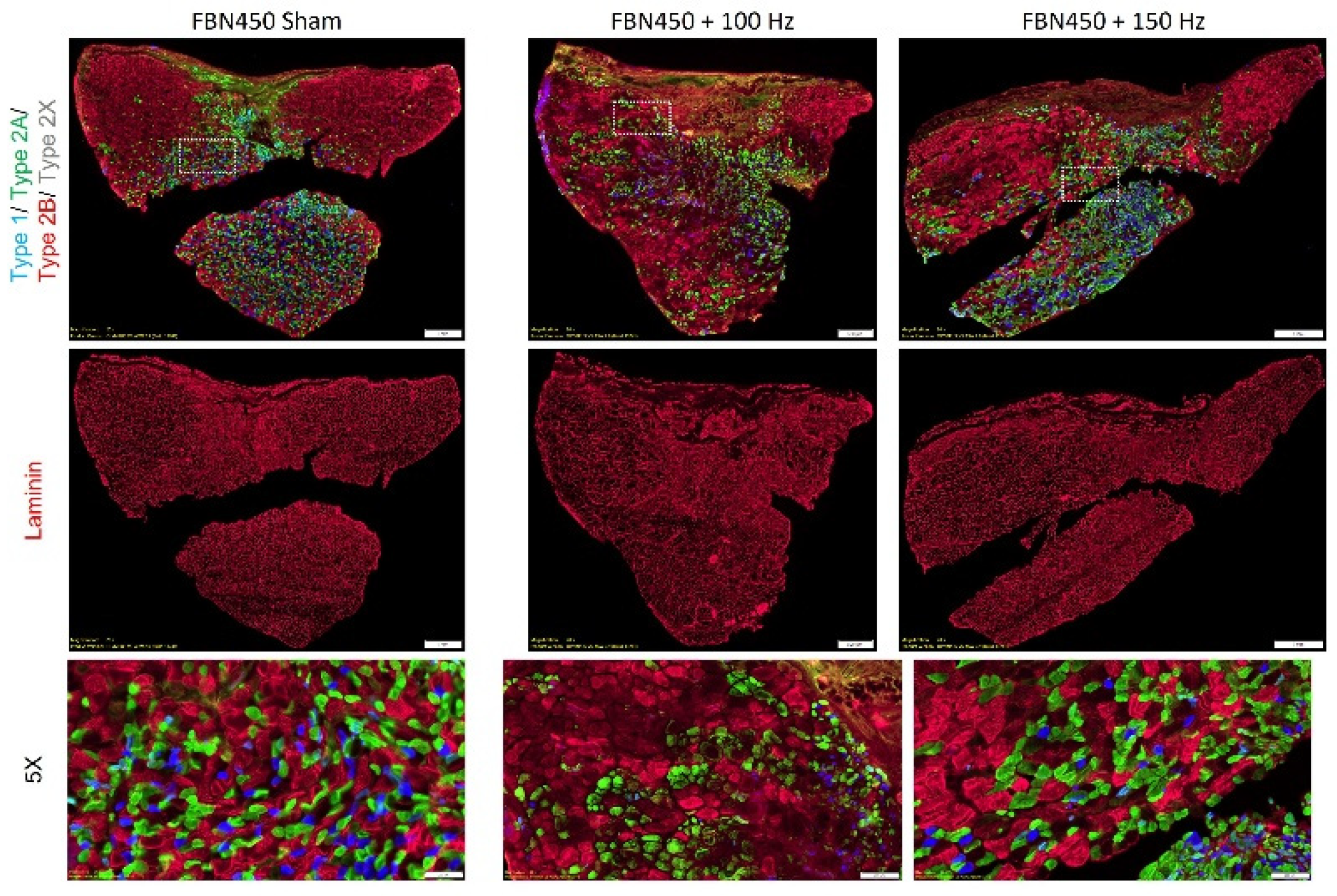
2.5. Histological and Immunohistochemistry Analysis
2.6. Gene Expression Analysis
2.7. Statistical Analysis
3. Results
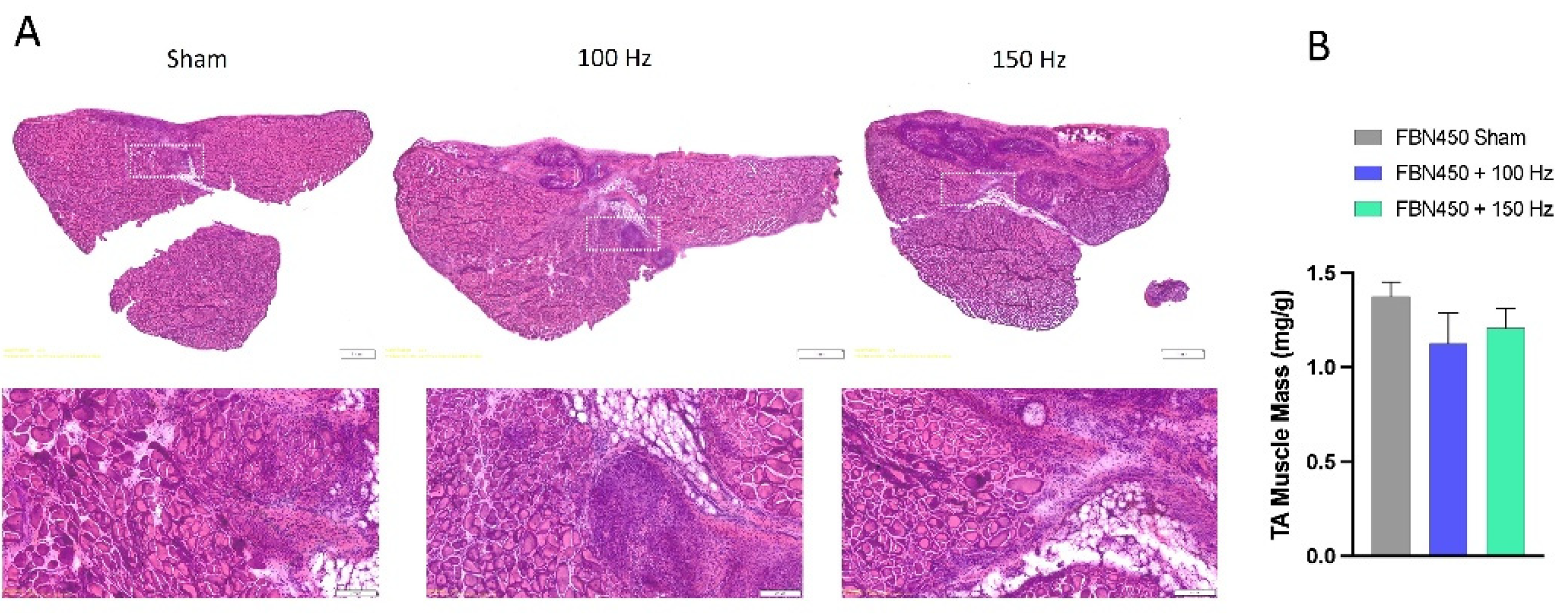
3.1. Muscle Mass
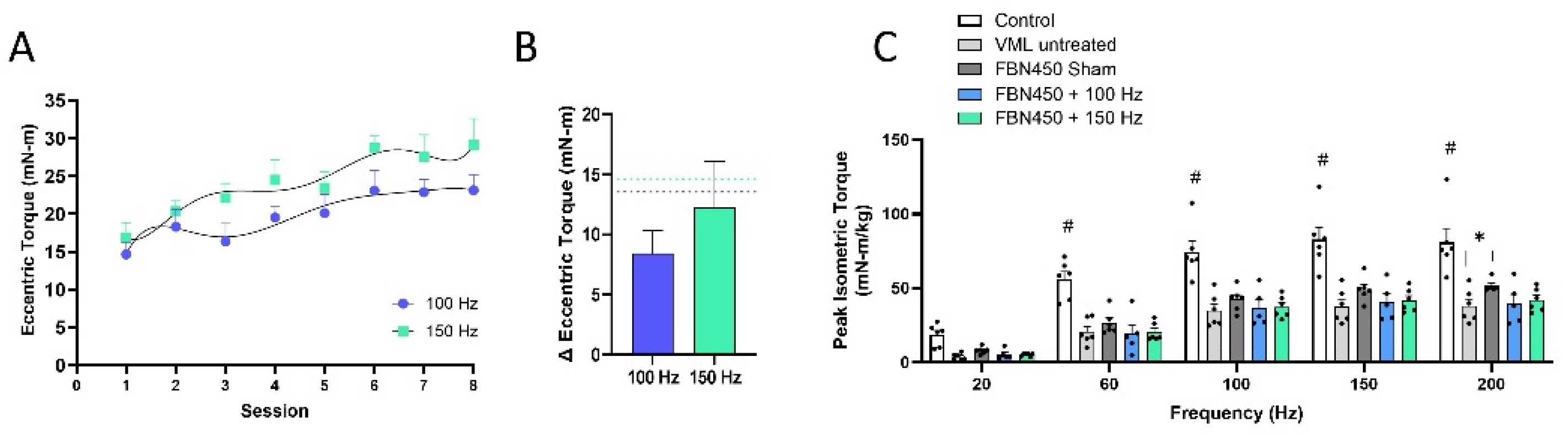
3.2. Eccentric Torque and Muscle Strength
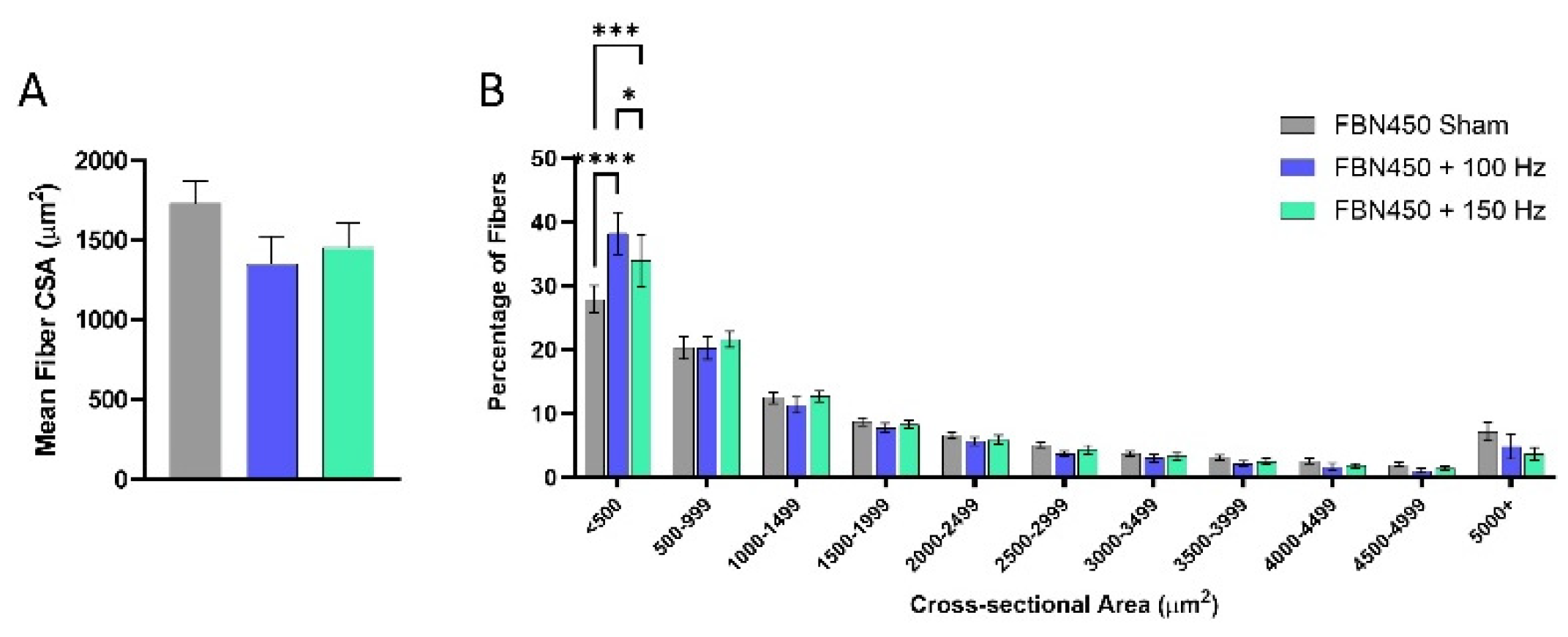
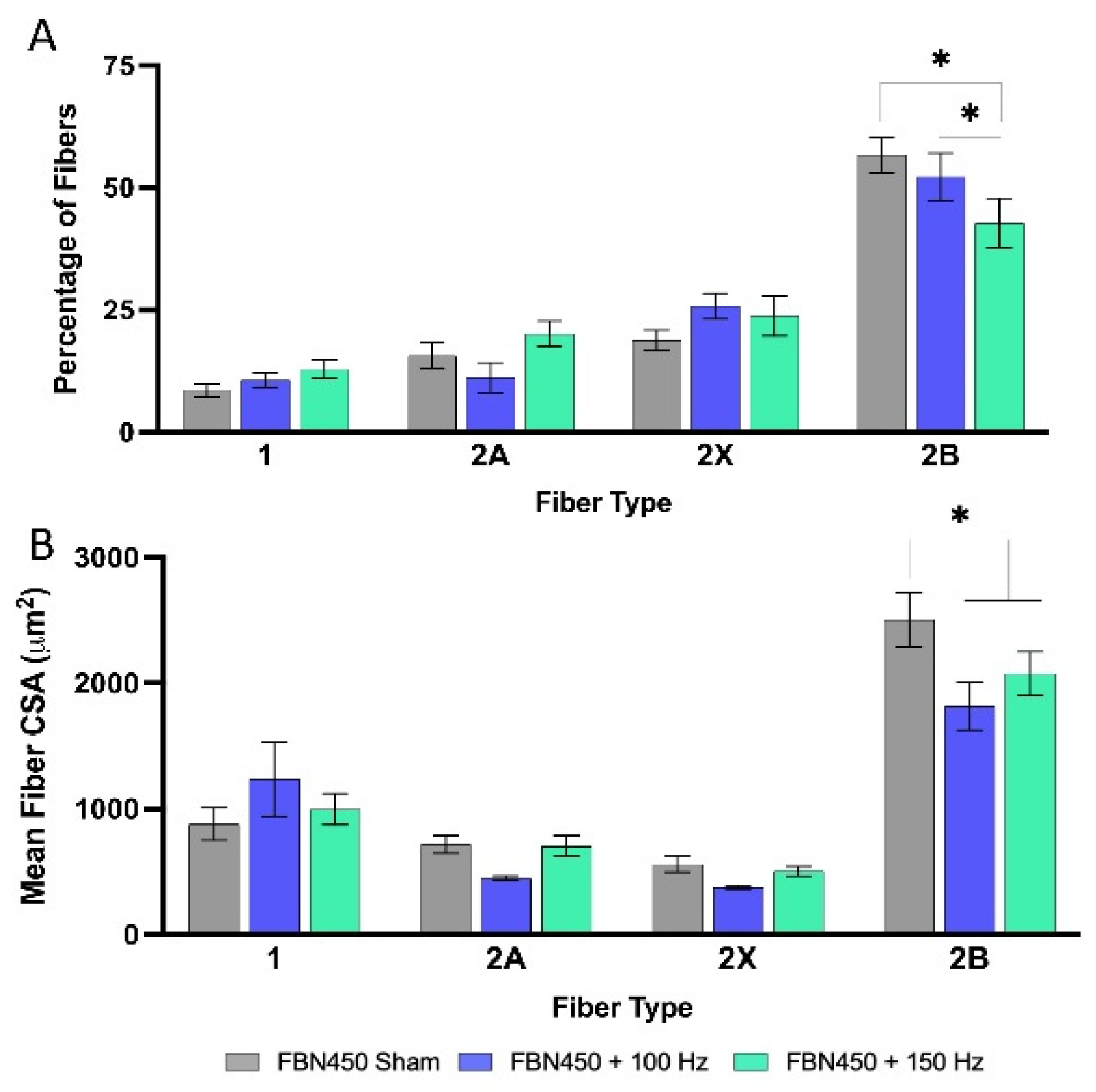
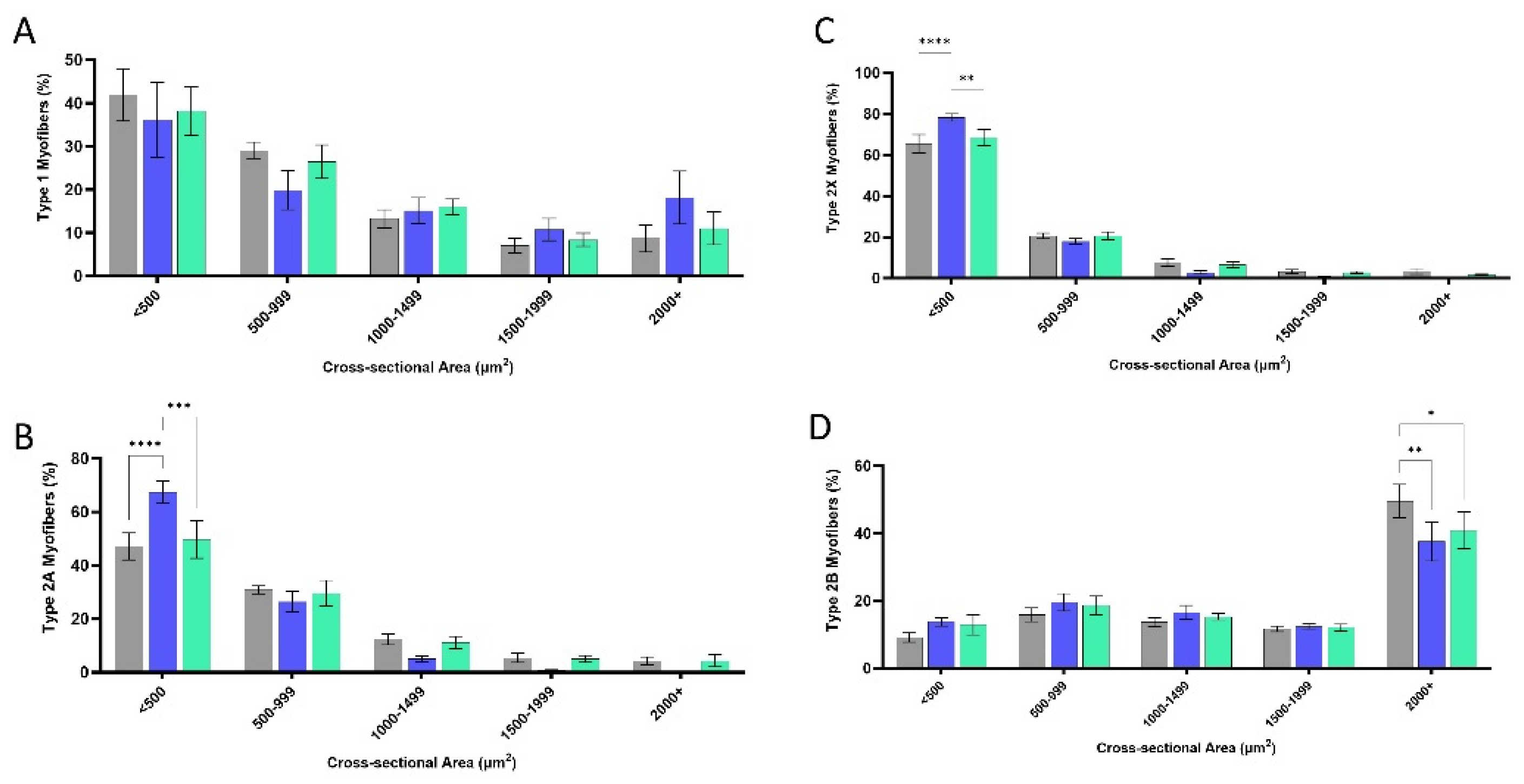
3.3. Myofiber Cross-Sectional Area
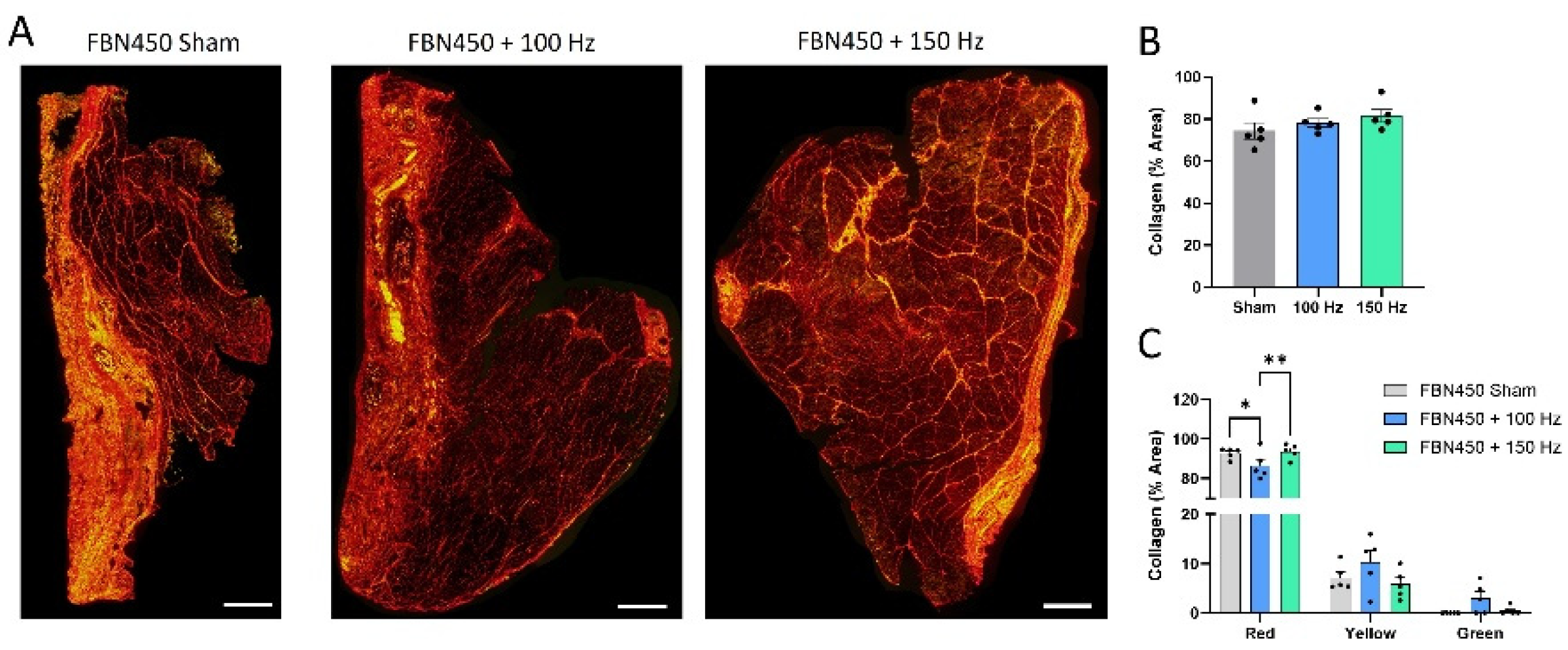
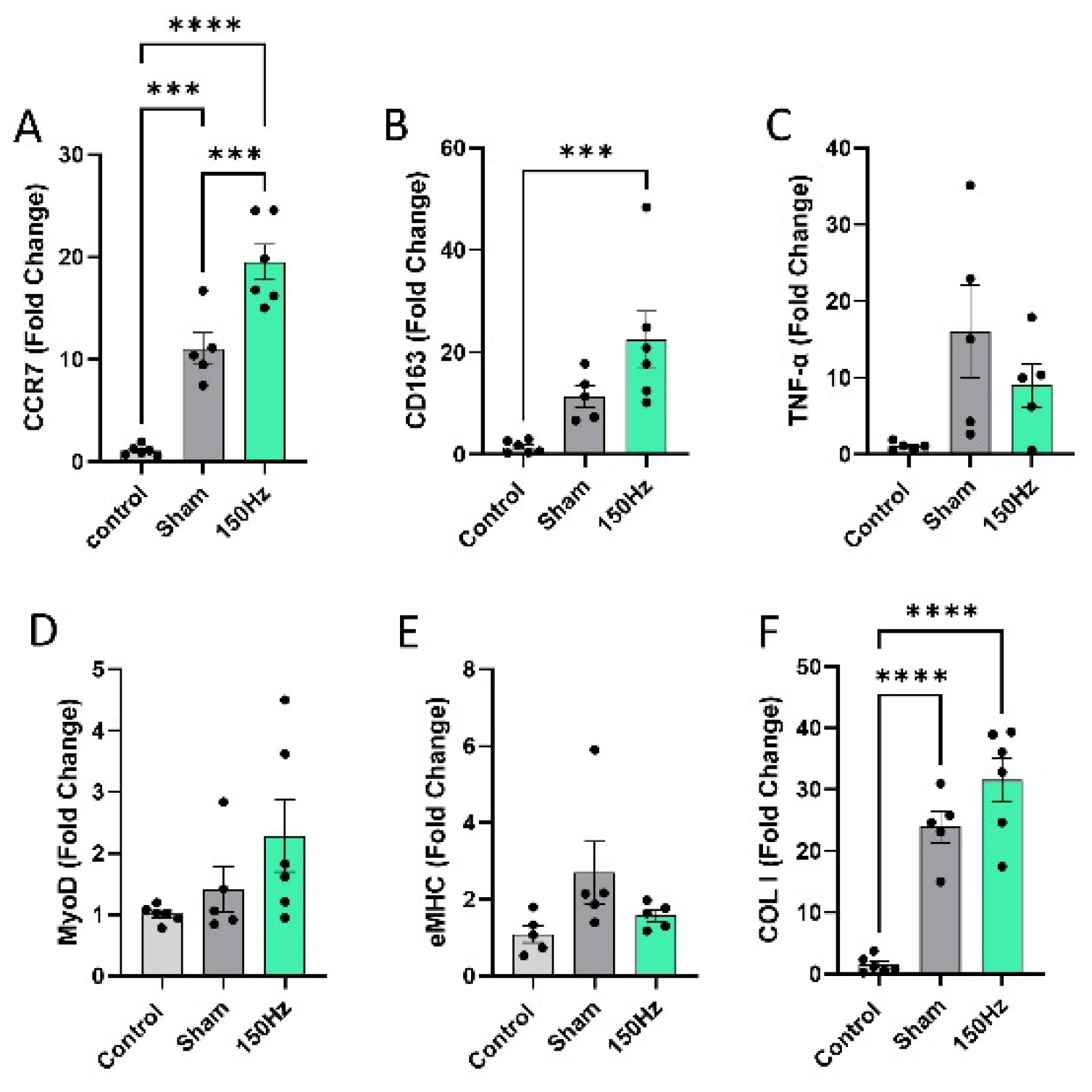
3.4. Collagen Deposition and Gene Expression Analysis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- B.F. Grogan, J.R. Hsu, Volumetric muscle loss, J Am Acad Orthop Surg 19 Suppl 1 (2011) S35-7.
- B.T. Corona, J.C. Rivera, J.C. Owens, J.C. Wenke, C.R. Rathbone, Volumetric Muscle Loss Leads To Permanent Disability Following Extremity Trauma J Rehabil Res Dev In Press (2015).
- Greising, S.M.; Rivera, J.C.; Goldman, S.M.; Watts, A.; Aguilar, C.A.; Corona, B.T. Unwavering Pathobiology of Volumetric Muscle Loss Injury. Sci. Rep. 2017, 7, 13179. [CrossRef]
- Corona, B.T.; Rivera, J.C.; Owens, J.G.; Wenke, J.C.; Rathbone, C.R. Volumetric muscle loss leads to permanent disability following extremity trauma. J. Rehabil. Res. Dev. 2015, 52, 785–792. [CrossRef]
- Ziemkiewicz, N.; Hilliard, G.M.; Dunn, A.J.; Madsen, J.; Haas, G.; Au, J.; Genovese, P.C.; Chauvin, H.M.; West, C.; Paoli, A.; et al. Laminin-111-Enriched Fibrin Hydrogels Enhance Functional Muscle Regeneration Following Trauma. Tissue Eng. Part A 2022, 28, 297–311. [CrossRef]
- Marcinczyk, M.; Dunn, A.; Haas, G.; Madsen, J.; Scheidt, R.; Patel, K.; Talovic, M.; Garg, K. The Effect of Laminin-111 Hydrogels on Muscle Regeneration in a Murine Model of Injury. Tissue Eng. Part A 2019, 25, 1001–1012. [CrossRef]
- Marcinczyk, M.; Elmashhady, H.; Talovic, M.; Dunn, A.; Bugis, F.; Garg, K. Laminin-111 enriched fibrin hydrogels for skeletal muscle regeneration. Biomaterials 2017, 141, 233–242. [CrossRef]
- Johnson, D.; Tobo, C.; Au, J.; Nagarapu, A.; Ziemkiewicz, N.; Chauvin, H.; Robinson, J.; Shringarpure, S.; Tadiwala, J.; Brockhouse, J.; et al. Combined regenerative rehabilitation improves recovery following volumetric muscle loss injury in a rat model. J. Biomed. Mater. Res. Part B: Appl. Biomater. 2024, 112, e35438–e35438. [CrossRef]
- Ambrosio, F.; Wolf, S.L.; Delitto, A.; Fitzgerald, G.K.; Badylak, S.F.; Boninger, M.L.; Russell, A.J. The Emerging Relationship Between Regenerative Medicine and Physical Therapeutics. Phys. Ther. 2010, 90, 1807–1814. [CrossRef]
- Ambrosio, F.; Russell, A. Regenerative rehabilitation: A call to action. J. Rehabilitation Res. Dev. 2010, 47. [CrossRef]
- C. Perez-Terzic, M.K. Childers, Regenerative rehabilitation: a new future?, Am J Phys Med Rehabil 93(11 Suppl 3) (2014) S73-8.
- Rando, T.A.; Ambrosio, F. Regenerative Rehabilitation: Applied Biophysics Meets Stem Cell Therapeutics. Cell Stem Cell 2018, 22, 306–309. [CrossRef]
- Garg, K.; Brockhouse, J.; McAndrew, C.M.; Reiter, A.J.; Owens, J.G.; Mueller, R.J.; Pena, G.; Ridolfo, A.; Johnson, D.L. Regenerative rehabilitation: Navigating the gap between preclinical promises and clinical realities for treating trauma-induced volumetric muscle loss. J. Physiol. 2025, 603, 7421–7439. [CrossRef]
- Greising, S.M.; Dearth, C.L.; Corona, B.T. Regenerative and Rehabilitative Medicine: A Necessary Synergy for Functional Recovery from Volumetric Muscle Loss Injury. Cells Tissues Organs 2016, 202, 237–249. [CrossRef]
- Ziemkiewicz, N.; Au, J.; Chauvin, H.M.; Garg, K. Electrically stimulated eccentric contraction training enhances muscle mass, function, and size following volumetric muscle loss. J. Orthop. Res. 2023, 41, 2588–2598. [CrossRef]
- A Standardized Rat Model of Volumetric Muscle Loss Injury for the Development of Tissue Engineering Therapies, BioResearch Open Access 1(6) (2012) 280-290.
- Vijayan, K.; Thompson, J.L.; Norenberg, K.M.; Fitts, R.H.; Riley, D.A. Fiber-type susceptibility to eccentric contraction-induced damage of hindlimb-unloaded rat AL muscles. J. Appl. Physiol. 2001, 90, 770–776. [CrossRef]
- Hurtgen, B.J.; Henderson, B.E.P.; Ward, C.L.; Goldman, S.M.; Garg, K.; McKinley, T.O.; Greising, S.M.; Wenke, J.C.; Corona, B.T. Impairment of early fracture healing by skeletal muscle trauma is restored by FK506. BMC Musculoskelet. Disord. 2017, 18, 253. [CrossRef]
- Garg, K.; Ward, C.L.; Rathbone, C.R.; Corona, B.T. Transplantation of devitalized muscle scaffolds is insufficient for appreciable de novo muscle fiber regeneration after volumetric muscle loss injury. Cell Tissue Res. 2014, 358, 857–873. [CrossRef]
- Garg, K.; Corona, B.T.; Walters, T.J. Losartan administration reduces fibrosis but hinders functional recovery after volumetric muscle loss injury. J. Appl. Physiol. 2014, 117, 1120–1131. [CrossRef]
- Hurtgen, B.J.; Ward, C.L.; Leopold Wager, C.M.; Garg, K.; Goldman, S.M.; Henderson, B.E.P.; McKinley, T.O.; Greising, S.M.; Wenke, J.C.; Corona, B.T. Autologous minced muscle grafts improve endogenous fracture healing and muscle strength after musculoskeletal trauma. Physiol. Rep. 2017, 5, e13362. [CrossRef]
- Madsen, J.; Haas, G.; Dunn, A.; Ziemkiewicz, N.; Johnson, D.; Garg, K. MyoQuant: An optimized image analysis algorithm for quantitative analysis of skeletal muscle fibers. MethodsX 2025, 15, 103645. [CrossRef]
- Smith, L.R.; Barton, E.R. Collagen content does not alter the passive mechanical properties of fibrotic skeletal muscle in mdx mice. Am. J. Physiol. Physiol. 2014, 306, C889–C898. [CrossRef]
- Schiaffino, S.; Reggiani, C. Fiber Types in Mammalian Skeletal Muscles. Physiol. Rev. 2011, 91, 1447–1531. [CrossRef]
- Badylak, S.F.; Valentin, J.E.; Ravindra, A.K.; McCabe, G.P.; Stewart-Akers, A.M. Macrophage Phenotype as a Determinant of Biologic Scaffold Remodeling. Tissue Eng. Part A 2008, 14, 1835–1842. [CrossRef]
- Tidball, J.G.; Villalta, S.A. Regulatory interactions between muscle and the immune system during muscle regeneration. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1173–R1187. [CrossRef]
- Dziki, J.; Badylak, S.; Yabroudi, M.; Sicari, B.; Ambrosio, F.; Stearns, K.; Turner, N.; Wyse, A.; Boninger, M.L.; Brown, E.H.P.; et al. An acellular biologic scaffold treatment for volumetric muscle loss: results of a 13-patient cohort study. npj Regen. Med. 2016, 1, 16008. [CrossRef]
- Aurora, A.; Roe, J.L.; Corona, B.T.; Walters, T.J. An acellular biologic scaffold does not regenerate appreciable de novo muscle tissue in rat models of volumetric muscle loss injury. Biomaterials 2015, 67, 393–407. [CrossRef]
- Quarta, M.; Cromie, M.; Chacon, R.; Blonigan, J.; Garcia, V.; Akimenko, I.; Hamer, M.; Paine, P.; Stok, M.; Shrager, J.B.; et al. Bioengineered constructs combined with exercise enhance stem cell-mediated treatment of volumetric muscle loss. Nat. Commun. 2017, 8, 15613. [CrossRef]
| Gene | Forward sequence (5’-3’) | Reverse sequence (3’-5’) | Amplicon length (bp) |
| 18s | GGCCCGAAGCGTTTACTT | ACCTCTAGCGGCGCAATAC | 173 |
| CCR7 | GCTCTCCTGGTCATTTTCCA | AAGCACACCGACTCATACAGG | 107 |
| CD163 | TCATTTCGAAGAAGCCCAAG | CTCCGTGTTTCACTTCCACA | 101 |
| TNF-α | ACTCGAGTGACAAGCCCGTA | CCTTGTCCCTTGAAGAGAACC | 184 |
| MyoD | CGTGGCAGTGAGCACTACAG | TGTAGTAGGCGGCGTCGTA | 133 |
| eMHC | TGGAGGACCAAATATGAGACG | CACCATCAAGTCCTCCACCT | 180 |
| COL1 | CTGGTGAACGTGGTGCAG | GACCAATGGGACCAGTCAGA | 123 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




