Submitted:
15 December 2025
Posted:
16 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Raw Materials and Preparation of Sorbents
2.2. Assessment of Structural, Chemical, and Microstructural Characteristics
2.3. Specific Surface Area and Sorption Tests
3. Results and Discussion
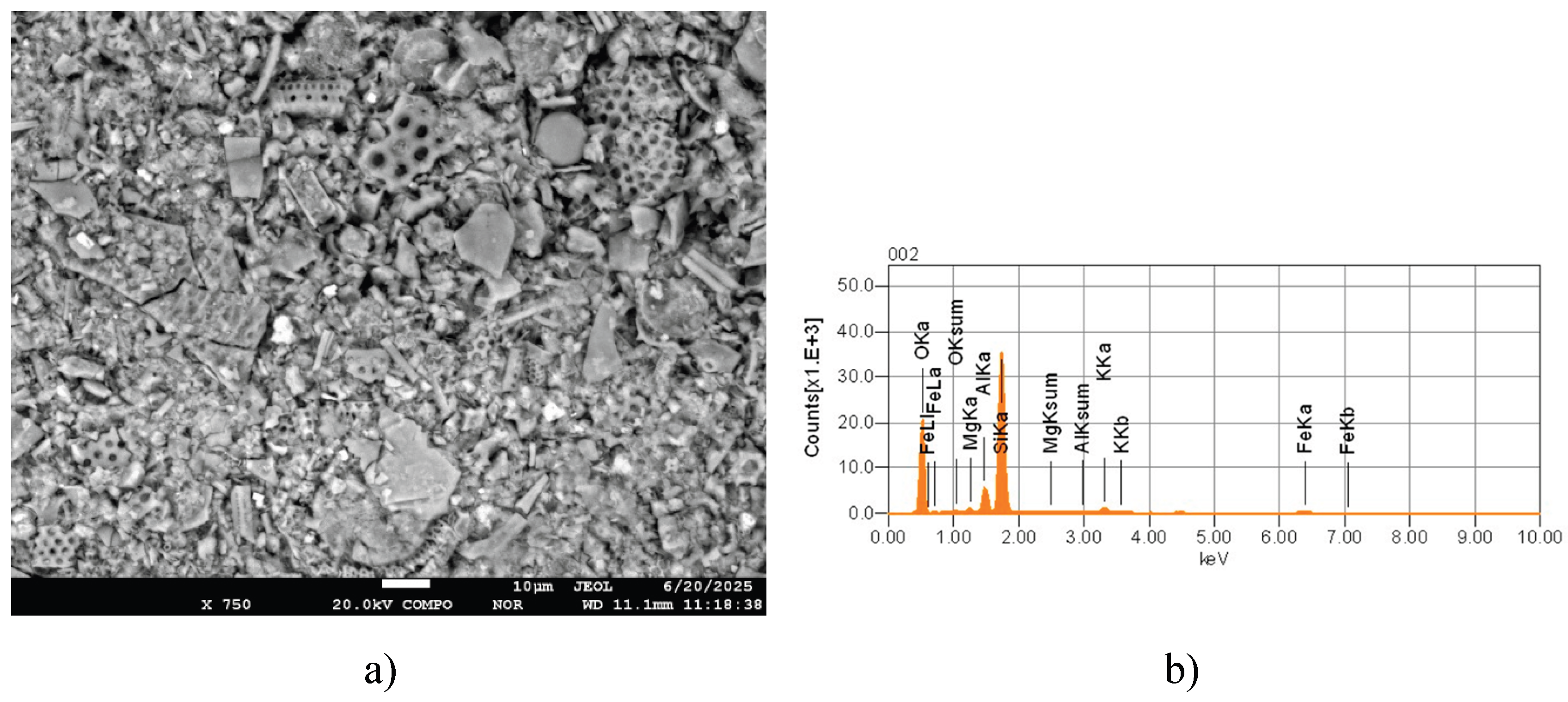
3.1. Structural-Morphological and Elemental-Phase Characteristics of Minerals
3.2. The Effect of Heat Treatment on the Oxide Composition of Diatomite
3.3. FTIR Analysis Results
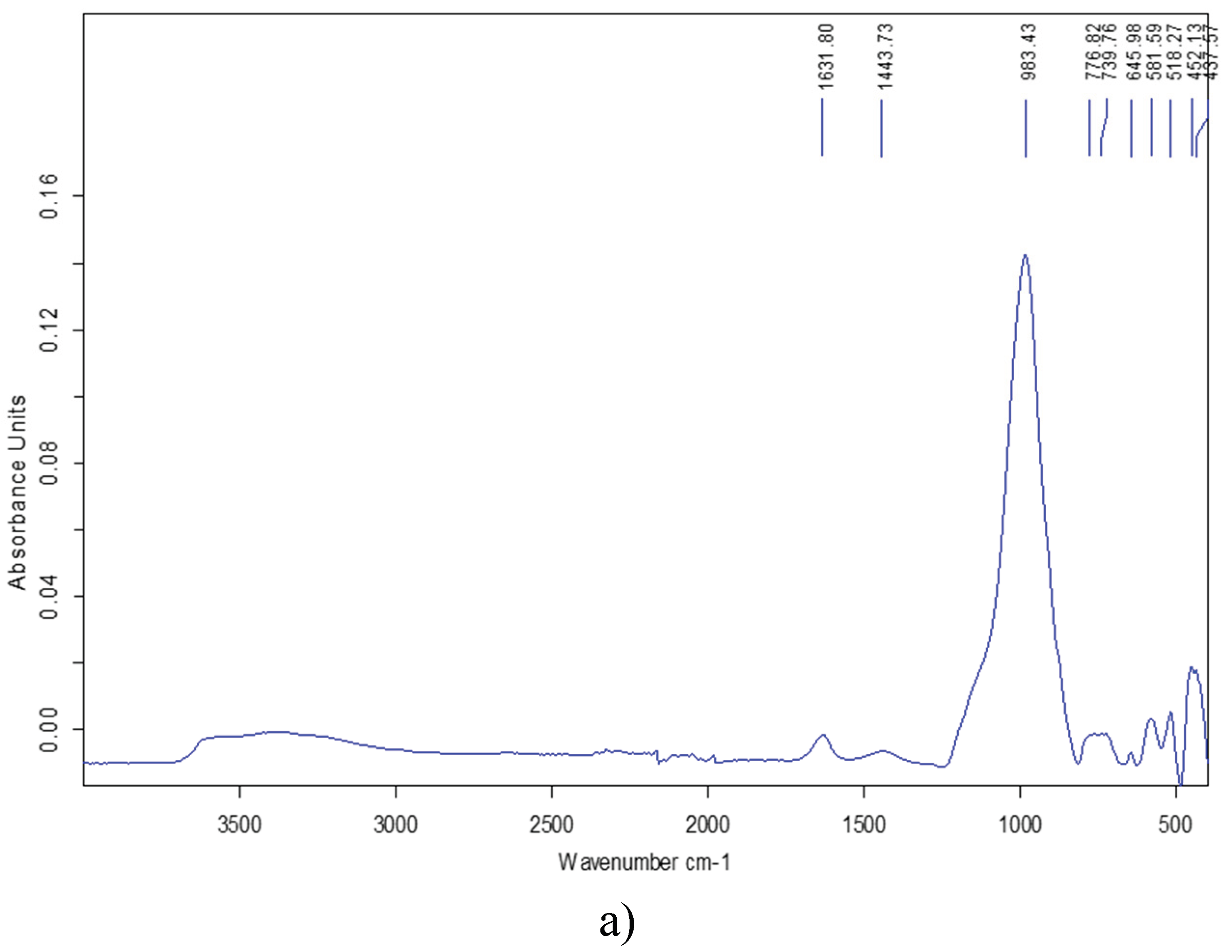
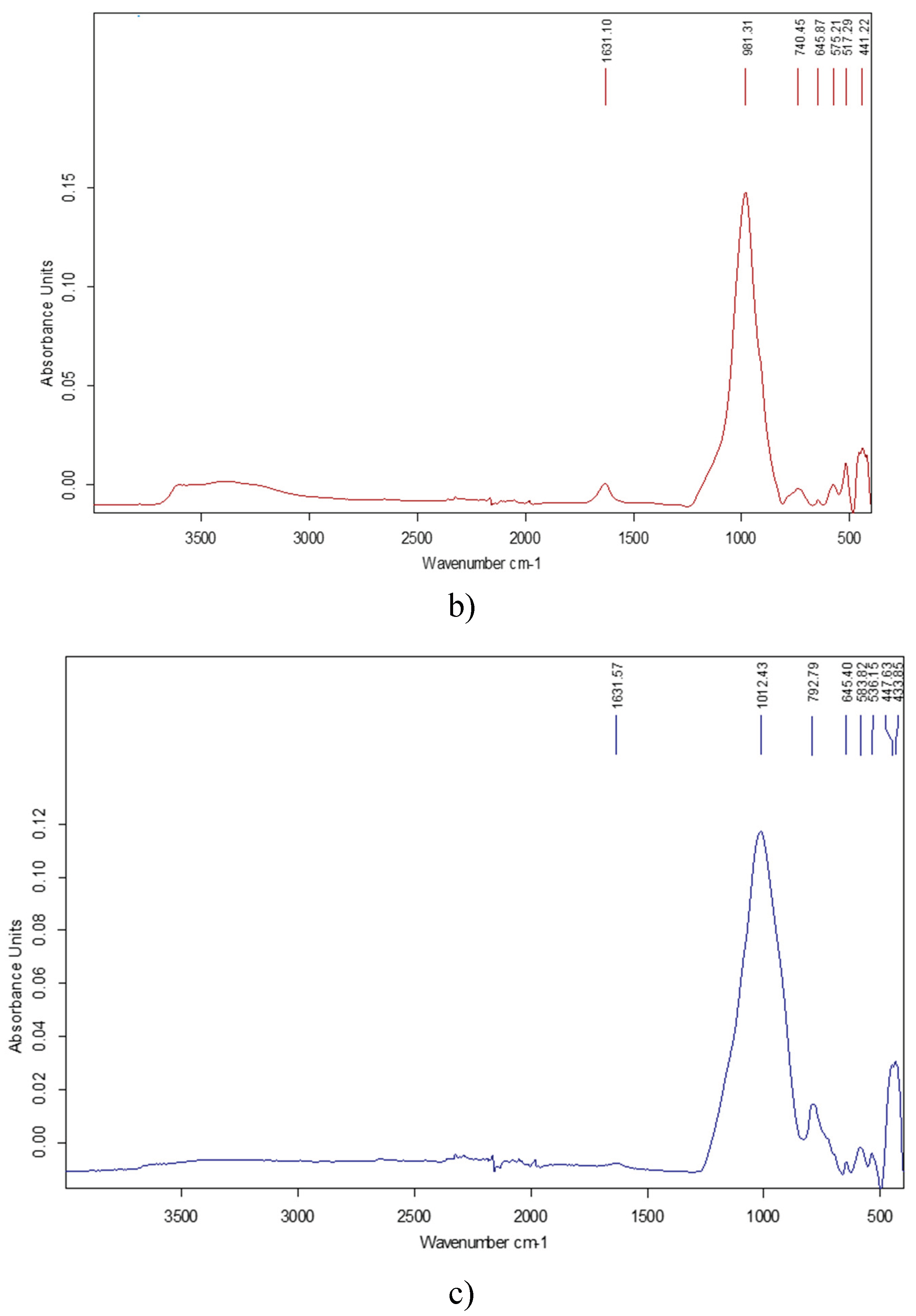
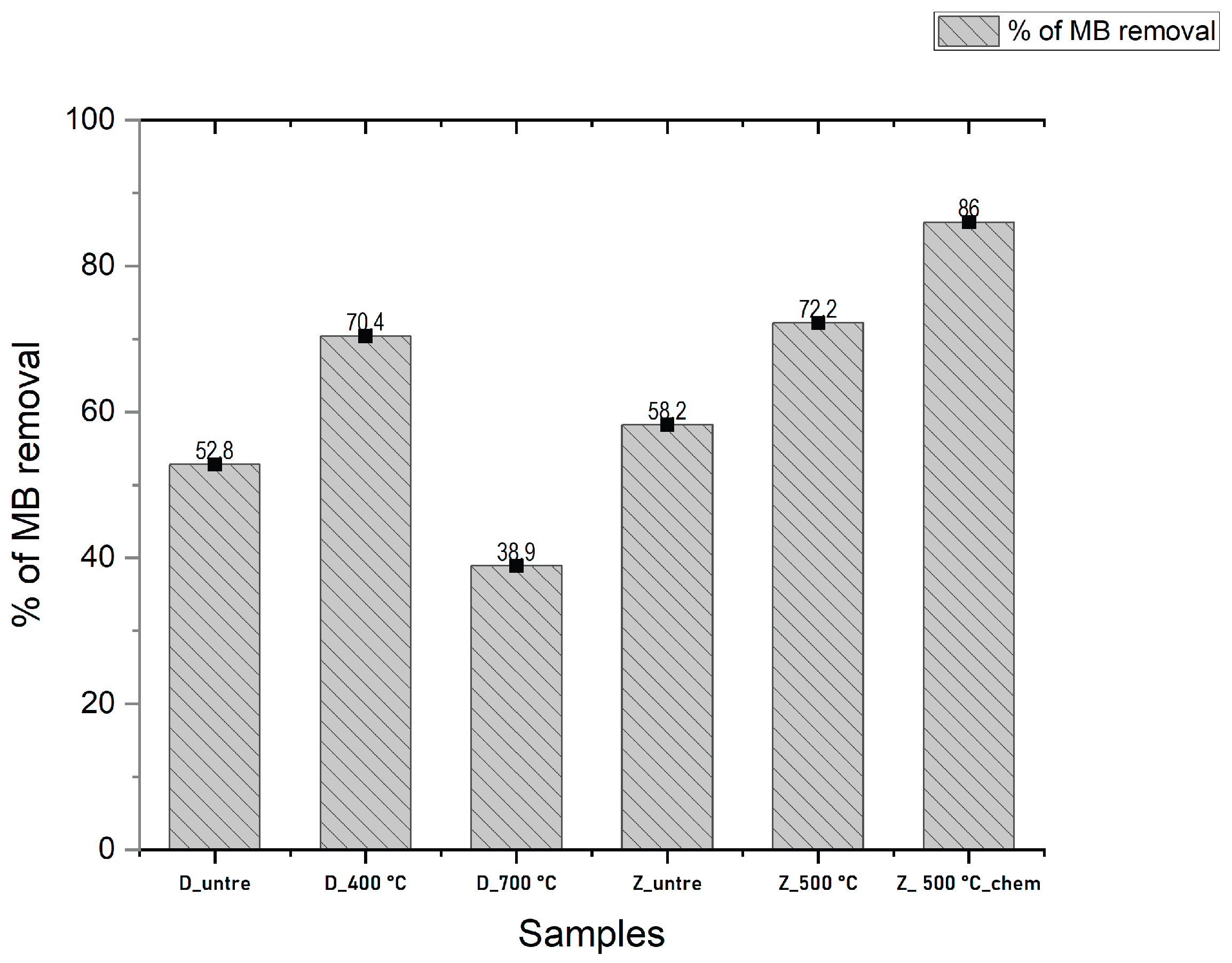
3.4. BET-Analysis and Testing of Dynamic MB Adsorption
| Indicators | Value | ||||||
| Samples | D_400 °C | D_700 °C | D_untre | Z_untre | Z_500 °C | Z_500 °C_chem | |
| Initial concentration C₀, mg/l | 820 | 243 | 501 | 387 | 343 | 228 | 115 |
| Weight of sorbent (m_sorbent), g | 10 | ||||||
| Sorbent fraction, mm | 0,5-1 | ||||||
| Column diameter D, cm | 4,0 | ||||||
| Layer height H, cm | 2,5 | ||||||
| Volume of the supplied solution, ml | 200,0 | ||||||
| Flow rate Q, ml/min | 4 | ||||||
| Feed type (gravity-fed / pump-driven) | gravity-fed | - | - | ||||
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| D_400 °C | Diatomite samples with heat treatment at 400 °C |
| D_500 °C | Diatomite samples with heat treatment at 500 °C |
| D_untre | Samples of untreated natural diatomite |
| Z_500 °C | Zeolite samples with heat treatment at 500 °C |
| Z_500 °C_chem | Zeolite samples with chemical activation and subsequent heat treatment at 500°C |
| Z_untre | Samples of untreated natural zeolite |
References
- Grizzetti, B.; Lanzanova, D.; Liquete, C.; Reynaud, A.; Cardoso, A. C. Assessing water ecosystem services for water resource management. Environ. Sci. Policy 2016, 61, 194–203. [Google Scholar] [CrossRef]
- Choudhary, M.; Peter, C. N.; Shukla, S. K.; Govender, P. P.; Joshi, G. M.; Wang, R. Environmental issues: a challenge for wastewater treatment. In Green Materials for Wastewater Treatment; Springer International Publishing: Cham, 2019; pp. 1–12. [Google Scholar] [CrossRef]
- Xu, Zihan; Peng, Jian. Recognizing ecosystem service's contribution to SDGs: Ecological foundation of sustainable develop-ment. Geo. and Sustain 2024, 5, 511–525. [Google Scholar] [CrossRef]
- Salgot, M.; Folch, M. Wastewater treatment and water reuse. Curr. Opin. Environ. Sci. Health 2018, 2, 64–74. [Google Scholar] [CrossRef]
- Dutta, Deblina; Arya, Shashi; Kumar, Sunil. Industrial wastewater treatment: Current trends, bottlenecks, and best practices. Chemosphere 2021, 285, 131245. [Google Scholar] [CrossRef]
- Musina, U. Sh. Environmental properties of shale shungite in the treatment of wastewater of various technogenic origins ( 2015, (2–2), 12. Available online: https://science-education.ru/ru/article/view?id=21572 (accessed on 12 June 2025).
- Caputo, D.; Pepe, F. Experiments and data processing of ion exchange equilibria involving Italian natural zeolites: a review. Microporous Mesoporous Mater. 2007, 105, 222–231. [Google Scholar] [CrossRef]
- Sabitov, A.; Atamanov, M.; Doszhanov, O.; Saurykova, K.; Tazhu, K.; Kerimkulova, A.; Orazbayev, A.; Doszhanov, Y. Surface Characteristics of Activated Carbon Sorbents Obtained from Biomass for Cleaning Oil-Contaminated Soils. Molecules 2024, 29, 3786. [Google Scholar] [CrossRef]
- Bizhanova, G. Z.; Abdibattaeva, M. M.; Musina, U. Sh.; Kurbanova, L. S.; Tusupova, B. Kh. Natural sorbents: an envi-ronmentally friendly alternative for water treatment. Geography and water resources (In Kazakh). 2025, № 3, 23–33 р. [Google Scholar] [CrossRef]
- Zahid, M.; Doszhanov, Ye.; Saurykova, K.; Ahmadi, N.; Bolatova, D.; Kurmanbayeva, M.; Aydarbek, A.; Ihsas, R.; Seitzhanova, M.; Akhmetzhanova, D.; et al. Modification and Application of Natural Clinoptilolite and Mordenite from Almaty Region for Drinking Water Purification. Molecules 2025, 30, 2021. [Google Scholar] [CrossRef] [PubMed]
- El Sayed, E. E. Natural diatomite as an effective adsorbent for heavy metals in water and wastewater treatment (a batch study). Water Sci. 2018, 32, 32–43. [Google Scholar] [CrossRef]
- Łach, M.; Pławecka, K.; Marczyk, J.; Ziejewska, C.; Hebdowska-Krupa, M.; Nykiel, M.; Hebda, M.; Miernik, K.; Mierzwiński, D.; Korniejenko, K.; Mikuła, J.; Smoroń, K. Use of diatomite from Polish fields in sustainable development as a sorbent for petroleum substances. J. Clean. Prod. 2023, 389, 136100. [Google Scholar] [CrossRef]
- Myrzalieva, S.; Bagasharova, J.; Akilbekova, S.; Serikbayev, P. Natural mineral raw materials as granular filtering materials in industrial and waste water treatment. Kompleksnoe Ispolzovanie Mineralnogo Syra = Complex Use of Mineral Resources 2024, 332(1), 70–78. [Google Scholar] [CrossRef]
- Nikolaeva, L. A.; Aikenova, N. E.; Demin, A. V. Purification of wastewater of industrial enterprises from phenols by modified energy waste. RUDN J. Ecol. Life Saf. 2021, 29, 174–181. [Google Scholar] [CrossRef]
- Zhao, Y.; Tian, G.; Duan, X.; Liang, X.; Meng, J.; Liang, J. Environmental Applications of Diatomite Minerals in Removing Heavy Metals from Water. Ind. Eng. Chem. Res. 2019, 58, 11638–11652. [Google Scholar] [CrossRef]
- Nurgain, A.; Nazhipkyzy, M.; Özsin, G.; Zhaparova, A. A.; Apaydın-Varol, E. Efficient Pb(II) Adsorption by Natural Mugaldzhar Diatomite: Isotherm, Kinetic, and Thermodynamic Analysis. J. Compos. Sci. 2025, 9, 625. [Google Scholar] [CrossRef]
- Mansour, H.; Nassar, H. F.; Zaghloul, A.; et al. Preparation and evaluation of altered zeolite and diatomite as affordable adsorbents in contaminated water treatment. Appl. Water Sci. 2025, 15, 160. [Google Scholar] [CrossRef]
- Yang, Q.; Nong, J.; Xie, Q.; Chen, N. Research progress on application of modified diatomite in wastewater treatment. New Chem. Mater. 2022, 50(1), 298–302. [Google Scholar] [CrossRef]
- Michel, M. M.; Reczek, L.; Papciak, D.; Włodarczyk-Makuła, M.; Siwiec, T.; Trach, Y. Mineral Materials Coated with and Consisting of MnOx—Characteristics and Application of Filter Media for Groundwater Treatment: A Review. Materials 2020, 13, 2232. [Google Scholar] [CrossRef] [PubMed]
- Shewatatek, S.; Gonfa, G.; Hailegiorgis, S. M.; Tessema, B. Adsorptive removal of lead ions from wastewater using modified diatomite. J. Hazard. Mater. Adv. 2025, 20, 100900. [Google Scholar] [CrossRef]
- Wu, Z.; Han, Y.; Zan, F.; Ye, Y.; Ren, Y.; Han, K.; Liu, D.; Jiang, W. Highly efficient removal of phosphate by La–diatomite and sodium alginate composite hydrogel beads. Environ. Sci.: Water Res. Technol. 2023, 4, article. [Google Scholar] [CrossRef]
- Fang, S.; Li, G.; Shi, H.; Ye, J.; Wang, H.; Ding, X.; Luo, L.; Li, G.; Yang, M. Preparation of low-cost functionalized diatomite and its effective removal of ammonia nitrogen from wastewater. Environ. Sci. Pollut. Res. 2023, 30, 98881–98894. [Google Scholar] [CrossRef]
- Dehmani, Y.; Ba Mohammed, B.; Oukhrib, R.; Dehbi, A.; Lamhasni, T.; Brahmi, Y.; El-Kordy, A.; Franco, D. S. P.; Georgin, J.; Lima, E. C.; et al. Adsorption of various inorganic and organic pollutants by natural and synthetic zeolites: A critical review. Arab. J. Chem. 2024, 17, 105474. [Google Scholar] [CrossRef]
- Doszhanov, Ye.; Zahid, M.; Ahmadi, N.; Ihsas, R.; Kerimkulova, A.; Doszhanov, O.; Zhumazhanov, A.; Baizakova, N.; Akhmetzhanova, D.; Saurykova, K.; Aydarbek, A. Investigation of ion-exchange properties of zeolite modified with hydrochloric acid for water purification. Hydrometeorology and ecology 2025, (3), 78–87. [Google Scholar] [CrossRef]
- Bare, S. R.; Knop-Gericke, A.; Teschner, D.; Hävacker, M.; Blume, R.; Rocha, T.; Schlögl, R.; Chan, A. S. Y.; Blackwell, N.; Charochak, M. E.; et al. Surface analysis of zeolites: an XPS, variable kinetic energy XPS, and low energy ion scattering study. Surf. Sci. 2016, 648, 376–382. [Google Scholar] [CrossRef]
- Misaelides, P. Application of natural zeolites in environmental remediation: A short review. Microporous Mesoporous Mater. 2011, 144, 15–18. [Google Scholar] [CrossRef]
- Kuldeyev, E.; Seitzhanova, M.; Tanirbergenova, S.; Tazhu, K.; Doszhanov, E.; Mansurov, Z.; Azat, S.; Nurlybaev, R.; Berndtsson, R. Modifying Natural Zeolites to Improve Heavy Metal Adsorption. Water 2023, 15, 2215. [Google Scholar] [CrossRef]
- Mumpton, F. A. La roca magica: uses of natural zeolites in agriculture and industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef]
- Li, M.; Zhu, X.; Zhu, F.; Ren, G.; Cao, G.; Song, L. Application of modified zeolite for ammonium removal from drinking water. Desalination 2011, 271, 295–300. [Google Scholar] [CrossRef]
- Margeta, K. Z. N.; Siljeg, M.; Farkas, A. Natural zeolites in water treatment. How Effective is Their Use. In Water Treatment; Elshorbagy, W., Chowdhury, R., Eds.; 2013; pp. 81–112. [Google Scholar]
- Vladimirovich, B. V.; Semenovich, K. A.; Nikolaevich, K. V.; Aleksandrovich, Z. A. Natural sorbents and their use for wastewater treatment. Water Magazine 2017 (6), (118). 2017, p. (118). Available online: https://watermagazine.ru/nauchnye-stati2/novye-stati/23081-prirodnye-sorbenty-i-ikh-ispolzovanie-dlya-ochistki-stochnykh-vod.html (accessed on 15 July 2025).
- Zhang, B.; Wang, X.; Li, S.; Liu, Y.; An, Y.; Zheng, X. Preferable Adsorption of Nitrogen and Phosphorus from Agricultural Wastewater Using Thermally Modified Zeolite–Diatomite Composite Adsorbent. Water 2019, 11, 2053. [Google Scholar] [CrossRef]
- Seitzhanova, M.; Doszhanov, Ye.; Kuldeev, E.; Mansurov, Z.; Tazhu, K.; Tanirbergenova, S.; Kanzharkan, E.; Tajkenova, G. Influence of thermal treatment on sorption characteristics of zeolite used in water treatment. Combust. Plasma Chem. 2023, 21(3), 173–179. [Google Scholar] [CrossRef]
- Korzhynbayeva, K.; Medetkhan, R.; Diaz, M.; Tazhibayeva, S.; Musabekov, K. Peculiarities of methylene blue adsorption on the diatomite surface. Chem. Bull. Kazakh Natl. Univ. 2013, 72(4), 64–69. [Google Scholar] [CrossRef]
- Mohamed, E. A.; Selim, A. Q.; Zayed, A. M.; Komarneni, S.; Mobarak, M.; Seliem, M. K. Enhancing adsorption capacity of Egyptian diatomaceous earth by thermo-chemical purification: Methylene blue uptake. J. Colloid Interface Sci. 2019, 534, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, P.; Kumar, A. Diatomite chemical activation for effective adsorption of methylene blue dye from model textile wastewater. Int. J. Environ. Sci. Dev. 2021, 12(1), 23–28. [Google Scholar] [CrossRef]
- Tanirbergenova, S.; Tugelbayeva, D.; Zhylybayeva, N.; Aitugan, A.; Tazhu, K.; Moldazhanova, G.; Mansurov, Z. Effect of Acid Treatment on the Structure of Natural Zeolite from the Shankhanai Deposit. Processes 2025, 13, 2896. [Google Scholar] [CrossRef]

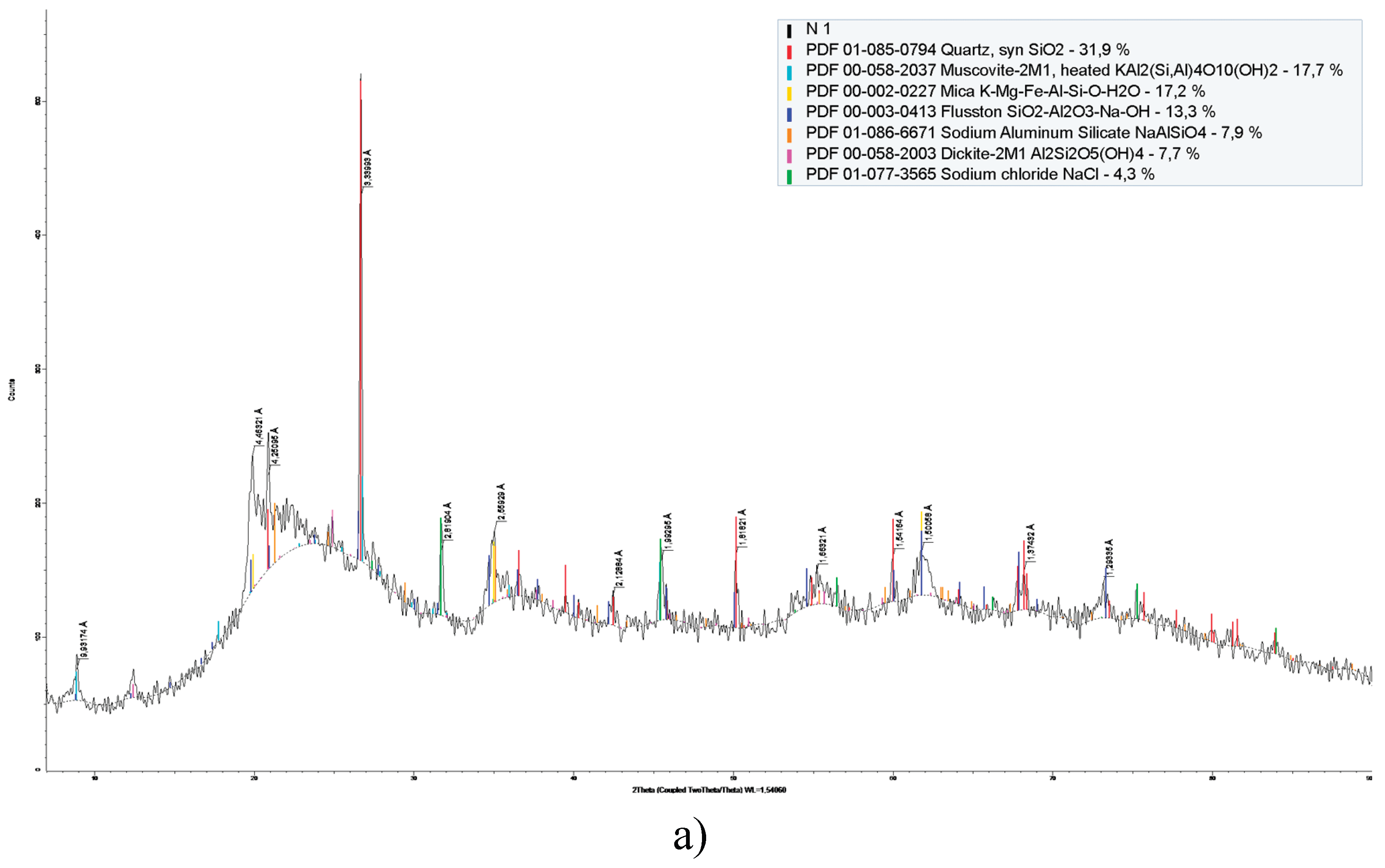
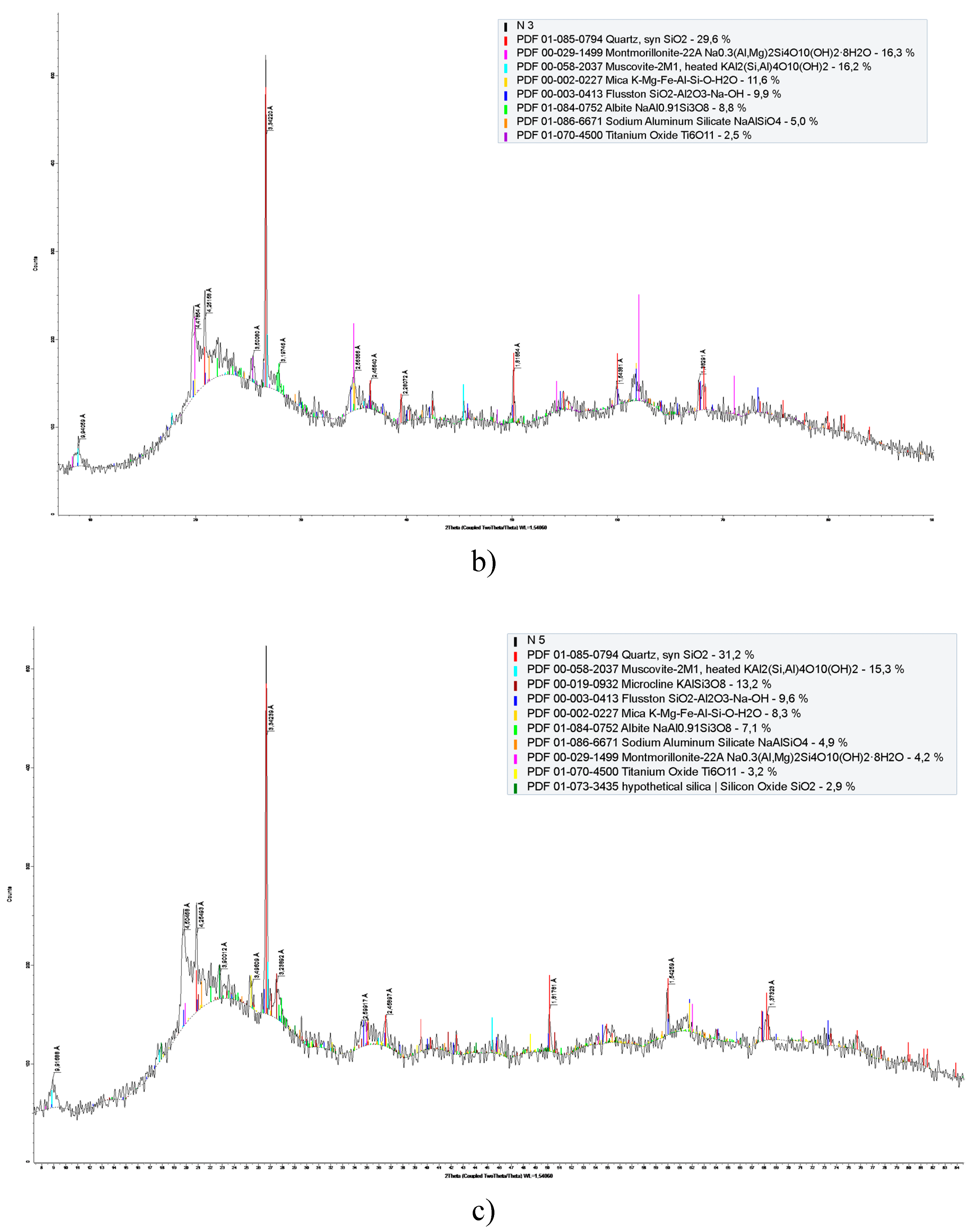
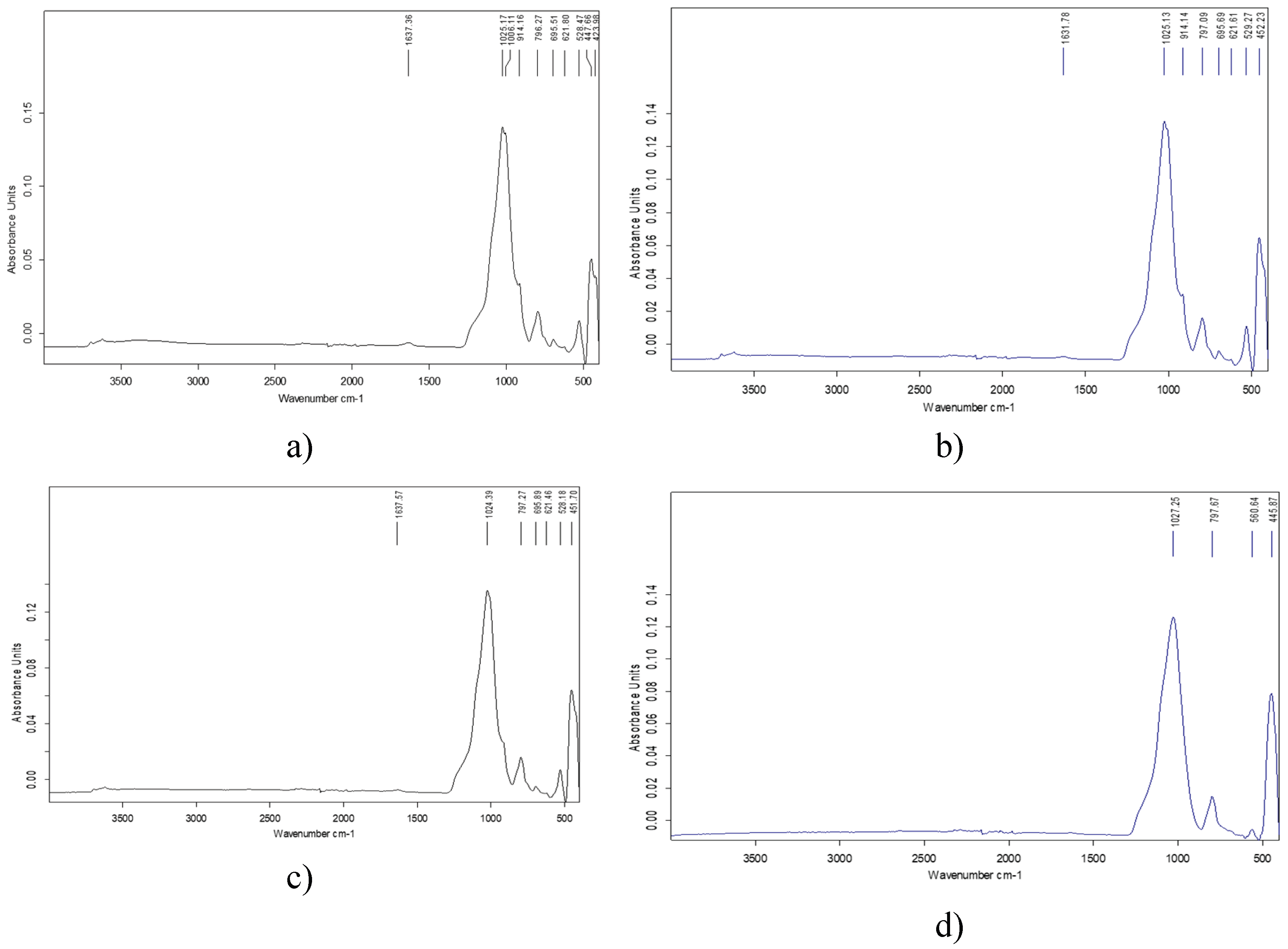
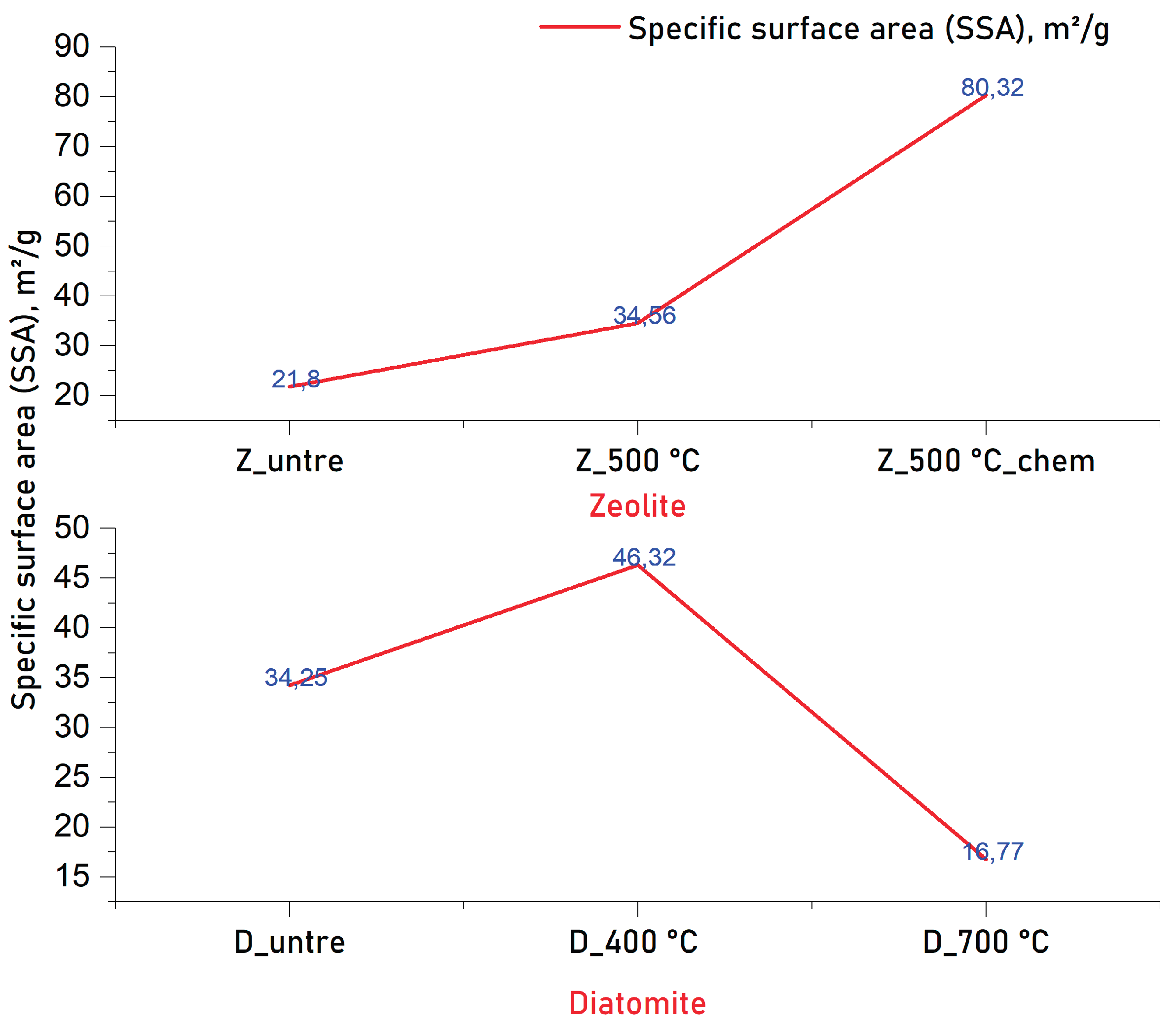
| Component | Concentration, % | |
| Diatomite (Utesai deposit) |
Zeolite (Shanghanai deposit) [26] |
|
| SiO2 | 82,630 | 60,0-74,0 |
| Al2O3 | 8,622 | 14,0-15,0 |
| Fe2O3 | 2,819 | 1,40-5,83 |
| MgO | 1,089 | 0-2,120 |
| K2O | 0,646 | 0,660-4,030 |
| CaO | 0,191 | 0,130-6,400 |
| TiO2 | 1,320 | 0,070-0,700 |
| Na2O | 1,380 | 0,610-5,450 |
| P2O5 | 0,031 | 0,012-0,173 |
| SO3 | 0,221 | - |
| V2O5 | 0,030 | - |
| Cl | 0,936 | - |
| Cr2O3 | 0,025 | - |
| MnO | - | 0,067-0,199 |
| Component | Concentration, % | ||
| 400 °C | 500 °C | 700 °C | |
| exposure | 60 minutes | ||
| SiO2 | 83,443 | 83,791 | 83,502 |
| Al2O3 | 8,602 | 8,331 | 8,650 |
| Fe2O3 | 2,686 | 2,627 | 2,633 |
| MgO | 1,093 | 1,121 | 1,151 |
| K2O | 0,807 | 0,628 | 0,633 |
| CaO | 0,225 | 0,272 | 0,302 |
| TiO2 | 1,226 | 1,309 | 1,298 |
| Na2O | 1,051 | 1,257 | 1,330 |
| P2O5 | 0,030 | 0,029 | 0,027 |
| SO3 | 0,285 | 0,328 | 0,389 |
| V2O5 | 0,020 | - | 0,034 |
| Cl | 0,506 | 0,259 | 0,017 |
| Cr2O3 | - | 0,024 | 0,014 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




