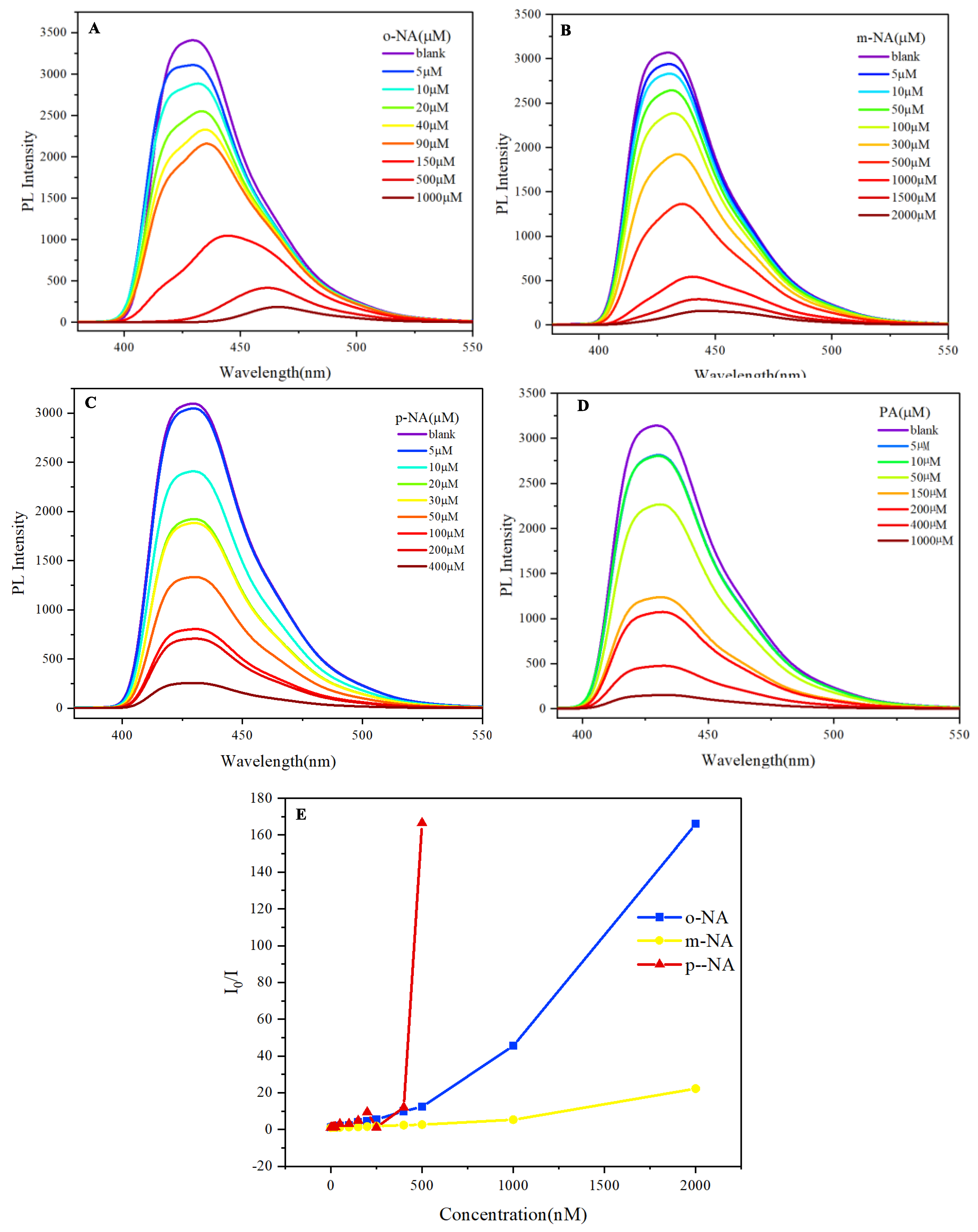
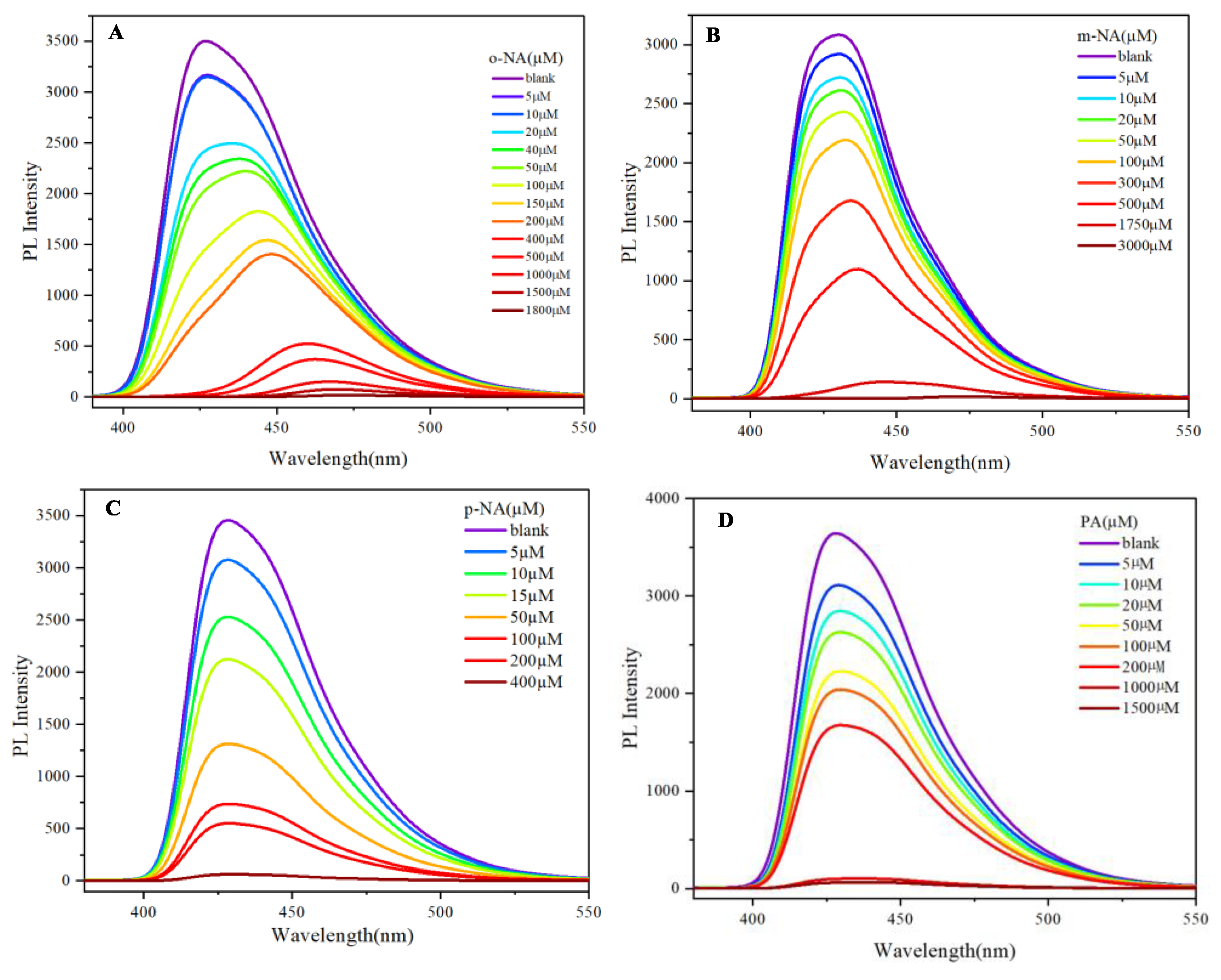
2. Aggregation phenomenon
The investigation of the ultraviolet (UV) absorption spectra of naphthalene and anthracene revealed intriguing insights into their aggregation behavior. Both naphthalene and anthracene displayed slight aggregation-induced absorbance, characterized by subtle shifts in their UV spectra as the water fraction increased. This phenomenon is indicative of the formation of an excimer or aggregate species, which can lead to changes in electronic transitions and, consequently, the absorbance characteristics.
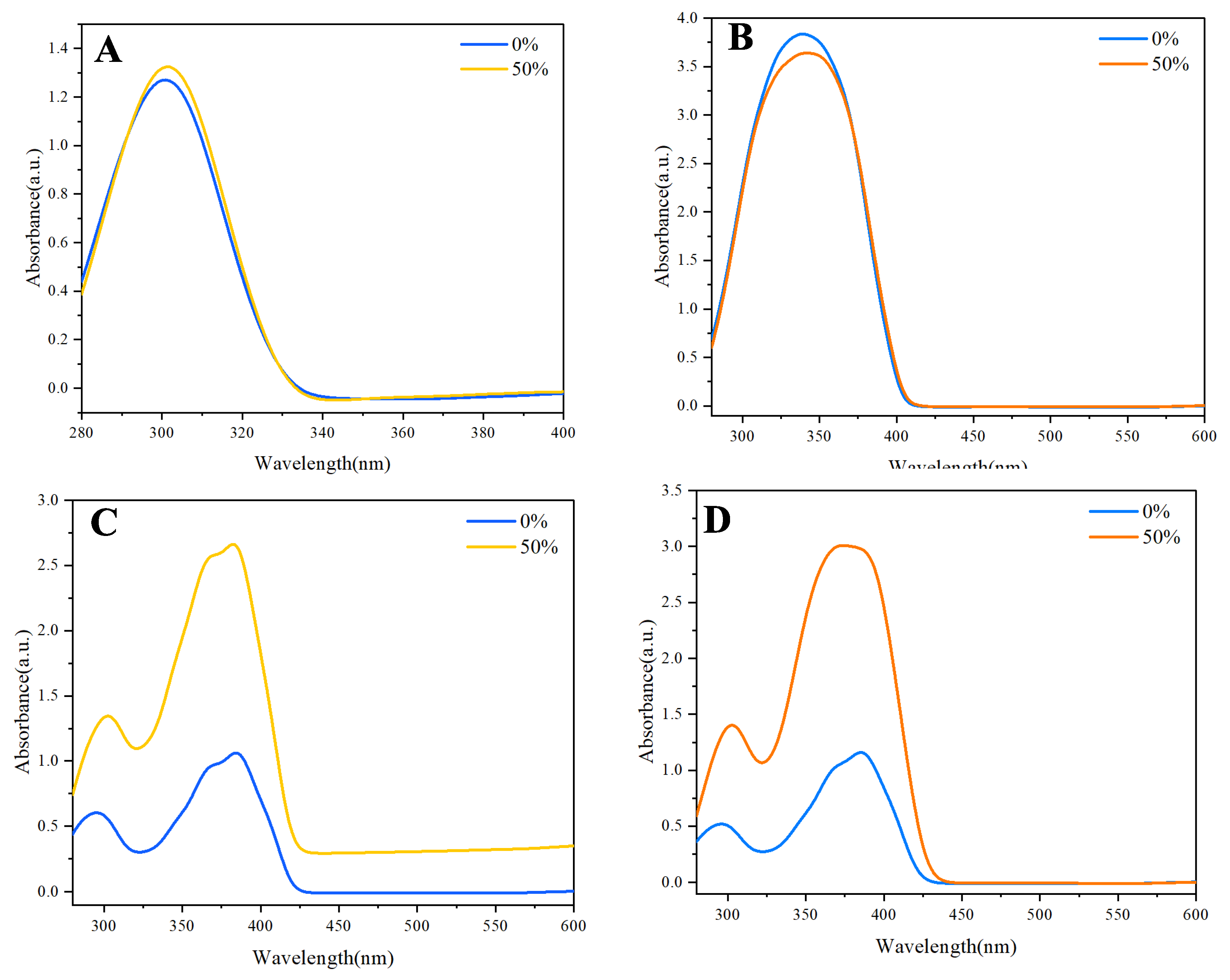
In the UV spectrum of naphthalene, a relatively sharp absorption peak is observed (
Figure 2A), corresponding to its π-π* transitions. However, as the water fraction increases, a slight broadening of the absorption band and a tailing effect becomes evident. This tailing is likely due to aggregated species forming, which can absorb light at slightly different wavelengths than the monomeric form. The aggregation leads to a distribution of energy states, causing a shift in the absorption maximum and the appearance of a broader spectral profile.
Similarly, anthracene exhibited a comparable trend, albeit with more pronounced effects (
Figure 2B). The absorption spectrum of anthracene also showed a clear peak associated with its π-π* transitions. However, the aggregation-induced effects are not as noticeable as naphthalene, but there is broader absorption band and significant tailing observed as the water fraction increased. The aggregation of anthracene molecules can result in the formation of excimers, which are well-known to exhibit distinct absorption characteristics compared to their monomeric counterparts. This excimer formation is attributed to the close proximity of anthracene molecules, allowing for intermolecular interactions that alter their electronic properties.
In contrast, when examining the UV spectra of the synthesized derivatives—9,10-di(naphthalen-1-yl)anthracene
3 and 9-(naphthalen-1-yl)-10-(naphthalen-2-yl)anthracene
5, the phenomenon of aggregation was significantly more pronounced (
Figure 2C and
Figure 2D). The absorption spectra of compound
4 were omitted from
Figure 2 as it showed no notable tailing or intensity changes with water fraction, indicating negligible aggregation behavior compared to derivatives 3 and 5. The UV absorption spectra of these compounds
1,
2,
3, and
5 displayed markedly increased absorbance and more extensive tailing, suggesting a higher propensity for aggregation compared to naphthalene and anthracene alone. The UV spectra exhibited broader absorption bands, indicating the presence of multiple electronic states due to aggregation. This behavior underscores the importance of molecular design in influencing the optical properties of organic compounds. The UV spectral analysis of naphthalene, anthracene, and their derivatives reveals significant insights into the aggregation-induced absorbance and tailing phenomenon. The slight aggregation observed in naphthalene and anthracene becomes markedly pronounced in their derivatives, highlighting the influence of molecular structure on aggregation behavior. The observed spectral broadening in the UV and photoluminescence spectra of naphthalene, anthracene, and their derivatives can be attributed to aggregation effects rather than instrumental limitations. Instrumental factors, such as slit width or detector resolution, typically produce consistent broadening across all samples, which was not observed here. Instead, the broadening and tailing effects were concentration- and solvent-dependent, aligning with the formation of aggregates or excimers. For instance, the broadening in naphthalene and anthracene spectra at higher water fractions (
Figure 2A, 2B) reflects the distribution of electronic states due to intermolecular interactions in aggregated species. This is further supported by the derivatives (e.g., compounds 3 and 5), where structural modifications enhanced aggregation, leading to more pronounced spectral changes (
Figure 2C, 2D). The absence of such effects in compound 4 underscores the role of molecular design. These findings are consistent with AIE literature, where aggregation restricts intramolecular motion, altering emission profiles. Thus, the broadening is a intrinsic property of the aggregated states, providing a clear distinction from instrumental artifacts.
The absorption spectra were measured at 50% water content due to experimental constraints at higher water fractions (e.g., 70%), where excessive scattering and precipitation interfered with spectral clarity. Despite this, the 50% condition provided sufficient aggregation to observe meaningful absorbance shifts, aligning with the AIE trends observed in photoluminescence studies at higher water fractions.
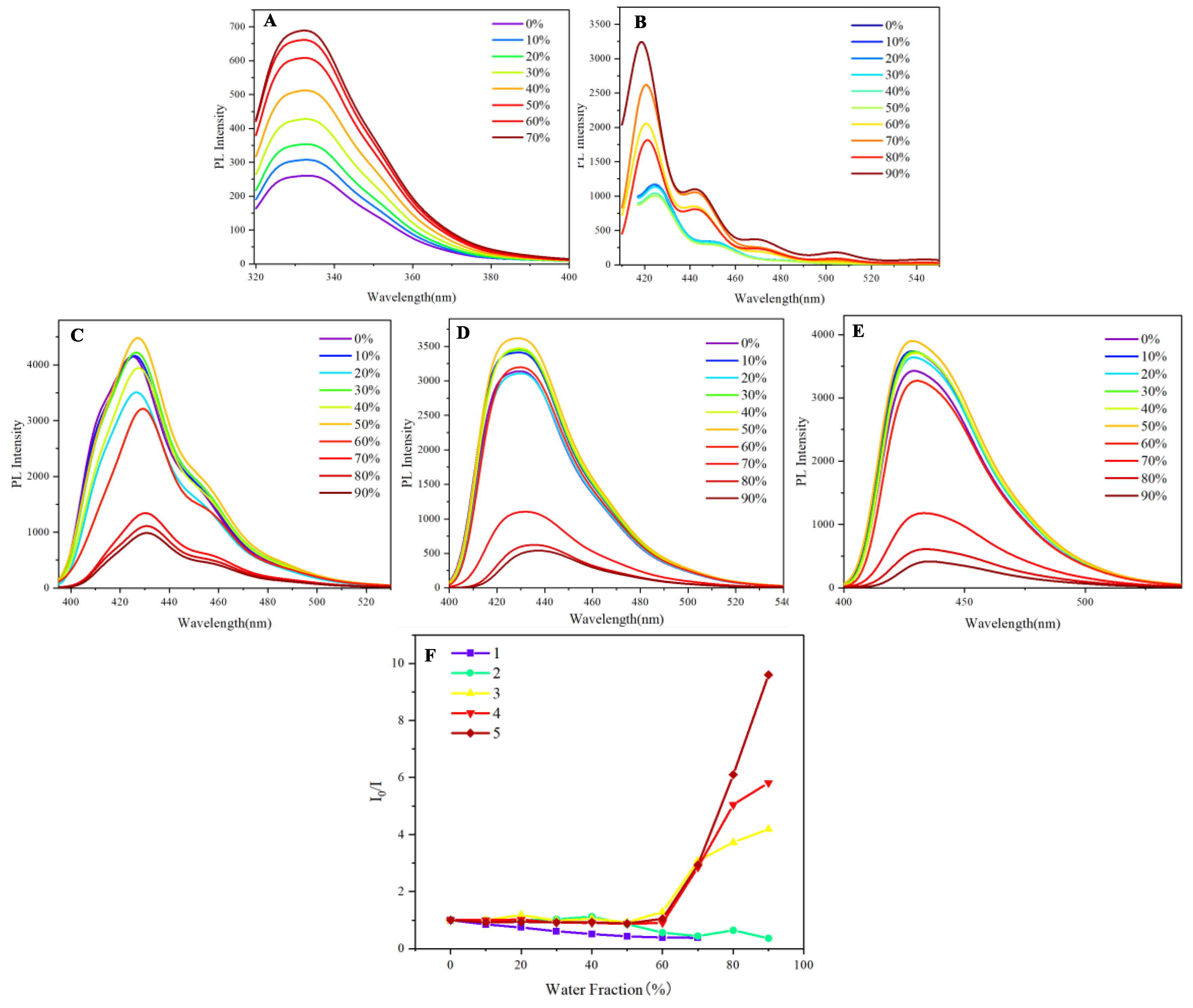
Having identified the aggregation behavior through UV spectra, the subsequent step involves measuring photoluminescence to assess the limitations imposed by aggregation. In the photoluminescence measurements, naphthalene and anthracene exhibited a pronounced aggregation-induced emission (AIE) phenomenon as the water fraction increased from 0% to 70%. Initially, both compounds demonstrated weak emission in pure organic solvents due to non-radiative decay processes prevalent in their dissolved states. However, as the water fraction was gradually increased, a significant enhancement in luminescence was observed, indicative of AIE (
Figure 3A and
Figure 3B). This enhancement can be attributed to the aggregation of the naphthalene and anthracene molecules, driven by hydrophobic interactions in the presence of water. At approximately 70% water content, the emission intensity peaked, suggesting an optimal aggregation state that effectively restricted intramolecular motion and minimized energy loss through non-radiative pathways. Beyond this threshold, further increases in water content led to a decline in luminescence, likely due to excessive aggregation or solubility issues. These results highlight the critical role of solvent composition in modulating the photophysical properties of naphthalene and anthracene (
Figure 3A and
Figure 3B). The observed bathochromic shift of approximately 5-10 nm in the emission maximum of anthracene (
2) as the water fraction increases from 0-50% to 60-90% is indicative of aggregate formation. This redshift is characteristic of the formation of J-aggregates or excimers, where the close packing of molecules in the aggregated state results in a lower energy excited state. The more pronounced shift at higher water fractions (60-90%) suggests the formation of larger or more ordered aggregates, which exhibit stronger intermolecular interactions and thus a greater stabilization of the excited state compared to the initial aggregates formed at lower water content.
In exploring the aggregation-induced emission (AIE) characteristics of naphthalene and anthracene, we extended our investigation to three derivatives: 9,10-di(naphthalen-1-yl)anthracene
3, 9,10-di(naphthalen-2-yl)anthracene
4, and 9-(naphthalen-1-yl)-10-(naphthalen-2-yl)anthracene
5. Unlike the pronounced AIE observed in naphthalene and anthracene, the derivatives displayed only a slight AIE phenomenon when the water fraction ranged between 0% and 50% (
3 in
Figure 3C,
4 in
Figure 3D, and
5 in
Figure 3E). This modest enhancement in fluorescence intensity can be attributed to the limited aggregation of the molecules, which, while promoting some degree of restriction on intramolecular motion, did not reach the optimal conditions for significant AIE. As the water fraction continued to rise beyond 50%, a noticeable quenching of fluorescence intensity was observed, culminating in a significant decrease as the water fraction approached 90% (
3 in
Figure 3C,
4 in
Figure 3D, and
5 in
Figure 3E). This quenching could be linked to excessive aggregation and potential solubility issues that disrupt the compact, rigid structure necessary for efficient radiative transitions. The observed slight AIE phenomenon in these derivatives suggests a scientific relationship with the AIE behavior of naphthalene and anthracene. While the derivatives exhibit reduced AIE efficiency, their photophysical properties still reflect the underlying principles of AIE, indicating that the structural modifications imparted by the naphthalene-anthracene combination influence their aggregation behavior and emission characteristics. The luminescence spectra of naphthalene and anthracene in THF/water mixtures exhibit broadened emission profiles, which may arise from the formation of aggregates or excimers under high water fractions. While literature reports distinct emission maxima for these compounds in monomeric form (e.g., naphthalene at 321, 334 nm and anthracene at 375, 396, 420 nm in pure solvents), the observed spectral similarity in our study likely reflects the dominant contribution of aggregated species under AIE conditions. This behavior aligns with previous studies on AIE-active systems, where aggregation can lead to significant spectral shifts and broadening.[
19]
-[
21]
Figure 3 shows that derivatives 3-5 exhibit minimal emission enhancement (≤10%) within the 0–50% water fraction range, within experimental error margins (±3%). This suggests their AIE activity is significantly weaker compared to 1-2, likely due to structural constraints limiting aggregation-induced rigidification. The observed trends align with their modest spectral shifts in UV studies (
Figure 2).