Submitted:
10 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
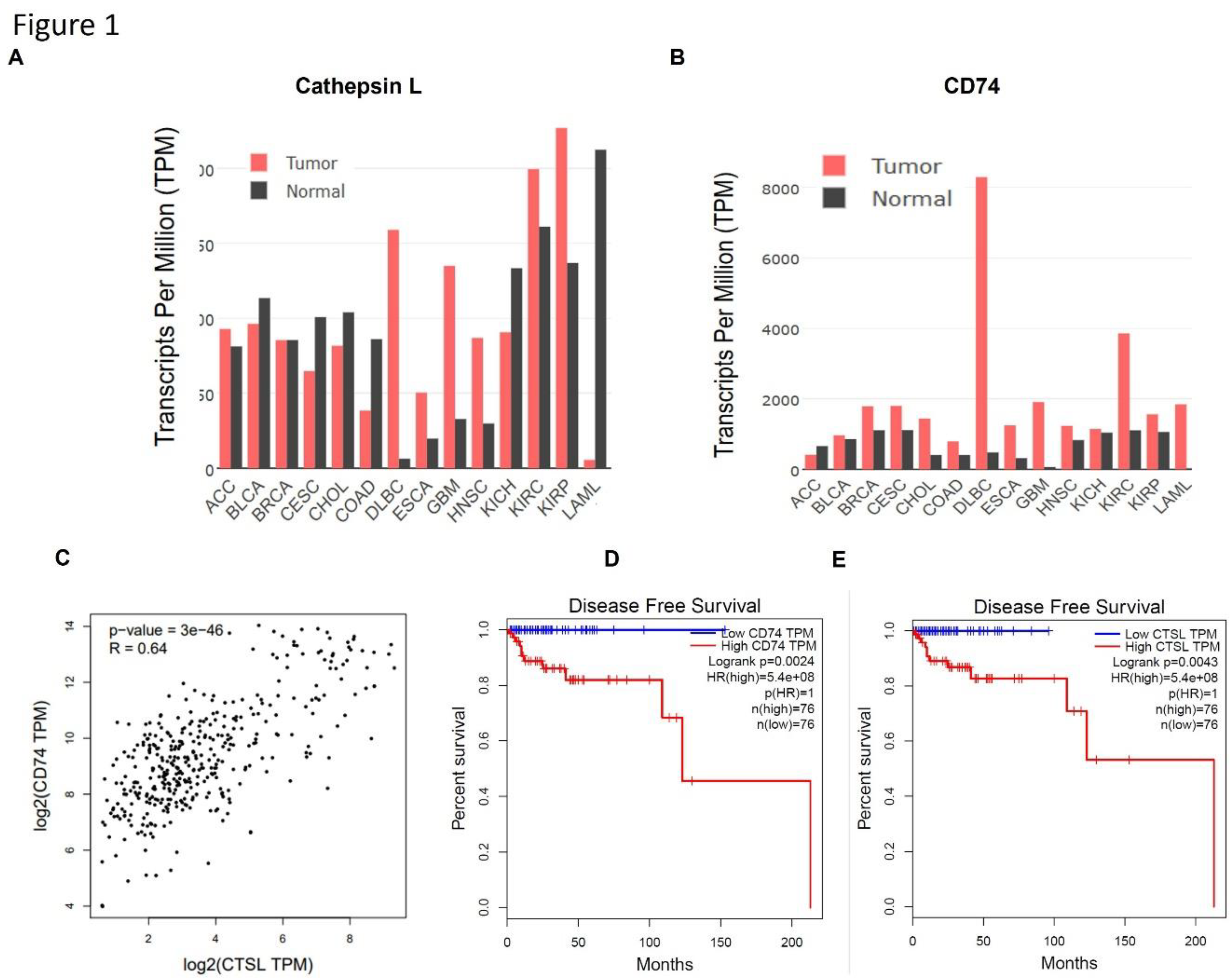
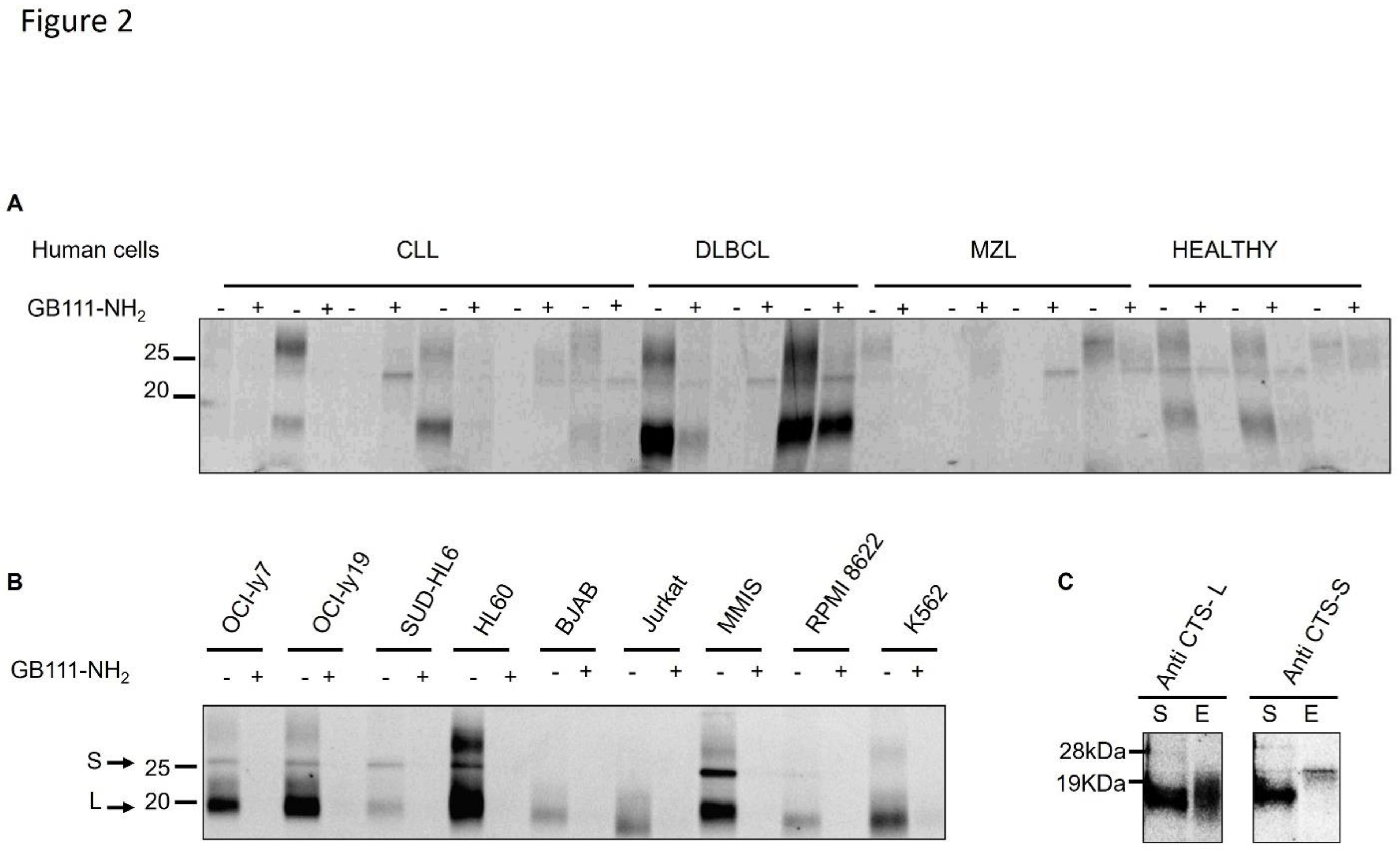
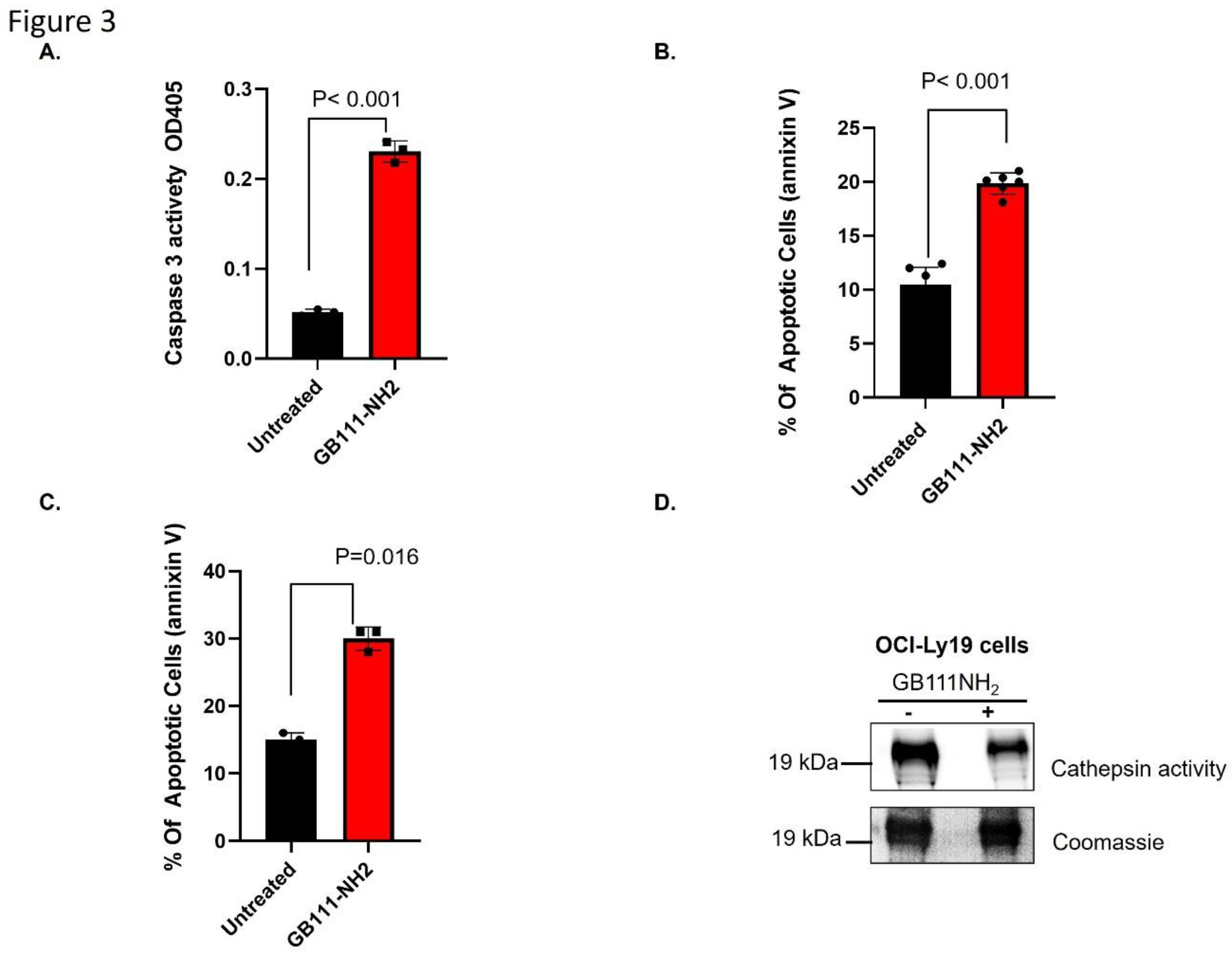
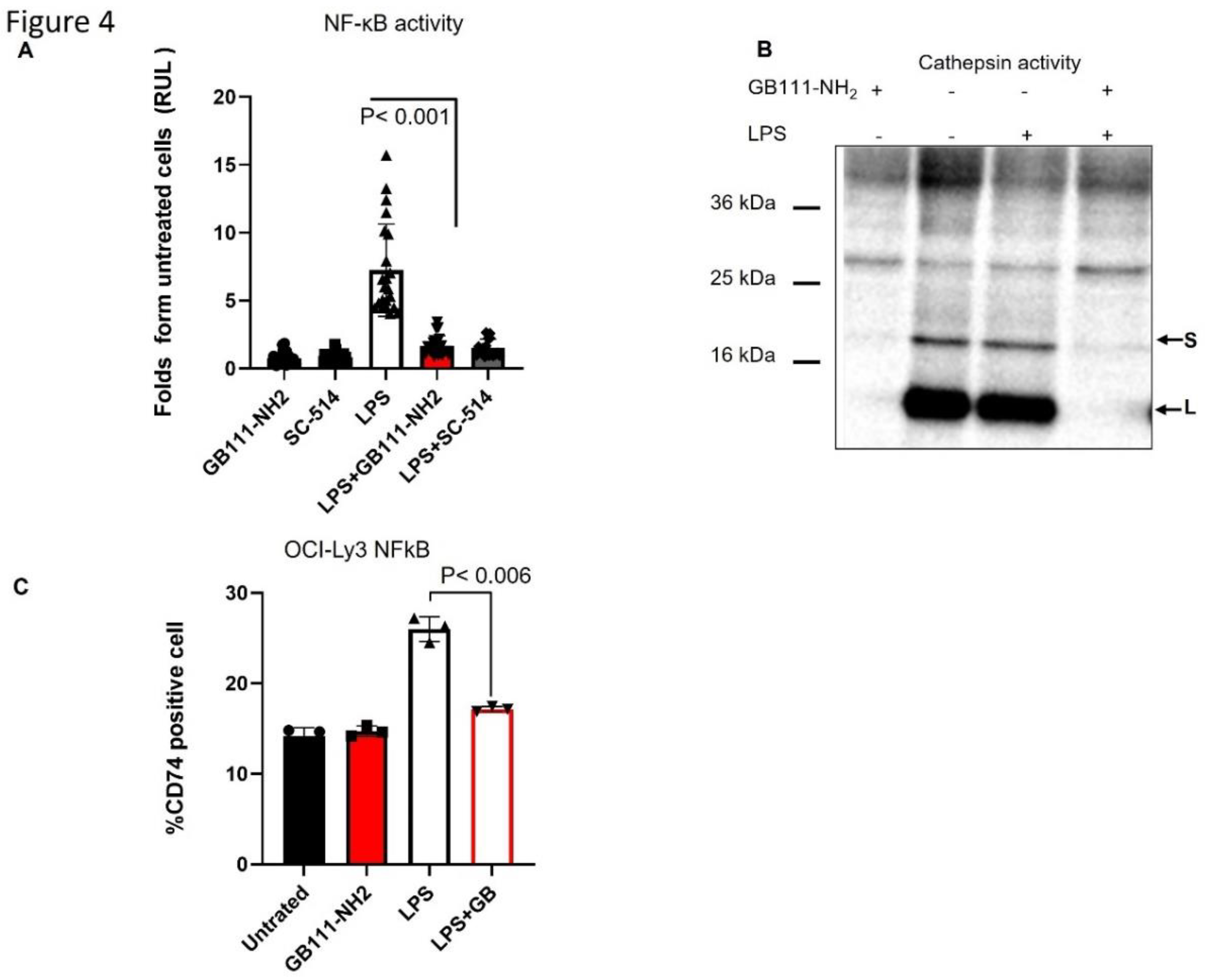
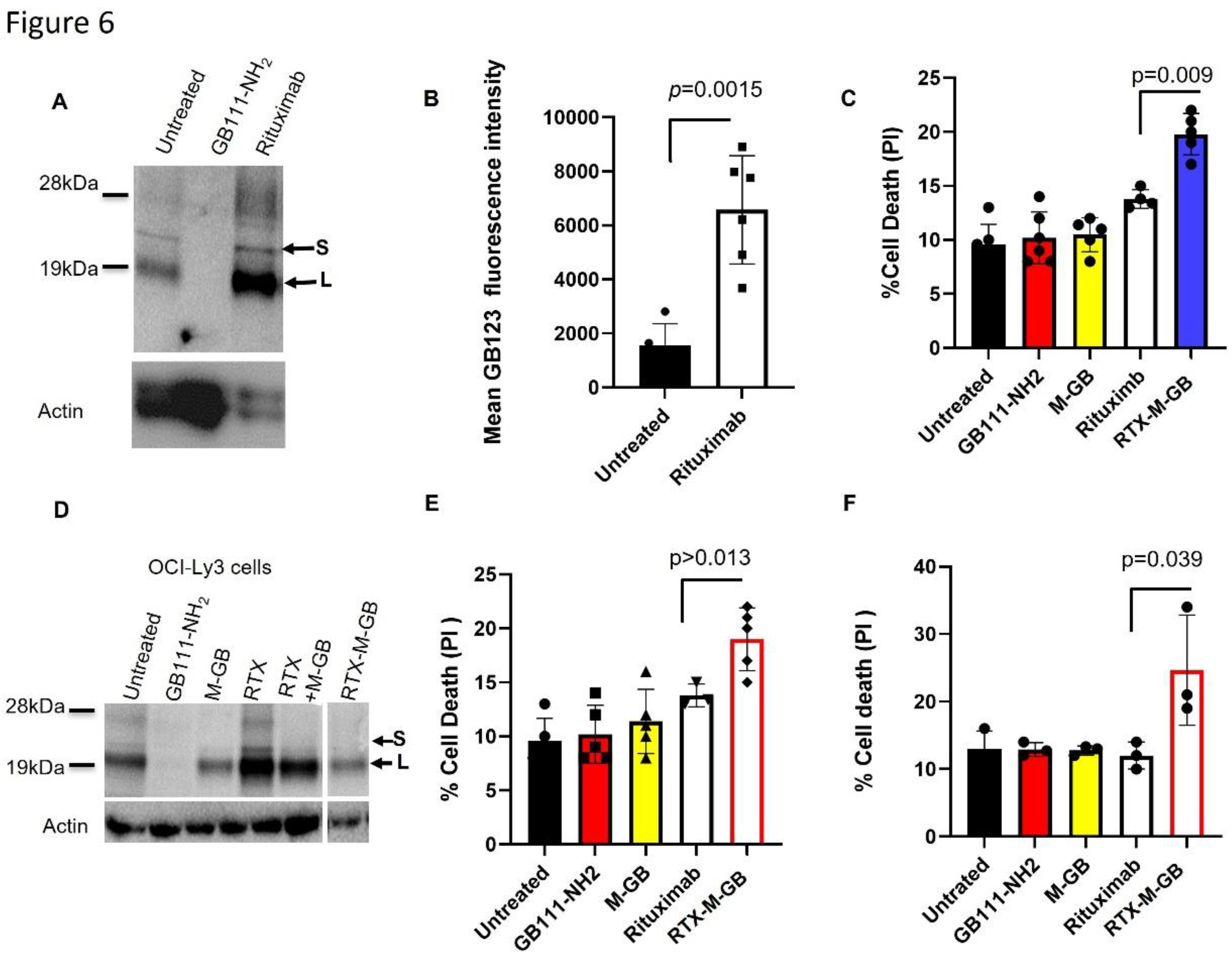
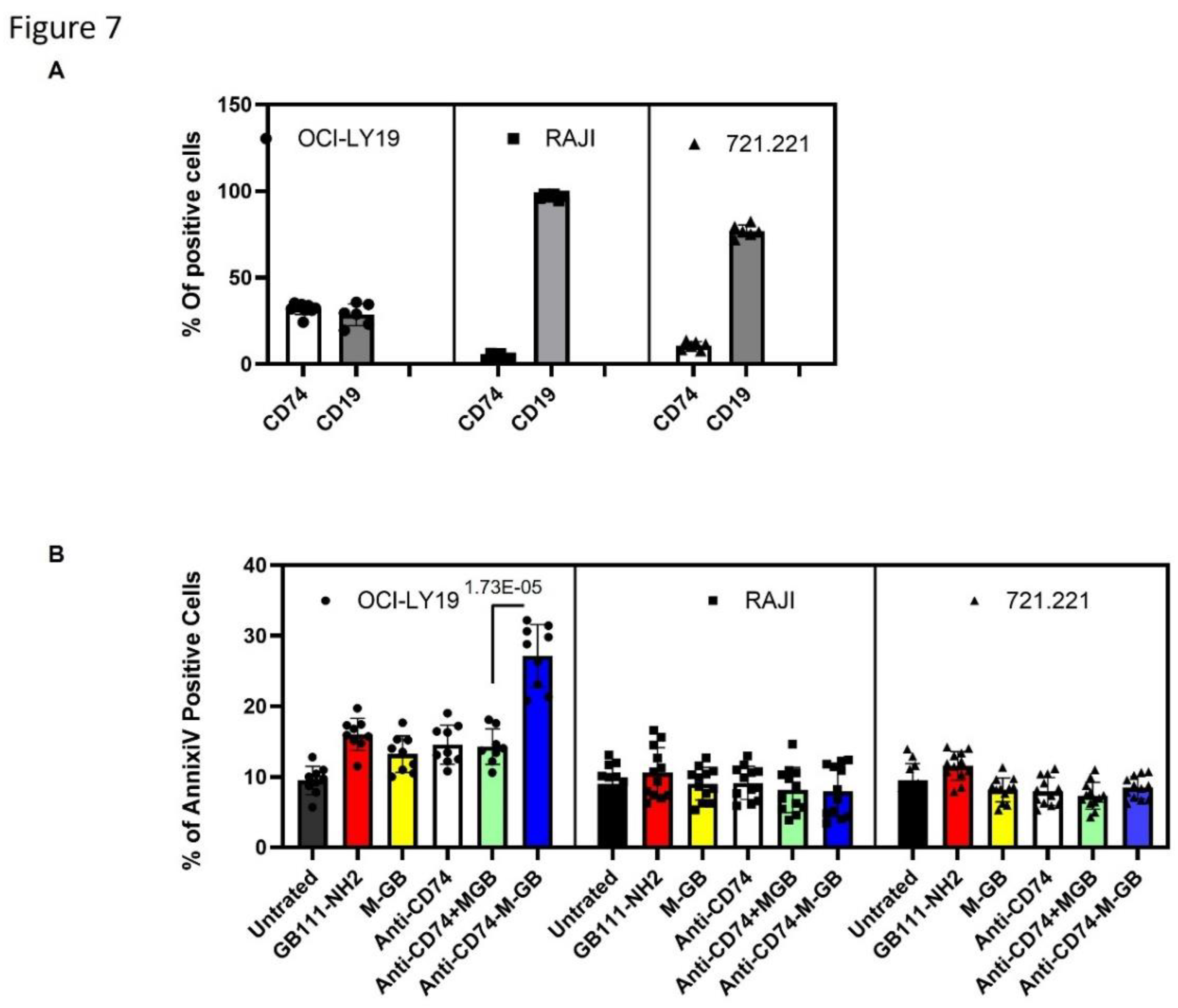
Results
Discussion
Ethical approval and consent to participate
Consent for publication
Availability of data and materials
Funding
Author Contributions
Competing interests
References
- Susanibar-Adaniya, S. & Barta, S. K. 2021 Update on Diffuse large B cell lymphoma: A review of current data and potential applications on risk stratification and management. Am J Hematol 96, 617-629 (2021). [CrossRef]
- Coiffier, B. & Sarkozy, C. Diffuse large B-cell lymphoma: R-CHOP failure-what to do? Hematology Am Soc Hematol Educ Program 2016, 366-378 (2016). [CrossRef]
- Schmitz, R. et al. Burkitt lymphoma pathogenesis and therapeutic targets from structural and functional genomics. Nature 490, 116-120 (2012). [CrossRef]
- Stiff, P. J. et al. Autologous transplantation as consolidation for aggressive non-Hodgkin's lymphoma. N Engl J Med 369, 1681-1690 (2013). [CrossRef]
- Li, X. et al. Targeting CD74 in B-cell non-Hodgkin lymphoma with the antibody-drug conjugate STRO-001. Oncotarget 14, 1-13 (2023). [CrossRef]
- Morin, R. D., Arthur, S. E. & Hodson, D. J. Molecular profiling in diffuse large B-cell lymphoma: why so many types of subtypes? Br J Haematol 196, 814-829 (2022). [CrossRef]
- Bock, A. M. & Epperla, N. Therapeutic landscape of primary refractory and relapsed diffuse large B-cell lymphoma: Recent advances and emerging therapies. J Hematol Oncol 18, 68 (2025). [CrossRef]
- Thiruvengadam, S. K. et al. Cost-effectiveness of polatuzumab vedotin combined with chemoimmunotherapy in untreated diffuse large B-cell lymphoma. Blood 140, 2697-2708 (2022). [CrossRef]
- Song, Y. Q. et al. Glofitamab monotherapy induces high complete response rates and manageable safety in Chinese patients with heavily pretreated relapsed or refractory diffuse large B-cell lymphoma. Haematologica 109, 1269-1273 (2024). [CrossRef]
- García-Sancho, A. M., Cabero, A. & Gutiérrez, N. C. Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma: New Approved Options. J Clin Med 13 (2023). [CrossRef]
- Löser, R. & Pietzsch, J. Cysteine cathepsins: their role in tumor progression and recent trends in the development of imaging probes. Front Chem 3, 37 (2015). [CrossRef]
- Radisky, E. S. Extracellular proteolysis in cancer: Proteases, substrates, and mechanisms in tumor progression and metastasis. J Biol Chem 300, 107347 (2024). [CrossRef]
- Almalki, A. A. et al. Targeting Cathepsin L in Cancer Management: Leveraging Machine Learning, Structure-Based Virtual Screening, and Molecular Dynamics Studies. Int J Mol Sci 24 (2023). [CrossRef]
- Zhao, K., Sun, Y., Zhong, S. & Luo, J. L. The multifaceted roles of cathepsins in immune and inflammatory responses: implications for cancer therapy, autoimmune diseases, and infectious diseases. Biomark Res 12, 165 (2024). [CrossRef]
- Wilkinson, R. D. et al. CCL2 is transcriptionally controlled by the lysosomal protease cathepsin S in a CD74-dependent manner. Oncotarget 6, 29725-29739 (2015). [CrossRef]
- Colella, R. et al. Induction of cell death in neuroblastoma by inhibition of cathepsins B and L. Cancer Lett 294, 195-203 (2010). [CrossRef]
- Zhao, S. et al. High frequency of CD74 expression in lymphomas: implications for targeted therapy using a novel anti-CD74-drug conjugate. J Pathol Clin Res 5, 12-24 (2019). [CrossRef]
- Berkova, Z. et al. CD74 interferes with the expression of fas receptor on the surface of lymphoma cells. J Exp Clin Cancer Res 33, 80 (2014). [CrossRef]
- Wang, R. et al. Antibody–Drug Conjugates (ADCs): current and future biopharmaceuticals. Journal of Hematology & Oncology 18, 51 (2025). [CrossRef]
- Blum, G. et al. Dynamic imaging of protease activity with fluorescently quenched activity-based probes. Nature Chemical Biology 1, 203-209 (2005).
- Blum, G., von Degenfeld, G., Merchant, M. J., Blau, H. M. & Bogyo, M. Noninvasive optical imaging of cysteine protease activity using fluorescently quenched activity-based probes. Nature Chemical Biology 3, 668-677 (2007).
- Grønbaek, K. & Jäättelä, M. Engaging the lysosomal compartment to combat B cell malignancies. J Clin Invest 119, 2133-2136 (2009). [CrossRef]
- Chavez, J. C. & Locke, F. L. CAR T cell therapy for B-cell lymphomas. Best Pract Res Clin Haematol 31, 135-146 (2018). [CrossRef]
- Rudzińska, M. et al. The Role of Cysteine Cathepsins in Cancer Progression and Drug Resistance. Int J Mol Sci 20 (2019). [CrossRef]
- Michot, J. M. et al. Clinical significance of the loss of CD20 antigen on tumor cells in patients with relapsed or refractory follicular lymphoma. Cancer Drug Resist 4, 710-718 (2021). [CrossRef]
- Zhang, J. et al. Cystatin m: a novel candidate tumor suppressor gene for breast cancer. Cancer Res 64, 6957-6964 (2004). [CrossRef]
- Mohamed, M. M. & Sloane, B. F. Cysteine cathepsins: multifunctional enzymes in cancer. Nat Rev Cancer 6, 764-775 (2006). [CrossRef]
- Zhang, L., Wang, H. & Xu, J. Cathepsin S as a cancer target. Neoplasma 62, 16-26 (2015).
- Neumann, C. S. et al. Targeted Delivery of Cytotoxic NAMPT Inhibitors Using Antibody-Drug Conjugates. Mol Cancer Ther 17, 2633-2642 (2018). [CrossRef]
- Conilh, L., Sadilkova, L., Viricel, W. & Dumontet, C. Payload diversification: a key step in the development of antibody–drug conjugates. Journal of Hematology & Oncology 16, 3 (2023). [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



