Submitted:
08 April 2026
Posted:
10 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
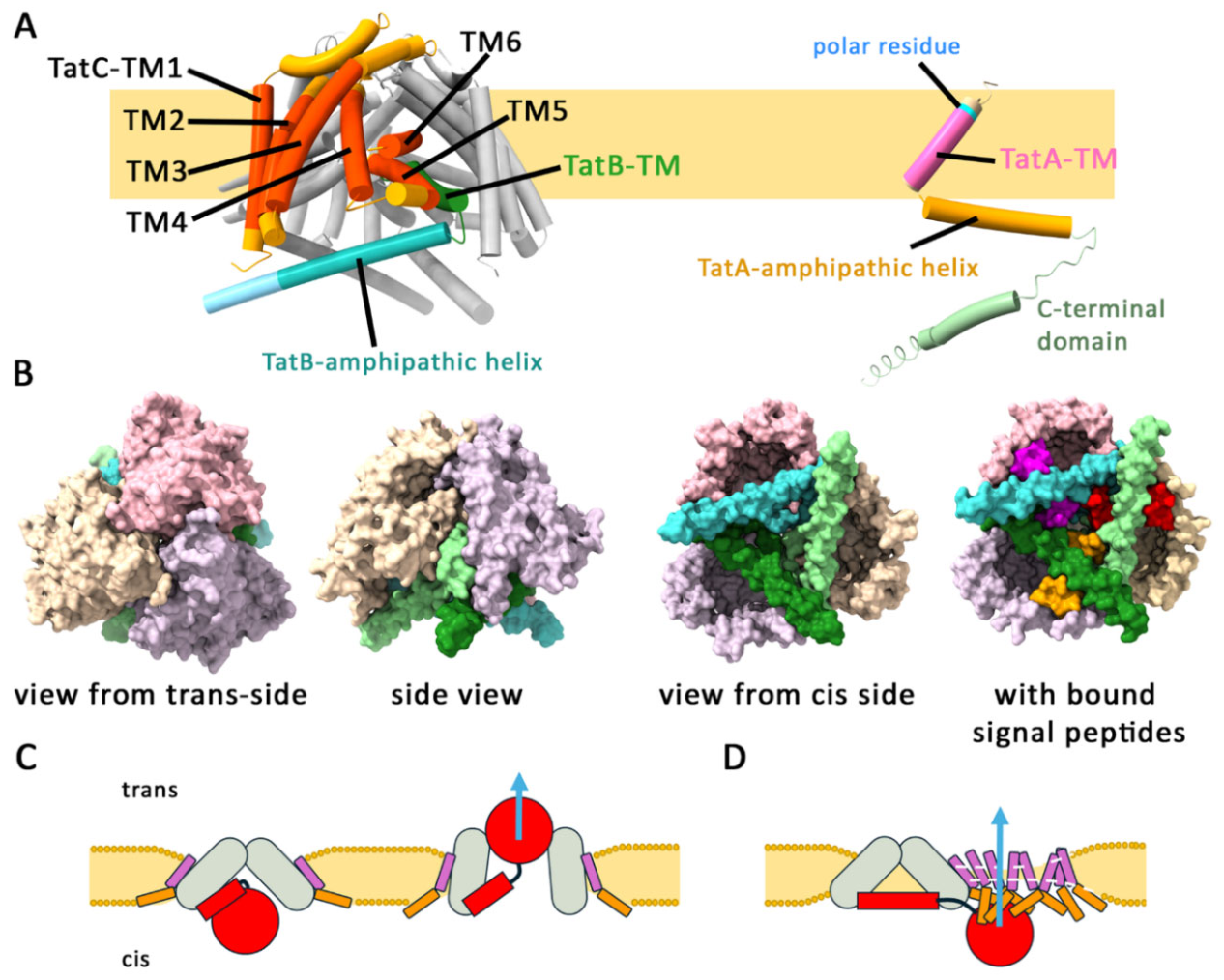
Tat Transport Cannot Occur Through a Central Pore Generated by a TatB3C3 Complex
All Substrate-Binding Sites Can Be Equally Functional
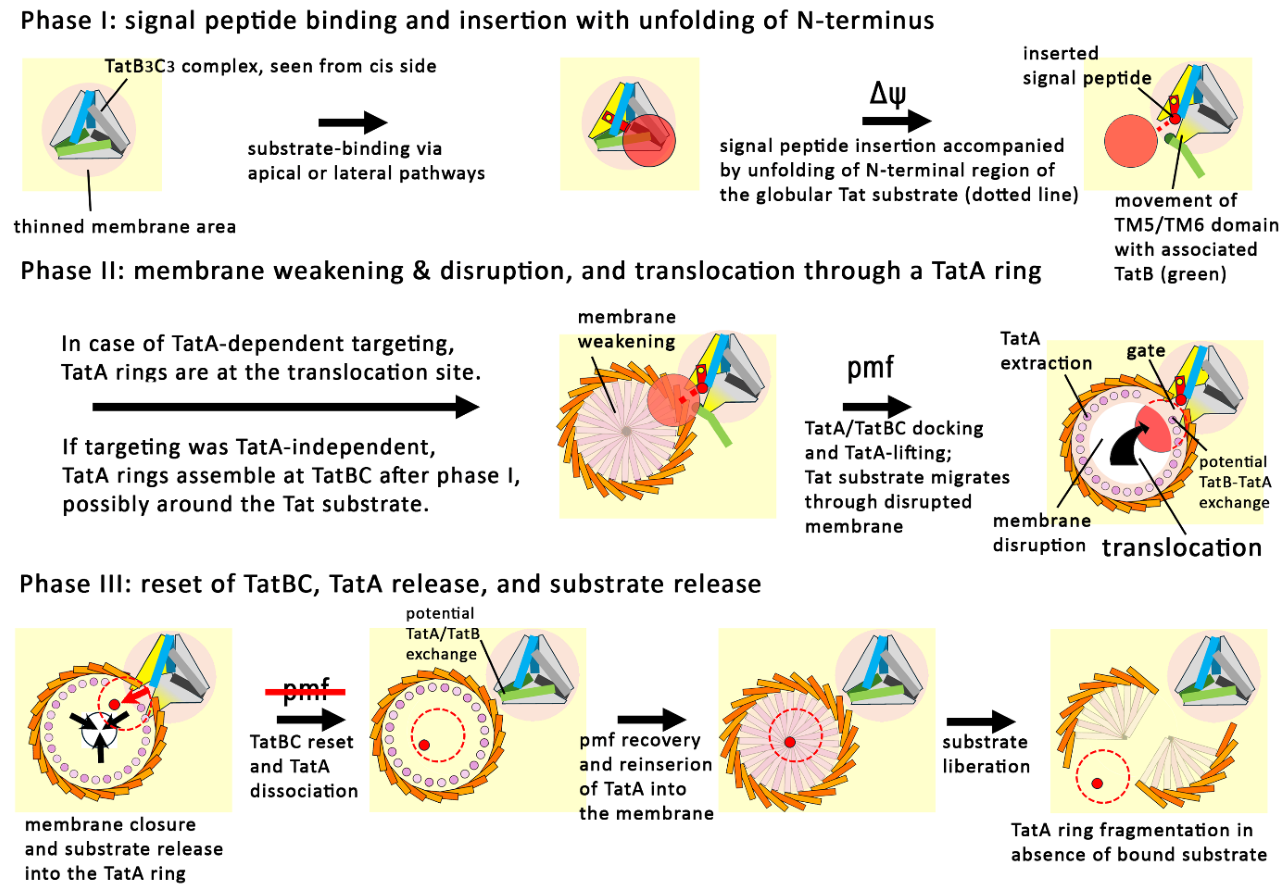
Phase I of Tat Transport: Signal Peptide Insertion by TatBC Complexes
Previously Unanswered Key Questions That Helped to Uncover the TatBC Complex Mechanism
- -
- How can TatC catalyze the insertion of signal peptides without transport of the mature domain, and why does TatB prevent this insertase activity, as observed by Fröbel et al. [33]?
- -
- If this insertase activity is important for the mechanism, how can the signal peptide insertion be catalyzed without liberating the twin-arginine motif from its initial binding site, as observed by Gerard et al. [34]?
- -
- How can signal peptide-binding to TatBC complexes alone already cause the association of TatA clusters, as observed by Dabney-Smith et al. [15]?
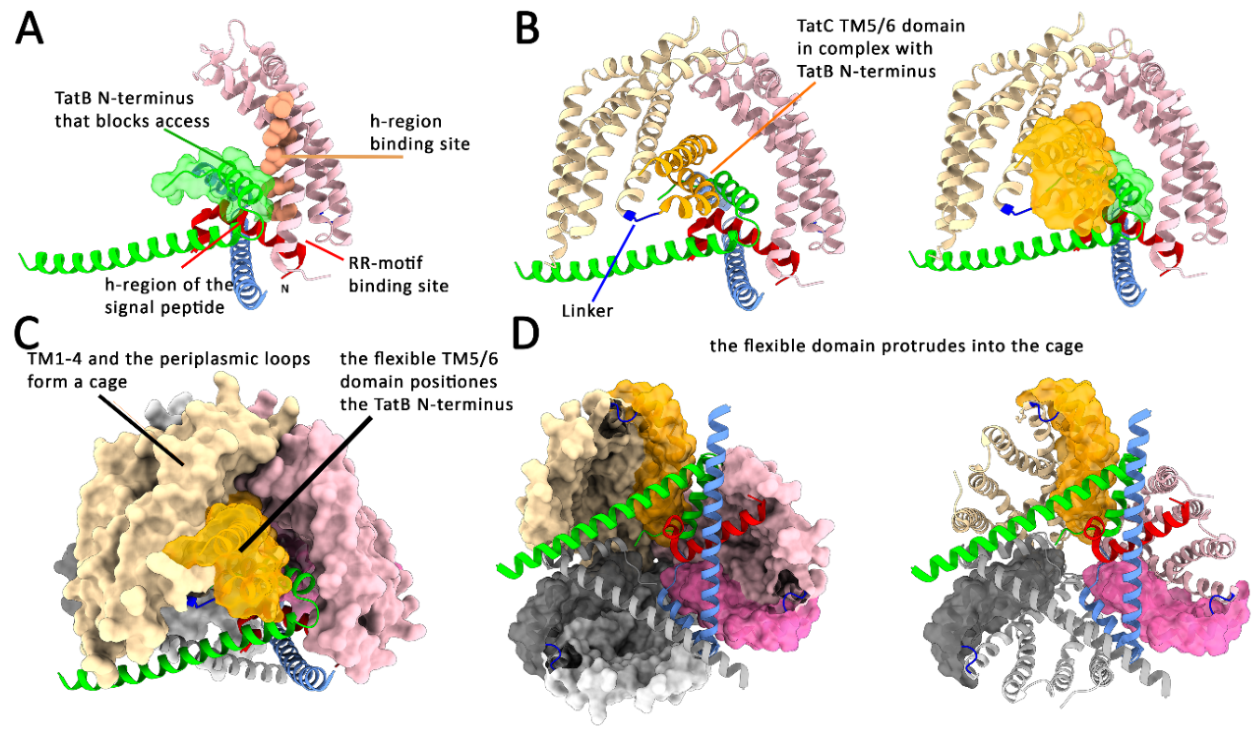
TatB N-Terminal Helices Block Access to the Signal Peptide H-Region-Binding Site in TatC
Biochemical and Structural Evidence for a Signal Peptide-Induced Domain Movement in TatBC
The Domain Movement Requires Membrane Energetization
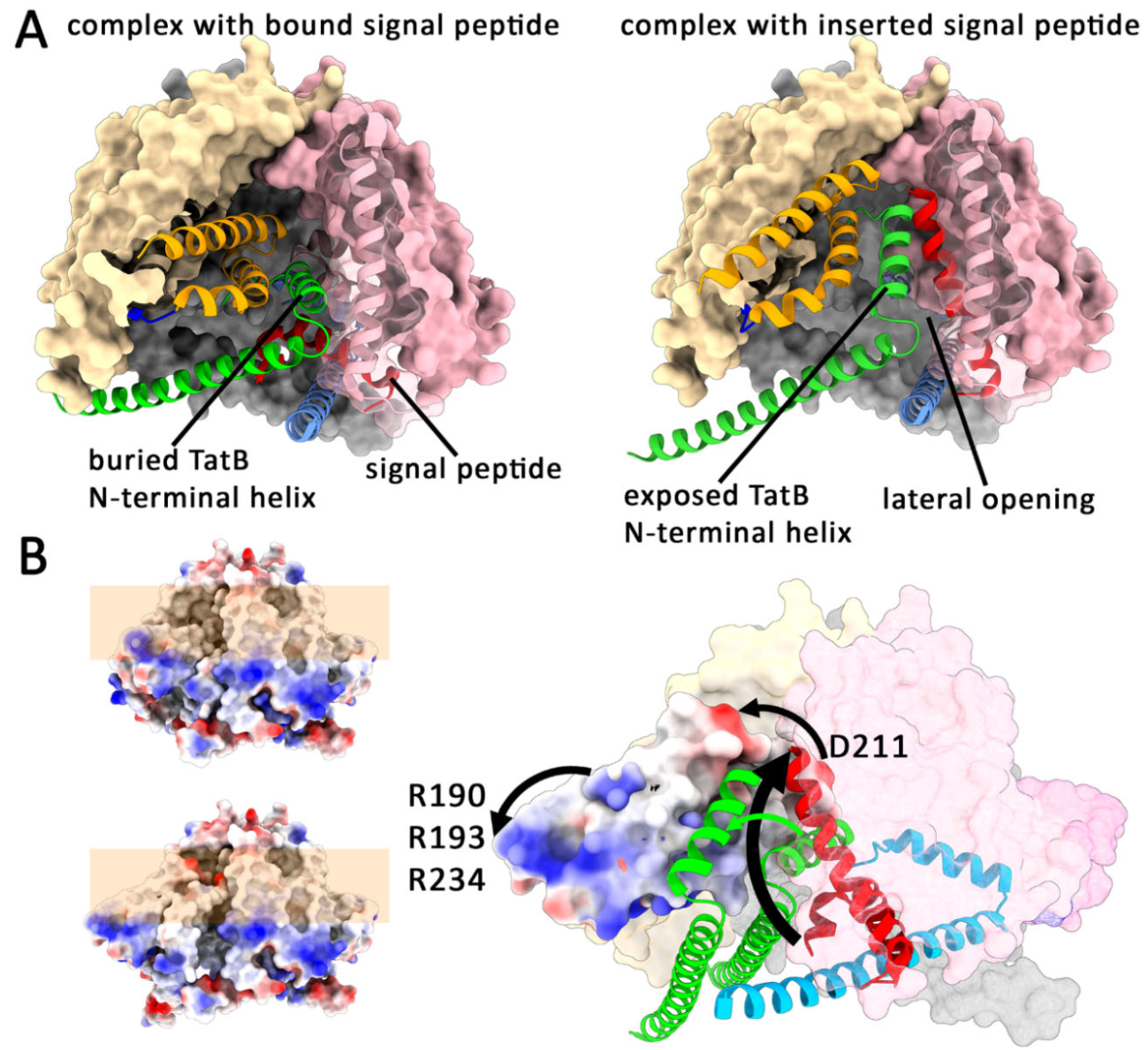
Upon Signal Peptide Insertion, the Globular Domain Relocates to the Translocation Site
Phase II of Tat Transport: Docking of TatA, Membrane-Disruption and Directional Translocation
Previously Unanswered Key Questions that Helped to Understand TatA-Dependent Translocation
- -
- -
- What are the structural characteristics of the ordered cluster of at least 16 TatA protomers that is generated at TatBC upon RR-dependent binding of the signal peptide to TatBC, as observed by Carole Dabney Smith et al. [15]?
- -
- -
- Why does substrate-interaction with E. coli TatA quantitatively protect a specific region of the TatA amphipathic helix in a TatBC-dependent way, as shown by us [21]?
- -
- Why do large TatA assemblies diffuse either slowly, as expected for membrane-integral proteins, or rapidly, as expected for membrane-associated proteins without transmembrane helix, as shown by Yves Bollen et al. [54]?
- -
- What is the energy-demanding step in phase II of Tat transport [46]?
A Simple Pulling of the Folded Protein Through a Disordered TatA Assembly Cannot Explain Tat Transport
TatA Likely Forms a More Defined Structure to Permit Tat Transport
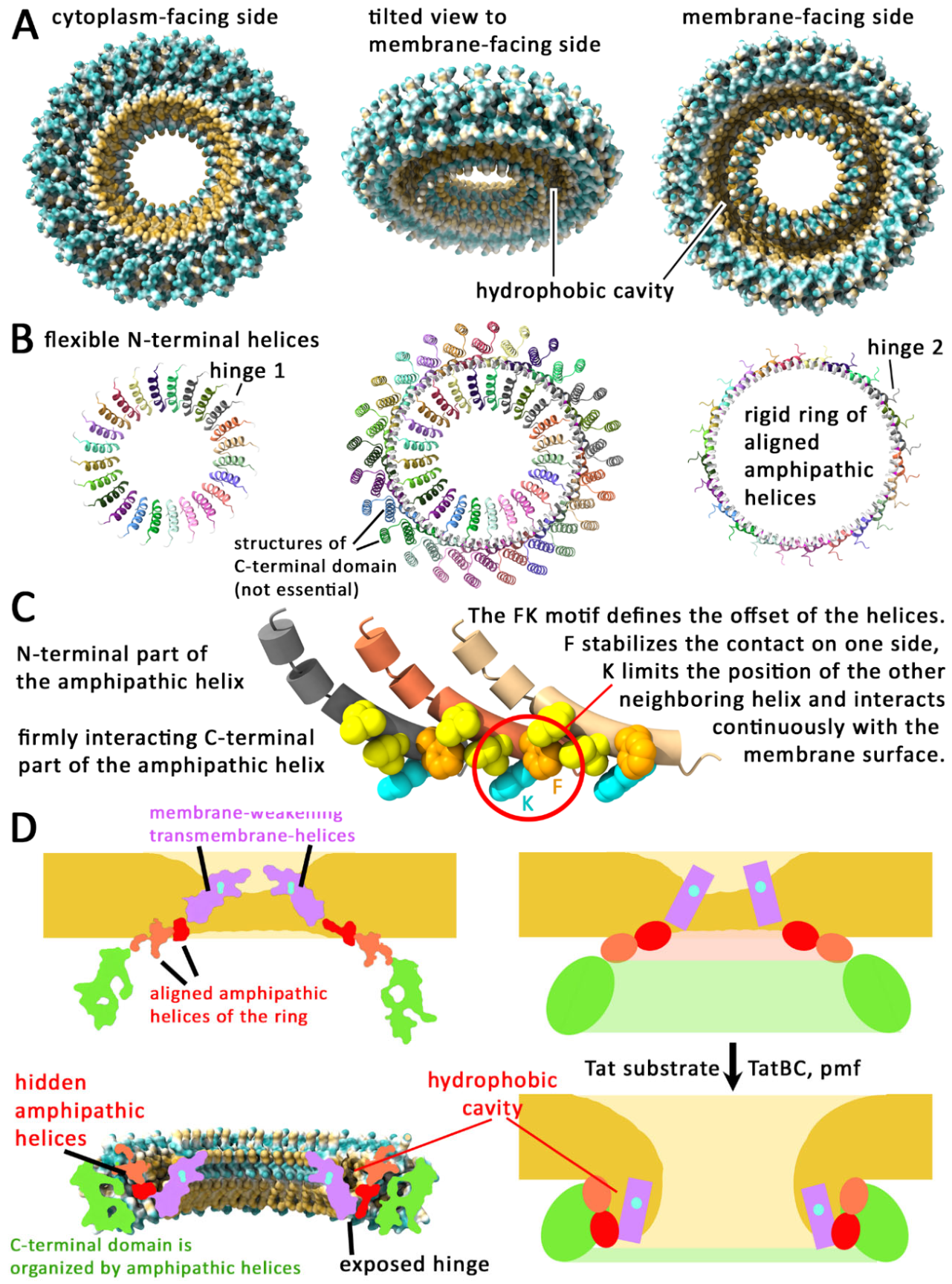
TatA Forms Rings That Could Allow a Controlled TatBC-Dependent Membrane Perforation
AlphaFold 3 Predicts TatA Rings That Explain So Far Not Understood Findings
The Predicted TatA Rings Suggest a Membrane Disruption Mechanism That Is Experimentally Supported
The Membrane Disruption Mechanism Explains the TatBC Dependence and the 2nd Energy Requirement
Experimental Evidence for a Transient Lifting of TatA Rings onto the Membrane Surface
Priming of TatA Rings by Bound Substrate, and TatBC-Dependent Membrane Disruption by TatA
Phase III: Closing of the Catalytic Cycle and Resetting of the System
Release of the Signal Peptide into the Membrane, and Reset of the TatBC Complex
Reset of TatA Rings to Membrane-Inserted Clusters That Can Interact with Tat Substrates
The Tat System Requires a Functional Asymmetry
Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Natale, P; Brüser, T; Driessen, AJM. Sec- and Tat-mediated protein secretion across the bacterial cytoplasmic membrane - distinct translocases and mechanisms. Biochim Biophys Acta 2008, 1778(9), 1735–1756. [Google Scholar] [CrossRef]
- Yen, M-R; Tseng, Y-H; Nguyen, EH; Wu, L-F; Saier, MH. Sequence and phylogenetic analyses of the twin-arginine targeting (Tat) protein export system. Arch Microbiol 2002, 177(6), 441–450. [Google Scholar] [CrossRef] [PubMed]
- Lee, PA; Buchanan, G; Stanley, NR; Berks, BC; Palmer, T. Truncation analysis of TatA and TatB defines the minimal functional units required for protein translocation. J Bacteriol 2002, 184(21), 5871–5879. [Google Scholar] [CrossRef] [PubMed]
- Warren, G; Oates, J; Robinson, C; Dixon, AM. Contributions of the transmembrane domain and a key acidic motif to assembly and function of the TatA complex. J Mol Biol 2009, 388(1), 122–132. [Google Scholar] [CrossRef] [PubMed]
- Barnett, JP; Eijlander, RT; Kuipers, OP; Robinson, C. A minimal Tat system from a gram-positive organism: a bifunctional TatA subunit participates in discrete TatAC and TatA complexes. J Biol Chem 2008, 283(5), 2534–2542. [Google Scholar] [CrossRef]
- Jongbloed, JD; van der Ploeg, R; van Dijl, JM. Bifunctional TatA subunits in minimal Tat protein translocases. Trends Microbiol 2006, 14(1), 2–4. [Google Scholar] [CrossRef]
- Jongbloed, JDH; Grieger, U; Antelmann, H; Hecker, M; Nijland, R; Bron, S; van Dijl, JM. Two minimal Tat translocases in Bacillus. Mol Microbiol 2004, 54(5), 1319–1325. [Google Scholar] [CrossRef]
- Zhao, Z; Sazanov, LA. Structure of E. coli Twin-arginine translocase (Tat) complex with bound cargo. BioRxiv 2025. [Google Scholar] [CrossRef]
- Deme, JC; Oj, Bryant; Batista, M; Stansfeld, PJ; Berks, BC; Lea, SM. Structure and substrate recognition by the Twin-arginine translocation (Tat) pathway core complex. BioRxiv 2025. [Google Scholar] [CrossRef]
- Alami, M; Lüke, I; Deitermann, S; Eisner, G; Koch, HG; Brunner, J; Müller, M. Differential interactions between a twin-arginine signal peptide and its translocase in Escherichia coli. Mol Cell 2003, 12(4), 937–946. [Google Scholar] [CrossRef]
- Kreutzenbeck, P; Kroger, C; Lausberg, F; Blaudeck, N; Sprenger, GA; Freudl, R. Escherichia coli twin arginine (Tat) mutant translocases possessing relaxed signal peptide recognition specificities. J Biol Chem 2007, 282(11), 7903–7911. [Google Scholar] [CrossRef]
- Lausberg, F; Fleckenstein, S; Kreutzenbeck, P; Frobel, J; Rose, P; Muller, M; Freudl, R. Genetic evidence for a tight cooperation of TatB and TatC during productive recognition of twin-arginine (Tat) signal peptides in Escherichia coli. PLoS ONE 2012, 7(6), e39867. [Google Scholar] [CrossRef]
- Ma, X; Cline, K. Mapping the signal peptide binding and oligomer contact sites of the core subunit of the pea twin arginine protein translocase. Plant Cell 2013, 25(3), 999–1015. [Google Scholar] [CrossRef]
- Strauch, EM; Georgiou, G. Escherichia coli tatC mutations that suppress defective twin-arginine transporter signal peptides. J Mol Biol 2007, 374(2), 283–291. [Google Scholar] [CrossRef]
- Dabney-Smith, C; Cline, K. Clustering of C-terminal stromal domains of Tha4 homo-oligomers during translocation by the Tat protein transport system. Mol Biol Cell 2009. [Google Scholar] [CrossRef] [PubMed]
- Lüke, I; Handford, JI; Palmer, T; Sargent, F. Proteolytic processing of Escherichia coli twin-arginine signal peptides by LepB. Arch Microbiol 2009, 191(12), 919–925. [Google Scholar] [CrossRef]
- Hatzixanthis, K; Palmer, T; Sargent, F. A subset of bacterial inner membrane proteins integrated by the twin-arginine translocase. Mol Microbiol 2003, 49(5), 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Goosens, VJ; Monteferrante, CG; van Dijl, JM. Co-factor insertion and disulfide bond requirements for twin-arginine translocase-dependent export of the Bacillus subtilis Rieske protein QcrA. J Biol Chem 2014, 289(19), 13124–13131. [Google Scholar] [CrossRef]
- Hopkins, A; Buchanan, G; Palmer, T. Role of the twin arginine protein transport pathway in the assembly of the Streptomyces coelicolor cytochrome bc1 complex. J Bacteriol 2014, 196(1), 50–59. [Google Scholar] [CrossRef] [PubMed]
- Brüser, T; Sanders, C. An alternative model of the twin arginine translocation system. Microbiol Res 2003, 158(1), 7–17. [Google Scholar] [CrossRef] [PubMed]
- Hou, B; Heidrich, ES; Mehner-Breitfeld, D; Brüser, T. The TatA component of the twin-arginine translocation system locally weakens the cytoplasmic membrane of Escherichia coli upon protein substrate binding. J Biol Chem 2018, 293(20), 7592–7605. [Google Scholar] [CrossRef] [PubMed]
- Asher, AH; Theg, SM. Electrochromic shift supports the membrane destabilization model of Tat-mediated transport and shows ion leakage during Sec transport. Proc Natl Acad Sci U S A 2021, 118((12). [Google Scholar] [CrossRef]
- McNeilage, R; Ganesan, I; Keilman, J; Theg, SM. Cell-penetrating peptides stimulate protein transport on the Twin-arginine translocation pathway: evidence for a membrane thinning and toroidal pore mechanism. BioRxiv 2023, 07.08.548235. [Google Scholar] [CrossRef]
- Rodriguez, F; Rouse, SL; Tait, CE; Harmer, J; de, Riso A; Timmel, CR; Sansom, MS; Berks, BC; Schnell, JR. Structural model for the protein-translocating element of the twin-arginine transport system. Proc Natl Acad Sci U S A 2013, 110(12), E1092-101. [Google Scholar] [CrossRef]
- Mehner-Breitfeld, D; Ringel, MT; Tichy, DA; Endter, LJ; Stroh, KS; Lünsdorf, H; Risselada, HJ; Brüser, T. TatA and TatB generate a hydrophobic mismatch important for the function and assembly of the Tat translocon in Escherichia coli. J Biol Chem 2022, 298, 102236. [Google Scholar] [CrossRef]
- Bolhuis, A; Mathers, JE; Thomas, JD; Barrett, CM; Robinson, C. TatB and TatC form a functional and structural unit of the twin-arginine translocase from Escherichia coli. J Biol Chem 2001, 276(23), 20213–20219. [Google Scholar] [CrossRef]
- Behrendt, J; Brüser, T. The TatBC complex of the Tat protein translocase in Escherichia coli and its transition to the substrate-bound TatABC complex. Biochemistry 2014, 53(14), 2344–2354. [Google Scholar] [CrossRef] [PubMed]
- Aldridge, C; Ma, X; Gerard, F; Cline, K. Substrate-gated docking of pore subunit Tha4 in the TatC cavity initiates Tat translocase assembly. J Cell Biol 2014, 205(1), 51–65. [Google Scholar] [CrossRef] [PubMed]
- Habersetzer, J; Moore, K; Cherry, J; Buchanan, G; Stansfeld, PJ; Palmer, T. Substrate-triggered position switching of TatA and TatB during Tat transport in Escherichia coli. Open Biol 2017, 7((8). [Google Scholar] [CrossRef]
- Werner, M-H; Mehner-Breitfeld, D; Brüser, T. A larger TatBC complex associates with TatA clusters for transport of folded proteins across the bacterial cytoplasmic membrane. Sci Rep 2024, 14(1), 13754. [Google Scholar] [CrossRef]
- Barrett, CM; Mangels, D; Robinson, C. Mutations in subunits of the Escherichia coli twin-arginine translocase block function via differing effects on translocation activity or Tat complex structure. J Mol Biol 2005, 347(2), 453–463. [Google Scholar] [CrossRef] [PubMed]
- Celedon, JM; Cline, K. Stoichiometry for binding and transport by the twin arginine translocation system. J Cell Biol 2012, 197(4), 523–534. [Google Scholar] [CrossRef] [PubMed]
- Fröbel, J; Rose, P; Lausberg, F; Blümmel, A-S; Freudl, R; Müller, M. Transmembrane insertion of twin-arginine signal peptides is driven by TatC and regulated by TatB. Nat Commun 2012, 3, 1311. [Google Scholar] [CrossRef]
- Gerard, F; Cline, K. Efficient twin arginine translocation (Tat) Pathway transport of a precursor protein covalently anchored to its initial cpTatC binding site. J Biol Chem 2006, 281(10), 6130–6135. [Google Scholar] [CrossRef]
- Bageshwar, UK; Whitaker, N; Liang, FC; Musser, SM. Interconvertibility of lipid- and translocon-bound forms of the bacterial Tat precursor pre-SufI. Mol Microbiol 2009, 74(1), 209–226. [Google Scholar] [CrossRef]
- Ramasamy, S; Abrol, R; Suloway, CJM; Clemons, WM. The glove-like structure of the conserved membrane protein TatC provides insight into signal sequence recognition in twin-arginine translocation. Structure 2013, 21(5), 777–788. [Google Scholar] [CrossRef]
- Taubert, J; Hou, B; Risselada, HJ; Mehner, D; Lünsdorf, H; Grubmüller, H; Brüser, T. TatBC-independent TatA/Tat substrate interactions contribute to transport efficiency. PLoS ONE 2015, 10(3), e0119761. [Google Scholar] [CrossRef]
- Rathmann, C; Schlösser, AS; Schiller, J; Bogdanov, M; Brüser, T. Tat transport in Escherichia coli requires zwitterionic phosphatidylethanolamine but no specific negatively charged phospholipid. FEBS Lett 2017, 591(18), 2848–2858. [Google Scholar] [CrossRef] [PubMed]
- Fröbel, J; Rose, P; Müller, M. Early contacts between substrate proteins and TatA translocase component in twin-arginine translocation. J Biol Chem 2011, 286(51), 43679–43689. [Google Scholar] [CrossRef]
- Blaudeck, N; Kreutzenbeck, P; Müller, M; Sprenger, GA; Freudl, R. Isolation and characterization of bifunctional Escherichia coli TatA mutant proteins that allow efficient Tat-dependent protein translocation in the absence of TatB. J Biol Chem 2005, 280(5), 3426–3432. [Google Scholar] [CrossRef] [PubMed]
- Taubert, J; Brüser, T. Twin-arginine translocation-arresting protein regions contact TatA and TatB. Biol Chem 2014, 395(7-8), 827–836. [Google Scholar] [CrossRef] [PubMed]
- Klösgen, RB; Brock, IW; Herrmann, RG; Robinson, C. Proton gradient-driven import of the 16 kDa oxygen-evolving complex protein as the full precursor protein by isolated thylakoids. Plant Mol Biol 1992, 18(5), 1031–1034. [Google Scholar] [CrossRef]
- Cline, K; Ettinger, WF; Theg, SM. Protein-specific energy requirements for protein transport across or into thylakoid membranes. Two lumenal proteins are transported in the absence of ATP. J Biol Chem 1992, 267(4), 2688–2696. [Google Scholar] [CrossRef]
- Braun, NA; Davis, AW; Theg, SM. The chloroplast Tat pathway utilizes the transmembrane electric potential as an energy source. Biophys J 2007, 93(6), 1993–1998. [Google Scholar] [CrossRef]
- Theg, SM; Cline, K; Finazzi, G; Wollman, FA. The energetics of the chloroplast Tat protein transport pathway revisited. Trends Plant Sci 2005, 10(4), 153–154. [Google Scholar] [CrossRef]
- Bageshwar, UK; Musser, SM. Two electrical potential-dependent steps are required for transport by the Escherichia coli Tat machinery. J Cell Biol 2007, 179(1), 87–99. [Google Scholar] [CrossRef]
- Buchanan, G; Leeuw, E; Stanley, NR; Wexler, M; Berks, BC; Sargent, F; Palmer, T. Functional complexity of the twin-arginine translocase TatC component revealed by site-directed mutagenesis. Mol Microbiol 2002, 43(6), 1457–1470. [Google Scholar] [CrossRef]
- Maurer, C; Panahandeh, S; Jungkamp, A-C; Moser, M; Müller, M. TatB Functions as an Oligomeric Binding Site for Folded Tat Precursor Proteins. Mol Biol Cell 2010, 21, 4151–4161. [Google Scholar] [CrossRef] [PubMed]
- Berghöfer, J; Klösgen, RB. Two distinct translocation intermediates can be distinguished during protein transport by the TAT (Dph) pathway across the thylakoid membrane. FEBS Lett 1999, 460(2), 328–332. [Google Scholar] [CrossRef] [PubMed]
- Gerard, F; Cline, K. The thylakoid proton gradient promotes an advanced stage of signal peptide binding deep within the Tat pathway receptor complex. J Biol Chem 2007. [Google Scholar] [CrossRef] [PubMed]
- Lindenstrauss, U; Brüser, T. Tat transport of linker-containing proteins in Escherichia coli. FEMS Microbiol Lett 2009, 295(1), 135–140. [Google Scholar] [CrossRef]
- Cline, K; McCaffery, M. Evidence for a dynamic and transient pathway through the TAT protein transport machinery. EMBO J. 2007, 26(13), 3039–3049. [Google Scholar] [CrossRef]
- Aldridge, C; Storm, A; Cline, K; Dabney-Smith, C. The chloroplast twin arginine transport (Tat) component, Tha4, undergoes conformational changes leading to Tat protein transport. J Biol Chem 2012, 287(41), 34752–34763. [Google Scholar] [CrossRef]
- Varadarajan, A; Oswald, F; Lill, H; Peterman, EJ; Bollen, YJM. Rapid diffusion of large TatA complexes detected using single particle tracking microscopy. BioRxiv 2020, 2020.05.14.095463. [Google Scholar] [CrossRef]
- Hao, B; Zhou, W; Theg, SM. Hydrophobic mismatch is a key factor in protein transport across lipid bilayer membranes via the Tat pathway. J Biol Chem 2022, 298, 101991. [Google Scholar] [CrossRef] [PubMed]
- Fan, E; Jakob, M; Klösgen, RB. One signal is enough: Stepwise transport of two distinct passenger proteins by the Tat pathway across the thylakoid membrane. Biochem Biophys Res Commun 2010, 398(3), 438–443. [Google Scholar] [CrossRef]
- Dabney-Smith, C; Mori, H; Cline, K. Oligomers of Tha4 organize at the thylakoid Tat translocase during protein transport. J Biol Chem 2006, 281(9), 5476–5483. [Google Scholar] [CrossRef]
- Gohlke, U; Pullan, L; McDevitt, CA; Porcelli, I; Leeuw, E; de; Palmer, T; Saibil, HR; Berks, BC. The TatA component of the twin-arginine protein transport system forms channel complexes of variable diameter. Proc Natl Acad Sci U S A 2005, 102(30), 10482–10486. [Google Scholar] [CrossRef] [PubMed]
- Beck, D; Vasisht, N; Baglieri, J; Monteferrante, CG; van Dijl, JM; Robinson, C; Smith, CJ. Ultrastructural characterisation of Bacillus subtilis TatA complexes suggests they are too small to form homooligomeric translocation pores. Biochim Biophys Acta 2013, 1833(8), 1811–1819. [Google Scholar] [CrossRef]
- Berthelmann, F; Mehner, D; Richter, S; Lindenstrauss, U; Lünsdorf, H; Hause, G; Brüser, T. Recombinant expression of tatABC and tatAC results in the formation of interacting cytoplasmic TatA tubes in Escherichia coli. J Biol Chem 2008, 283(37), 25281–25289. [Google Scholar] [CrossRef] [PubMed]
- Jormakka, M; Törnroth, S; Byrne, B; Iwata, S. Molecular basis of proton motive force generation: structure of formate dehydrogenase-N. Science 2002, 295(5561), 1863–1868. [Google Scholar] [CrossRef]
- Abramson, J; Adler, J; Dunger, J; Evans, R; Green, T; Pritzel, A; Ronneberger, O; Willmore, L; Ballard, AJ; Bambrick, J; Bodenstein, SW; Evans, DA; Hung, C-C; O'Neill, M; Reiman, D; Tunyasuvunakool, K; Wu, Z; Žemgulytė, A; Arvaniti, E; Beattie, C; Bertolli, O; Bridgland, A; Cherepanov, A; Congreve, M; Cowen-Rivers, AI; Cowie, A; Figurnov, M; Fuchs, FB; Gladman, H; Jain, R; et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 2024, 630(8016), 493–500. [Google Scholar] [CrossRef]
- Lange, C; Müller, SD; Walther, TH; Bürck, J; Ulrich, AS. Structure analysis of the protein translocating channel TatA in membranes using a multi-construct approach. Biochim Biophys Acta 2007, 1768(10), 2627–2634. [Google Scholar] [CrossRef]
- Zhang, Y; Hu, Y; Li, H; Jin, C. Structural basis for TatA oligomerization: an NMR study of Escherichia coli TatA dimeric structure. PLoS ONE 2014, 9(8), e103157. [Google Scholar] [CrossRef]
- Hou, B; Brüser, T. The Tat-dependent protein translocation pathway. Biomol Concepts 2011, 2(6), 507–523. [Google Scholar] [CrossRef]
- Hicks, MG; de, Leeuw E; Porcelli, I; Buchanan, G; Berks, BC; Palmer, T. The Escherichia coli twin-arginine translocase: conserved residues of TatA and TatB family components involved in protein transport. FEBS Lett 2003, 539(1-3), 61–67. [Google Scholar] [CrossRef]
- Walther, TH; Grage, SL; Roth, N; Ulrich, AS. Membrane alignment of the pore-forming component TatA(d) of the twin-arginine translocase from Bacillus subtilis resolved by solid-state NMR spectroscopy. J Am Chem Soc 2010, 132(45), 15945–15956. [Google Scholar] [CrossRef] [PubMed]
- Stockwald, ER; Steger, LME; Vollmer, S; Gottselig, C; Grage, SL; Bürck, J; Afonin, S; Fröbel, J; Blümmel, A-S; Setzler, J; Wenzel, W; Walther, TH; Ulrich, AS. Length matters: Functional flip of the short TatA transmembrane helix. Biophys J 2022, 122(11), 2125–2146. [Google Scholar] [CrossRef] [PubMed]
- Richter, S; Lindenstrauss, U; Lücke, C; Bayliss, R; Brüser, T. Functional Tat transport of unstructured, small, hydrophilic proteins. J Biol Chem 2007, 282(46), 33257–33264. [Google Scholar] [CrossRef] [PubMed]
- Alder, NN; Theg, SM. Energetics of protein transport across biological membranes. a study of the thylakoid ΔpH-dependent/cpTat pathway. Cell 2003, 112(2), 231–242. [Google Scholar] [CrossRef] [PubMed]
- Mehner, D; Osadnik, H; Lunsdorf, H; Brüser, T. The Tat system for membrane translocation of folded proteins recruits the membrane-stabilizing Psp machinery in Escherichia coli. J Biol Chem 2012, 287(33), 27834–27842. [Google Scholar] [CrossRef]
- Bernal-Cabas, M; Miethke, M; Antelo-Varela, M; Aguilar Suárez, R; Neef, J; Schön, L; Gabarrini, G; Otto, A; Becher, D; Wolf, D; van Dijl, JM. Functional association of the stress-responsive LiaH protein and the minimal TatAyCy protein translocase in Bacillus subtilis. Biochim Biophys Acta 2020, 1867(8), 118719. [Google Scholar] [CrossRef] [PubMed]
- Fincher, V; McCaffery, M; Cline, K. Evidence for a loop mechanism of protein transport by the thylakoid ΔpH pathway. FEBS Lett 1998, 423(1), 66–70. [Google Scholar] [CrossRef]
- Hou, B; Frielingsdorf, S; Klösgen, RB. Unassisted membrane insertion as the initial step in ΔpH/Tat-dependent protein transport. J Mol Biol 2006, 355(5), 957–967. [Google Scholar] [CrossRef]
- Richter, S; Brüser, T. Targeting of unfolded PhoA to the TAT translocon of Escherichia coli. J Biol Chem 2005, 280(52), 42723–42730. [Google Scholar] [CrossRef]
- Di Cola, A; Robinson, C. Large-scale translocation reversal within the thylakoid Tat system in vivo. J Cell Biol 2005, 171(2), 281–289. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, N; Bageshwar, UK; Musser, SM. Kinetics of precursor interactions with the bacterial Tat translocase detected by real-time FRET. J Biol Chem 2012, 287(14), 11252–11260. [Google Scholar] [CrossRef] [PubMed]
- Alcock, F; Baker, MAB; Greene, NP; Palmer, T; Wallace, MI; Berks, BC. Live cell imaging shows reversible assembly of the TatA component of the twin-arginine protein transport system. Proc Natl Acad Sci U S A 2013, 110(38), E3650-9. [Google Scholar] [CrossRef] [PubMed]
- Shanmugham, A; Wong Fong Sang, H. W.; Bollen, YJ; Lill, H. Membrane binding of twin arginine preproteins as an early step in translocation. Biochemistry 2006, 45(7), 2243–2249. [Google Scholar] [CrossRef]
- Pop, OI; Westermann, M; Volkmer-Engert, R; Schulz, D; Lemke, C; Schreiber, S; Gerlach, R; Wetzker, R; Müller, JP. Sequence-specific binding of prePhoD to soluble TatAd indicates protein-mediated targeting of the Tat export in Bacillus subtilis. J Biol Chem 2003, 278(40), 38428–38436. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S; Stengel, R; Westermann, M; Volkmer-Engert, R; Pop, OI; Müller, JP. Affinity of TatCd for TatAd elucidates its receptor function in the Bacillus subtilis Tat translocase system. J Biol Chem 2006, 281(29), 19977–19984. [Google Scholar] [CrossRef]
- de, Keersmaeker S; van Mellaert, L; Lammertyn, E; Vrancken, K; Anne, J; Geukens, N. Functional analysis of TatA and TatB in Streptomyces lividans. Biochem Biophys Res Commun 2005, 335(3), 973–982. [Google Scholar] [CrossRef]
- Dilks, K; Gimenez, MI; Pohlschröder, M. Genetic and biochemical analysis of the twin-arginine translocation pathway in halophilic archaea. J Bacteriol 2005, 187(23), 8104–8113. [Google Scholar] [CrossRef]
- Frielingsdorf, S; Jakob, M; Klösgen, RB. A stromal pool of TatA promotes Tat-dependent protein transport across the thylakoid membrane. J Biol Chem 2008, 283(49), 33838–33845. [Google Scholar] [CrossRef]
- Pettersson, P; Patrick, J; Jakob, M; Jacobs, M; Klösgen, RB; Wennmalm, S; Mäler, L. Soluble TatA forms oligomers that interact with membranes: Structure and insertion studies of a versatile protein transporter. Biochim Biophys Acta 2021, 1863(2), 183529. [Google Scholar] [CrossRef]
- 86. Keersmaeker S de; van Mellaert, L; Schaerlaekens, K; van Dessel, W; Vrancken, K; Lammertyn, E; Anne, J; Geukens, N. Structural organization of the twin-arginine translocation system in Streptomyces lividans. FEBS Lett 2005, 579(3), 797–802. [Google Scholar] [CrossRef]
- Thomas, JR; Bolhuis, A. The tatC gene cluster is essential for viability in halophilic archaea. FEMS Microbiol Lett 2006, 256(1), 44–49. [Google Scholar] [CrossRef] [PubMed]
- Goddard, TD; Huang, CC; Meng, EC; Pettersen, EF; Couch, GS; Morris, JH; Ferrin, TE. UCSF ChimeraX: Meeting modern challenges in visualization and analysis. Protein Sci 2018, 27(1), 14–25. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



