Submitted:
11 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
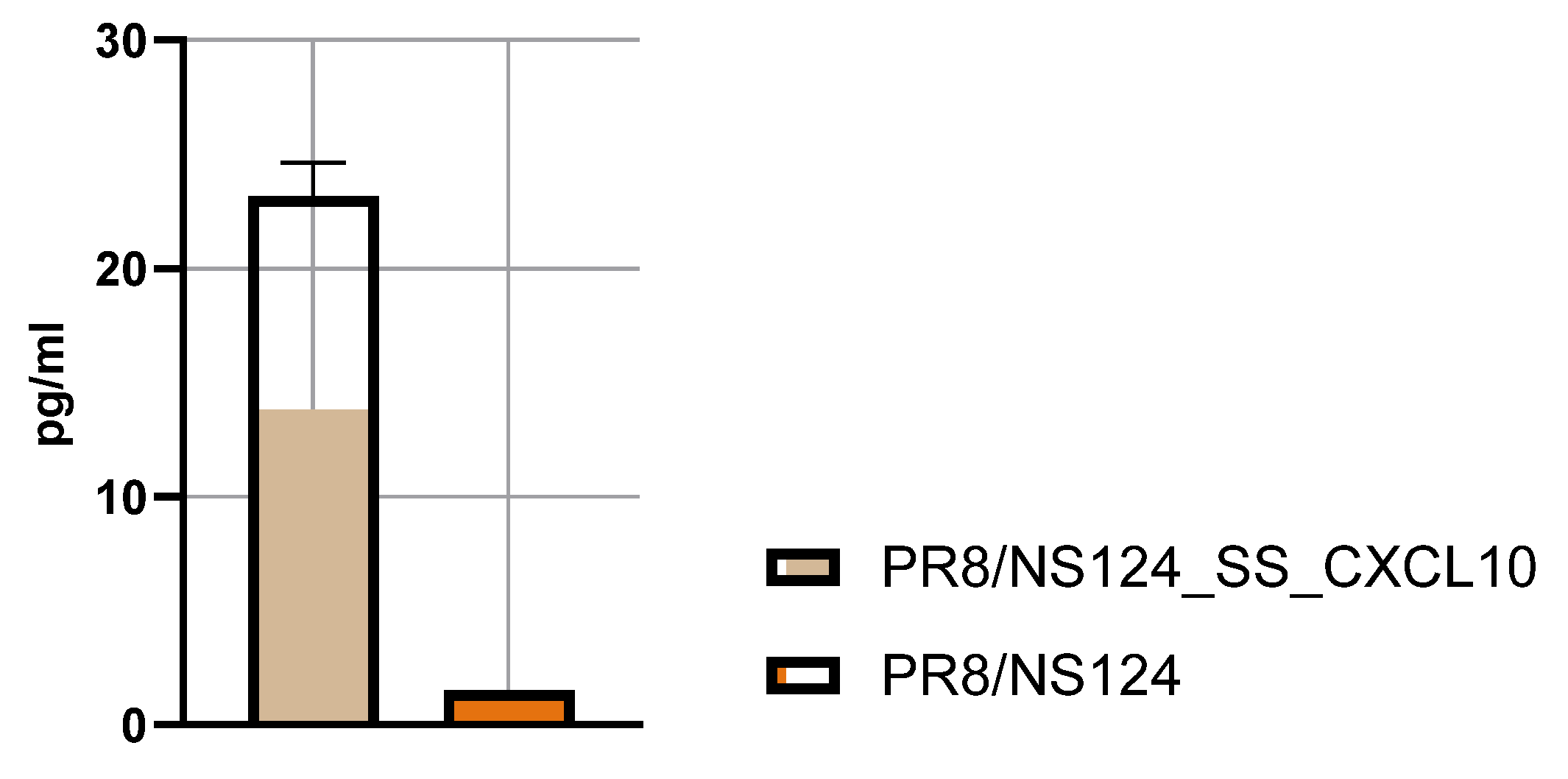
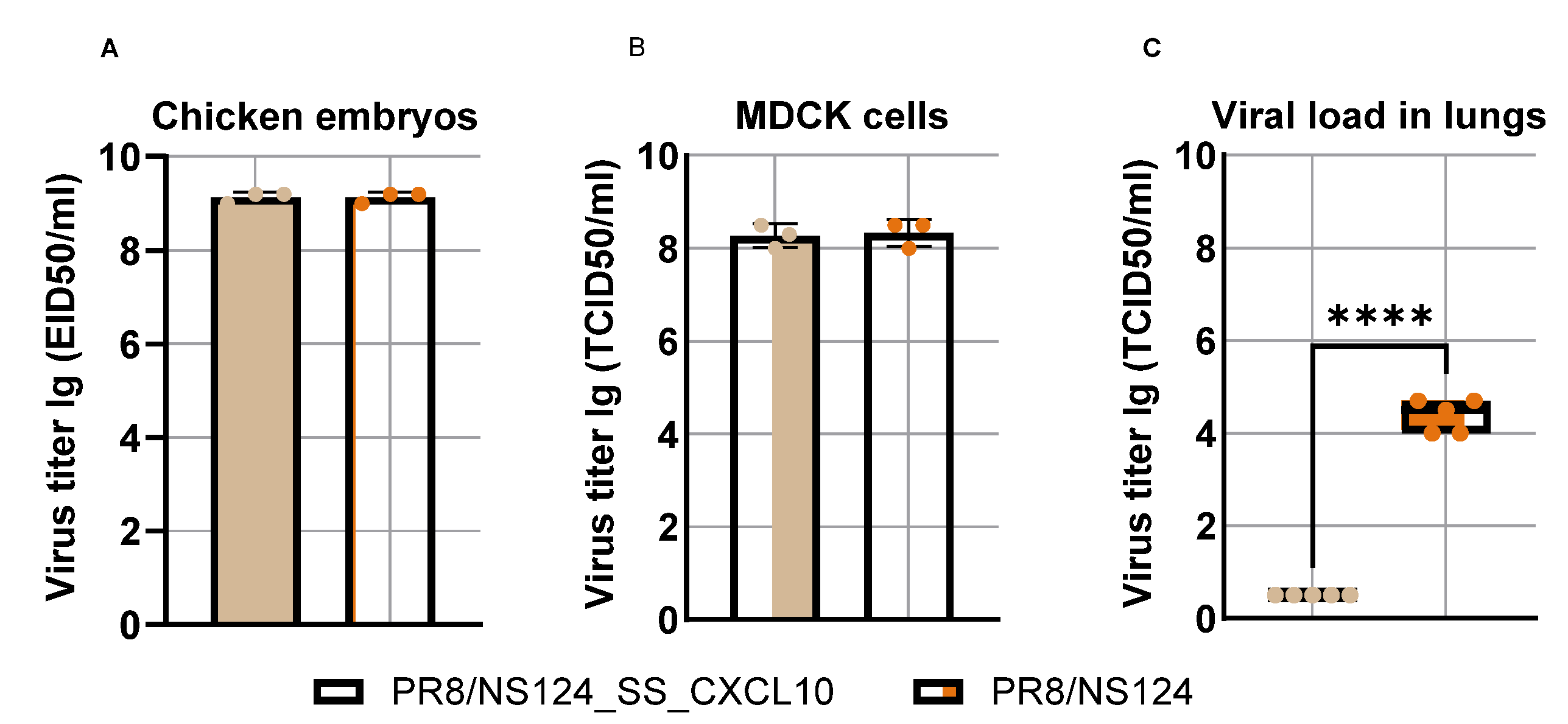
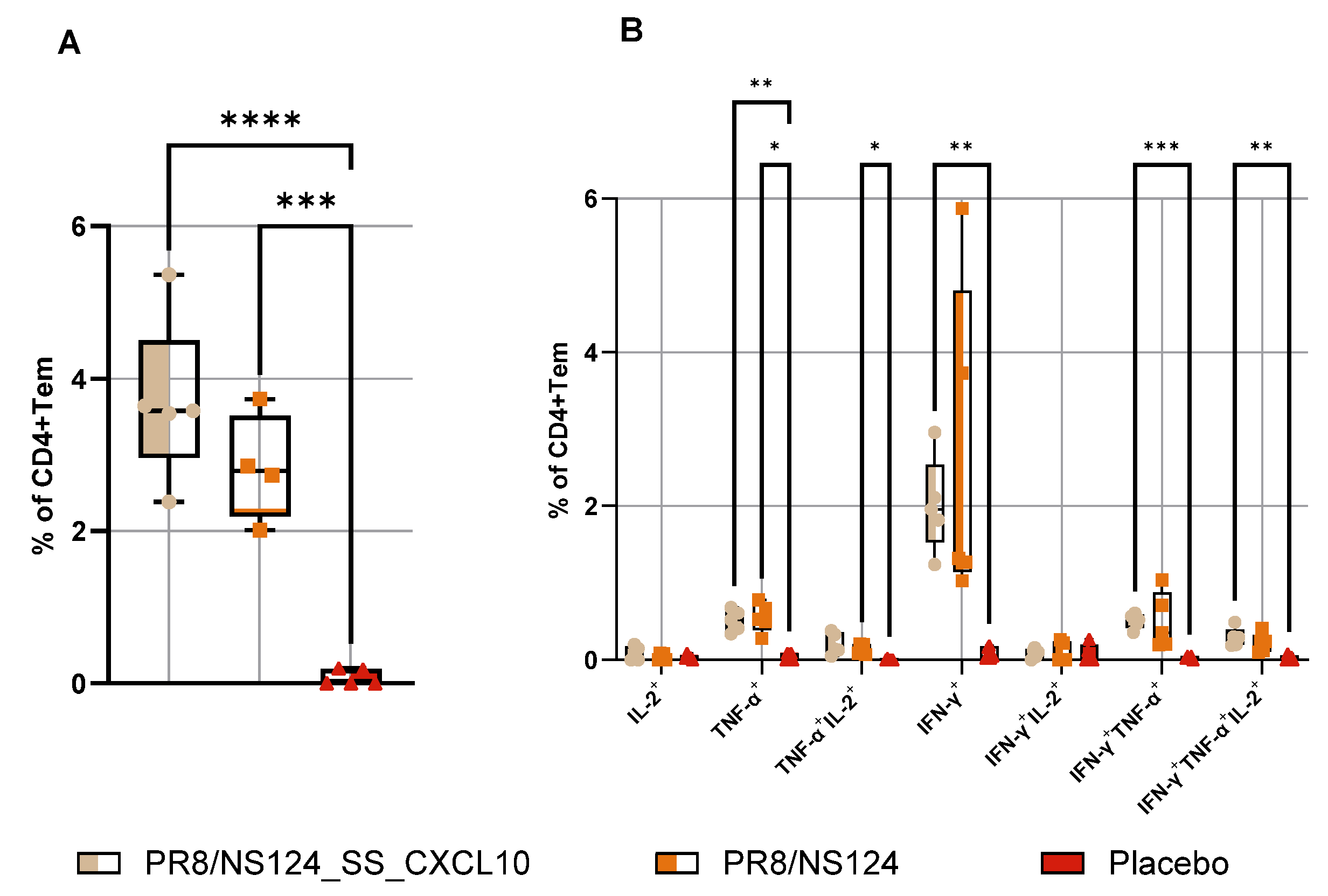
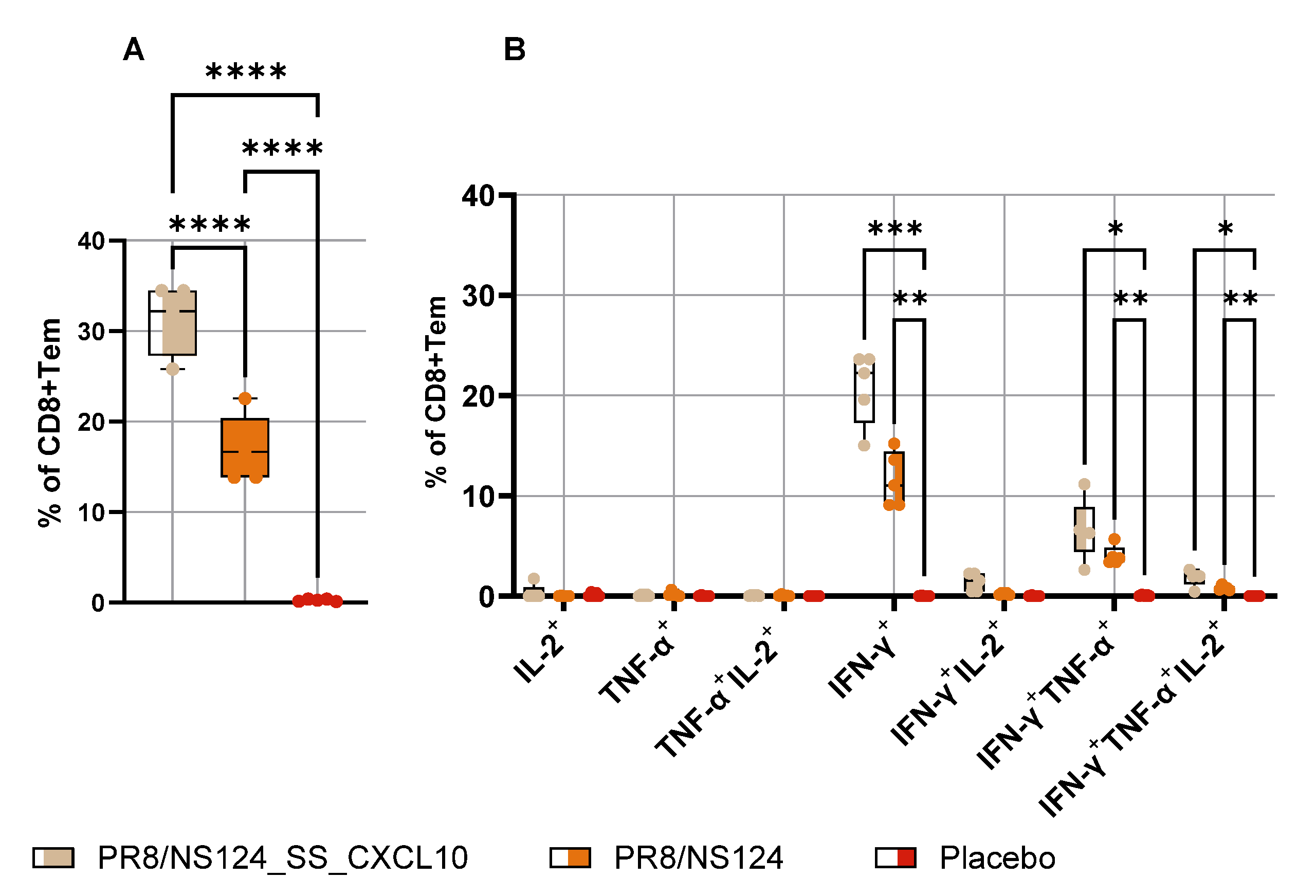
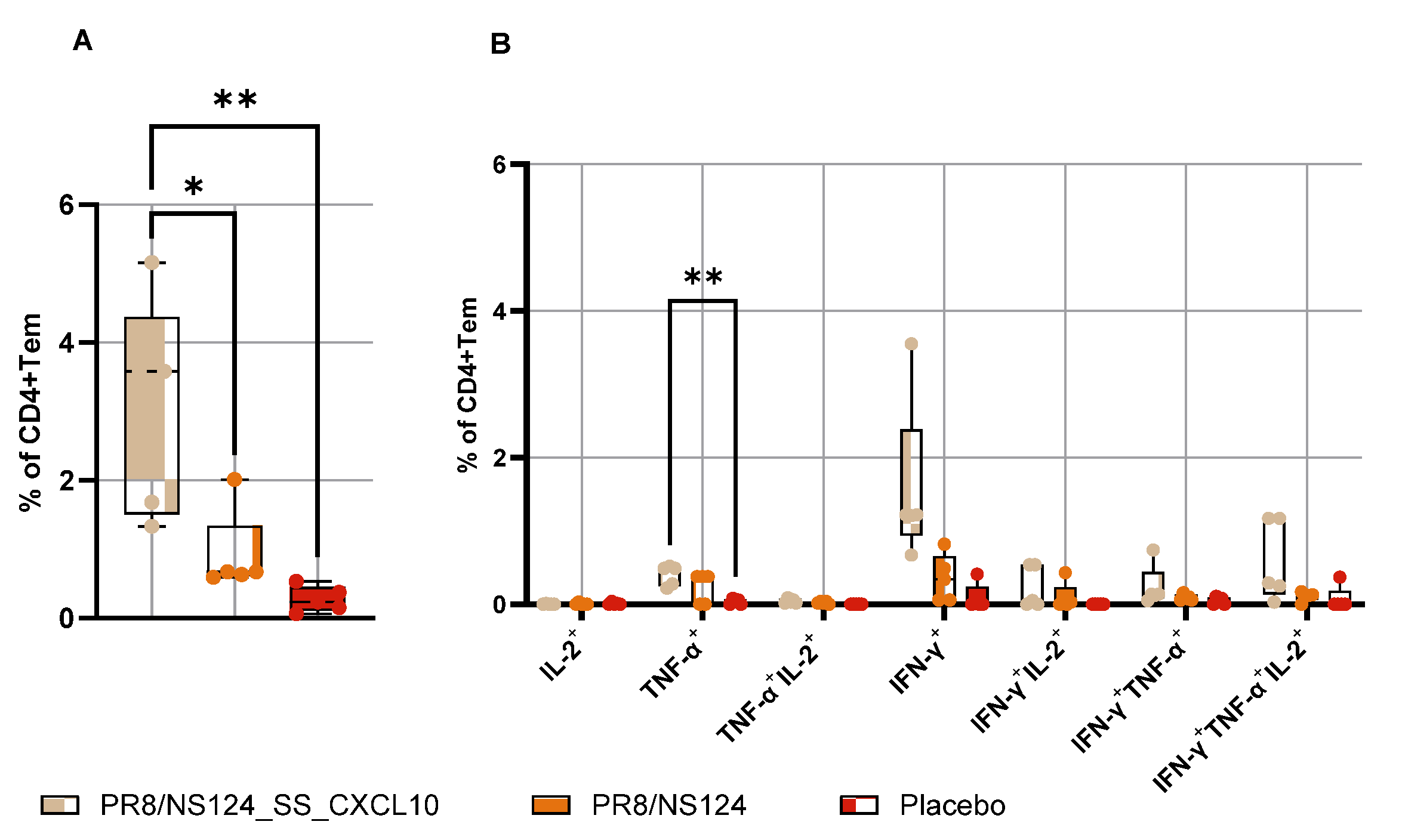
Influenza A virus (IAV) vectors with truncated NS1 proteins combine strong innate adjuvanticity with genetic flexibility and are attractive platforms for immune modulation. We engineered an NS1-truncated A/Puerto Rico/8/34 (H1N1) virus, PR8/NS124_SS_CXCL10, to express human CXCL10 from the NS segment and compared its biological and immunological properties with the parental NS124 vector in mice. The CXCL10-expressing virus replicated efficiently in embryonated chicken eggs and MDCK cells and secreted high levels of CXCL10, but showed profoundly reduced replication in mouse lungs and peritoneal cavity, indicating a strongly attenuated in vivo phenotype. After intraperitoneal immunization, both vectors induced rapid local cytokine and innate-cell recruitment, although early inflammatory responses and viral RNA loads were lower with PR8/NS124_SS_CXCL10 than with NS124. Despite this attenuation, PR8/NS124_SS_CXCL10 elicited significantly higher frequencies of systemic antigen-specific CD8⁺ and CD4⁺ effector-memory T cells producing IFN-γ, TNF-α, and IL-2, and promoted robust recall CD8⁺ and CD4⁺ T-cell responses in the lungs following low-dose homologous challenge. In a stringent heterologous challenge model with A/Aichi/2/68 (H3N2), however, mice primed intranasally with the CXCL10 vector experienced greater weight loss than NS124-primed animals, consistent with enhanced T-cell–driven immunopathology in the context of insufficient early viral control. These data show that CXCL10 expression in an NS1-attenuated IAV backbone simultaneously enforces replication restriction and amplifies T-cell immunogenicity, supporting its potential as a chemokine-armed platform for immune modulation and oncolytic virotherapy while underscoring the need to carefully balance mucosal priming and recall in chemokine-expressing influenza vaccines.
Keywords:
1. Introduction
2. Materials and Methods
Cells
Generation of Recombinant Virus
Animals
Viral Infectious Activity Analysis
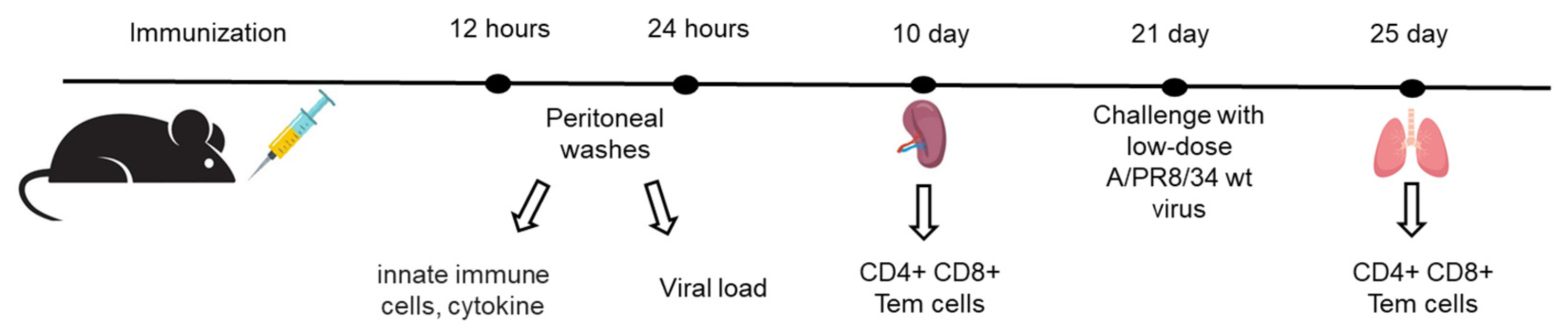
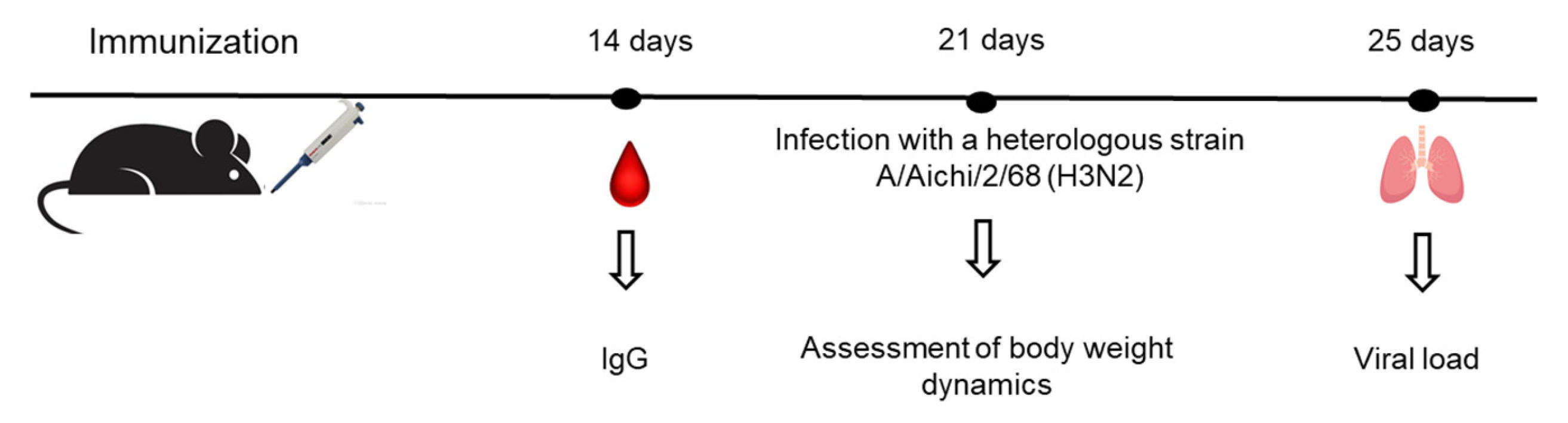
Immunization
Real-Time PCR for Detection of Influenza A
Cytokine Production Analysis
Leukocytes Isolation and Stimulation
Flow Cytometry
Challenge with Influenza Viruses
Enzyme-Linked Immunosorbent Assay (ELISA)
Statistical Analysis
3. Results
3.1. Construction of Influenza Vectors Expressing CXCR3 Receptor Ligands
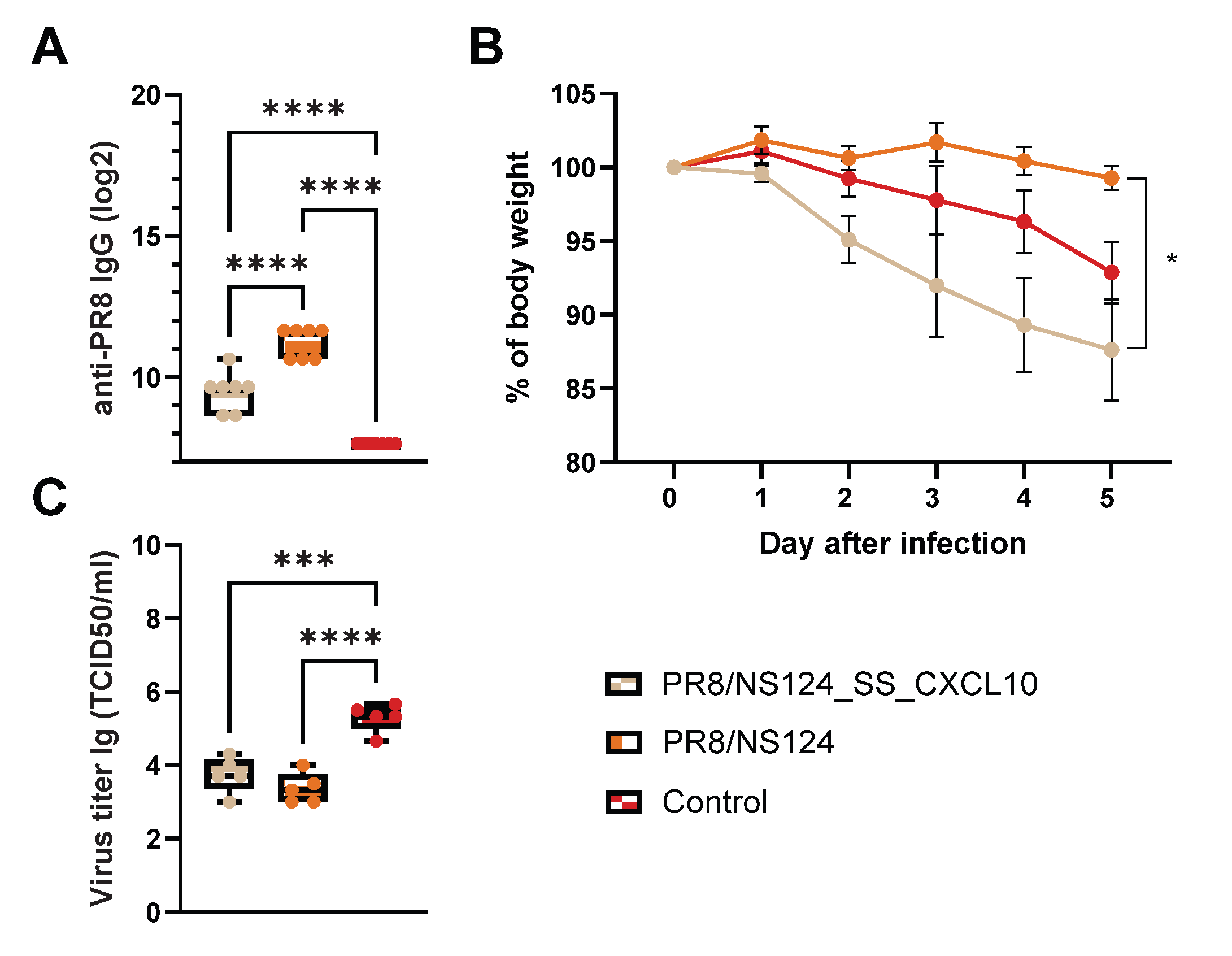
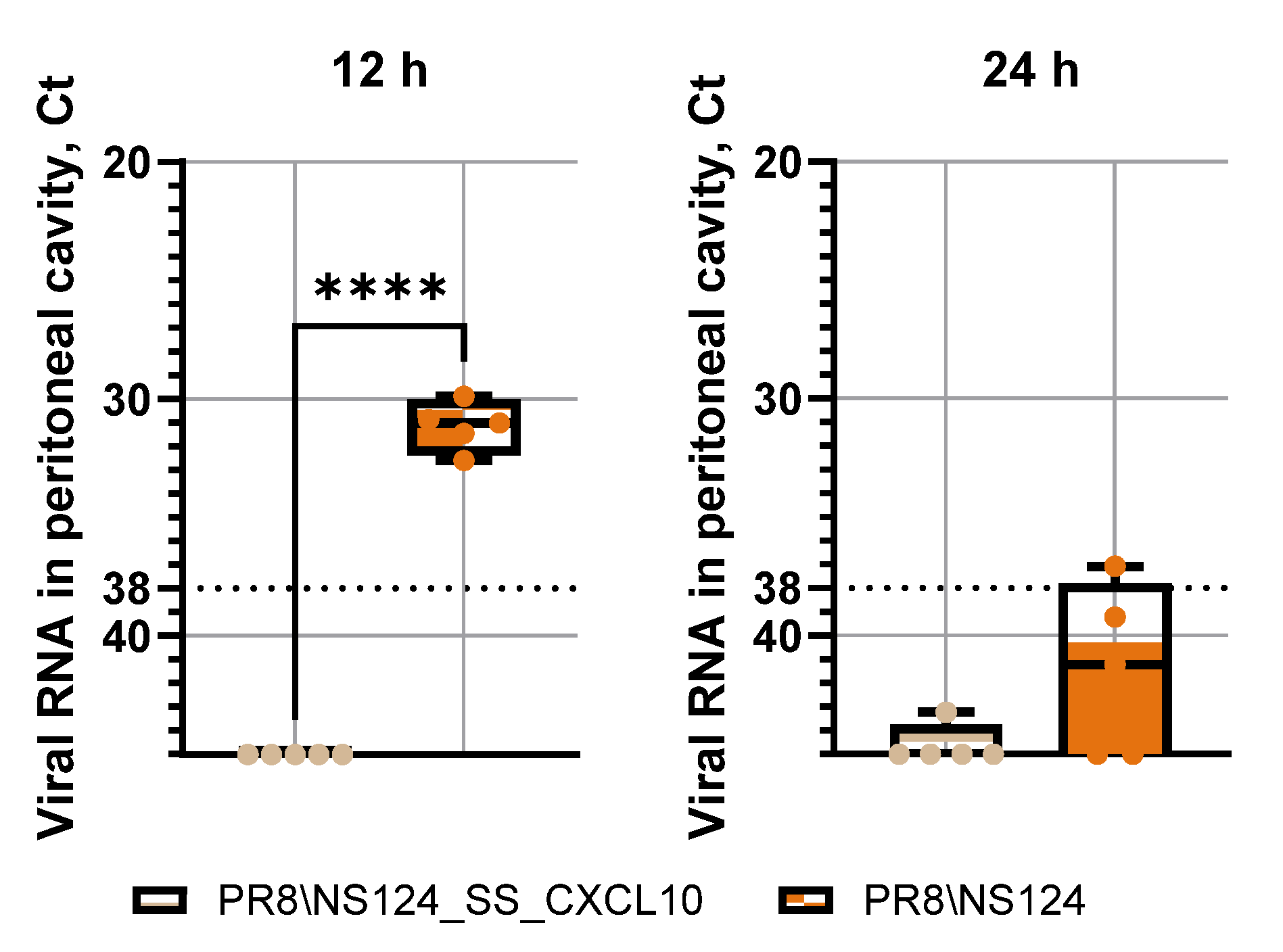
3.2. Attenuation of the PR8/NS124_SS_CXCL10 Vector in Mice
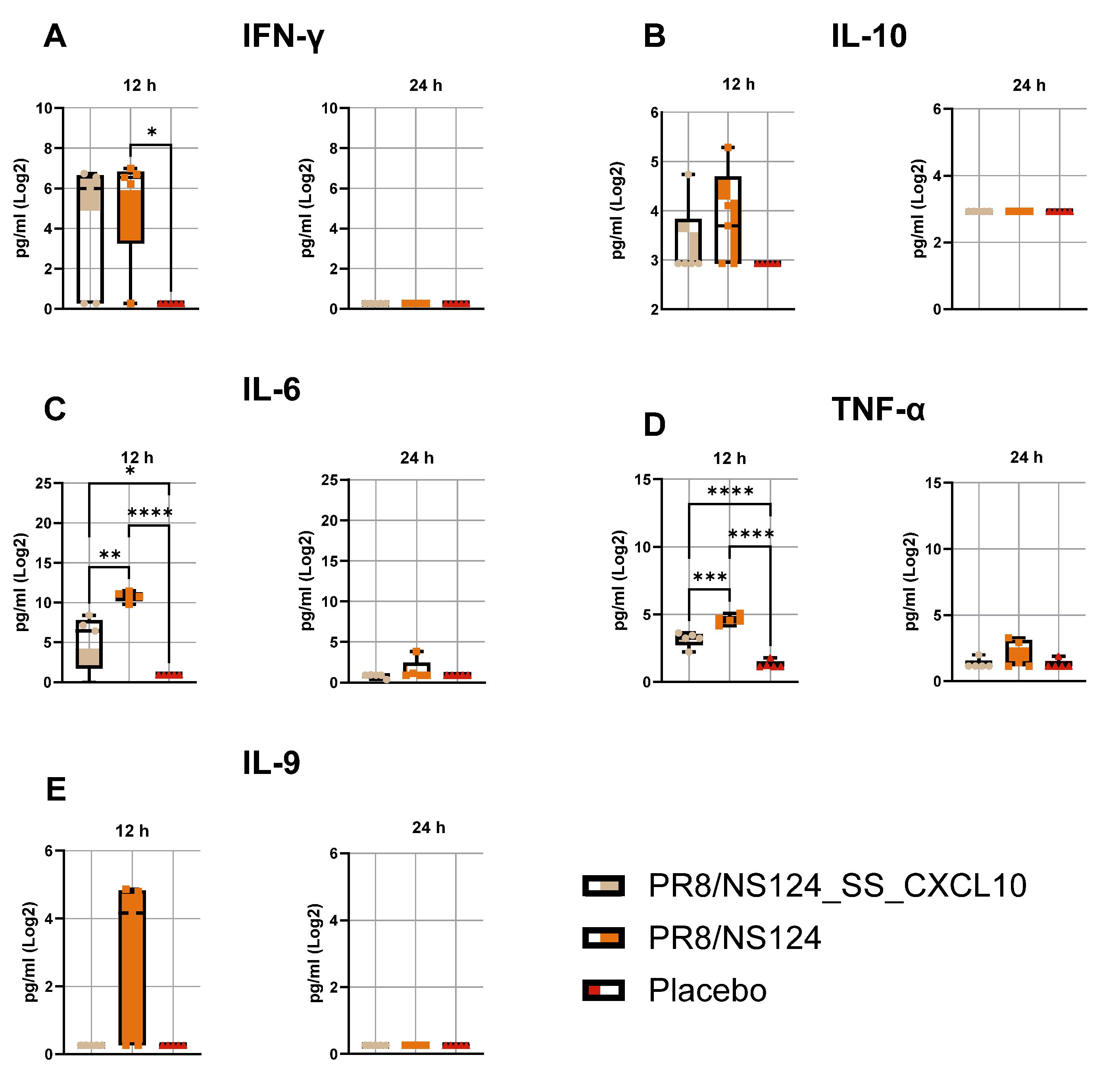
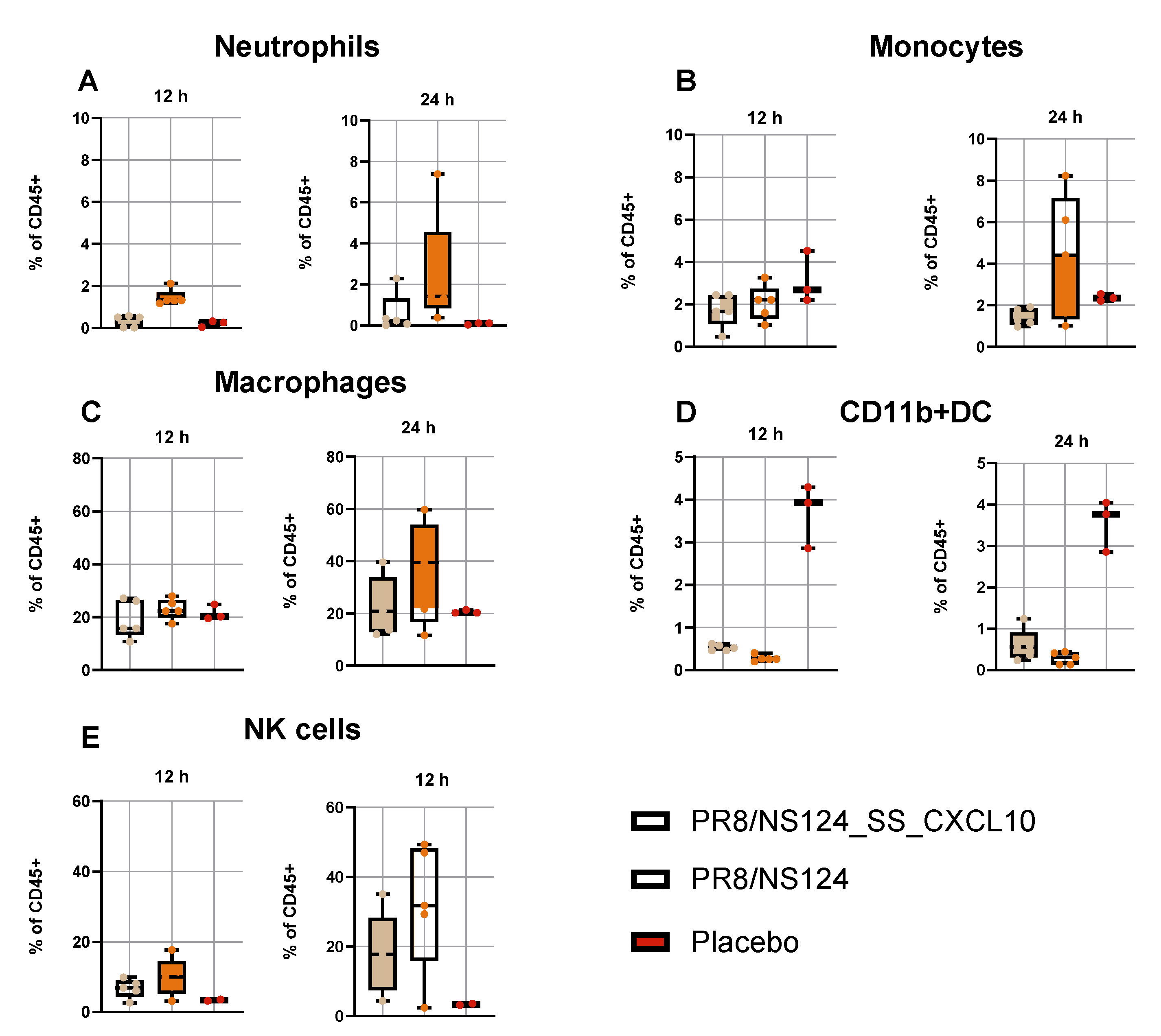
3.3. Innate Immune Response
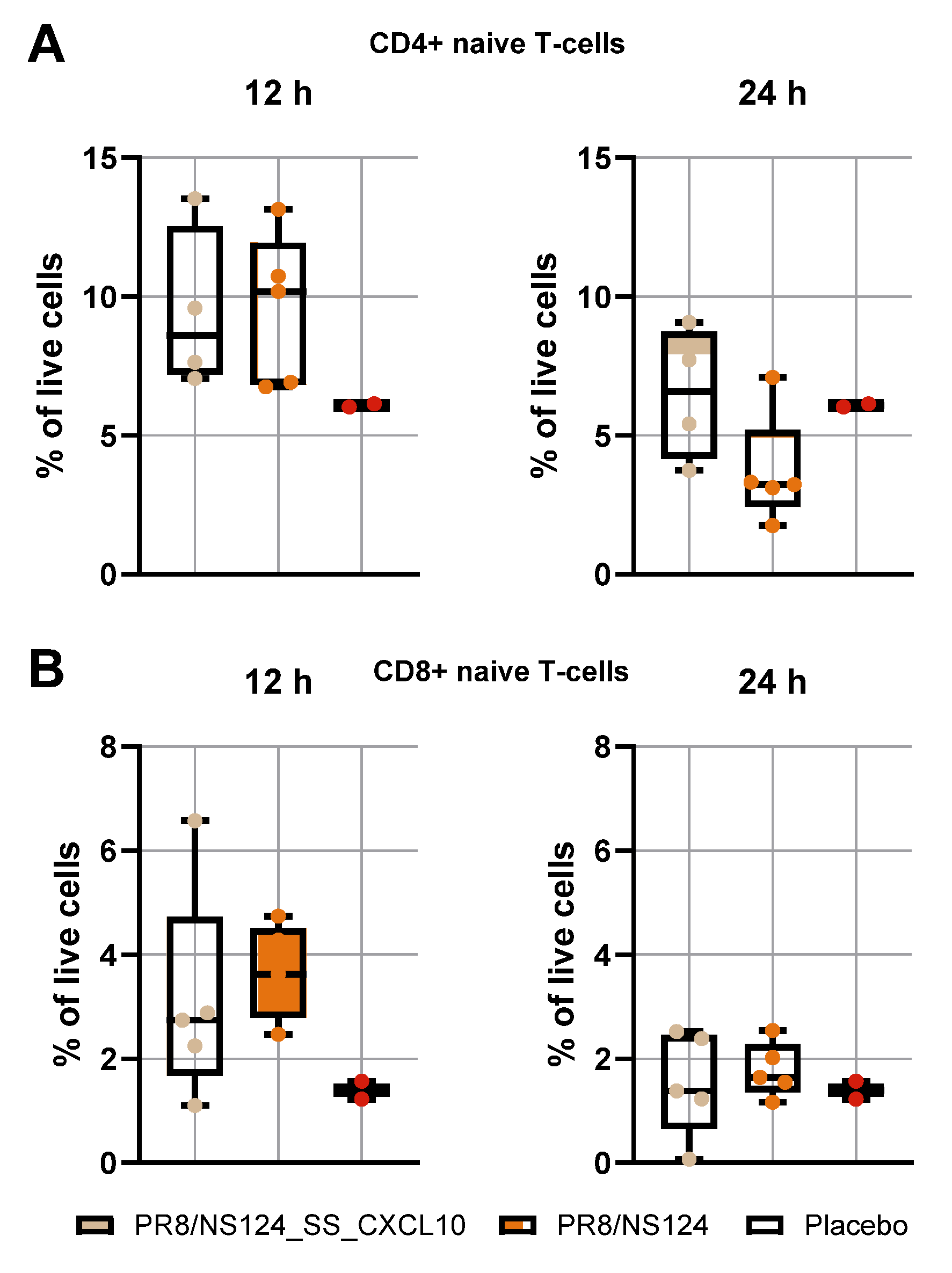
3.4. Adaptive Immune Response
3.4. Intranasal Immunization with CXCL10 Expressing Vector Provoked Lung Damage in Mice
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| IAV | Influenza A virus |
| ANOVA | analysis of variance |
| DC | Dendritic cells |
| CXCR3 | C-X-C motif receptor 3 |
| CXCL10 | High serum C-X-C motif chemokine ligand 10 |
| NK | Natural killer |
| NS | Nonstructural protein |
| CE | Chicken embryos |
| EID50 | 50% embryonic infectious dose |
| TCID50 | 50% culture infectious dose |
| TMB | 3,3′,5,5′-Tetramethylbenzidine |
| MDCK | Madin-Darby canine kidney cell |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| MLD50 | 50% mice lethal Dose |
| PCR | polymerase chain reaction |
| HSV-1, | Human herpesvirus 1 |
| VAERD | Vaccine-associated enhanced respiratory disease |
| VSV | Vesicular stomatitis virus |
| SEM | standard error of the mean |
| SD | standard deviation |
| PBS | Phosphate-buffered saline |
References
- Bian, C.; Liu, S.; Liu, N.; Zhang, G.; Xing, L.; Song, Y.; Duan, Y.; Gu, H.; Zhou, Y.; Zhang, P.; et al. Influenza Virus Vaccine Expressing Fusion and Attachment Protein Epitopes of Respiratory Syncytial Virus Induces Protective Antibodies in BALB/c Mice. Antiviral Res 2014, 104, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Bugybayeva, D.; Kydyrbayev, Z.; Zinina, N.; Assanzhanova, N.; Yespembetov, B.; Kozhamkulov, Y.; Zakarya, K.; Ryskeldinova, S.; Tabynov, K. A New Candidate Vaccine for Human Brucellosis Based on Influenza Viral Vectors: A Preliminary Investigation for the Development of an Immunization Schedule in a Guinea Pig Model. Infect Dis Poverty 2021, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Kittel, C.; Ferko, B.; Kurz, M.; Voglauer, R.; Sereinig, S.; Romanova, J.; Stiegler, G.; Katinger, H.; Egorov, A. Generation of an Influenza A Virus Vector Expressing Biologically Active Human Interleukin-2 from the NS Gene Segment. J Virol 2005, 79, 10672–10677. [Google Scholar] [CrossRef]
- Egorov, A.; Brandt, S.; Sereinig, S.; Romanova, J.; Ferko, B.; Katinger, D.; Grassauer, A.; Alexandrova, G.; Katinger, H.; Muster, T. Transfectant Influenza A Viruses with Long Deletions in the NS1 Protein Grow Efficiently in Vero Cells. J Virol 1998, 72, 6437–6441. [Google Scholar] [CrossRef]
- Shurygina, A.P.; Shuklina, M.; Ozhereleva, O.; Romanovskaya-Romanko, E.; Kovaleva, S.; Egorov, A.; Lioznov, D.; Stukova, M. Truncated NS1 Influenza A Virus Induces a Robust Antigen-Specific Tissue-Resident T-Cell Response and Promotes Inducible Bronchus-Associated Lymphoid Tissue Formation in Mice. Vaccines (Basel) 2025, 13. [Google Scholar] [CrossRef]
- Stukova, M.A.; Sereinig, S.; Zabolotnyh, N. V; Ferko, B.; Kittel, C.; Romanova, J.; Vinogradova, T.I.; Katinger, H.; Kiselev, O.I.; Egorov, A. Vaccine Potential of Influenza Vectors Expressing Mycobacterium Tuberculosis ESAT-6 Protein. Tuberculosis 2006, 86, 236–246. [Google Scholar] [CrossRef]
- Luytjes, W.; Krystai, M.; Enami, M.; Parvin, J.D.; Ralese, P. Amplification, Expression, and Packaging of a Foreign Gene by Influenza Virus. Cell 1989, 59, 1107–1113. [Google Scholar] [CrossRef]
- Ferko, B.; Stasakova, J.; Romanova, J.; Kittel, C.; Sereinig, S.; Katinger, H.; Egorov, A. Immunogenicity and Protection Efficacy of Replication-Deficient Influenza A Viruses with Altered NS1 Genes. J Virol 2004, 78, 13037–13045. [Google Scholar] [CrossRef]
- Ferko, B.; Kittel, C.; Romanova, J.; Sereinig, S.; Katinger, H.; Egorov, A. Live Attenuated Influenza Virus Expressing Human Interleukin-2 Reveals Increased Immunogenic Potential in Young and Aged Hosts. J Virol 2006, 80, 11621–11627. [Google Scholar] [CrossRef]
- García-Sastre, A.; Egorov, A.; Matassov, D.; Brandt, S.; Levy, D.E.; Durbin, J.E.; Palese, P.; Muster, T. Influenza A Virus Lacking the NS1 Gene Replicates in Interferon-Deficient Systems. Virology 1998, 252, 324–330. [Google Scholar] [CrossRef]
- Vasilyev, K.A.; Yukhneva, M.A.; Shurygina, A.-P.S.; Stukova, M.A.; Egorov, A.Y. Enhancement of the Immunogenicity of Influenza A Virus by the Inhibition of Immunosuppressive Function of NS1 Protein. Microbiology Independent Research Journal (MIR Journal) 2018, 5. [Google Scholar] [CrossRef]
- Muster, T.; Rajtarova, J.; Sachet, M.; Unger, H.; Fleischhacker, R.; Romirer, I.; Grassauer, A.; Url, A.; García-Sastre, A.; Wolff, K.; et al. Interferon Resistance Promotes Oncolysis by Influenza Virus NS1-Deletion Mutants. Int J Cancer 2004, 110, 15–21. [Google Scholar] [CrossRef] [PubMed]
- van Rikxoort, M.; Michaelis, M.; Wolschek, M.; Muster, T.; Egorov, A.; Seipelt, J.; Doerr, H.W.; Cinatl, J. Oncolytic Effects of a Novel Influenza A Virus Expressing Interleukin-15 from the NS Reading Frame. PLoS One 2012, 7, e36506. [Google Scholar] [CrossRef] [PubMed]
- Loetscher, P.; Pellegrino, A.; Gong, J.H.; Mattioli, I.; Loetscher, M.; Bardi, G.; Baggiolini, M.; Clark-Lewis, I. The Ligands of CXC Chemokine Receptor 3, I-TAC, Mig, and IP10, Are Natural Antagonists for CCR3. Journal of Biological Chemistry 2001, 276, 2986–2991. [Google Scholar] [CrossRef]
- Groom, J.R.; Luster, A.D. CXCR3 Ligands: Redundant, Collaborative and Antagonistic Functions. Immunol Cell Biol 2011, 89, 207–215. [Google Scholar] [CrossRef]
- Ren, G.; Doshi, M.; Hack, B.K.; Alexander, J.J.; Quigg, R.J. Isolation and Characterization of a Novel Rat Factor H-Related Protein That Is up-Regulated in Glomeruli under Complement Attack. Journal of Biological Chemistry 2002, 277, 48351–48358. [Google Scholar] [CrossRef]
- Mikucki, M.E.; Fisher, D.T.; Matsuzaki, J.; Skitzki, J.J.; Gaulin, N.B.; Muhitch, J.B.; Ku, A.W.; Frelinger, J.G.; Odunsi, K.; Gajewski, T.F.; et al. Non-Redundant Requirement for CXCR3 Signalling during Tumoricidal T-Cell Trafficking across Tumour Vascular Checkpoints. Nat Commun 2015, 6, 7458. [Google Scholar] [CrossRef]
- Trifilo, M.J.; Lane, T.E. Adenovirus-Mediated Expression of CXCL10 in the Central Nervous System Results in T-Cell Recruitment and Limited Neuropathology. J Neurovirol 2003, 9, 315–324. [Google Scholar] [CrossRef]
- Li, X.; Lu, M.; Yuan, M.; Ye, J.; Zhang, W.; Xu, L.; Wu, X.; Hui, B.; Yang, Y.; Wei, B.; et al. CXCL10-Armed Oncolytic Adenovirus Promotes Tumor-Infiltrating T-Cell Chemotaxis to Enhance Anti-PD-1 Therapy. Oncoimmunology 2022, 11. [Google Scholar] [CrossRef]
- Feodoroff, M.; Hamdan, F.; Antignani, G.; Feola, S.; Fusciello, M.; Russo, S.; Chiaro, J.; Välimäki, K.; Pellinen, T.; Branca, R.M.; et al. Enhancing T-Cell Recruitment in Renal Cell Carcinoma with Cytokine-Armed Adenoviruses. Oncoimmunology 2024, 13. [Google Scholar] [CrossRef]
- von Roemeling, C.A.; Patel, J.A.; Carpenter, S.L.; Yegorov, O.; Yang, C.; Bhatia, A.; Doonan, B.P.; Russell, R.; Trivedi, V.S.; Klippel, K.; et al. Adeno-Associated Virus Delivered CXCL9 Sensitizes Glioblastoma to Anti-PD-1 Immune Checkpoint Blockade. Nat Commun 2024, 15, 5871. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, E.; Neumann, G.; Hobom, G.; Webster, R.G.; Kawaoka, Y. “Ambisense” Approach for the Generation of Influenza A Virus: VRNA and MRNA Synthesis from One Template. Virology 2000, 267, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.J.; Muench, H. A Simple Method of Estimating Fifty per Cent Endpoints. Am J Epidemiol 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Vasilyev, K.; Shurygina, A.-P.; Sergeeva, M.; Stukova, M.; Egorov, A. Intranasal Immunization with the Influenza A Virus Encoding Truncated NS1 Protein Protects Mice from Heterologous Challenge by Restraining the Inflammatory Response in the Lungs. Microorganisms 2021, Vol. 9, 2021, 9. [Google Scholar] [CrossRef]
- Tokunaga, R.; Zhang, W.; Naseem, M.; Puccini, A.; Berger, M.D.; Soni, S.; McSkane, M.; Baba, H.; Lenz, H.J. CXCL9, CXCL10, CXCL11/CXCR3 Axis for Immune Activation – A Target for Novel Cancer Therapy. Cancer Treat Rev 2018, 63, 40–47. [Google Scholar] [CrossRef]
- Srivastava, R.; Khan, A.A.; Chilukuri, S.; Syed, S.A.; Tran, T.T.; Furness, J.; Bahraoui, E.; BenMohamed, L. CXCL10/CXCR3-Dependent Mobilization of Herpes Simplex Virus-Specific CD8 + T EM and CD8 + T RM Cells within Infected Tissues Allows Efficient Protection against Recurrent Herpesvirus Infection and Disease. J Virol 2017, 91, 278–295. [Google Scholar] [CrossRef]
- Steain, M.; Gowrishankar, K.; Rodriguez, M.; Slobedman, B.; Abendroth, A. Upregulation of CXCL10 in Human Dorsal Root Ganglia during Experimental and Natural Varicella-Zoster Virus Infection. J Virol 2011, 85, 626–631. [Google Scholar] [CrossRef]
- Xue, W.; Zhang, J.; Tong, H.; Xie, T.; Chen, X.; Zhou, B.; Wu, P.; Zhong, P.; Du, X.; Guo, Y.; et al. Effects of BMPER, CXCL10, and HOXA9 on Neovascularization During Early-Growth Stage of Primary High-Grade Glioma and Their Corresponding MRI Biomarkers. Front Oncol 2020, 10, 497108. [Google Scholar] [CrossRef]
- Maru, S. V.; Holloway, K.A.; Flynn, G.; Lancashire, C.L.; Loughlin, A.J.; Male, D.K.; Romero, I.A. Chemokine Production and Chemokine Receptor Expression by Human Glioma Cells: Role of CXCL10 in Tumour Cell Proliferation. J Neuroimmunol 2008, 199, 35–45. [Google Scholar] [CrossRef]
- Altenburg, A.F.; Rimmelzwaan, G.F.; de Vries, R.D. Virus-Specific T Cells as Correlate of (Cross-)Protective Immunity against Influenza. Vaccine 2015, 33, 500–506. [Google Scholar] [CrossRef]
- Grebe, K.M.; Yewdell, J.W.; Bennink, J.R. Heterosubtypic Immunity to Influenza A Virus: Where Do We Stand? Microbes Infect 2008, 10, 1024–1029. [Google Scholar] [CrossRef]
- Ullah, M.A.; Rittchen, S.; Li, J.; Hasnain, S.Z.; Phipps, S. DP1 Prostanoid Receptor Activation Increases the Severity of an Acute Lower Respiratory Viral Infection in Mice via TNF-α-Induced Immunopathology. Mucosal Immunol 2021, 14, 963–972. [Google Scholar] [CrossRef]
- Sun, L.; Su, Y.; Jiao, A.; Wang, X.; Zhang, B. T Cells in Health and Disease. Signal Transduction and Targeted Therapy 2023, 2023 8:1 8, 235. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



