Submitted:
13 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Historical Development and Persistent Challenges
1.2. Criteria for Element Classification
1.3. Anomalies and Alternative Representations
1.4. Central Positioning and Spiral Representations
1.5. Contemporary Perspectives
1.6. Scope and Objectives
1.7. Information Theory
1.8. Objectives and Scope
2. Computational Methodology
3. Information-Theoretic Measures
3.1. Electronic Density Representations
3.2. Shannon Entropy
3.3. Fisher Information
3.4. Disequilibrium
3.5. Complexity Measures
4. Statistical Method: Student’s t-test
4.1. Shapiro-Wilk Test for Normality
4.2. Probability Plots
4.3. Welch’s t-test
5. Results and Discussion
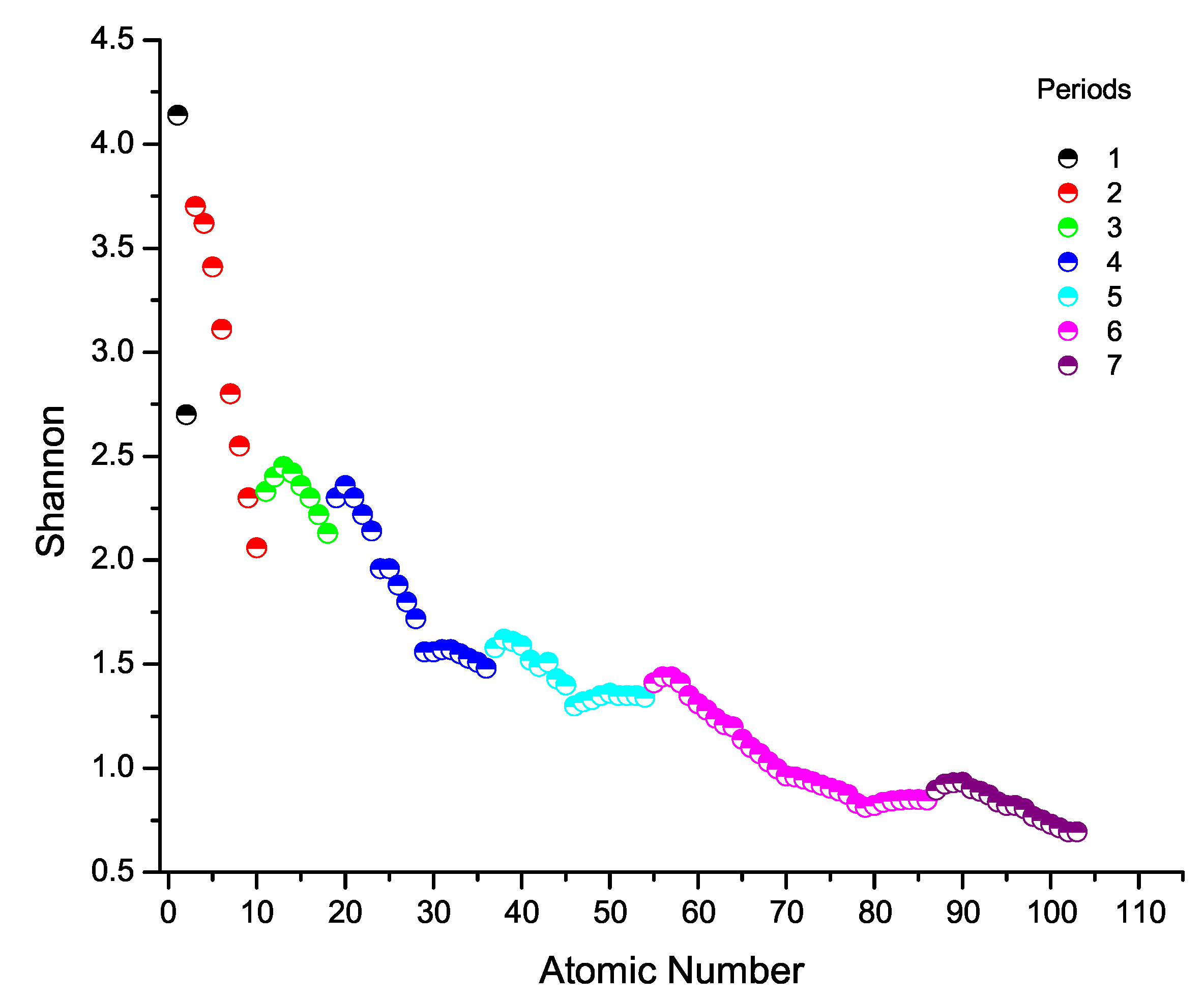
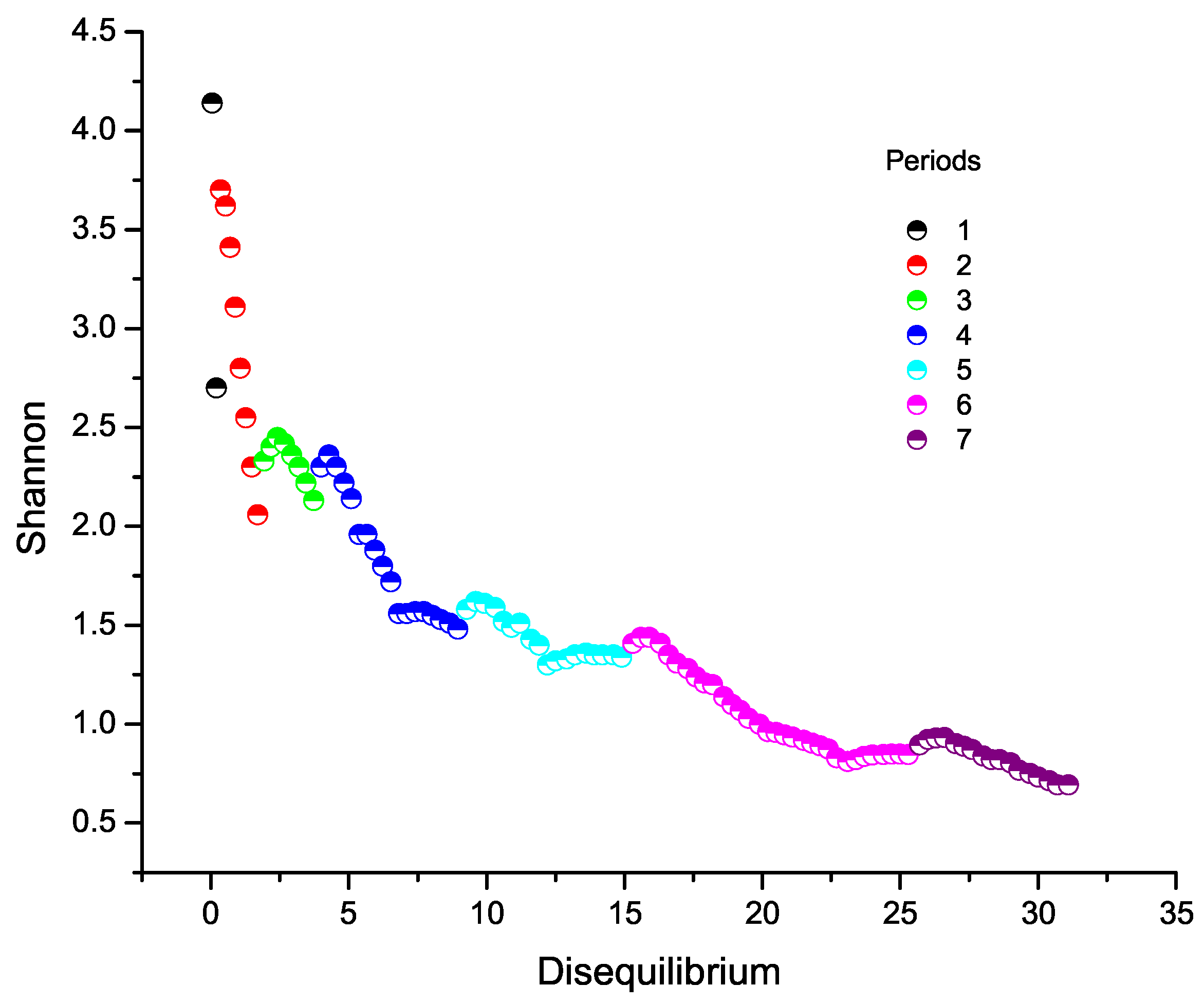
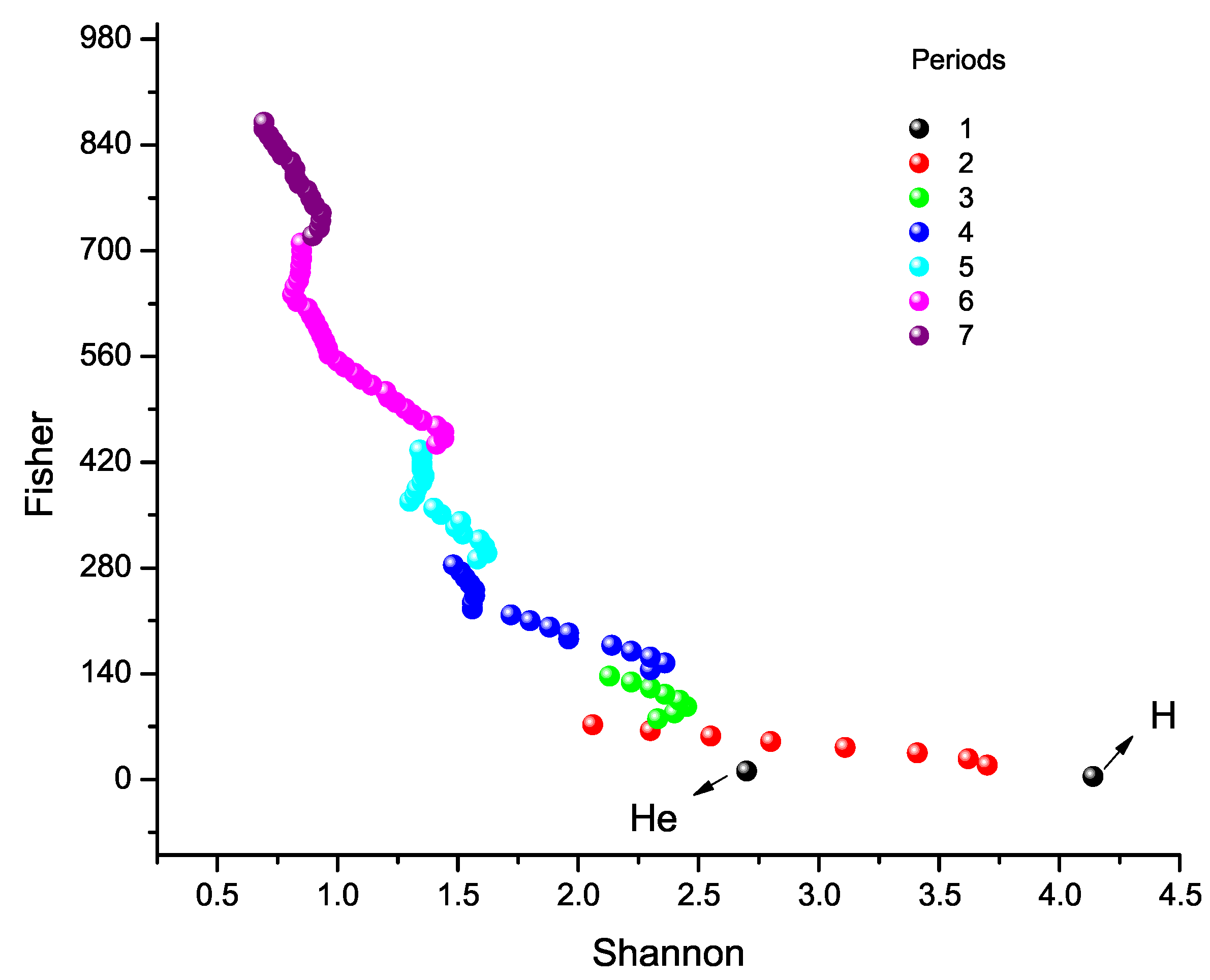
6. Shannon Entropy Across the Periodic Table
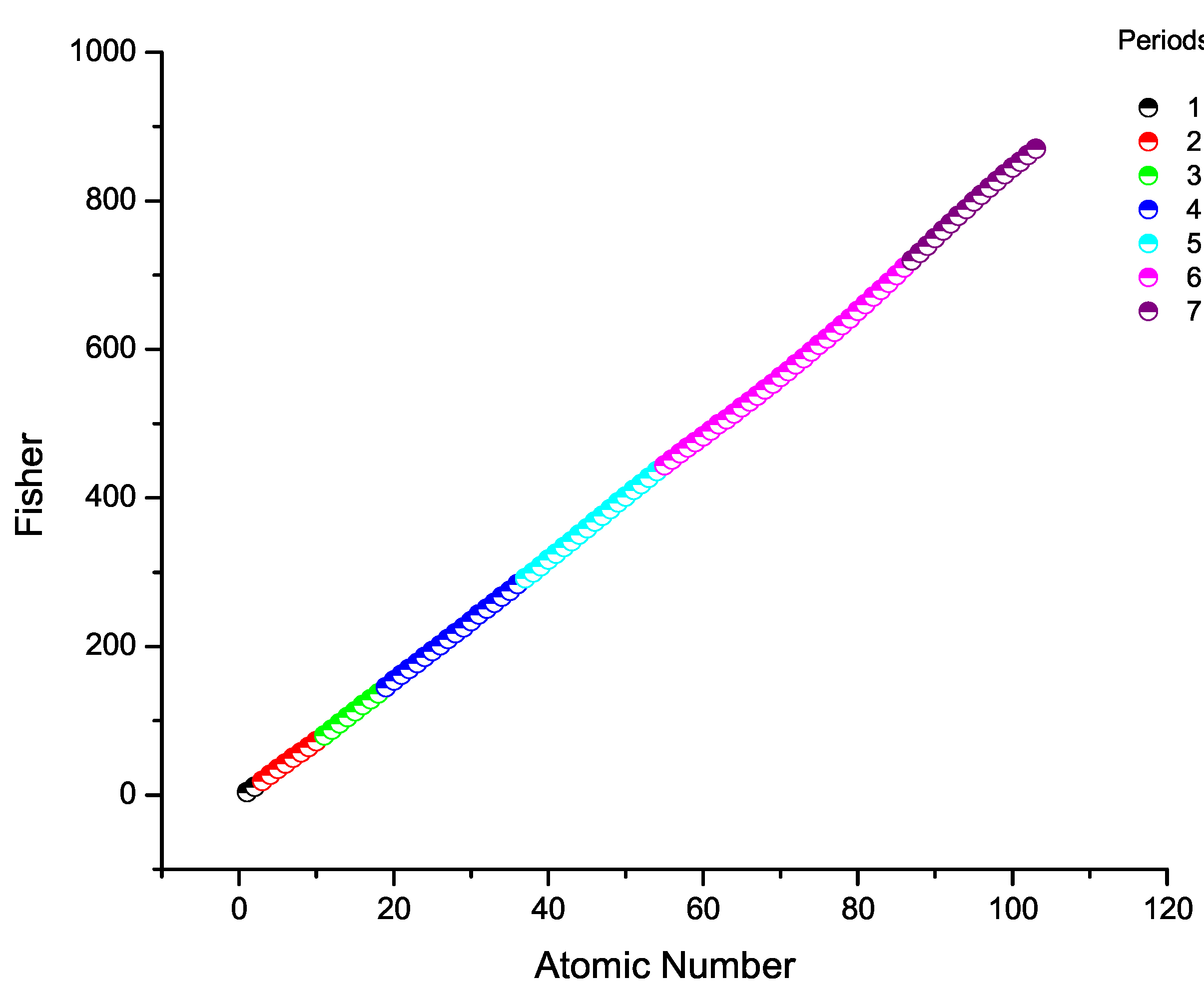
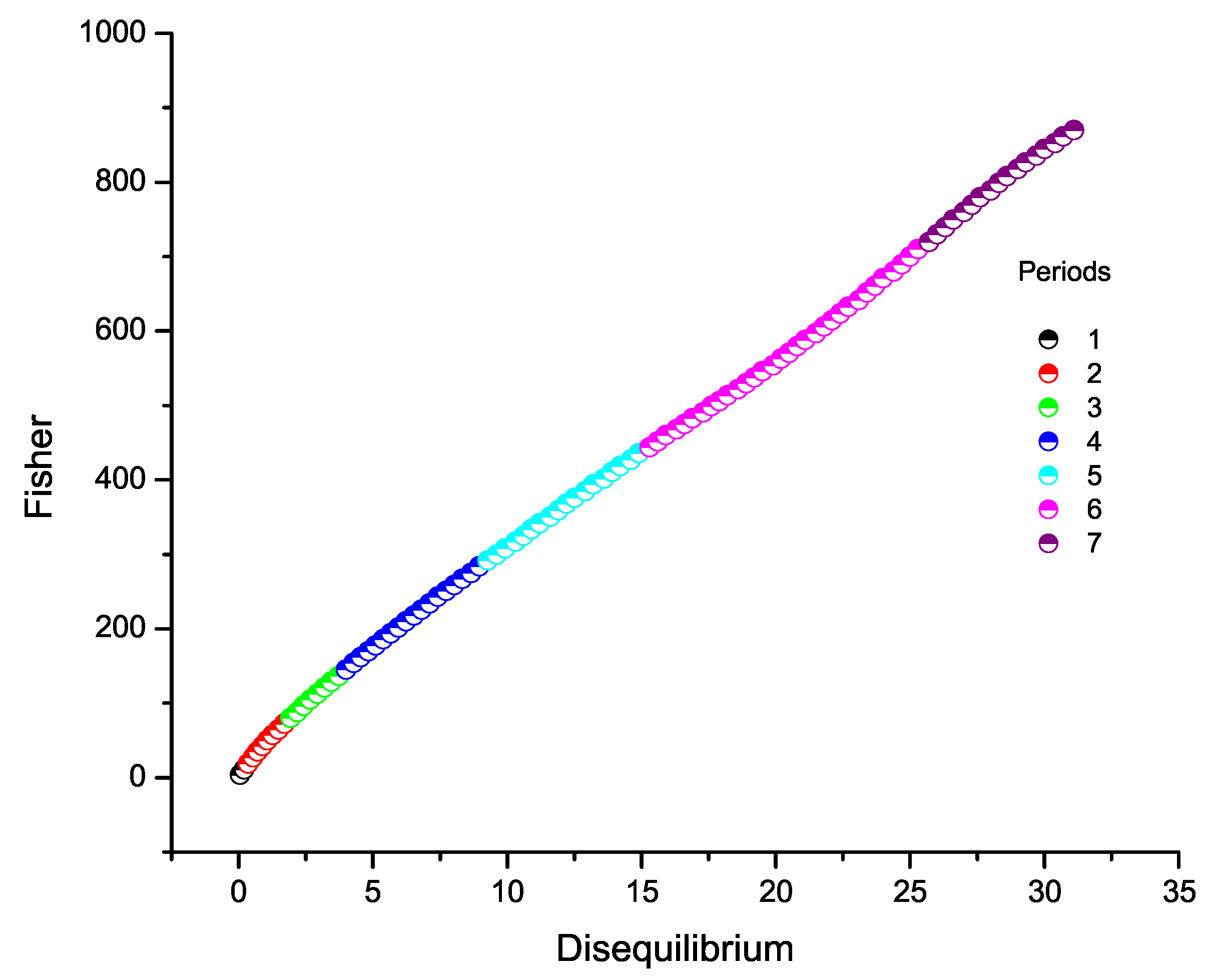
7. Fisher Information Across the Periodic Table
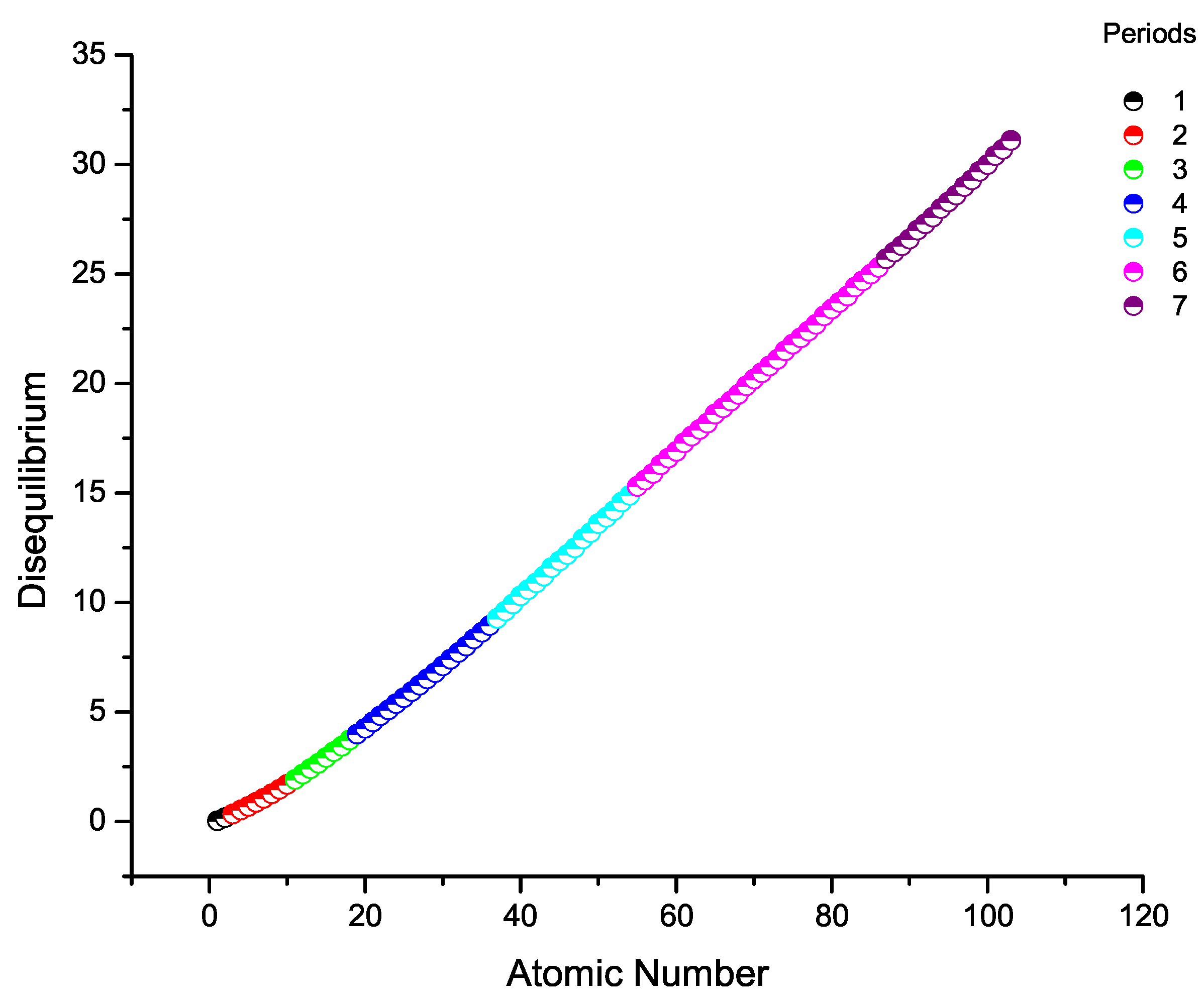
8. Disequilibrium Across the Periodic Table
9. Relationships Between Information-Theoretic Measures
9.1. Shannon Entropy versus Disequilibrium
9.2. Fisher Information versus Disequilibrium
9.3. Fisher Information versus Shannon Entropy
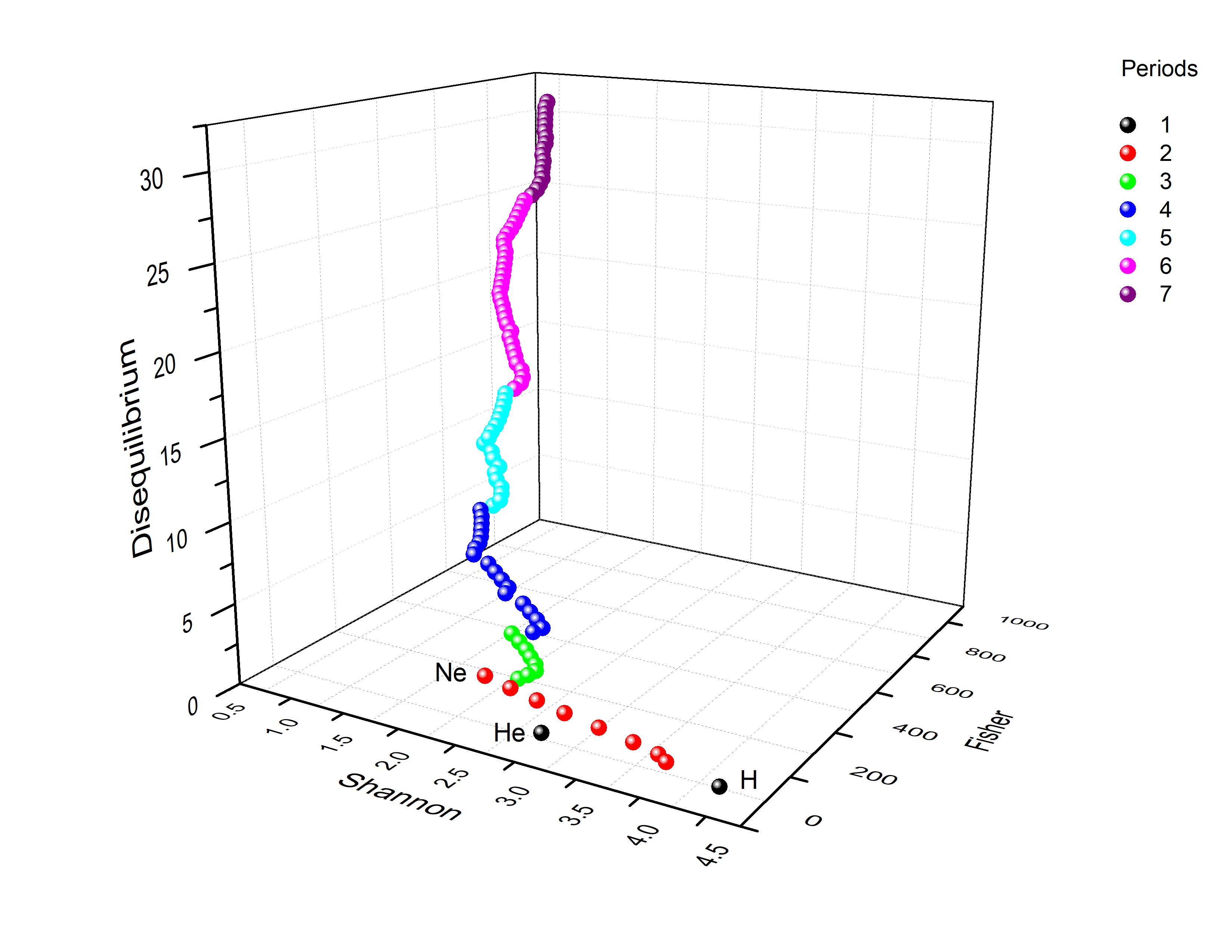
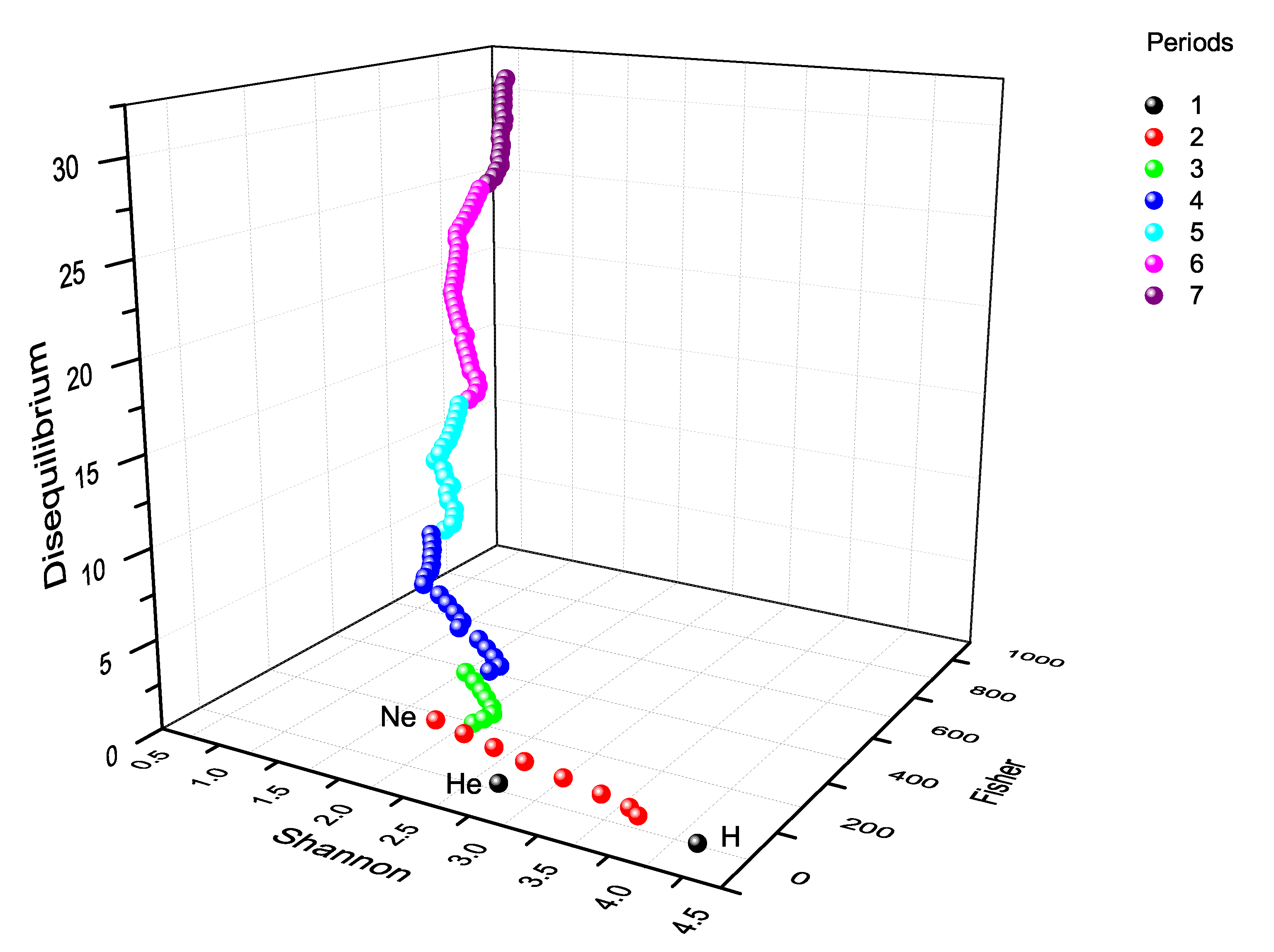
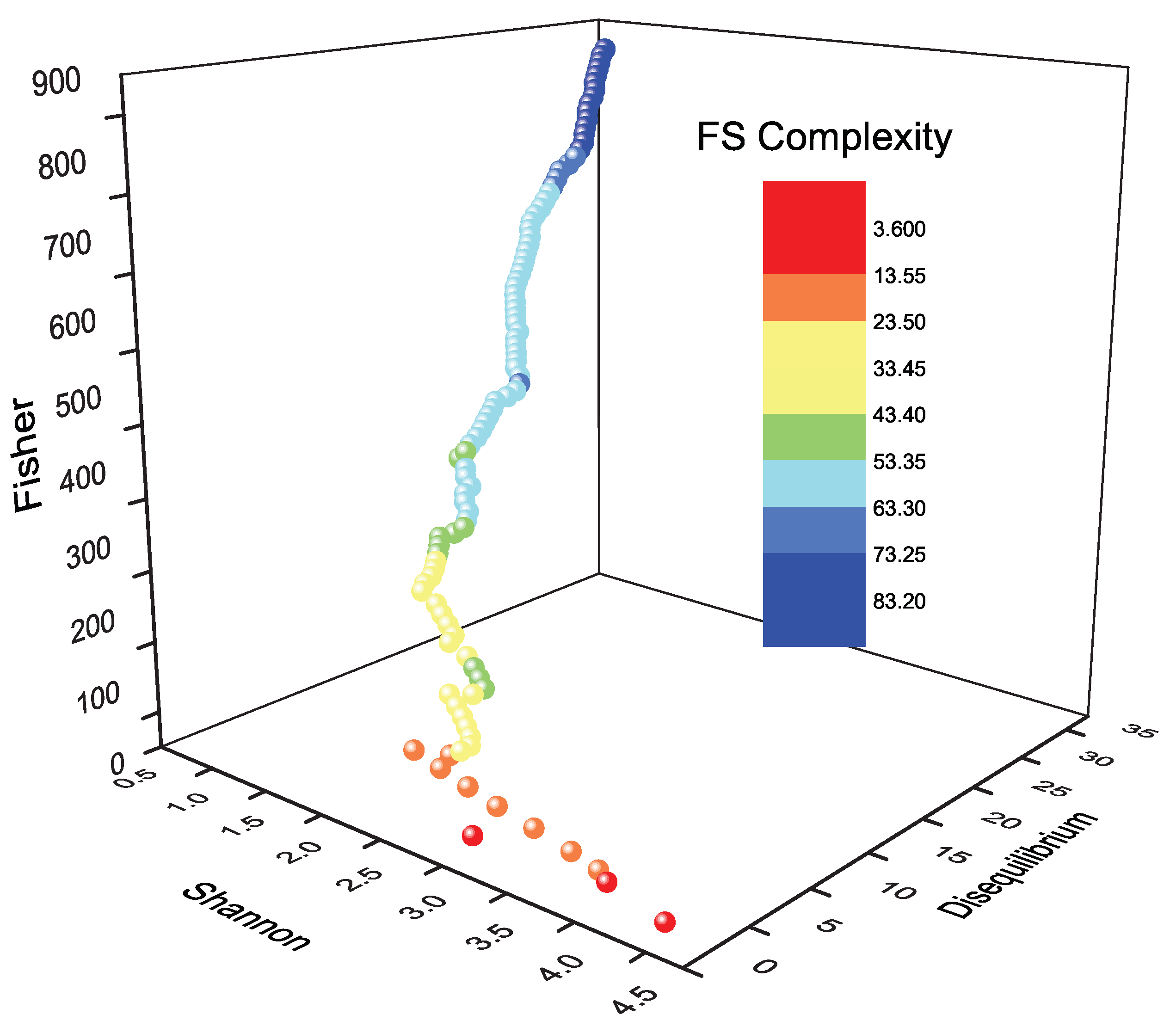
10. Three-Dimensional Information-Theoretic Landscape
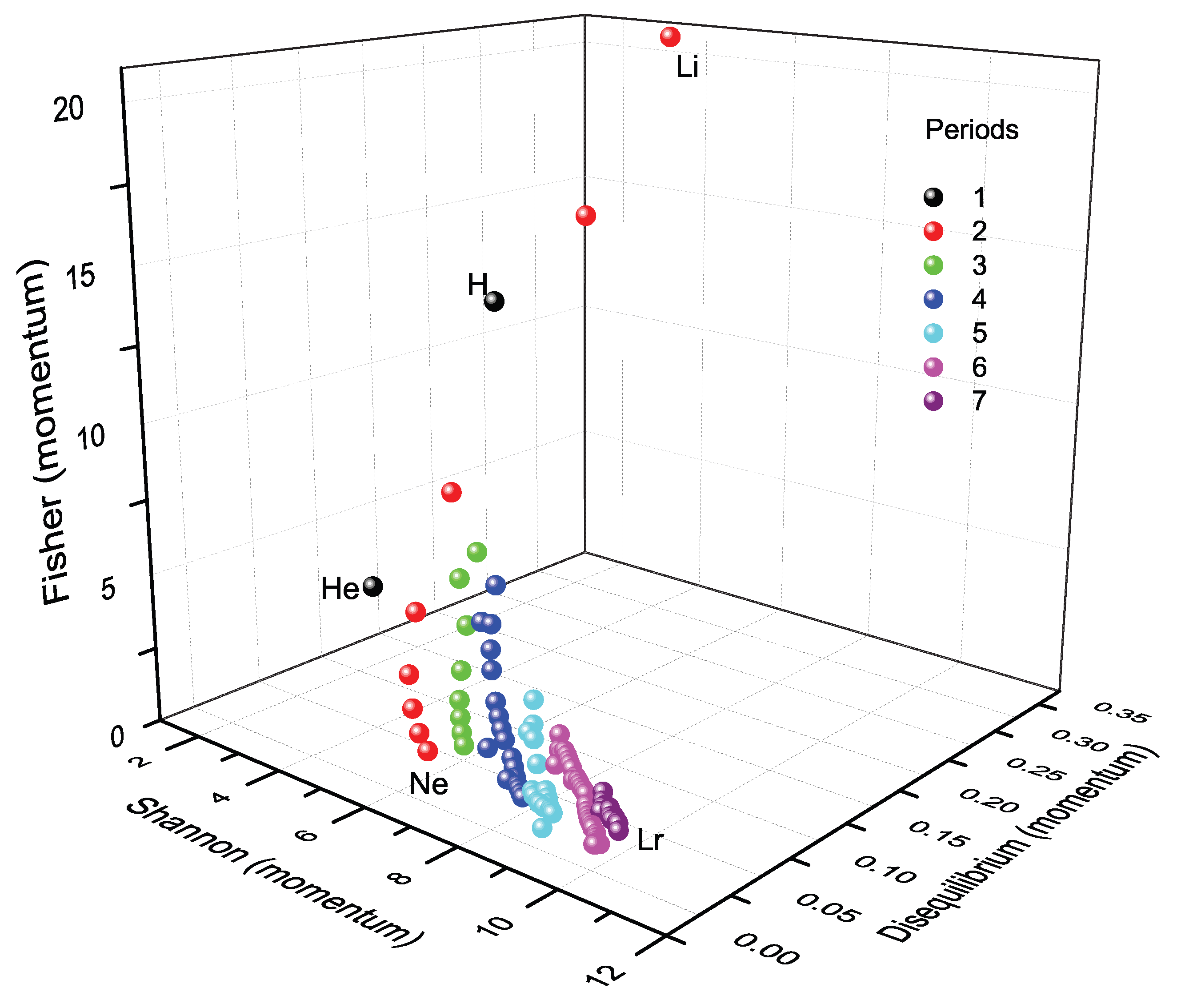
10.1. Periodic Representation
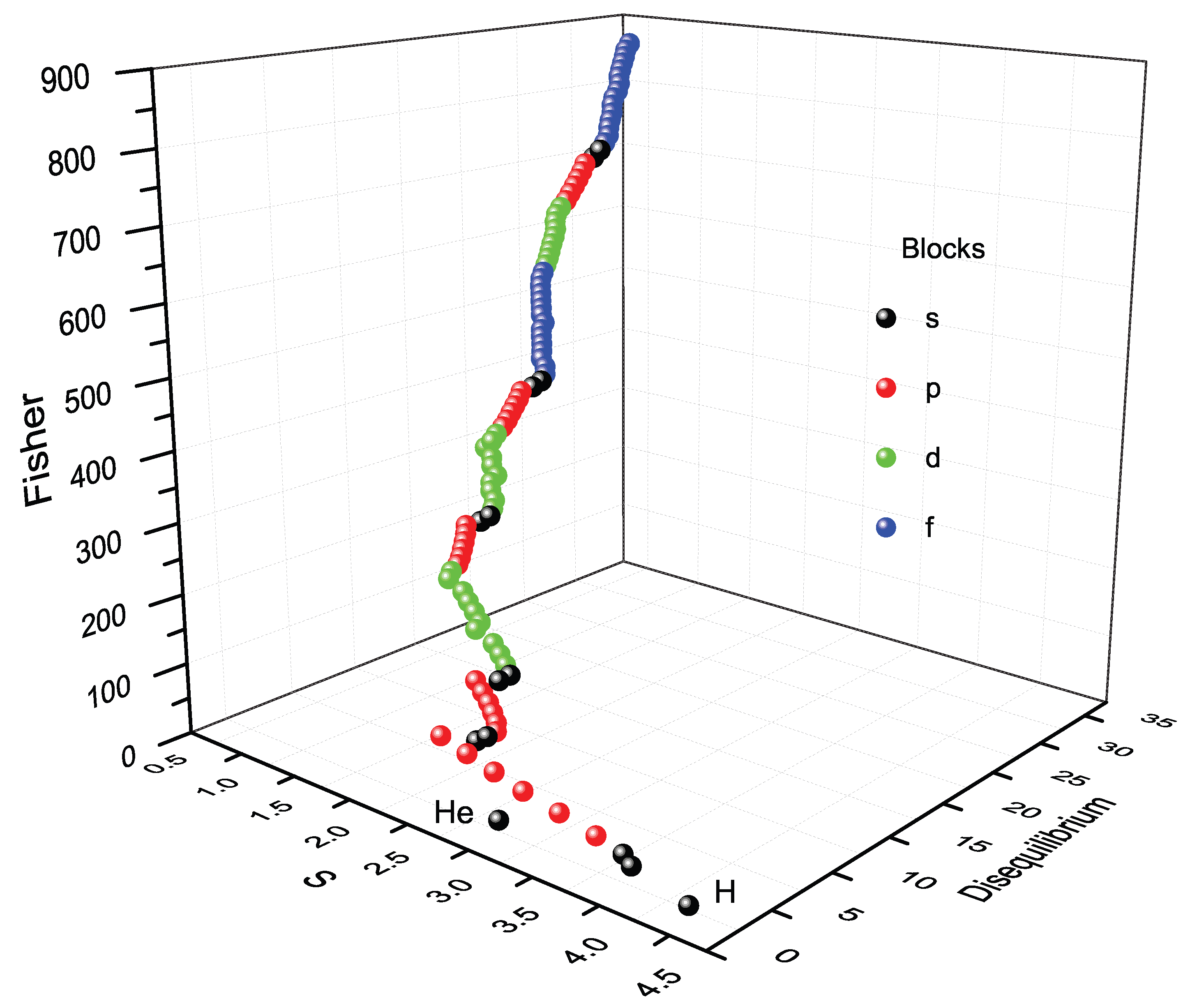
10.2. Block Representation
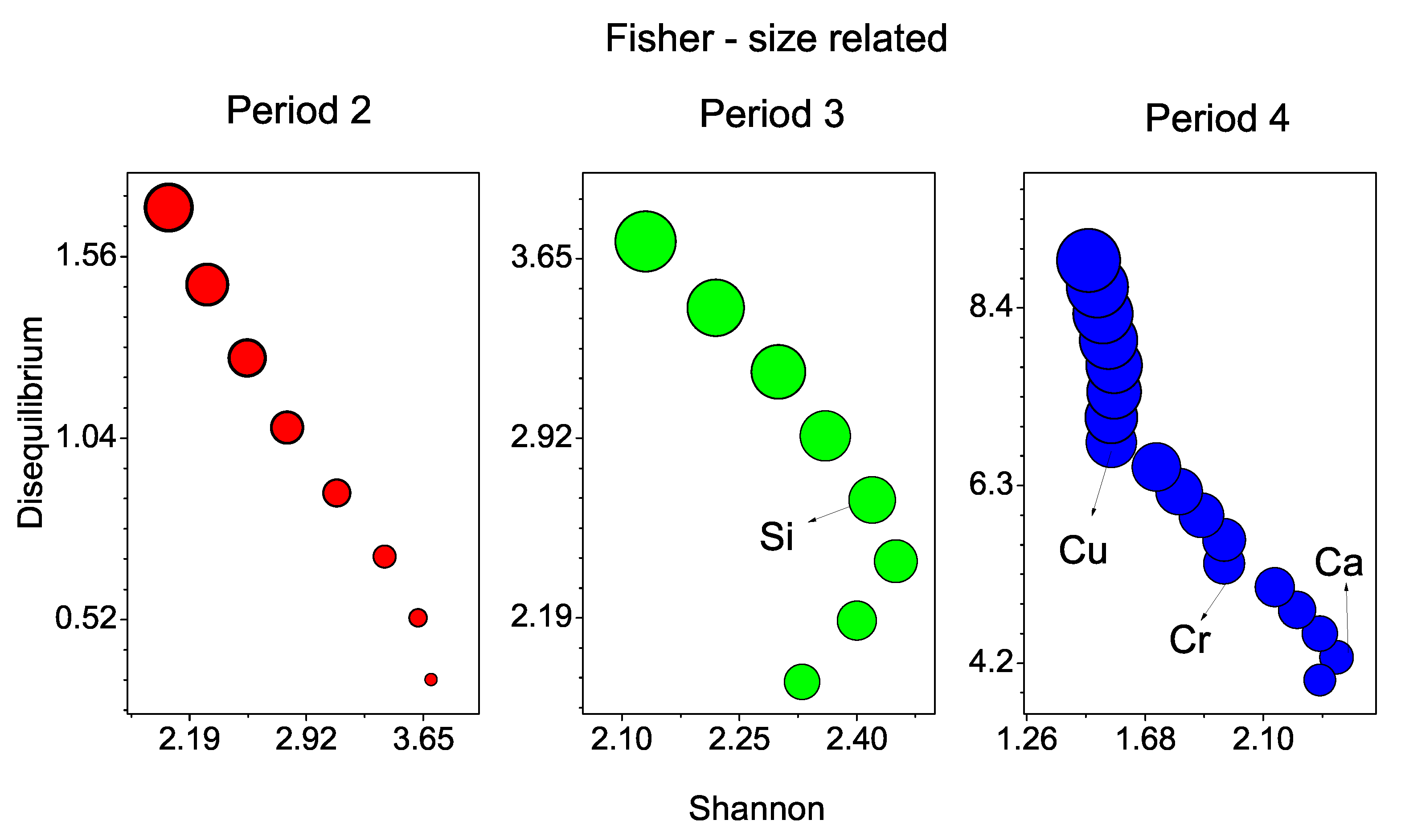
11. Chemical Anomalies in Periods 2–4
12. Momentum Space Analysis
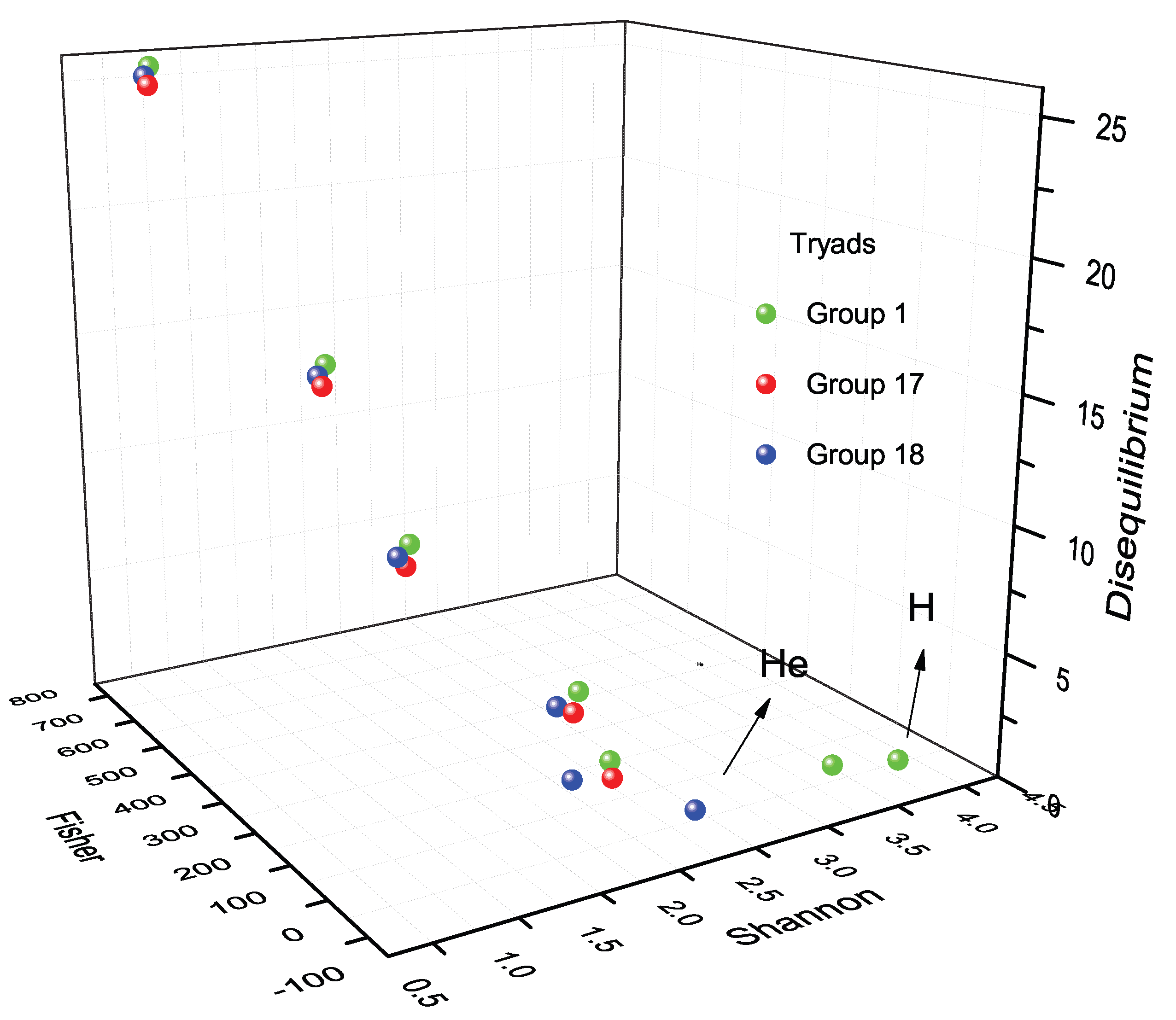
13. Triadic Analysis Around Halogens
14. Complexity Measures
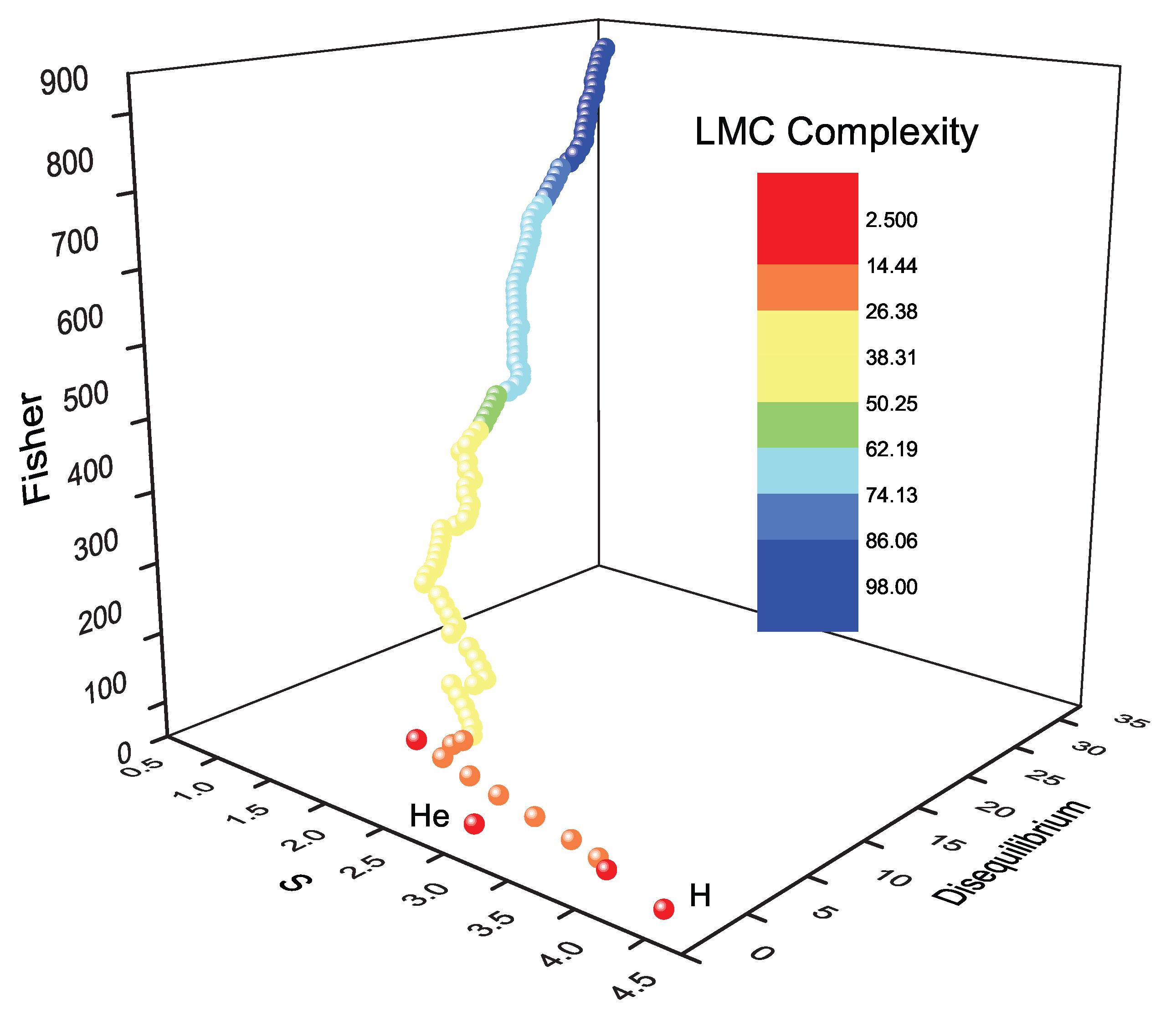
14.1. López-Mancini-Ruiz (LMC) Complexity
14.2. Fisher-Shannon (FS) Complexity
15. Statistical Correlation Validation
15.1. Methodology
15.2. Wilks’ statistics analysis
15.2.1. Random forest analysis
| Classification | Accuracy (%,r) | Accuracy (%,p) |
|---|---|---|
| Periods | 78.7 | 69.0 |
| Numeric Groups | 24.2 | 28.0 |
| Chemical Families | 49.4 | 49.6 |
16. Conclusions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mendeleev, D.I. Sootnoshenie svoistv s atomnym vesom elementov. Zhurnal Russkogo Fiziko-Khimicheskogo Obshchestva 1869, 1, 60–79. [Google Scholar]
- Mendeleev, D.I. Estestvennaya sistema elementov i primenieee k ukazaniyu svoistv neotkrytykh elementov. Zhurnal Russkogo Fiziko-Khimicheskogo Obshchestva 1871, 3, 25–56. [Google Scholar]
- Van Spronsen, J.W. The Periodic System of Chemical Elements: A History of the First Hundred Years; Elsevier: Amsterdam, 1969. [Google Scholar]
- Scerri, E.R. The Periodic Table: Its Story and Its Significance; Oxford University Press: Oxford, 2007. [Google Scholar]
- Petruševski, V.M.; Cvetković, J. On the ’True Position’ of Hydrogen in the Periodic Table. Contributions, Section of Natural, Mathematical and Biotechnical Sciences, MASA 2017, 38, 83–90. [Google Scholar] [CrossRef]
- Labarca, M.; Srivaths, A. On the Placement of Hydrogen and Helium in the Periodic System: A New Approach. Chemistry: Bulgarian Journal of Science Education 2016, 25, 514–530. [Google Scholar]
- Dash, H.H. Constant Energy Differences in Atomic Structure. Nature 1963, 198, 25–26. [Google Scholar] [CrossRef]
- Dash, H.H. Position of Hydrogen in the Periodic System of Elements. Nature 1964, 202, 1001–1003. [Google Scholar] [CrossRef]
- Sacks, L.J. Concerning the Position of Hydrogen in the Periodic Table. Foundations of Chemistry 2006, 8, 31–35. [Google Scholar] [CrossRef]
- Langmuir, I. The Arrangements of Electrons in Atoms and Molecules. Journal of the American Chemical Society 1919, 41, 868–934. [Google Scholar] [CrossRef]
- Luder, W.F. Electron Configuration as the Basis of the Periodic Table. Journal of Chemical Education 1943, 20, 21–26. [Google Scholar] [CrossRef]
- Janet, C. La classification hélicoïdale des éléments chimiques. 1928. [Google Scholar]
- Janet, C. The Helicoidal Classification of the Elements. Chemical News 1929, 138, 372–374, 388–393. [Google Scholar]
- Katz, G. The Periodic Table: An Eight-Period Table for the 21st Century. Chemical Educator 2001, 6, 324–332. [Google Scholar] [CrossRef]
- Bohr, N. The Theory of Spectra and Atomic Constitution. 1922. [Google Scholar]
- Scerri, E.R. Chemistry, Spectroscopy, and the Question of Reduction. Journal of Chemical Education 1991, 68, 122–126. [Google Scholar] [CrossRef]
- Simmons, L.M. A Modification of the Periodic Table. Journal of Chemical Education 1947, 24, 588–591. [Google Scholar] [CrossRef]
- Schwarz, W.H.E. The Full Story of Electron Configurations. Journal of Chemical Education 2010, 87, 444–448. [Google Scholar] [CrossRef]
- Sanderson, R.T. A Rational Periodic Table. Journal of Chemical Education 1964, 41, 187–189. [Google Scholar] [CrossRef]
- Allen, L.C. Electronegativity is the Average One-Electron Energy of the Valence-Shell Electrons in Ground-State Free Atoms. Journal of the American Chemical Society 1989, 111, 9003–9014. [Google Scholar] [CrossRef]
- Leach, M.R. Concerning Electronegativity as a Basic Elemental Property and Why the Periodic Table is Usually Represented in Its Medium Form. Foundations of Chemistry 2013, 15, 13–29. [Google Scholar] [CrossRef]
- Rayner-Canham, G.; Overton, T. Descriptive Inorganic Chemistry. 2010. [Google Scholar] [CrossRef]
- Scerri, E.R. The Role of Triads in the Evolution of the Periodic Table: Past and Present. Journal of Chemical Education 2008, 85, 585–589. [Google Scholar] [CrossRef]
- Scerri, E.R. Explaining the Periodic Table, and the Role of Chemical Triads. Foundations of Chemistry 2010, 12, 69–83. [Google Scholar] [CrossRef]
- Bent, H.A. New Ideas in Chemistry from Fresh Energy for the Periodic Law. 2006. [Google Scholar]
- Firsching, F.H. Anomalies in the Periodic Table. Journal of Chemical Education 1981, 58, 478–479. [Google Scholar] [CrossRef]
- Mendeleev, D.I. Die periodische Gesetzmässigkeit der chemischen Elemente. Annalen der Chemie und Pharmacie 1870, Supplementband 8, 133–229. [Google Scholar]
- Stewart, P.J. A Century on from Dmitrii Mendeleev: Tables and Spirals, Noble Gases and Nobel Prizes. Foundations of Chemistry 2007, 9, 235–245. [Google Scholar] [CrossRef]
- Kaesz, H.; Atkins, P.W. A Central Position for Hydrogen in the Periodic Table. Chemistry International 2003, 25, 14. [Google Scholar] [CrossRef]
- Rodríguez Peña, M.; García Guerra, J.A. The Periodic Spiral of Elements. Foundations of Chemistry 2024, 26, 315–321. [Google Scholar] [CrossRef]
- Imyanitov, N.S. Spiral as the Fundamental Graphic Representation of the Periodic Law. Blocks of Elements as the Autonomic Parts of the Periodic System. Foundations of Chemistry 2016, 18, 153–173. [Google Scholar] [CrossRef]
- Scherer, J.A. Models of a Spiral Periodic Chart. Journal of Chemical Education 1949, 26, 113–117. [Google Scholar] [CrossRef]
- Stewart, P.J. Charles Janet: Unrecognized Genius of the Periodic System. Foundations of Chemistry 2010, 12, 5–15. [Google Scholar] [CrossRef]
- Janet, C. Concordance de l’arrangement quantique de base des électrons planétaires des atomes avec la classification scalariforme, hélicoïdale des elements chimiques. 1930. [Google Scholar]
- Tsimmerman, V. ADOMAH Periodic Table. 2013. [Google Scholar]
- Kurushkin, M. Viatscheslaw Romanoff: Unknown Genius of the Periodic System. Pure and Applied Chemistry 2019, 91, 1921–1928. [Google Scholar] [CrossRef]
- Petruševski, V.M.; Cvetković, J. On the ’True Position’ of Hydrogen in the Periodic Table. Foundations of Chemistry 2018, 20, 251–260. [Google Scholar] [CrossRef]
- Laing, M. Where to Put Hydrogen in a Periodic Table? Foundations of Chemistry 2007, 9, 127–137. [Google Scholar] [CrossRef]
- Rich, R.L. Are Some Elements More Equal Than Others? Journal of Chemical Education 2005, 82, 1761–1763. [Google Scholar] [CrossRef]
- Scerri, E.R. Mendeleev’s Periodic Table is Finally Completed and What to Do About Group 3. Chemistry International 2012, 34, 28–31. [Google Scholar] [CrossRef]
- Esquivel, R.O.; Angulo, J.C.; Antolín, J.; Dehesa, J.S.; López-Rosa, S.; Flores-Gallegos, N. Analysis of complexity measures and information planes of selected molecules in position and momentum spaces. Physical Chemistry Chemical Physics 2010, 12, 7108. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, R.O.; López-Rosa, S.; Molina-Espíritu, M.; Angulo, J.C.; Dehesa, J.S. Information-theoretic space from simple atomic and molecular systems to biological and pharmacological molecules. Theoretical Chemistry Accounts 2016, 135, 253. [Google Scholar] [CrossRef]
- Esquivel, R.O.; Flores-Gallegos, N.; Iuga, C.; Carrera, E.M.; Angulo, J.C.; Antolín, J. Phenomenological description of the transition state, and the bond breaking and bond forming processes of selected elementary chemical reactions: an information-theoretic study. Theoretical Chemistry Accounts 2009, 124, 445–460. [Google Scholar] [CrossRef]
- López-Rosa, S.; Esquivel, R.O.; Angulo, J.C.; Antolín, J.; Dehesa, J.S.; Flores-Gallegos, N. Fisher Information Study in Position and Momentum Spaces for Elementary Chemical Reactions. Journal of Chemical Theory and Computation 2010, 6, 145–154. [Google Scholar] [CrossRef]
- Vázquez-Hernández, H.; Esquivel, R.O. Phenomenological description of the acidity of the citric acid and its deprotonated species: informational-theoretical study. Journal of Molecular Modeling 2023, 29, 253. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, R.O.; Molina-Espíritu, M.; López-Rosa, S.; Soriano-Correa, C.; Barrientos-Salcedo, C.; Kohout, M.; Dehesa, J.S. Predominant Information Quality Scheme for the Essential Amino Acids: An Information-Theoretical Analysis. ChemPhysChem 2015, 16, 2571–2581. [Google Scholar] [CrossRef]
- Demirtaş, K.; Erman, B.; Haliloğlu, T. Dynamic correlations: exact and approximate methods for mutual information. Bioinformatics 2024, 40, btae076. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Wang, X.; Wu, Z.; Li, C. Study of the Allosteric Mechanism of Human Mitochondrial Phenylalanyl-tRNA Synthetase by Transfer Entropy via an Improved Gaussian Network Model and Co-evolution Analyses. The Journal of Physical Chemistry Letters 2023, 14, 3452–3460. [Google Scholar] [CrossRef] [PubMed]
- Hong, Q.J.; Liu, Z.K. Generalized approach for rapid entropy calculation of liquids and solids. Physical Review Research 2025, 7, L012030. [Google Scholar] [CrossRef]
- Giulini, M.; Menichetti, R.; Shell, M.S.; Potestio, R. An Information-Theory-Based Approach for Optimal Model Reduction of Biomolecules. Journal of Chemical Theory and Computation 2020, 16, 6795–6813. [Google Scholar] [CrossRef]
- Schwalbe-Koda, D.; Hamel, S.; Sadigh, B.; Zhou, F.; Lordi, V. Model-free estimation of completeness, uncertainties, and outliers in atomistic machine learning using information theory. Nature Communications 2025, 16, 4014. [Google Scholar] [CrossRef]
- Angulo, J.C.; Antolín, J.; Esquivel, R.O. Atomic and molecular complexities: Their physical and chemical interpretations. In Statistical Complexities: Applications in electronic structures; Sen, K.D., Ed.; Springer: Berlin, 2010. [Google Scholar]
- Rawlings, D.C.; Davidson, E.R. Molecular electron density distributions in position and momentum space. The Journal of Physical Chemistry 1985, 89, 969–974. [Google Scholar] [CrossRef]
- Kaijser, P.; Smith, V.H. Evaluation of Momentum Distributions and Compton Profiles for Atomic and Molecular Systems. In Advances in Quantum Chemistry; Elsevier, 1977; Vol. 10, pp. 37–76. [Google Scholar] [CrossRef]
- Baerends, E.J.; Aguirre, N.F.; Austin, N.D.; Autschbach, J.; Bickelhaupt, F.M.; Bulo, R.; Cappelli, C.; Van Duin, A.C.T.; Egidi, F.; Fonseca Guerra, C.; et al. The Amsterdam Modeling Suite. The Journal of Chemical Physics 2025, 162, 162501. [Google Scholar] [CrossRef]
- Shannon, C.E. A Mathematical Theory of Communication. Bell System Technical Journal 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Fisher, R.A. Theory of Statistical Estimation. Mathematical Proceedings of the Cambridge Philosophical Society 1925, 22, 700–725. [Google Scholar] [CrossRef]
- Frieden, B.R. Science from Fisher information: a unification; Cambridge University Press: Cambridge, 2004. [Google Scholar]
- Carbo, R.; Leyda, L.; Arnau, M. How similar is a molecule to another? An electron density measure of similarity between two molecular structures. International Journal of Quantum Chemistry 1980, 17, 1185–1189. [Google Scholar] [CrossRef]
- Onicescu, O. Théorie de l’information. In Énergie informationnelle. C. R. Acad. Sci., Paris, Sér. A; 1966; Volume 263, pp. 841–842. [Google Scholar]
- López-Ruiz, R.; Mancini, H.; Calbet, X. A statistical measure of complexity. Physics Letters A 1995, 209, 321–326. [Google Scholar] [CrossRef]
- Anteneodo, C.; Plastino, A. Some features of the López-Ruiz-Mancini-Calbet (LMC) statistical measure of complexity. Physics Letters A 1996, 223, 348–354. [Google Scholar] [CrossRef]
- Romera, E.; Dehesa, J.S. The Fisher–Shannon information plane, an electron correlation tool. The Journal of Chemical Physics 2004, 120, 8906–8912. [Google Scholar] [CrossRef] [PubMed]
- Statistical Complexity: Applications in Electronic Structure; Sen, K., Ed.; Springer Netherlands: Dordrecht, 2011. [Google Scholar] [CrossRef]
- Yamano, T. A statistical complexity measure with nonextensive entropy and quasi-multiplicativity. Journal of Mathematical Physics 2004, 45, 1974–1987. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). Biometrika 1965, 52, 591. [Google Scholar] [CrossRef]
- Filliben, J.J. The Probability Plot Correlation Coefficient Test for Normality. Technometrics 1975, 17, 111–117. [Google Scholar] [CrossRef]
- Welch, B.L. The Generalization of `Student’s’ Problem when Several Different Population Variances are Involved. Biometrika 1947, 34, 28. [Google Scholar] [CrossRef]
- Satterthwaite, F.E. An Approximate Distribution of Estimates of Variance Components. Biometrics Bulletin 1946, 2, 110. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: fundamental algorithms for scientific computing in Python. Nature Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Wilks, S.S. Certain generalizations in the analysis of variance. Biometrika 1932, 24, 471–494. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Machine learning 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Seabold, S.; Perktold, J. statsmodels: Econometric and statistical modeling with python. In Proceedings of the 9th Python in Science Conference, 2010. [Google Scholar]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. Journal of Machine Learning Research 2011, 12, 2825–2830. [Google Scholar]
| Classification | ||
|---|---|---|
| Periods | 0.0732 | 0.0452 |
| Numeric Groups | 0.844 | 0.795 |
| Chemical Families | 0.953 | 0.639 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




