Submitted:
30 December 2025
Posted:
31 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
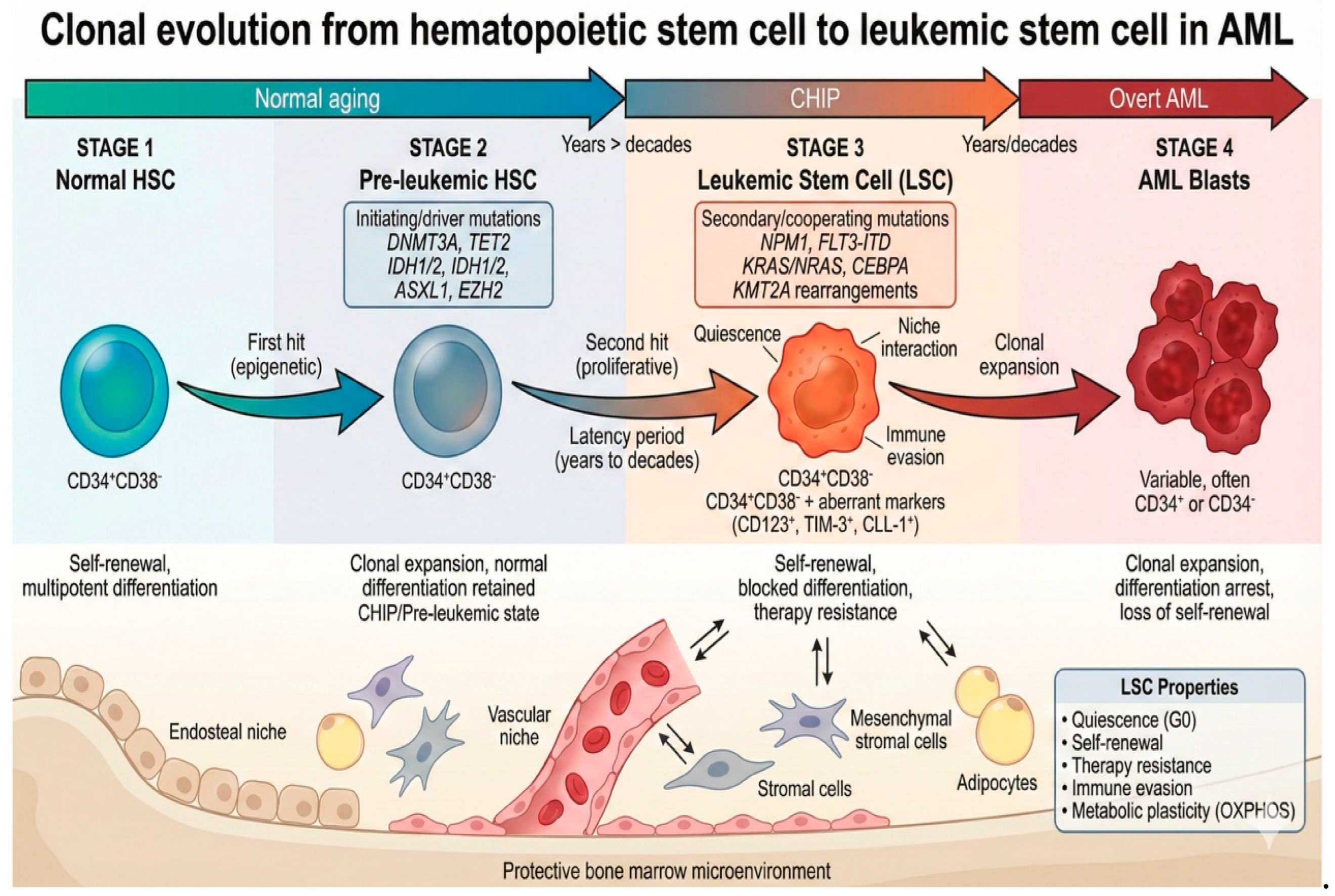
2. Biological Features of LSCs
3. The Role of LSCs and the BMM in Leukemogenesis
3.1. LSCs and Leukemogenesis
3.2. BM Niche and Leukemogenesis
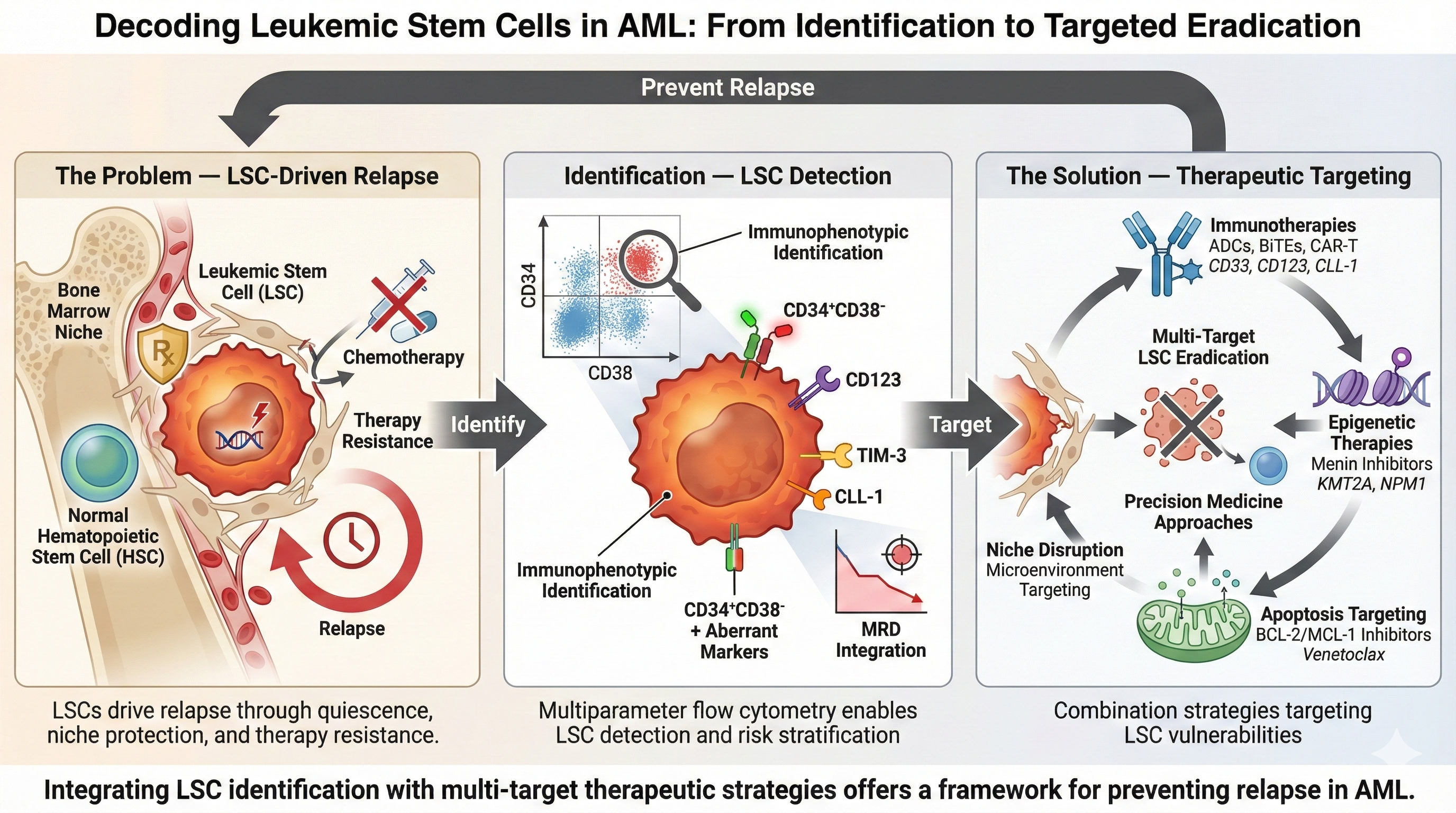
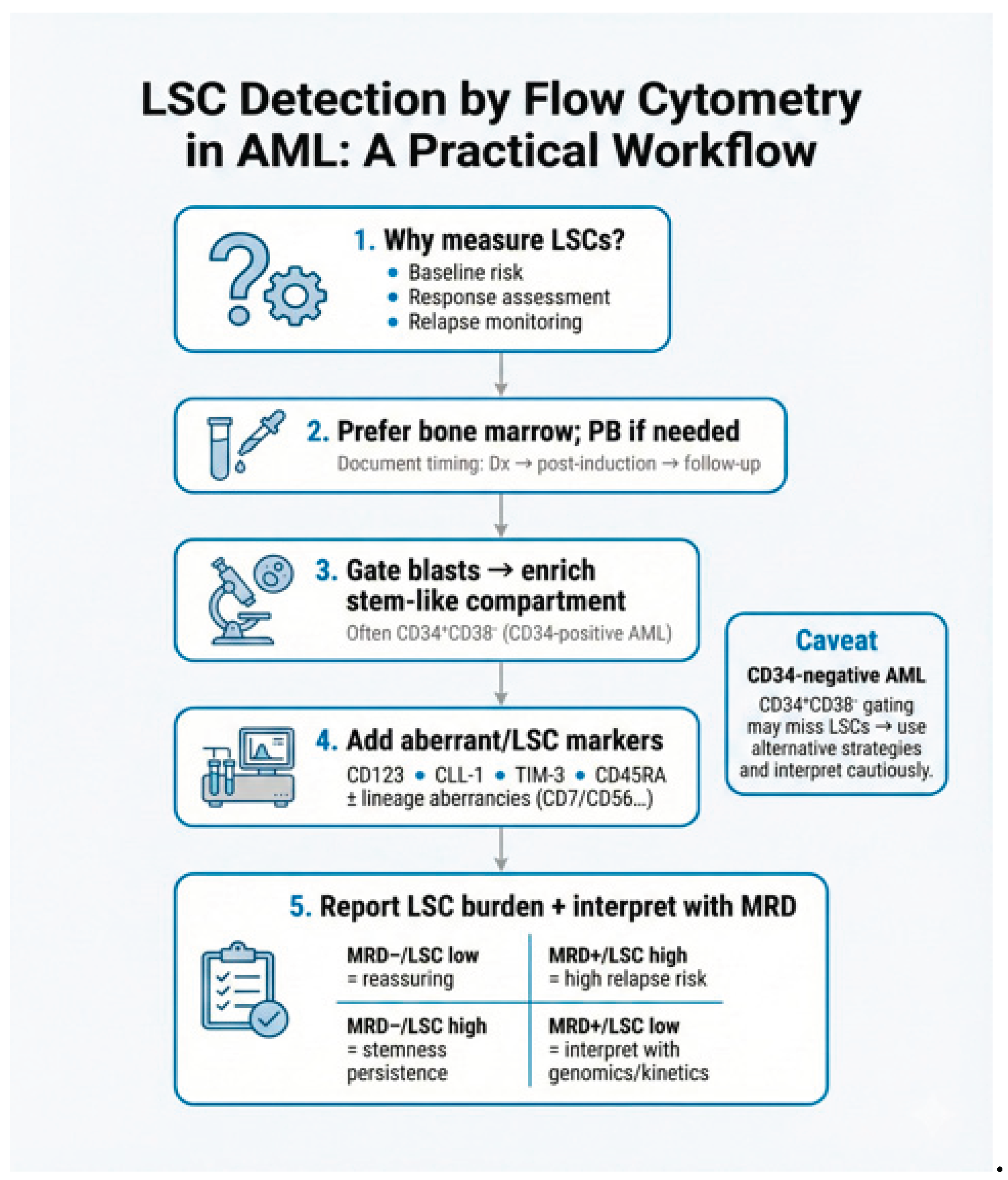
4. Immunophenotypic Identification of LSCs
5. The Contribution of LSCs in Relapse and AML Progression
6. LSCs in Other Hematological Malignancies
7. Prognostic and Predictive Implications of LSCs in AML
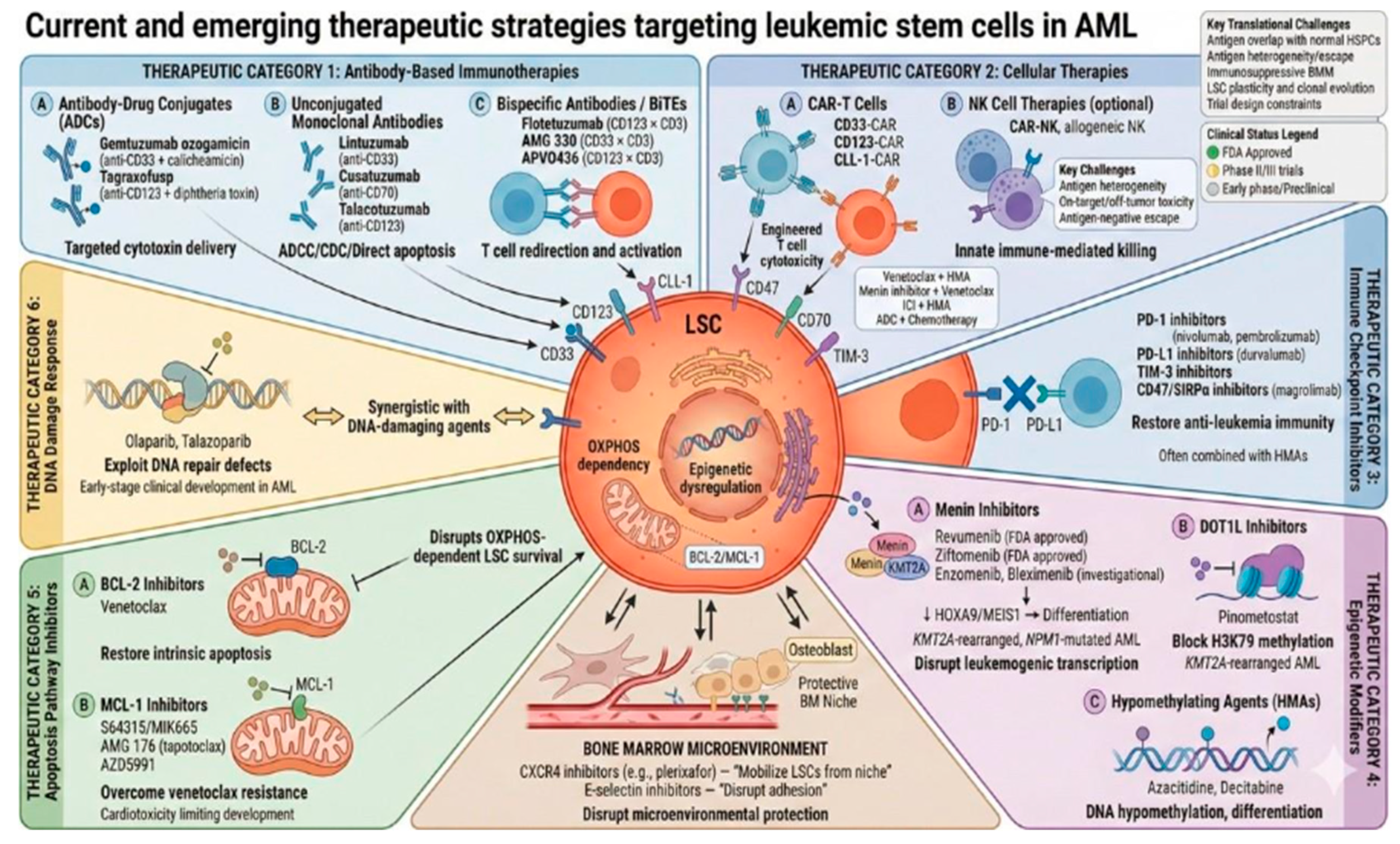
8. Therapeutic Targeting of LSCs in AML
8.1. Immunotherapies
8.1.1. Antibody–Drug Conjugates (ADCs)
8.1.2. Unconjugated Monoclonal Antibodies
8.1.3. Bispecific Antibodies/BiTEs
- Immune checkpoint inhibitors (ICIs)
- Cellular therapies (CAR-T and related platforms)
8.2. Epigenetic Modifiers
8.2.1. Menin Inhibitors
8.2.2. DOT1L Inhibitors
8.3. Targeting Apoptosis Pathways
8.4. DNA Damage Response Targeting: PARP Inhibitors
9. Why Promising Targets Fail in AML
- Absence of an “ideal” AML-specific antigen: on-target/off-tumor toxicity. The most significant barrier for antibody-based and cellular therapies in AML is the lack of a surface antigen uniformly expressed on blasts and LSCs but absent from normal hematopoietic stem and progenitor cells (HSPCs). Commonly targeted antigens—CD33, CD123, and CLL-1—are also expressed on normal myeloid progenitors, creating inherent risk for prolonged myeloablation, severe cytopenias, and infectious complications when potent targeting is achieved. This shared-antigen problem represents the central limitation for CAR-based approaches in AML and explains the tight coupling between efficacy and toxicity in this disease [89,90].
- Antigen heterogeneity and antigen-negative escape: AML is biologically heterogeneous both across patients and within individual patients, characterized by dynamic subclonal evolution and variable antigen density. Single-antigen targeting strategies are therefore vulnerable to antigen-low subpopulations and antigen-negative relapse, particularly under therapeutic selective pressure. This limitation is well recognized in the CAR-T AML literature and has been specifically documented in the context of CD123-directed immunotherapies [91].
- LSC plasticity and clonal evolution under therapy: A key reason that promising therapeutic targets fail to deliver durable responses is that the LSC state is not static. LSCs can alter their phenotype and transcriptional programs in response to treatment, and relapse frequently reflects selection of resistant subclones rather than regrowth of the original dominant population. Current LSC-focused research highlights that stemness traits are shaped by both global and subtype-specific features, and that this heterogeneity fundamentally limits the effectiveness of single-pathway or single-marker targeting strategies [92].
- An immunosuppressive BMM: The BMM in AML undermines immune effector function through multiple mechanisms, including dysfunctional antigen presentation, suppressive myeloid populations, inhibitory cytokines, and metabolic constraints. Baseline T-cell dysfunction and the immunosuppressive tumor microenvironment are increasingly recognized as key factors contributing to the limited and variable clinical activity observed with AML immunotherapies, including CD123-directed approaches [91,93].
- High-risk disease biology can overwhelm single-agent promise (example: TP53-mutated AML): Clinical failures are often most evident in genomically adverse disease, where aggressive biology and profound treatment resistance limit the ability of any single mechanism to meaningfully improve survival. The phase 3 ENHANCE-2 trial of magrolimab plus azacitidine in previously untreated TP53-mutated AML provides a recent example: despite encouraging signals from non-randomized studies, the combination failed to improve OS compared with physician's choice [94].
- Trial-design and implementation constraints in AML: Even effective modalities face significant practical barriers in AML: patients may deteriorate rapidly, have active infections, or carry substantial prior-treatment burdens that compromise immune function and organ reserve. For cellular therapies, logistical challenges including manufacturing time, bridging therapy requirements, and toxicity management, combined with the need to avoid prolonged aplasia, can limit feasibility and confound outcome interpretation. These factors represent core obstacles to the broad applicability of CAR-based approaches in AML.
10. Discussion
11. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chen, Y.; Li, J.; Xu, L.; Găman, M.-A.; Zou, Z. The genesis and evolution of acute myeloid leukemia stem cells in the microenvironment: From biology to therapeutic targeting. Cell Death Discovery 2022, 8, 397. [Google Scholar] [CrossRef]
- Urs, A.P.; Goda, C.; Kulkarni, R. Remodeling of the bone marrow microenvironment during acute myeloid leukemia progression. Ann Transl Med 2024, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Stelmach, P.; Trumpp, A. Leukemic stem cells and therapy resistance in acute myeloid leukemia. Haematologica 2023, 108, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Lapidot, T.; Sirard, C.; Vormoor, J.; Murdoch, B.; Hoang, T.; Caceres-Cortes, J.; Minden, M.; Paterson, B.; Caligiuri, M.A.; Dick, J.E. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994, 367, 645–648. [Google Scholar] [CrossRef]
- Bonnet, D.; Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nature Medicine 1997, 3, 730–737. [Google Scholar] [CrossRef]
- Barreto, I.V.; Pessoa, F.M.C.d.P.; Machado, C.B.; Pantoja, L.d.C.; Ribeiro, R.M.; Lopes, G.S.; Amaral de Moraes, M.E.; de Moraes Filho, M.O.; de Souza, L.E.B.; Burbano, R.M.R.; et al. Leukemic Stem Cell: A Mini-Review on Clinical Perspectives. Frontiers in Oncology 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.-Y. Human acute myeloid leukemia stem cells: evolution of concept. BR 2022, 57, S67–S74. [Google Scholar] [CrossRef]
- Hansen, Q.; Bachas, C.; Smit, L.; Cloos, J. Characteristics of leukemic stem cells in acute leukemia and potential targeted therapies for their specific eradication. Cancer Drug Resistance 2022, 5, 344–367. [Google Scholar] [CrossRef]
- Cozzio, A.; Passegué, E.; Ayton, P.M.; Karsunky, H.; Cleary, M.L.; Weissman, I.L. Similar MLL-associated leukemias arising from self-renewing stem cells and short-lived myeloid progenitors. Genes Dev 2003, 17, 3029–3035. [Google Scholar] [CrossRef]
- van der Werf, I.; Mondala, P.K.; Steel, S.K.; Balaian, L.; Ladel, L.; Mason, C.N.; Diep, R.H.; Pham, J.; Cloos, J.; Kaspers, G.J.L.; et al. Detection and targeting of splicing deregulation in pediatric acute myeloid leukemia stem cells. Cell Reports Medicine 2023, 4, 100962. [Google Scholar] [CrossRef]
- Patel, S.B.; Nemkov, T.; D'Alessandro, A.; Welner, R.S. Deciphering Metabolic Adaptability of Leukemic Stem Cells. Front Oncol 2022, 12, 846149. [Google Scholar] [CrossRef]
- Rattigan, K.M.; Zarou, M.M.; Helgason, G.V. Metabolism in stem cell–driven leukemia: parallels between hematopoiesis and immunity. Blood 2023, 141, 2553–2565. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, R.; Ghosh, S.; Nath, A.; Basu, A.; Biswas, O.; Patil, C.R.; Kundu, C.N. Theragnostic strategies harnessing the self-renewal pathways of stem-like cells in the acute myeloid leukemia. Critical Reviews in Oncology/Hematology 2022, 177, 103753. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Huang, S.; Chen, J.-L. Understanding of leukemic stem cells and their clinical implications. Molecular Cancer 2017, 16, 2. [Google Scholar] [CrossRef]
- Kollmann, S.; Grausenburger, R.; Klampfl, T.; Prchal-Murphy, M.; Bastl, K.; Pisa, H.; Knab, V.M.; Brandstoetter, T.; Doma, E.; Sperr, W.R.; et al. A STAT5B–CD9 axis determines self-renewal in hematopoietic and leukemic stem cells. Blood 2021, 138, 2347–2359. [Google Scholar] [CrossRef] [PubMed]
- Joshi, K.; Zhang, L.; Breslin S.J, P.; Zhang, J. Leukemia Stem Cells in the Pathogenesis, Progression, and Treatment of Acute Myeloid Leukemia. In Leukemia Stem Cells in Hematologic Malignancies; Zhang, H., Li, S., Eds.; Springer Singapore: Singapore, 2019; pp. 95–128. [Google Scholar]
- Azizidoost, S.; Nasrolahi, A.; Sheykhi-Sabzehpoush, M.; Anbiyaiee, A.; Khoshnam, S.E.; Farzaneh, M.; Uddin, S. Signaling pathways governing the behaviors of leukemia stem cells. Genes & Diseases 2024, 11, 830–846. [Google Scholar] [CrossRef]
- Queiroz, K.C.S.; Ruela-de-Sousa, R.R.; Fuhler, G.M.; Aberson, H.L.; Ferreira, C.V.; Peppelenbosch, M.P.; Spek, C.A. Hedgehog signaling maintains chemoresistance in myeloid leukemic cells. Oncogene 2010, 29, 6314–6322. [Google Scholar] [CrossRef]
- Kellaway, S.G.; Potluri, S.; Keane, P.; Blair, H.J.; Ames, L.; Worker, A.; Chin, P.S.; Ptasinska, A.; Derevyanko, P.K.; Adamo, A.; et al. Leukemic stem cells activate lineage inappropriate signalling pathways to promote their growth. Nature Communications 2024, 15, 1359. [Google Scholar] [CrossRef]
- Bouligny, I.M.; Maher, K.R.; Grant, S. Augmenting Venetoclax Activity Through Signal Transduction in AML. J Cell Signal 2023, 4, 1–12. [Google Scholar] [CrossRef]
- Valent, P.; Sadovnik, I.; Eisenwort, G.; Bauer, K.; Herrmann, H.; Gleixner, K.V.; Schulenburg, A.; Rabitsch, W.; Sperr, W.R.; Wolf, D. Immunotherapy-Based Targeting and Elimination of Leukemic Stem Cells in AML and CML. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Dunn, W.G.; McLoughlin, M.A.; Vassiliou, G.S. Clonal hematopoiesis and hematological malignancy. J Clin Invest 2024, 134. [Google Scholar] [CrossRef]
- Choi, H.-S.; Kim, B.S.; Yoon, S.; Oh, S.-O.; Lee, D. Leukemic Stem Cells and Hematological Malignancies. International Journal of Molecular Sciences 2024, 25, 6639. [Google Scholar] [CrossRef]
- Marchand, T.; Pinho, S. Leukemic Stem Cells: From Leukemic Niche Biology to Treatment Opportunities. Frontiers in Immunology 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Tasian, S.K.; Bornhäuser, M.; Rutella, S. Targeting Leukemia Stem Cells in the Bone Marrow Niche. Biomedicines 2018, 6, 22. [Google Scholar] [CrossRef]
- Karantanou, C.; Godavarthy, P.S.; Krause, D.S. Targeting the bone marrow microenvironment in acute leukemia. Leuk Lymphoma 2018, 59, 2535–2545. [Google Scholar] [CrossRef]
- Semedo, C.; Caroço, R.; Almeida, A.; Cardoso, B.A. Targeting the bone marrow niche, moving towards leukemia eradication. Frontiers in Hematology 2024, 3–2024. [Google Scholar] [CrossRef]
- Soto, C.A.; Lo Celso, C.; Purton, L.E.; Frisch, B.J. From the niche to malignant hematopoiesis and back: reciprocal interactions between leukemia and the bone marrow microenvironment. JBMR Plus 2021, 5. [Google Scholar] [CrossRef]
- Yao, Y.; Li, F.; Huang, J.; Jin, J.; Wang, H. Leukemia stem cell-bone marrow microenvironment interplay in acute myeloid leukemia development. Experimental Hematology & Oncology 2021, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Zeijlemaker, W.; Kelder, A.; Oussoren-Brockhoff, Y.J.; Scholten, W.J.; Snel, A.N.; Veldhuizen, D.; Cloos, J.; Ossenkoppele, G.J.; Schuurhuis, G.J. A simple one-tube assay for immunophenotypical quantification of leukemic stem cells in acute myeloid leukemia. Leukemia 2016, 30, 439–446. [Google Scholar] [CrossRef]
- Zeijlemaker, W.; Grob, T.; Meijer, R.; Hanekamp, D.; Kelder, A.; Carbaat-Ham, J.C.; Oussoren-Brockhoff, Y.J.M.; Snel, A.N.; Veldhuizen, D.; Scholten, W.J.; et al. CD34+CD38− leukemic stem cell frequency to predict outcome in acute myeloid leukemia. Leukemia 2019, 33, 1102–1112. [Google Scholar] [CrossRef]
- Das, N.; Panda, D.; Gajendra, S.; Gupta, R.; Thakral, D.; Kaur, G.; Khan, A.; Singh, V.K.; Vemprala, A.; Bakhshi, S.; et al. Immunophenotypic characterization of leukemic stem cells in acute myeloid leukemia using single tube 10-colour panel by multiparametric flow cytometry: Deciphering the spectrum, complexity and immunophenotypic heterogeneity. International Journal of Laboratory Hematology 2024, 46, 646–656. [Google Scholar] [CrossRef]
- Reuvekamp, T.; Janssen, L.L.G.; Ngai, L.L.; Carbaat-Ham, J.; den Hartog, D.; Scholten, W.J.; Kelder, A.; Hanekamp, D.; Wensink, E.; van Gils, N.; et al. The role of the primitive marker CD133 in CD34-negative acute myeloid leukemia for the detection of leukemia stem cells. Cytometry Part B: Clinical Cytometry 2025, 108, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.A.; Bill, M.; Rosenberg, C.A. OMIP 072: A 15-color panel for immunophenotypic identification, quantification, and characterization of leukemic stem cells in children with acute myeloid leukemia. Cytometry Part A 2021, 99, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Meddi, E.; Savi, A.; Moretti, F.; Mallegni, F.; Palmieri, R.; Paterno, G.; Buzzatti, E.; Del Principe, M.I.; Buccisano, F.; Venditti, A.; et al. Measurable Residual Disease (MRD) as a Surrogate Efficacy-Response Biomarker in AML. International Journal of Molecular Sciences 2023, 24, 3062. [Google Scholar] [CrossRef]
- Chea, M.; Rigolot, L.; Canali, A.; Vergez, F. Minimal Residual Disease in Acute Myeloid Leukemia: Old and New Concepts. International Journal of Molecular Sciences 2024, 25, 2150. [Google Scholar] [CrossRef]
- Heuser, M.; Freeman, S.D.; Ossenkoppele, G.J.; Buccisano, F.; Hourigan, C.S.; Ngai, L.L.; Tettero, J.M.; Bachas, C.; Baer, C.; Béné, M.C.; et al. 2021 Update on MRD in acute myeloid leukemia: a consensus document from the European LeukemiaNet MRD Working Party. Blood 2021, 138, 2753–2767. [Google Scholar] [CrossRef]
- Schuurhuis, G.J.; Heuser, M.; Freeman, S.; Béné, M.C.; Buccisano, F.; Cloos, J.; Grimwade, D.; Haferlach, T.; Hills, R.K.; Hourigan, C.S.; et al. Minimal/measurable residual disease in AML: a consensus document from the European LeukemiaNet MRD Working Party. Blood 2018, 131, 1275–1291. [Google Scholar] [CrossRef]
- Reuvekamp, T.; Bachas, C.; Cloos, J. Immunophenotypic features of early haematopoietic and leukaemia stem cells. Int J Lab Hematol 2024, 46, 795–808. [Google Scholar] [CrossRef]
- Cao, S.; Wang, Q.; Zhu, G. From Chemotherapy to Targeted Therapy: Unraveling Resistance in Acute Myeloid Leukemia Through Genetic and Non-Genetic Insights. International Journal of Molecular Sciences 2025, 26, 4005. [Google Scholar] [CrossRef]
- van Gils, N.; Denkers, F.; Smit, L. Escape From Treatment; the Different Faces of Leukemic Stem Cells and Therapy Resistance in Acute Myeloid Leukemia. Frontiers in Oncology 2021, Volume 11 - 2021. [Google Scholar] [CrossRef] [PubMed]
- Niu, J.; Peng, D.; Liu, L. Drug Resistance Mechanisms of Acute Myeloid Leukemia Stem Cells. Frontiers in Oncology 2022, Volume 12 - 2022. [Google Scholar] [CrossRef]
- Mojtahedi, H.; Yazdanpanah, N.; Rezaei, N. Chronic myeloid leukemia stem cells: targeting therapeutic implications. Stem Cell Research & Therapy 2021, 12, 603. [Google Scholar] [CrossRef]
- Alvarez-Calderon, F.; Gregory, M.A.; Pham-Danis, C.; DeRyckere, D.; Stevens, B.M.; Zaberezhnyy, V.; Hill, A.A.; Gemta, L.; Kumar, A.; Kumar, V.; et al. Tyrosine kinase inhibition in leukemia induces an altered metabolic state sensitive to mitochondrial perturbations. Clin Cancer Res 2015, 21, 1360–1372. [Google Scholar] [CrossRef]
- Pellicano, F.; Scott, M.T.; Helgason, G.V.; Hopcroft, L.E.; Allan, E.K.; Aspinall-O'Dea, M.; Copland, M.; Pierce, A.; Huntly, B.J.; Whetton, A.D.; et al. The antiproliferative activity of kinase inhibitors in chronic myeloid leukemia cells is mediated by FOXO transcription factors. Stem Cells 2014, 32, 2324–2337. [Google Scholar] [CrossRef]
- Zhang, B.; Nguyen, L.X.T.; Li, L.; Zhao, D.; Kumar, B.; Wu, H.; Lin, A.; Pellicano, F.; Hopcroft, L.; Su, Y.L.; et al. Bone marrow niche trafficking of miR-126 controls the self-renewal of leukemia stem cells in chronic myelogenous leukemia. Nat Med 2018, 24, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Salati, S.; Salvestrini, V.; Carretta, C.; Genovese, E.; Rontauroli, S.; Zini, R.; Rossi, C.; Ruberti, S.; Bianchi, E.; Barbieri, G.; et al. Deregulated expression of miR-29a-3p, miR-494-3p and miR-660-5p affects sensitivity to tyrosine kinase inhibitors in CML leukemic stem cells. Oncotarget 2017, 8, 49451–49469. [Google Scholar] [CrossRef] [PubMed]
- Yung, Y.; Lee, E.; Chu, H.T.; Yip, P.K.; Gill, H. Targeting Abnormal Hematopoietic Stem Cells in Chronic Myeloid Leukemia and Philadelphia Chromosome-Negative Classical Myeloproliferative Neoplasms. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Bernt, K.M.; Armstrong, S.A. Leukemia stem cells and human acute lymphoblastic leukemia. Semin Hematol 2009, 46, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Cox, C.V.; Evely, R.S.; Oakhill, A.; Pamphilon, D.H.; Goulden, N.J.; Blair, A. Characterization of acute lymphoblastic leukemia progenitor cells. Blood 2004, 104, 2919–2925. [Google Scholar] [CrossRef]
- le Viseur, C.; Hotfilder, M.; Bomken, S.; Wilson, K.; Röttgers, S.; Schrauder, A.; Rosemann, A.; Irving, J.; Stam, R.W.; Shultz, L.D.; et al. In childhood acute lymphoblastic leukemia, blasts at different stages of immunophenotypic maturation have stem cell properties. Cancer Cell 2008, 14, 47–58. [Google Scholar] [CrossRef]
- Castor, A.; Nilsson, L.; Astrand-Grundström, I.; Buitenhuis, M.; Ramirez, C.; Anderson, K.; Strömbeck, B.; Garwicz, S.; Békássy, A.N.; Schmiegelow, K.; et al. Distinct patterns of hematopoietic stem cell involvement in acute lymphoblastic leukemia. Nat Med 2005, 11, 630–637. [Google Scholar] [CrossRef]
- Chiu, P.P.L.; Jiang, H.; Dick, J.E. Leukemia-initiating cells in human T-lymphoblastic leukemia exhibit glucocorticoid resistance. Blood 2010, 116, 5268–5279. [Google Scholar] [CrossRef]
- Johnsen, H.E.; Bøgsted, M.; Schmitz, A.; Bødker, J.S.; El-Galaly, T.C.; Johansen, P.; Valent, P.; Zojer, N.; Van Valckenborgh, E.; Vanderkerken, K.; et al. The myeloma stem cell concept, revisited: from phenomenology to operational terms. Haematologica 2016, 101, 1451–1459. [Google Scholar] [CrossRef]
- Basak, G.W.; Carrier, E. The search for multiple myeloma stem cells: the long and winding road. Biol Blood Marrow Transplant 2010, 16, 587–594. [Google Scholar] [CrossRef]
- Huff, C.A.; Matsui, W. Multiple myeloma cancer stem cells. J Clin Oncol 2008, 26, 2895–2900. [Google Scholar] [CrossRef]
- Gunes, E.G.; Gunes, M.; Yu, J.; Janakiram, M. Targeting cancer stem cells in multiple myeloma. Trends in Cancer 2024, 10, 733–748. [Google Scholar] [CrossRef]
- Xu, S.; De Veirman, K.; De Becker, A.; Vanderkerken, K.; Van Riet, I. Mesenchymal stem cells in multiple myeloma: a therapeutical tool or target? Leukemia 2018, 32, 1500–1514. [Google Scholar] [CrossRef] [PubMed]
- Terwijn, M.; Zeijlemaker, W.; Kelder, A.; Rutten, A.P.; Snel, A.N.; Scholten, W.J.; Pabst, T.; Verhoef, G.; Löwenberg, B.; Zweegman, S.; et al. Leukemic Stem Cell Frequency: A Strong Biomarker for Clinical Outcome in Acute Myeloid Leukemia. PLOS ONE 2014, 9, e107587. [Google Scholar] [CrossRef] [PubMed]
- Canali, A.; Vergnolle, I.; Bertoli, S.; Largeaud, L.; Nicolau, M.L.; Rieu, J.B.; Tavitian, S.; Huguet, F.; Picard, M.; Bories, P.; et al. Prognostic Impact of Unsupervised Early Assessment of Bulk and Leukemic Stem Cell Measurable Residual Disease in Acute Myeloid Leukemia. Clin Cancer Res 2023, 29, 134–142. [Google Scholar] [CrossRef]
- Vergez, F.; Nicolau-Travers, M.L.; Bertoli, S.; Rieu, J.B.; Tavitian, S.; Bories, P.; Luquet, I.; Mas, V.; Largeaud, L.; Sarry, A.; et al. CD34(+)CD38(-)CD123(+) Leukemic Stem Cell Frequency Predicts Outcome in Older Acute Myeloid Leukemia Patients Treated by Intensive Chemotherapy but Not Hypomethylating Agents. Cancers (Basel) 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Darwish, N.H.; Sudha, T.; Godugu, K.; Elbaz, O.; Abdelghaffar, H.A.; Hassan, E.E.; Mousa, S.A. Acute myeloid leukemia stem cell markers in prognosis and targeted therapy: potential impact of BMI-1, TIM-3 and CLL-1. Oncotarget 2016, 7, 57811–57820. [Google Scholar] [CrossRef] [PubMed]
- Yabushita, T.; Satake, H.; Maruoka, H.; Morita, M.; Katoh, D.; Shimomura, Y.; Yoshioka, S.; Morimoto, T.; Ishikawa, T. Expression of multiple leukemic stem cell markers is associated with poor prognosis in de novo acute myeloid leukemia. Leukemia & Lymphoma 2018, 59, 2144–2151. [Google Scholar] [CrossRef]
- Mohamed, M.M.I.; Aref, S.; Agdar, M.A.; Mabed, M.; El-Sokkary, A.M.A. Leukemic Stem Cell (CD34+/CD38–/TIM3+) Frequency in Patients with Acute Myeloid Leukemia: Clinical Implications. Clinical Lymphoma, Myeloma and Leukemia 2021, 21, 508–513. [Google Scholar] [CrossRef]
- Huang, W.; Zheng, S.; Wang, Q.; Zhao, N.; Long, Z. Identification and validation of a prognostic risk-scoring model based on the level of TIM-3 expression in acute myeloid leukemia. Scientific Reports 2023, 13, 15658. [Google Scholar] [CrossRef]
- Wu, Y.; Li, Y.; Gao, Y.; Zhang, P.; Jing, Q.; Zhang, Y.; Jin, W.; Wang, Y.; Du, J.; Wu, G. Immunotherapies of acute myeloid leukemia: Rationale, clinical evidence and perspective. Biomed Pharmacother 2024, 171, 116132. [Google Scholar] [CrossRef]
- Ebinama, U.; George, B. Revolutionizing acute myeloid leukemia treatment: a systematic review of immune-based therapies. Discov Oncol 2025, 16, 89. [Google Scholar] [CrossRef]
- Bruzzese, A.; Martino, E.A.; Labanca, C.; Mendicino, F.; Lucia, E.; Olivito, V.; Neri, A.; Imovilli, A.; Morabito, F.; Vigna, E.; et al. Tagraxofusp in myeloid malignancies. Hematological Oncology 2024, 42, e3234. [Google Scholar] [CrossRef] [PubMed]
- Kubicka, E.; Lum, L.G.; Huang, M.; Thakur, A. Bispecific antibody-targeted T-cell therapy for acute myeloid leukemia. Front Immunol 2022, 13, 899468. [Google Scholar] [CrossRef]
- Uy, G.L.; Aldoss, I.; Foster, M.C.; Sayre, P.H.; Wieduwilt, M.J.; Advani, A.S.; Godwin, J.E.; Arellano, M.L.; Sweet, K.L.; Emadi, A.; et al. Flotetuzumab as salvage immunotherapy for refractory acute myeloid leukemia. Blood 2021, 137, 751–762. [Google Scholar] [CrossRef]
- Damiani, D.; Tiribelli, M. Checkpoint Inhibitors in Acute Myeloid Leukemia. Biomedicines 2023, 11, 1724. [Google Scholar] [CrossRef] [PubMed]
- Michelozzi, I.M.; Kirtsios, E.; Giustacchini, A. Driving CAR T Stem Cell Targeting in Acute Myeloid Leukemia: The Roads to Success. Cancers 2021, 13, 2816. [Google Scholar] [CrossRef]
- Yokoyama, A.; Somervaille, T.C.; Smith, K.S.; Rozenblatt-Rosen, O.; Meyerson, M.; Cleary, M.L. The menin tumor suppressor protein is an essential oncogenic cofactor for MLL-associated leukemogenesis. Cell 2005, 123, 207–218. [Google Scholar] [CrossRef]
- Argiropoulos, B.; Humphries, R.K. Hox genes in hematopoiesis and leukemogenesis. Oncogene 2007, 26, 6766–6776. [Google Scholar] [CrossRef]
- Borkin, D.; He, S.; Miao, H.; Kempinska, K.; Pollock, J.; Chase, J.; Purohit, T.; Malik, B.; Zhao, T.; Wang, J.; et al. Pharmacologic inhibition of the Menin-MLL interaction blocks progression of MLL leukemia in vivo. Cancer Cell 2015, 27, 589–602. [Google Scholar] [CrossRef]
- Kuhn, M.W.; Song, E.; Feng, Z.; Sinha, A.; Chen, C.W.; Deshpande, A.J.; Cusan, M.; Farnoud, N.; Mupo, A.; Grove, C.; et al. Targeting Chromatin Regulators Inhibits Leukemogenic Gene Expression in NPM1 Mutant Leukemia. Cancer Discov 2016, 6, 1166–1181. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, L.; Gundry, M.C.; Sorcini, D.; Guzman, A.G.; Huang, Y.H.; Ramabadran, R.; Gionfriddo, I.; Mezzasoma, F.; Milano, F.; Nabet, B.; et al. Mutant NPM1 Maintains the Leukemic State through HOX Expression. Cancer Cell 2018, 34, 499–512 e499. [Google Scholar] [CrossRef] [PubMed]
- (FDA); U.S.F.a.D.A. FDA approves revumenib for relapsed or refractory acute leukemia with a KMT2A. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-revumenib-relapsed-or-refractory-acute-leukemia-kmt2a-translocation (accessed on 27 Dec 2025).
- (FDA); U.S.F.a.D.A. FDA approves revumenib for relapsed or refractory acute myeloid leukemia with a susceptible NPM1 mutation. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-revumenib-relapsed-or-refractory-acute-myeloid-leukemia-susceptible-npm1-mutation (accessed on 27 December 2025).
- Administration; U.S.F.a.D. FDA approves ziftomenib for relapsed or refractory acute myeloid leukemia with a NPM1 mutation. Available online: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-ziftomenib-relapsed-or-refractory-acute-myeloid-leukemia-npm1-mutation (accessed on 27 December 2025).
- Nadiminti, K.V.G.; Sahasrabudhe, K.D.; Liu, H. Menin inhibitors for the treatment of acute myeloid leukemia: challenges and opportunities ahead. Journal of Hematology & Oncology 2024, 17, 113. [Google Scholar] [CrossRef]
- Dali, S.A.; Al-Mashdali, A.F.; Kalfah, A.; Mohamed, S.F. Menin Inhibitors in KMT2A-Rearranged and NPM1-Mutated Acute Leukemia: A Scoping Review of Safety and Efficacy. Critical Reviews in Oncology/Hematology 2025, 104783. [Google Scholar] [CrossRef] [PubMed]
- Candoni, A.; Coppola, G. A 2024 Update on Menin Inhibitors. A New Class of Target Agents against KMT2A-Rearranged and NPM1-Mutated Acute Myeloid Leukemia. Hematology Reports 2024, 16, 244–254. [Google Scholar] [CrossRef]
- Alexandrova, E.; Salvati, A.; Pecoraro, G.; Lamberti, J.; Melone, V.; Sellitto, A.; Rizzo, F.; Giurato, G.; Tarallo, R.; Nassa, G.; et al. Histone Methyltransferase DOT1L as a Promising Epigenetic Target for Treatment of Solid Tumors. Frontiers in Genetics 2022, Volume 13 - 2022. [Google Scholar] [CrossRef]
- Menghrajani, K.; Cai, S.F.; Devlin, S.M.; Armstrong, S.A.; Piekarz, R.; Rudek, M.; Tallman, M.S.; Stein, E.M. A Phase Ib/II Study of the Histone Methyltransferase Inhibitor Pinometostat in Combination with Azacitidine in Patients with 11q23-Rearranged Acute Myeloid Leukemia. Blood 2019, 134, 2655. [Google Scholar] [CrossRef]
- Nwosu, G.O.; Ross, D.M.; Powell, J.A.; Pitson, S.M. Venetoclax therapy and emerging resistance mechanisms in acute myeloid leukaemia. Cell Death & Disease 2024, 15, 413. [Google Scholar] [CrossRef]
- Murphy, L.A.; Winters, A.C. Emerging and Future Targeted Therapies for Pediatric Acute Myeloid Leukemia: Targeting the Leukemia Stem Cells. Biomedicines 2023, 11, 3248. [Google Scholar] [CrossRef]
- Wei, A.H.; Roberts, A.W. BCL2 Inhibition: A New Paradigm for the Treatment of AML and Beyond. Hemasphere 2023, 7, e912. [Google Scholar] [CrossRef]
- Zugasti, I.; Espinosa-Aroca, L.; Fidyt, K.; Mulens-Arias, V.; Diaz-Beya, M.; Juan, M.; Urbano-Ispizua, A.; Esteve, J.; Velasco-Hernandez, T.; Menendez, P. CAR-T cell therapy for cancer: current challenges and future directions. Signal Transduct Target Ther 2025, 10, 210. [Google Scholar] [CrossRef]
- Buldini, B.; Varotto, E. Moving forward in target antigen discovery for immunotherapy in acute myeloid leukemia. Haematologica 2024, 109, 3088–3090. [Google Scholar] [CrossRef] [PubMed]
- Dreyzin, A.; Holtzman, N.G.; Bonifant, C.L. CD123-targeting immunotherapeutic approaches in acute myeloid leukaemia. Br J Haematol 2025, 207, 1178–1191. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, K.; Deshpande, A.J. Therapeutic targeting of leukemia stem cells in acute myeloid leukemia. Front Oncol 2023, 13, 1204895. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Wang, T.; Wei, J. Strategic innovations: Tackling challenges of immunotherapy in acute myeloid leukemia. Chin J Cancer Res 2025, 37, 490–504. [Google Scholar] [CrossRef]
- Zeidner, J.F.; Sallman, D.A.; Recher, C.; Daver, N.G.; Leung, A.Y.H.; Hiwase, D.K.; Subklewe, M.; Pabst, T.; Montesinos, P.; Larson, R.A.; et al. Magrolimab plus azacitidine vs physician's choice for untreated TP53-mutated acute myeloid leukemia: the ENHANCE-2 study. Blood 2025, 146, 590–600. [Google Scholar] [CrossRef]
- Bonnet, D.; Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997, 3, 730–737. [Google Scholar] [CrossRef]
- Ishikawa, F.; Yoshida, S.; Saito, Y.; Hijikata, A.; Kitamura, H.; Tanaka, S.; Nakamura, R.; Tanaka, T.; Tomiyama, H.; Saito, N.; et al. Chemotherapy-resistant human AML stem cells home to and engraft within the bone-marrow endosteal region. Nat Biotechnol 2007, 25, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Lagadinou, E.D.; Sach, A.; Callahan, K.; Rossi, R.M.; Neering, S.J.; Minhajuddin, M.; Ashton, J.M.; Pei, S.; Grose, V.; O'Dwyer, K.M.; et al. BCL-2 inhibition targets oxidative phosphorylation and selectively eradicates quiescent human leukemia stem cells. Cell Stem Cell 2013, 12, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Kamel, A.M.; Elsharkawy, N.M.; Kandeel, E.Z.; Hanafi, M.; Samra, M.; Osman, R.A. Leukemia Stem Cell Frequency at Diagnosis Correlates With Measurable/Minimal Residual Disease and Impacts Survival in Adult Acute Myeloid Leukemia. Front Oncol 2022, 12, 867684. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan Rajsri, K.; Roy, N.; Chakraborty, S. Acute Myeloid Leukemia Stem Cells in Minimal/Measurable Residual Disease Detection. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Trumpp, A.; Essers, M.; Wilson, A. Awakening dormant haematopoietic stem cells. Nat Rev Immunol 2010, 10, 201–209. [Google Scholar] [CrossRef]
- Saito, Y.; Uchida, N.; Tanaka, S.; Suzuki, N.; Tomizawa-Murasawa, M.; Sone, A.; Najima, Y.; Takagi, S.; Aoki, Y.; Wake, A.; et al. Induction of cell cycle entry eliminates human leukemia stem cells in a mouse model of AML. Nat Biotechnol 2010, 28, 275–280. [Google Scholar] [CrossRef]
|
Marker Category |
Marker |
Expression on LSC |
Expression on HSC |
Distinction Role & Clinical Relevance |
|
Backbone & Stemness |
CD34 | + (variable) |
++ | Present in both LSCs and HSCs, requires additional markers for specificity, starting marker for gating |
| CD38 | - | + | LSCs are CD34⁺CD38⁻, while HSCs express CD38 | |
| CD90 (Thy-1) | - | + | Critical negative marker. Distinguishes LSCs from normal HSCs (CD90+). Essential marker for LSC-MRD detection | |
| CD45RA | +/- | - | Key aberrant marker; helps distinguish LSCs from normal HSCs, enriched in certain AML subtypes | |
|
Therapeutic targets |
CD123 (IL-3Rα) |
++ | dim | Overexpressed in LSCs and target of tagraxofusp and CD123-CAR-T cells |
| CLL-1 (CLEC12A) | + | - | Absent in HSCs. Ideal target for CAR-T cell therapy | |
| TIM-3 | ++ | - | Enriched in LSCs. Targeted by TIM-3 inhibitors (Sabatolimab) | |
|
Immune evasion |
CD200 |
++ |
+ |
“Don’t kill me” signal. Suppresses T-cell & NK-cell activity and helps LSCs evade immune responses. Associated with poor prognosis. |
| CD47 | ++ | + | "Don't eat me" signal. Overexpressed in LSCs, allowing them to evade immune clearance. Target of magrolimab. High expression links to venetoclax resistance |
|
| Other markers | CD96 | +/- | dim | Higher in LSCs, implicated in adhesion and survival |
| CD117 (c-KIT) | +/- | + | Expressed in some LSC populations | |
| CD44 | ++ | + | Crucial for LSC interaction with BM niche | |
| CD7 | + | - | Aberrant expression in AML, linked to poor prognosis and therapy resistance | |
| CD70 | ++ | - | Supports LSC survival via immune modulation | |
| CD9 | ++ | +/- | Associated with drug resistance and stemness in LSCs | |
| HLA-DR | +/- | + | Associated with poor prognosis in HLA-DR negative AML subtypes | |
| CD99 | ++ | dim | LSC adhesion, migration, and potential immune evasion | |
| IL1RAP | ++ | - | Potential therapeutic target | |
| CD244 | + | +/- | Immune evasion and survival of LSCs. Potential therapeutic target | |
| CD56 | ++ | +/- | Promotes leukemogenesis. Potential therapeutic target |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




