Submitted:
19 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
Tau protein is a nonspecific marker of neurodegeneration, and its phosphorylated form, ptau-181, is specifically associated with Alzheimer's disease (AD). Calculating the ratio of the phosphorylated form to total tau protein can help distinguish AD from other tauopathies or neurodegeneration, as well as reduce the impact of individual differences in total tau protein levels. This also allows for monitoring and comparing the dynamics of changes within the same patient. For this purpose, two SPRi biosensors were constructed, sensitive to the proteins described: total tau and ptau-181 for plasma determinations. The use of these biosensors requires prior sensor validation, during which specific parameters of the analytical method are established. A study of the optimal concentration of the receptor layer in which particular antibodies were immobilized found that the optimal concentration for total tau protein determinations was 1000 ng/mL. For ptau-181, it was 90 ng/mL. Biosensor layer formation was confirmed by analysis over a wide angle range, which enabled the generation of SPR curves. The dynamic range of the sensors is 1–50 pg/mL for total tau and 1–100 pg/mL for ptau-181. The limits of detection are 0.18 pg/mL and 0.037 pg/mL, respectively. Low standard deviation (SD) and coefficient of variation (CV) values indicate good precision and accuracy of the results obtained using the SPRi biosensors. Specificity testing confirmed that no interferents influenced the assay. The method is therefore suitable for researching biological materials, such as blood plasma. Proteins were thus measured in the blood plasma of AD patients and controls. Statistical analysis revealed significant differences in the concentrations of tau and ptau-181 protein in both groups. The calculated ptau/total tau ratio for both sample groups also demonstrated high statistical significance. This suggests that a high ratio may be characteristic of AD. However, more extensive analysis is needed to obtain cutoff values. The ROC curves indicate that both biosensors have good diagnostic utility, with lower specificity for total tau.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Subsection
2.2. Biological Material
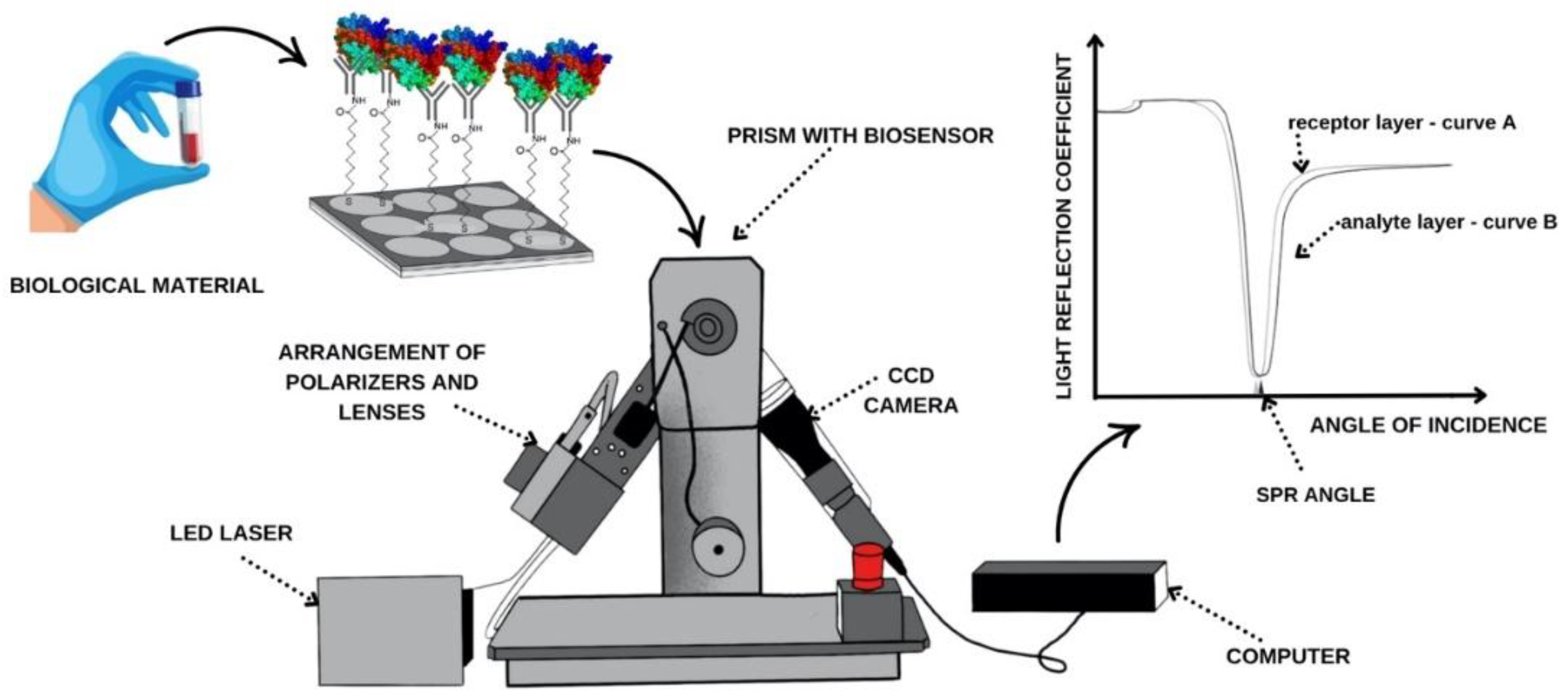
2.3. SPRi Device and Measurement Methodology
3. Experimental
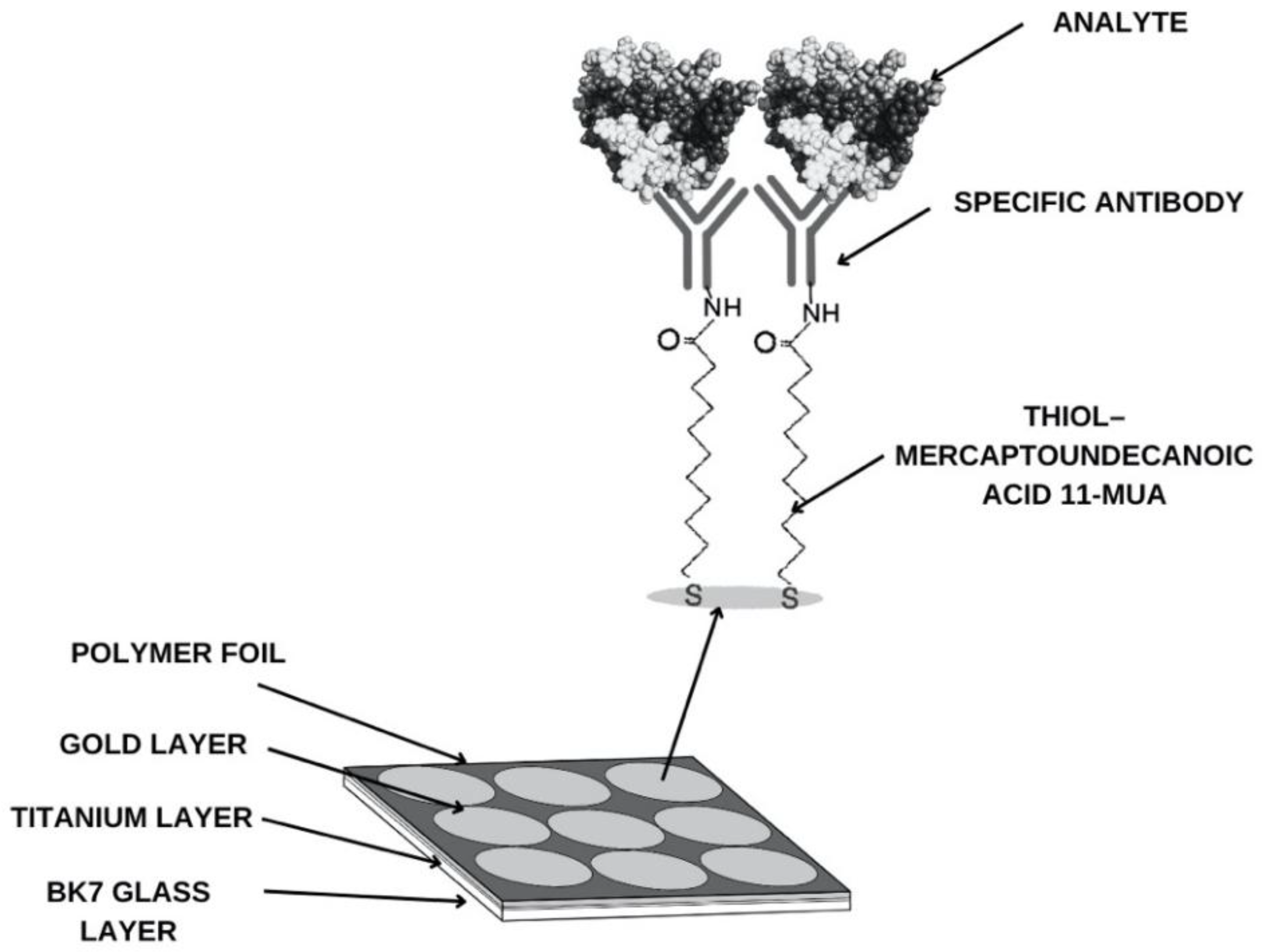
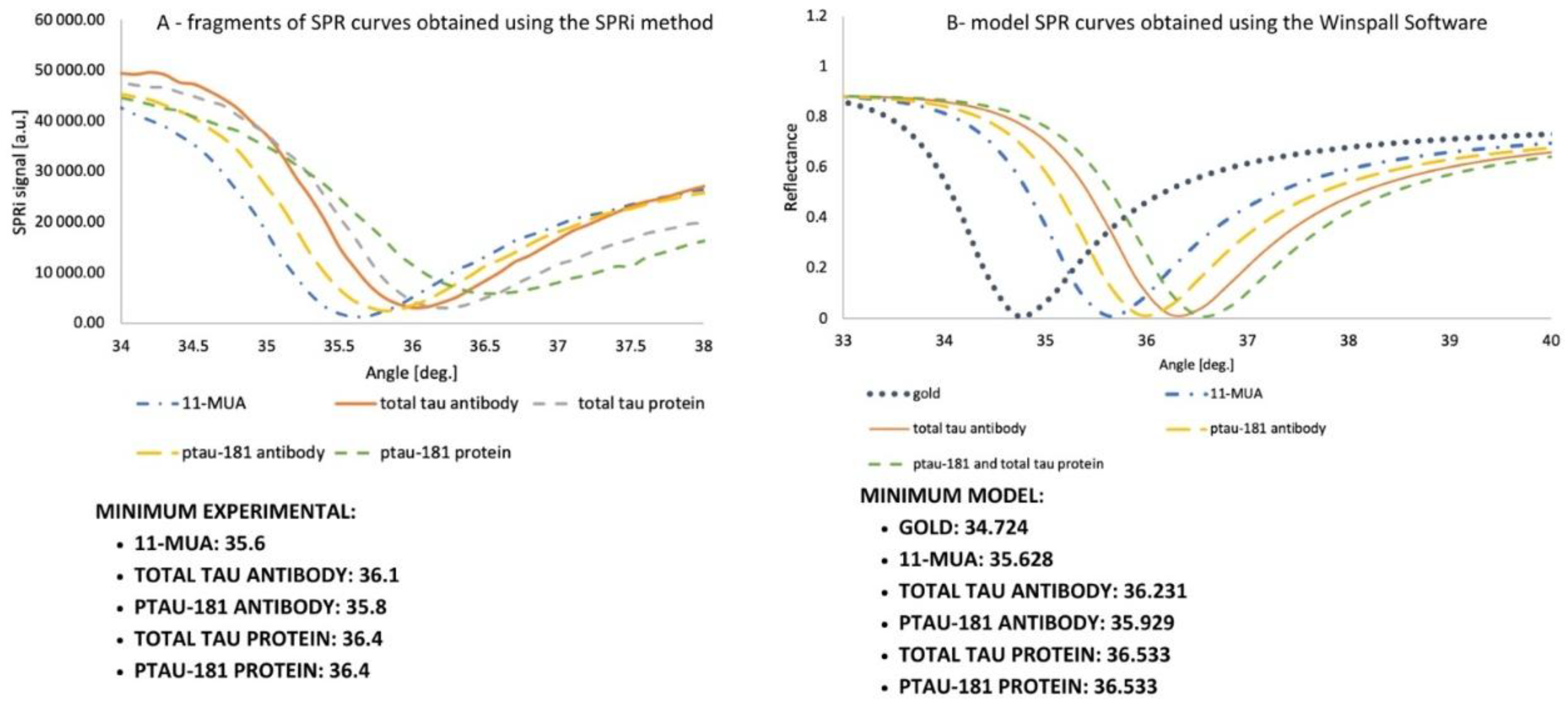
3.1. Confirmation of Layer Formation on the Sensor Surface
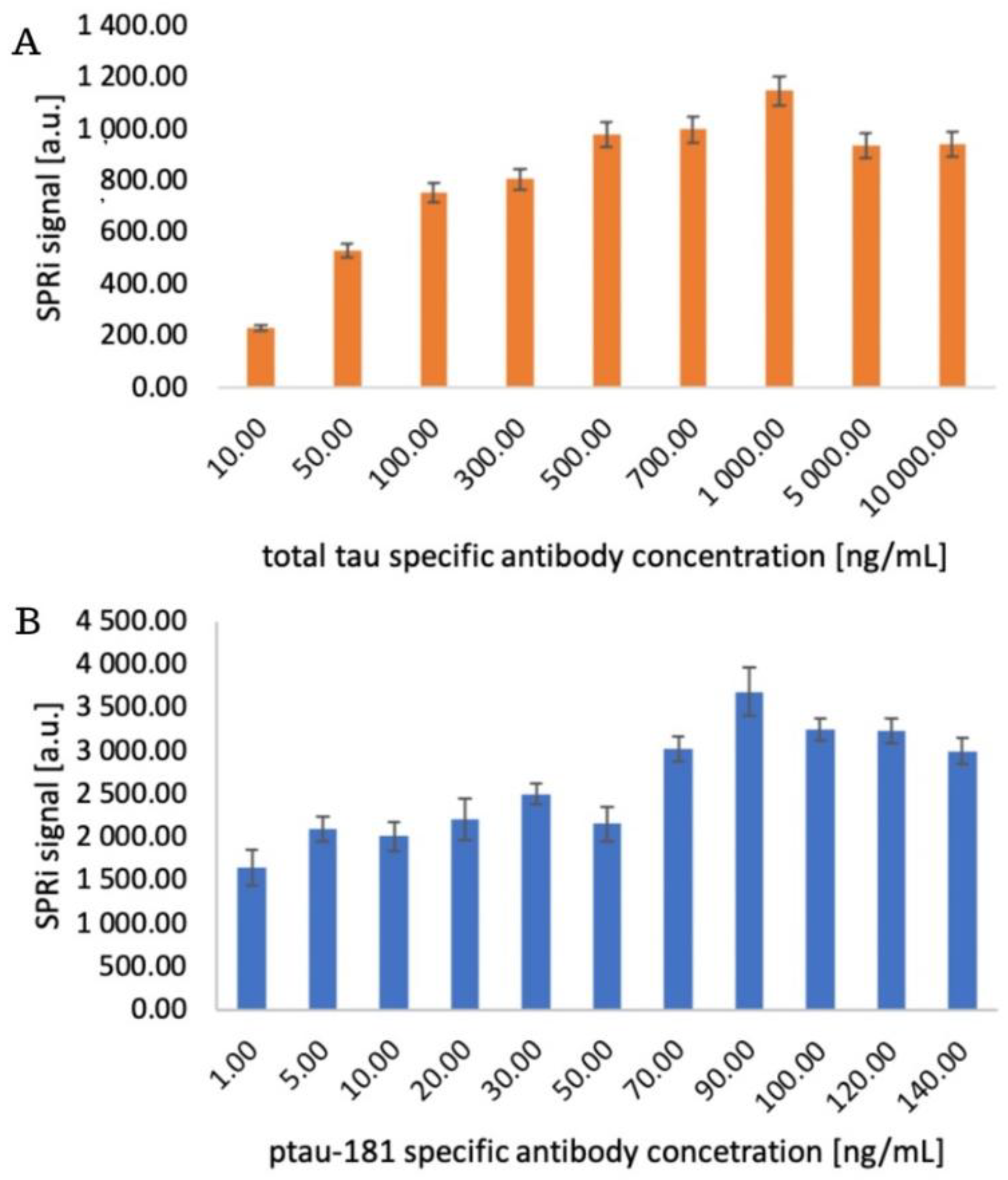
3.2. Structure of the Receptor Layer. Selection of the Appropriate Antibody Concentration
3.3. Analytical Response of the Biosensor. Limit of Detection and Quantification
3.4. Precision and Accuracy, Repeatability of the Method
3.5. Selectivity of the Method
3.6. Determination of Proteins in Patients' Blood Plasma
4. Statistical Analysis and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Number of Sample from Patient with AD | Concentration of Ptau-181 [pg/mL] | Concentration of Total tau [pg/mL] | Ptau-181/Total tau Ratio | Number of Sample from Healthy Patient | Concentration of Ptau-181 [pg/mL] | Concentration of Total tau [pg/mL] | Ptau-181/Total tau Ratio |
|---|---|---|---|---|---|---|---|
| 1 | 41.77 | 13.02 | 3.21 | 1C | 2.31 | 3.19 | 0.72 |
| 2 | 100.76 | 45.26 | 2.23 | 2C | 5.14 | 2.76 | 1.87 |
| 3 | 70.25 | 6.93 | 10.14 | 3C | 1.95 | 5.11 | 0.38 |
| 4 | 47.71 | 3.35 | 14.22 | 4C | 3.42 | 7.57 | 0.45 |
| 5 | 92.58 | 27.54 | 3.36 | 5C | 1.19 | 3.71 | 0.32 |
| 6 | 101.92 | 6.26 | 16.28 | 6C | 11.03 | 5.35 | 2.06 |
| 7 | 36.55 | 28.10 | 1.30 | 7C | 1.60 | 5.69 | 0.28 |
| 8 | 94.99 | 37.83 | 2.51 | 8C | 0.24 | 8.28 | 0.03 |
| 9 | 72.88 | 15.07 | 4.84 | 9C | 5.26 | 0.07 | 72.68 |
| 10 | 15.53 | 17.21 | 0.90 | 10C | 0.55 | 2.58 | 0.21 |
| 11 | 60.73 | 51.83 | 1.17 | 11C | 21.55 | 2.21 | 9.74 |
| 12 | 79.74 | 21.38 | 3.73 | 12C | 0.36 | 2.98 | 0.12 |
| 13 | 42.95 | 3.55 | 12.09 | 13C | 9.24 | 6.19 | 1.49 |
| 14 | 33.54 | 32.13 | 1.04 | 14C | 1.36 | 7.58 | 0.18 |
| 15 | 87.67 | 14.41 | 6.09 | 15C | 0.99 | 5.67 | 0.17 |
| 16 | 38.44 | 40.87 | 0.94 | 16C | 0.38 | 5.63 | 0.07 |
| 17 | 101.96 | 36.33 | 2.81 | 17C | 2.01 | 0.59 | 3.41 |
| 18C | 0.40 | 1.21 | 0.33 |
References
- Klyucherev, T.O.; Olszewski, P.; Shalimova, A.A. Advances in the development of new biomarkers for Alzheimer’s disease. Transl. Neurodegener. 2022, 11, 25. [CrossRef]
- Gunes, S.; Aizawa, Y.; Sugashi, T.; Sugimoto, M.; Rodrigues, P.P. Biomarkers for Alzheimer’s Disease in the Current State: A Narrative Review. Int. J. Mol. Sci. 2022, 23, 4962. [CrossRef]
- Turk, K.W.; Geada, A.; Alvarez, V.E. A comparison between tau and amyloid-β cerebrospinal fluid biomarkers in chronic traumatic encephalopathy and Alzheimer disease. Alz. Res. Therapy 2022, 14, 28. [CrossRef]
- Braak, H.; Braak, E. Staging of Alzheimer’s disease-related neurofibrillary changes. Neurobiol. Aging 1995, 16(3), 271–8, discussion 8-84. [CrossRef]
- Leuzy, A.; Ashton, N.J.; Mattsson-Carlgren, N.; Dodich, A.; Boccardi, M.; Corre, J.; Drzezga, A.; Nordberg, A.; Ossenkoppele, R.; Zetterberg, H. 2020 update on the clinical validity of cerebrospinal fluid amyloid, tau, and phospho-tau as biomarkers for Alzheimer’s disease in the context of a structured 5-phase development framework. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 2121–2139. [CrossRef]
- Florean, I.; Penolazzi, B.; Menichelli, A.; Pastore, M.; Cattaruzza, T.; Mazzon, G.; Manganotti, P. Using the ATN system as a guide for the neuropsychological assessment of Alzheimer’s disease. J. Clin. Exp. Neuropsychol. 2022, 43, 1–18. [CrossRef]
- Hansson, O.; Blennow, K.; Zetterberg, H.; Dage, J. Blood biomarkers for Alzheimer’s disease in clinical practice and trials. Nat Aging 2023, 3, 506–519. [CrossRef]
- Sinsky, J.; Pichlerova, K.; Hanes, J. Tau Protein Interaction Partners and Their Roles in Alzheimer’s Disease and Other Tauopathies. Int. J. Mol. Sci. 2021, 22, 9207. [CrossRef]
- Karikari, T.K.; Pascoal, T.A.; Ashton, N.J.; Janelidze, S.; Benedet, A.L.; Rodriguez, J.L.; Chamoun, M.; Savard, M.; Kang, M.S.; Therriault, J.; Schöll, M.; Massarweh, G.; Soucy, J.P.; Höglund, K.; Brinkmalm, G.; Mattsson, N.; Palmqvist, S.; Gauthier, S.; Stomrud, E.; Zetterberg, H.; Hansson, O.; Rosa-Neto, P.; Blennow, K. Blood phosphorylated tau 181 as a biomarker for Alzheimer's disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet. Neuro.l 2020, 19(5), 422-433. [CrossRef]
- Hong, X.; Huang, L.; Lei, F.; Li, T.; Luo, Y.; Zeng, M.; Wang, Z. The Role and Pathogenesis of Tau Protein in Alzheimer’s Disease. Biomolecules 2025, 15, 824. [CrossRef]
- Dage, J.L.; Wennberg, A.M.V.; Airey, D.C.; Hagen, C.E.; Knopman, D.S.; Machulda, M.M.; Roberts, R.O.; Jack Jr, C.R.; Petersen R.C.; Mielke, M.M. Levels of tau protein in plasma are associated with neurodegeneration and cognitive function in a population-based elderly cohort. Alzheimers Dement. 2016, 12(12), 1226–34. [CrossRef]
- Janelidze, S.; Stomrud, E.; Smith, R.; Palmqvist, S.; Mattsson, N.; Airey, D.C.; Proctor, N.K.; Chai, X.; Shcherbinin, S.; Sims, J.R.; Triana-Baltzer, G.; Theumis, C.; Slemmon, R.; Mercken, M.; Kolb, H.; Dage, J.L.; Hannson, O. Cerebrospinal fluid p-tau217 performs better than p-tau181 as a biomarker of Alzheimer's disease. Nat Commun. 2020, 11(1), 1683. [CrossRef]
- Blennow, K.; Hampel, H. CSF markers for incipient Alzheimer's disease. Lancet Neurol. 2003, 2(10), 605-13. [CrossRef]
- Krawczuk, D.; Kulczyńska-Przybik, A.; Mroczko, B. Clinical Application of Blood Biomarkers in Neurodegenerative Diseases—Present and Future Perspectives. Int. J. Mol. Sci. 2024, 25, 8132. [CrossRef]
- Chadha, U.; Bhardwaj, P.; Agarwal, R.; Rawat, P.; Agarwal, R.; Gupta, I.; Panjwani, M.; Singh, S.; Ahuja, C.; Selvaraj, S.K.; Banavoth, M.; Sonar, P.; Badoni, B.; Chakravorty, A. Recent progress and growth in biosensors technology: A critical review. J. Ind. Eng. Chem. 2022, 109, 21-51. [CrossRef]
- Huan,g Z.; Li, M.; Zhang, L.; Liu, Y.; Electrochemical immunosensor based on superwettable microdroplet array for detecting multiple Alzheimer’s disease biomarkers. Front. Bioeng. Biotechnol. 2022, 10, 1029428. [CrossRef]
- Kim, K.; Kim, M.J.; Kim, D.W.; Kim, S.Y.; Park, S.; Park, C.B. Clinically accurate diagnosis of Alzheimer’s disease via multiplexed sensing of core biomarkers in human plasma. Nat Commun, 2020, 11, 119. [CrossRef]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: review. Sens. Actuat. B-Chem. 1999, 54(1–2), 3-15. [CrossRef]
- Homola, J.; Piliarik, M. (2006). Surface Plasmon Resonance (SPR) Sensors. In: Homola, J. (eds) Surface Plasmon Resonance Based Sensors. Springer Series on Chemical Sensors and Biosensors 2006, vol 4. Springer, Berlin, Heidelberg.
- Bocková, M.; Slabý, J.; Špringer, S.; Homola, J. Advances in Surface Plasmon Resonance Imaging and Microscopy and Their Biological Applications. Annu. Rev. Anal. Chem. 2019, 12, 151-176. [CrossRef]
- Hutapea, T. P. H.; Madurani, K. A.; Syahputra, M. Y.; Hudha, M. N.; Asriana, A. N. Suprapto; Kurniawan, F. Albumin: Source, preparation, determination, applications, and prospects. J. Sci. Adv. Mater. Devices 2023, 8(2), 100549. [CrossRef]
- Vu Nu, T.T.; Tran, N.H.T.; Nam, E.; Nguyen, T.T.; Yoon, W.J.; Cho, S.; Kim, J.; Chang, K.-A.; Ju, H. Blood-Based Immunoassay of Tau Proteins for Early Diagnosis of Alzheimer’s Disease Using Surface Plasmon Resonance Fiber Sensors. RSC Adv 2018, 8, 7855–7862. [CrossRef]
- Kaushik, A.; Jayant, R.D.; Tiwari, S.; Vashist, A.; Nair, M. Electrochemical Immunosensor for Phosphorylated Tau-181 Detection toward Alzheimer’s Disease Diagnosis. Biosens. Bioelectron 2021, 183, 113450.
- Tieu, M.V.; Choi, S.H.; Le, H.T.N.; Cho, S. Electrochemical impedance-based biosensor for label-free determination of plasma P-tau181 levels for clinically accurate diagnosis of mild cognitive impairment and Alzheimer's disease. Anal Chim Acta. 2023, 1273, 341535. [CrossRef]
- Tijms, B.M.; Teunissen, C.E. Concatenating plasma p-tau to Alzheimer’s disease. Brain 2021, 144(1), 14–17. [CrossRef]
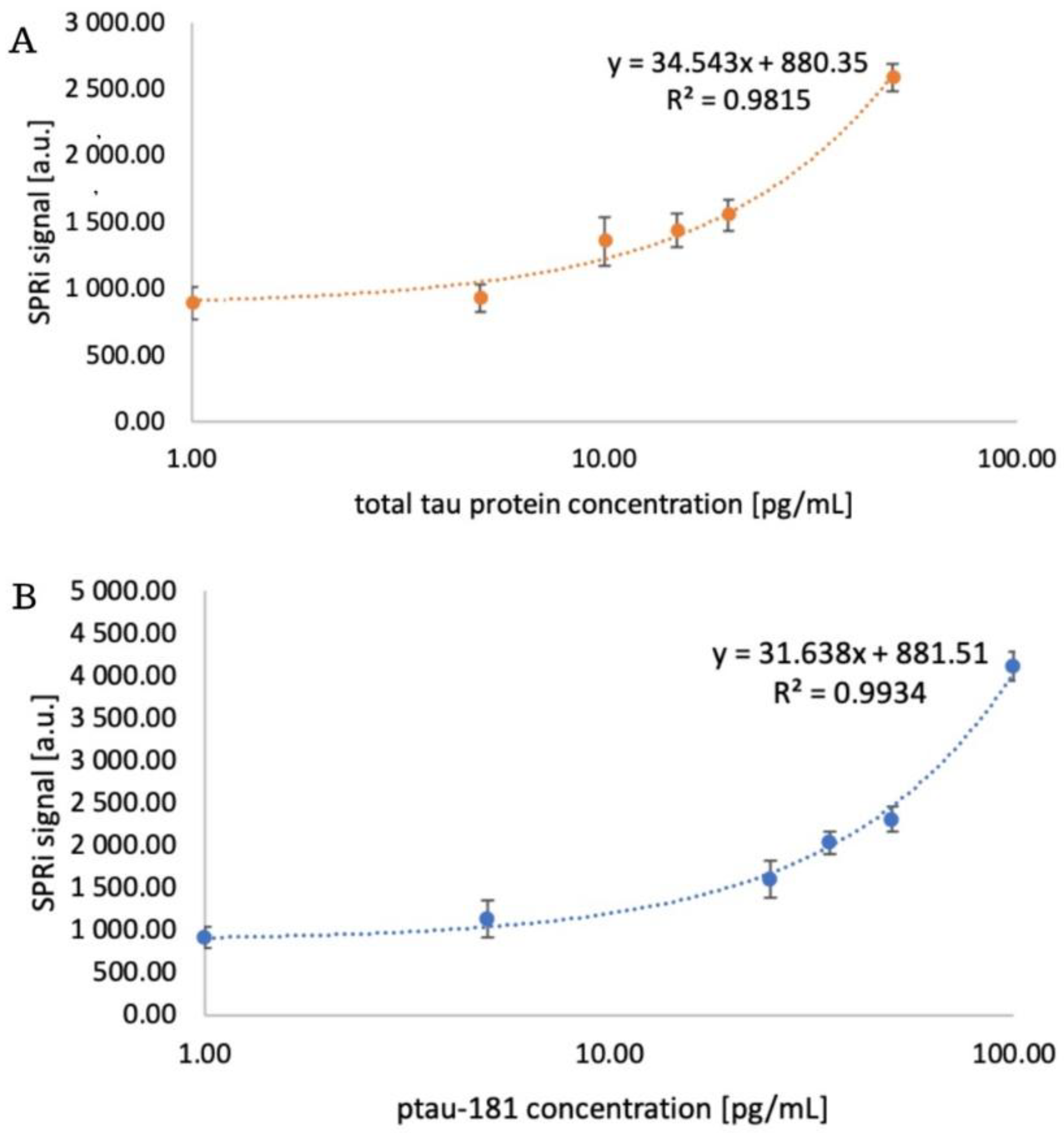
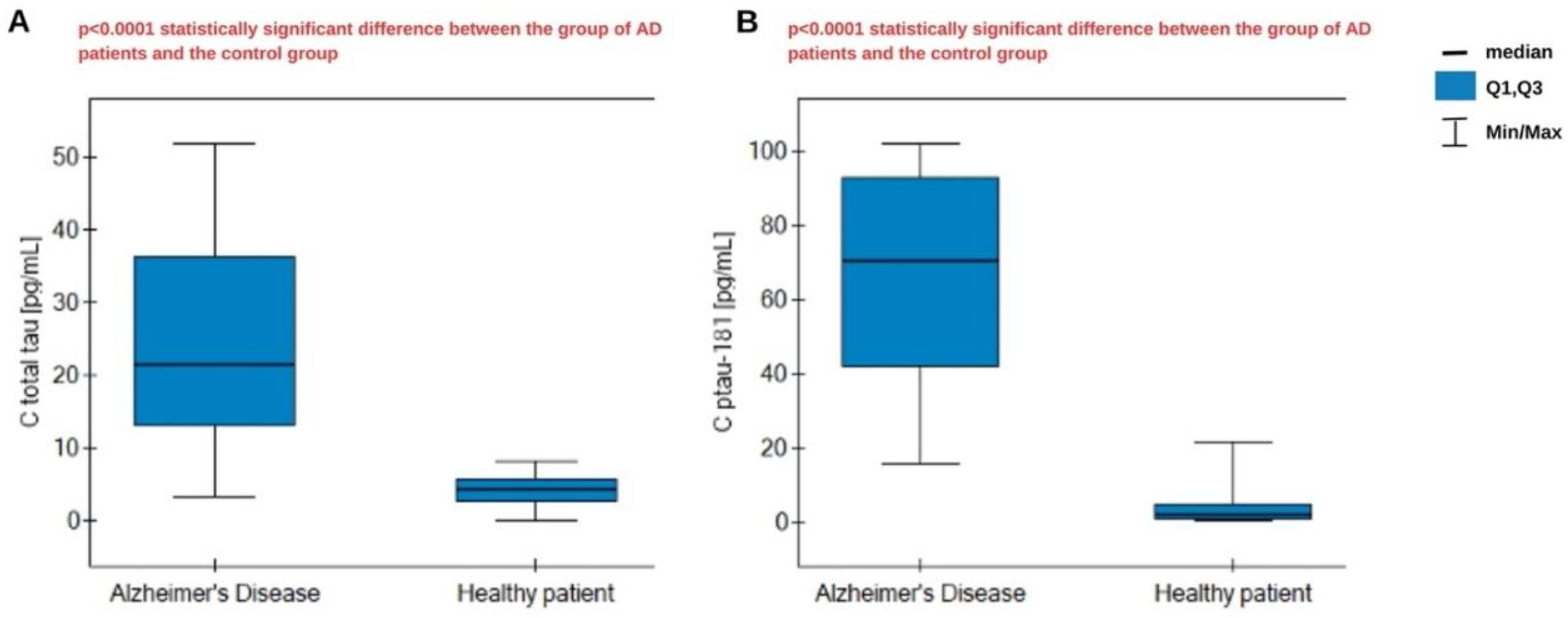
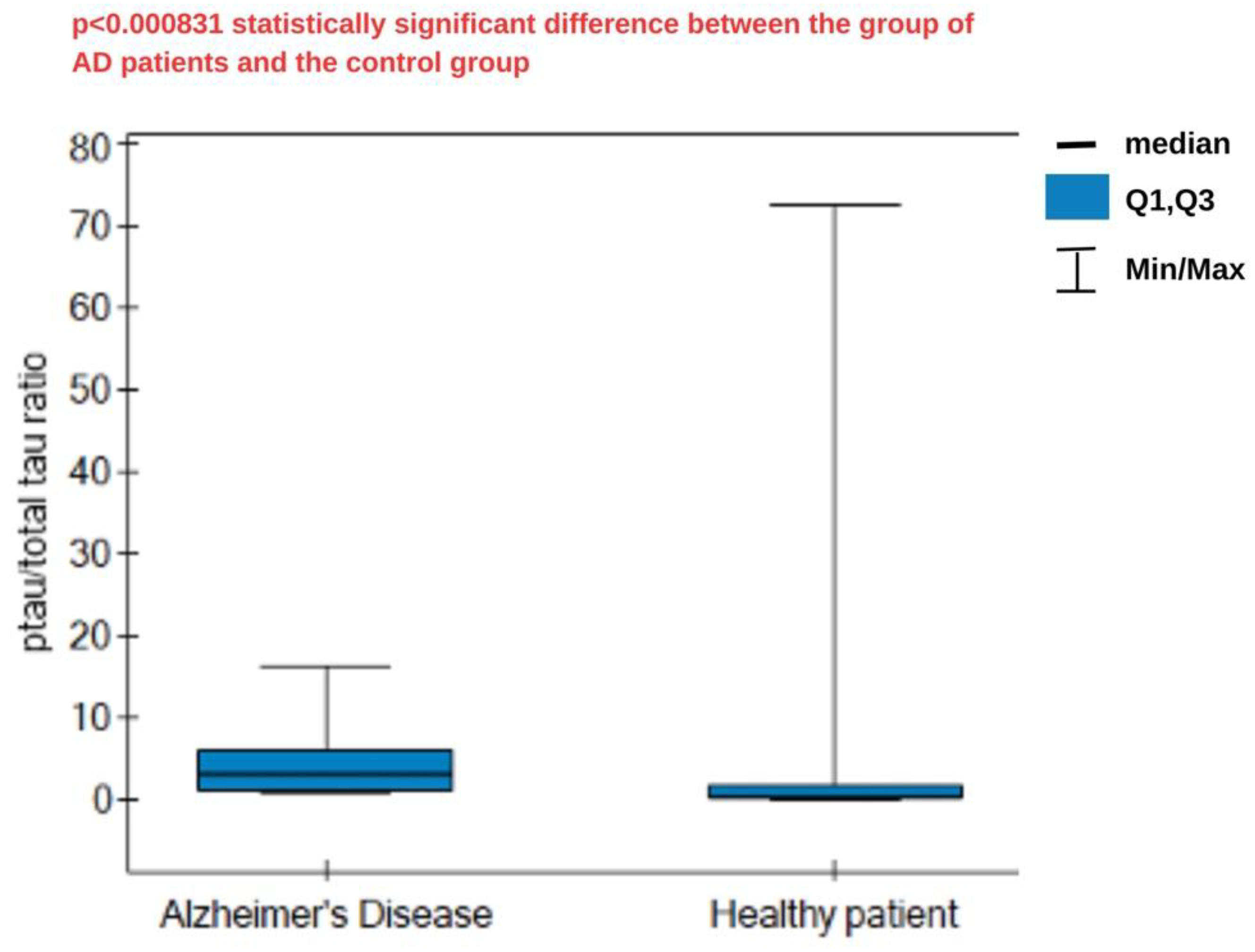
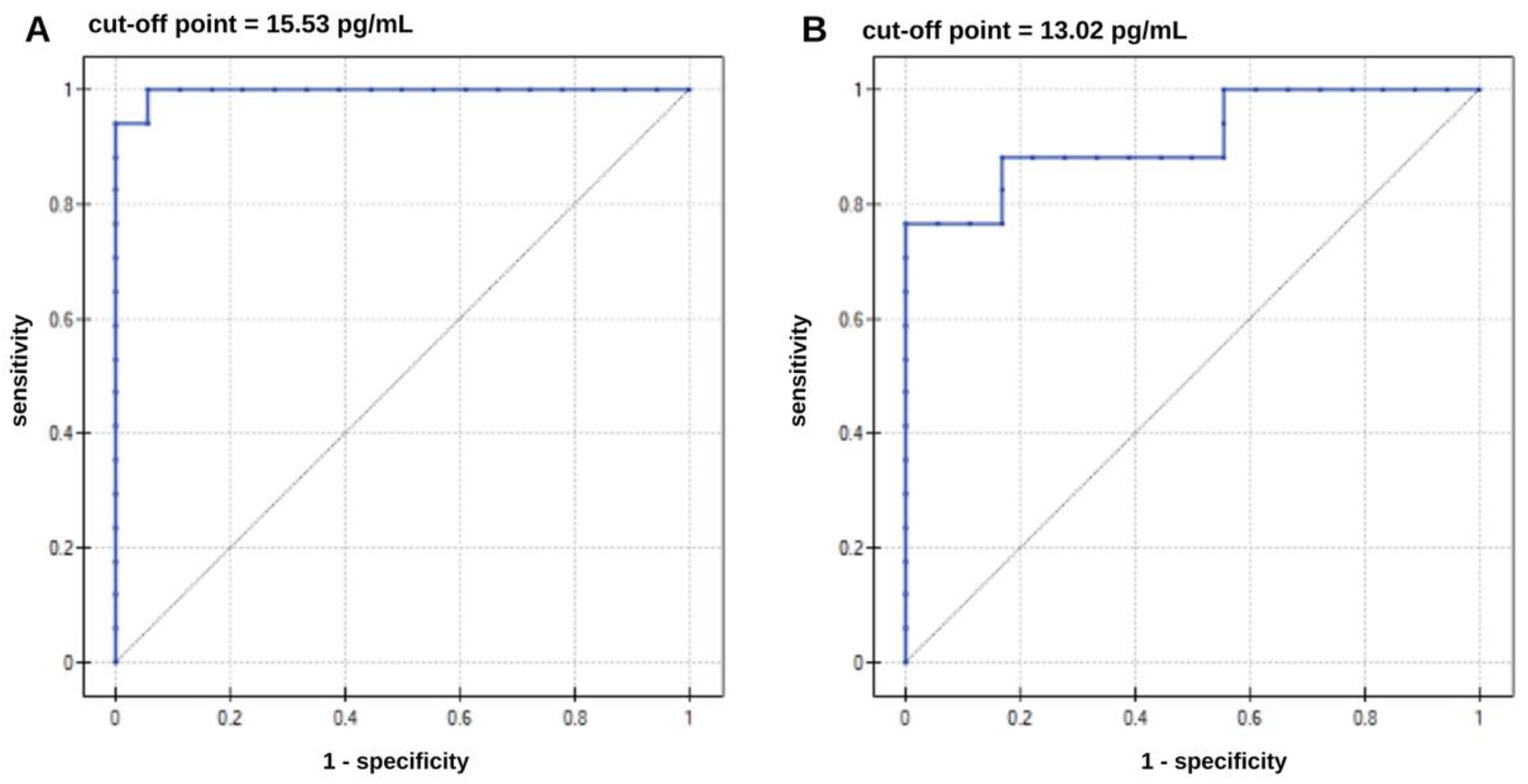
| Parameter | Total tau | Ptau-181 |
|---|---|---|
| LOD [pg/mL] | 0.18 | 0.037 |
| LOQ [pg/mL] | 0.85 | 0.112 |
| TOTAL TAU PROTEIN | ||||||||
| Concentration of standard solution [pg/mL] | Analysis 1 | Analysis 2 | Analysis 3 | Analysis 4 | SD [pg/mL] | MEAN [pg/mL] | CV [%] | RE [%] |
| 0.85 | 0.87 | 0.84 | 0.86 | 0.83 | 0.02 | 0.85 | 1.83 | 0.25 |
| 1.00 | 1.18 | 1.11 | 0.96 | 1.18 | 0.09 | 1.11 | 8.25 | 10.70 |
| 10.00 | 13.85 | 10.61 | 9.31 | 9.97 | 1.75 | 10.93 | 15.97 | 9.34 |
| 20.00 | 19.57 | 18.73 | 18.56 | 19.68 | 0.49 | 19.14 | 2.59 | 4.32 |
| 50.00 | 49.41 | 44.69 | 50.06 | 47.57 | 2.08 | 47.93 | 4.35 | 4.14 |
| PTAU-181 PROTEIN | ||||||||
| Concentration of standard solution [pg/mL] | Analysis 1 | Analysis 2 | Analysis 3 | Analysis 4 | SD [pg/mL] | MEAN [pg/mL] | CV [%] | RE [%] |
| 0.112 | 0.12 | 0.14 | 0.13 | 0.13 | 0.01 | 0.13 | 6.09 | 14.93 |
| 1.00 | 1.03 | 1.27 | 1.05 | 1.13 | 0.11 | 1.12 | 9.63 | 11.99 |
| 25.00 | 23.19 | 24.72 | 24.23 | 21.40 | 1.47 | 23.39 | 6.28 | 6.45 |
| 50.00 | 45.41 | 49.02 | 48.32 | 46.15 | 1.72 | 47.22 | 3.65 | 5.55 |
| 100.00 | 102.10 | 98.54 | 104.42 | 101.08 | 2.44 | 101.54 | 2.40 | 1.54 |
| TOTAL TAU | ||||
| Potential interferent | Concentration of standard solution [pg/mL] | Ctotal tau vs. Cinterferent | Cfound [pg/mL] | Recovery [%] |
| Ptau-181 | 100 | 1 to 10 | 98.66 | 98.66 |
| 1 to 100 | 108.84 | 108.84 | ||
| erythropoietin | 1 to 10 | 101.55 | 101.55 | |
| 1 to 100 | 101.02 | 101.02 | ||
| human albumin | 1 to 10 | 104.16 | 104.16 | |
| 1 to 100 | 101.73 | 101.73 | ||
| PTAU-181 | ||||
| Potential interferent | Concentration of standard solution [pg/mL] | Cptau-181 vs. Cinterferent | Cfound [pg/mL] | Recovery [%] |
| total tau | 50 | 1 to10 | 51.12 | 102.24 |
| 1 to 100 | 52.48 | 104.96 | ||
| erythropoietin | 1 to 10 | 48.90 | 97.80 | |
| 1 to 100 | 52.60 | 105.20 | ||
| albumin | 1 to 10 | 52.22 | 104.44 | |
| 1 to 100 | 49.19 | 98.38 | ||
| Protein | Direction of the Diagnostic Variable | AUC | Sensitivity [%] | Specificity] [% | PPV [%] | NPV [%] | Cutt-of Point [pg/mL] | p-Value |
|---|---|---|---|---|---|---|---|---|
| ptau-181 | stimulant | 0.92 | 76 | 100 | 100 | 81 | 13.02 | p< 0.000028 |
| total tau | stimulant | 0.99 | 100 | 94 | 94 | 100 | 15.53 | p<0.000001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




