Submitted:
18 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Background
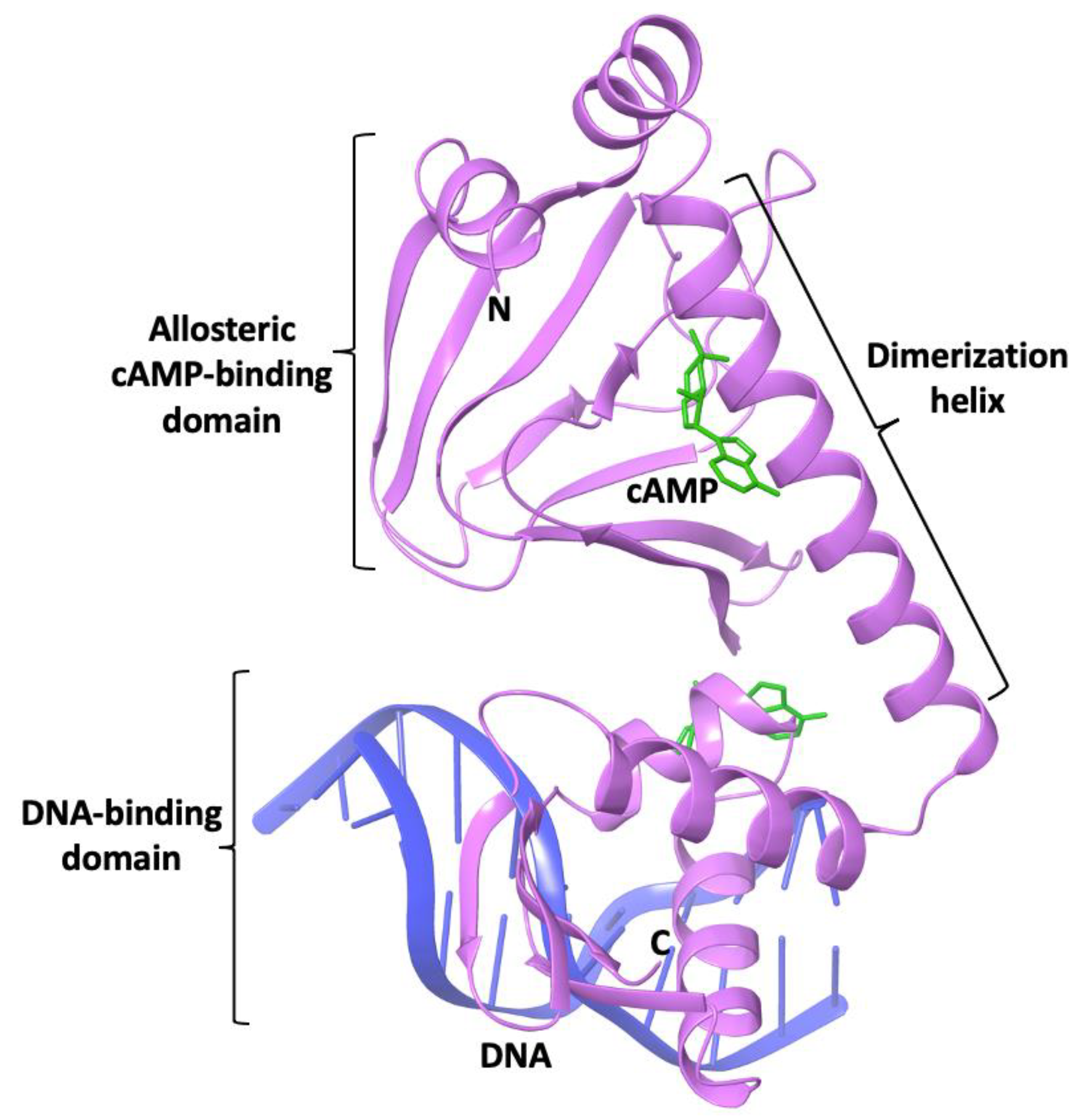
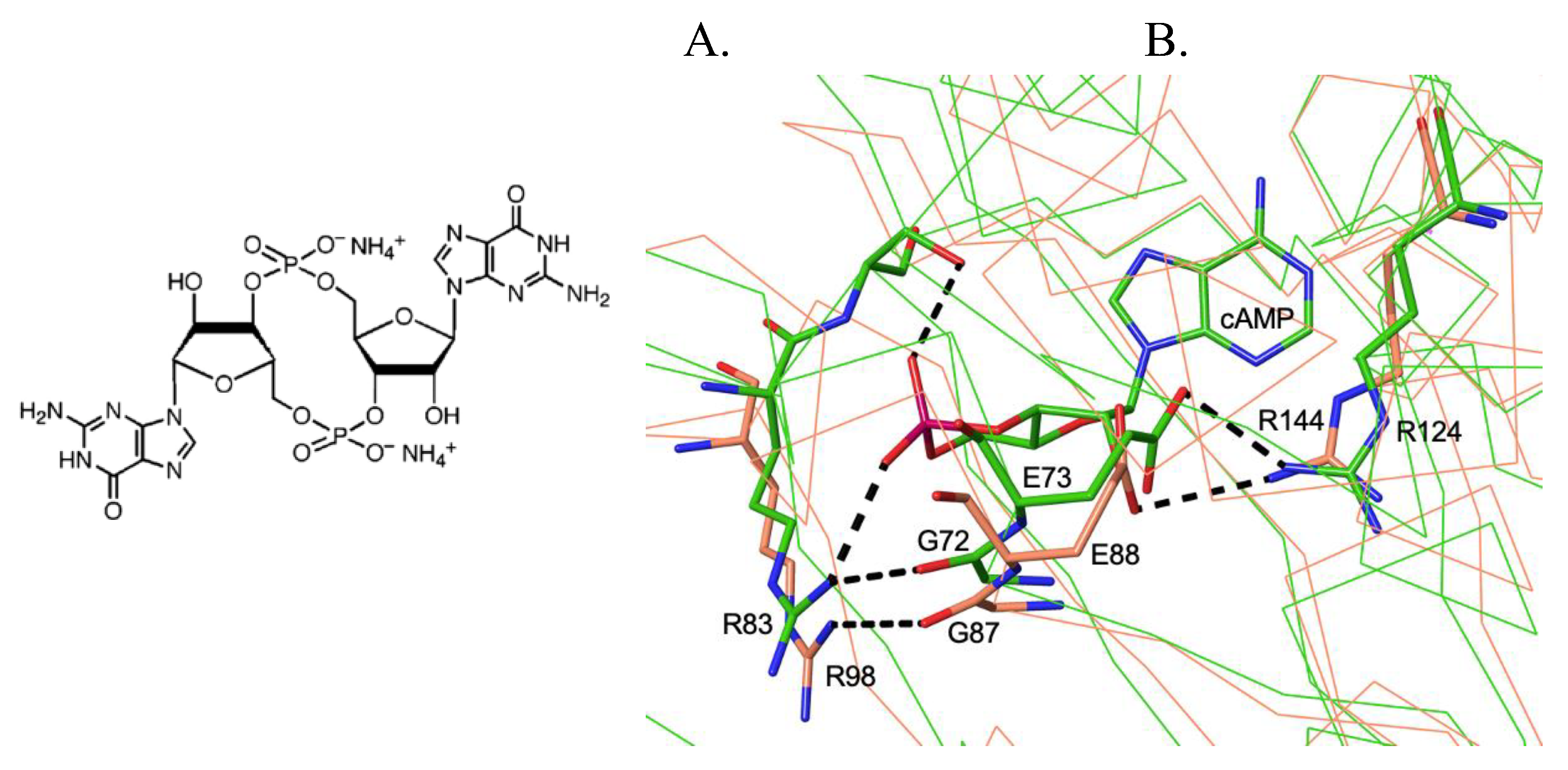
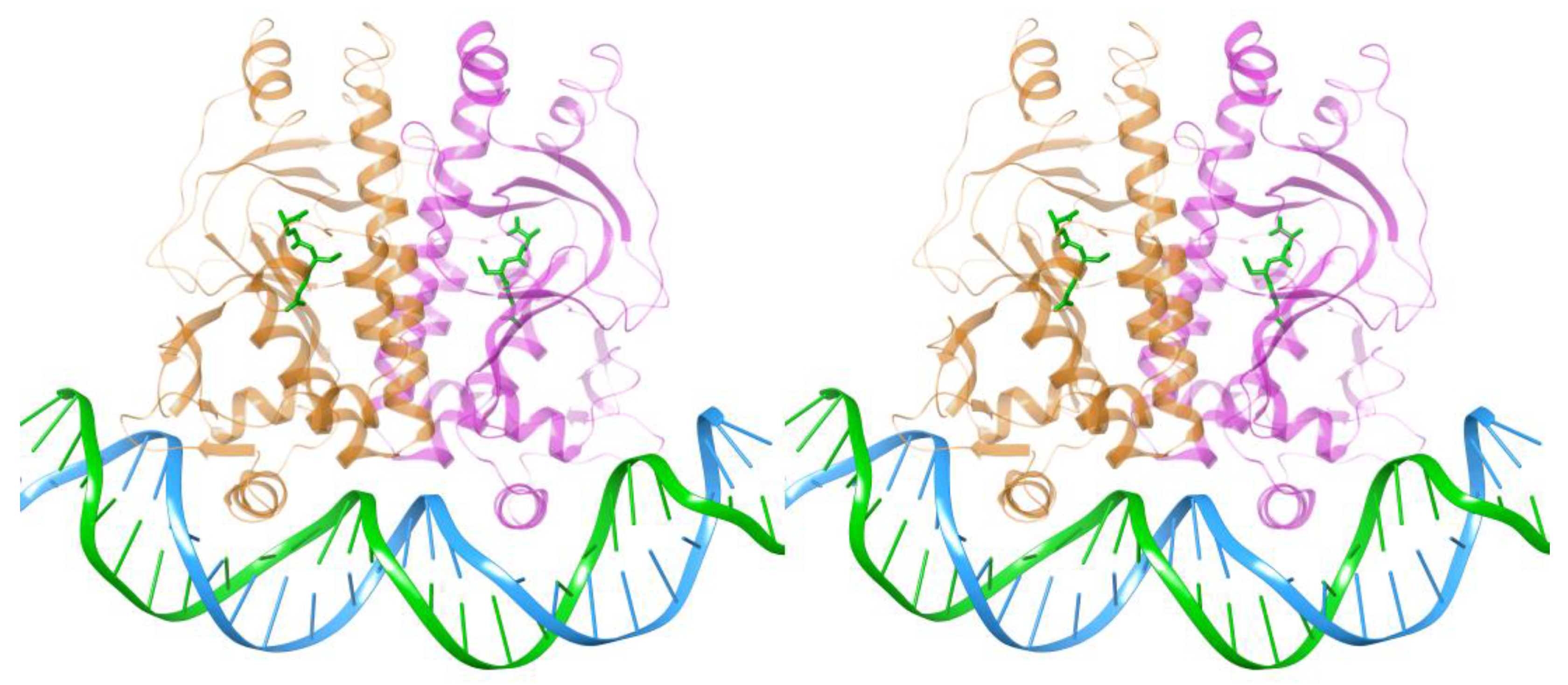
3. Structural Similarities and Differences in the Cavity of the N-Terminal Domain
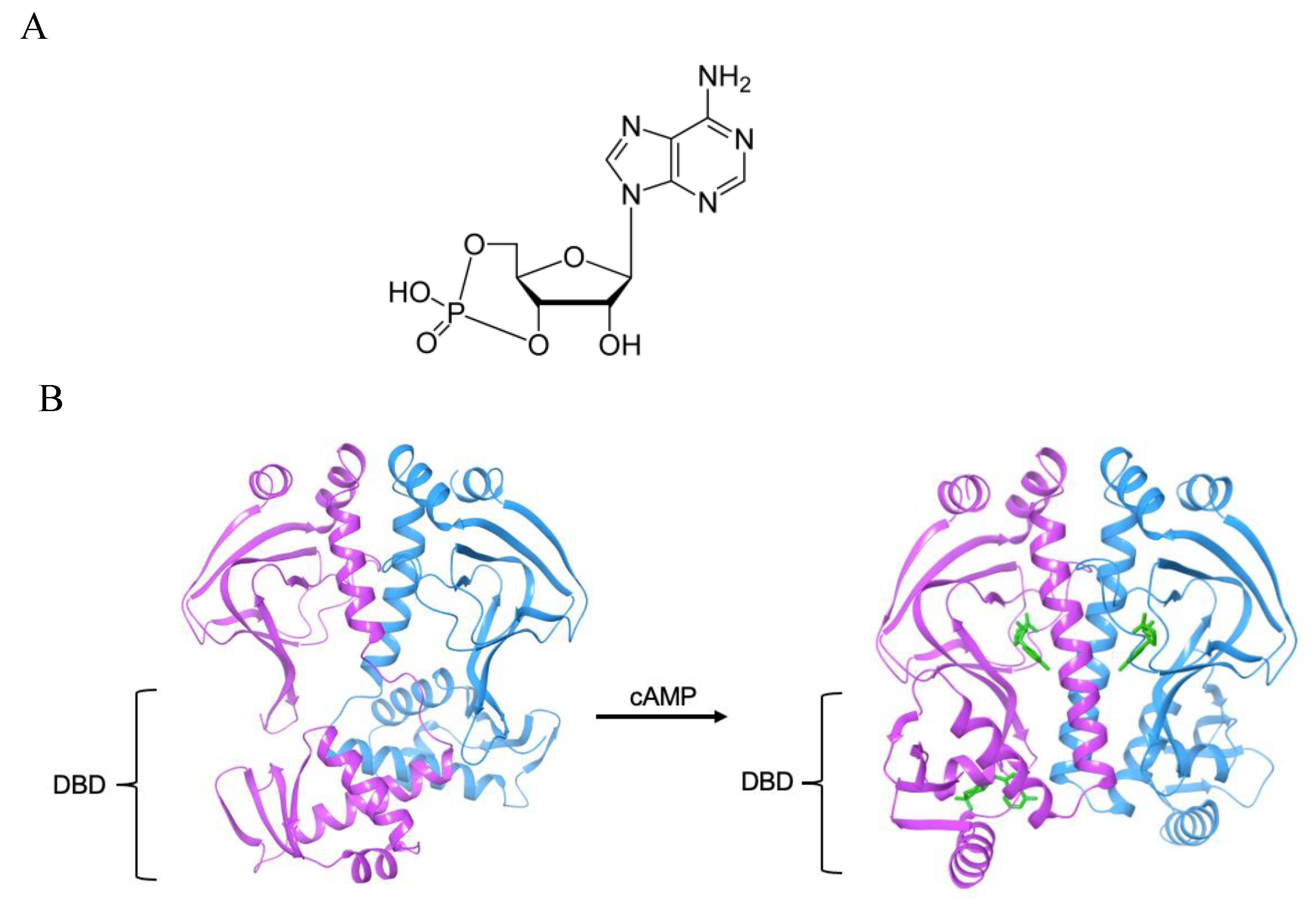
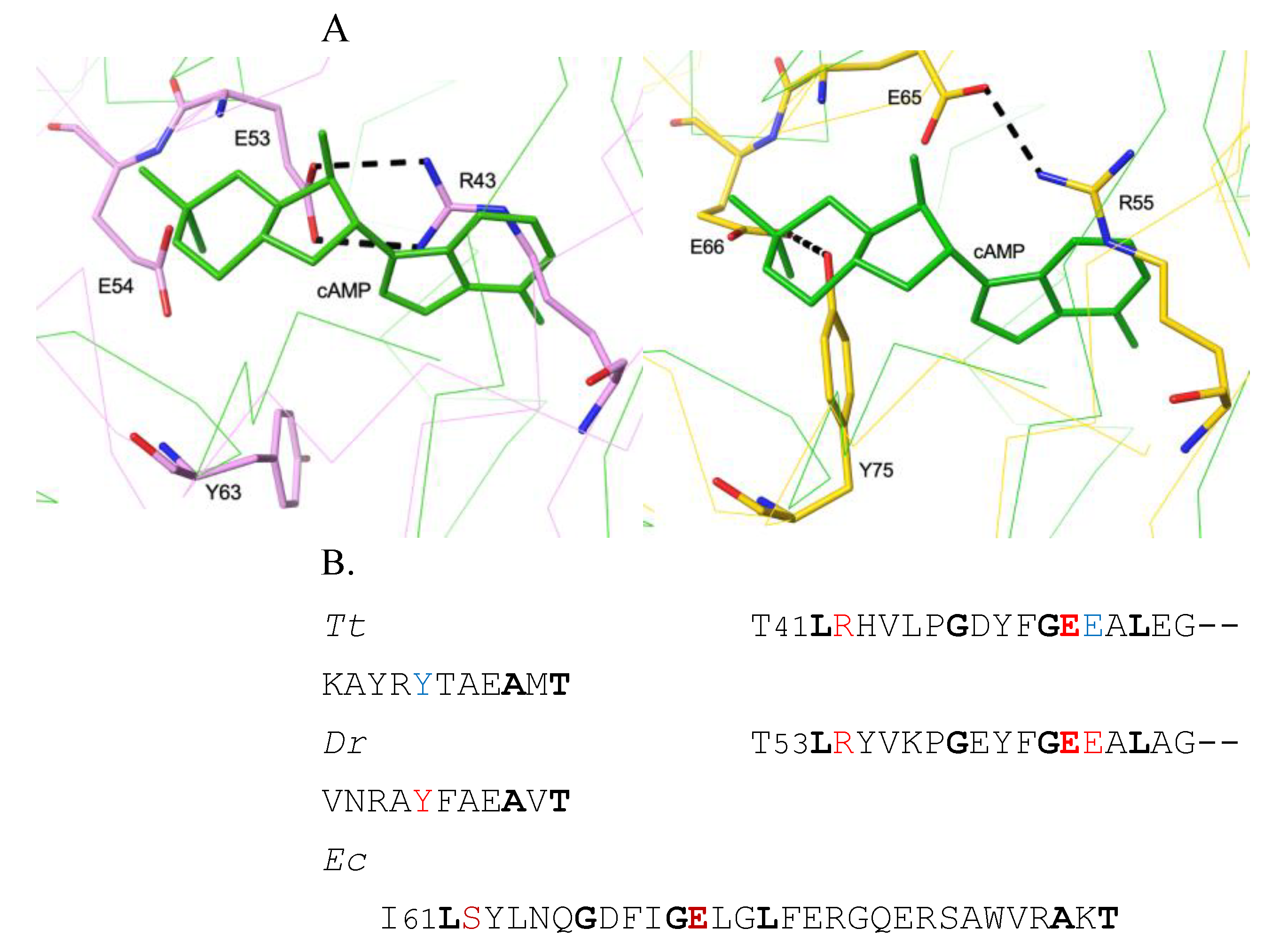
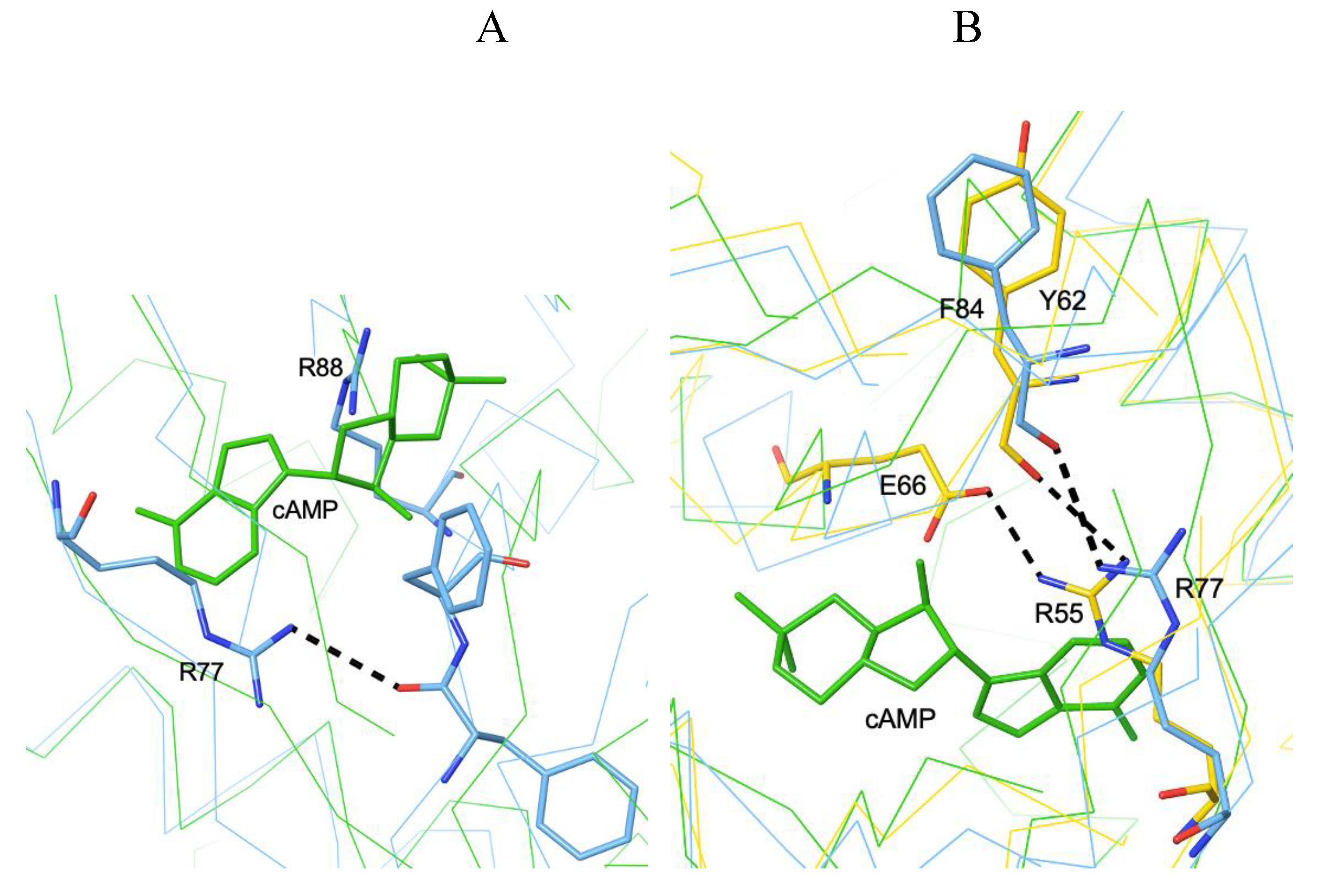
3.1. The Allosteric Cavity in Members of the FNR/CRP Family
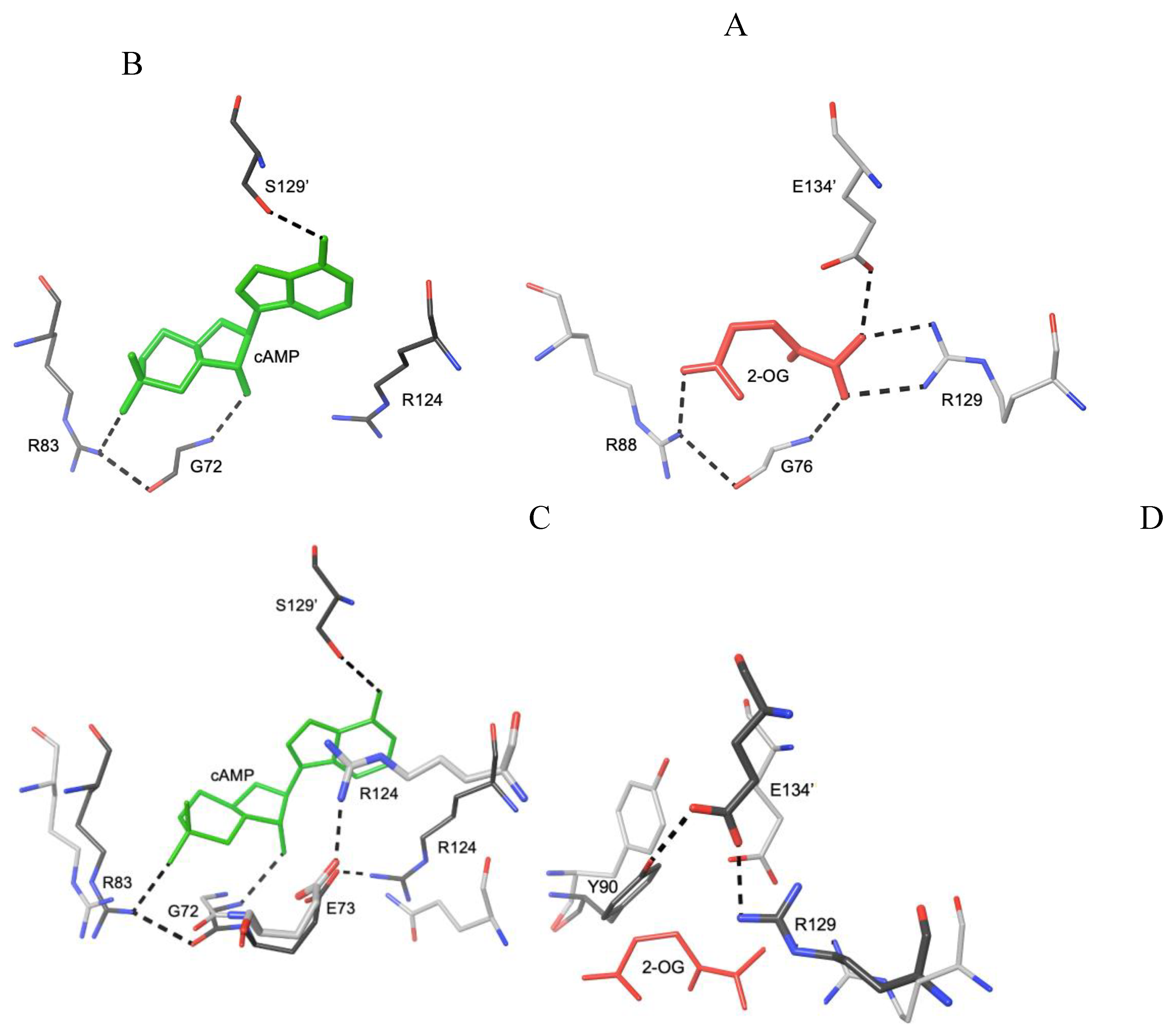
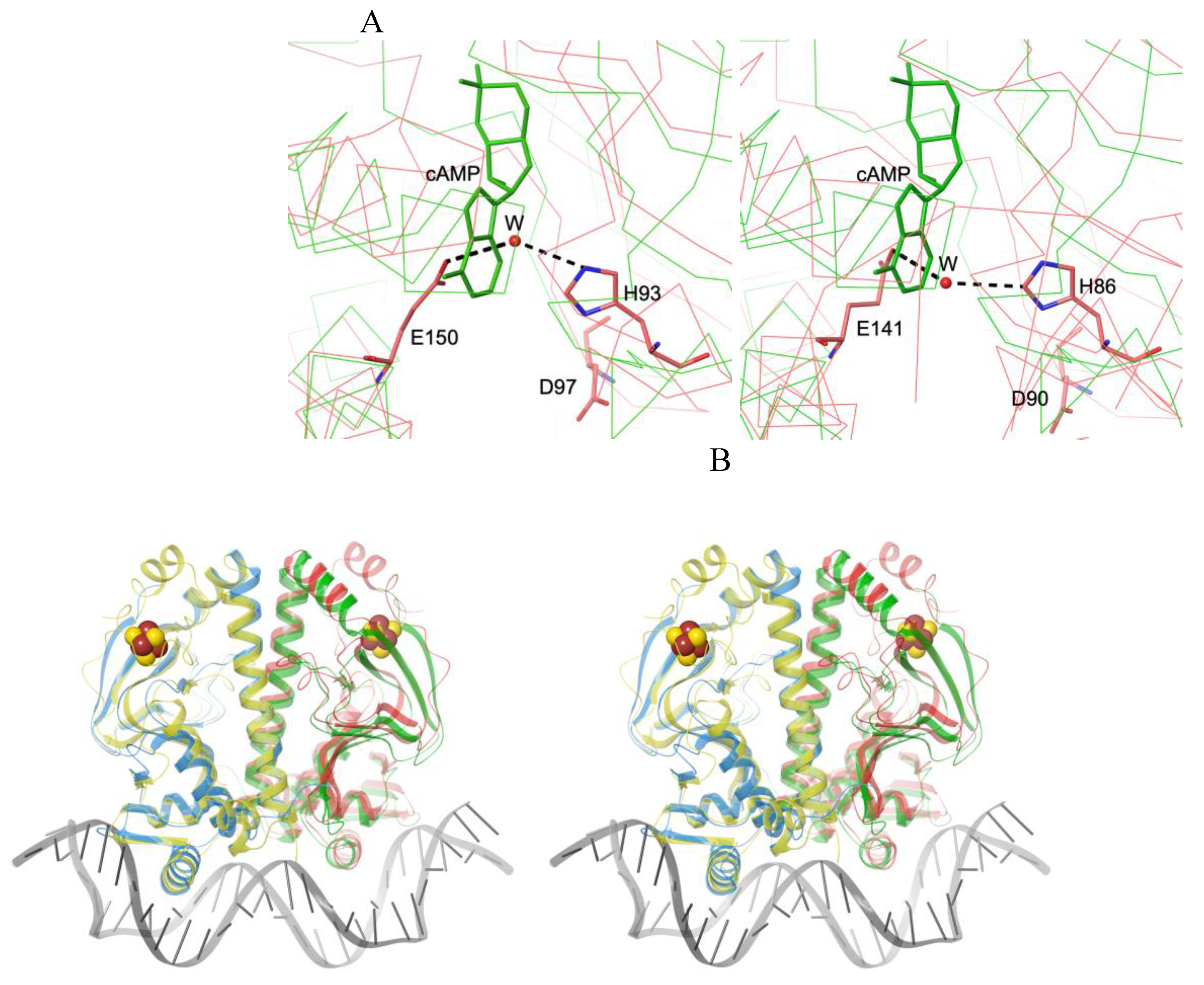
3.2. Negative Allostery in the FNR/CRP Family
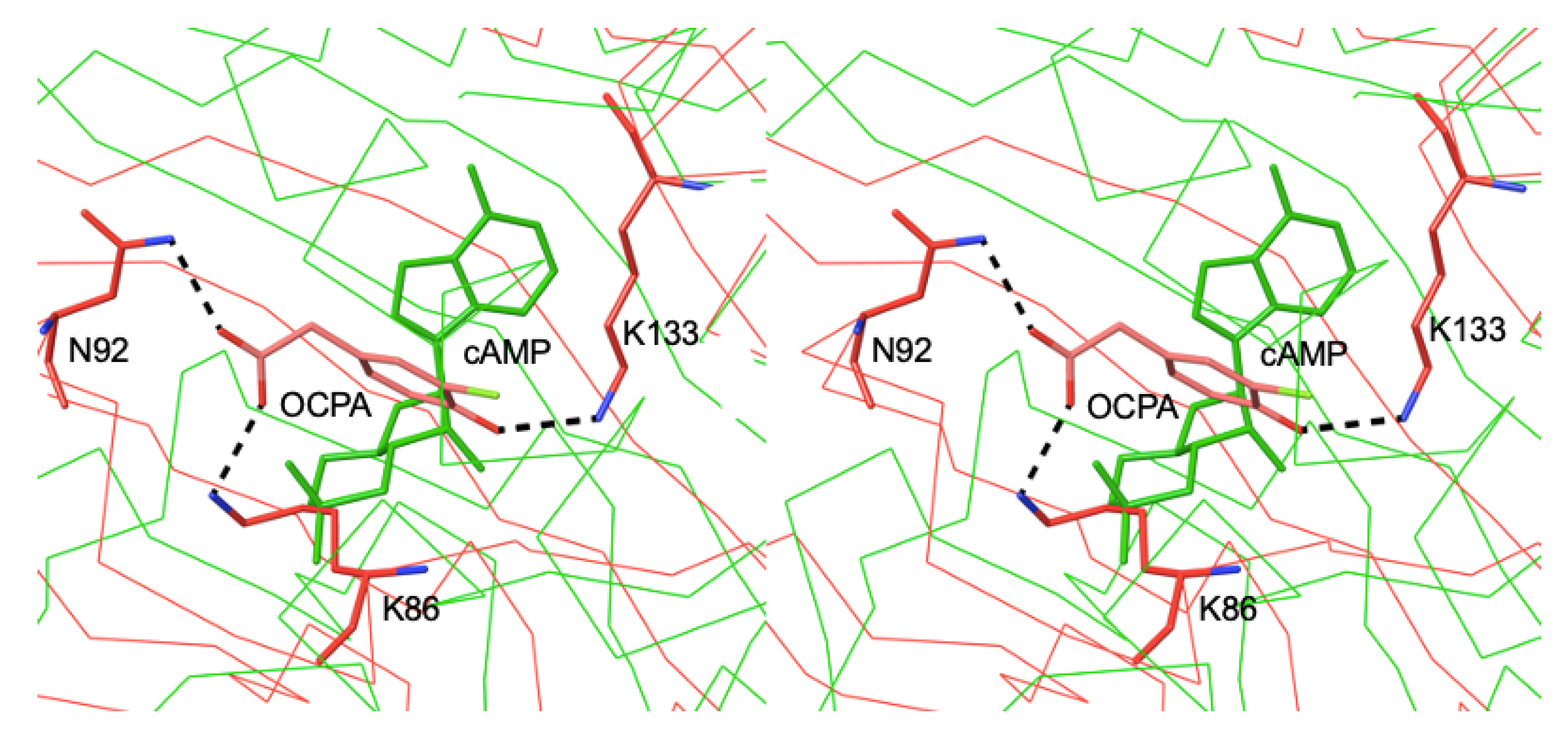
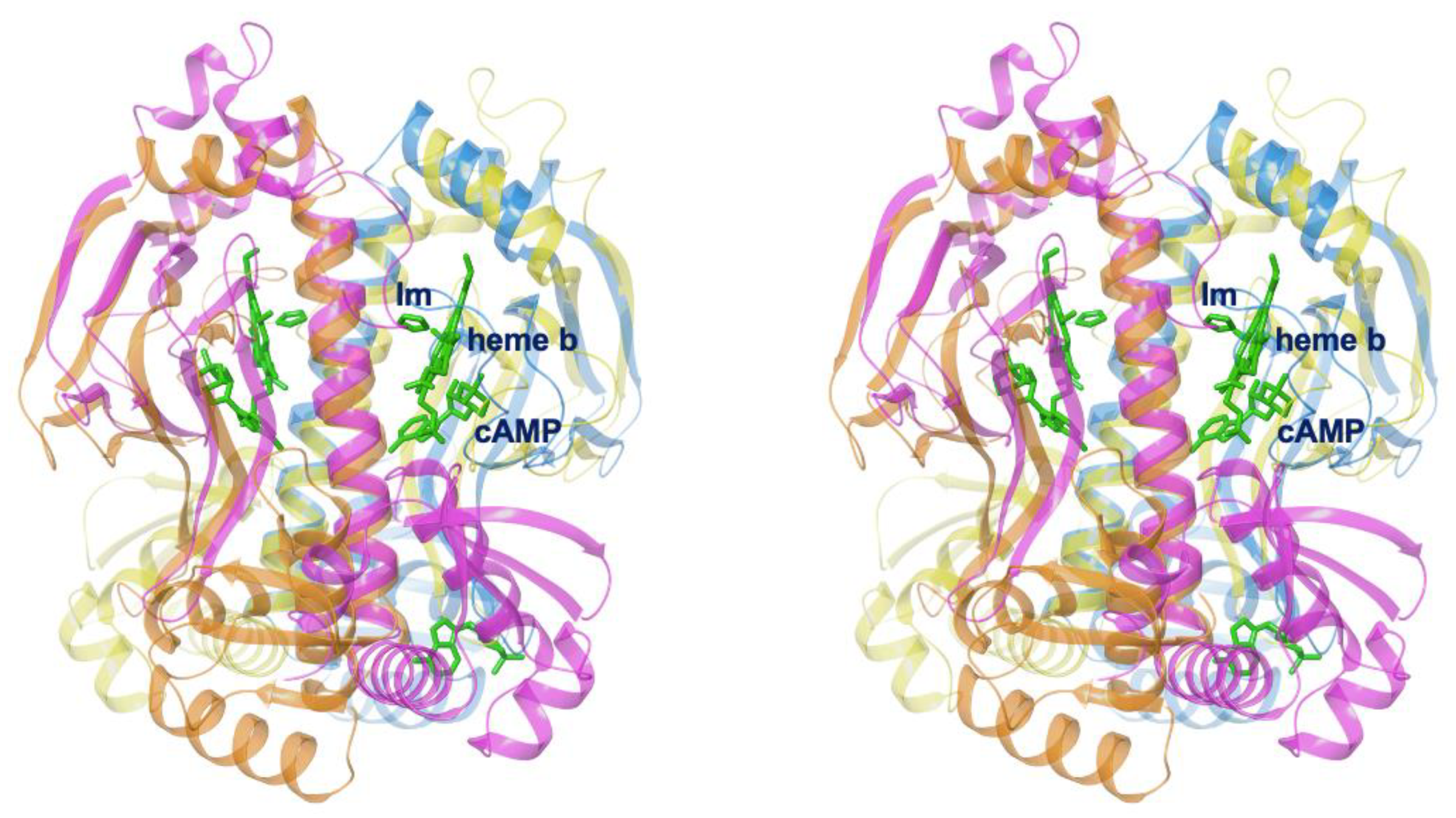
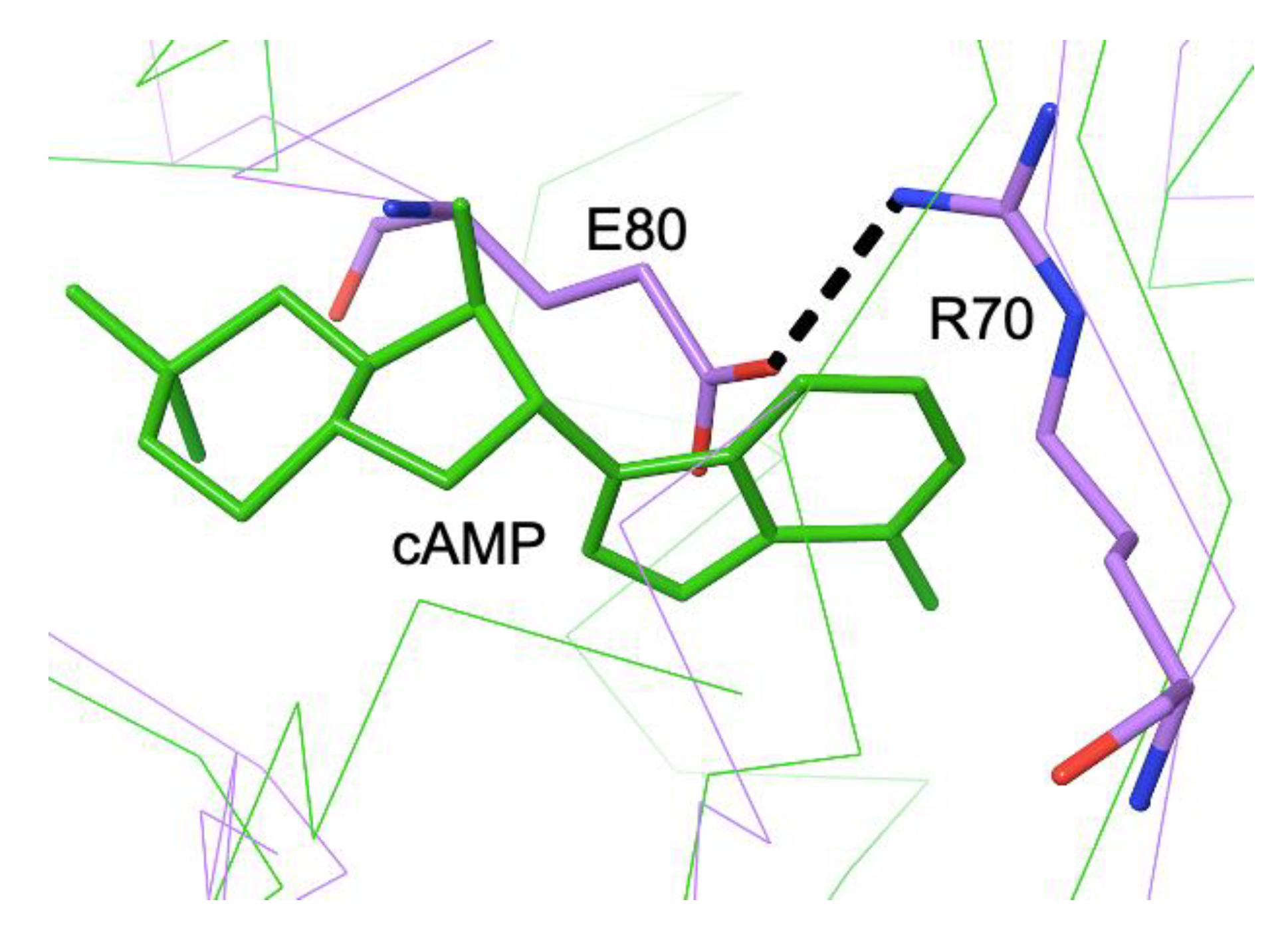
3.3. Different Allostery in Other Members of the FNR/CRP Family
3.4. Non-Allosteric Members of the FNR/CRP Family
3.5. Non-Allosteric O2-Sensing Members of the FNR/CRP Family

4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Blattner, F.R.; Plunkett, G.; Bloch, C.A.; Perna, N.T.; Burland, V.; Riley, M.; Collado-Vides, J.; Glasner, J.D.; Rode, C.K.; Mayhew, G.F.; et al. The Complete Genome Sequence of Escherichia coli K-12. Science 1997, 277, 1453–1462. [Google Scholar] [CrossRef] [PubMed]
- Murphy, L.D.; Zimmerman, S.B. Condensation and Cohesion of λ DNA in Cell Extracts and Other Media: Implications for the Structure and Function of DNA in Prokaryotes. Biophys Chem 1995, 57, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Visweswariah, S.S.; Busby, S.J.W. Evolution of Bacterial Transcription Factors: How Proteins Take on New Tasks, but Do Not Always Stop Doing the Old Ones. Trends Microbiol. 2015, 23, 463–467. [Google Scholar] [CrossRef]
- Brosius, J. Exaptation at the Molecular Genetic Level. Sci China Life Sci 2019, 62, 437–452. [Google Scholar] [CrossRef]
- Gould, S.J. Exaptation: A Crucial Tool for an Evolutionary Psychology. J. Soc. Issues 1991, 47, 43–65. [Google Scholar] [CrossRef]
- Dorman, C.J.; Schumacher, M.A.; Bush, M.J.; Brennan, R.G.; Buttner, M.J. When Is a Transcription Factor a NAP? Curr. Opin. Microbiol. 2020, 55, 26–33. [Google Scholar] [CrossRef]
- McKay, D.B.; Steitz, T.A. Structure of Catabolite Gene Activator Protein at 2.9 A Resolution Suggests Binding to Left-Handed B-DNA. Nature 1981, 290, 744–749. [Google Scholar] [CrossRef]
- Berman, H.M.; Ten Eyck, L.F.; Goodsell, D.S.; Haste, N.M.; Kornev, A.; Taylor, S.S. The cAMP Binding Domain: An Ancient Signaling Module. Proc. Natl. Acad. Sci. U.S.A. 2005, 102, 45–50. [Google Scholar] [CrossRef]
- Weber, I.T.; Takio, K.; Titani, K.; Steitz, T.A. The cAMP-Binding Domains of the Regulatory Subunit of cAMP-Dependent Protein Kinase and the Catabolite Gene Activator Protein Are Homologous. Proc. Natl. Acad. Sci. U.S.A 1982, 79, 7679–7683. [Google Scholar] [CrossRef]
- Ulrich, L.E.; Koonin, E.V.; Zhulin, I.B. One-Component Systems Dominate Signal Transduction in Prokaryotes. Trends Microbiol. 2005, 13, 52–56. [Google Scholar] [CrossRef]
- Zschiedrich, C.P.; Keidel, V.; Szurmant, H. Molecular Mechanisms of Two-Component Signal Transduction. J. Mol. Biol. 2016, 428, 3752–3775. [Google Scholar] [CrossRef]
- Krishnaswamy, M.; Seshasayee, A.S.N. The Evolution of Function in the DNA Binding Domain of the CRP/FNR Family. Genome Biol Evol. 2025, 17(10), evaf182. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Tomita, M.; Kanai, A. Comprehensive Computational Analysis of Bacterial CRP/FNR Superfamily and Its Target Motifs Reveals Stepwise Evolution of Transcriptional Networks. Genome Biol Evol. 2013, 5, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Omelchenko, M.V.; Wolf, Y.I.; Gaidamakova, E.K.; Matrosova, V.Y.; Vasilenko, A.; Zhai, M.; Daly, M.J.; Koonin, E.V.; Makarova, K.S. Comparative Genomics of Thermus Thermophilus and Deinococcus Radiodurans: Divergent Routes of Adaptation to Thermophily and Radiation Resistance. BMC Evol Biol 2005, 5, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Choi, I.-G.; Kim, S.-H. Evolution of Protein Structural Classes and Protein Sequence Families. Proc. Natl. Acad. Sci. U.S.A 2006, 103, 14056–14061. [Google Scholar] [CrossRef]
- Dinardo, S.; Voelkel, K.A.; Sternglanz, R.; Reynolds, A.E.; Wright, A. Escherichia coli DNA Topoisomerase I Mutants Have Compensatory Mutations in DNA Gyrase Genes. Cell 1982, 31, 43–51. [Google Scholar] [CrossRef]
- Heyde, S.A.H.; Frendorf, P.O.; Lauritsen, I.; Nørholm, M.H.H. Restoring Global Gene Regulation through Experimental Evolution Uncovers a NAP (Nucleoid-Associated Protein)-Like Behavior of Crp/Cap. mBio 2021, 12. [Google Scholar] [CrossRef]
- Hołówka, J.; Zakrzewska-Czerwińska, J. Nucleoid Associated Proteins: The Small Organizers That Help to Cope With Stress. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Sharma, H.; Yu, S.; Kong, J.; Wang, J.; Steitz, T.A. Structure of Apo-CAP Reveals That Large Conformational Changes Are Necessary for DNA Binding. Proc. Natl Acad Sci U S A 2009, 106, 16604–16609. [Google Scholar] [CrossRef]
- Vega-Palas, M.A.; Madueño, F.; Herrero, A.; Flores, E. Identification and Cloning of a Regulatory Gene for Nitrogen Assimilation in the Cyanobacterium Synechococcus sp. Strain PCC 7942. J Bacteriol 1990, 172, 643–647. [Google Scholar] [CrossRef]
- Forcada-Nadal, A.; Bibak, S.; Salinas, P.; Contreras, A.; Rubio, V.; Llácer, J.L. Structures of the Cyanobacterial Nitrogen Regulators NtcA and PipX Complexed to DNA Shed Light on DNA Binding by NtcA and Implicate PipX in the Recruitment of RNA Polymerase. Nucleic Acids Res. 2025, 53, gkaf096. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.-X.; Jiang, Y.-L.; He, Y.-X.; Chen, Y.-F.; Teng, Y.-B.; Chen, Y.; Zhang, C.-C.; Zhou, C.-Z. Structural Basis for the Allosteric Control of the Global Transcription Factor NtcA by the Nitrogen Starvation Signal 2-Oxoglutarate. Proc Natl Acad Sci U S A 2010, 107, 12487–12492. [Google Scholar] [CrossRef] [PubMed]
- Körner, H.; Sofia, H.J.; Zumft, W.G. Phylogeny of the Bacterial Superfamily of Crp-Fnr Transcription Regulators: Exploiting the Metabolic Spectrum by Controlling Alternative Gene Programs. FEMS Microbiol. Rev. 2003, 27, 559–592. [Google Scholar] [CrossRef] [PubMed]
- Canaves, J.M.; Taylor, S.S. Classification and Phylogenetic Analysis of the cAMP-Dependent Protein Kinase Regulatory Subunit Family. J Mol Evol 2002, 54, 17–29. [Google Scholar] [CrossRef]
- Smidt, H.; Vos, W.M. de Anaerobic Microbial Dehalogenation. Annu. Rev. Microbiol. 2004, 58, 43–73. [Google Scholar] [CrossRef]
- Villemur, R.; Lanthier, M.; Beaudet, R.; Lépine, F. The Desulfitobacterium genus. FEMS Microbiol. Rev. 2006, 30, 706–733. [Google Scholar] [CrossRef]
- Smidt, H.; van Leest, M.; van der Oost, J.; de Vos, W.M. Transcriptional Regulation of the Cpr Gene Cluster in ortho-Chlorophenol-Respiring Desulfitobacterium dehalogenans. J. Bacteriol. 2000, 182, 5683–5691. [Google Scholar] [CrossRef]
- Levy, C.; Pike, K.; Heyes, D.J.; Joyce, M.G.; Gabor, K.; Smidt, H.; van der Oost, J.; Leys, D. Molecular Basis of Halorespiration Control by CprK, a CRP-FNR Type Transcriptional Regulator. Mol. Microbiol. 2008, 70, 151–167. [Google Scholar] [CrossRef]
- Kemp, L.R.; Dunstan, M.S.; Fisher, K.; Warwicker, J.; Leys, D. The Transcriptional Regulator CprK Detects Chlorination by Combining Direct and Indirect Readout Mechanisms. In Philos. Trans. R. Soc. London, Ser B: Biol. Sci.; 2013. [Google Scholar] [CrossRef]
- Joyce, M.G.; Levy, C.; Gábor, K.; Pop, S.M.; Biehl, B.D.; Doukov, T.I.; Ryter, J.M.; Mazon, H.; Smidt, H.; van den Heuvel, R.H.H.; et al. CprK Crystal Structures Reveal Mechanism for Transcriptional Control of Halorespiration*. J. Biol. Chem. 2006, 281, 28318–28325. [Google Scholar] [CrossRef]
- Gábor, K.; Veríssimo, C.S.; Cyran, B.C.; Ter Horst, P.; Meijer, N.P.; Smidt, H.; de Vos, W.M.; van der Oost, J. Characterization of CprK1, a CRP/FNR-Type Transcriptional Regulator of Halorespiration from Desulfitobacterium hafniense. J Bacteriol 2006, 188, 2604–2613. [Google Scholar] [CrossRef]
- Xu, G.; Wang, B.-G. Independent Evolution of Six Families of Halogenating Enzymes. PLoS One 2016, 11, e0154619. [Google Scholar] [CrossRef] [PubMed]
- Chin, K.-H.; Lee, Y.-C.; Tu, Z.-L.; Chen, C.-H.; Tseng, Y.-H.; Yang, J.-M.; Ryan, R.P.; McCarthy, Y.; Dow, J.M.; Wang, A.H.-J.; et al. The cAMP Receptor-Like Protein CLP Is a Novel c-di-GMP Receptor Linking Cell–Cell Signaling to Virulence Gene Expression in Xanthomonas campestris. J. Mol. Biol. 2010, 396, 646–662. [Google Scholar] [CrossRef]
- Leduc, J.L.; Roberts, G.P. Cyclic di-GMP Allosterically Inhibits the CRP-like Protein (Clp) of Xanthomonas Axonopodis pv. citri. J Bacteriol 2009, 191, 7121–7122. [Google Scholar] [CrossRef] [PubMed]
- Gomelsky, M. Cyclic-Di-GMP-Binding CRP-Like Protein: A Spectacular New Role for a Veteran Signal Transduction Actor. J. Bact. 2009, 191, 6785–6787. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, T.; Leimeister-Wächter, M.; Domann, E.; Hartl, M.; Goebel, W.; Nichterlein, T.; Notermans, S. Coordinate Regulation of Virulence Genes in Listeria Monocytogenes Requires the Product of the prfA Gene. J Bacteriol 1992, 174, 568–574. [Google Scholar] [CrossRef]
- Eiting, M.; Hagelüken, G.; Schubert, W.-D.; Heinz, D.W. The Mutation G145S in PrfA, a Key Virulence Regulator of Listeria monocytogenes, Increases DNA-Binding Affinity by Stabilizing the HTH Motif. Mol. Microbiol. 2005, 56, 433–446. [Google Scholar] [CrossRef]
- Reniere, M.L.; Whiteley, A.T.; Hamilton, K.L.; John, S.M.; Lauer, P.; Brennan, R.G.; Portnoy, D.A. Glutathione Activates Virulence Gene Expression of an Intracellular Pathogen. Nature 2015, 517, 170–173. [Google Scholar] [CrossRef]
- Deponte, M. Glutathione Catalysis and the Reaction Mechanisms of Glutathione-Dependent Enzymes. Biochim. Biophys. Acta 2013, 1830, 3217–3266. [Google Scholar] [CrossRef]
- Wang, Y.; Feng, H.; Zhu, Y.; Gao, P. Structural Insights into Glutathione-Mediated Activation of the Master Regulator PrfA in Listeria monocytogenes. Protein Cell 2017, 8, 308–312. [Google Scholar] [CrossRef]
- Hall, M.; Grundström, C.; Begum, A.; Lindberg, M.J.; Sauer, U.H.; Almqvist, F.; Johansson, J.; Sauer-Eriksson, A.E. Structural Basis for Glutathione-Mediated Activation of the Virulence Regulatory Protein PrfA in Listeria. Proc Natl Acad Sci U S A 2016, 113, 14733–14738. [Google Scholar] [CrossRef]
- Aono, S.; Nakajima, H.; Saito, K.; Okada, M. A Novel Heme Protein That Acts as a Carbon Monoxide-Dependent Transcriptional Activator in Rhodospirillum rubrum. Biochem. Biophys. Res. Commun. 1996, 228, 752–756. [Google Scholar] [CrossRef]
- Lanzilotta, W.N.; Schuller, D.J.; Thorsteinsson, M.V.; Kerby, R.L.; Roberts, G.P.; Poulos, T.L. Structure of the CO Sensing Transcription Activator CooA. Nat Struct Mol Biol 2000, 7, 876–880. [Google Scholar] [CrossRef]
- Komori, H.; Inagaki, S.; Yoshioka, S.; Aono, S.; Higuchi, Y. Crystal Structure of CO-Sensing Transcription Activator CooA Bound to Exogenous Ligand Imidazole. J. Mol. Biol. 2007, 367, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Dent, M.R.; Weaver, B.R.; Roberts, M.G.; Burstyn, J.N. Carbon Monoxide-Sensing Transcription Factors: Regulators of Microbial Carbon Monoxide Oxidation Pathway Gene Expression. J Bacteriol 205 e00332-22. [CrossRef] [PubMed]
- Borjigin, M.; Li, H.; Lanz, N.D.; Kerby, R.L.; Roberts, G.P.; Poulos, T.L. Structure-Based Hypothesis on the Activation of the CO-Sensing Transcription Factor CooA. Acta Crystallogr D Biol Crystallogr 2007, 63, 282–287. [Google Scholar] [CrossRef]
- Tripathi, S.; Poulos, T.L. Testing the N-Terminal Velcro Model of CooA Carbon Monoxide Activation. Biochemistry 2018, 57, 3059–3064. [Google Scholar] [CrossRef]
- Agari, Y.; Kashihara, A.; Yokoyama, S.; Kuramitsu, S.; Shinkai, A. Global Gene Expression Mediated by Thermus thermophilus SdrP, a CRP/FNR Family Transcriptional Regulator. Mol. Microbiol. 2008, 70, 60–75. [Google Scholar] [CrossRef]
- Agari, Y.; Kuramitsu, S.; Shinkai, A. X-Ray Crystal Structure of TTHB099, a CRP/FNR Superfamily Transcriptional Regulator from Thermus thermophilus HB8, Reveals a DNA-Binding Protein with No Required Allosteric Effector Molecule. Proteins 2012, 80, 1490–1494. [Google Scholar] [CrossRef]
- Wang, Y.; Hu, J.; Gao, X.; Cao, Y.; Ye, S.; Chen, C.; Wang, L.; Xu, H.; Guo, M.; Zhang, D.; et al. cAMP-Independent DNA Binding of the CRP Family Protein DdrI from Deinococcus radiodurans. mBio 2024, 15, e01144-24. [Google Scholar] [CrossRef]
- Nishida, H.; Abe, R.; Nagayama, T.; Yano, K. Genome Signature Difference between Deinococcus radiodurans and Thermus thermophilus. Int. J. Evol. Biol 2012, 2012, 205274. [Google Scholar] [CrossRef]
- Śmiga, M.; Roszkiewicz, E.; Ślęzak, P.; Tracz, M.; Olczak, T. cAMP-Independent Crp Homolog Adds to the Multi-Layer Regulatory Network in Porphyromonas gingivalis. Front. Cell. Infect. Microbiol. 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Townsend, P.D.; Rodgers, T.L.; Glover, L.C.; Korhonen, H.J.; Richards, S.A.; Colwell, L.J.; Pohl, E.; Wilson, M.R.; Hodgson, D.R.W.; McLeish, T.C.B.; et al. The Role of Protein-Ligand Contacts in Allosteric Regulation of the Escherichia coli Catabolite Activator Protein*. J. Biol. Chem. 2015, 290, 22225–22235. [Google Scholar] [CrossRef] [PubMed]
- Mysak, J.; Podzimek, S.; Sommerova, P.; Lyuya-Mi, Y.; Bartova, J.; Janatova, T.; Prochazkova, J.; Duskova, J. Porphyromonas gingivalis: Major Periodontopathic Pathogen Overview. J Immunol Res 2014, 2014, 476068. [Google Scholar] [CrossRef] [PubMed]
- Kiley, P.J.; Beinert, H. Oxygen Sensing by the Global Regulator, FNR: The Role of the Iron-Sulfur Cluster. FEMS Microbiol. Rev. 1998, 22, 341–352. [Google Scholar] [CrossRef]
- Reents, H.; Gruner, I.; Harmening, U.; Böttger, L.H.; Layer, G.; Heathcote, P.; Trautwein, A.X.; Jahn, D.; Härtig, E. Bacillus subtilis Fnr Senses Oxygen via a [4Fe-4S] Cluster Coordinated by Three Cysteine Residues without Change in the Oligomeric State. Mol. Microbiol. 2006, 60, 1432–1445. [Google Scholar] [CrossRef]
- Bonnet, M.; Kurz, M.; Mesa, S.; Briand, C.; Hennecke, H.; Grütter, M.G. The Structure of Bradyrhizobium japonicum Transcription Factor FixK2 Unveils Sites of DNA Binding and Oxidation. J. Biol. Chem. 2013, 288, 14238–14246. [Google Scholar] [CrossRef]
- Volbeda, A.; Darnault, C.; Renoux, O.; Nicolet, Y.; Fontecilla-Camps, J.C. The Crystal Structure of the Global Anaerobic Transcriptional Regulator FNR Explains Its Extremely Fine-Tuned Monomer-Dimer Equilibrium. Sci Adv 2015, 1, e1501086. [Google Scholar] [CrossRef]
- Mesa, S.; Reutimann, L.; Fischer, H.-M.; Hennecke, H. Posttranslational Control of Transcription Factor FixK2, a Key Regulator for the Bradyrhizobium japonicum–Soybean Symbiosis. Proc. Natl. Acad. Sci. U.S.A. 2009, 106, 21860. [Google Scholar] [CrossRef]
- The UniProt Consortium UniProt: The Universal Protein Knowledgebase in 2025. Nucleic Acids Res 2025, 53, D609–D617. [CrossRef]
- Pi, H.-W.; Lin, J.-J.; Chen, C.-A.; Wang, P.-H.; Chiang, Y.-R.; Huang, C.-C.; Young, C.-C.; Li, W.-H. Origin and Evolution of Nitrogen Fixation in Prokaryotes. Mol Biol Evol 2022, 39, msac181. [Google Scholar] [CrossRef]
- Davín, A.A.; Woodcroft, B.J.; Soo, R.M.; Morel, B.; Murali, R.; Schrempf, D.; Clark, J.W.; Álvarez-Carretero, S.; Boussau, B.; Moody, E.R.R.; et al. A Geological Timescale for Bacterial Evolution and Oxygen Adaptation. Science 2025, 388, eadp1853. [Google Scholar] [CrossRef]
- Schrödinger Release 2019-2. Schrödinger, LLC: New York, NY, 2019.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



