Submitted:
19 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
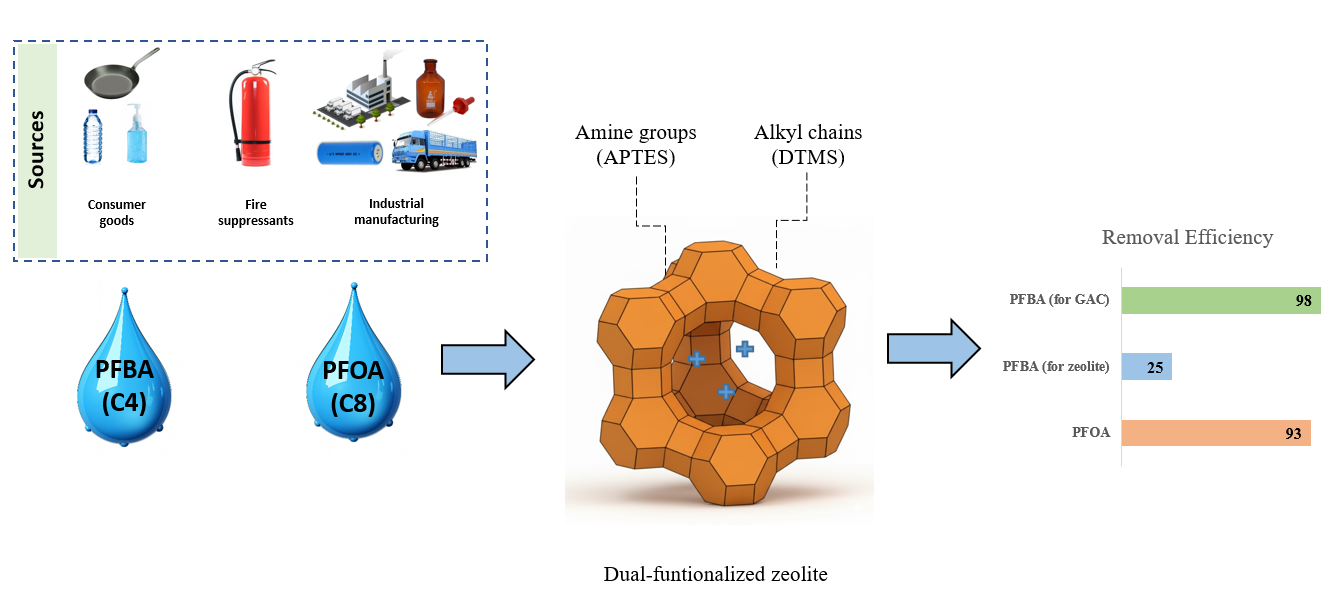
Keywords:
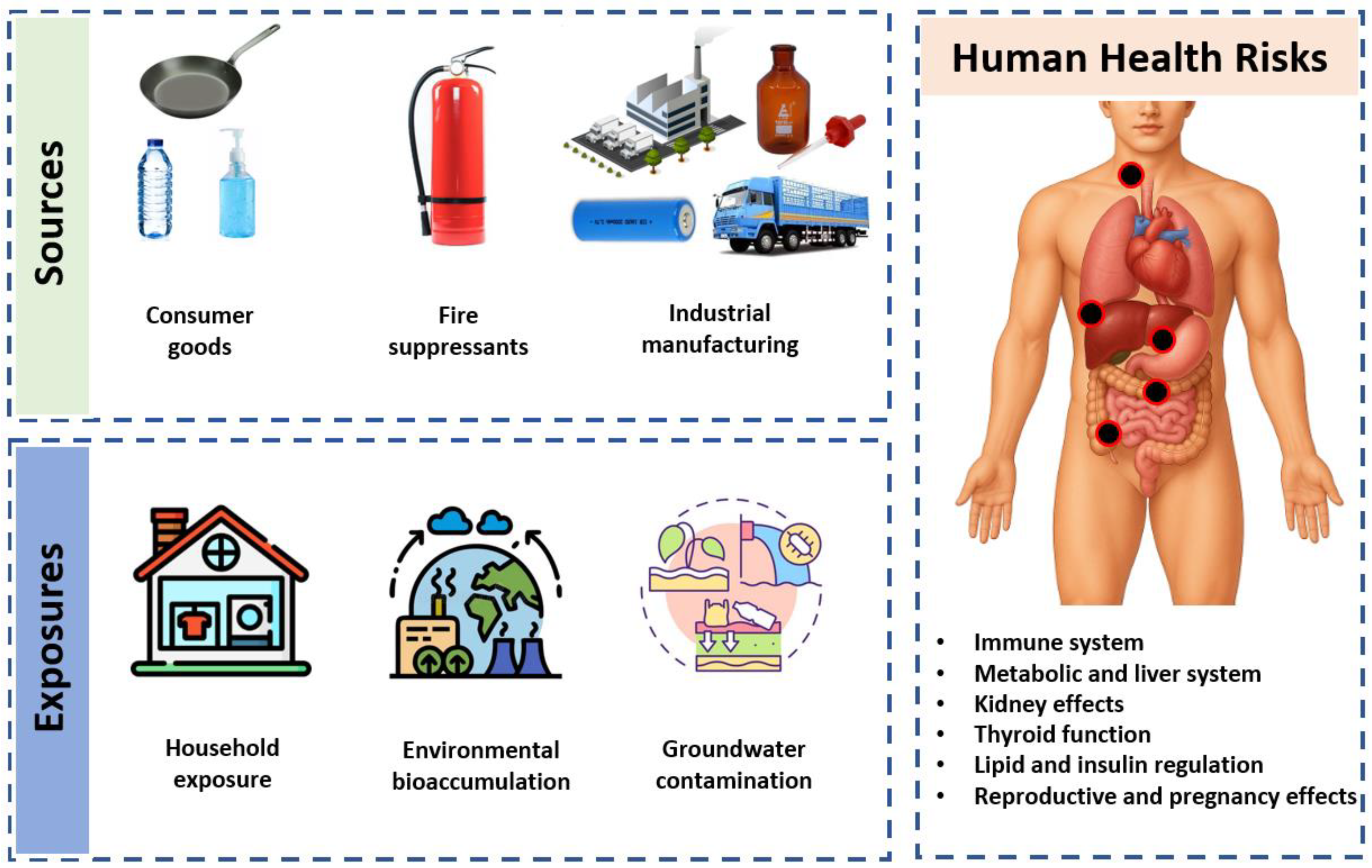
1. Introduction
2. Materials and Methods
2.1. Materials and Chemicals
2.2. Zeolite Surface Modification Procedures
2.2.1. Lanthanum Coated Zeolite (AZ-La)
2.2.2. Iron Treatment (AZ-Fe)
2.2.3. APTES Grafting (AZ-APTES)
2.2.4. DTMS Grafting (AZ-DTMS)
2.2.5. Combined APTES + DTMS Functionalization
2.2.6. Graphene Oxide Coating (AZ-GO)
2.3. Batch Adsorption Experiments
2.4. Analytical Method
3. Results
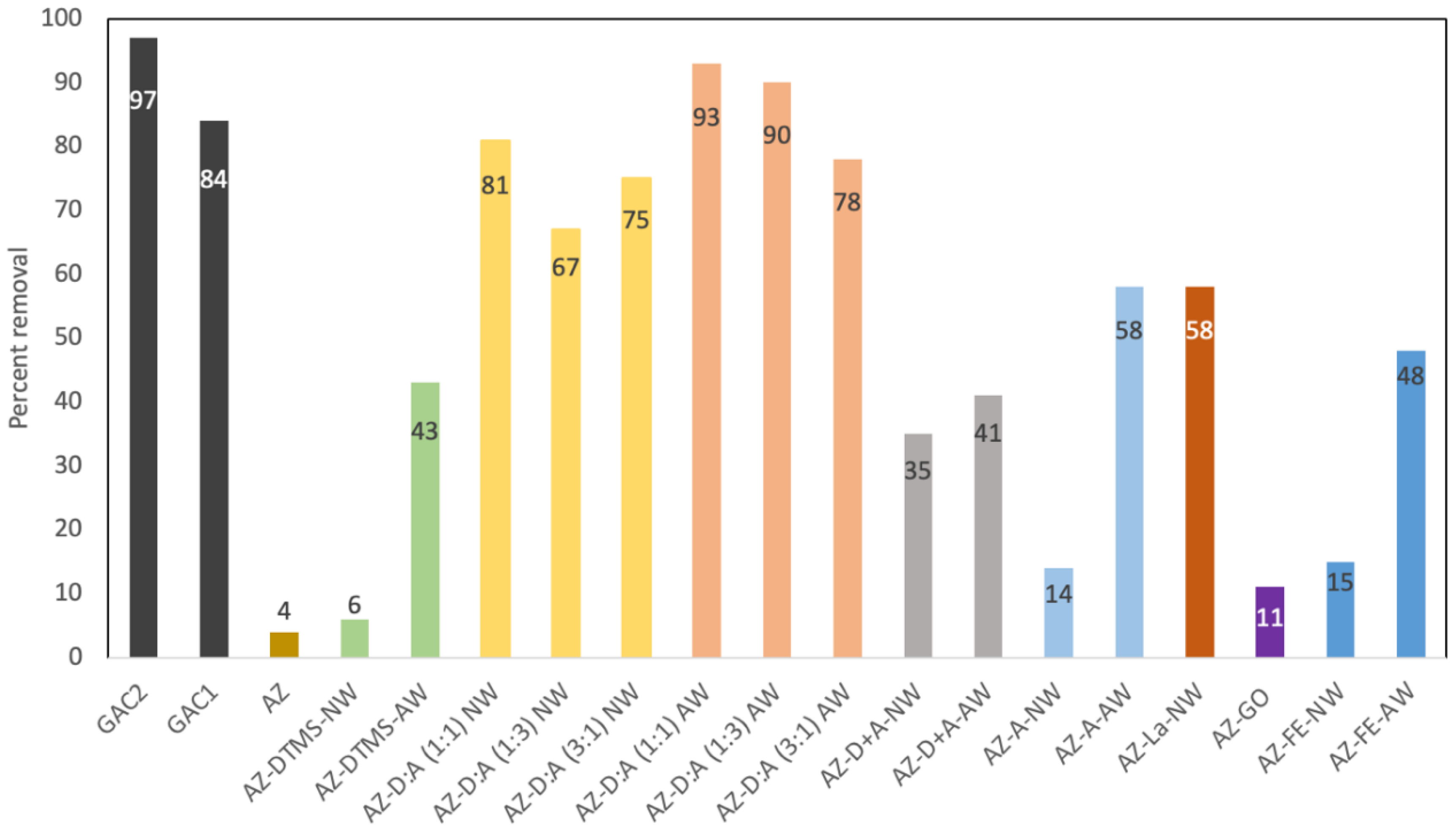
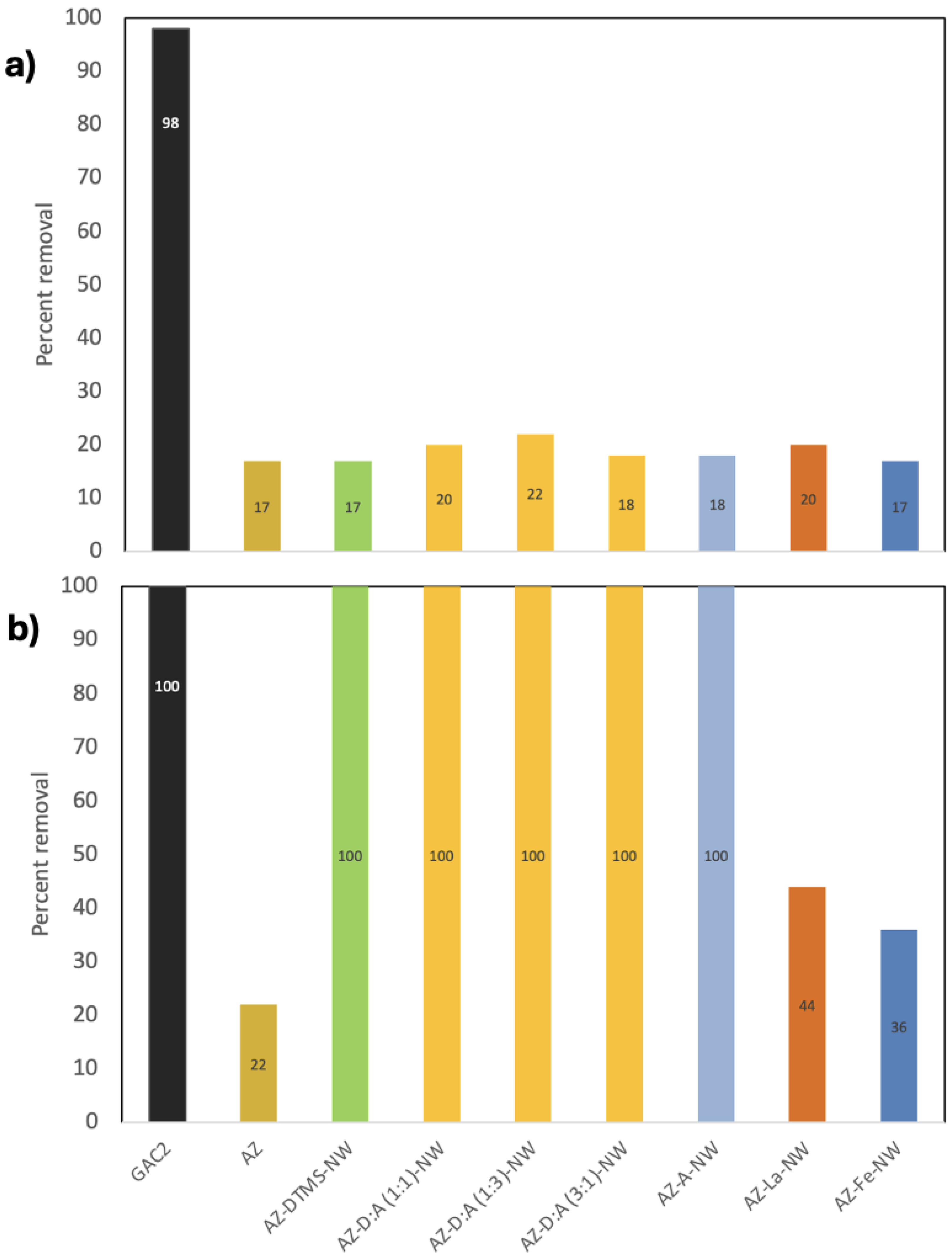
3.1. PFOA Removal at High Concentration (1 ppm Screening)
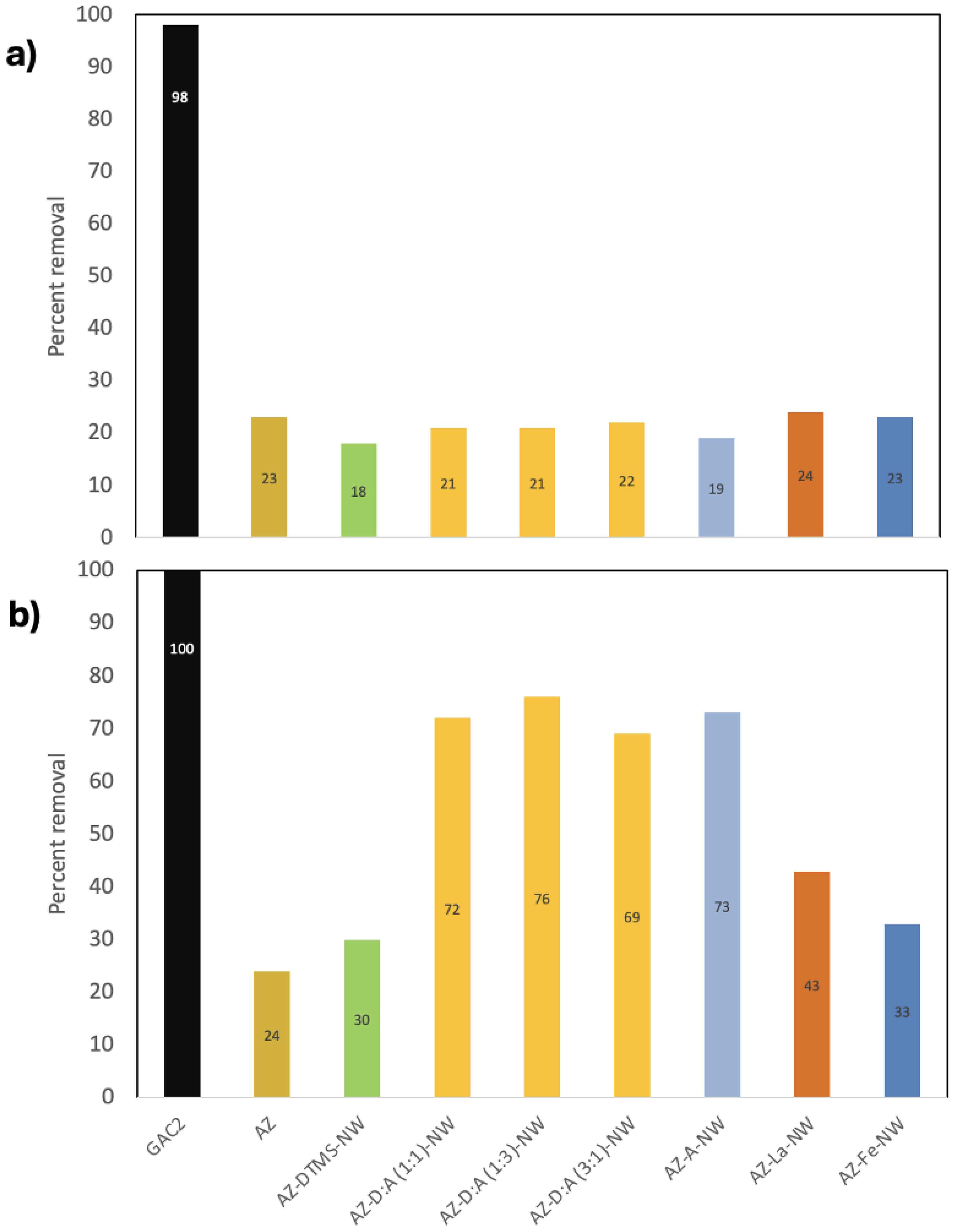
3.2. PFAS Removal at 100 μg/L – Single-Compound Tests
3.3. PFAS Removal from a Mixed Solution (Competitive Effects)
3.4. Mechanistic Insights and Discussion of Adsorbent Performance
3.5. Implications for Practical Use and Regeneration
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| PFAS | Per- and Polyfluoroalkyl Substances |
| GAC | Granular Activated Carbon |
| PFOA | Perfluorooctanoic Acid |
| PFBA | Perfluorobutanoic Acid |
| PFTDA | Perfluorotetradecanoic Acid |
| AZ | Australian Zeolite (Clinoptilolite) |
| NW | Normal Washed |
| AW | Acid Washed |
| APTES | (3-Aminopropyl) triethoxysilane |
| DTMS | Dodecyltrimethoxysilane |
| GO | Graphene Oxide |
| IX | Ion Exchange |
| LC-MS/MS | Liquid Chromatography–Tandem Mass Spectrometry |
| NOM | Natural Organic Matter |
| EPA | Environmental Protection Agency |
| GAC1 | Coconut-Based Granular Activated Carbon |
| GAC2 | Coal-Based Granular Activated Carbon |
| PFAS | Per- and Polyfluoroalkyl Substances |
References
- Giesy, J.P.; Kannan, K. Global distribution of perfluorooctane sulfonate in wildlife. Environmental science & technology 2001, 35(7), 1339–1342. [Google Scholar]
- Evich, M.G.; et al. Per-and polyfluoroalkyl substances in the environment. Science 2022, 375(6580), eabg9065. [Google Scholar] [CrossRef]
- Buck, R.C.; et al. Perfluoroalkyl and polyfluoroalkyl substances in the environment: terminology, classification, and origins. Integrated environmental assessment and management 2011, 7(4), 513–541. [Google Scholar] [CrossRef]
- Leung, S.C.E.; et al. Physicochemical properties and interactions of perfluoroalkyl substances (PFAS)-Challenges and opportunities in sensing and remediation. Science of the Total Environment 2023, 905, 166764. [Google Scholar] [CrossRef]
- Brunn, H.; et al. PFAS: forever chemicals—persistent, bioaccumulative and mobile. Reviewing the status and the need for their phase out and remediation of contaminated sites. Environmental Sciences Europe 2023, 35(1), 1–50. [Google Scholar]
- Grandjean, P.; Budtz-Jørgensen, E. Immunotoxicity of perfluorinated alkylates: calculation of benchmark doses based on serum concentrations in children. Environmental Health 2013, 12, 1–7. [Google Scholar] [CrossRef]
- Post, G.B.; Cohn, P.D.; Cooper, K.R. Perfluorooctanoic acid (PFOA), an emerging drinking water contaminant: a critical review of recent literature. Environmental research 2012, 116, 93–117. [Google Scholar] [CrossRef]
- Steenland, K.; Zhao, L.; Winquist, A. A cohort incidence study of workers exposed to perfluorooctanoic acid (PFOA). Occupational and environmental medicine 2015, 72(5), 373–380. [Google Scholar] [CrossRef]
- Ibrahim, M.; et al. Per-and polyfluoroalkyl substances and global water resources: an in-depth review of existing regulatory frameworks worldwide. International Journal of Environmental Science and Technology 2025, 1–22. [Google Scholar] [CrossRef]
- Epa, U. Drinking water health advisories for PFOA and PFOS; United States Environmental Protection Agency: USA, 2016. [Google Scholar]
- Takdastan, A.; et al. Perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) in water and edible fish species of Karun River, Ahvaz, Iran: spatial distribution, human health, and ecological risk assessment. International Journal of Environmental Health Research 2024, 34(2), 803–814. [Google Scholar] [CrossRef] [PubMed]
- Wanninayake, D.M. Comparison of currently available PFAS remediation technologies in water: A review. Journal of Environmental Management 2021, 283, 111977. [Google Scholar] [CrossRef] [PubMed]
- Lei, X.; et al. A review of PFAS adsorption from aqueous solutions: Current approaches, engineering applications, challenges, and opportunities. Environmental Pollution 2023, 321, 121138. [Google Scholar] [CrossRef]
- Murray, C.C.; et al. PFAS treatment with granular activated carbon and ion exchange resin: Comparing chain length, empty bed contact time, and cost. Journal of Water Process Engineering 2021, 44, 102342. [Google Scholar] [CrossRef]
- Park, M.; et al. Adsorption of perfluoroalkyl substances (PFAS) in groundwater by granular activated carbons: Roles of hydrophobicity of PFAS and carbon characteristics. Water research 2020, 170, 115364. [Google Scholar] [CrossRef]
- Riegel, M.; Haist-Gulde, B.; Sacher, F. Sorptive removal of short-chain perfluoroalkyl substances (PFAS) during drinking water treatment using activated carbon and anion exchanger. Environmental Sciences Europe 2023, 35(1), 12. [Google Scholar] [CrossRef]
- Murray, C.C.; et al. Removal of per-and polyfluoroalkyl substances using super-fine powder activated carbon and ceramic membrane filtration. Journal of Hazardous Materials 2019, 366, 160–168. [Google Scholar] [CrossRef]
- Nakazawa, Y.; et al. Maximum desorption of perfluoroalkyl substances adsorbed on granular activated carbon used in full-scale drinking water treatment plants. Water Research 2024, 254, 121396. [Google Scholar] [CrossRef]
- Zhang, Y.; et al. Coexisting ions and long-chain per-and polyfluoroalkyl substances (PFAS) inhibit the adsorption of short-chain PFAS by granular activated carbon. Journal of Hazardous Materials 2023, 460, 132378. [Google Scholar] [CrossRef]
- Chen, R.; et al. Performance of in-service granular activated carbon for perfluoroalkyl substances removal under changing water quality conditions. Science of The Total Environment 2022, 848, 157723. [Google Scholar] [CrossRef] [PubMed]
- Gagliano, E.; et al. State of the research on regeneration and reactivation techniques for per-and polyfluoroalkyl substances (PFAS)-laden granular activated carbons (GACs). Current Opinion in Chemical Engineering 2023, 42, 100955. [Google Scholar] [CrossRef]
- Vakili, M.; et al. Regeneration of exhausted adsorbents after PFAS adsorption: A critical review. Journal of Hazardous Materials 2024, 134429. [Google Scholar] [CrossRef] [PubMed]
- Mian, M.M.; et al. Recent advances in activated carbon driven PFAS removal: structure-adsorption relationship and new adsorption mechanisms. Frontiers of Environmental Science & Engineering 2025, 19(6), 78. [Google Scholar] [CrossRef]
- Ellis, A.C.; et al. Life cycle assessment and life cycle cost analysis of anion exchange and granular activated carbon systems for remediation of groundwater contaminated by per-and polyfluoroalkyl substances (PFASs). Water Research 2023, 243, 120324. [Google Scholar] [CrossRef]
- Karbassiyazdi, E.; et al. A juxtaposed review on adsorptive removal of PFAS by metal-organic frameworks (MOFs) with carbon-based materials, ion exchange resins, and polymer adsorbents. Chemosphere 2023, 311, 136933. [Google Scholar] [CrossRef]
- Chow, S.J.; et al. Comparative investigation of PFAS adsorption onto activated carbon and anion exchange resins during long-term operation of a pilot treatment plant. Water research 2022, 226, 119198. [Google Scholar] [CrossRef]
- Woodard, S.; Berry, J.; Newman, B. Ion exchange resin for PFAS removal and pilot test comparison to GAC. Remediation Journal 2017, 27(3), 19–27. [Google Scholar] [CrossRef]
- Zeidabadi, F.A.; et al. Managing PFAS exhausted Ion-exchange resins through effective regeneration/electrochemical process. Water Research 2024, 255, 121529. [Google Scholar] [CrossRef] [PubMed]
- Boyer, T.H.; et al. Life cycle environmental impacts of regeneration options for anion exchange resin remediation of PFAS impacted water. Water research 2021, 207, 117798. [Google Scholar] [CrossRef]
- Liu, Y.-L.; Sun, M. Ion exchange removal and resin regeneration to treat per-and polyfluoroalkyl ether acids and other emerging PFAS in drinking water. Water research 2021, 207, 117781. [Google Scholar] [CrossRef]
- Mastropietro, T.F.; et al. Reverse osmosis and nanofiltration membranes for highly efficient PFASs removal: overview, challenges and future perspectives. Dalton Transactions 2021, 50(16), 5398–5410. [Google Scholar] [CrossRef]
- Liu, C.; et al. Evaluating the efficiency of nanofiltration and reverse osmosis membrane processes for the removal of per-and polyfluoroalkyl substances from water: A critical review. Separation and Purification Technology 2022, 302, 122161. [Google Scholar] [CrossRef]
- Ma, Q., et al., Evaluation of Commercial Nanofiltration and Reverse Osmosis Membrane Filtration to Remove Pfass: Effects of Transmembrane Pressures and Water Matrices. Available at SSRN 4524126, 2023.
- Cardoso, I.M.; Pinto da Silva, L.; Esteves da Silva, J.C. Nanomaterial-based advanced oxidation/reduction processes for the degradation of PFAS. Nanomaterials 2023, 13(10), 1668. [Google Scholar] [CrossRef]
- Ahmed, M.B.; et al. Advanced treatment technologies efficacies and mechanism of per-and poly-fluoroalkyl substances removal from water. Process Safety and Environmental Protection 2020, 136, 1–14. [Google Scholar] [CrossRef]
- Duinslaeger, N.; Radjenovic, J. Electrochemical degradation of per-and polyfluoroalkyl substances (PFAS) using low-cost graphene sponge electrodes. Water research 2022, 213, 118148. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; et al. Sonochemical degradation of poly-and perfluoroalkyl substances–A review. Ultrasonics Sonochemistry 2020, 69, 105245. [Google Scholar] [CrossRef]
- Benettayeb, A.; et al. Natural adsorbents for the removal of emerging pollutants and its adsorption mechanisms, in Sustainable remediation technologies for emerging pollutants in aqueous environment; 2024; Elsevier; pp. 63–78. [Google Scholar]
- Kordala, N.; Wyszkowski, M. Zeolite properties, methods of synthesis, and selected applications. Molecules 2024, 29(5), 1069. [Google Scholar] [CrossRef]
- Krysenko, D.; Demchenko, V.Y. Effect of the Composition of the Zeolite Framework of Natural Clinoptilolites on the Exchange of Cations Na+ On Ca2+. Theoretical and Experimental Chemistry 2023, 59(3), 222–230. [Google Scholar] [CrossRef]
- Willett, A. The Versatility of Surface-Modified Clay Adsorbents for PFAS Treatment. in 2022 Emerging Contaminants in the Environment Conference (ECEC22). 2022.
- Khodabakhshloo, N.; Biswas, B. Adsorption of aqueous perfluorooctane sulfonate by raw and oleylamine-modified Iranian diatomite and zeolite: Material and application insight. Applied Clay Science 2023, 244, 107101. [Google Scholar] [CrossRef]
- Mukhopadhyay, R.; et al. Natural and engineered clays and clay minerals for the removal of poly-and perfluoroalkyl substances from water: State-of-the-art and future perspectives. Advances in Colloid and Interface Science 2021, 297, 102537. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; et al. Molecular mechanisms of per-and polyfluoroalkyl substances on a modified clay: a combined experimental and molecular simulation study. Water Research 2020, 184, 116166. [Google Scholar] [CrossRef]
- Stebel, E.K.; et al. Absorption of short-chain to long-chain perfluoroalkyl substances using swellable organically modified silica. Environmental Science: Water Research & Technology 2019, 5(11), 1854–1866. [Google Scholar]
- Mohammadzadeh Kakhki, R.; Zirjanizadeh, S.; Mohammadpoor, M. A review of clinoptilolite, its photocatalytic, chemical activity, structure and properties: in time of artificial intelligence. Journal of Materials Science 2023, 58(26), 10555–10575. [Google Scholar] [CrossRef]
- Grifasi, N.; et al. Fundamental properties and sustainable applications of the natural zeolite clinoptilolite. Environmental Science and Pollution Research 2024, 1–36. [Google Scholar] [CrossRef]
- Ledesma, B.; et al. Fundamental study on the thermal regeneration stages of exhausted activated carbons: kinetics. Journal of Thermal Analysis and Calorimetry 2014, 115(1), 537–543. [Google Scholar] [CrossRef]
- McNamara, J.D.; et al. Comparison of activated carbons for removal of perfluorinated compounds from drinking water. Journal-American Water Works Association 2018, 110(1), E2–E14. [Google Scholar] [CrossRef]
- He, Y.; et al. Advancing PFAS sorbent design: mechanisms, challenges, and perspectives. ACS Materials Au 2023, 4(2), 108–114. [Google Scholar] [CrossRef]
- Lenka, S.P.; et al. Adsorption mechanisms of short-chain and ultrashort-chain PFAS on anion exchange resins and activated carbon. Environmental Science: Water Research & Technology 2024, 10(5), 1280–1293. [Google Scholar]
- Geneyton, A.; et al. Synergistic adsorption of lanthanum ions and fatty acids for efficient rare-earth phosphate recovery: Surface analysis and ab initio molecular dynamics studies. Applied Surface Science 2020, 526, 146725. [Google Scholar] [CrossRef]
- Penchoff, D.A.; et al. Evaluations of molecular modeling and machine learning for predictive capabilities in binding of lanthanum and actinium with carboxylic acids. Journal of Radioanalytical and Nuclear Chemistry 2022, 331(12), 5469–5485. [Google Scholar] [CrossRef]
- Sthoer, A.; et al. La3+ and Y3+ interactions with the carboxylic acid moiety at the liquid/vapor interface: Identification of binding complexes, charge reversal, and detection limits. Journal of Colloid and Interface Science 2022, 608, 2169–2180. [Google Scholar] [CrossRef]
- Sontakke, A.D.; Tiwari, S.; Purkait, M.K. A comprehensive review on graphene oxide-based nanocarriers: Synthesis, functionalization and biomedical applications. FlatChem 2023, 38, 100484. [Google Scholar] [CrossRef]
- Mahanty, B.; et al. Efficient removal of perfluorooctanoic acid from aqueous matrices using cationic surfactant functionalized graphene oxide nanocomposite: RSM and ANN modeling, and adsorption behaviour. Journal of Water Process Engineering 2024, 68, 106448. [Google Scholar] [CrossRef]
- Xu, D.; et al. Graphene oxide with 1-nm-thick adlayer for efficient and near-instant removal of per-and polyfluoroalkyl substances. National Science Review 2025, 12(5), nwaf092. [Google Scholar] [CrossRef]
- Roy, J.W.; et al. Per-and poly-fluoroalkyl substances (PFAS) contamination of surface waters by historic landfills via groundwater plumes: ecosystem exposure and downstream mass loading. Environmental Science: Processes & Impacts 2025, 27(4), 1074–1087. [Google Scholar]
- Ersoy, B.; Çelik, M.S. Electrokinetic properties of clinoptilolite with mono-and multivalent electrolytes. Microporous and Mesoporous Materials 2002, 55(3), 305–312. [Google Scholar] [CrossRef]
- Ponge, C.A.; et al. Designing zeolites for the removal of aqueous PFAS: a perspective. Industrial Chemistry & Materials 2024, 2(2), 270–275. [Google Scholar]
- Burkhardt, J.B.; et al. Polanyi adsorption potential theory for estimating PFAS treatment with granular activated carbon. Journal of water process engineering 2023, 53, 103691. [Google Scholar] [CrossRef]
- Sayadi, S.; Brouillette, F. Silylation of phosphorylated cellulosic fibers with an aminosilane. Carbohydrate Polymers 2024, 343, 122500. [Google Scholar] [CrossRef]
- Dunmyer, M.; et al. Molecular scale adsorption behavior of per-and poly-fluoroalkyl substances (PFAS) on model surfaces. Chemical Engineering Journal 2024, 497, 154286. [Google Scholar] [CrossRef]
- Welchert, J.; et al. Investigation into the adhesion properties of PFAS on model surfaces. RSC Applied Interfaces 2024, 1(6), 1265–1275. [Google Scholar] [CrossRef]
- Gao, Y.; Du, J.; Gu, T. Hemimicelle formation of cationic surfactants at the silica gel–water interface. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases 1987, 83(8), 2671–2679. [Google Scholar] [CrossRef]
- Qu, Y.; et al. Hydrothermal synthesis, structures and spectroscopy of 2D lanthanide coordination polymers built from helical chains:[Ln2 (BDC) 3 (H2O) 2] n (Ln= Sm, 1; Ln= Eu, 2; BDC= 1, 3-benzenedicarboxylate). Journal of molecular structure 2005, 734(1-3), 7–13. [Google Scholar] [CrossRef]
- Huang, H.-H.; Twidwell, L.; Young, C. Point of Zero Charge (PZC) and Double Layer Adsorption–An Equilibrium Calculation Approach. Computational Analysis in Hydrometallurgy, COM, 2005.
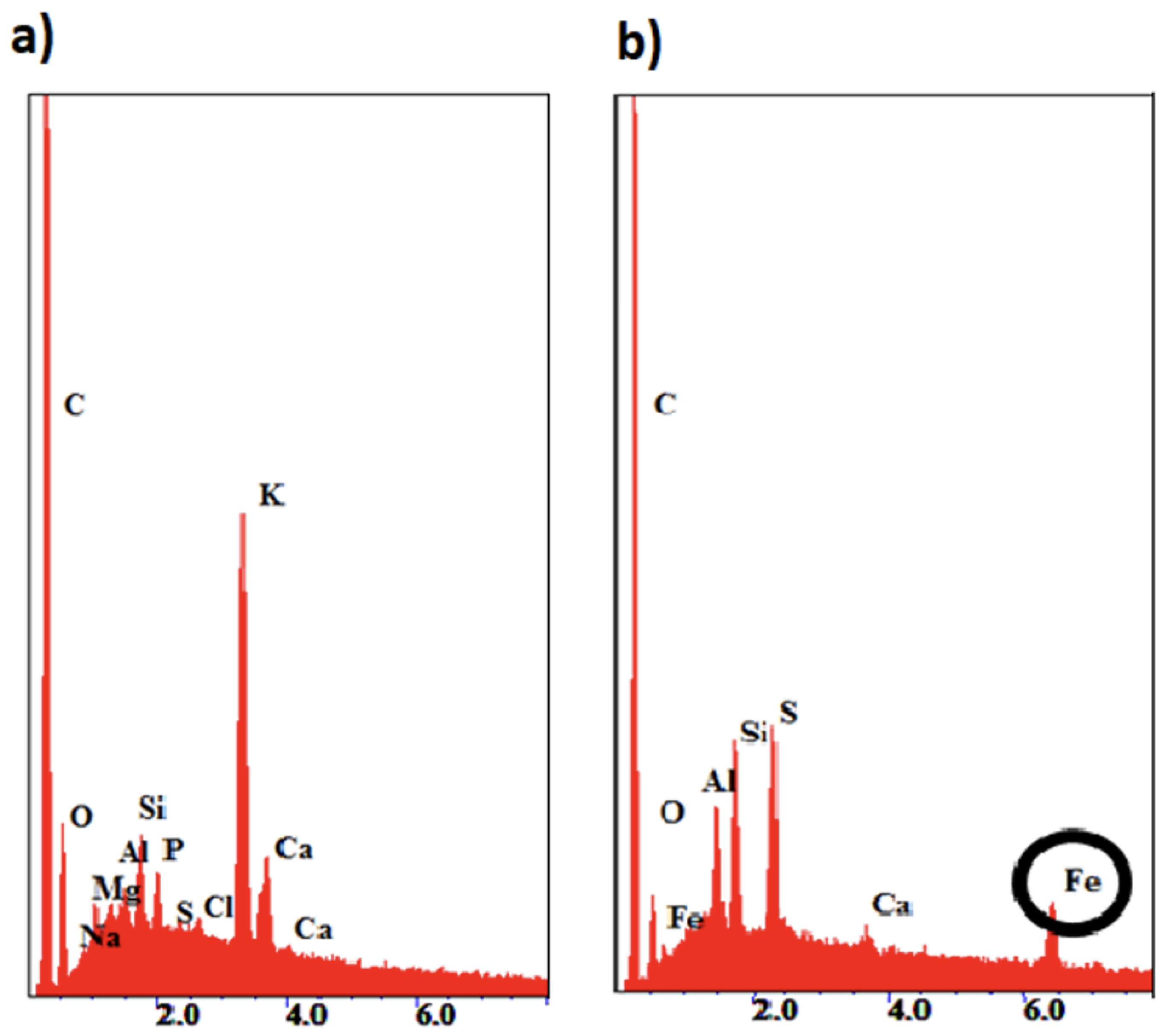
| Sample name | Description |
|---|---|
| GAC1 | Coconut based granular activated carbon, washed with hot water |
| GAC2 | Coal based (source 1) granular activated carbo, washed with hot water |
| AZ | Australian zeolite, normal washed |
| AZ-DTMS-NW | Australian zeolite coated with DTMS normal washed |
| AZ-DTMS-AW | Australian zeolite coated with DTMS acid washed |
| AZ-A-NW | Australian zeolite coated with APTES normal washed |
| AZ-A-AW | Australian zeolite coated with APTES acid washed |
| AZ-D:A (1:1)-NW | Normal washed Australian zeolite coated with a mixture of 1 to 1 DTMS and APTES |
| AZ-D:A (1:3)-NW | Normal washed Australian zeolite coated with a mixture of 1 to 3 DTMS and APTES |
| AZ-D:A (3:1)-NW | Normal washed Australian zeolite coated with a mixture of 3 to 1 DTMS and APTES |
| AZ-D:A (1:1)-AW | Acid washed Australian zeolite coated with a mixture of 1 to 1 DTMS and APTES |
| AZ-D:A (1:3)-AW | Acid washed Australian zeolite coated with a mixture of 1 to 3 DTMS and APTES |
| AZ-D:A (3:1)-AW | Acid washed Australian zeolite coated with a mixture of 3 to 1 DTMS and APTES |
| AZ-D+A-NW | Normal washed zeolite, the sample is consisted of ratio of 1 portion APTES coated grains and 1 portion DTMS coated grains |
| AZ-D+A-AW | Acid washed zeolite, the sample is consisted of ratio of 1 portion APTES coated grains and 1 portion DTMS coated grains |
| AZ-FE-AW | Acid washed Australian zeolite impregnated with Fe |
| AZ-FE-NW | Normal washed Australian zeolite impregnated with Fe |
| AZ-La-NW | Normal washed Australian zeolite, coated with La |
| AZ-GO | Normal washed Australian zeolite, coated with graphene oxide |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




