Submitted:
14 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. MRI Data Acquisition
- 3D T2-FLAIR: TR = 5000 ms, TE = 390 ms, TI = 1800 ms;
- 3D T1-weighted: TR = 16 ms, TE = 4.76 ms;
- 3D T2-weighted: TR=3000 ms, TE=335 ms;
- MT-weighted: TR = 20 ms, echo time (TE) = 4.76 ms, flip angle (FA) = 8°, scan time 5 min 40 s;
- T1-weighted: TR =16 ms, TE = 4.76 ms, FA =18°, scan time 4 min 32 s;
- Proton-density-weighted: TR= 16 ms, TE = 4.76 ms, FA= 3°, scan time 4 min 32 s.
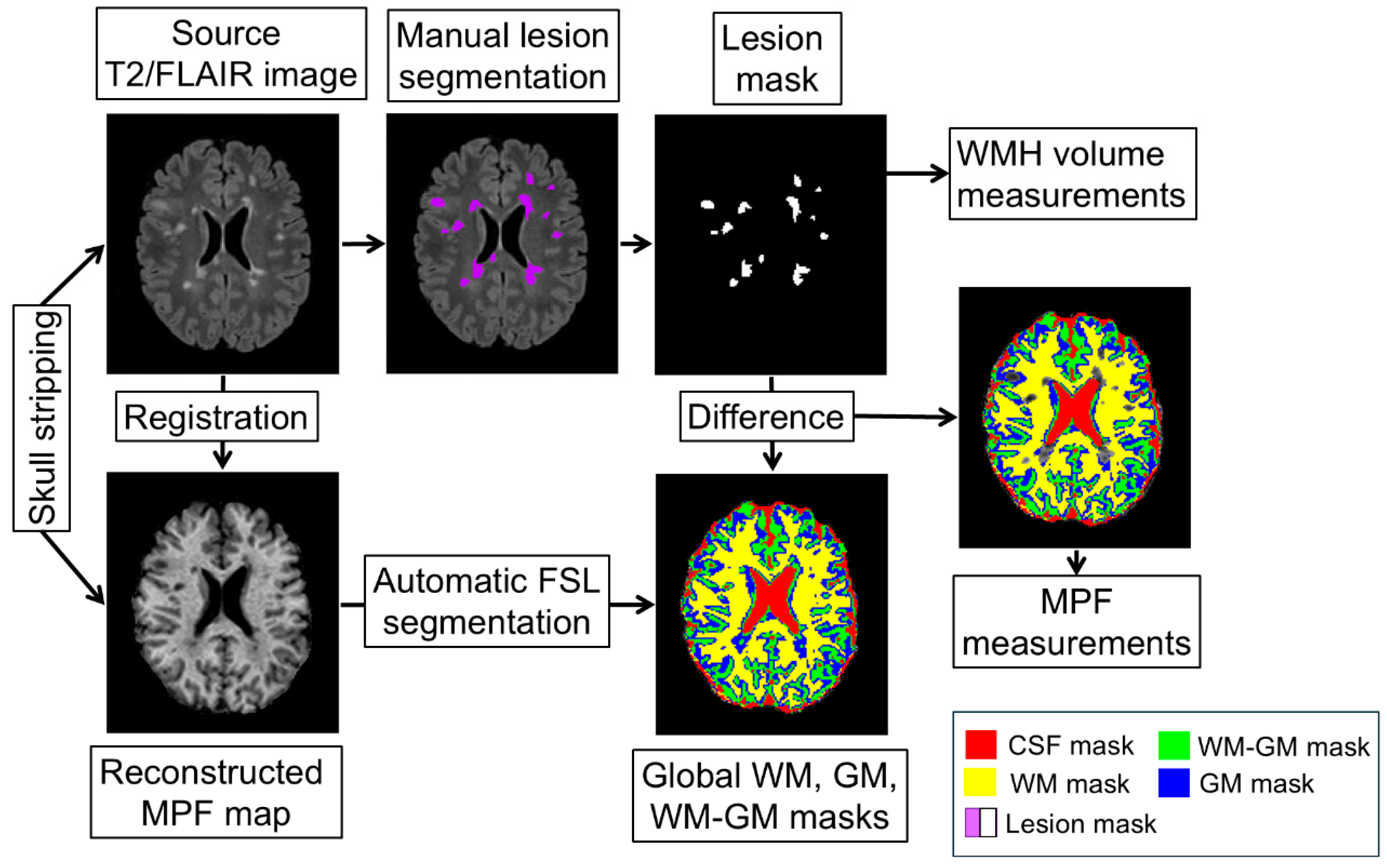
2.3. Image Processing
- 1)
- Skull stripping was performed using a mask, which was obtained by applying the BET algorithm to the PD-weighted images in the MRIcro application [48]. The mask was converted to a binary image and applied to the MPF maps to remove extracerebral tissue.
- 2)
- WMH were outlined manually by one operator blinded to the subject information on T2/FLAIR images with the guidance of T2-weighted and T1-weighted images.
- 3)
- T2/FLAIR images were registered to MPF maps using ITK-snap software to measure WMH volumes and mean MPF values in the outlined areas.
- 4)
- 5)
- To obtain MPF measurements of normal-appearing white and grey matter outside of focal lesions, WMH masks were subtracted from global compartment masks. The resulting masks were used to measure average MPF values for global compartments. The measurements were carried out using ITK-SNAP application [50].
2.4. Statistical Analysis
3. Results
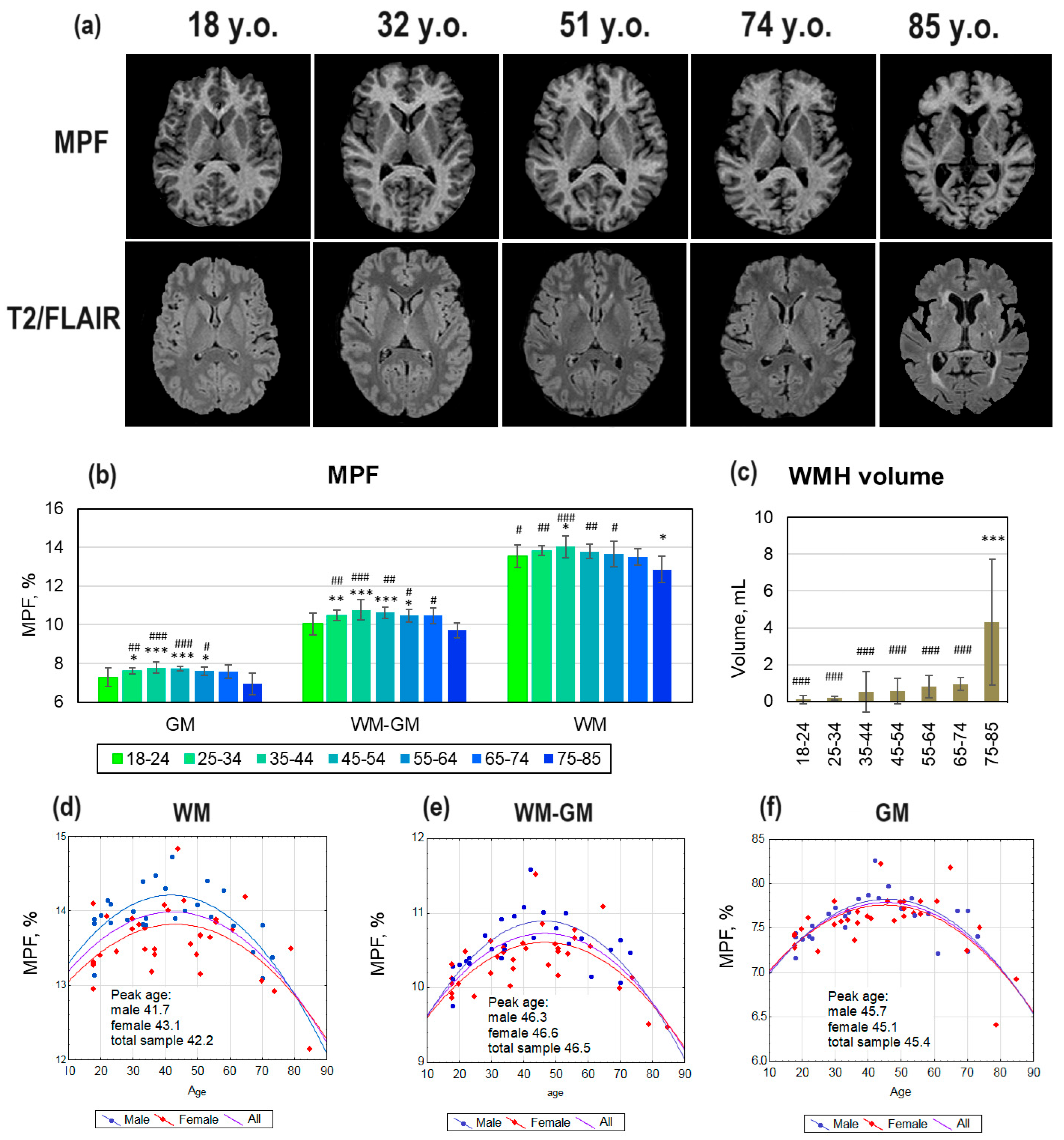
3.1. Age-Related Global Changes in the Brain Myelination
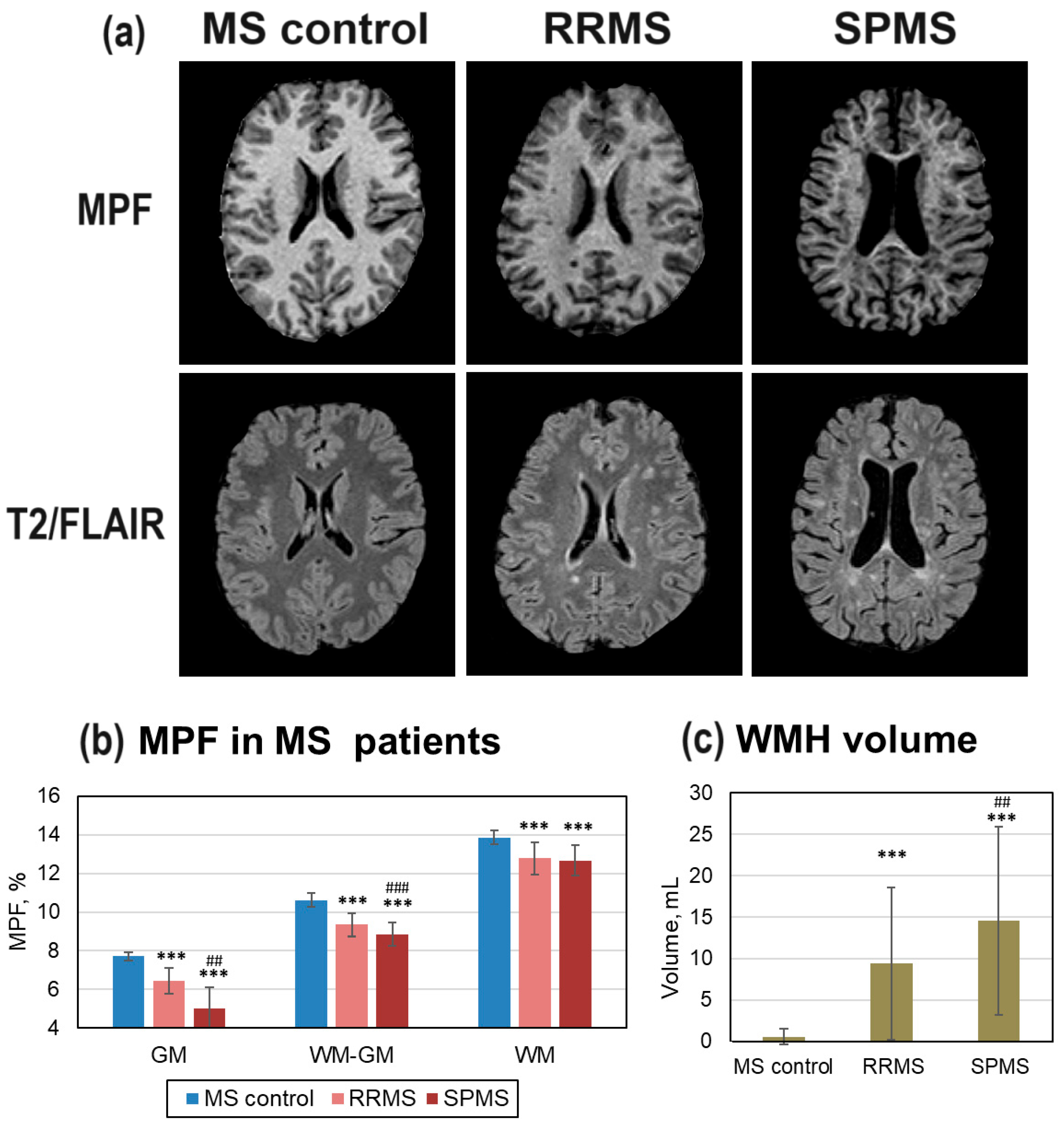
3.2. Diffuse and Focal Demyelination in MS Patients
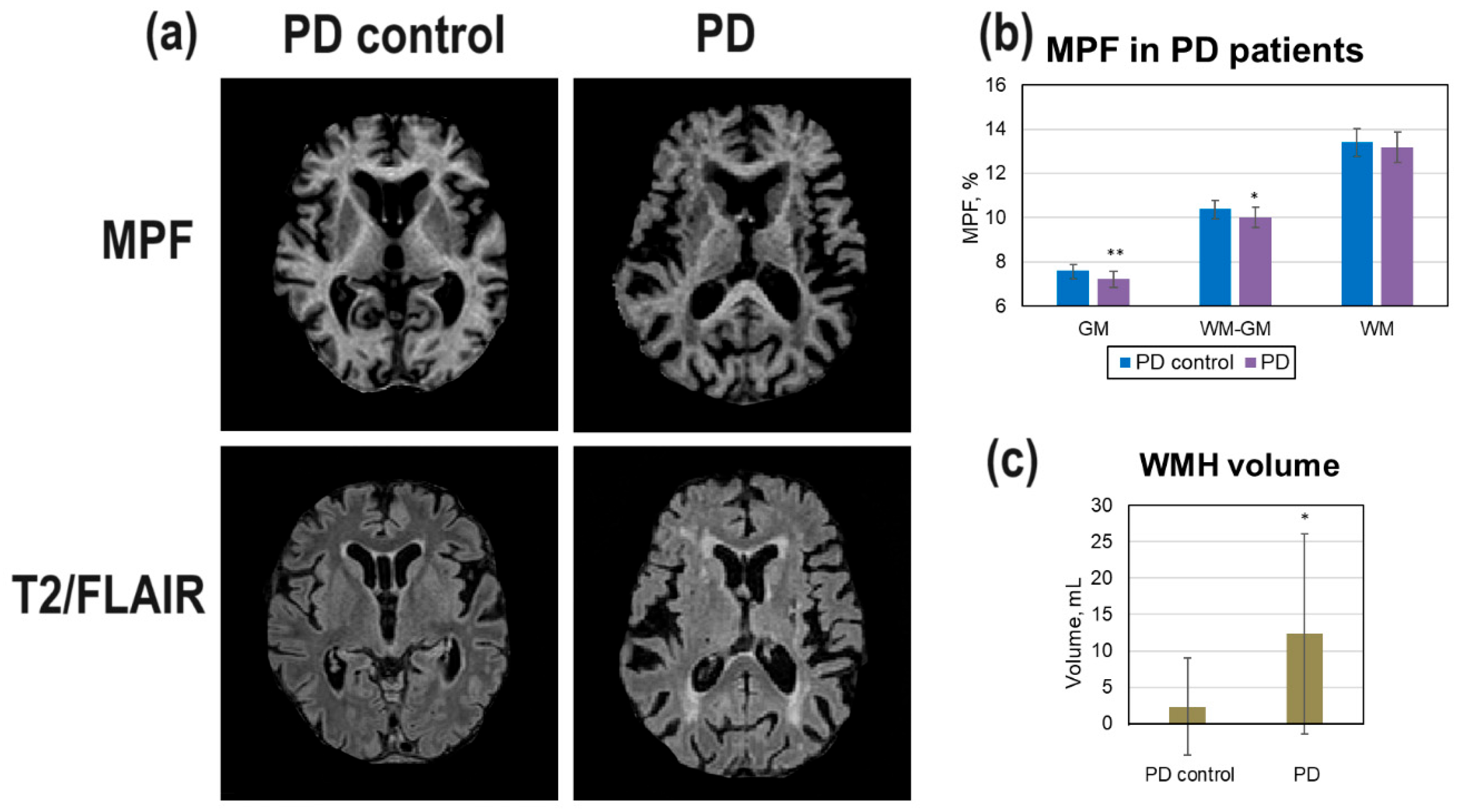
3.3. Diffuse and Focal Demyelination in PD Patients
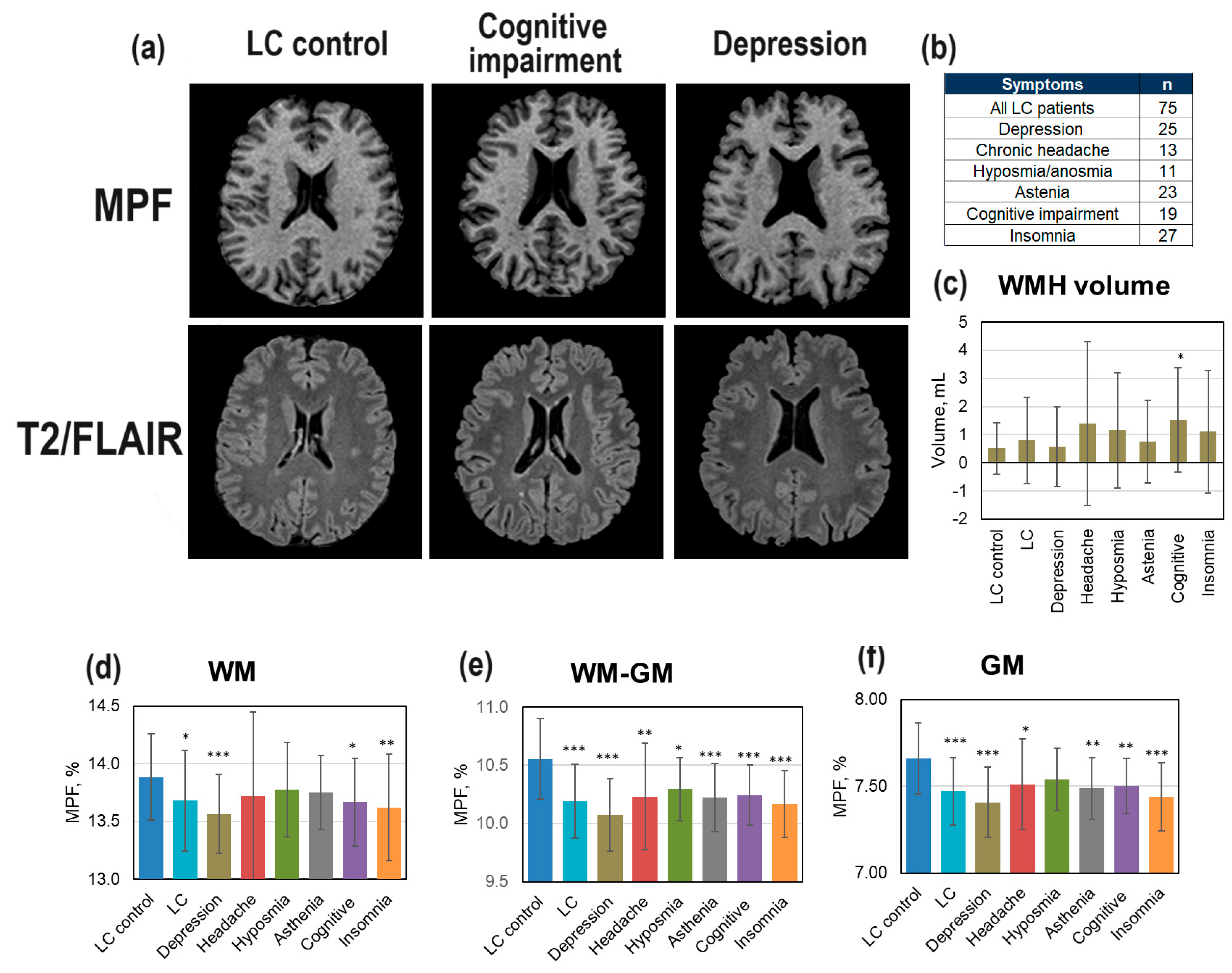
3.4. Demyelination in LC Patients
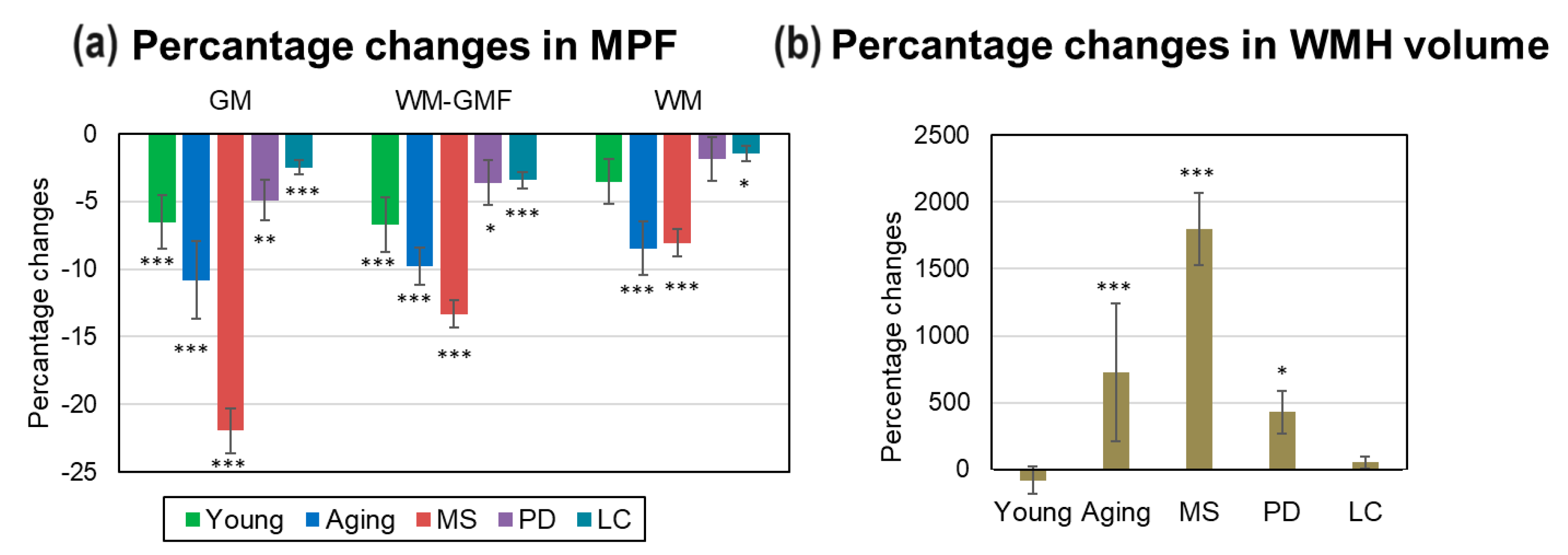
3.5. Comparison of Global Demyelinationand WMH Volume in MS, PD, LC Patients, and Normal Aging
4. Discussion
5. Conclusions
6. Study Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pivneva, T.A. Mechanisms Underlying the Process of Demyelination in Multiple Sclerosis. Neurophysiology 2009, 41, 365–373. [Google Scholar] [CrossRef]
- Bradl, M.; Lassmann, H. Oligodendrocytes: Biology and Pathology. Acta Neuropathol. 2010, 119, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Lassmann, H. Multiple Sclerosis Pathology. Cold Spring Harb. Perspect. Med. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Bakirtzis, C.; Lima, M.; De Lorenzo, S.S.; Artemiadis, A.; Theotokis, P.; Kesidou, E.; Konstantinidou, N.; Sintila, S.A.; Boziki, M.K.; Parissis, D.; et al. Secondary Central Nervous System Demyelinating Disorders in the Elderly: A Narrative Review. Healthc. 2023, 11, 1–19. [Google Scholar] [CrossRef]
- Yang, K.; Wu, Z.; Long, J.; Li, W.; Wang, X.; Hu, N.; Zhao, X.; Sun, T. White Matter Changes in Parkinson’s Disease. npj Park. Dis. 2023, 9, 1–10. [Google Scholar] [CrossRef]
- Faulkner, M.E.; Gong, Z.; Guo, A.; Laporte, J.P.; Bae, J.; Bouhrara, M. Harnessing Myelin Water Fraction as an Imaging Biomarker of Human Cerebral Aging, Neurodegenerative Diseases, and Risk Factors Influencing Myelination: A Review. J. Neurochem. 2024, 168, 2243–2263. [Google Scholar] [CrossRef]
- Bergamino, M.; Keeling, E.G.; Mishra, V.R.; Stokes, A.M.; Walsh, R.R. Assessing White Matter Pathology in Early-Stage Parkinson Disease Using Diffusion Mri: A Systematic Review. Front. Neurol. 2020, 11, 1–21. [Google Scholar] [CrossRef]
- Zeis, T.; Graumann, U.; Reynolds, R.; Schaeren-Wiemers, N. Normal-Appearing White Matter in Multiple Sclerosis Is in a Subtle Balance between Inflammation and Neuroprotection. Brain 2008, 131, 288–303. [Google Scholar] [CrossRef]
- Gallego-Delgado, P.; James, R.; Browne, E.; Meng, J.; Umashankar, S.; Tan, L.; Picon, C.; Mazarakis, N.D.; Faisal, A.A.; Howell, O.W.; et al. Neuroinflammation in the Normal-Appearing White Matter (NAWM) of the Multiple Sclerosis Brain Causes Abnormalities at the Nodes of Ranvier; 2020; Vol. 18, ISBN 1111111111. [Google Scholar]
- Groh, J.; Simons, M. White Matter Aging and Its Impact on Brain Function. Neuron 2025, 113, 127–139. [Google Scholar] [CrossRef]
- Chapman, T.W.; Hill, R.A. Myelin Plasticity in Adulthood and Aging. Neurosci. Lett. 2020, 715, 134645. [Google Scholar] [CrossRef]
- Braga, J.; Lepra, M.; Kish, S.J.; Rusjan, P.M.; Nasser, Z.; Verhoeff, N.; Vasdev, N.; Bagby, M.; Boileau, I.; Husain, M.I.; et al. Neuroinflammation after COVID-19 with Persistent Depressive and Cognitive Symptoms. JAMA Psychiatry 2023, 80, 787–795. [Google Scholar] [CrossRef]
- Sriwastava, S.; Tandon, M.; Podury, S.; Prasad, A.; Wen, S.; Guthrie, G.; Kakara, M.; Jaiswal, S.; Subedi, R.; Elkhooly, M.; et al. COVID-19 and Neuroinflammation: A Literature Review of Relevant Neuroimaging and CSF Markers in Central Nervous System Inflammatory Disorders from SARS-COV2. J. Neurol. 2021, 268, 4448–4478. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.; Kamaeva, D.A.; Naumova, A.V. Role of Demyelination in the Persistence of Neurological and Mental Impairments after COVID-19. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef]
- Linortner, P.; McDaniel, C.; Shahid, M.; Levine, T.F.; Tian, L.; Cholerton, B.; Poston, K.L. White Matter Hyperintensities Related to Parkinson’s Disease Executive Function. Mov. Disord. Clin. Pract. 2020, 7, 629–638. [Google Scholar] [CrossRef]
- Fang, E.; Fartaria, M.J.; Ann, C.N.; Maréchal, B.; Kober, T.; Lim, J.X.; Ooi, L.Q.R.; Chen, C.; Lim, S.L.; Tan, E.K.; et al. Clinical Correlates of White Matter Lesions in Parkinson’s Disease Using Automated Multi-Modal Segmentation Measures. J. Neurol. Sci. 2021, 427. [Google Scholar] [CrossRef]
- Rovaris, M.; Bozzali, M.; Iannucci, G.; Ghezzi, A.; Caputo, D.; Montanari, E.; Bertolotto, A.; Bergamaschi, R.; Capra, R.; Mancardi, G.L.; et al. Assessment of Normal-Appearing White and Gray Matter in Patients with Primary Progressive Multiple Sclerosis: A Diffusion-Tensor Magnetic Resonance Imaging Study. Arch. Neurol. 2002, 59, 1406–1412. [Google Scholar] [CrossRef]
- De Groot, M.; Verhaaren, B.F.J.; De Boer, R.; Klein, S.; Hofman, A.; Van Der Lugt, A.; Ikram, M.A.; Niessen, W.J.; Vernooij, M.W. Changes in Normal-Appearing White Matter Precede Development of White Matter Lesions. Stroke 2013, 44, 1037–1042. [Google Scholar] [CrossRef]
- Vavasour, I.M.; Huijskens, S.C.; Li, D.K.; Traboulsee, A.L.; Mädler, B.; Kolind, S.H.; Rauscher, A.; Moore, G.W.; MacKay, A.L.; Laule, C. Global Loss of Myelin Water over 5 Years in Multiple Sclerosis Normal-Appearing White Matter. Mult. Scler. J. 2017, 24, 1557–1568. [Google Scholar] [CrossRef]
- Rahmanzadeh, R.; Lu, P.J.; Barakovic, M.; Weigel, M.; Maggi, P.; Nguyen, T.D.; Schiavi, S.; Daducci, A.; La Rosa, F.; Schaedelin, S.; et al. Myelin and Axon Pathology in Multiple Sclerosis Assessed by Myelin Water and Multi-Shell Diffusion Imaging. Brain 2021, 144, 1684–1696. [Google Scholar] [CrossRef]
- Muñoz González, G.; ’t Hart, B.A.; Bugiani, M.; Plemel, J.R.; Schenk, G.J.; Kooij, G.; Luchicchi, A. A Focus on the Normal-Appearing White and Gray Matter within the Multiple Sclerosis Brain: A Link to Smoldering Progression. Acta Neuropathol. 2025, 150, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Yarnykh, V.L.L.; Bowen, J.D.D.; Henson, L.K.J.J.; Samsonov, A.; Repovic, P.; Mayadev, A.; Qian, P.; Gangadharan, B.; Keogh, B.P.P.; Maravilla, K.R.R.; et al. Fast Whole-Brain Three-Dimensional Macromolecular Proton Fraction Mapping in Multiple Sclerosis. Radiology 2014, 274, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Luo, C.; Li, Q.; Hu, N.; Xiao, Y.; Liu, N.; Lui, S.; Gong, Q. White Matter Abnormalities in Patients With Parkinson’s Disease: A Meta-Analysis of Diffusion Tensor Imaging Using Tract-Based Spatial Statistics. Front. Aging Neurosci. 2021, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gattellaro, G.; Minati, L.; Grisoli, M.; Mariani, C.; Carella, F.; Osio, M.; Ciceri, E.; Albanese, A.; Bruzzone, M.G. White Matter Involvement in Idiopathic Parkinson Disease: A Diffusion Tensor Imaging Study. Am. J. Neuroradiol. 2009, 30, 1222–1226. [Google Scholar] [CrossRef]
- Smirnova, L.P.; Yarnykh, V.L.; Parshukova, D.A.; Kornetova, E.G.; Semke, A.V.; Usova, A.V.; Pishchelko, A.O.; Khodanovich, M.Y.; Ivanova, S.A. Global Hypomyelination of the Brain White and Gray Matter in Schizophrenia: Quantitative Imaging Using Macromolecular Proton Fraction. Transl. Psychiatry 2021, 11, 365. [Google Scholar] [CrossRef]
- Khodanovich, M.; Svetlik, M.; Kamaeva, D.; Usova, A.; Kudabaeva, M.; Anan’ina, T.; Vasserlauf, I.; Pashkevich, V.; Moshkina, M.; Obukhovskaya, V.; et al. Demyelination in Patients with POST-COVID Depression. Biomedicines 2024, 13, 1–23. [Google Scholar] [CrossRef]
- Faizy, T.D.; Thaler, C.; Broocks, G.; Flottmann, F.; Leischner, H.; Kniep, H.; Nawabi, J.; Schön, G.; Stellmann, J.-P.; Kemmling, A.; et al. The Myelin Water Fraction Serves as a Marker for Age-Related Myelin Alterations in the Cerebral White Matter – A Multiparametric MRI Aging Study. Front. Neurosci. 2020, 14, 1–14. [Google Scholar] [CrossRef]
- Faizy, T.D.; Kumar, D.; Broocks, G.; Thaler, C.; Flottmann, F.; Leischner, H.; Kutzner, D.; Hewera, S.; Dotzauer, D.; Reddy, R.; et al. Age-Related Measurements of the Myelin Water Fraction Derived from 3D Multi-Echo GRASE Reflect Myelin Content of the Cerebral White Matter. Sci. Rep. 2018, 1–8. [Google Scholar] [CrossRef]
- James, S.N.; Manning, E.N.; Storey, M.; Nicholas, J.M.; Coath, W.; Keuss, S.E.; Cash, D.M.; Lane, C.A.; Parker, T.; Keshavan, A.; et al. Neuroimaging, Clinical and Life Course Correlates of Normal-Appearing White Matter Integrity in 70-Year-Olds. Brain Commun. 2023, 5, 1–14. [Google Scholar] [CrossRef]
- Khodanovich, M.; Svetlik, M.; Naumova, A.; Kamaeva, D.; Usova, A.; Kudabaeva, M.; Anan’ina, T.; Wasserlauf, I.; Pashkevich, V.; Moshkina, M.; et al. Age-Related Decline in Brain Myelination: Quantitative Macromolecular Proton Fraction Mapping, T2-FLAIR Hyperintensity Volume, and Anti-Myelin Antibodies Seven Years Apart. Biomedicines 2024, 12. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.; Svetlik, M.V.; Naumova, A.V.; Usova, A.V.; Pashkevich, V.Y.; Moshkina, M.V.; Shadrina, M.M.; Kamaeva, D.A.; Obukhovskaya, V.B.; Kataeva, N.G.; et al. Global and Regional Sex-Related Di Ff Erences, Asymmetry, and Peak Age of Brain Myelination in Healthy Adults. J. Clin. Med. 2024, 13, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Yarnykh, V.L.; Krutenkova, E.P.; Aitmagambetova, G.; Henson, L.K.J.; Piedmont, H.; Repovic, P.; Mayadev, A.; Qian, P.; Gangadharan, B. Iron-Insensitive Quantitative Assessment of Subcortical Gray Matter Demyelination in Multiple Sclerosis Using Macromolecular Proton Fraction. Am. J. Neuroradiol. 2018, 39, 618–625. [Google Scholar] [CrossRef] [PubMed]
- Kisel, A.A.; Naumova, A.V.; Yarnykh, V.L. Macromolecular Proton Fraction as a Myelin Biomarker: Principles, Validation, and Applications. Front. Neurosci. 2022, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Seiler, S.; Ropele, S.; Schmidt, R. Magnetization Transfer Imaging for in Vivo Detection of Microstructural Tissue Changes in Aging and Dementia: A Short Literature Review. J. Alzheimer’s Dis. 2014, 42, 229–237. [Google Scholar] [CrossRef]
- Underhill, H.R.; Yuan, C.; Yarnykh, V.L. Direct Quantitative Comparison between Cross-Relaxation Imaging and Diffusion Tensor Imaging of the Human Brain at 3.0 T. Neuroimage 2009, 47, 1568–1578. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.Y.; Sorokina, I.V.V.; Glazacheva, V.Y.Y.; Akulov, A.E.E.; Nemirovich-Danchenko, N.M.M.; Romashchenko, A.V. V.; Tolstikova, T.G.G.; Mustafina, L.R.R.; Yarnykh, V.L.L. Histological Validation of Fast Macromolecular Proton Fraction Mapping as a Quantitative Myelin Imaging Method in the Cuprizone Demyelination Model. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.; Pishchelko, A.O.; Glazacheva, V.Y.; Pan, E.S.; Akulov, A.E.; Svetlik, M.V.; Tyumentseva, Y.A.; Anan’ina, T.V.; Yarnykh, V.L. Quantitative Imaging of White and Gray Matter Remyelination in the Cuprizone Demyelination Model Using the Macromolecular Proton Fraction. Cells 2019, 8, 1204. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.; Kisel, A.A.; Akulov, A.E.; Atochin, D.N.; Kudabaeva, M.S.; Glazacheva, V.Y.; Svetlik, M.V.; Medvednikova, Y.A.; Mustafina, L.R.; Yarnykh, V.L. Quantitative Assessment of Demyelination in Ischemic Stroke in Vivo Using Macromolecular Proton Fraction Mapping. J. Cereb. Blood Flow Metab. 2018, 38, 919–931. [Google Scholar] [CrossRef]
- Khodanovich, M.Y.; Gubskiy, I.L.; Kudabaeva, M.S.; Namestnikova, D.D.; Kisel, A.A.; Anan’ina, T.V.; Tumentceva, Y.A.; Mustafina, L.R.; Yarnykh, V.L. Long-Term Monitoring of Chronic Demyelination and Remyelination in a Rat Ischemic Stroke Model Using Macromolecular Proton Fraction Mapping. J. Cereb. Blood Flow Metab. 2021, 41, 2856–2869. [Google Scholar] [CrossRef]
- Drobyshevsky, A.; Synowiec, S.; Goussakov, I.; Lu, J.; Gascoigne, D.; Aksenov, D.P.; Yarnykh, V. NeuroImage Temporal Trajectories of Normal Myelination and Axonal Development Assessed by Quantitative Macromolecular and Diffusion MRI: Ultrastructural and Immunochemical Validation in a Rabbit Model. Neuroimage 2023, 270, 119974. [Google Scholar] [CrossRef]
- Naumova, A.V.; Akulov, A.E.; Khodanovich, M.Y.; Yarnykh, V.L. High-Resolution Three-Dimensional Macromolecular Proton Fraction Mapping for Quantitative Neuroanatomical Imaging of the Rodent Brain in Ultra-High Magnetic Fields. Neuroimage 2017, 147, 985–993. [Google Scholar] [CrossRef]
- Yarnykh, V.L.; Kisel, A.A.; Khodanovich, M.Y. Scan–Rescan Repeatability and Impact of B0 and B1 Field Nonuniformity Corrections in Single-Point Whole-Brain Macromolecular Proton Fraction Mapping. J. Magn. Reson. Imaging 2020, 51, 1789–1798. [Google Scholar] [CrossRef] [PubMed]
- Yarnykh, V.L. Time-Efficient, High-Resolution, Whole Brain Three-Dimensional Macromolecular Proton Fraction Mapping. Magn. Reson. Med. 2016, 75, 2100–2106. [Google Scholar] [CrossRef] [PubMed]
- Yarnykh, V.L.L.; Prihod’ko, I.Y.Y.; Savelov, A.A.A.; Korostyshevskaya, A.M.M. Quantitative Assessment of Normal Fetal Brain Myelination Using Fast Macromolecular Proton Fraction Mapping. Am. J. Neuroradiol. 2018, 39, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Petrie, E.C.; Cross, D.J.; Yarnykh, V.L.; Richards, T.; Martin, N.M.; Pagulayan, K.; Hoff, D.; Hart, K.; Mayer, C.; Tarabochia, M.; et al. Neuroimaging, Behavioral, and Psychological Sequelae of Repetitive Combined Blast/Impact Mild Traumatic Brain Injury in Iraq and Afghanistan War Veterans. J. Neurotrauma 2014, 31, 425–436. [Google Scholar] [CrossRef]
- Huber, E.; Corrigan, N.M.; Yarnykh, V.L.; Ramírez, N.F.; Kuhl, P.K. Language Experience during Infancy Predicts White Matter Myelination at Age 2 Years. J. Neurosci. 2023, 43, 1590–1599. [Google Scholar] [CrossRef]
- Corrigan, N.M.; Yarnykh, V.L.; Hippe, D.S.; Owen, J.P.; Huber, E.; Zhao, T.C.; Kuhl, P.K. Myelin Development in Cerebral Gray and White Matter during Adolescence and Late Childhood. Neuroimage 2021, 227, 117678. [Google Scholar] [CrossRef]
- Rorden, C.; Brett, M. Stereotaxic Display of Brain Lesions. Behav. Neurol. 2000, 12, 191–200. [Google Scholar] [CrossRef]
- Jenkinson, M.; Beckmann, C.F.; Behrens, T.E.J.; Woolrich, M.W.; Smith, S.M. FSL. Neuroimage 2012, 62, 782–790. [Google Scholar] [CrossRef]
- Yushkevich, P.A.; Piven, J.; Hazlett, C.; Smith, G.; Ho, S.; Gee, J.C.; Gerig, G. User-Guided 3D Active Contour Segmentation of Anatomical Structures: Significantly Improved Efficiency and Reliability. Neuroimage 2006, 31, 1116–1128. [Google Scholar] [CrossRef]
- de Schipper, L.J.; Hafkemeijer, A.; Bouts, M.J.R.J.; van der Grond, J.; Marinus, J.; Henselmans, J.M.L.; van Hilten, J.J. Age- and Disease-Related Cerebral White Matter Changes in Patients with Parkinson’s Disease. Neurobiol. Aging 2019, 80, 203–209. [Google Scholar] [CrossRef]
- Rektor, I.; Svátková, A.; Vojtíšek, L.; Zikmundová, I.; Vaníček, J.; Király, A.; Szabó, N. White Matter Alterations in Parkinson’s Disease with Normal Cognition Precede Grey Matter Atrophy. PLoS One 2018, 13, 1–15. [Google Scholar] [CrossRef]
- Mohsen, M.; Mohamed, N.A.E.; Bedir, A.E.T.M.; Razek, A.A.K.A.; Saied, A.E.A.M. Advanced MRI-Based Evaluation of Gray and White Matter Changes in Parkinson’s Disease. Egypt. J. Radiol. Nucl. Med. 2024, 55. [Google Scholar] [CrossRef]
- Zhang, J.; Tao, R.; Liu, C.; Wu, W.; Zhang, Y.; Cui, J.; Wang, J. Possible Effects of Iron Deposition on the Measurement of DTI Metrics in Deep Gray Matter Nuclei: An in Vitro and in Vivo Study. Neurosci. Lett. 2013, 551, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Khodanovich, M.; Naumova, A.; Kamaeva, D.; Obukhovskaya, V.; Vasilieva, S.; Schastnyy, E.; Kataeva, N.; Levina, A.; Kudabaeva, M.; Pashkevich, V.; et al. Neurocognitive Changes in Patients with Post-COVID Depression. J. Clin. Med. 2024, 13, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Nelson, B.K.; Farah, L.N.; Saint, S.A.; Song, C.; Field, T.S.; Sossi, V.; Stoessl, A.J.; Wellington, C.; Honer, W.G.; Lang, D.; et al. Diffusion Tensor Imaging after COVID-19 Infection: A Systematic Review. Neuroimage 2025, 310, 121150. [Google Scholar] [CrossRef]
- Yuan, M.; Lu, R.; Liu, Y.; Zhu, H.; Wang, H.; Wang, J.; Song, Y.; Yang, L.; Xiao, M. White Matter Changes in Recovered COVID-19 Patients: Insights from DTI, DKI, and NODDI Metrics. Front. Neurol. 2025, 16, 1–11. [Google Scholar] [CrossRef]
- Cui, Y.; Wang, Y.; Lu, L.; Zhang, Y.; Yu, Q.; Dong, S.; Wang, Y.; Liu, Y.; Dong, G.; Dai, J.; et al. Long-Term White Matter Changes in Patients Who Develop Severe Depression after Multiple COVID-19 Infections: A 3–6-Month Study. Quant. Imaging Med. Surg. 2025, 15, 4364–4374. [Google Scholar] [CrossRef]
- Huang, S.; Zhou, X.; Zhao, W.; Du, Y.; Yang, D.; Huang, Y.; Chen, Y.; Zhang, H.; Yang, G.; Liu, J.; et al. Dynamic White Matter Changes in Recovered COVID-19 Patients: A Two-Year Follow-up Study. Theranostics 2023, 13, 724–735. [Google Scholar] [CrossRef]
- Huang, S.; Zhou, Z.; Yang, D.; Zhao, W.; Zeng, M.; Xie, X.; Du, Y.; Jiang, Y.; Zhou, X.; Yang, W.; et al. Persistent White Matter Changes in Recovered COVID-19 Patients at the 1-Year Follow-Up. Brain 2022, 145, 1830–1838. [Google Scholar] [CrossRef]
- Qin, Y.; Wu, J.; Chen, T.; Li, J.; Zhang, G.; Wu, D.; Zhou, Y.; Zheng, N.; Cai, A.; Ning, Q.; et al. Long-Term Microstructure and Cerebral Blood Flow Changes in Patients Recovered from COVID-19 without Neurological Manifestations. J. Clin. Invest. 2021, 131. [Google Scholar] [CrossRef]
- Bispo, D.D. de C.; Brandão, P.R. de P.; Pereira, D.A.; Maluf, F.B.; Dias, B.A.; Paranhos, H.R.; von Glehn, F.; de Oliveira, A.C.P.; Regattieri, N.A.T.; Silva, L.S.; et al. Brain Microstructural Changes and Fatigue after COVID-19. Front. Neurol. 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Nelson, B.K.; Farah, L.N.; Grier, A.; Su, W.; Chen, J.; Sossi, V.; Sekhon, M.S.; Stoessl, A.J.; Wellington, C.; Honer, W.G.; et al. Differences in Brain Structure and Cognitive Performance between Patients with Long-COVID and Those with Normal Recovery. Neuroimage 2024, 300, 120859. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, F.; Palladini, M.; Paolini, M.; Melloni, E.; Vai, B.; De Lorenzo, R.; Furlan, R.; Rovere-Querini, P.; Falini, A.; Mazza, M.G. Brain Correlates of Depression, Post-Traumatic Distress, and Inflammatory Biomarkers in COVID-19 Survivors: A Multimodal Magnetic Resonance Imaging Study. Brain, Behav. Immun. - Heal. 2021, 18, 100387. [Google Scholar] [CrossRef]
- Qin, H.; Duan, G.; Zhou, K.; Qin, L.; Lai, Y.; Liu, Y.; Lu, Y.; Peng, B.; Zhang, Y.; Zhou, X.; et al. Alteration of White Matter Microstructure in Patients with Sleep Disorders after COVID-19 Infection. Sleep Med. 2024, 114, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Hsu, J.L.; Leemans, A.; Bai, C.H.; Lee, C.H.; Tsai, Y.F.; Chiu, H.C.; Chen, W.H. Gender Differences and Age-Related White Matter Changes of the Human Brain: A Diffusion Tensor Imaging Study. Neuroimage 2008, 39, 566–577. [Google Scholar] [CrossRef]
- Cercignani, M.; Giulietti, G.; Dowell, N.G.; Gabel, M.; Broad, R.; Leigh, P.N.; Harrison, N.A.; Bozzali, M. Characterizing Axonal Myelination within the Healthy Population: A Tract-by-Tract Mapping of Effects of Age and Gender on the Fiber g-Ratio. Neurobiol. Aging 2017, 49, 109–118. [Google Scholar] [CrossRef]
- Kitamura, S.; Morikawa, M.; Kiuchi, K.; Taoka, T.; Fukusumi, M.; Kichikawa, K.; Kishimoto, T. Asymmetry, Sex Differences and Age-Related Changes in the White Matter in the Healthy Elderly: A Tract-Based Study. BMC Res. Notes 2011, 4. [Google Scholar] [CrossRef]
- Salat, D.H.; Tuch, D.S.; Greve, D.N.; Kouwe, A.J.W.; Van Der; Hevelone, N.D.; Zaleta, A.K.; Rosen, B.R.; Fischl, B.; Corkin, S.; Rosas, H.D.; et al. Age-Related Alterations in White Matter Microstructure Measured by Diffusion Tensor Imaging. Neurobiol. Aging 2005, 26, 1215–1227. [Google Scholar] [CrossRef]
- Brenner, E.K.; Bangen, K.J.; Clark, A.L.; Delano-Wood, L.; Evangelista, N.D.; Edwards, L.; Sorg, S.F.; Jak, A.J.; Bondi, M.W.; Deoni, S.C.L.; et al. Sex Moderates the Association between Age and Myelin Water Fraction in the Cingulum and Fornix among Older Adults without Dementia. Front. Aging Neurosci. 2023, 15, 1–7. [Google Scholar] [CrossRef]
- Inano, S.; Takao, H.; Hayashi, N.; Abe, O.; Ohtomo, K. Effects of Age and Gender on White Matter Integrity. Am. J. Neuroradiol. 2011, 32, 2103–2109. [Google Scholar] [CrossRef]
- Lebel, C.; Caverhill-Godkewitsch, S.; Beaulieu, C. Age-Related Regional Variations of the Corpus Callosum Identified by Diffusion Tensor Tractography. Neuroimage 2010, 52, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; Treit, S.; Beaulieu, C. A Review of Diffusion MRI of Typical White Matter Development from Early Childhood to Young Adulthood. NMR Biomed. 2019, 32. [Google Scholar] [CrossRef] [PubMed]
- Westlye, L.T.; Walhovd, K.B.; Dale, A.M.; Bjørnerud, A.; Due-tønnessen, P.; Engvig, A.; Tamnes, C.K.; Østby, Y. Life-Span Changes of the Human Brain White Matter: Diffusion Tensor Imaging ( DTI ) and Volumetry. Cereb. Cortex 2010, 20, 2055–2068. [Google Scholar] [CrossRef] [PubMed]
- Arshad, M.; Stanley, J.A.; Raz, N. Adult Age Differences in Subcortical Myelin Content Are Consistent with Protracted Myelination and Unrelated to Diffusion Tensor Imaging Indices. Neuroimage 2016, 143, 26–39. [Google Scholar] [CrossRef]
- Kochunov, P.; Williamson, D.E.; Lancaster, J.; Fox, P.; Cornell, J.; Blangero, J.; Glahn, D.C. Fractional Anisotropy of Water Diffusion in Cerebral White Matter across the Lifespan. Neurobiol. Aging 2012, 33, 9–20. [Google Scholar] [CrossRef]
- O’Sullivan, M.; Jones, D.K.; Summers, P.E.; Morris, R.G.; Williams, S.C.R.; Markus, H.S. Evidence for Cortical “Disconnection” as a Mechanism of Age-Related Cognitive Decline. Neurology 2001, 57, 632–638. [Google Scholar] [CrossRef]
- Bartzokis, G.; Lu, P.H.; Tingus, K.; Mendez, M.F.; Richard, A.; Peters, D.G.; Oluwadara, B.; Barrall, K.A.; Finn, J.P.; Villablanca, P.; et al. Lifespan Trajectory of Myelin Integrity and Maximum Motor Speed. Neurobiol. Aging 2010, 31, 1554–1562. [Google Scholar] [CrossRef]
- Kohama, S.G.; Rosene, D.L.; Sherman, L.S. Age-Related Changes in Human and Non-Human Primate White Matter: From Myelination Disturbances to Cognitive Decline. Age (Omaha). 2012, 34, 1093–1110. [Google Scholar] [CrossRef]
- Armstrong, C.L.; Traipe, E.; Hunter, J.V.; Haselgrove, J.C.; Ledakis, G.E.; Tallent, E.M.; van Buchem, M.; Shera, D. Age-Related, Regional, Hemispheric, and Medial-Lateral Differences in Myelin Integrity in Vivo in the Normal Adult Brain. Am. J. Neuroradiol. 2021, 25, 977–984. [Google Scholar] [CrossRef]
- Garnier-Crussard, A.; Bougacha, S.; Wirth, M.; André, C.; Delarue, M.; Landeau, B.; Mézenge, F.; Kuhn, E.; Gonneaud, J.; Chocat, A.; et al. White Matter Hyperintensities across the Adult Lifespan: Relation to Age, Aβ Load, and Cognition. Alzheimer’s Res. Ther. 2020, 12, 1–11. [Google Scholar] [CrossRef]
- Bartzokis, G. Age-Related Myelin Breakdown: A Developmental Model of Cognitive Decline and Alzheimer’s Disease. Neurobiol. Aging 2004, 25, 5–18. [Google Scholar] [CrossRef]
| Parameter | MS | PD | LC | Control |
|---|---|---|---|---|
| Sample size | 42 | 16 | 75 | 63 |
| Male (%)/Female (%) | 9(21)/33(79) | 5(31)/11(69) | 25(17)/50(83) | 27(43)/36(57) |
| Age, years±SD | 39.0±9.5 | 69.7±9.1 | 40.9±9.9 | 41.7±18.9 |
| Age, median (min-max) | 39(25-67) | 70(51-85) | 42(19-61) | 40(18-85) |
| Disease duration, years±SD | 9.0±5.7 | 9.8±3.9 | 1.8±0.82 | - |
| Disease severity, parameters (%) |
RR/SP (71/29) |
stage1 1/2/3/4 (6/19/50/25) | mild/moderate/severe/critical3 (75/11/11/3) |
- |
| Disease | Sample | n | Male, n (%) | Female, n (%) | Age, years±SD | Age, median (min-max) |
| MS | patients | 42 | 9 (21) | 33(79) | 39.0±9.5 | 39 (25 - 67) |
| control | 36 | 12 (33) | 24 (67) | 41.1±10.4 | 40 (22 – 67) | |
| PD | patients | 16 | 5(31) | 11(69) | 69.7±9.1 | 70 (51 – 85) |
| control | 17 | 6 (35) | 11 (65) | 65.3±9.7 | 67 (51 – 85) | |
| LC | patients | 75 | 25 (33) | 50 (67) | 40.9±9.9 | 42 (19 – 61) |
| control | 42 | 17 (40) | 25 (60) | 39.9±12.0 | 40 (18 – 61) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




