Submitted:
18 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. The ME/CFS Enigma
1.2. The ACE Connection
1.3. Preliminary Pharmacological Observations
1.4. Scope and Structure
2. Neurobiological Foundations
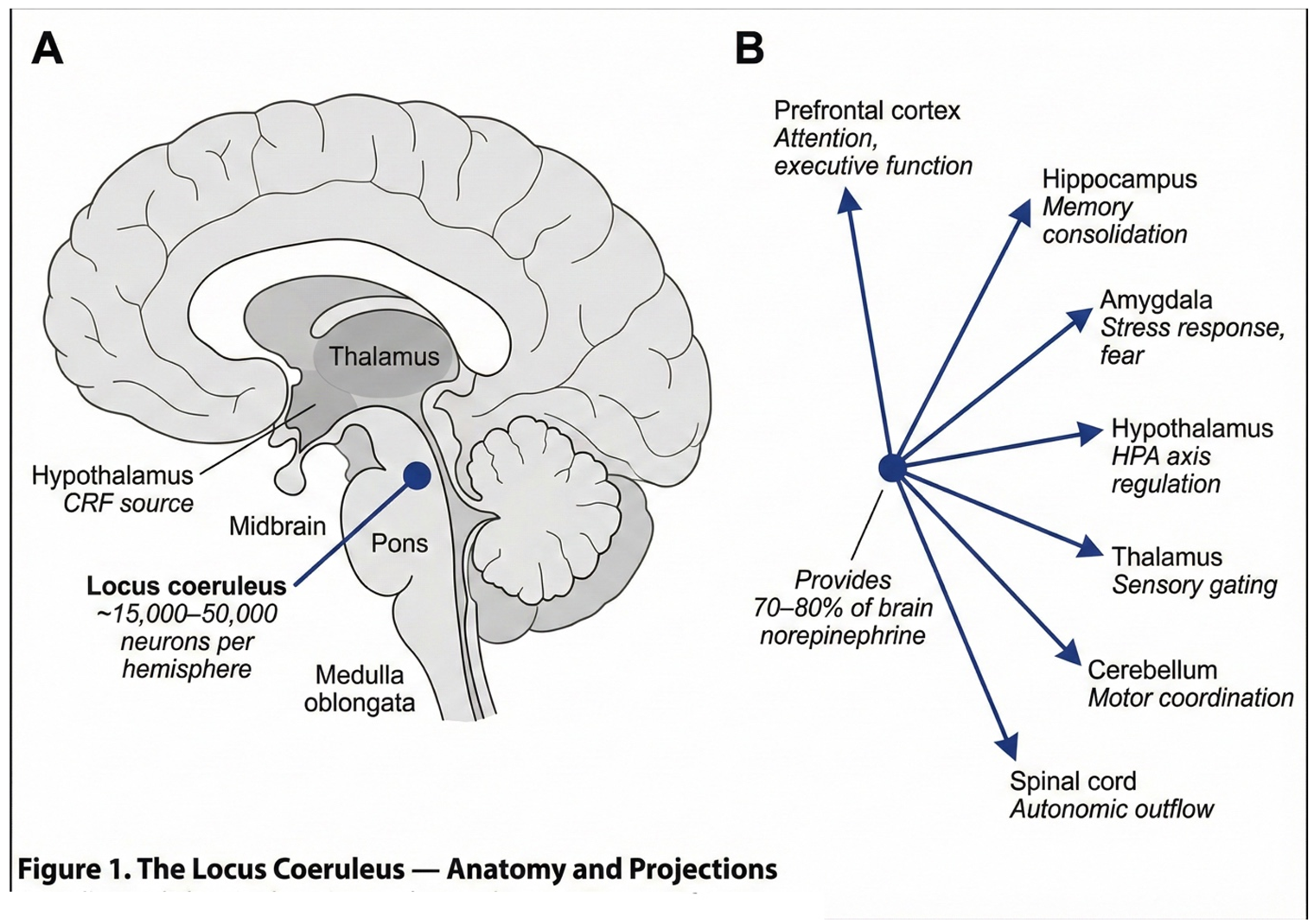
2.1. The Locus Coeruleus: Anatomy and Function
2.2. The CRF-LC Stress Axis
2.3. ACEs: Programming the Stress System
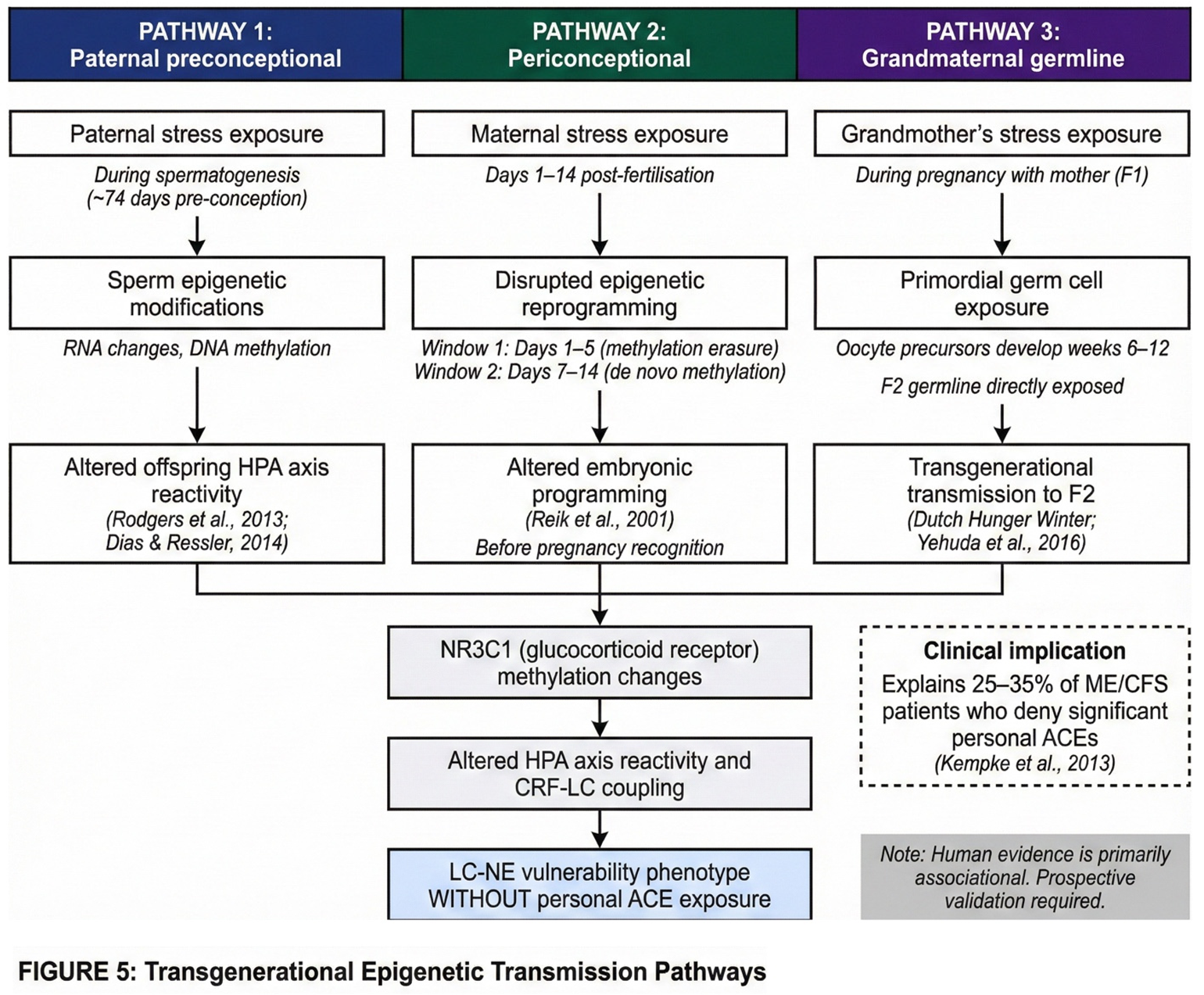
2.4. Transgenerational Epigenetic Transmission: The 'ACE-Negative' Paradox
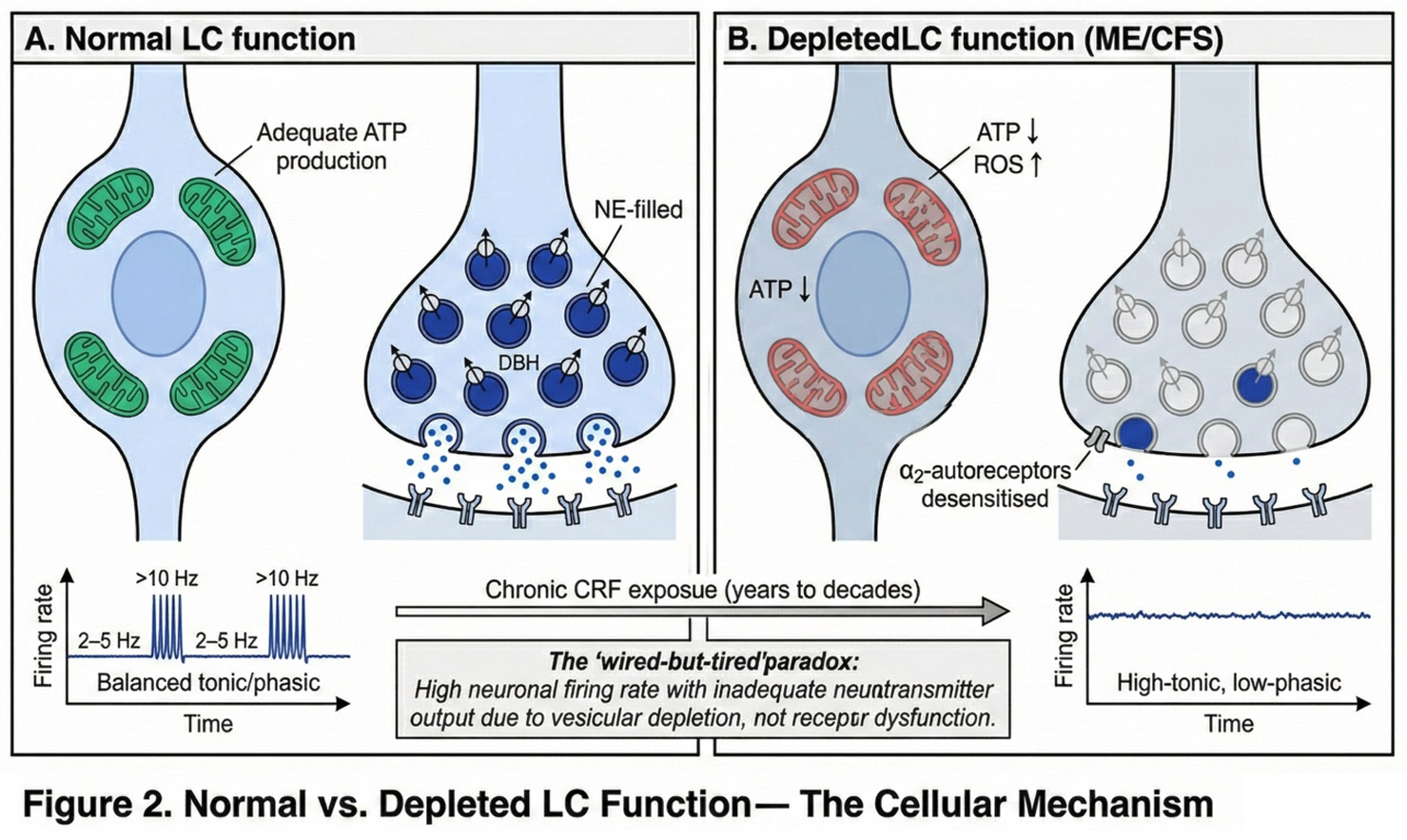
2.5. LC Vulnerability to Chronic Stress
2.6. The Glymphatic Connection
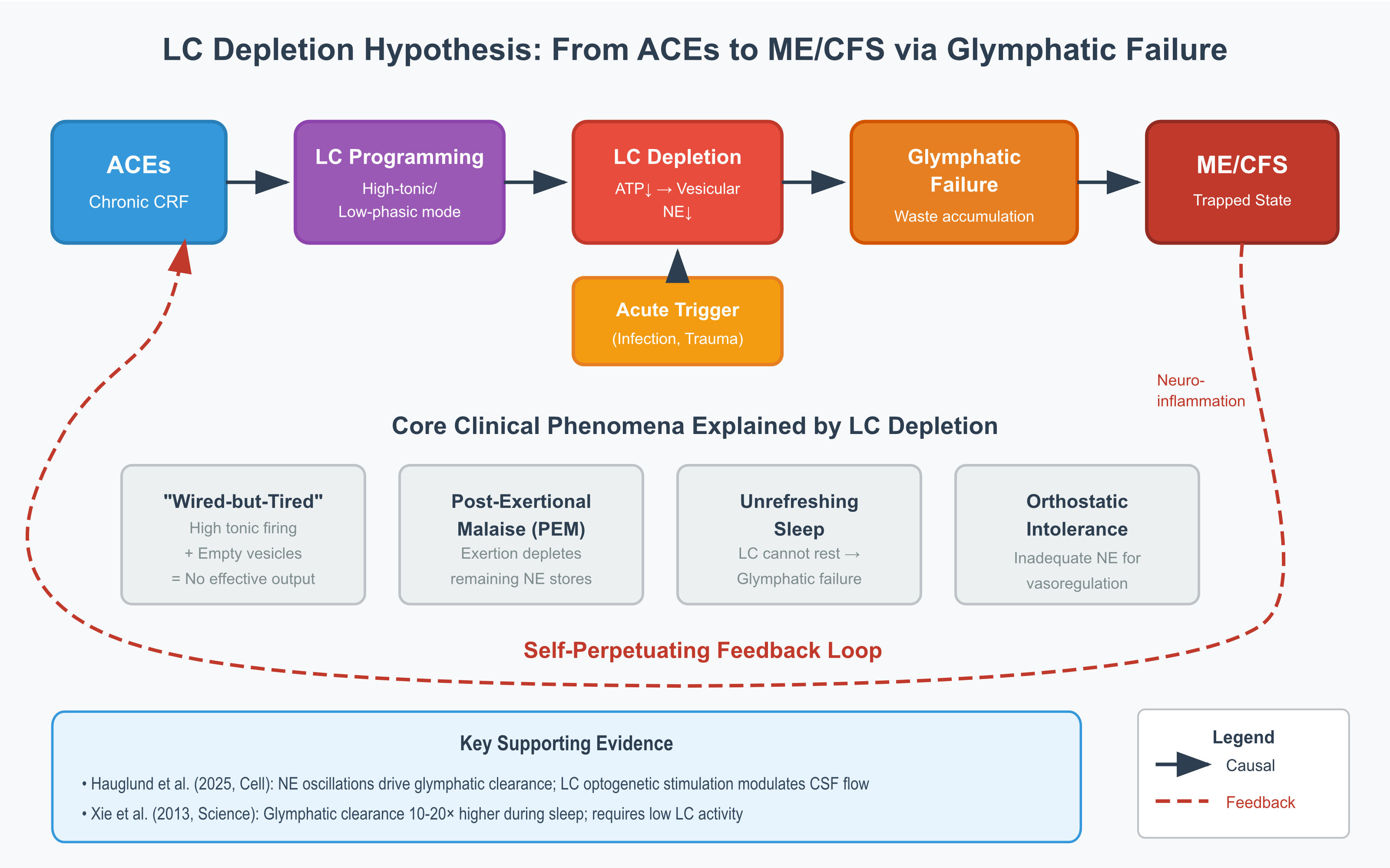
3. The Proposed Mechanism
3.1. Multi-Stage Model
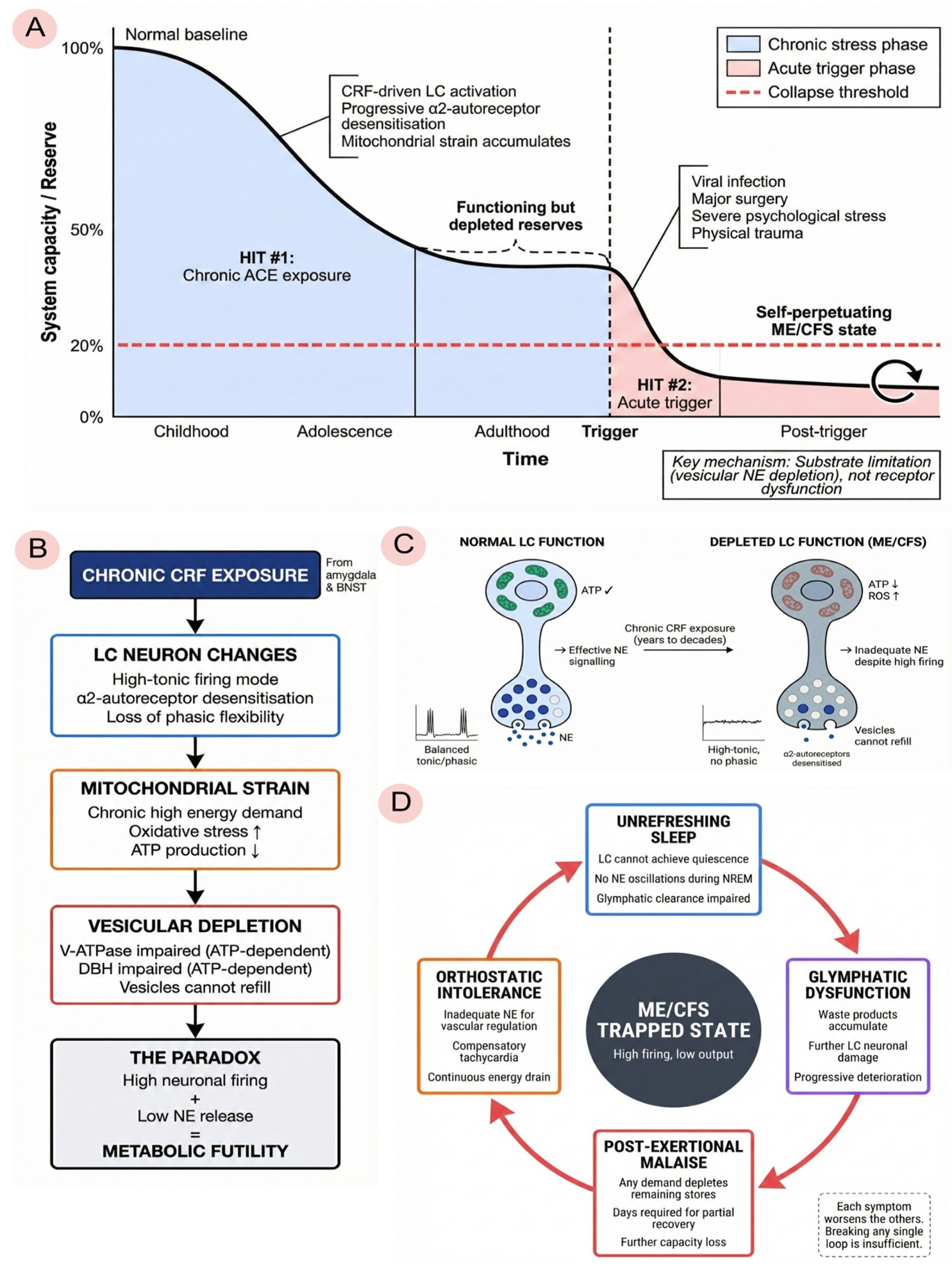
3.2. Differentiation from Existing Two-Hit Models
4. Clinical Phenomena Through the LC Depletion Lens
4.1. The 'Wired-but-Tired' Paradox
4.2. Post-Exertional Malaise
4.3. Orthostatic Intolerance
4.4. Unrefreshing Sleep
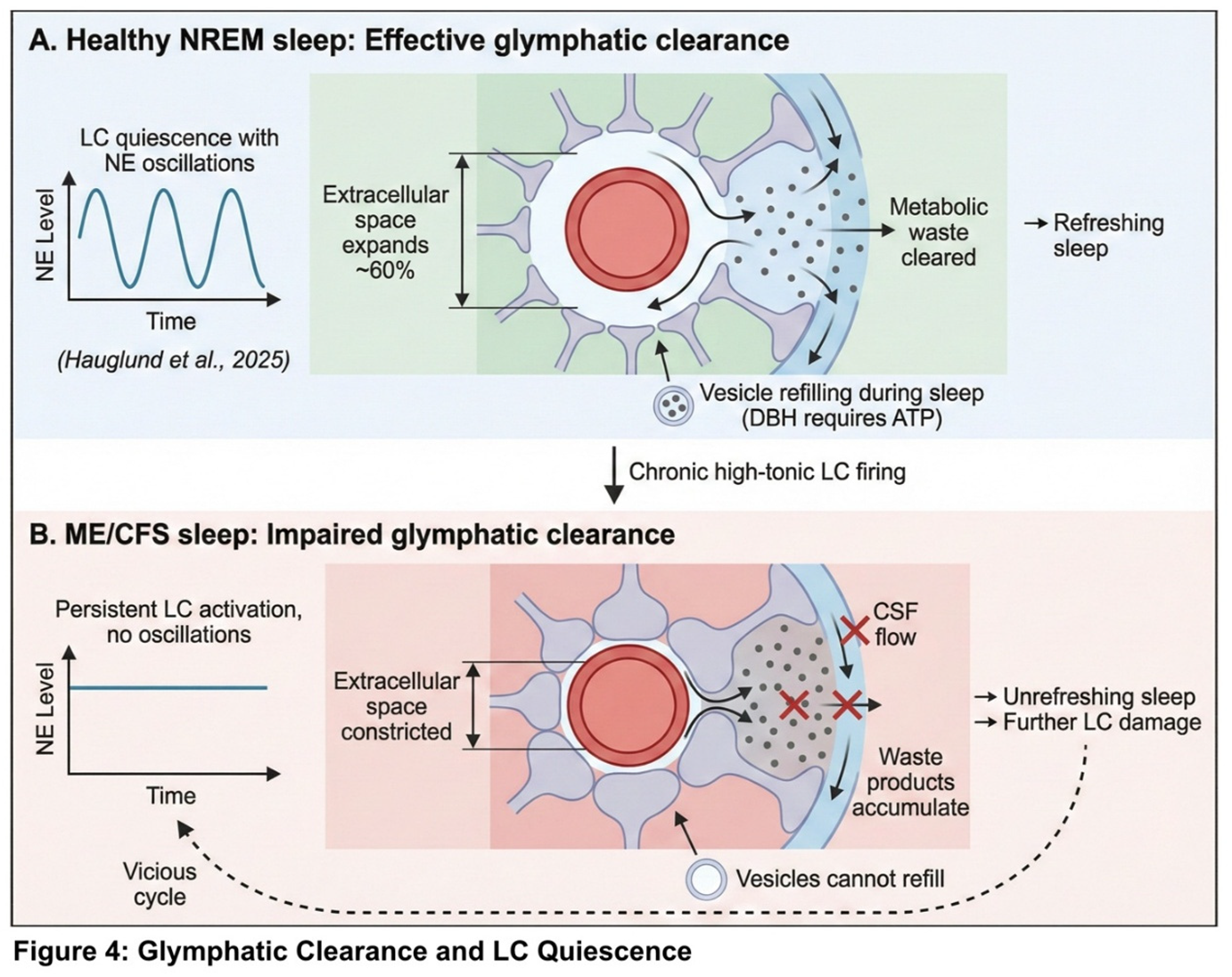
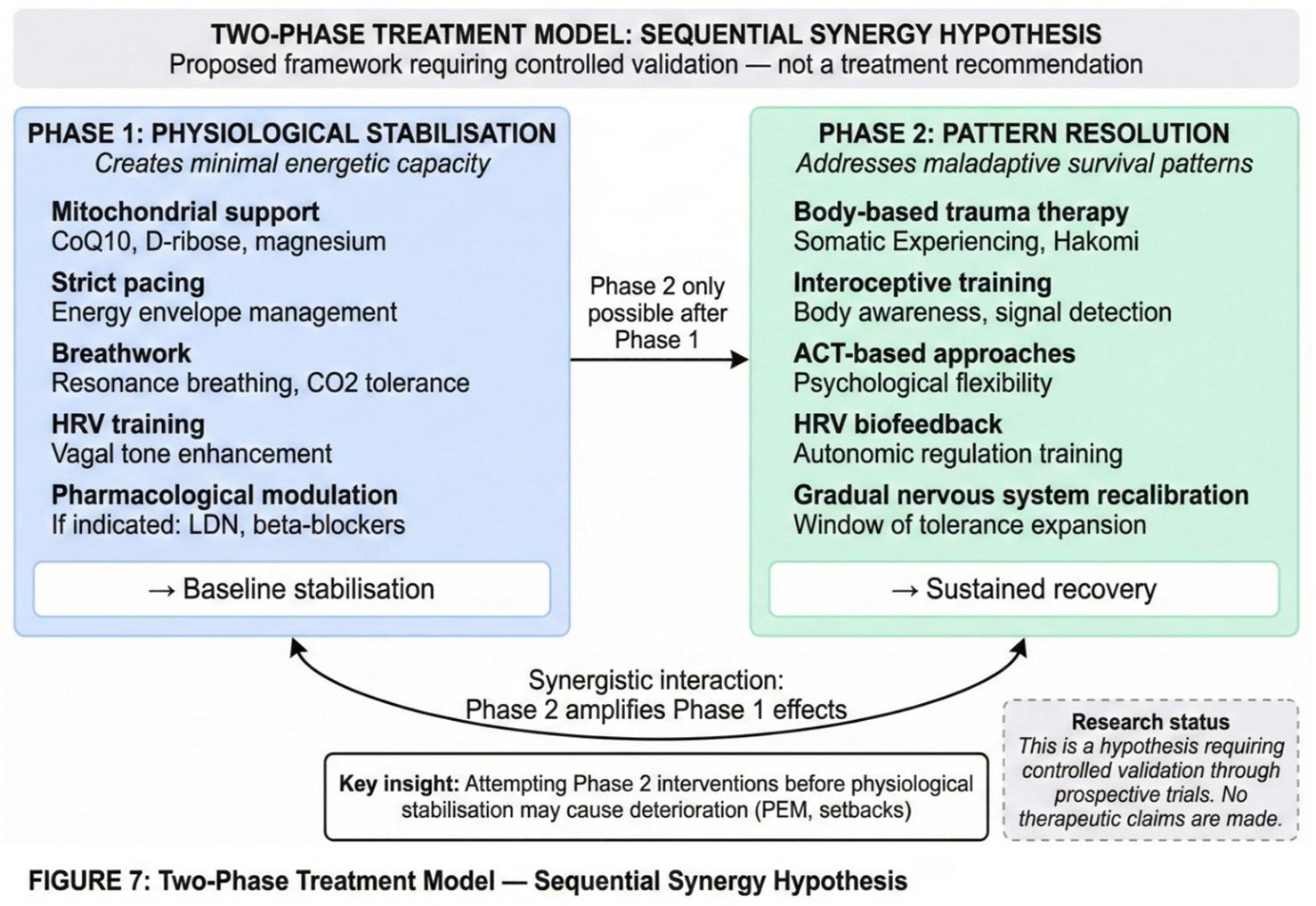
5. Supporting Evidence and Alternative Explanations
5.1. Evidence Supporting the Hypothesis
5.2. Alternative and Complementary Mechanisms
5.3. Limitations and Uncertainties
6. Testable Predictions and Research Agenda
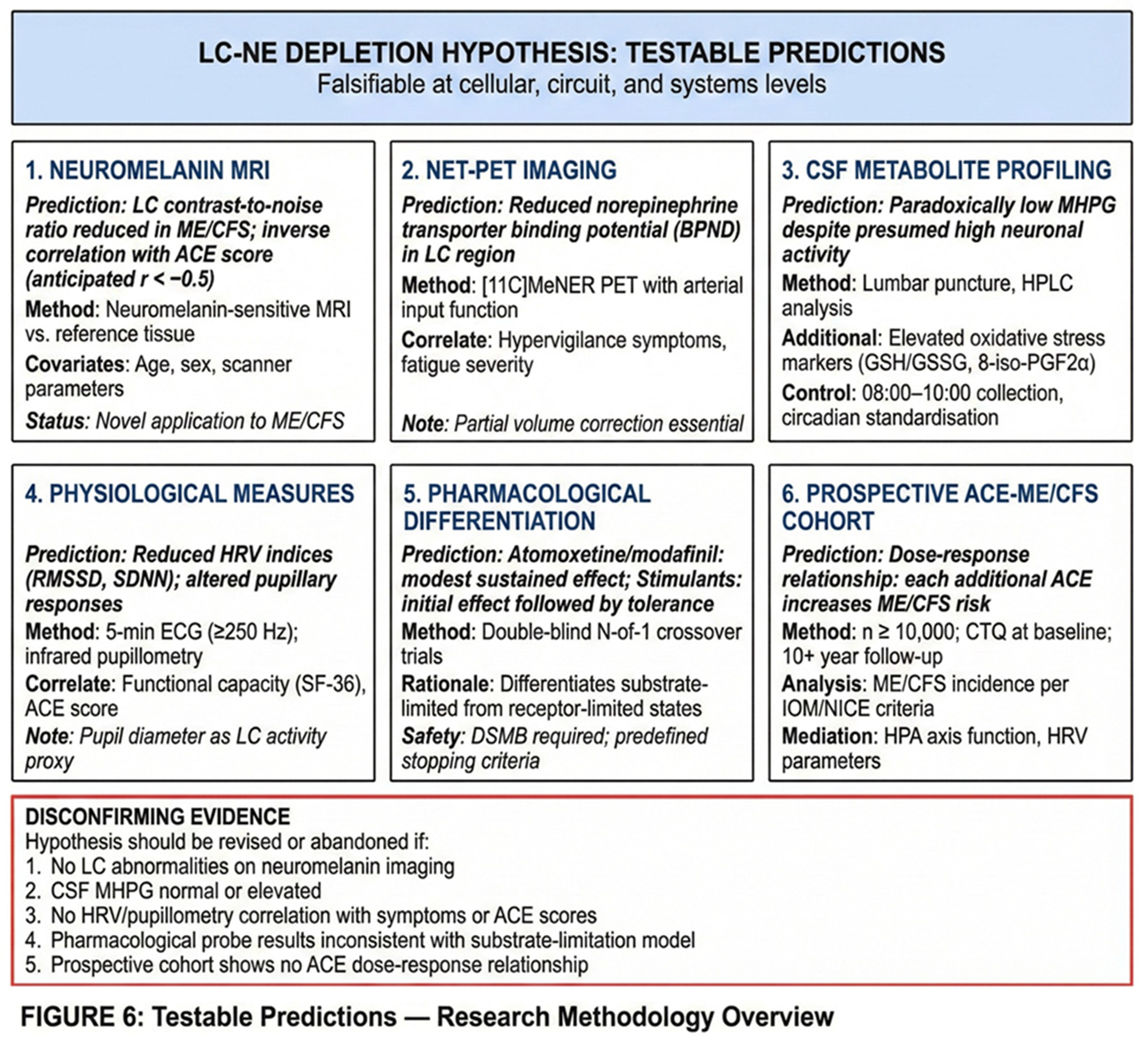
6.1. Neuromelanin-Sensitive MRI of the LC
6.2. NET-PET Imaging
6.3. CSF Metabolite Profiling
6.4. Physiological Measures: Heart Rate Variability and Pupillometry
6.5. Pharmacological Differentiation: N-of-1 Trials
6.6. Transcutaneous Auricular Vagus Nerve Stimulation: A Therapeutic Window to the LC-NE System
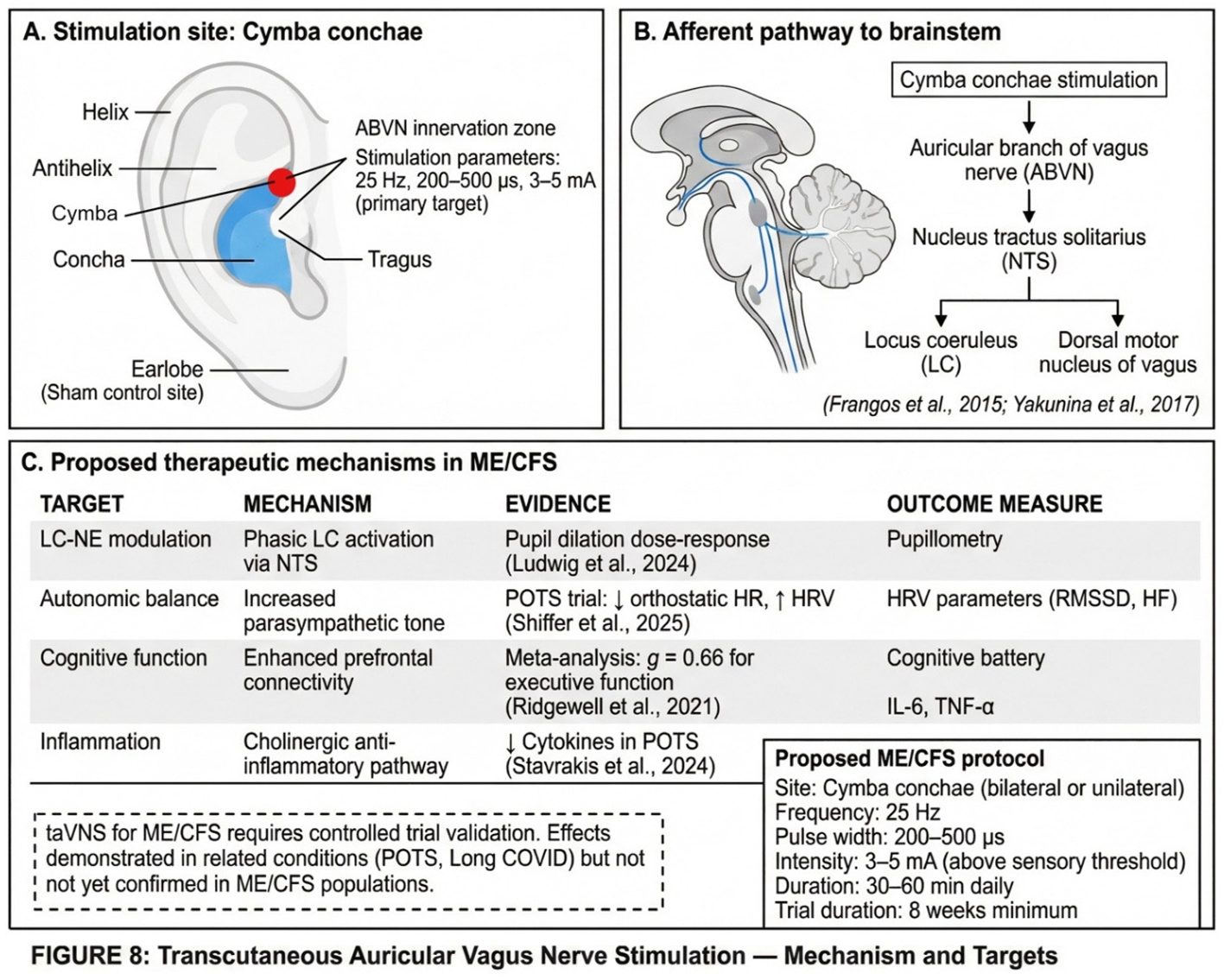
6.6.1. Pupillary Responses as LC Activity Proxy
6.6.2. Orthostatic Intolerance: Direct Evidence from POTS
6.6.3. Cognitive Function and Brain Fog
6.6.4. Stimulation Laterality: Unilateral vs. Bilateral
6.6.5. Proposed Study Design for ME/CFS
6.7. Prospective ACE-ME/CFS Cohort
7. Potential Disconfirming Evidence
8. Discussion
8.1. Strengths
8.2. Weaknesses
8.3. Implications if Hypothesis Is Correct
9. Conclusion
Funding
Acknowledgments
Conflicts of Interest
Data Availability
Ethics
Abbreviations
References
- Amann, R.P. (2008). The cycle of the seminiferous epithelium in humans: A need to revisit? Journal of Andrology, 29(5), 469–487.
- Aston-Jones, G., & Cohen, J.D. (2005). An integrative theory of locus coeruleus-norepinephrine function: Adaptive gain and optimal performance. Annual Review of Neuroscience, 28, 403–450.
- Badran, B.W., Dowdle, L.T., Mithoefer, O.J., LaBate, N.T., Coatsworth, J., Brown, J.C., DeVries, W.H., Austelle, C.W., McTeague, L.M., & George, M.S. (2018). Neurophysiologic effects of transcutaneous auricular vagus nerve stimulation (taVNS) via electrical stimulation of the tragus: A concurrent taVNS/fMRI study and review. Brain Stimulation, 11(3), 492–500.
- Bangasser, D.A., & Valentino, R.J. (2020). The locus coeruleus-norepinephrine system in stress and arousal: Unraveling historical, current, and future perspectives. Frontiers in Psychiatry, 11, 601519.
- Baumeister, D., Akhtar, R., Ciufolini, S., Pariante, C.M., & Mondelli, V. (2016). Childhood trauma and adulthood inflammation: A meta-analysis. Molecular Psychiatry, 21(5), 642–649.
- Betts, M.J., Kirilina, E., Otaduy, M.C.G., et al. (2019). Locus coeruleus imaging as a biomarker for noradrenergic dysfunction in neurodegenerative diseases. Brain, 142(9), 2558–2571.
- Blockmans, D., Persoons, P., Van Houdenhove, B., & Bobbaers, H. (2006). Does methylphenidate reduce the symptoms of chronic fatigue syndrome? The American Journal of Medicine, 119(12), 1047–1053.
- Borges, U., Knops, L., Laborde, S., Klatt, S., & Raab, M. (2020). Transcutaneous vagus nerve stimulation may enhance only specific aspects of the core executive functions: A randomized crossover trial. Frontiers in Neuroscience, 14, 523.
- Bunea, I.M., Szentagotai-Tatar, A., & Miu, A.C. (2017). Early-life adversity and cortisol response to social stress: A meta-analysis. Translational Psychiatry, 7(12), 1274.
- Burkusis, M., Plaks, V., Michael, A., et al. (2015). Environment and the developing embryo. In Principles of Developmental Genetics (2nd ed., pp. 143–154). Academic Press.
- Da Silva, J.P., et al. (2025). Delineating clinical phenotypes and HPA-axis dysfunction in ME/CFS. Presented at IACFS/ME 2025 Conference [conference abstract—not peer-reviewed].
- Dias, B.G., & Ressler, K.J. (2014). Parental olfactory experience influences behavior and neural structure in subsequent generations. Nature Neuroscience, 17(1), 89–96.
- Edwards, R.H. (2007). The neurotransmitter cycle and quantal size. Neuron, 55(6), 835–849.
- Eiden, L.E., & Weihe, E. (2011). VMAT2: A dynamic regulator of brain monoaminergic neuronal function interacting with drugs of abuse. Annals of the New York Academy of Sciences, 1216, 86–98.
- Fang, J., Egorova, N., Rong, P., Liu, J., Hong, Y., Fan, Y., Wang, X., Wang, H., Yu, Y., Ma, Y., Xu, C., Li, S., Zhao, J., Luo, M., Zhu, B., & Kong, J. (2017). Early cortical biomarkers of longitudinal transcutaneous vagus nerve stimulation treatment success in depression. NeuroImage: Clinical, 14, 105–111.
- Felitti, V.J., Anda, R.F., Nordenberg, D., et al. (1998). Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. American Journal of Preventive Medicine, 14(4), 245–258.
- Frangos, E., Ellrich, J., & Komisaruk, B.R. (2015). Non-invasive access to the vagus nerve central projections via electrical stimulation of the external ear: fMRI evidence in humans. Brain Stimulation, 8(3), 624–636.
- Goldstein, D.S., & Aregawi, D. (2025). Low brain norepinephrine in ME/CFS. Presented at IACFS/ME 2025 Conference [conference abstract—not peer-reviewed].
- Hauglund, N.L., Andersen, M., Tokarska, K., et al. (2025). Norepinephrine-mediated slow vasomotion drives glymphatic clearance during sleep. Cell, 188(3), 606–622.
- Hendrix, J., et al. (2025). Sympathetic dysfunction in ME/CFS: A meta-analysis. Presented at IACFS/ME 2025 Conference [conference abstract—not peer-reviewed].
- Jacobs, H.I.L., Riphagen, J.M., Razat, C.M., Wiese, S., & Sack, A.T. (2015). Transcutaneous vagus nerve stimulation boosts associative memory in older individuals. Neurobiology of Aging, 36(5), 1860–1867.
- Joshi, S., Li, Y., Kalwani, R.M., & Gold, J.I. (2016). Relationships between pupil diameter and neuronal activity in the locus coeruleus, colliculi, and cingulate cortex. Neuron, 89(1), 221–234.
- Joshi, S., & Gold, J.I. (2020). Pupil size as a window on neural substrates of cognition. Trends in Cognitive Sciences, 24(6), 466–480.
- Kempke, S., Luyten, P., Claes, S., et al. (2013). Self-critical perfectionism and fatigue and pain in chronic fatigue syndrome. Psychological Medicine, 43(5), 995–1002.
- Keute, M., Demirezen, M., Graf, A., Mueller, N.G., & Zaehle, T. (2019). No modulation of pupil size and event-related pupil response by transcutaneous auricular vagus nerve stimulation (taVNS). Scientific Reports, 9, 11452.
- Komaroff, A.L., & Lipkin, W.I. (2021). Insights from myalgic encephalomyelitis/chronic fatigue syndrome may help unravel the pathogenesis of postacute COVID-19 syndrome. Trends in Molecular Medicine, 27(9), 895–906.
- Ludwig, M., Wienke, C., Betts, M.J., Zaehle, T., & Hämmerer, D. (2024). Current and future directions in transcutaneous vagus nerve stimulation: Research, application, and effects on the locus coeruleus-norepinephrine system. Neuroscience & Biobehavioral Reviews, 159, 105607.
- Lumey, L.H., Stein, A.D., & Susser, E. (2011). Prenatal famine and adult health. Annual Review of Public Health, 32, 237–262.
- McGowan, P.O., Sasaki, A., D'Alessio, A.C., et al. (2009). Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nature Neuroscience, 12(3), 342–348.
- Murphy, P.R., O'Connell, R.G., O'Sullivan, M., Robertson, I.H., & Balsters, J.H. (2014). Pupil diameter covaries with BOLD activity in human locus coeruleus. Human Brain Mapping, 35(8), 4140–4154.
- Nakatomi, Y., Mizuno, K., Ishii, A., et al. (2014). Neuroinflammation in patients with chronic fatigue syndrome/myalgic encephalomyelitis: An [11C-(R)]PK11195 PET study. Journal of Nuclear Medicine, 55(6), 945–950.
- Naviaux, R.K., Naviaux, J.C., Li, K., et al. (2016). Metabolic features of chronic fatigue syndrome. Proceedings of the National Academy of Sciences USA, 113(37), E5472–E5480.
- Nedergaard, M. (2013). Garbage truck of the brain. Science, 340(6140), 1529–1530.
- Nelson, M.J., Bahl, J.S., Buckley, J.D., Thomson, R.L., & Davison, K. (2019). Evidence of altered cardiac autonomic regulation in ME/CFS: A systematic review and meta-analysis. Medicine, 98(43), e17600.
- Painter, R.C., Osmond, C., Gluckman, P., et al. (2008). Transgenerational effects of prenatal exposure to the Dutch famine on neonatal adiposity and health in later life. BJOG, 115(10), 1243–1249.
- Perroud, N., Paoloni-Giacobino, A., Prada, P., et al. (2011). Increased methylation of glucocorticoid receptor gene (NR3C1) in adults with a history of childhood maltreatment. Translational Psychiatry, 1(12), e59.
- Pietrzak, R.H., Gallezot, J.D., Ding, Y.S., et al. (2013). Association of PTSD with reduced in vivo norepinephrine transporter availability in the locus coeruleus. JAMA Psychiatry, 70(11), 1199–1205.
- Priovoulos, N., Jacobs, H.I.L., Ivanov, D., Uludag, K., Verhey, F.R.J., & Poser, B.A. (2018). High-resolution in vivo imaging of human locus coeruleus by magnetization transfer MRI at 3T and 7T. NeuroImage, 168, 427–436.
- Redgrave, J.N., Moore, L., Oyekunle, T., Ebrahim, M., Falidas, K., Snowdon, N., Ali, A., & Majid, A. (2022). Transcutaneous auricular vagus nerve stimulation with concurrent upper limb repetitive task practice for poststroke motor recovery: A pilot study. Journal of Stroke and Cerebrovascular Diseases, 27(7), 1998–2005.
- Reik, W., Dean, W., & Walter, J. (2001). Epigenetic reprogramming in mammalian development. Science, 293(5532), 1089–1093.
- Ridgewell, C., Heaton, K.J., Hildebrandt, A., Couse, J., Leber, D., & Neumeier, W.H. (2021). The effects of transcutaneous auricular vagal nerve stimulation on cognition in healthy individuals: A meta-analysis. Neuropsychology, 35(4), 352–365.
- Rodgers, A.B., Morgan, C.P., Bronson, S.L., et al. (2013). Paternal stress exposure alters sperm microRNA content and reprograms offspring HPA stress axis regulation. Journal of Neuroscience, 33(21), 9003–9012.
- Sara, S.J. (2009). The locus coeruleus and noradrenergic modulation of cognition. Nature Reviews Neuroscience, 10(3), 211–223.
- Sasaki, H., & Matsui, Y. (2008). Epigenetic events in mammalian germ-cell development: Reprogramming and beyond. Nature Reviews Genetics, 9(2), 129–140.
- Schwarz, L.A., & Luo, L. (2015). Organization of the locus coeruleus-norepinephrine system. Current Biology, 25(21), R1051–R1056.
- Shiffer, D., Rigo, S., Minonzio, M., Yarsuvat, D.T., Tobaldini, E., Furlan, L., Montano, N., Cairo, B., Porta, A., Zamunér, A.R., Bonovas, S., Urechie, V., Biaggioni, I., Diedrich, A., & Furlan, R. (2025). Short and long term effects of a two-week transcutaneous vagus nerve stimulation in hyperadrenergic postural orthostatic tachycardia syndrome: A proof-of-concept trial. European Journal of Internal Medicine. [CrossRef]
- Shungu, D.C., Weiduschat, N., Murrough, J.W., et al. (2012). Increased ventricular lactate in chronic fatigue syndrome. III. Relationships to cortical glutathione and clinical symptoms. NMR in Biomedicine, 25(9), 1073–1087.
- Stavrakis, S., Stoner, J.A., Humphrey, M.B., Morris, L., Filiberti, A., Reynolds, J.C., Elkholey, K., Javed, I., Twidale, N., Riha, P., Varahan, S., Scherlag, B.J., Jackman, W.M., Dasari, T.W., & Po, S.S. (2020). TREAT AF (Transcutaneous Electrical Vagus Nerve Stimulation to Suppress Atrial Fibrillation): A randomized clinical trial. JACC: Clinical Electrophysiology, 6(3), 282–291.
- Valentino, R.J., & Van Bockstaele, E. (2008). Convergent regulation of locus coeruleus activity as an adaptive response to stress. European Journal of Pharmacology, 583(2–3), 194–203.
- van Campen, C.L.M.C., Rowe, P.C., & Visser, F.C. (2020). Cerebral blood flow is reduced in severe ME/CFS patients during mild orthostatic stress testing. Healthcare, 8(2), 169.
- Van Houdenhove, B., Neerinckx, E., Lysens, R., et al. (2001). Victimization in chronic fatigue syndrome and fibromyalgia in tertiary care. Psychosomatics, 42(1), 21–28.
- Ventura-Bort, C., Garcia de Gurtubay, I., Bermejo, P., et al. (2023). Immediate effects and duration of a short and single application of transcutaneous auricular vagus nerve stimulation on P300 event related potential. Frontiers in Neuroscience, 17, 1096865.
- Villani, V., Tsakiris, M., & Azevedo, R.T. (2022). Transcutaneous vagus nerve stimulation modulates pupil size and interoceptive attention. Psychophysiology, 59(4), e13983.
- Warren, C.M., Tona, K.D., Ber, L., van den Brink, R.L., van Nieuwenhoven, L., de Geus, E.J.C., & Nieuwenhuis, S. (2019). The neuromodulatory and hormonal effects of transcutaneous vagus nerve stimulation as evidenced by salivary alpha amylase, salivary cortisol, pupil diameter, and the P3 event-related potential. Brain Stimulation, 12(3), 635–642.
- Wirth, K., & Scheibenbogen, C. (2020). A unifying hypothesis of the pathophysiology of ME/CFS: Insights from autoantibodies against beta2-adrenergic receptors. Autoimmunity Reviews, 19(6), 102527.
- Xie, L., Kang, H., Xu, Q., et al. (2013). Sleep drives metabolite clearance from the adult brain. Science, 342(6156), 373–377.
- Yakunina, N., Kim, S.S., & Nam, E.C. (2017). Optimization of transcutaneous vagus nerve stimulation using functional MRI. Neuromodulation: Technology at the Neural Interface, 20(3), 290–300.
- Yehuda, R., Daskalakis, N.P., Bierer, L.M., et al. (2016). Holocaust exposure induced intergenerational effects on FKBP5 methylation. Biological Psychiatry, 80(5), 372–380.
- Yehuda, R., & Lehrner, A. (2018). Intergenerational transmission of trauma effects: Putative role of epigenetic mechanisms. World Psychiatry, 17(3), 243–257.
- Young, J.L. (2013). Use of lisdexamfetamine dimesylate in treatment of executive functioning deficits and chronic fatigue syndrome: A double-blind, placebo-controlled study. Psychiatry Research, 207(1–2), 127–133.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



