1. Introduction
Intraventricular tumors represent a minority in the field of brain neoplasms, especially meningiomas (less than 4% of all meningiomas in average) [
1], but their treatment is particularly complex, due to the fine vascularization and their deep location. In clinical practice there has been an evolution towards minimally invasive techniques with the aim to preserve the neurological function: an example of this concept is the technique proposed by Yasargil regarding the use of trans cisternal trajectories rather than the usual transcortical surgical corridors [
2]. The characteristics of these tumors, when located deep in the brain, make them ideal candidates for the resection using a tubular retractor. This procedure grants direct access to the tumoral mass and the possibility to minimize the injury to the surrounding brain tissue [
3]. In addition, it represents a versatile technique which adapts to both the microscopic and the endoscopic instrumentation. Intraventricular meningiomas originate from the choroid plexus’ stroma: arachnoid cells here are found secondary to the embryologic origin of the choroid plexus [
4]. Jamshidi et al. and Lin et al. published the first reported series of intraventricular meningiomas and to date, to the best of our knowledge, most of the literature regarding the use of tubular retractors in intraventricular tumors is still based on individual case reports [
1,
2,
3,
4,
5]. The aim of this study is to describe the surgical technique and present our experience in 11 cases of intraventricular meningiomas managed through the use of tubular retractors.
2. Materials and Methods
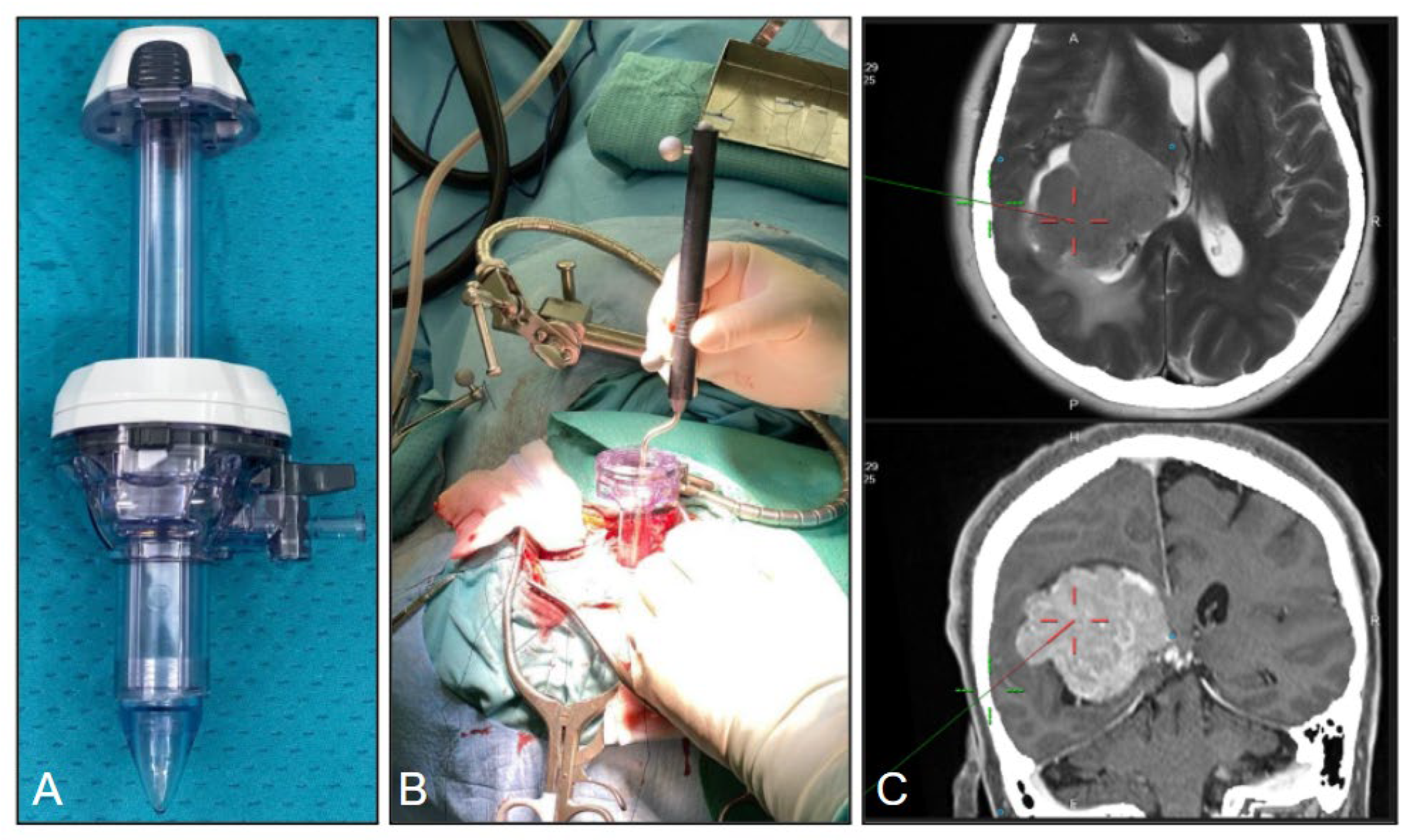
A retrospective 1-year multi-institution study (from January 2022 to December 2022) was conducted, including a series of 11 patients with intraventricular meningioma operated by the use of the tubular retractor. Surgical resection was performed with the assistance of the neuronavigation system, microscope, neuroendoscope, selective ultrasonic aspirator and tubular retractors. EVD was not used routinely. Surgical resection was assisted with a Leica surgical microscope (Leica Microsystems, Wetzlar, Germany) and a Stryker (Kalamazoo, Michigan, USA) rigid endoscope with a 0-degree optic × 2 mm in diameter × 12 cm in length. The tubular retractor system was obtained by the general surgery equipment and secured to the skull clamp with a Greenberg flexible arm: ENDOPATH XCEL 15 mm trocar (Ethicon, Johnson & Johnson MedTech, New Jersey USA) [
Figure 1] or, in a minority of cases (n=3) by the NICO System BrainPath (NICO Corporation, Indianapolis USA).
We describe the technique regarding the excision of the tumoral mass using the tubular retractor. Patient’s demographics, onset and follow up, clinical data (symptoms and neurological status), operative and postoperative details, histopathological diagnosis, and radiological imaging, including a study of the depth of the tumor, were analyzed to define outcome.
2.1. Perioperative Management
All patients underwent magnetic resonance imaging with and without contrast, in the majority of cases (n=8) supplemented by tractography, along with the neuronavigation protocol, to determine the surgical trajectory and tumor detailed position (BrainLAB neuronavigation system, Munich, Germany). Measurements of the tumor were made on the contrast-enhanced T1-weighted MRI sequences, as well as its depth from the entry point on the cortical surface. The anatomic location of the tumor and its dimensions were determined, in order to choose a patient-tailored surgical trajectory. An immediate CT scan and a follow-up MRI was carried out after 3 months and 1 year from surgery.
2.2. Operative Technique
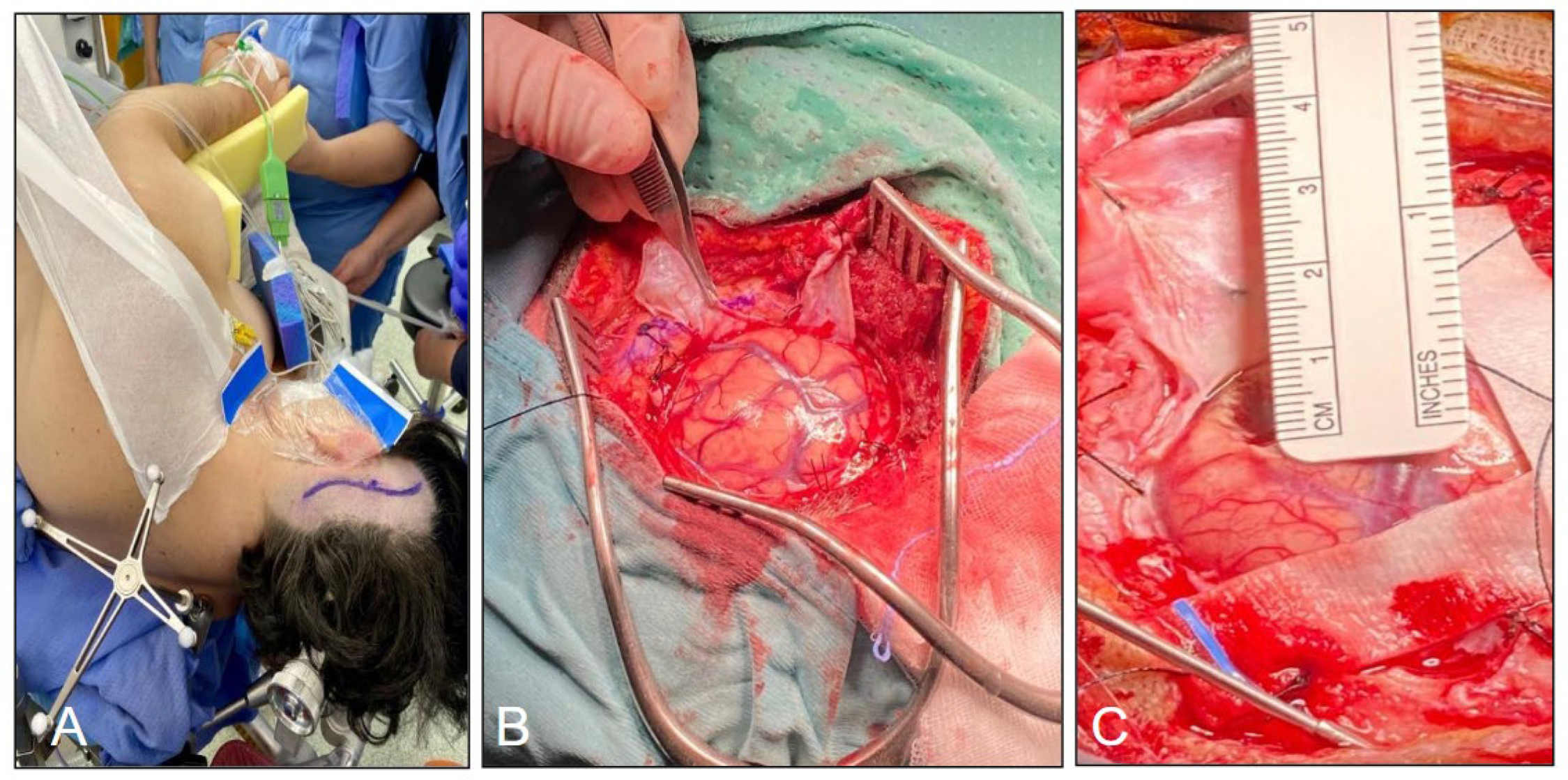
After general anesthesia, we adopted for the patients a supine, lateral or prone position according to the location of the tumor and the tailored approach. The Mayfield (Ohio Medical Instrument Co., Cincinnati, Ohio, USA) skull clamp was used and a surface neuronavigation registration was performed with the BrainLAB system. A linear or lazy S incision, in association with a round 3-4 cm craniotomy, was planned in all cases. Durotomy was preferably performed in a x-shaped fashion or in a semilunar manner [
Figure 2].
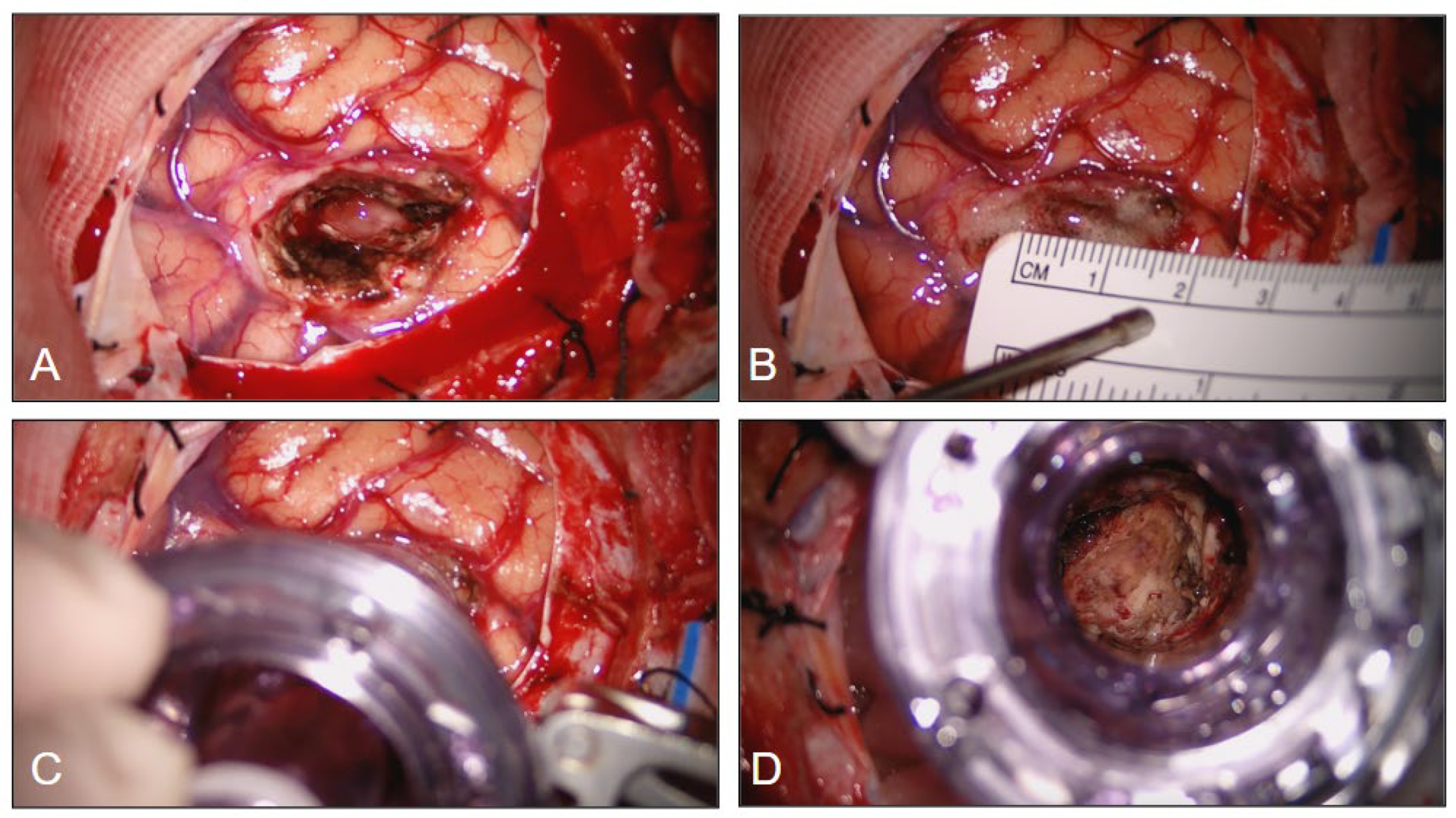
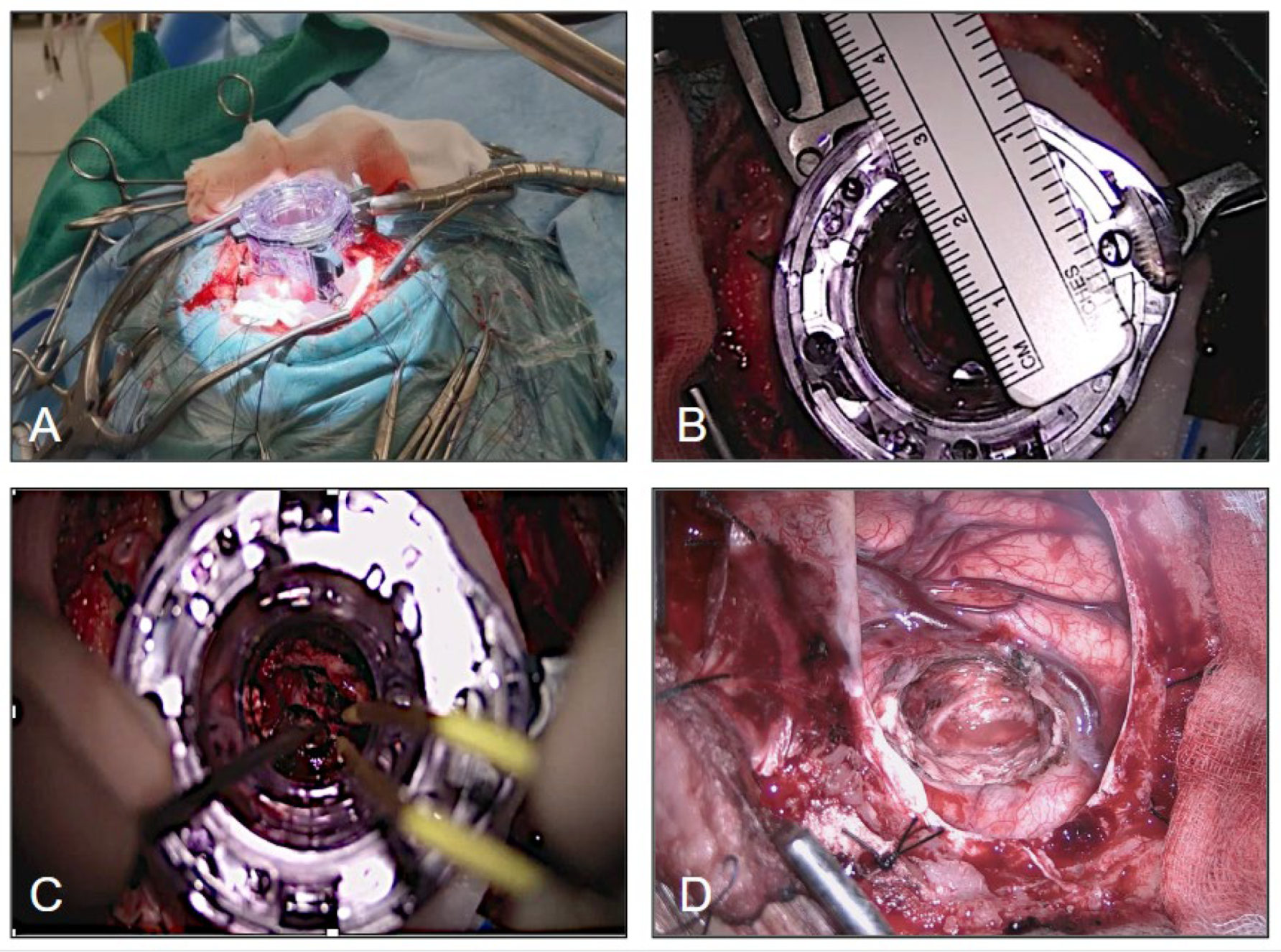
A transcortical approach was used in our series and, after identifying the reference gyrus and sulci, a trans-sulcal approach or a corticectomy no larger than 15-20 mm was performed. The trocar tubular retractor was introduced (the working lumen on top, once the blunt introducer is removed, is about 20 mm), perpendicular to the cortical surface, and inserted gently and in rotation into the brain tissue, always guided by the neuronavigation probe inside the port. These maneuvers bluntly spread the white matter fibers, permit a dynamic retraction and minimize brain tissue disruption, avoiding high and prolonged brain retraction pressure as an effect of the 360 degrees force distribution. Once the tumor is reached, the blunt introducer, used for the blunt dissection of the white matter tracts, is removed creating the direct angle working space. The working angle can eventually be modified during surgery, to explore all the possible tumor perimeter. A Greenberg flexible arm is then secured to the skull clamp and attached to the trocar system [
Figure 3 and
Figure 4].
The choice of the port diameter is influenced by the diameter of the lesion and by the preoperative radiological measurements. We performed the tumor resection by means of microscope and endoscope. The tumor was debulked using the ultrasonic selective aspirator and angulation of the tubular retractor in multiple directions. Once tumor resection and hemostasis were achieved, the tubular retractor was slowly removed, in a rotational way, as it was inserted. In none of the cases an EVD (external ventricular drain) was placed routinely before the procedure [
Figure 4]. The study was conducted in accordance with the Declaration of Helsinki and Ethical approval was waived by our local ethics committee in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
3. Results
3.1. Patient Overview
During 2022, 11 patients (5 males and 6 female), with an overall mean age of 72.6 years (range 56–83), were operated for an intraventricular meningioma by means of the tubular retractor system. In 4 patients the tumor was located in the trigonal area of the right lateral ventricle, in 5 patients, the tumor was in the left trigonal area of the ventricle, in 2 patients in the region of the foramen of Monro. In all patients the resection was gross total. In no case, before or after the operation, was it necessary to use an EVD. None of the patients, except one patient with postoperative right-side hemiplegia probably caused by venous infarction, experienced permanent neurological deficits. Four patients presented transitory post-operative agitation, visual blurring and transient hemiparesis. There were less than 10% of permanent major complications related to surgery (one patient with postoperative right-side hemiplegia probably caused by venous infarction) and no mortalities. Surgical procedures were performed with microsurgical vision and neuroendoscopic assistance in all cases. Mean tumor depth was 1.72 cm and the average preoperative tumor volume for the series was 45.1 cm3; tumor types included: N=9 WHO grade I and N=2 grade II meningiomas [
Table 1].
3.2. Illustrative Cases
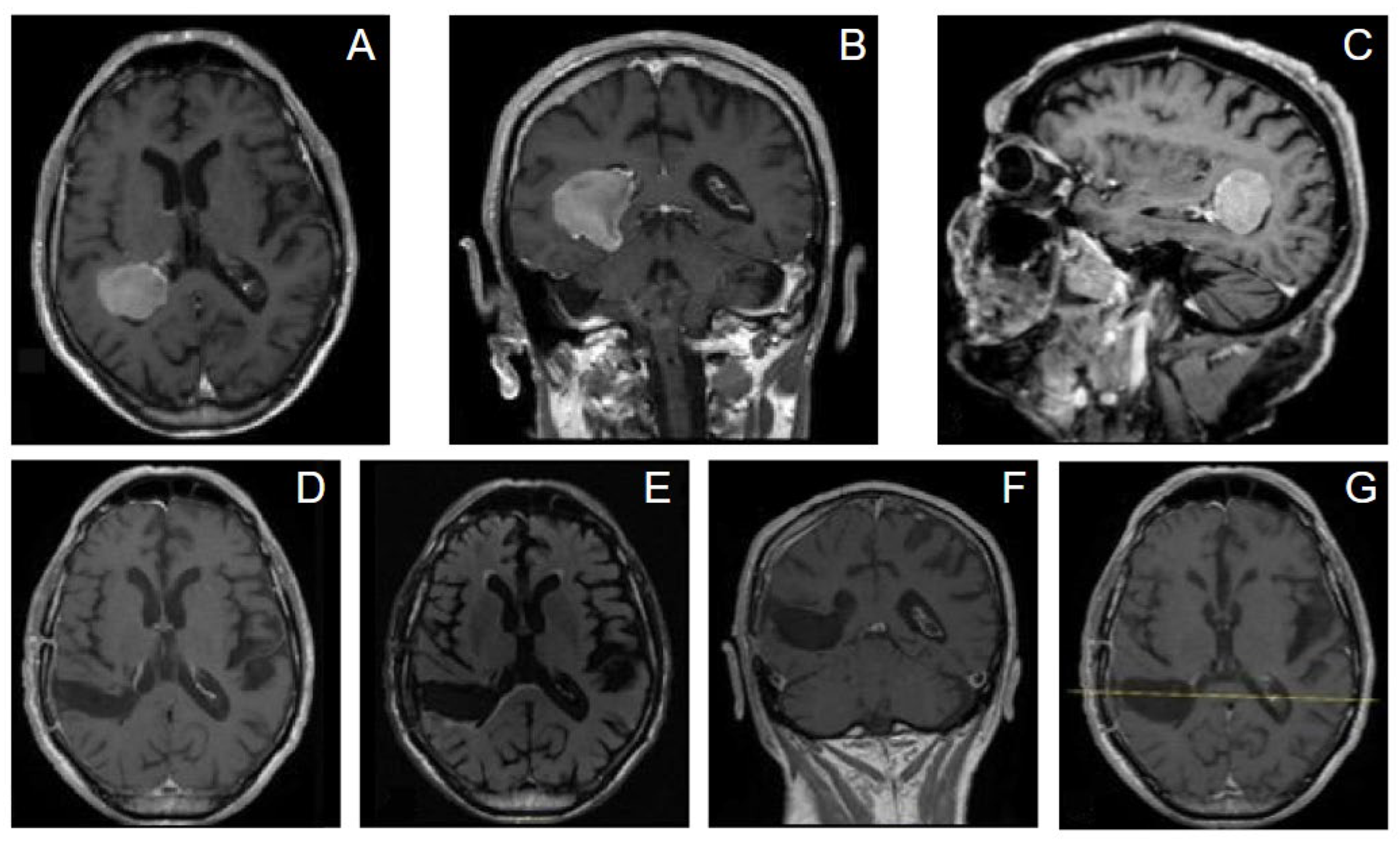
Case 1. An 83-year-old man presented with a history of loss of consciousness episodes and ideomotor slowdown. He was otherwise neurologically intact. MRI revealed a large right-sided intraventricular mass in the trigone of the right lateral ventricle consistent with meningioma. We adopted for the patient a supine position and a minimally invasive tumor excision was performed using the tubular retractor system assisted by neuronavigation. Gross total resection (GTR) was achieved and pathology confirmed WHO grade 1 meningioma. He had an uneventful recovery and remained neurologically intact after surgery. At 3 months from surgery there is no evidence of tumor recurrence, no evidence of brain injury or progressive neurological deficits [
Figure 5].
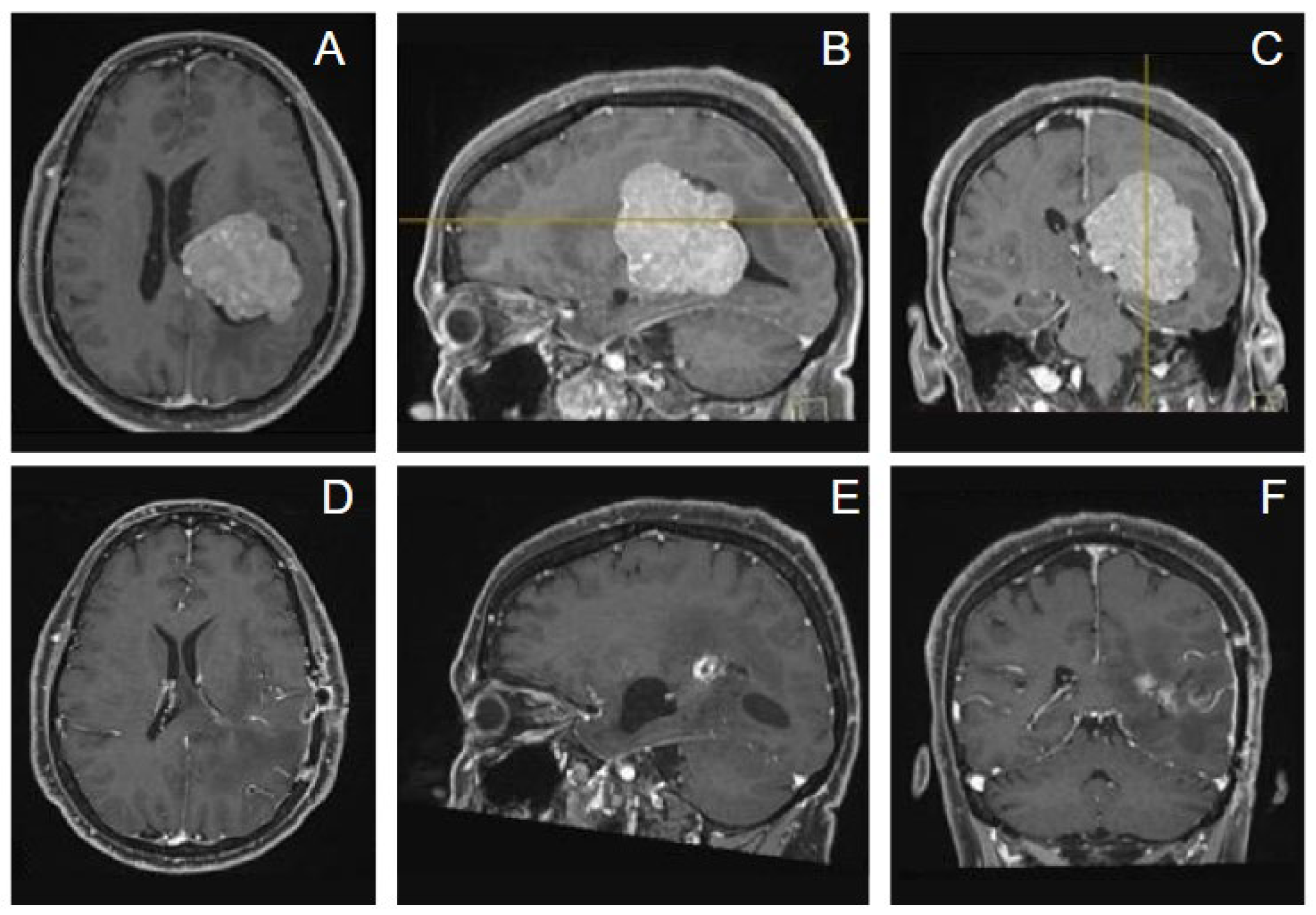
Case 2. A 56-year-old woman presented with memory impairment, dysarthria and neurological exam revealed hyposthenia at the right limbs. A contrast-enhanced MRI showed a large intraventricular mass in the left trigone with peritrigonal extension consistent with meningioma [
Figure 6]. We adopted for the patient a lateral position and a trans-sulcal temporal approach with port technique, neuronavigation-assisted. Surgery was uneventful and a gross total resection was obtained. Pathology revealed a WHO grade 2 meningioma. Postoperative MRI confirmed complete resection of the tumor. Adjuvant radiation therapy was not recommended due to complete tumor resection, the patient was neurologically intact and followed up.
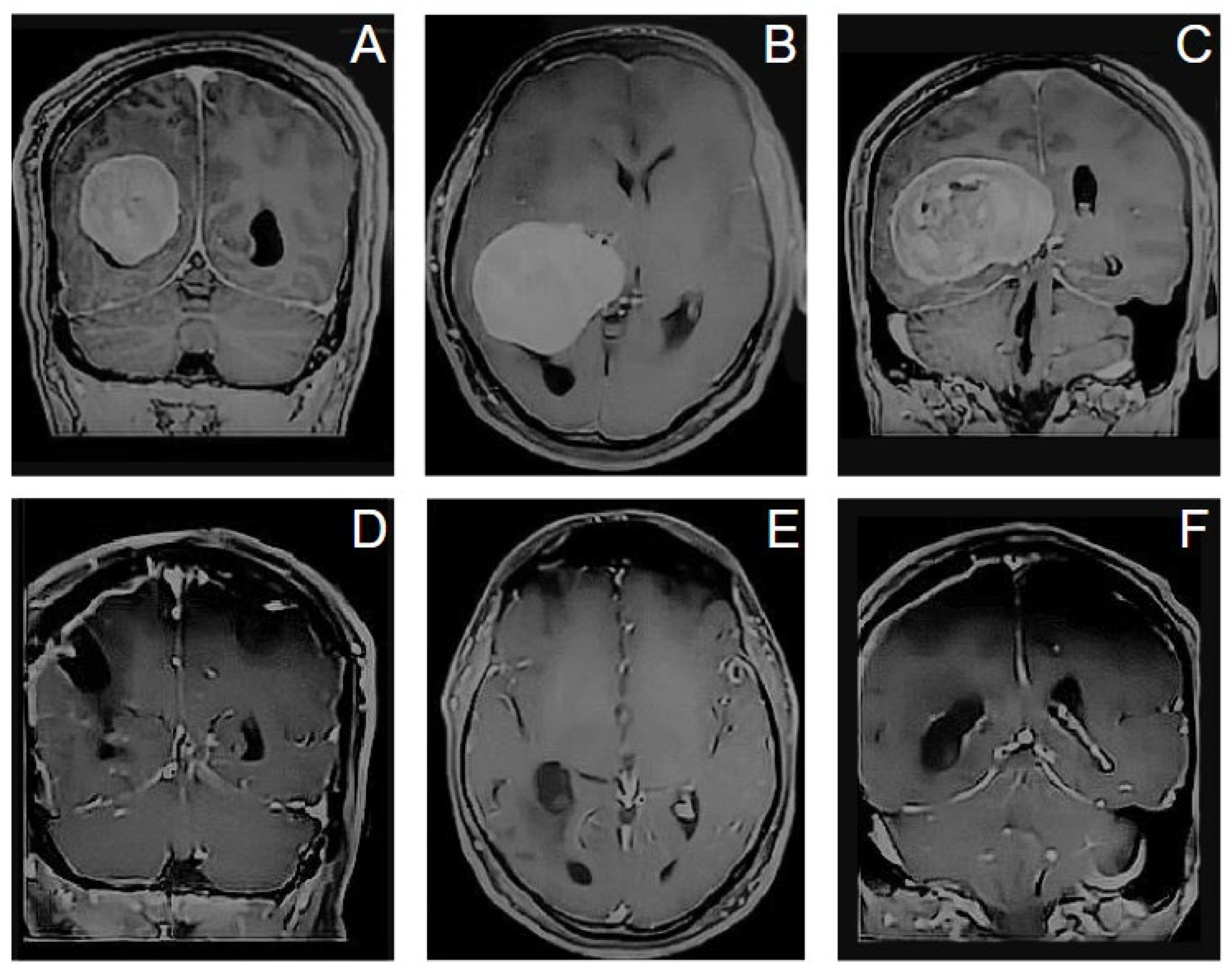
Case 3. A 79-year-old man presented with memory impairment, ideomotor slowdown and left hemitemporal hemianopsia; he was otherwise neurologically intact. Routine magnetic resonance imaging (MRI) revealed a large homogeneously enhancing tumor involving the right ventricular trigone that appeared consistent with a meningioma. He was scheduled for operation, and we adopted for the patient a prone position. Resection was achieved via the posterior aspect of the superior parietal lobule with a neuronavigated port technique. Surgery was uneventful and a total resection was obtained. Pathology revealed a WHO grade I meningioma. He recovered uneventfully and at 1-year postoperative follow-up there was no evidence of tumor recurrence, no evidence of brain injury or progressive neurological deficits [
Figure 7].
4. Discussion
In cerebral intraparenchymal tumors, brain injury related to retraction is a frequent complication with the onset of postoperative seizures, neurological deficits, brain edema and bad clinical outcome [
5,
6,
7]. Minimally invasive techniques, in transcortical approaches, allow to reduce damage to the surrounding brain maintaining good visualization fie these deep-located tumors. In this light the use of circular or tubular retractors, known since 1988 [
8], meets these purposes by dissipating the amount of retraction force in a circumferential manner reducing the cortex under retraction’s amount of force per unit [
5,
8,
9]. The circumferential shape of the tubular port, theoretically, redistributes pressure equally across the surrounding tissue, in a 360◦ dispersion pattern, and produces less direct cutting and tearing trauma to the underlying brain tissue [
10,
11,
12,
13]. In our preliminary experience and in accordance with the literature, it was a safe technique, but there are authors who, in order to avoid a potential complication such as shear injuries, have proposed alternative techniques which are matter of further investigation [
14,
15,
16].
Literature reports the use of different types of tubular retractor systems commercially available: as previously reviewed by Okasha et al., the most frequently mentioned tubular retractor system in the current literature is the ViewSite Brain Access System manufactured by Vycor Medical (Florida, United States) [
5]; other authors used the BrainPath (BrainPath NICO Corporation, Indianapolis, Indiana, USA) [
17], proposed a new mini tubular port like Liu et al. [
10] or modified preexisting equipment like syringes and spinal tubular retractor [
9,
18]. In our case, except few cases where the BrainPath was available, we modified a preexisting general surgery trocar as tubular brain retractor. We used the ENDOPATH XCEL 15 mm trocar: this laparoscopic instrumentation is widespread and well described in the context of general surgery and, according to the literature, it is a safe guide that potentially guarantees a low insertion force [
19]. The technical features of the trocar seem to have met the expectations of reducing tearing trauma to the brain surrounding the lesion, due to the 360◦ pression dispersion pattern. Further studies to confirm our preliminary results are needed.
Deep seated tumors could benefit from a minimally invasive approach with tubular retractors because it provides a small corticectomy and direct path to the lesion; this technique minimizes the brain injury without adhering to the brain [
3]. The transparency of most of the retractors offers a detailed visualization of surrounding tissue and the technique is versatile, using microscope, endoscope and the routine instruments (such as the ultrasonic aspirator) as needed. Often, intraventricular tumors represent a challenge due to the poor visualization of the surgical field: tubular retractors, in addition to less invasive corticectomy and stability on surgical corridors, allow the use of both hands of the surgeon to manage other instruments when needed (microscope, exoscope, ultrasonic aspirator) [
20,
21,
22,
23,
24,
25].
The need for a minimally invasive technique, aimed at improving the outcome and reducing complications, is particularly necessary in elderly patients and reflects a modern concept of neurosurgery: our series included a population with an average age of 72.6 years. In our experience, according to the literature, older age did not seem to be a limit and promising technologies such as tractography or Extend Reality could additionally help [
26,
27,
28].
We believe and prefer, according with the literature [
3], using both the microsurgical technique and the endoscopic assistance in the management of intraventricular tumors. The micro-endoscopic approach, in our preliminary experience, seemed to be the fastest approach related to the greatest possibility of a complete deep tumor resection. The purpose of this study is to describe our preliminary experience with intraventricular meningiomas approached through tubular retractors. As documented in recent studies in the literature; our, even if preliminary, experience seems to confirm that this technique is safe and allows the total removal of intraventricular meningioma.
5. Conclusions
Our preliminary experience suggests that the use of tubular retractors in intraventricular neoplastic diseases, such as meningiomas, is versatile, feasible and safe. The use of this minimally invasive technique allows the reduction of damage to the cerebral parenchyma by displacing the fibers surrounding the tubular retractor in a 360-degree manner. It also made it possible to work, easily, with all the main ordinary surgical instrumentation, the selective ultrasonic aspirator, along with the microscope and the endoscope, through its narrow lumen of a few centimeters.
Author Contributions
Conceptualization, A.I., V.L., M.I; methodology, M.G, C.E.M, A.F., G.L. H.M.; validation, R.T., M.L., A.C., L.D.S., A.M., D.A. and M.C.; formal analysis, A.I., V.L., M.C., D.A.; investigation, A.I., V.L., M.C., D.A; data curation, A.I., V.L., M.C., D.A; writing—original draft preparation, A.I., V.L., M.C., D.A.; writing—review and editing, A.I., V.L., M.C., D.A, M.I., M.G.; visualization, M.I; supervision, M.I. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and Ethical approval was waived by our local ethics committee in view of the retrospective nature of the study and all the procedures being performed were part of the routine care.
Informed Consent Statement
Written informed consent has been obtained from the patients to publish this paper.
Data Availability Statement
The original data presented in this study are available on reasonable request from the corresponding author. The data are not publicly available due to privacy concerns.
Acknowledgments
We would like to thank Stefano Vecchioni from the Department of Neurosurgery. Stefano Bruni, Umberto Russo, Elena Gambelli and Riccardo Gigli from the Department of Neuroradiology. Edoardo Barboni, Maria Elena Bagnarelli, Andrea Vito and Agnese Damia Paciarini from the Department of Neuroanesthesia.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| WHO |
World Health Organization grading system |
| Fig. |
figure |
| MRI |
Magnetic Resonance Imaging |
| CT |
computed tomography |
| Tab. |
table |
| d |
days |
| Y |
years |
| M |
male |
| F |
female |
| R |
retractor used (1: Endopath, 2: NICO) |
| P |
patient |
| A |
age |
| GTR |
gross total removal |
| L |
localization |
| L |
left |
| R |
right |
| PTV |
preoperative tumor volume |
| D |
tumor depth |
| EOR |
extent of resection |
| NB |
neurological status before surgery |
| LOC |
loss of consciousness |
| LOR |
length of retractor |
| COR |
corticectomy dimension |
| NA |
neurological status after surgery |
| LH |
length of hospital stay |
| FU |
months of follow-up |
| LP |
late postoperative complications. |
References
- Jamshidi, AO; Beer-Furlan, A; Hardesty, DA; Ditzel Filho, LFS; Prevedello, LM; Prevedello, DM. Management of large intraventricular meningiomas with minimally invasive port technique: a three-case series. Neurosurg Rev. 2021, 44(4), 2369–2377. [Google Scholar] [CrossRef] [PubMed]
- Yaşargil, MG; Abdulrauf, SI. Surgery of intraventricular tumors. Neurosurgery 2008, 62((6) Suppl 3, 1029-40; discussion 1040-1. [Google Scholar] [CrossRef] [PubMed]
- Chuchuca, AV; Hasing, LM; Wong-Achi, X; Egas, M. Minimally invasive surgery with tubular retractor system for deep-seated or intraventricular brain tumors: Report of 13 cases and technique description. Interdisciplinary Neurosurgery 2021, Volume 25(2021), 101260. [Google Scholar] [CrossRef]
- Mayol Del Valle, M; De Jesus, O. Intraventricular Meningioma. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 12 Feb 2023; Available online: https://www.ncbi.nlm.nih.gov/books/NBK562202/.
- Lin, M; Bakhsheshian, J; Strickland, B; Rennert, RC; Chu, RM; Chaichana, KL; Zada, G. Exoscopic resection of atrial intraventricular meningiomas using a navigation-assisted channel-based trans-sulcal approach: Case series and literature review. J Clin Neurosci;Epub 2020, 71, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Okasha, M; Ineson, G; Pesic-Smith, J; Surash, S. Transcortical Approach to Deep-Seated Intraventricular and Intra-axial Tumors Using a Tubular Retractor System: A Technical Note and Review of the Literature. J Neurol Surg A Cent Eur Neurosurg 2021, 82(3), 270–277. [Google Scholar] [CrossRef] [PubMed]
- Harada, S; Nakamura, T. Retraction induced brain edema. Acta Neurochir Suppl (Wien) 1994, 60, 449–51. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J; Dujovny, M; Perlin, AR; Perez-Arjona, E; Park, HK; Diaz, FG. Brain retraction injury. Neurol Res 2003, 25(8), 831–8. [Google Scholar] [CrossRef] [PubMed]
- Kelly, PJ; Goerss, SJ; Kall, BA. The stereotaxic retractor in computer-assisted stereotaxic microsurgery. Technical note. J Neurosurg 1988, 69(2), 301–6. [Google Scholar] [CrossRef] [PubMed]
- Singh, H; Patir, R; Vaishya, S; Miglani, R; Kaur, A. Syringe Port: A Convenient, Safe, and Cost-Effective Tubular Retractor for Transportal Removal of Deep-Seated Lesions of the Brain. World Neurosurg. 2018, 114, 117–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, X; Qiu, Y; Zhang, F; Wei, X; Zhou, Z; Zhang, F; Xue, Y; Ma, Z; Wang, X; Shen, H; Lin, Z; Shi, H; Liu, L. Combined intra- and extra-endoscopic techniques for endoscopic intraventricular surgery with a new mini-tubular port. Front Surg 2022, 9, 933726. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bander, ED; Jones, SH; Kovanlikaya, I; Schwartz, TH. Utility of tubular retractors to minimize surgical brain injury in the removal of deep intraparenchymal lesions: a quantitative analysis of FLAIR hyperintensity and apparent diffusion coefficient maps. J Neurosurg 2016, 124(4), 1053–60. [Google Scholar] [CrossRef] [PubMed]
- Almenawer, SA; Crevier, L; Murty, N; Kassam, A; Reddy, K. Minimal access to deep intracranial lesions using a serial dilatation technique: case-series and review of brain tubular retractor systems. Neurosurg Rev. 2013, 36(2), 321-9; discussion 329-30. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Gadol, A. Minitubular transcortical microsurgical approach for gross total resection of third ventricular colloid cysts: Technique and assessment. World Neurosurg. 2013, 79(1), 7–10. [Google Scholar] [CrossRef]
- Newman, WC; Engh, JA. Stereotactic-Guided Dilatable Endoscopic Port Surgery for Deep-Seated Brain Tumors: Technical Report with Comparative Case Series Analysis. World Neurosurg;Epub 2019, 125, e812–e819. [Google Scholar] [CrossRef] [PubMed]
- Assina, R.; Rubino, S.; Sarris, C.; Gandhi, C.; Prestigiacomo, C. The history of brain retractors throughout the development of neurological surgery. Neurosurg. Focus 2014, 36(4), 1–12. [Google Scholar] [CrossRef] [PubMed]
- Eichberg, D.; Buttrick, S.; Brusko, D.; Ivan, M.; Starke, R.; Komotar, R. Use of tubular retractor for resection of deep-seated cerebral tumors and colloid cysts: single surgeon experience and review of the literature. World Neurosurg. 2018, 112, e50–e60. [Google Scholar] [CrossRef]
- Klotz, E; Towers, W; Kurtom, K. Minimizing cortical disturbance to access ventricular subependymoma - A novel approach utilizing spinal minimally invasive tubular retractor system. Surg Neurol Int. 2019, 10, 95. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shun, Wu; Wong, Felix. A safe optically guided entry technique using Endopath Xcel Trocars in laparoscopic surgery: A personal series of 821 patients. Gynecology and Minimally Invasive Therapy 2013, Volume 2(Issue 1), Pages 30–33. [Google Scholar] [CrossRef]
- Li, B; Kim, MG; Dominguez, J; Feldstein, E; Kleinman, G; Hanft, S. Intraventricular Choroid Plexus Cavernoma Resection Using Tubular Retractor System and Exoscope Visualization: A Technical Case Report. Oper Neurosurg (Hagerstown) 2022, 22(3), e134–e137. [Google Scholar] [CrossRef] [PubMed]
- Bahar, M; Hashem, H; Tekautz, T; Worley, S; Tang, A; de Blank, P. Choroid plexus tumors in adult and pediatric populations:The Cleveland clinic and university hospitals experience. J Neurooncol 2017, 132, 427–32. [Google Scholar] [CrossRef]
- Eliyas, JK; Glynn, R; Kulwin, CG; Rovin, R; Young, R; Alzate, J. Minimally invasive transsulcal resection of intraventricular and periventricular lesions through a tubular retractor system: Multicentric experience and results. World Neurosurg. 2016, 90, 556–64. [Google Scholar] [CrossRef] [PubMed]
- Gungör, A; Baydin, S; Middlebrooks, EH; Tanriover, N; Isler, C; Rhoton, AL. The white matter tracts of the cerebrum in ventricular surgery and hydrocephalus. J Neurosurg 2017, 126, 945 71. [Google Scholar] [CrossRef] [PubMed]
- Marenco-Hillembrand, L; Prevatt, C; Suarez-Meade, P; Ruiz-Garcia, H; Quinones-Hinojosa, A; Chaichana, KL. Minimally invasive surgical outcomes for deep-seated brain lesions treated with different tubular retraction systems: A systematic review and meta-analysis. World Neurosurg 2020, 143, 537–45.e3. [Google Scholar] [CrossRef]
- Shapiro, SZ; Sabacinski, KA; Mansour, SA; Echeverry, NB; Shah, SS; Stein, AA; et al. Use of Vycor tubular retractors in the management of deep brain lesions: A review of current studies. World Neurosurg 2020, 133, 283–90. [Google Scholar] [CrossRef]
- Iacoangeli, A; Capelli, S; Held, A; Barba, M; Marasi, A; DE Benedictis, A; Iacoangeli, M; Locatelli, D; Fontanella, MM; Giussani, CG; Luglietto, D; Rossi Espagnet, MC; Marras, CE. Pre-surgical planning with extended reality in neurosurgery: a survey-based study describing a preliminary experience in Italy. J Neurosurg Sci Epub ahead of print. 2025. [Google Scholar] [CrossRef] [PubMed]
- Iacoangeli, A; et al. Minimally Invasive Endonasal Endoscopic Approaches in the Grand Elderly. J Neurol Surg B Skull Base 2023, 84, S1–S344. [Google Scholar] [CrossRef]
- da Silva, C.E.; de Freitas, P.E.P. Surgical Removal of Skull Base Meningiomas in Symptomatic Elderly Patients. PMID: 30218795.28842239. World Neurosurg. 2018, 120, 120:e1149–120:e1155. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).