Submitted:
31 December 2025
Posted:
01 January 2026
You are already at the latest version
Abstract
The valorization of wine by-products aligns with circular bioeconomy principles. This study investigates the ultrasound-assisted aqueous extraction (UAE) of bioactive compounds and cell wall polysaccharides from Syrah grape stems (Vitis vinifera L.) to produce cellulose-rich gels with enhanced antioxidant properties. Extractions were performed at three temperatures (10, 20, and 50 °C) and three ultrasonic power densities (120, 206, and 337 W/L), and compared to conventional extraction (CE, 200 rpm). The results demonstrated that UAE significantly accelerated the extraction kinetics for total phenolics (TP), flavonols, and antioxidant capacity (ABTS, FRAP), achieving up to a 3.1-fold increase in TP yield at 20°C. Notably, UAE at 337 W/L and 20 °C produced antioxidant levels equivalent to those obtained by CE at 50 °C, enabling high efficiency at lower, compound-preserving temperatures. Carbohydrate analysis revealed that the extracts were inherently "cellulose-rich" (glucose ~49–52 mol%), with co-extracted pectins and hemicelluloses forming a composite hydrogel matrix. While total polysaccharide yield was maximized at 10 °C, UAE's primary role was the structural modification of polymers rather than increasing bulk yield. The process reduced extraction times by 3- to over 6-fold to achieve equivalent bioactive yields compared to CE. This work establishes UAE with water as a green, efficient strategy for the integrated, one-step recovery of antioxidant phenolics and gel-forming polysaccharides from grape stems, transforming this underutilized residue into a multifunctional, value-added ingredient for food and pharmaceutical applications.
Keywords:
1. Introduction
2. Results and Discussion
2.1. Carbohydrate Composition
2.2. Extraction Kinetics and Process Efficiency: Phenolics, Flavonols and Antioxidant Capacity
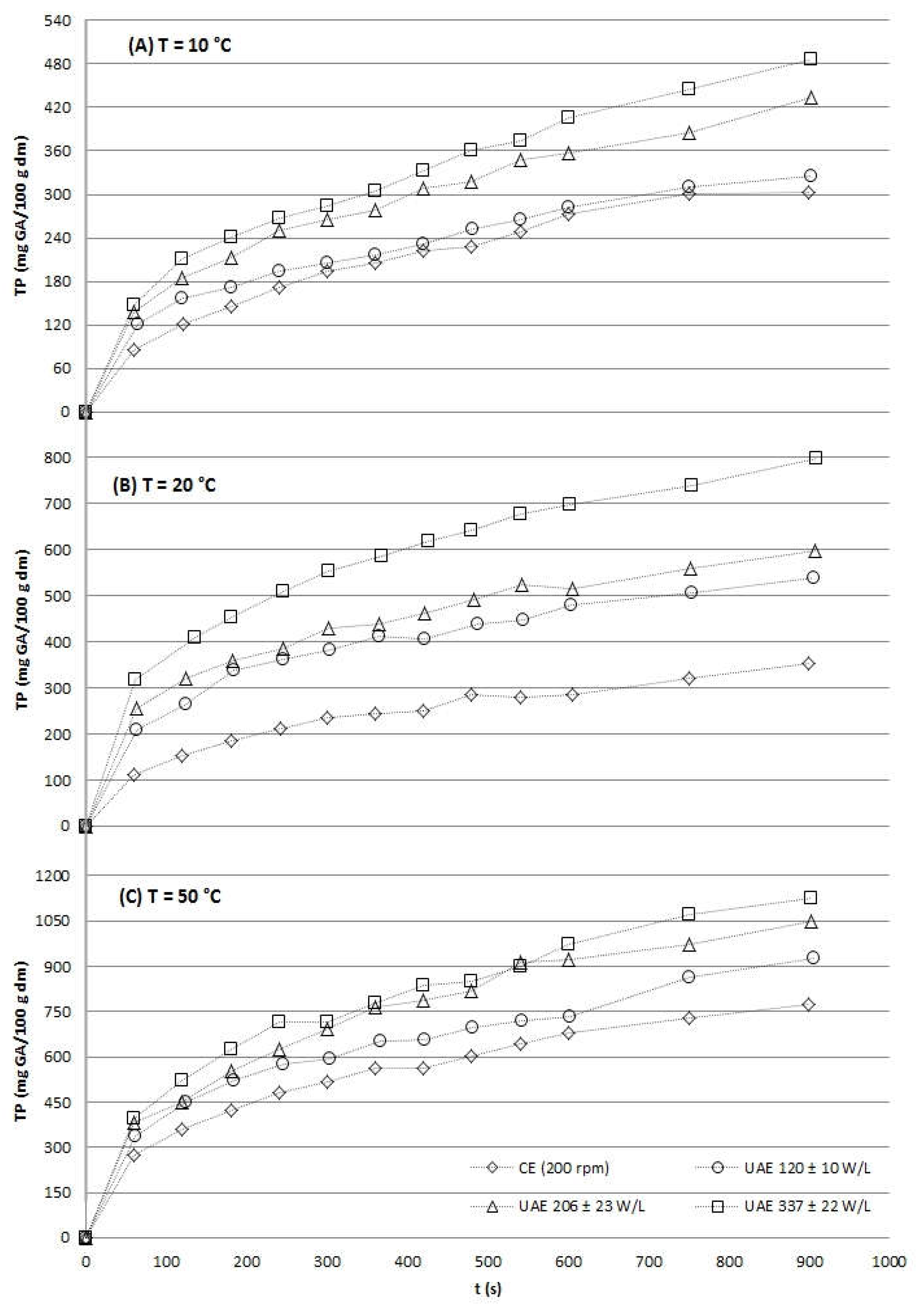
2.2.1. Total Phenolics
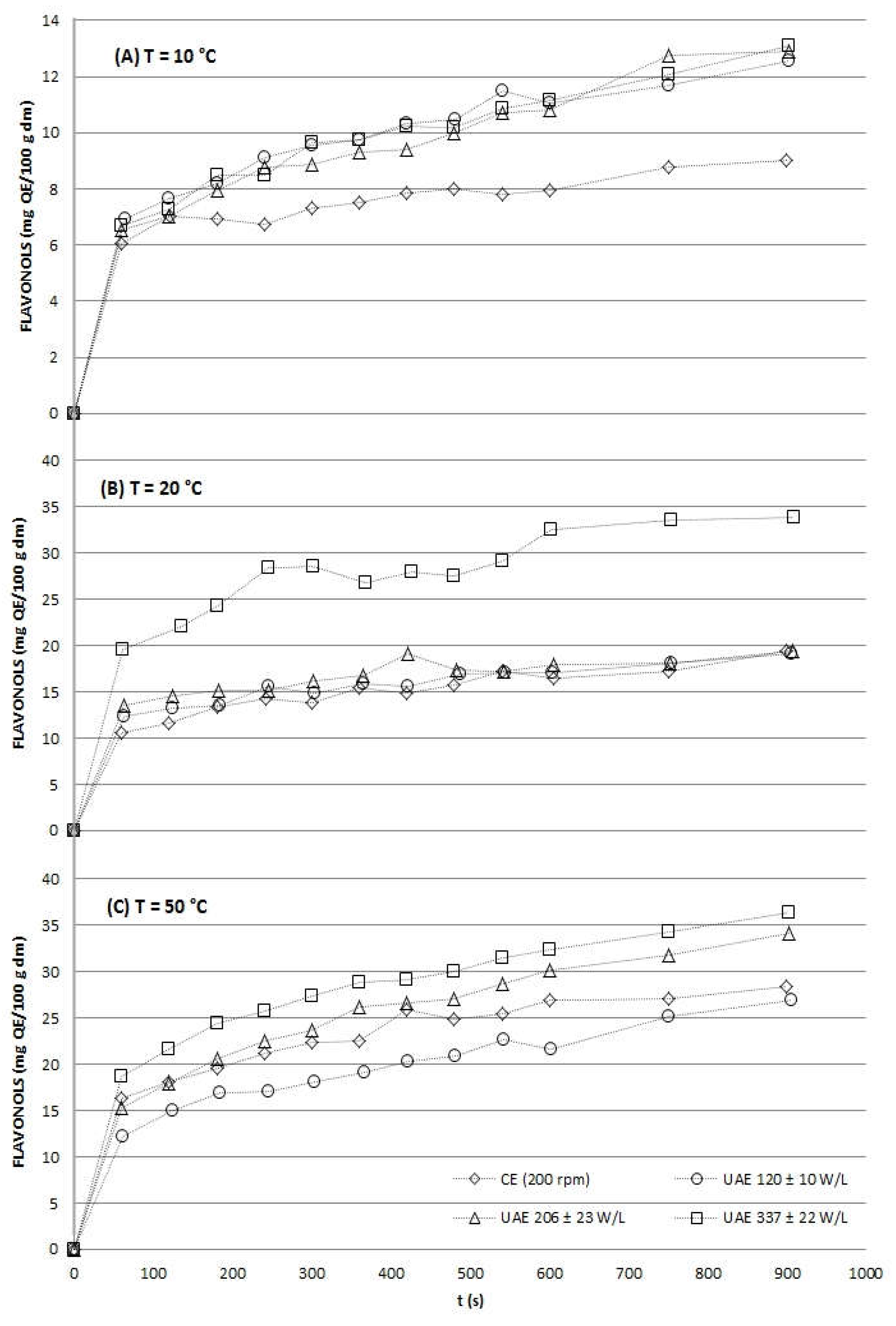
2.2.2. Flavonols Totals
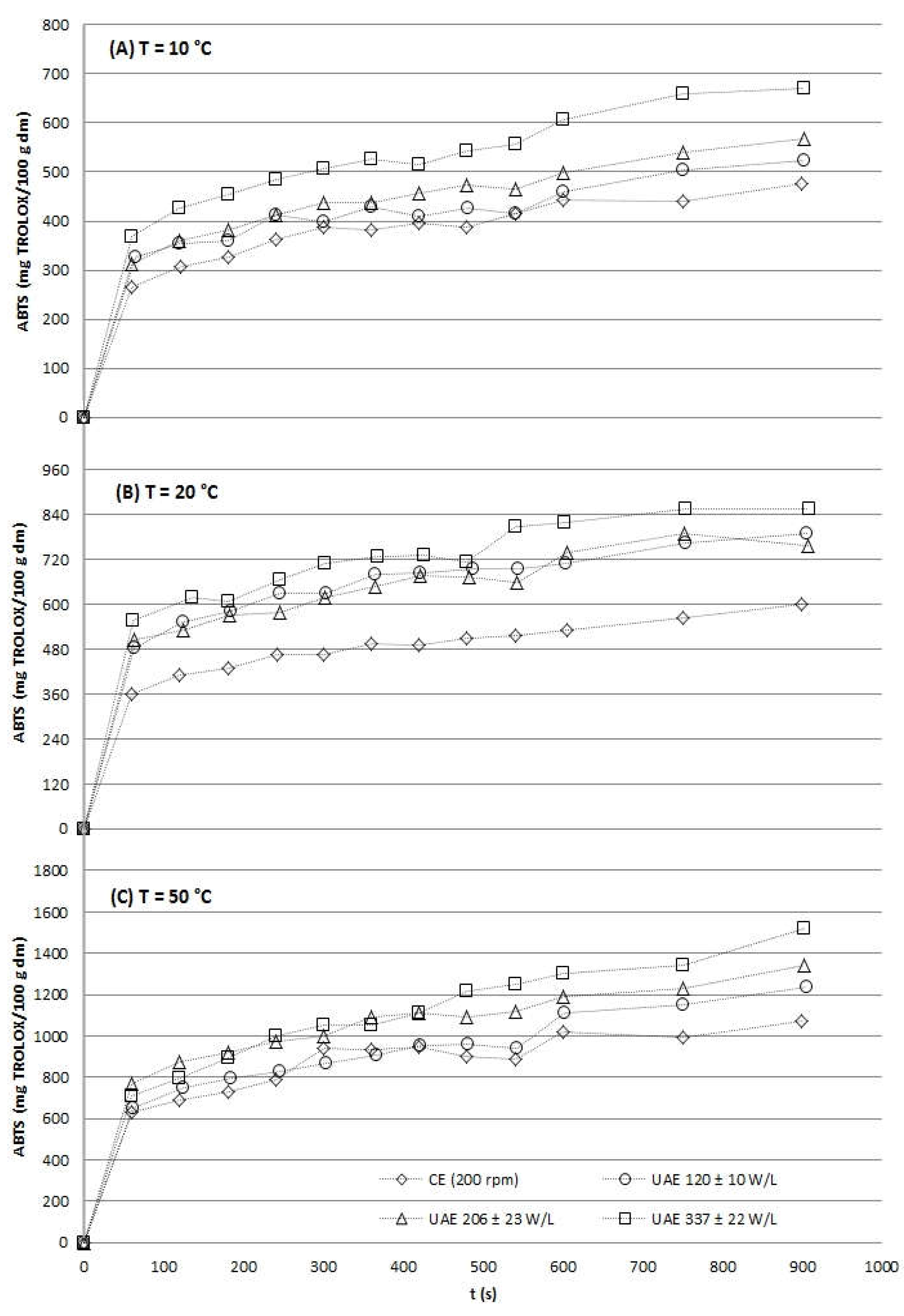
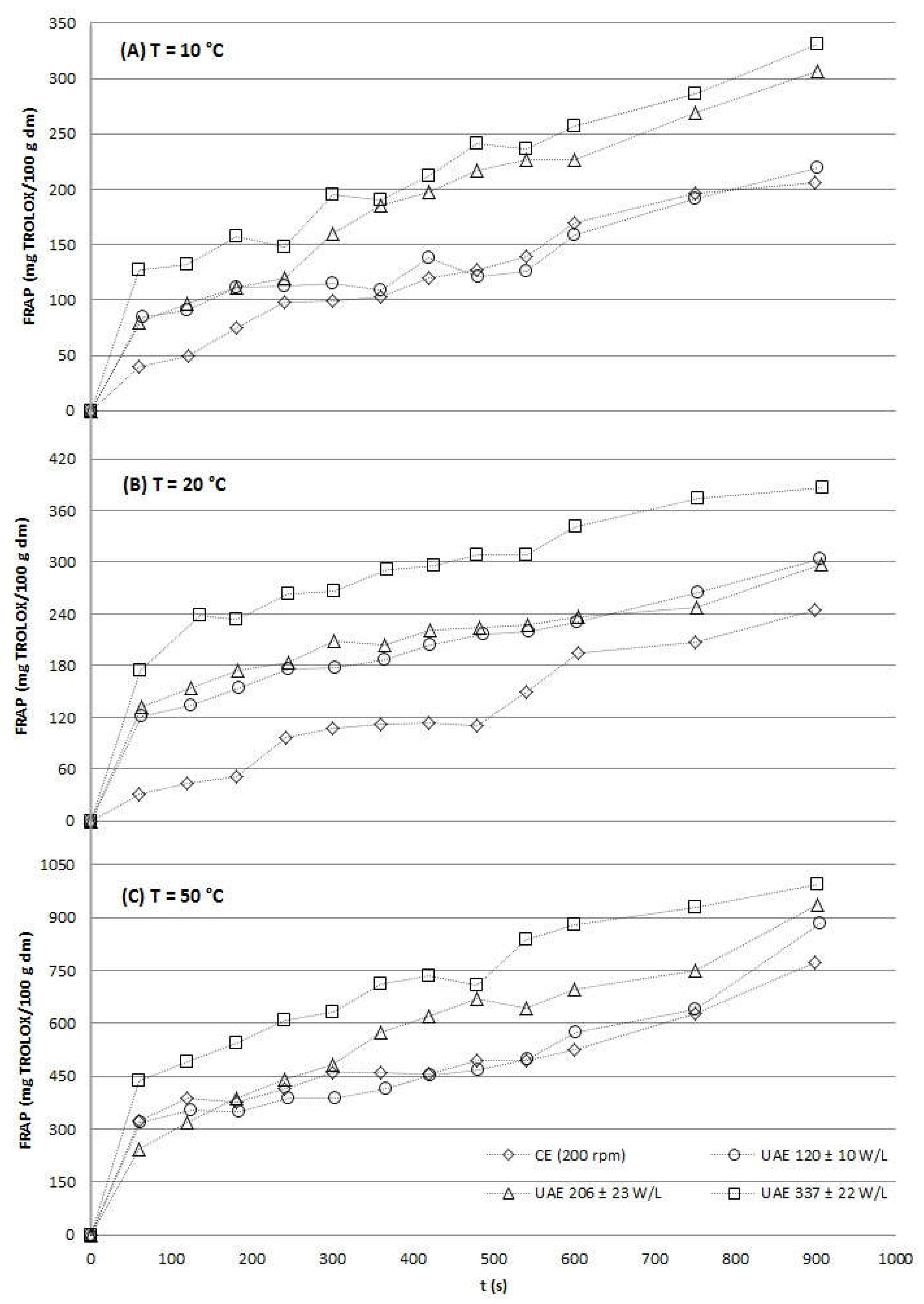
2.2.3. Antioxidant Activity
2.2.4. Process Efficiency
3. Conclusions
4. Materials and Methods
4.1. Samples
4.2. Ultrasound-Assisted Extraction Process
4.2.1. Acoustic Field Characterization
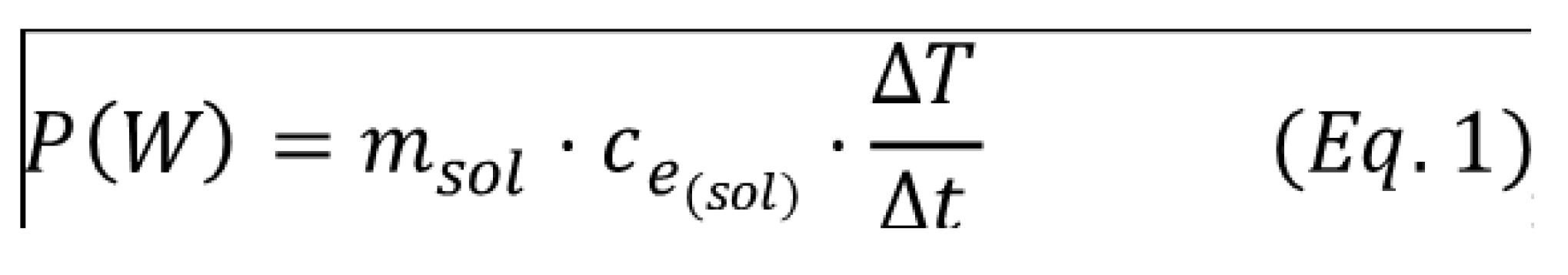
4.3. Conventional Extraction
4.4. Alcohol Insoluble Residues (AIRs) and Analysis of Carbohydrate Composition
4.5. Determination of Bioactive Compounds from Grape Stems Extracts
4.5.1. Determination of Total Phenolics
4.5.2. Determination of Flavonols Content
4.6. Antioxidant Capacity of Grape Stem Extracts
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Commission. A new Circular Economy Action Plan: For a cleaner and more competitive Europe. COM/2020/98 final.
- Sharma, S.; Sharma, U.; Kumar, R. Exploring the valorization of fruit bio-waste for sustainable utilization in agro-food industries: A Comprehensive Review. Food Biosci. 2025, 59, 104575. [Google Scholar] [CrossRef]
- Spigno, G.; Marinoni, L.; Garrido, G. D. State of the art in grape processing by-products. In Handbook of Grape Processing By-Products; Academic Press: Cambridge, MA, USA, 2017; pp. 1–27. [Google Scholar]
- Moreira, M.M.; Rodrigues, F.; Barros, L. Winery by-products as a valuable source of bioactive compounds for food and pharmaceutical industries. In Valorization of Agri-Food Wastes and By-Products; Academic Press: Cambridge, MA, USA, 2023; pp. 555–578. [Google Scholar]
- Shawky, E.; Gibbons, S.; Selim, D.A. Bio-sourcing from byproducts: A comprehensive review of bioactive molecules in Agri-Food Waste (AFW) streams for valorization and sustainable applications. Bioresour. Technol. 2025, 415, 131885. [Google Scholar] [CrossRef]
- Ribeiro, T.P.; Lima, M.J.R.; Lima, Á.S.; Rocha, C.M.R. Grape stems as a source of bioactive compounds: A review. Food Biosci. 2022, 49, 101927. [Google Scholar]
- Devi, M.; Gidado, M.J. Sustainable valorisation of postharvest waste for disease control and quality preservation in fruits and vegetables. Waste Manag. 2026, 186, 300–312. [Google Scholar] [CrossRef]
- Gullón, B.; Gagaoua, M.; Barba, F. J.; Gullón, P.; Zhang, W.; Lorenzo, J. M. Seaweeds as promising resource of bioactive compounds: Overview of novel extraction strategies and design of tailored meat products. Trends Food Sci. Technol. 2020, 100, 1–18. [Google Scholar] [CrossRef]
- Ahmed, M.M.; Nabi, M.H.B.; Zzaman, W. Valorization of plant-based agro-waste into sustainable food packaging materials: Current approaches and functional applications. Appl. Food Res. 2025, 5, 100780. [Google Scholar] [CrossRef]
- Marić, M.; Grassino, A.N.; Zhu, Z.; Barba, F.J.; Brnčić, M.; Rimac Brnčić, S. An overview of the traditional and innovative approaches for pectin extraction from plant food wastes and by-products: Ultrasound-, microwaves-, and enzyme-assisted extraction. Trends Food Sci. Technol. 2018, 76, 28–37. [Google Scholar] [CrossRef]
- Arévalo-Ortiz, F.H.; Palma, J.C.; Visitación-Figueroa, L. Valorization of postharvest waste from table grapes through the production of spirits and distillate by-products based on their antioxidant capacity. Waste Manag. Bull. 2025, 3, 105–115. [Google Scholar] [CrossRef]
- Chennat, F.; Vian, M. A.; Fabiano-Tixier, A. S.; Nutrizio, M.; Jambrak, A. R.; Munekata, P. E. S. A review of sustainable and intensified techniques for extraction of food and natural products. Green Chem. 2020, 22, 2325–2353. [Google Scholar] [CrossRef]
- Tiwari, B.K. Ultrasound: A clean, green extraction technology. TrAC Trends Anal. Chem. 2015, 71, 100–109. [Google Scholar] [CrossRef]
- Gómez, L.; Tiwari, B.; García-Vaquero, M. Emerging extraction techniques: Ultrasonic-assisted extraction. In Sustainable Seaweed Technologies; Elsevier: Amsterdam, The Netherlands, 2022; pp. 171–189. [Google Scholar]
- Egüés, I.; Hernandez-Ramos, F.; Rivilla, I.; Labidi, J. Optimization of Ultrasound Assisted Extraction of Bioactive Compounds from Apple Pomace. Molecules 2021, 26, 3783. [Google Scholar] [CrossRef]
- Pan, Y.; Wang, K.; Huang, S.; Wang, H.; Mu, X.; He, C.; Ji, X.; Zhang, J.; Huang, F. Antioxidant activity of microwave-assisted extract of longan (Dimocarpus longan Lour.) peel. Food Chem. 2008, 106, 1264–1270. [Google Scholar] [CrossRef]
- López-García, J.; Rodríguez-Seoane, P.; Díaz-Reinoso, B.; Moure, A.; Domínguez, H. Hydrogels from winemaking waste streams: Production, tuning of properties, and potential applications. Food Hydrocoll. 2024, 149, 109567. [Google Scholar]
- Rivas, M. Á.; Cásedas, G.; Les, F.; Gómez-Serranillos, M. P.; López, V. Bioactivity and cell-compatibility of β-glucan and pectin-based hydrogels for wound healing applications. Int. J. Biol. Macromol. 2023, 242, 124843. [Google Scholar]
- González-Centeno, M. R.; Rosselló, C.; Simal, S.; Garau, M. C.; López, F.; Femenia, A. Physico-chemical properties of cell wall materials obtained from ten grape varieties and their byproducts: grape pomaces and stems. LWT-Food Sci. Technol. 2010, 43, 1580–1586. [Google Scholar] [CrossRef]
- Yapo, B.M.; Robert, C.; Etienne, I.; Wathelet, B.; Paquot, M. Effect of extraction conditions on the yield, purity and surface properties of sugar beet pulp pectin extracts. Food Chem. 2007, 100, 1356–1364. [Google Scholar] [CrossRef]
- Chan, S.Y.; Choo, W.S.; Young, D.J.; Loh, X.J. Pectin as a rheology modifier: Origin, structure, commercial production and rheology. Carbohydr. Polym. 2017, 161, 118–139. [Google Scholar] [CrossRef]
- Femenia, A.; Lefebvre, A.C.; Thebaudin, J.Y.; Robertson, J.A.; Bourgeois, C.M. Physical and sensory properties of model foods supplemented with cauliflower fiber. J. Food Sci. 1997, 62, 635–639. [Google Scholar] [CrossRef]
- Chennat, F.; Rombaut, N.; Sicaire, A.G.; Meullemiestre, A.; Fabiano-Tixier, A.S.; Abert-Vian, M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef]
- Sarkar, P.; Bosneaga, E. Plant cell walls throughout evolution: towards a molecular understanding of their design principles. J. Exp. Bot. 2009, 60, 3615–3635. [Google Scholar] [CrossRef]
- Wang, X.; Chen, Q.; Lü, X. Pectin extracted from apple pomace and citrus peel by subcritical water. Food Hydrocoll. 2014, 38, 129–137. [Google Scholar] [CrossRef]
- Ebringerová, A.; Heinze, T. Xylan and xylan derivatives – biopolymers with valuable properties, 1. Naturally occurring xylans structures, isolation procedures and properties. Macromol. Rapid Commun. 2000, 21, 542–556. [Google Scholar] [CrossRef]
- Ashokkumar, M. The characterization of acoustic cavitation bubbles – An overview. Ultrason. Sonochem. 2011, 18, 864–872. [Google Scholar] [CrossRef]
- González-Centeno, M.R.; Knoerzer, K.; Sabarez, H.; Simal, S.; Rosselló, C.; Femenia, A. Effect of acoustic frequency and power density on the aqueous ultrasonic-assisted extraction of grape pomace phenolics. Ultrason. Sonochem. 2014, 21, 2176–2184. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Growth of the plant cell wall. Nat. Rev. Mol. Cell Biol. 2005, 6, 850–861. [Google Scholar] [CrossRef] [PubMed]
- Virot, M.; Tomao, V.; Le Bourvellec, C.; Renard, C.M.; Chemat, F. Towards the industrial production of antioxidants from food processing by-products with ultrasound-assisted extraction. Ultrason. Sonochem. 2010, 17, 1066–1074. [Google Scholar] [CrossRef]
- Rostagno, M.A.; Palma, M.; Barroso, C.G. Ultrasound-assisted extraction of soy isoflavones. J. Chromatogr. A 2003, 1012, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, B.; Yao, S. Application of ultrasonic technique for extracting chlorogenic acid from Eucommia ulmodies Oliv. (E. ulmodies). Ultrason. Sonochem. 2005, 12, 295–300. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis of AOAC International, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Spigno, G.; De Faveri, D.M. Antioxidants from grape stalks and marc: Influence of extraction procedure on yield, purity and antioxidant power of the extracts. J. Food Eng. 2007, 78, 793–801. [Google Scholar] [CrossRef]
- Mason, T.J.; Chemat, F.; Vinatoru, M. The extraction of natural products using ultrasound or microwaves. Curr. Org. Chem. 2011, 15, 237–247. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Margulis, M.A.; Margulis, I.M. Calorimetric method for measurement of acoustic power absorbed in a volume of a liquid. Ultrason. Sonochem. 2003, 10, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Barba, F.J.; Zhu, Z.; Koubaa, M.; Sant’Ana, A.S.; Orlien, V. Green alternative methods for the extraction of antioxidant bioactive compounds from winery wastes and by-products: A review. Trends Food Sci. Technol. 2016, 49, 96–109. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Miller, J.N.; Miller, J.C. Statistics and Chemometrics for Analytical Chemistry, 6th ed.; Pearson Education Limited: Harlow, UK, 2010. [Google Scholar]
- Femenia, A.; García-Pascual, P.; Simal, S.; Rosselló, C. Effects of heat treatment on bioactive polysaccharide acemannan and cell wall polymers from Aloe barbadensis Miller. Carbohydr. Polym. 2003, 51, 397–405. [Google Scholar] [CrossRef]
- Blumenkrantz, N.; Asboe-Hansen, G. New method for quantitative determination of uronic acids. Anal. Biochem. 1973, 54, 484–489. [Google Scholar] [CrossRef]
- Englyst, H.N.; Cummings, J.H. Simplified method for the measurement of total non-starch polysaccharides by gas-liquid chromatography of constituent sugars as alditol acetates. Analyst 1984, 109, 937–942. [Google Scholar] [CrossRef]
- Waterhouse, A.L. Determination of total phenolics. In Current Protocols in Food Analytical Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002. [Google Scholar]
- Escarpa, A.; González, M.C. Approach to the content of total extractable phenolic compounds from different food samples by comparison of chromatographic and spectrophotometric methods. Anal. Chim. Acta 2001, 427, 119–127. [Google Scholar] [CrossRef]
- ISO. Accuracy (Trueness and Precision) of Measurement Methods and Results—Part 1: General Principles and Definitions; ISO 5725-1:1994; International Organization for Standardization: Geneva, Switzerland, 1994. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- McKillup, S. Statistics Explained: An Introductory Guide for Life Scientists, 2nd ed.; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Saeman, J.F.; Moore, W.E.; Mitchell, R.L.; Millett, M.A. Techniques for the determination of pulp constituents by quantitative paper chromatography. Tappi 1954, 37, 336–343. [Google Scholar]
- Gómez, L.; Tiwari, B.; García-Vaquero, M. Emerging extraction techniques: Ultrasonic-assisted extraction. In Sustainable Seaweed Technologies; Elsevier: Amsterdam, The Netherlands, 2022; pp. 171–189. [Google Scholar]
- Virot, M.; Tomao, V.; Le Bourvellec, C.; Renard, C.M.; Chemat, F. Towards the industrial production of antioxidants from food processing by-products with ultrasound-assisted extraction. Ultrason. Sonochem. 2010, 17, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Rostagno, M.A.; Palma, M.; Barroso, C.G. Ultrasound-assisted extraction of soy isoflavones. J. Chromatogr. A 2003, 1012, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, B.; Yao, S. Application of ultrasonic technique for extracting chlorogenic acid from Eucommia ulmodies Oliv. (E. ulmodies). Ultrason. Sonochem. 2005, 12, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Margulis, M.A.; Margulis, I.M. Calorimetric method for measurement of acoustic power absorbed in a volume of a liquid. Ultrason. Sonochem. 2003, 10, 343–345. [Google Scholar] [CrossRef]
- McKillup, S. Statistics Explained: An Introductory Guide for Life Scientists, 2nd ed.; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
| 10 ºC | 20 ºC | 50 ºC | ||||||||||||||
| Fresh stems | CE | UAE120 | UAE206 | UAE337 | CE | UAE120 | UAE206 | UAE337 | CE | UAE120 | UAE206 | UAE337 | ||||
| Rha | 0.9 ± 0.2 | 1.0 ± 0.1 | 0.9 ± 0.2 | 0.9 ± 0.1 | 0.8 ± 0.1 | 0.9 ± 0.0 | 0.7 ± 0.1 | 0.9 ± 0.0 | 0.8 ± 0.1 | 0.7 ± 0.2 | 0.6 ± 0.1 | 0.5 ± 0.1 | 0.4 ± 0.1 | |||
| Fuc | 0.4 ± 0.0 | 0.5 ± 0.1 | 0.5 ± 0.0 | 0.4 ± 0.1 | 0.5 ± 0.1 | 0.5 ± 0.0 | 0.4 ± 0.0 | 0.5 ± 0.0 | 0.4 ± 0.1 | 0.6 ± 0.2 | 0.6 ± 0.0 | 0.4 ± 0.1 | 0.5 ± 0.0 | |||
| Ara | 4.1 ± 0.3 | 4.5 ± 0.1 | 4.9 ± 0.4 | 4.4 ± 0.1 | 4.5 ± 0.2 | 4.3 ± 0.0 | 4.5 ± 0.2 | 4.3 ± 0.2 | 4.6 ± 0.1 | 4.2 ± 0.2 | 4.1 ± 0.1 | 4.0 ± 0.2 | 4.1 ± 0.1 | |||
| Xyl | 15.9 ± 0.4 | 15.5 ± 0.2 | 15.7 ± 0.5 | 15.5 ± 0.3 | 15.1 ± 0.5 | 15.3 ± 0.3 | 15.6 ± 0.2 | 15.1 ± 0.5 | 15.2 ± 0.4 | 15.2 ± 0.4 | 14.8 ± 0.5 | 14.7 ± 0.4 | 14.9 ± 0.7 | |||
| Man | 1.9 ± 0.1 | 1.6 ± 00 | 1.8 ± 0.1 | 1.7 ± 0.1 | 1.9 ± 0.1 | 2.3 ± 0.2 | 2.1 ± 0.1 | 2.2 ± 0.1 | 2.0 ± 0.1 | 1.2 ± 0.0 | 1.3 ± 0.1 | 1.8 ± 0.1 | 1.4 ± 0.1 | |||
| Gal | 2.9 ± 0.3 | 3.5 ± 0.2 | 2.9 ± 0.1 | 3.4 ± 0.0 | 2.5 ± 0.0 | 3.3 ± 0.1 | 3.5 ± 0.1 | 4.2 ± 0.2 | 4.0 ± 0.0 | 3.7 ± 0.1 | 4.4 ± 0.2 | 3.9 ± 0.1 | 3.8 ± 0.2 | |||
| Glc | 49.5 ± 1.2 | 49.7 ± 0.9 | 51.2 ± 1.4 | 52.3 ± 0.9 | 52.5 ± 0.8 | 49.5 ± 0.6 | 49.9 ± 0.7 | 50.5 ± 1.2 | 51.2 ± 0.8 | 49.9 ± 0.5 | 49.5 ± 0.3 | 50.5 ± 0.6 | 50.9 ± 1.2 | |||
| Glc (1M) | (4.9 ± 0.4) | (4.5 ± 0.2) | (5.1 ± 0.2) | (4.4 ± 0.3) | (4.5 ± 0.1) | (4.6 ± 0.2) | (4.9 ± 0.3) | (4.4 ± 0.4) | (4.5 ± 0.3) | (4.4 ± 0.3) | (4.5 ± 0.4) | (4.7 ± 0.3) | (4.3 ± 0.5) | |||
| Uronic A. | 24.4 ± 1.0 | 23.7 ± 1.2 | 22.1 ± 1.6 | 21.4 ± 0.9 | 22.2 ± 1.8 | 23.9 ± 1.7 | 23.3 ± 1.5 | 22.3 ± 1.4 | 21.8 ± 1.3 | 24.5 ± 1.7 | 24.7 ± 0.9 | 24.2 ± 1.7 | 24.0 ± 1.5 | |||
| Total sugars* | 18.9 ± 1.1 | 18.7 ± 1.3 | 19.2 ± 1.1 | 18.7 ± 1.7 | 17.2 ± 1.5 | 17.9 ± 1.3 | 17.3 ± 1.0 | 17.0 ± 1.1 | 16.8 ± 1.5 | 16.7 ± 1.2 | 15.8 ± 0.8 | 15.4 ± 13 | 14.7 ± 1.7 | |||
| Temperature (ºC) | US power density (W/L) |
TPUAE at 15 min (mg GA/100 g dm) |
TimeCE to obtain TPUAE at 15 min (min) |
FlavonolsUAE at 15 min (mg QE/100 g dm) |
TimeCE to obtain FlavonolsUAE at 15 min (min) |
| 120 ± 10 | 326.05 ± 20.61 | 20 | 12.56 ± 0.75 | 45 | |
| 10 | 206 ± 23 | 434.02 ± 14.39 | 40 | 12.92 ± 1.68 | 50 |
| 337 ± 22 | 486.63 ± 12.79 | 50 | 13.08 ± 1.44 | 50 | |
| 120 ± 10 | 538.31 ± 57.85 | 45 | 19.08 ± 2.03 | 15 | |
| 20 | 206 ± 23 | 598.11 ± 29.84 | 60 | 19.42 ± 1.64 | 15 |
| 337 ± 22 | 797.49 ± 18.27 | > 90 | 33.79 ± 3.11 | 30 | |
| 120 ± 10 | 928.03 ± 49.10 | 30 | 27.00 ± 2.28 | 10 | |
| 30 | 206 ± 23 | 1049.84 ± 61.00 | 45 | 34.19 ± 3.35 | 25 |
| 337 ± 22 | 1127.02 ± 12.21 | 55 | 36.29 ± 3.02 | 30 | |
| Temperature (ºC) | US power density (W/L) | ABTSUAE at 15 min (mg TROLOX/100 g dm) | TimeCE to obtain ABTSUAE at 15 min (min) |
FRAPUAE at 15 min (mg TROLOX/100 g dm) |
TimeCE to obtain FRAPUAE at 15 min (min) |
| 120 ± 10 | 522.33 ± 13.03 | 20 | 219.49 ± 3.91 | 15 | |
| 10 | 206 ± 23 | 569.24 ± 36.62 | 20 | 306.44 ± 13.75 | 45 |
| 337 ± 22 | 670.88 ± 15.64 | 70 | 331.25 ± 23.58 | 50 | |
| 120 ± 10 | 788.66 ± 34.64 | 20 | 290.58 ± 9.15 | 20 | |
| 20 | 206 ± 23 | 790.56 ± 52.26 | 20 | 297.90 ± 25.43 | 25 |
| 337 ± 22 | 855.73 ± 7.33 | 25 | 386.69 ± 1.31 | 30 | |
| 120 ± 10 | 1234.17 ± 32.52 | 20 | 883.88 ± 33.10 | 20 | |
| 30 | 206 ± 23 | 1342.69 ± 20.98 | 25 | 938.31 ± 23.40 | 20 |
| 337 ± 22 | 1520.92 ± 11.46 | 30 | 993.99 ± 27.08 | 25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




