Figure 1.
Histamine induces melanin production in melanocytes. (A) Cell viability was assessed over 72 hours following treatment with increasing concentrations of histamine (0-100 μM) in B16F10 cells. Cell density was measured by optical density (OD) and normalized to the value of day 0. (B) Representative images showing B16F10 pellets treated with histamine (0-30 μM) for 48 hours. (C) Quantification of melanin content normalized to total protein in B16F10 cells. Data are expressed as fold change relative to untreated control (0 μM). (D) Representative images of NHEM pellets showing melanin accumulation at indicated time points (days 0, 2, 4, 6, and 8) in the absence (0 μM) or presence (10 μM) of histamine. (E) Quantification of melanin content normalized to total protein over the 8-day culture period in NHEMs. Data are expressed as fold change relative to control at day 0. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett’s post-hoc test, was used for multiple comparisons. *p < 0.05, ***p < 0.001, ns: not significant.
Figure 1.
Histamine induces melanin production in melanocytes. (A) Cell viability was assessed over 72 hours following treatment with increasing concentrations of histamine (0-100 μM) in B16F10 cells. Cell density was measured by optical density (OD) and normalized to the value of day 0. (B) Representative images showing B16F10 pellets treated with histamine (0-30 μM) for 48 hours. (C) Quantification of melanin content normalized to total protein in B16F10 cells. Data are expressed as fold change relative to untreated control (0 μM). (D) Representative images of NHEM pellets showing melanin accumulation at indicated time points (days 0, 2, 4, 6, and 8) in the absence (0 μM) or presence (10 μM) of histamine. (E) Quantification of melanin content normalized to total protein over the 8-day culture period in NHEMs. Data are expressed as fold change relative to control at day 0. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett’s post-hoc test, was used for multiple comparisons. *p < 0.05, ***p < 0.001, ns: not significant.
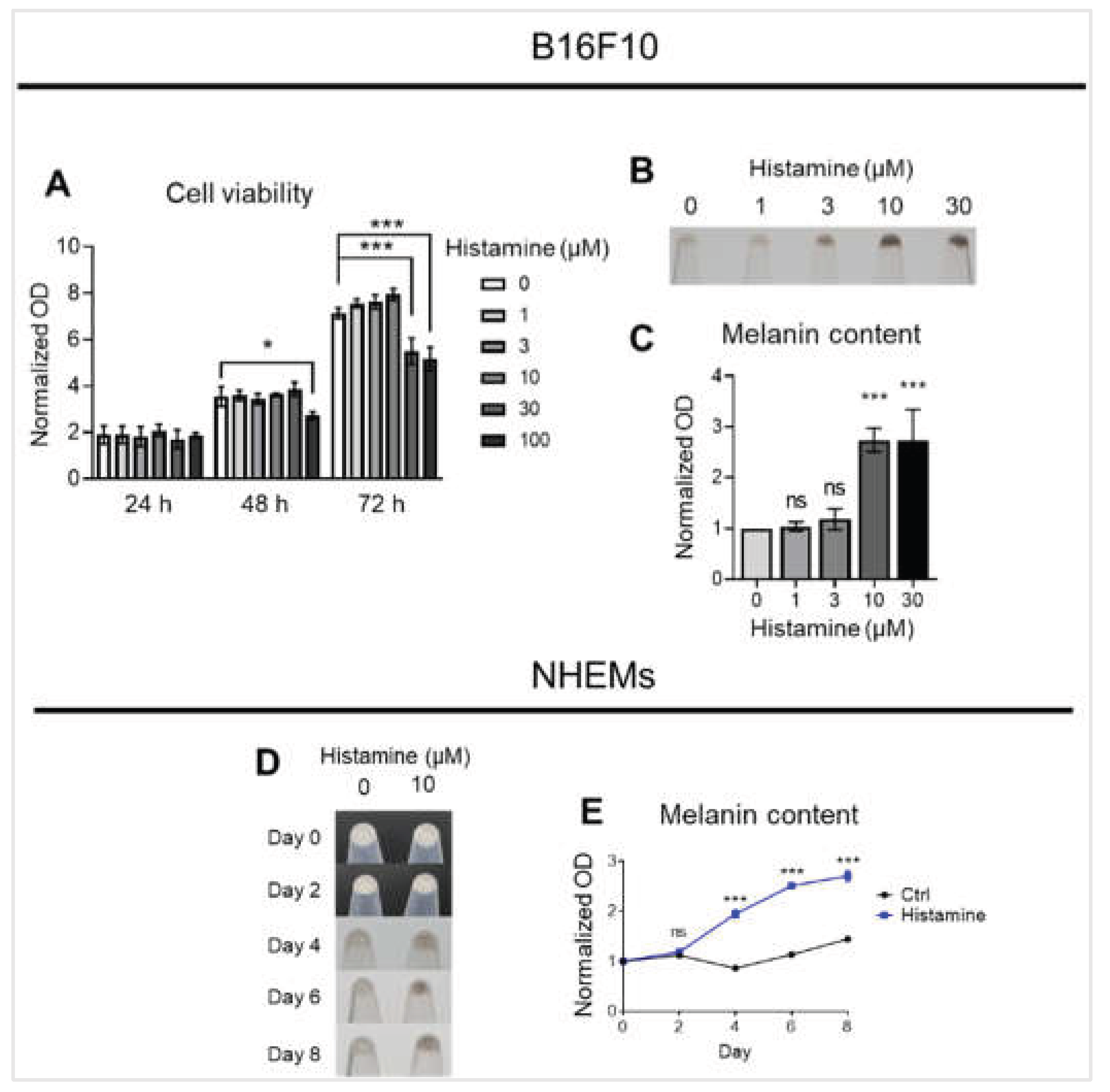
Figure 2.
Histamine induces melanogenesis in melanocytes through histamine H2 receptors. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), or histamine in combination with selective histamine receptor antagonists: pyrilamine (H1 antagonist, 10 μM), famotidine (H2 antagonist, 10 μM), or thioperamide (H3 antagonist, 10 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), or histamine in combination with pyrilamine (10 μM), famotidine (10 μM), or thioperamide (10 μM) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. *p < 0.05, **p < 0.01, ***p < 0.001, ns: not significant.
Figure 2.
Histamine induces melanogenesis in melanocytes through histamine H2 receptors. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), or histamine in combination with selective histamine receptor antagonists: pyrilamine (H1 antagonist, 10 μM), famotidine (H2 antagonist, 10 μM), or thioperamide (H3 antagonist, 10 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), or histamine in combination with pyrilamine (10 μM), famotidine (10 μM), or thioperamide (10 μM) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. *p < 0.05, **p < 0.01, ***p < 0.001, ns: not significant.
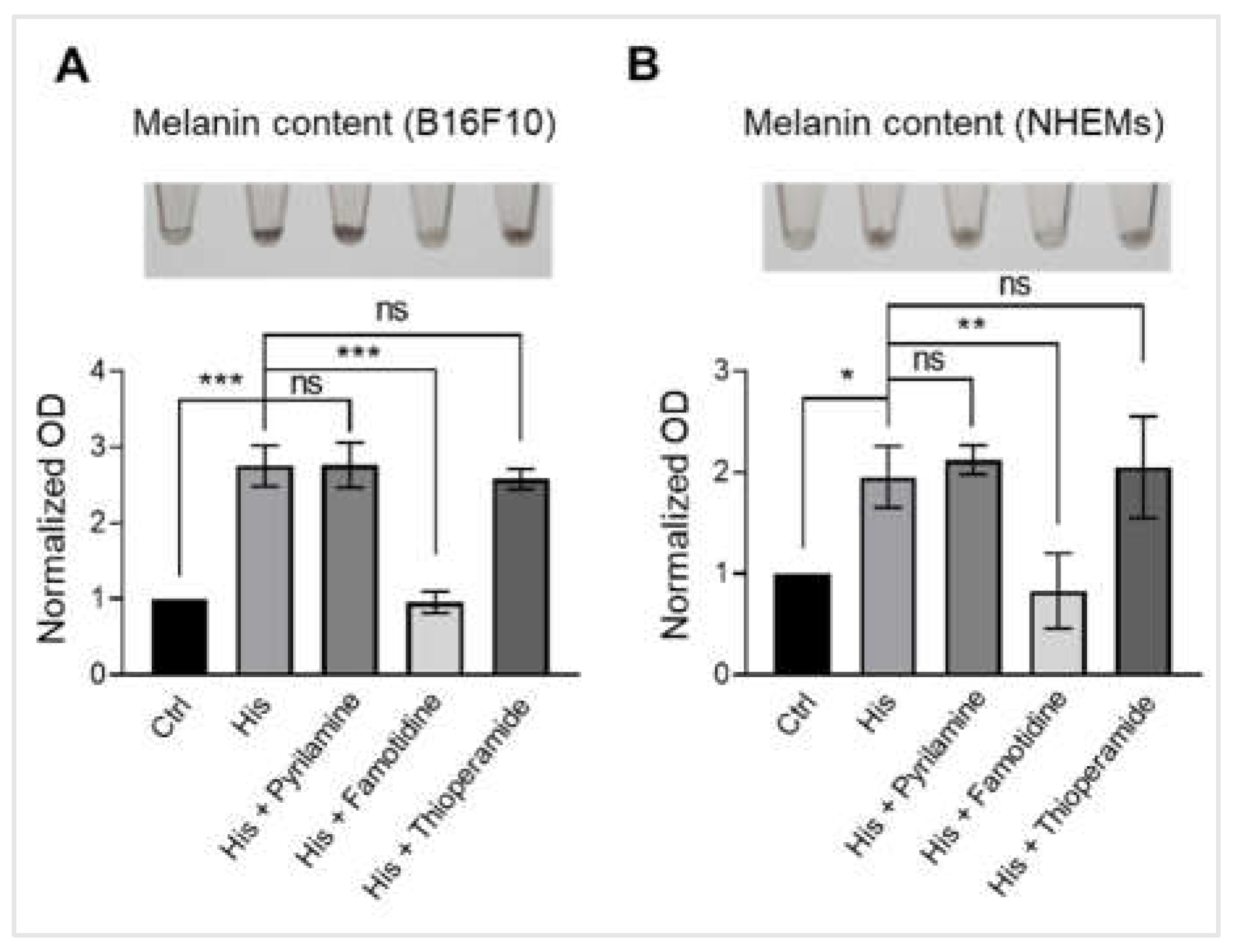
Figure 3.
Histamine enhances store-operated calcium entry (SOCE) in melanocytes. (A) Representative trace (left) and averaged peak value (right) of cytosolic Ca²⁺ levels in response to increasing concentrations of histamine (1, 3, 10, 30, and 100 μM) in the presence of extracellular calcium (1.3 mM Ca²⁺) in B16F10 cells (n = 24 cells). (B) Representative trace (left) and averaged peak value (right) of cytosolic Ca²⁺ levels in response to increasing concentrations of histamine (1, 3, 10, 30, and 100 μM) in the presence of extracellular calcium (1.3 mM Ca²⁺) in NHEMs (n = 24 cells). (C) Representative traces (left) show SOCE in B16F10 cells pretreated with vehicle control (Ctrl, black trace, n = 29 cells) or histamine (His, 10 μM, blue trace, n = 16 cells) for 48 hours. Thapsigargin (Tg, 1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺) to deplete intracellular Ca²⁺ stores, followed by the presence of extracellular calcium (1.3 mM Ca²⁺) to measure SOCE. Quantitative SOCE amplitude (ΔF340/F380) (right) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. (D) Representative traces show SOCE in NHEMs pretreated with vehicle control (Ctrl, left, n = 19 cells) or histamine (His, 10 μM, n = 18 cells) (middle) for 6 days. Tg (1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺), followed by the presence of extracellular calcium (1.3 mM Ca²⁺). Quantitative SOCE amplitude (ΔF340/F380) (right) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. Data are presented as mean ± SD. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. Student's t-test was used for comparison between two groups. **p< 0.01, ***p < 0.001, ns: not significant.
Figure 3.
Histamine enhances store-operated calcium entry (SOCE) in melanocytes. (A) Representative trace (left) and averaged peak value (right) of cytosolic Ca²⁺ levels in response to increasing concentrations of histamine (1, 3, 10, 30, and 100 μM) in the presence of extracellular calcium (1.3 mM Ca²⁺) in B16F10 cells (n = 24 cells). (B) Representative trace (left) and averaged peak value (right) of cytosolic Ca²⁺ levels in response to increasing concentrations of histamine (1, 3, 10, 30, and 100 μM) in the presence of extracellular calcium (1.3 mM Ca²⁺) in NHEMs (n = 24 cells). (C) Representative traces (left) show SOCE in B16F10 cells pretreated with vehicle control (Ctrl, black trace, n = 29 cells) or histamine (His, 10 μM, blue trace, n = 16 cells) for 48 hours. Thapsigargin (Tg, 1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺) to deplete intracellular Ca²⁺ stores, followed by the presence of extracellular calcium (1.3 mM Ca²⁺) to measure SOCE. Quantitative SOCE amplitude (ΔF340/F380) (right) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. (D) Representative traces show SOCE in NHEMs pretreated with vehicle control (Ctrl, left, n = 19 cells) or histamine (His, 10 μM, n = 18 cells) (middle) for 6 days. Tg (1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺), followed by the presence of extracellular calcium (1.3 mM Ca²⁺). Quantitative SOCE amplitude (ΔF340/F380) (right) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. Data are presented as mean ± SD. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. Student's t-test was used for comparison between two groups. **p< 0.01, ***p < 0.001, ns: not significant.
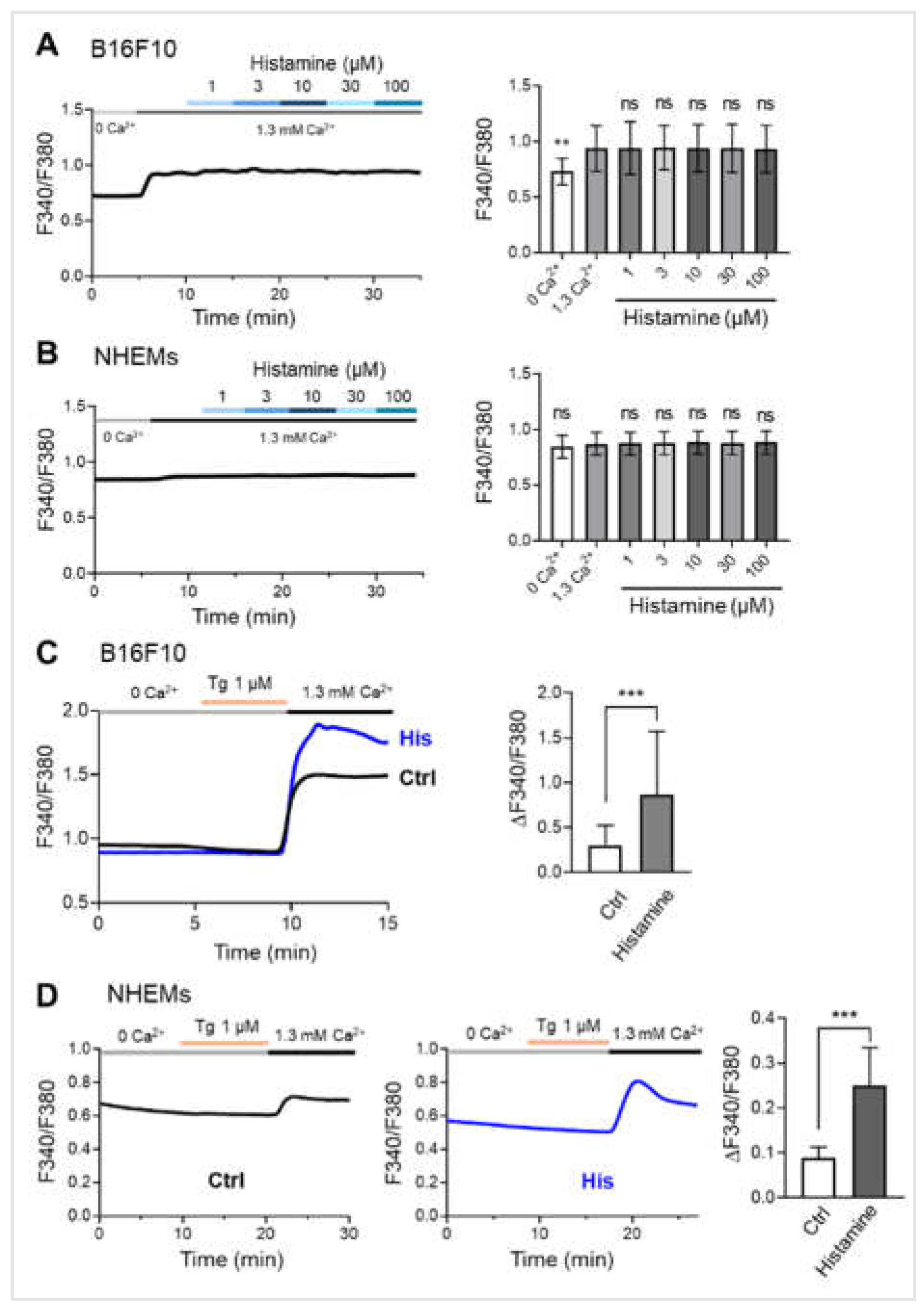
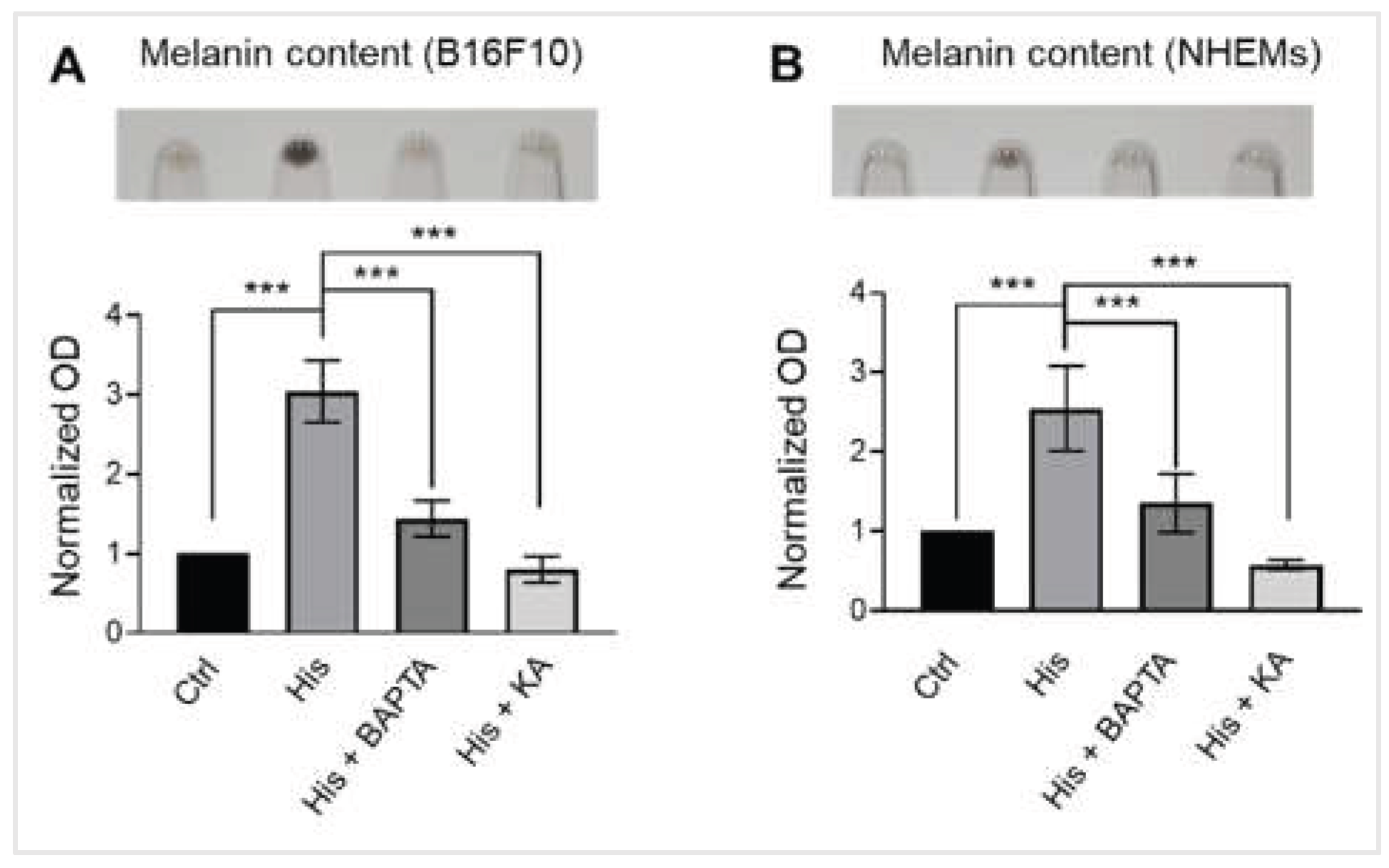
Figure 4.
Intracellular calcium is required for histamine-induced melanogenesis. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BAPTA-AM (intracellular calcium chelator, 10 μM) or Kojic acid (tyrosynase inhibitor, 50 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BAPTA-AM (10 μM), or KA (50 μM) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001.
Figure 4.
Intracellular calcium is required for histamine-induced melanogenesis. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BAPTA-AM (intracellular calcium chelator, 10 μM) or Kojic acid (tyrosynase inhibitor, 50 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BAPTA-AM (10 μM), or KA (50 μM) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001.
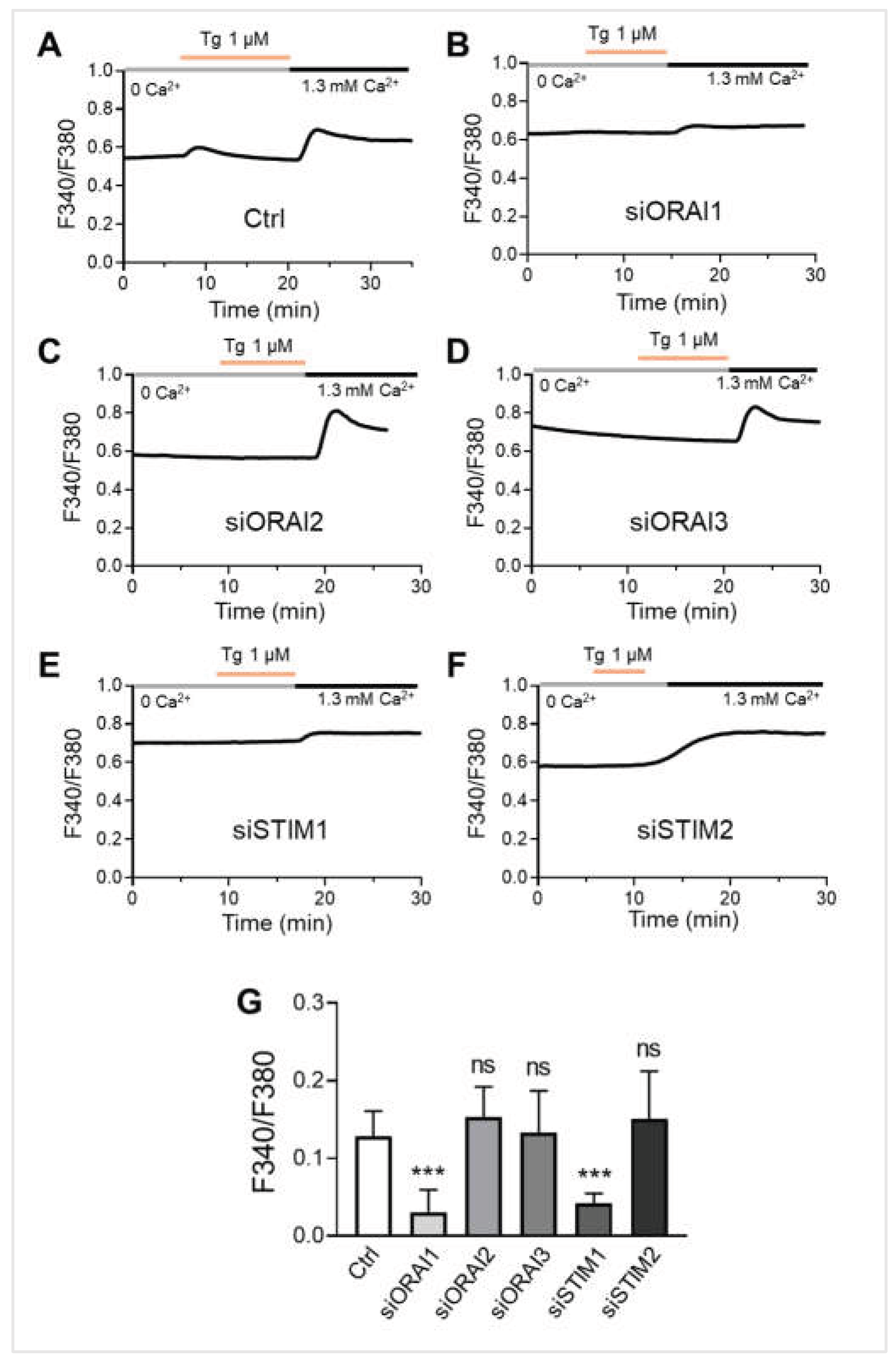
Figure 5.
ORAI1 and STIM1 are the essential molecular components of store-operated calcium entry (SOCE) in melanocytes. (A-F) Representative traces showing SOCE in NHEMs. Cells were transfected with siRNAs targeting ORAI1 (siORAI1, n = 36), ORAI2 (siORAI2, n = 21), ORAI3 (siORAI3, n = 27), STIM1 (siSTIM1, n = 37), or STIM2 (siSTIM2, n = 24) for 6 days. Thapsigargin (Tg, 1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺) to deplete intracellular Ca²⁺ stores, followed by the presence of extracellular calcium (1.3 mM Ca²⁺) to measure SOCE. (G) Quantitative SOCE amplitude (ΔF340/F380) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. Data are presented as mean ± SD. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001, ns: not significant.
Figure 5.
ORAI1 and STIM1 are the essential molecular components of store-operated calcium entry (SOCE) in melanocytes. (A-F) Representative traces showing SOCE in NHEMs. Cells were transfected with siRNAs targeting ORAI1 (siORAI1, n = 36), ORAI2 (siORAI2, n = 21), ORAI3 (siORAI3, n = 27), STIM1 (siSTIM1, n = 37), or STIM2 (siSTIM2, n = 24) for 6 days. Thapsigargin (Tg, 1 μM) was applied in the absence of extracellular calcium (0 Ca²⁺) to deplete intracellular Ca²⁺ stores, followed by the presence of extracellular calcium (1.3 mM Ca²⁺) to measure SOCE. (G) Quantitative SOCE amplitude (ΔF340/F380) shows the change in cytosolic calcium level before and after the presence of extracellular calcium. Data are presented as mean ± SD. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001, ns: not significant.
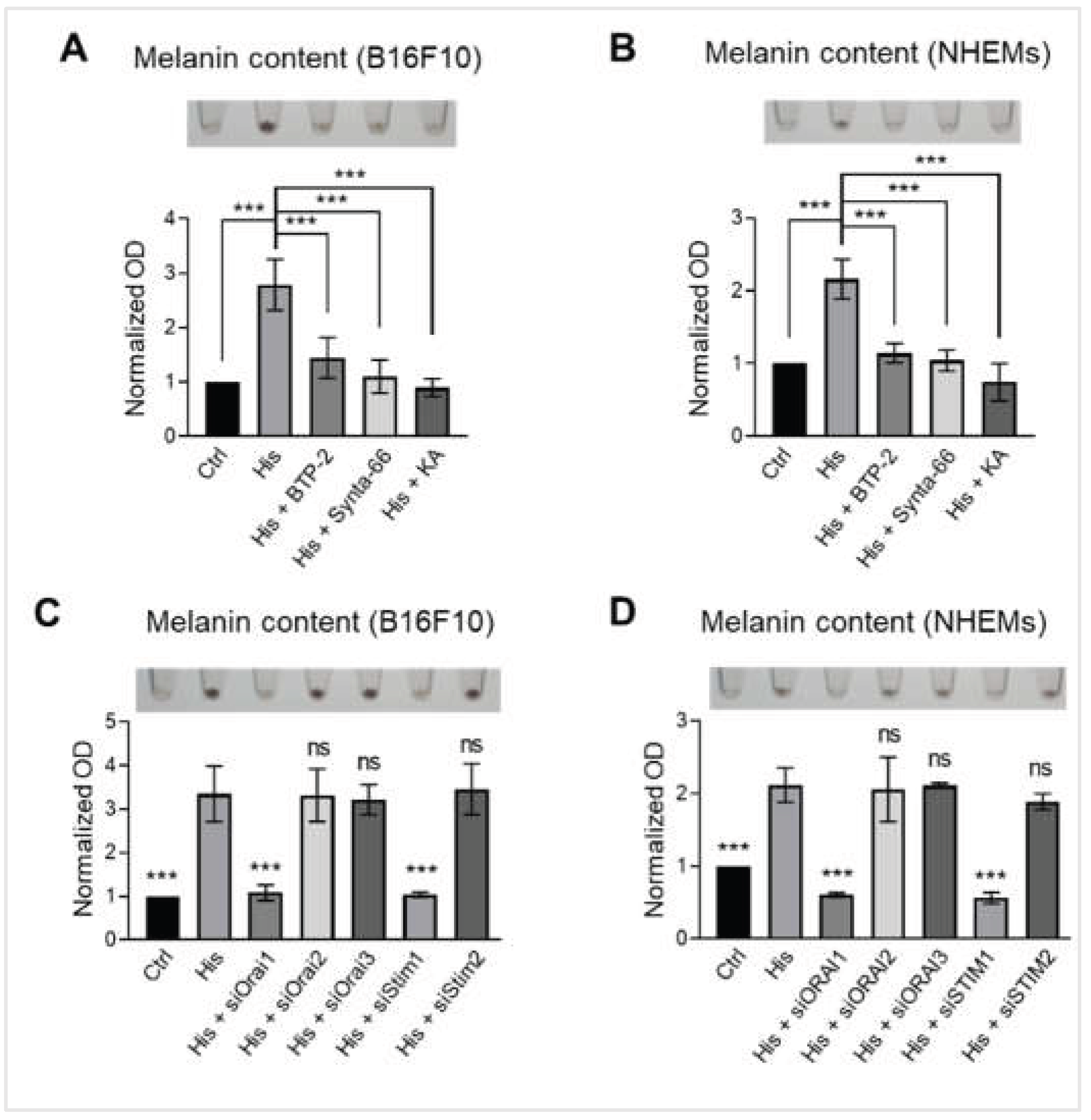
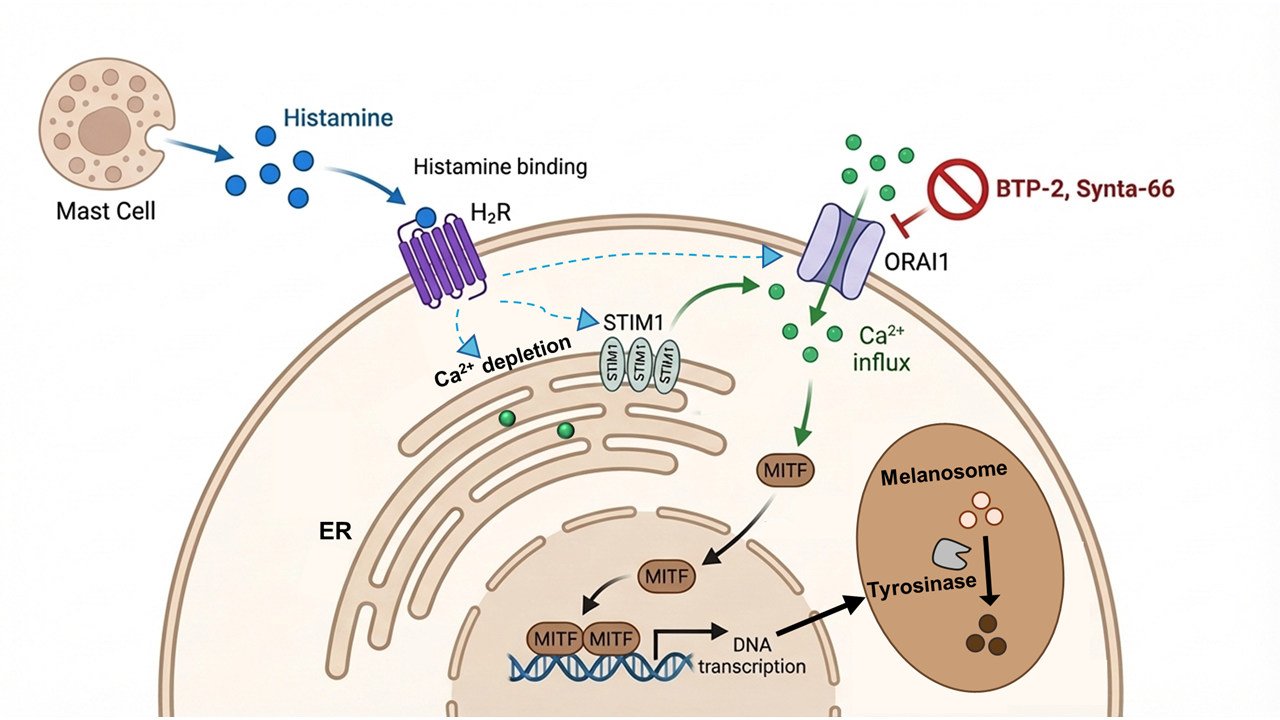
Figure 6.
Histamine-induced melanogenesis is specifically mediated through the ORAI1-STIM1 dependent SOCE pathway. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BTP-2 (CRAC channel inhibitor, 5 μM), Synta-66 (ORAI1 inhibitor, 10 μM), or Kojic acid (KA, tyrosinase inhibitor, 50 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images of NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BTP-2 (5 μM), Synta-66 (10 μM), or KA (50 μM) for 6 days. Data are expressed as fold change relative to vehicle control. (C) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with histamine (His, 10 μM) and transfected with siRNAs targeting Orai1 (siOrai1), Orai2 (siOrai2), Orai3 (siOrai3), Stim (siStim1), or Stim2 (siStim2) for 48 hours. Data are expressed as fold change relative to vehicle control. (D) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with histamine (His, 10 μM) and transfected with siRNAs targeting ORAI1 (siORAI1), ORAI2 (siORAI2), ORAI3 (siORAI3), STIM1 (siSTIM1), or STIM2 (siSTIM2) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001, ns: not significant.
Figure 6.
Histamine-induced melanogenesis is specifically mediated through the ORAI1-STIM1 dependent SOCE pathway. (A) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BTP-2 (CRAC channel inhibitor, 5 μM), Synta-66 (ORAI1 inhibitor, 10 μM), or Kojic acid (KA, tyrosinase inhibitor, 50 μM) for 48 hours. Data are expressed as fold change relative to vehicle control. (B) Representative images of NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with vehicle control (Ctrl), histamine (His, 10 μM), histamine and BTP-2 (5 μM), Synta-66 (10 μM), or KA (50 μM) for 6 days. Data are expressed as fold change relative to vehicle control. (C) Representative images showing B16F10 pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with histamine (His, 10 μM) and transfected with siRNAs targeting Orai1 (siOrai1), Orai2 (siOrai2), Orai3 (siOrai3), Stim (siStim1), or Stim2 (siStim2) for 48 hours. Data are expressed as fold change relative to vehicle control. (D) Representative images showing NHEM pellets (upper) and quantification of melanin content normalized to total protein (lower). Cells were treated with histamine (His, 10 μM) and transfected with siRNAs targeting ORAI1 (siORAI1), ORAI2 (siORAI2), ORAI3 (siORAI3), STIM1 (siSTIM1), or STIM2 (siSTIM2) for 6 days. Data are expressed as fold change relative to vehicle control. Data are presented as mean ± SD from three independent experiments. One-way analysis of variance (ANOVA), followed by Dunnett's post-hoc test, was used for multiple comparisons. ***p < 0.001, ns: not significant.