Submitted:
23 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
Identification of Contemporary Needs
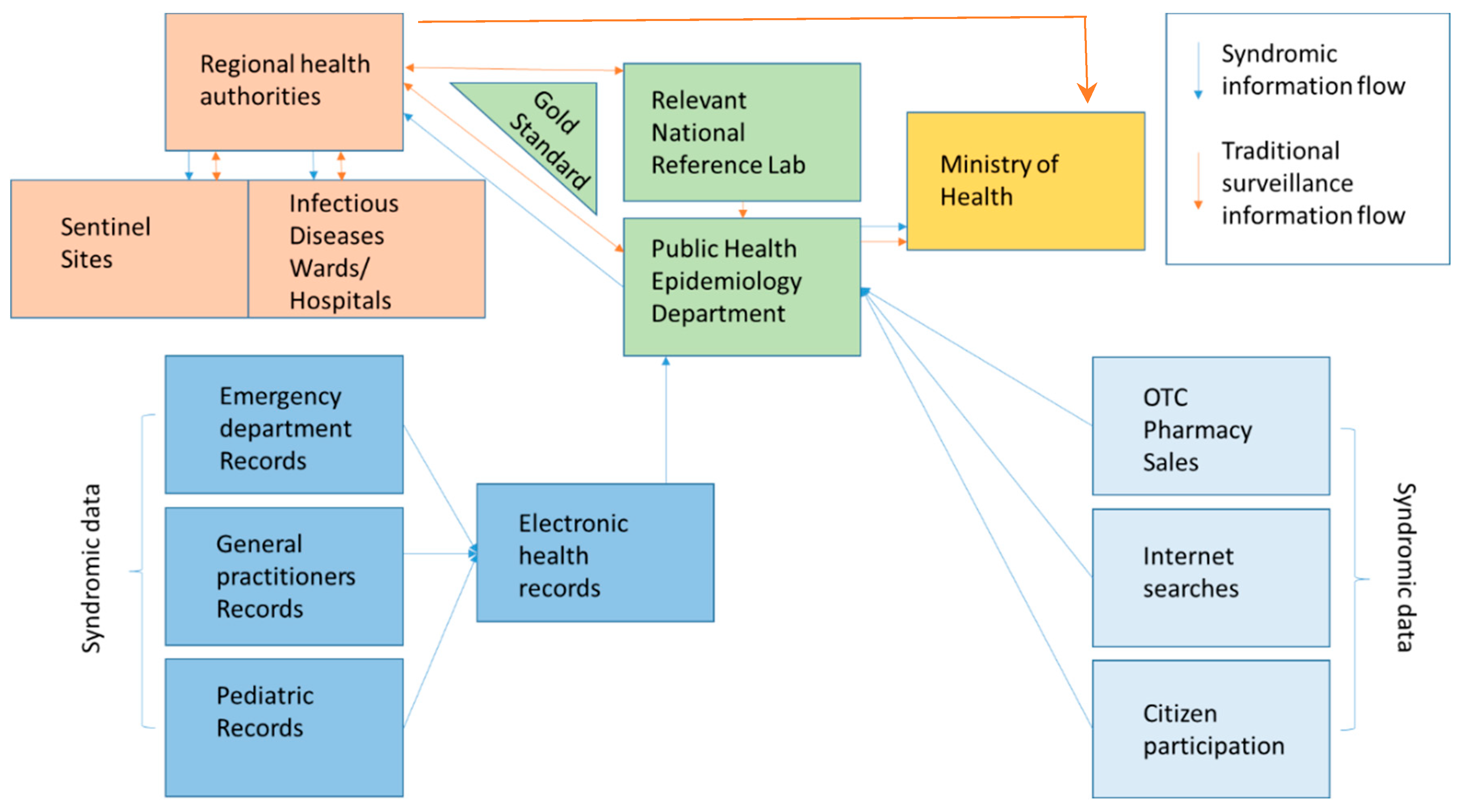
2. Materials and Methods
3. Results and Discussion
3.1. International Experience in Syndromic Surveillance Implementation
Syndromic Surveillance Based on Emergency Department and Hospital Data
3.2. Syndromic Surveillance – Basic Indicators, Approaches and Utility
3.3. Pharmacy and Over-the-Counter (OTC) Sales Data
3.4. Participatory and Citizen-Generated Data
3.5. Other Innovative and Hybrid Models
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thacker, S. B., & Berkelman, R. L. Public health surveillance in the United States. Epidemiologic reviews, 1988; 10(1):164-190.
- World Health Organization. (2008). International health regulations (2005). World Health Organization.
- Milinovich, G. J., Williams, G. M., Clements, A. C., & Hu, W.. Internet-based surveillance systems for monitoring emerging infectious diseases. The Lancet infectious diseases, 2014 Feb; 14(2): 160-168. [CrossRef]
- Djibuti, M., Rukhadze, N., Hotchkiss, D. R., Eisele, T. P., & Silvestre, E. A. Health systems barriers to effective use of infectious disease surveillance data in the context of decentralization in Georgia: A qualitative study. Health Policy, 2007; 83(2-3): 323–331. [CrossRef]
- Xu, L., Zhou, C., Luo, S., Chan, D. K., McLaws, M. L., & Liang, W. Modernising infectious disease surveillance and an early-warning system: the need for China’s action. The Lancet Regional Health–Western Pacific, 2022; 23. [CrossRef]
- Serres K, Erazo D, Despréaux G, Vincenti-González MF, Van Bortel W, Arsevska E, Dellicour S. Integrating indicator-based and event-based surveillance data for risk mapping of West Nile virus, Europe, 2006 to 2021. Euro Surveill. 2024 Oct;29(44):2400084. https://pmc.ncbi.nlm.nih.gov/articles/PMC11528904/. [CrossRef]
- Sala Soler M, Fouillet A, Viso AC, Josseran L, Smith GE, Elliot AJ, et al. Assessment of syndromic surveillance in Europe. Lancet 2011;378:1833–4.
- O.W. Morgan, R.W. Pinner. Surveillance of Infectious Diseases, Editor(s): Moselio Schaechter (2009). Encyclopedia of Microbiology (Third Edition), Academic Press, 759-774.
- Semenza JC, Paz S. Climate change and infectious disease in Europe: Impact, projection and adaptation, The Lancet Regional Health - Europe, 2021 Oct; Volume 9: 100230. [CrossRef]
- Getzova Zh, Hristova I, Kunchev A, Doycheva V, Bogdanov N, Sshalamanov D., Risks from the emergence of viral hemorrhagic fevers in Bulgaria and Europe, General Medicine, 2022;3:54:60.
- Rupasinghe R, Chomel BB, Martínez-López B.Climate change and zoonoses: A review of the current status, knowledge gaps, and future trends, Acta Tropica, 2022 Feb; (226): 106225. [CrossRef]
- UK Health Security Agency (2025) First detection of West Nile virus in UK mosquitoes, available at: https://www.gov.uk/government/news/first-detection-of-west-nile-virus-in-uk-mosquitoes, last accessed: 05.12.2025.
- Tambo E, El-Dessouky AG, Khater EIM, Xianonng Z, Enhanced surveillance and response approaches for pilgrims and local Saudi populations against emerging Nipah, Zika and Ebola viral diseases outbreaks threats, Journal of Infection and Public Health, 2020 May; 13:5:674-678. [CrossRef]
- Afrooghe A, Damavandi AR, Ahmadi E. Reverse zoonosis and monkeypox: Time for a more advanced global surveillance system for emerging pathogens, New Microbes and New Infections, 2022 July; (48):101013. [CrossRef]
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 Feb 15;395(10223):497-506. [CrossRef]
- Awadasseid A, Wu Y, Tanaka Y, Zhang W. Initial success in the identification and management of the coronavirus disease 2019 (COVID-19) indicates human-to-human transmission in Wuhan, China. Int J Biol Sci. 2020 Apr 6;16(11):1846-1860. [CrossRef]
- Wang H, Paulson KR, Pease S, et al. Estimating excess mortality due to the COVID-19 pandemic: a systematic analysis of COVID-19-related mortality, 2020–21, The Lancet, 2022 March; 399:10334:1513-1536. [CrossRef]
- Odone A, Delmonte D, Gaetti G, Signorelli C. Doubled mortality rate during the COVID-19 pandemic in Italy: quantifying what is not captured by surveillance, Public Health,2021 Jan; Volume 190:108-115. [CrossRef]
- Shah, M. R. T., Ahammed, T., Anjum, A., Chowdhury, A. A., & Suchana, A. J. Finding the real COVID-19 case-fatality rates for SAARC countries. Biosafety and health, 2021;3(3): 164-171. [CrossRef]
- WHO Director-General declares mpox outbreak a public health emergency of international concern- https://www.who.int/news/item/14-08-2024-who-director-general-declares-mpox-outbreak-a-public-health-emergency-of-international-concern, last accessed: 30 June 2025.
- O’Connell EK, Zhang G, Leguen F, et al. Innovative Uses for Syndromic Surveillance. Emerging Infectious Diseases. 2010;16(4):669-671. [CrossRef]
- Honeyman D, Gurdasani D, Notaras A, et al. Global Epidemiology of Outbreaks of Unknown Cause Identified by Open-Source Intelligence, 2020–2022. Emerging Infectious Diseases. 2025 Feb;31(2):298-308. [CrossRef]
- Barthell, E. N., Cordell, W. H., Moorhead, et al. The Frontlines of Medicine Project: A proposal for the standardized communication of emergency department data for public health uses including syndromic surveillance for biological and chemical terrorism. Annals of Emergency Medicine, 2002; 39:4:422–429. [CrossRef]
- Foldy, S., Biedrzycki, P. A., Barthell, E. N., Healy-Haney, N., Baker, B. K et al. Syndromic surveillance using regional emergency medicine internet. Annals of emergency medicine, 2004: 44(3), 242-246. [CrossRef]
- Gesteland, P. H., Gardner, R. M., Tsui, F. C., Espino, J., Rolfs, R.T. et al. Automated syndromic surveillance for the 2002 Winter Olympics. Journal of the American Medical Informatics Association, 2003; 10(6), 547-554. [CrossRef]
- Espino JU, Wagner MM. Accuracy of ICD-9-coded chief complaints and diagnoses for the detection of acute respiratory illness. Proc AMIA Symp., 2001; 164-8. PMID: 11833477.
- Reis, B. Y., & Mandl, K. D. Syndromic surveillance: the effects of syndrome grouping on model accuracy and outbreak detection. Annals of Emergency Medicine, 2004; 44:3: 235–241. [CrossRef]
- Morbey R. Communicating the detection capabilities of syndromic surveillance systems. Online J Public Health Inform. 2019 May 30;11(1):e304. https://pmc.ncbi.nlm.nih.gov/articles/PMC6606168/. [CrossRef]
- GUASTICCHI G, GIORGI ROSSI P, LORI G, et al. Syndromic surveillance: sensitivity and positive predictive value of the case definitions. Epidemiology and Infection. 2009;137(5):662-671. https://www.cambridge.org/core/journals/epidemiology-and-infection/article/syndromic-surveillance-sensitivity-and-positive-predictive-value-of-the-case-definitions/B7AC1092414E3A2D54A9A1C8B1548F9E. [CrossRef]
- Roberts, R.R, Einstein, A., Ahmad, I., Gore, R., Kampe, L.M. et al. Improving Syndrome Surveillance Case Definitions and Outcomes by Using Existing Data. Annals of Emergency Medicine, 2005; 46:3, 24. [CrossRef]
- Barthell E.N., Aronsky D., Cochrane D.G., Cable G. et al. For the Frontlines Work Group. The frontlines of medicine project progress report: standardized communication of emergency department triage data for syndromic surveillance. Annals of Emergency Medicine, 2004; 44:3, 247–252. [CrossRef]
- Lober W.B., Karras B.T., Wagner M.M., Overhage J.M., Davidson A.J., Fraser H. et al. Roundtable on bioterrorism detection: information system–based surveillance. Journal of the American Medical Informatics Association, 2002; 9(2), 105-115. [CrossRef]
- Wagner M.M., Tsui FC, Espino J.U. et al. The Emerging Science of Very Early Detection of Disease Outbreaks. Journal of Public Health Management and Practice, 2001; 7:6, 51–59. [CrossRef]
- Mandl, K.D.. Implementing Syndromic Surveillance: A Practical Guide Informed by the Early Experience. Journal of the American Medical Informatics Association, 2003; 11:2, 141–150. [CrossRef]
- Cameron AR, Meyer A, Faverjon C, Mackenzie C. Quantification of the sensitivity of early detection surveillance. Transbound Emerg Dis. 2020 Nov;67(6):2532-2543. [CrossRef]
- McNabb, S. J., Jajosky, R. A., Hall-Baker, P. A., Adams, D. A., Sharp, P., Anderson, W.J. et al. Centers for Disease Control and Prevention (CDC). Summary of notifiable diseases—United States, 2005. MMWR Morb Mortal Wkly Rep, 2007; 54(53), 1-92.
- Pang X, Wang X, Wang Q, Gao T, Li X Chapter 8 - Infectious Disease Surveillance and Early Warning System During Beijing Olympic Games, Editor(s): Weizhong Yang, Early Warning for Infectious Disease Outbreak, Academic Press, 2017; 163-180. [CrossRef]
- Sugishita Y., Sugawara T., Ohkusa Y., Ishikawa T., Yoshida M, Endo, H. Syndromic surveillance using ambulance transfer data in Tokyo, Japan. Journal of Infection and Chemotherapy, 2020; 26(1), 8-12. [CrossRef]
- Tarnas MC, Desai AN, Parker DM,Almhawish N, Zakieh O, Rayes D, et al. Syndromic surveillance of respiratory infections during protracted conflict: experiences from northern Syria 2016-2021, International Journal of Infectious Diseases, 2022; Volume 122, 337-344. [CrossRef]
- Welliver RC, Cherry JD, Boyer KM, et al. Sales of nonprescription cold remedies: a unique method of influenza surveillance. Pediatr Res 1979;13:1015--7.
- Angulo FJ, Tippen S, Sharp DJ, et al. A community waterborne outbreak of salmonellosis and the effectiveness of a boil water order. Am J Public Health 1997;87:580—4.
- Proctor ME, Blair KA, Davis JP. Surveillance data for waterborne illness detection: an assessment following a massive waterborne outbreak of Cryptosporidium infection. Epidemiol Infect 1998;120:43--54.
- Stirling R, Aramini J, Ellis A, et al. Waterborne cryptosporidiosis outbreak, North Battleford, Saskatchewan, Spring 2001. Can Commun Dis Rep 2001;27:185--92.
- Hogan WR, Tsui F-C, Ivanov O, et al. Early detection of pediatric respiratory and diarrheal outbreaks from retail sales of electrolyte products. J Am Med Inform Assoc 2003;10:555--62.
- Magruder S. Evaluation of over-the-counter pharmaceutical sales as a possible early warning indicator of public health. Johns Hopkins University Applied Physics Laboratory Technical Digest 2003; 24:349--53.
- Pivette, M., Mueller, J. E., Crépey, P., & Bar-Hen, A. (2014). Drug sales data analysis for outbreak detection of infectious diseases: a systematic literature review. BMC infectious diseases, 14, 604. [CrossRef]
- Das D, Metzger K, Heffernan R, Balter S, Weiss D, Mostashari F: Monitoring over-the-counter medication sales for early detection of disease outbreaks-New York City. MMWR Morb Mortal Wkly Rep. 2005, 54 (Suppl): 41-46.
- Yoshida M, Matsui T, Ohkusa Y, Kobayashi J, Ohyama T, Sugawara T, Yasui Y, Tachibana T, Okabe N: Seasonal influenza surveillance using prescription data for anti-influenza medications. Jpn J Infect Dis. 2009, 62: 233-235. [CrossRef]
- Chunara R., Goldstein E., Patterson-Lomba O. et al. Estimating influenza attack rates in the United States using a participatory cohort. Sci Rep 2015; 5, 9540. [CrossRef]
- Guerrisi C, Turbelin C, Blanchon T, et al. Participatory Syndromic Surveillance of Influenza in Europe, The Journal of Infectious Diseases, 2016; Volume 214, Issue suppl_4:S386–S392. [CrossRef]
- Mahmud AS, Chowdhury S, Sojib KH, et al.Participatory syndromic surveillance as a tool for tracking COVID-19 in Bangladesh, Epidemics, 2021; Volume 35, 100462. [CrossRef]
- Thomas, R.G., Hemphill, RR.,Aronsky, D. Syndromic surveillance for infectious disease outbreaks using chief complaint data. Annals of Emergency Medicine, 2004; 44(4), S124. [CrossRef]
- Bellazzini MA, Minor KD. ED syndromic surveillance for novel H1N1 spring 2009, The American Journal of Emergency Medicine, 2011; 29:1, 70-74. [CrossRef]
- Rosenkötter N, Ziemann A, Riesgo LG, Gillet JB, Vergeiner G, Krafft T, Brand H. Validity and timeliness of syndromic influenza surveillance during the autumn/winter wave of A (H1N1) influenza 2009: results of emergency medical dispatch, ambulance and emergency department data from three European regions. BMC Public Health. 2013 Oct 1;13:905. https://pmc.ncbi.nlm.nih.gov/articles/PMC3852468/. [CrossRef]
- Chikotie, T., Watson, B., Kimanuka, U., & Banda, T. (2025, May). Leveraging Electronic Syndromic Surveillance Synthetic Data to Predict Diarrhoea in Zimbabwean Children Under-Five: An Explainable AI Framework. In 2025 IST-Africa Conference (IST-Africa) (pp. 1-10). IEEE.
- Parums, D. V. (2023). infectious disease surveillance using artificial intelligence (AI) and its role in epidemic and pandemic preparedness. Medical science monitor: international medical journal of experimental and clinical research, 29, e941209-1.
- Lake, I. R., Colón-González, F. J., Barker, G. C., Morbey, R. A., Smith, G. E., & Elliot, A. J. (2019). Machine learning to refine decision making within a syndromic surveillance service. BMC Public Health, 19(1), 559.
| Data Source | Example System / Study | Lead Time (vs. Lab Confirmation) | Sensitivity / Specificity | Advantages | Limitations |
|---|---|---|---|---|---|
| Emergency Department Records | Espino et al. [26] | 4–7 days earlier | Sensitivity 44%, Specificity 97% | Timely, structured, standardized ICD coding | Requires integration across facilities; may miss mild cases |
| Emergency Department Records | Morbey R. [28] | Up to 7 days earlier | Sensitivity 100% for seasonal influenza and 0% for seasonal adenovirus | Early; adaptable | Detection highly dependent on system organization |
| Emergency Department Records | Reis, B. Y., & Mandl, K. D. [27] | 1-day detection approach | Sensitivity 26-47% Specificity 93% |
Early, information available in real-time, possibility to incorporate different data sources (symptom-based/diagnostic); temporal smoothing filters might reduce noise | Chief complaints lower accuracy |
| Emergency Department Records | Guasticchi G, Giorgi Rossi P, Lori G, et al. [29] | - | Variable, disease-specific: Sensitivity 22.2-90.2 |
Information routinely collected, Automated | Results are highly dependent on syndrome definition |
| Emergency Department Records | Rosenkötter N, Ziemann A, Riesgo LG et al.[54] | Up to 8 days earlier | Variable depending on country and source: Sensitivity 0-100%, Specificity 57.1-88.9% |
Strong validity and timeliness of data | Results differ across countries due to differences in catchment population. In some cases identification of events is later than references. |
| Pharmacy / OTC Sales | Das et al., 2005 [47]; Hogan et al., 2003 [44] | 2–14 days earlier | Variable, disease-specific | Early community signal; inexpensive; easy to automate | False positives; context-dependent on self-medication patterns |
| Participatory (Citizen Reports) | Chunara et al. 2015[49]; Mahmud et al., 2021 [51] | 3–5 days earlier | High correlation with lab-confirmed data | Engages public; rapid; adaptable | Participation bias; data validation required |
| Hybrid / Multi-source Systems | Foldy et al. 2004 [24] | 2–10 days earlier | Improved combined sensitivity | Comprehensive, scalable; high analytic power | Requires strong IT infrastructure and privacy safeguards |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




