Submitted:
10 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
Keywords:
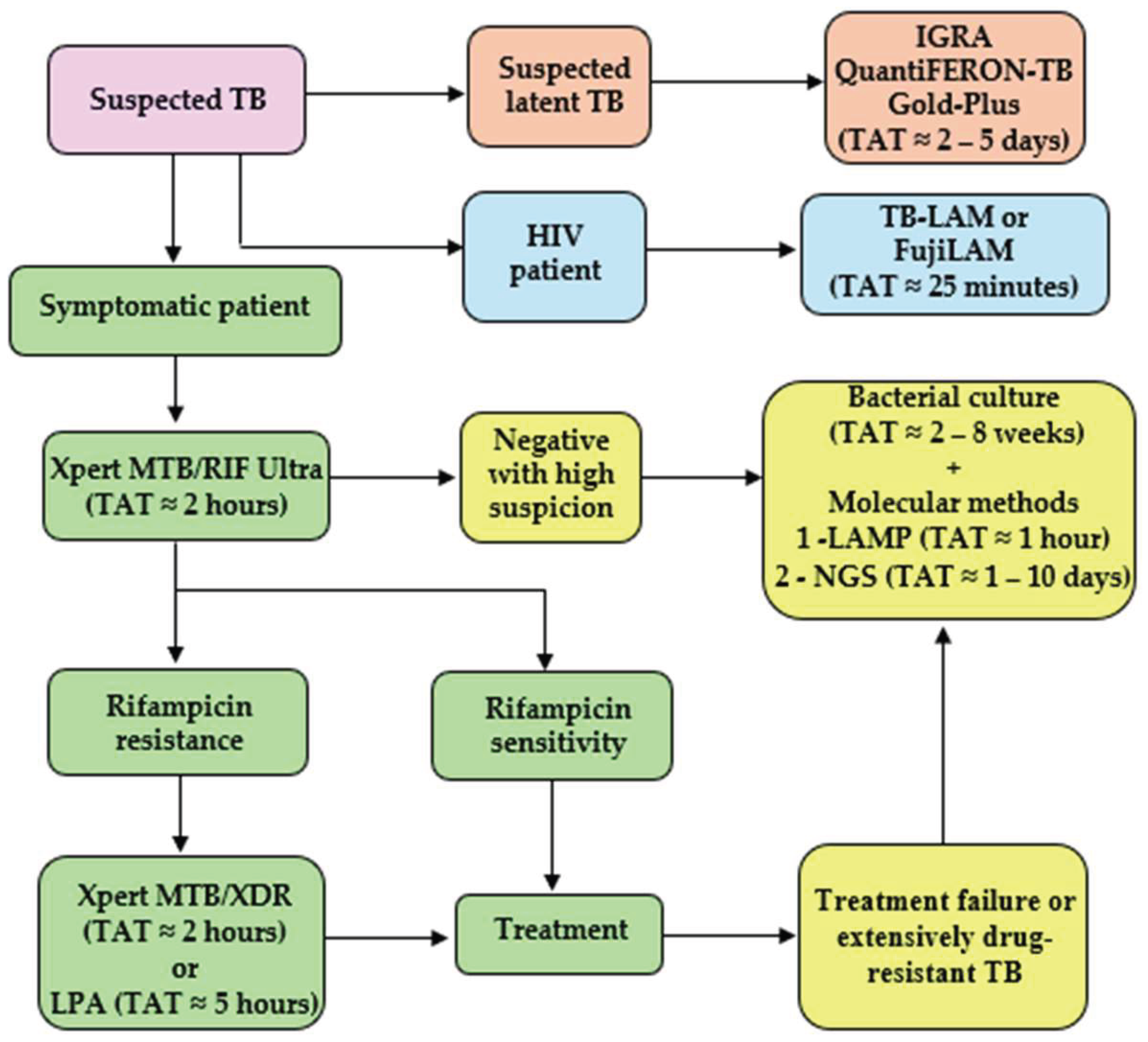
1. Introduction
2. Search Strategy
3. Classic Methods
3.1. Bacterial Culture
3.2. Smear Microscopy
4. Latent Tuberculosis Diagnosis
5. Lateral Flow Assays
6. Molecular Methods
6.1. Automated Nucleic Acid Amplification Tests (NAAT)
6.2. Loop-Mediated Isothermal Amplification (LAMP)
6.3. Line Probe Hybridization (LPA)
6.4. Next-Generation Sequencing (NGS)
7. Practical Implications for the Clinical Laboratory
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BCG | Bacillus Calmette -Guérin |
| DST | Drug susceptibility testing |
| ELISA | Enzyme-linked immunoassay |
| HIV | Human immunodeficiency virus |
| IGRA | Interferon-gamma release assay |
| IFN- γ | Interferon-gamma |
| LAM | Lipoarabinomannan |
| LAMP | Loop-mediated isothermal amplification |
| LF-LAM | Lateral flow immunoassay |
| LPA | Line probe assay |
| MDR | Multidrug resistance |
| MGIT | Mycobacteria Growth Indicator Tube |
| mNGS | Metagenomic next-generation sequencing |
| M. tuberculosis | Mycobacterium tuberculosis |
| NAAT | Automated nucleic acid amplification tests |
| NGS | Next-generation sequencing |
| PCR | Polymerase Chain Reaction |
| PPD | Purified protein derivative |
| RIF | Rifampicin |
| TB | Tuberculosis |
| tNGS | Targeted next-generation sequencing |
| UV | Ultraviolet |
| XDR | Extensively drug-resistant |
| WHO | World health organization |
References
- World Health Organization. Global Tuberculosis Report 2024. 1st ed. Geneva: World Health Organization; 2024. Accessed on: 21 September 2025. Available online: https://www.who.int/teams/global-programme-on-tuberculosis-and-lung-health/tb-reports/global-tuberculosis-report-2024.
- Tortora GJ. et al. Microbiologia. 14th ed. Porto Alegre: Artmed; 2025.
- Ortiz-Brizuela E., Menzies D., Behr M.A. Testing and Treating Mycobacterium tuberculosis Infection. Med Clin North Am 2022;106:929–47. [CrossRef]
- Houben R.M.G.J., Dodd P.J. The Global Burden of Latent Tuberculosis Infection: A Re-estimation Using Mathematical Modelling. PLOS Med 2016;13:e1002152. [CrossRef]
- Emery J.C., Richards A.S., Dale K.D., et al. Self-clearance of Mycobacterium tuberculosis infection: implications for lifetime risk and population at-risk of tuberculosis disease. Proc R Soc B Biol Sci 2021;288:20201635. [CrossRef]
- Nações Unidas Brasil. Objetivo de Desenvolvimento Sustentável 3: Saúde e Bem-Estar [Internet]. Brasília: ONU Brasil; Accessed on: 18 September 2025. Available online: https://brasil.un.org/pt-br/sdgs/3.
- World Health Organization. WHO consolidated guidelines on tuberculosis: rapid diagnostics for tuberculosis detection; Module 3: Diagnosis. Third edition. Geneva: World Health Organization; 2024. Accessed on: 21 September 2025. Available onlne: https://www.who.int/publications/i/item/9789240089488.
- Pati H.P., Singh G. Turnaround Time (TAT): Difference in Concept for Laboratory and Clinician. Indian J Hematol Blood Transfus 2014;30:81–4. [CrossRef]
- Kanade S., Mohammed Z., Kulkarni A., et al. Comparison of Xpert MTB/RIF Assay, Line Probe Assay, and Culture in Diagnosis of Pulmonary Tuberculosis on Bronchoscopic Specimen. Int J Mycobacteriology 2023;12:151. [CrossRef]
- Santos F.D.J., Tenorio J.E.D.O.S., Portugal L.G. A importância da realização de cultura de escarro para o diagnóstico de tuberculose pulmonar em pacientes paucibacilares. Rev Bras Análises Clínicas 2024;56:90–5. [CrossRef]
- Baquero-Artigao F., del Rosal T., Falcón-Neyra L., et al. Update on the diagnosis and treatment of tuberculosis. An Pediatría Engl Ed 2023;98:460–9. [CrossRef]
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Departamento de Doenças de Condições Crônicas e Infecções Sexualmente Transmissíveis. Manual de Recomendações para o Diagnóstico Laboratorial de Tuberculose e Micobactérias não Tuberculosas de Interesse em Saúde Pública no Brasil. – Brasília: Ministério da Saúde, 2022. Accessed on: 21 September 2025. Available online: https://www.gov.br/saude/pt-br/centrais-de-conteudo/publicacoes/svsa/tuberculose/manual-de-recomendacoes-e-para-diagnostico-laboratorial-de-tuberculose-e-micobacterias-nao-tuberculosas-de-interesse-em-saude-publica-no-brasil.pdf/view.
- Raj A., Baliga S., Shenoy M.S., et al. Validity of a CB-NAAT assay in diagnosing tuberculosis in comparison to culture: A study from an urban area of South India. J Clin Tuberc Mycobact Dis 2020;21:100198. [CrossRef]
- McNerney R., Clark T.G., Campino S., et al. Removing the bottleneck in whole genome sequencing of Mycobacterium tuberculosis for rapid drug resistance analysis: a call to action. Int J Infect Dis 2017;56:130–5. [CrossRef]
- Boldi M-O., Denis-Lessard J., Neziri R., et al. Performance of microbiological tests for tuberculosis diagnostic according to the type of respiratory specimen: A 10-year retrospective study. Front Cell Infect Microbiol 2023;13:1131241. [CrossRef]
- Siddiqi S.H., Ruesch-Gerdes S. MGIT procedure manual for BACTEC™ MGIT 960™ TB System (Also applicable for Manual MGIT): Mycobacteria Growth Indicator Tube (MGIT) Culture and Drug Susceptibility Demonstration Projects, 2006. Accessed on: 21 September 2025. Available online: https://www.finddx.org/wp-content/uploads/2023/02/20061101_rep_mgit_manual_FV_EN.pdf.
- Arora D., Dhanashree B. Utility of smear microscopy and GeneXpert for the detection of Mycobacterium tuberculosis in clinical samples. Germs 2020;10:81–7. [CrossRef]
- World Health Organization. Laboratory services in tuberculosis control. Part I: Organization and management. Geneva: World Health Organization, 1998. Accessed on: 21 September 2025. Available online: https://iris.who.int/bitstream/handle/10665/65942/WHO_TB_98.258_(part1).pdf.
- Singhal R., Myneedu V.P. Microscopy as a diagnostic tool in pulmonary tuberculosis. Int J Mycobacteriology 2015;4:1–6. [CrossRef]
- Deng Y., Duan Y., Gao S., et al. Comparison of LAMP, GeneXpert, Mycobacterial Culture, Smear Microscopy, TSPOT.TB, TBAg/PHA Ratio for Diagnosis of Pulmonary Tuberculosis. Curr Med Sci 2021;41:1023–8. [CrossRef]
- World Health Organization. Fluorescent light-emitting diode (LED) microscopy for diagnosis of tuberculosis: policy statement. Geneva: World Health Organization, 2011. Accessed on: 21 September 2025. Available online: https://iris.who.int/handle/10665/44602.
- Palanivel J., Sounderrajan V., Thangam T., et al. Latent Tuberculosis: Challenges in Diagnosis and Treatment, Perspectives, and the Crucial Role of Biomarkers. Curr Microbiol 2023;80:1–13. [CrossRef]
- Janssen S., Murphy M., Upton C., et al. Tuberculosis: An Update for the Clinician. Respirol Carlton Vic 2025;30:196–205. [CrossRef]
- Verma A., Kaur M., Singh L.V., et al. Reactivation of latent tuberculosis through modulation of resuscitation promoting factors by diabetes. Sci Rep 2021;11:19700. [CrossRef]
- World Health Organization. Rapid communication: TB antigen-based skin tests for the diagnosis of TB infection. Geneva: World Health Organization, 2022. Accessed on: 21 September 2025. Available online: https://www.who.int/publications/i/item/WHO-UCN-TB-2022.1.
- Krutikov M., Faust L., Nikolayevskyy V., et al. The diagnostic performance of novel skin-based in-vivo tests for tuberculosis infection compared with purified protein derivative tuberculin skin tests and blood-based in vitro interferon-γ release assays: a systematic review and meta-analysis. Lancet Infect Dis 2022;22:250–64. [CrossRef]
- Yang H., Kruh-Garcia N.A., Dobos K.M. Purified Protein Derivatives of Tuberculin - Past, Present, and Future. FEMS Immunol Med Microbiol 2012;66:273–80. [CrossRef]
- Hamada Y., Cirillo D.M., Matteelli A., et al. Tests for tuberculosis infection: landscape analysis. Eur Respir J 2021;58. [CrossRef]
- Starshinova A., Dovgalyk I., Malkova A., et al. Recombinant Tuberculosis Allergen (Diaskintest®) in Tuberculosis Diagnostic in Russia (Meta-Analysis). Int J Mycobacteriology 2020;9:335. [CrossRef]
- Meier N.R., Volken T., Geiger M., et al. Risk Factors for Indeterminate Interferon-Gamma Release Assay for the Diagnosis of Tuberculosis in Children—A Systematic Review and Meta-Analysis. Front Pediatr 2019;7:208. [CrossRef]
- Pai M., Zwerling A., Menzies D. Systematic Review: T-Cell–based Assays for the Diagnosis of Latent Tuberculosis Infection: An Update. Ann Intern Med 2008;149:177–84. [CrossRef]
- Goletti D., Delogu G., Matteelli A., et al. The role of IGRA in the diagnosis of tuberculosis infection, differentiating from active tuberculosis, and decision making for initiating treatment or preventive therapy of tuberculosis infection. Int J Infect Dis IJID Off Publ Int Soc Infect Dis 2022;124 Suppl 1:S12–9. [CrossRef]
- van Ingen J, de Zwaan R, Dekhuijzen R, et al. Region of Difference 1 in Nontuberculous Mycobacterium Species Adds a Phylogenetic and Taxonomical Character. J Bacteriol 2009;191:5865–7. [CrossRef]
- World Health Organization. Lateral flow urine lipoarabinomannan assay (LF-LAM) for the diagnosis of active tuberculosis in people living with HIV: policy update 2019. Geneva: World Health Organization; 2019. Accessed on: 21 September 2025. Available online: https://www.who.int/publications/i/item/9789241550604.
- Brennan P.J. Structure, function, and biogenesis of the cell wall of Mycobacterium tuberculosis. Tuberculosis (Edinb). 2003;83(1-3):91-7. [CrossRef]
- Flores J., Cancino J.C., Chavez-Galan L. Lipoarabinomannan as a Point-of-Care Assay for Diagnosis of Tuberculosis: How Far Are We to Use It? Front Microbiol 2021;12:638047. [CrossRef]
- Broger T., Nicol M.P., Sigal G.B., et al. Diagnostic accuracy of 3 urine lipoarabinomannan tuberculosis assays in HIV-negative outpatients. J Clin Invest 2020;130:5756–64. [CrossRef]
- Bulterys M.A., Wagner B., Redard-Jacot M., et al. Point-Of-Care Urine LAM Tests for Tuberculosis Diagnosis: A Status Update. J Clin Med 2019;9:111. [CrossRef]
- Broger T., Sossen B., du Toit E., et al. Novel lipoarabinomannan point-of-care tuberculosis test for people with HIV: a diagnostic accuracy study. Lancet Infect Dis 2019;19:852–61. [CrossRef]
- Das S., Mangold K.A., Shah N.S., et al. Performance and Utilization of a Laboratory-Developed Nucleic Acid Amplification Test (NAAT) for the Diagnosis of Pulmonary and Extrapulmonary Tuberculosis in a Low-Prevalence Area. Am J Clin Pathol 2020;154:115–23. [CrossRef]
- Schwab T.C., Perrig L., Göller P.C., et al. Targeted next-generation sequencing to diagnose drug-resistant tuberculosis: systematic review and test accuracy meta-analysis. Lancet Infect Dis 2024;24:1162–76. [CrossRef]
- MacLean E., Kohli M., Weber S.F., et al. Advances in Molecular Diagnosis of Tuberculosis. J Clin Microbiol 2020;58:e01582-19. [CrossRef]
- Alsharksi N.A., Sirekbasan S., Gürkök-Tan T., et al. From Tradition to Innovation: Diverse Molecular Techniques in the Fight Against Infectious Diseases. Diagnostics 2024;14:2876. [CrossRef]
- Espy M.J., Uhl J.R., Sloan L.M., et al. Real-Time PCR in Clinical Microbiology: Applications for Routine Laboratory Testing. Clin Microbiol Rev 2006;19:165–256. [CrossRef]
- World Health Organization. Automated real-time nucleic acid amplification technology for rapid and simultaneous detection of tuberculosis and rifampicin resistance: Xpert MTB/RIF system: policy statement. Geneva: World Health Organization; 2011. Accessed on: 21 september 2025. Available online: https://www.who.int/publications/i/item/9789241501545.
- Osei Sekyere J., Maphalala N., Malinga L.A., et al. A Comparative Evaluation of the New Genexpert MTB/RIF Ultra and other Rapid Diagnostic Assays for Detecting Tuberculosis in Pulmonary and Extra Pulmonary Specimens. Sci Rep 2019;9:16587. [CrossRef]
- World Health Organization. Rapid implementation of the Xpert MTB/RIF diagnostic test: technical and operational “How-to”; practical considerations. Geneva: World Health Organization; 2011. Accessed on: 21 September 2025. Available online: https://www.who.int/publications/i/item/9789241501569.
- Li Y., Jiao M., Liu Y., et al. Application of Metagenomic Next-Generation Sequencing in Mycobacterium tuberculosis Infection. Front Med 2022;9:802719. [CrossRef]
- Opota O., Mazza-Stalder J., Greub G., et al. The rapid molecular test Xpert MTB/RIF ultra: towards improved tuberculosis diagnosis and rifampicin resistance detection. Clin Microbiol Infect 2019;25:1370–6. [CrossRef]
- van Zyl-Smit R.N., Binder A., Meldau R., et al. Comparison of Quantitative Techniques including Xpert MTB/RIF to Evaluate Mycobacterial Burden. PLoS ONE 2011;6:e28815. [CrossRef]
- Horne D.J., Kohli M., Zifodya J.S., et al. Xpert MTB/RIF and Xpert MTB/RIF Ultra for pulmonary tuberculosis and rifampicin resistance in adults. Cochrane Database Syst Rev 2019;2019:CD009593. [CrossRef]
- Chakravorty S., Simmons A.M., Rowneki M., et al. The New Xpert MTB/RIF Ultra: Improving Detection of Mycobacterium tuberculosis and Resistance to Rifampin in an Assay Suitable for Point-of-Care Testing. mBio 2017;8:e00812-17. [CrossRef]
- Xie Y.L., Chakravorty S., Armstrong D.T., et al. Evaluation of a Rapid Molecular Drug-Susceptibility Test for Tuberculosis. N Engl J Med 2017;377:1043–54. [CrossRef]
- Cao Y. Xpert MTB/XDR: a 10-Color Reflex Assay Suitable for Point-of-Care Settings To Detect Isoniazid, Fluoroquinolone, and Second-Line-Injectable-Drug Resistance Directly from Mycobacterium tuberculosis-Positive Sputum. J Clin Microbiol 2021;59(3): e02314-2. [CrossRef]
- Rudeeaneksin J., Bunchoo S., Srisungngam S., et al. Rapid Identification of Mycobacterium tuberculosis in BACTEC MGIT960 Cultures by In-House Loop-Medicated Isothermal Amplification. Jpn J Infect Dis 2012;65:306–11. [CrossRef]
- Park J-W. Principles and Applications of Loop-Mediated Isothermal Amplification to Point-of-Care Tests. Biosensors 2022;12:857. [CrossRef]
- Ou X., Li Q., Xia H., et al. Diagnostic Accuracy of the PURE-LAMP Test for Pulmonary Tuberculosis at the County-Level Laboratory in China. PLoS ONE 2014;9:e94544. [CrossRef]
- Neonakis I.K., Spandidos D.A., Petinaki E. Use of loop-mediated isothermal amplification of DNA for the rapid detection of Mycobacterium tuberculosis in clinical specimens. Eur J Clin Microbiol Infect Dis 2011;30:937–42. [CrossRef]
- Kim S-H., Lee S-Y., Kim U., et al. Diverse methods of reducing and confirming false-positive results of loop-mediated isothermal amplification assays: A review. Anal Chim Acta 2023;1280:341693. [CrossRef]
- Nathavitharana R.R., Hillemann D., Schumacher S.G., et al. Multicenter Noninferiority Evaluation of Hain GenoType MTBDRplus Version 2 and Nipro NTM+MDRTB Line Probe Assays for Detection of Rifampin and Isoniazid Resistance. J Clin Microbiol 2016;54:1624–30. [CrossRef]
- World Health Organization. Line probe assays for detection of drug-resistant tuberculosis: interpretation and reporting manual for laboratory staff and clinicians. Geneva: World Health Organization; 2022. Accessed on: 21 September 2025. Available online: https://www.who.int/publications/i/item/9789240046665.
- Schünemann H.J., Mustafa R.A., Brozek J., et al. GRADE guidelines: 21 part 1. Study design, risk of bias, and indirectness in rating the certainty across a body of evidence for test accuracy. J Clin Epidemiol 2020;122:129–41. [CrossRef]
- Gu W., Miller S., Chiu C.Y. Clinical Metagenomic Next-Generation Sequencing for Pathogen Detection. Annu Rev Pathol 2019;14:319–38. [CrossRef]
- Jin W., Pan J., Miao Q., et al. Diagnostic accuracy of metagenomic next-generation sequencing for active tuberculosis in clinical practice at a tertiary general hospital. Ann Transl Med 2020;8:1065. [CrossRef]
- Gu W., Deng X., Lee M., et al. Rapid Pathogen Detection by Metagenomic Next-Generation Sequencing of Infected Body Fluids. Nat Med 2021;27:115–24. [CrossRef]
- Wang S., Chen Y., Wang D., et al. The Feasibility of Metagenomic Next-Generation Sequencing to Identify Pathogens Causing Tuberculous Meningitis in Cerebrospinal Fluid. Front Microbiol 2019;10. [CrossRef]
- Figueredo L.J.A., Miranda S.S., Santos L.B., et al. Cost analysis of smear microscopy and the Xpert assay for tuberculosis diagnosis: average turnaround time. Rev Soc Bras Med Trop 2020;53:e20200314. [CrossRef]
- Zhang C., Hu T., Xiu L., et al. Use of Ultra-Deep Sequencing in a Patient with Tuberculous Coxitis Shows Its Limitations in Extrapulmonary Tuberculosis Diagnostics: A Case Report. Infect Drug Resist 2019;12:3739–43. [CrossRef]
- Cabibbe A.M., Walker T.M., Niemann S., et al. Whole genome sequencing of Mycobacterium tuberculosis. Eur Respir J 2018;52. [CrossRef]
- de Araujo L., Cabibbe A.M., Mhuulu L., et al. Implementation of targeted next-generation sequencing for the diagnosis of drug-resistant tuberculosis in low-resource settings: a programmatic model, challenges, and initial outcomes. Front Public Health 2023;11:1204064. [CrossRef]
- World Health Organization. The use of next-generation sequencing technologies for the detection of mutations associated with drug resistance in Mycobacterium tuberculosis complex: technical guide. Geneva: World Health Organization; 2018. Accessed on: 21 September 2025. Available online: https://www.who.int/publications/i/item/WHO-CDS-TB-2018.19.
- Wylie T.N., Wylie K.M., Herter B.N., et al. Enhanced virome sequencing using targeted sequence capture. Genome Res 2015;25:1910–20. [CrossRef]
- Satam H., Joshi K., Mangrolia U., et al. Next-Generation Sequencing Technology: Current Trends and Advancements. Biology 2023;12:997. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



