Submitted:
11 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
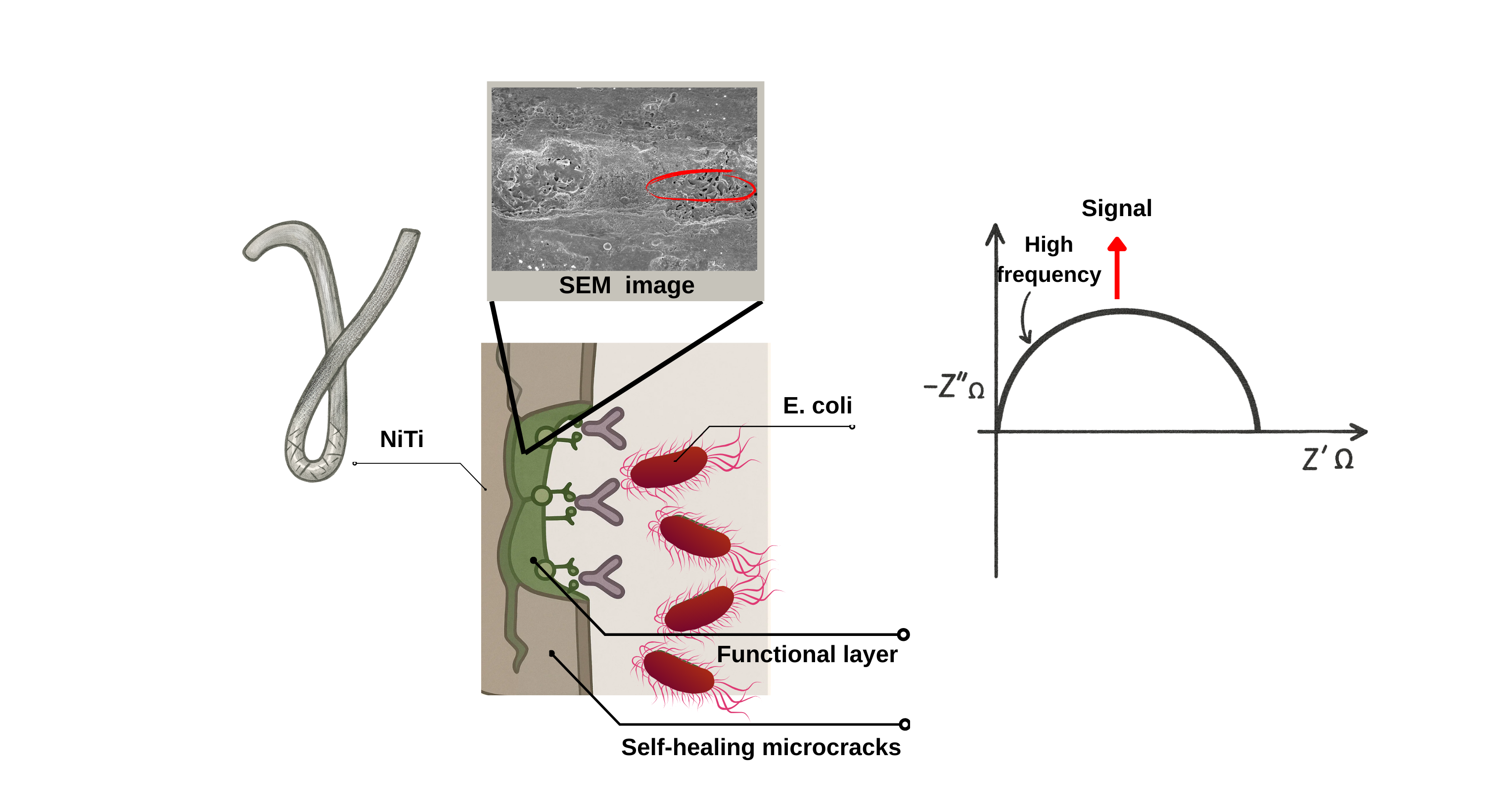
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of NiTi Microcrack Transducers
2.3. Surface Modification
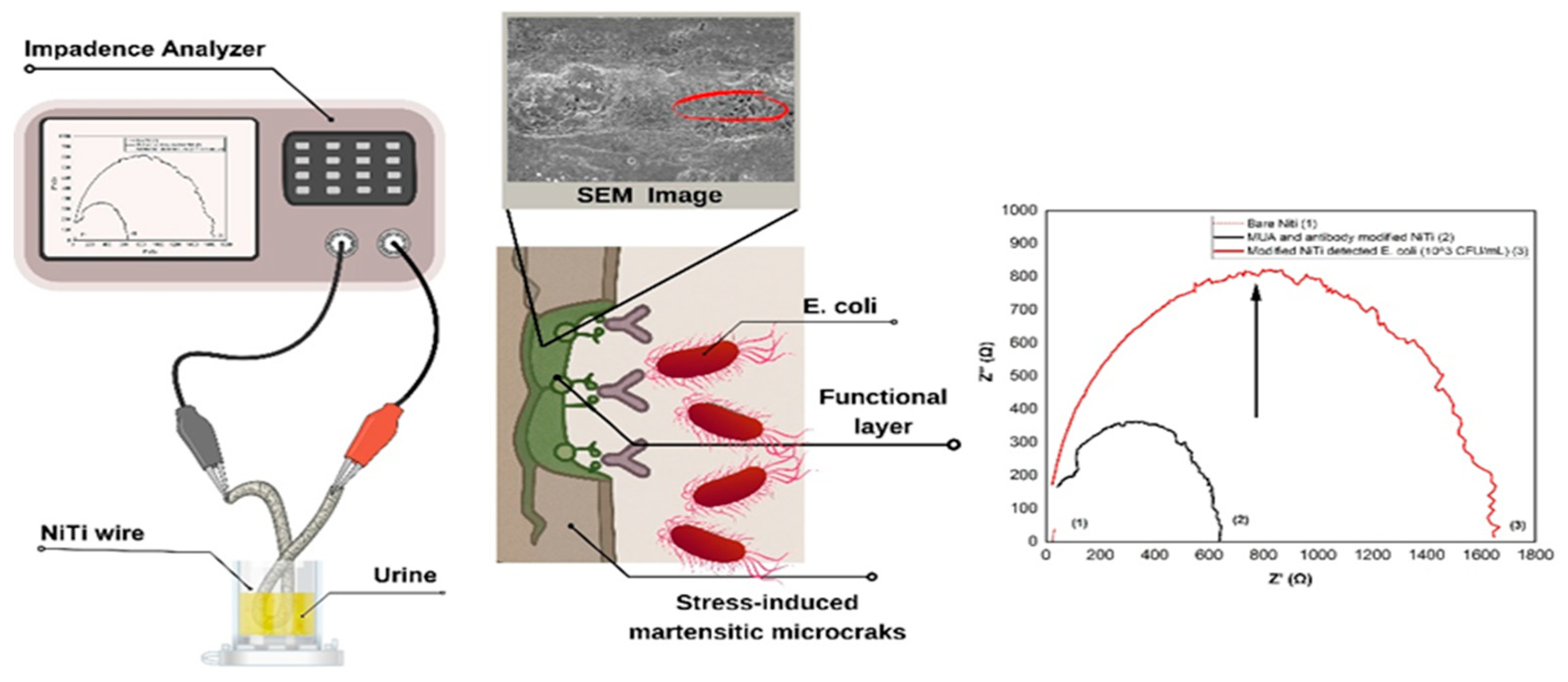
2.4. Impedance Measurements
2.5. Selectivity and Repeatability Tests
2.6. Characterization and Analysis
3. Results
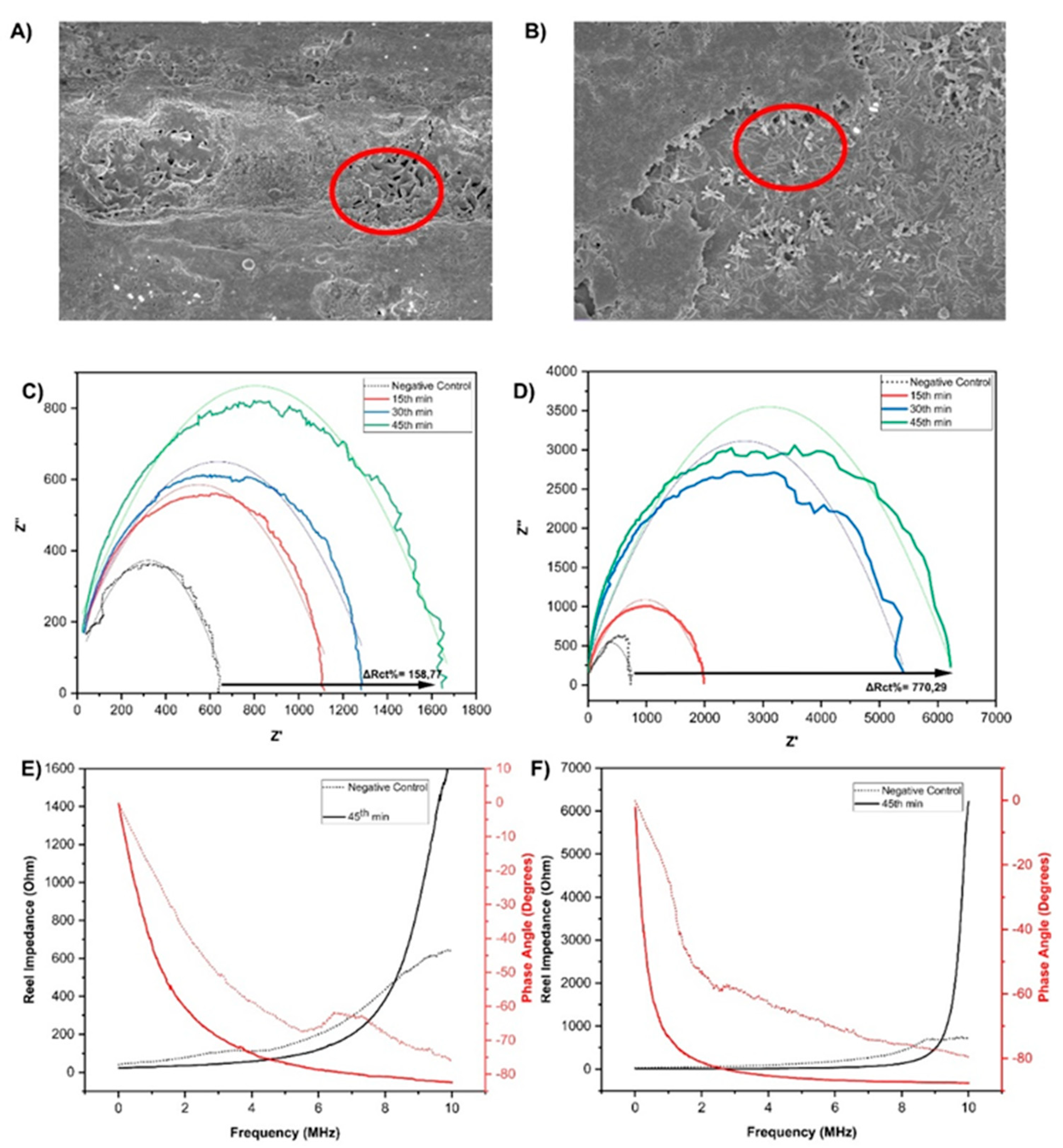
3.1. Morphological and Electrochemical Characterization of the NiTi Biosensor
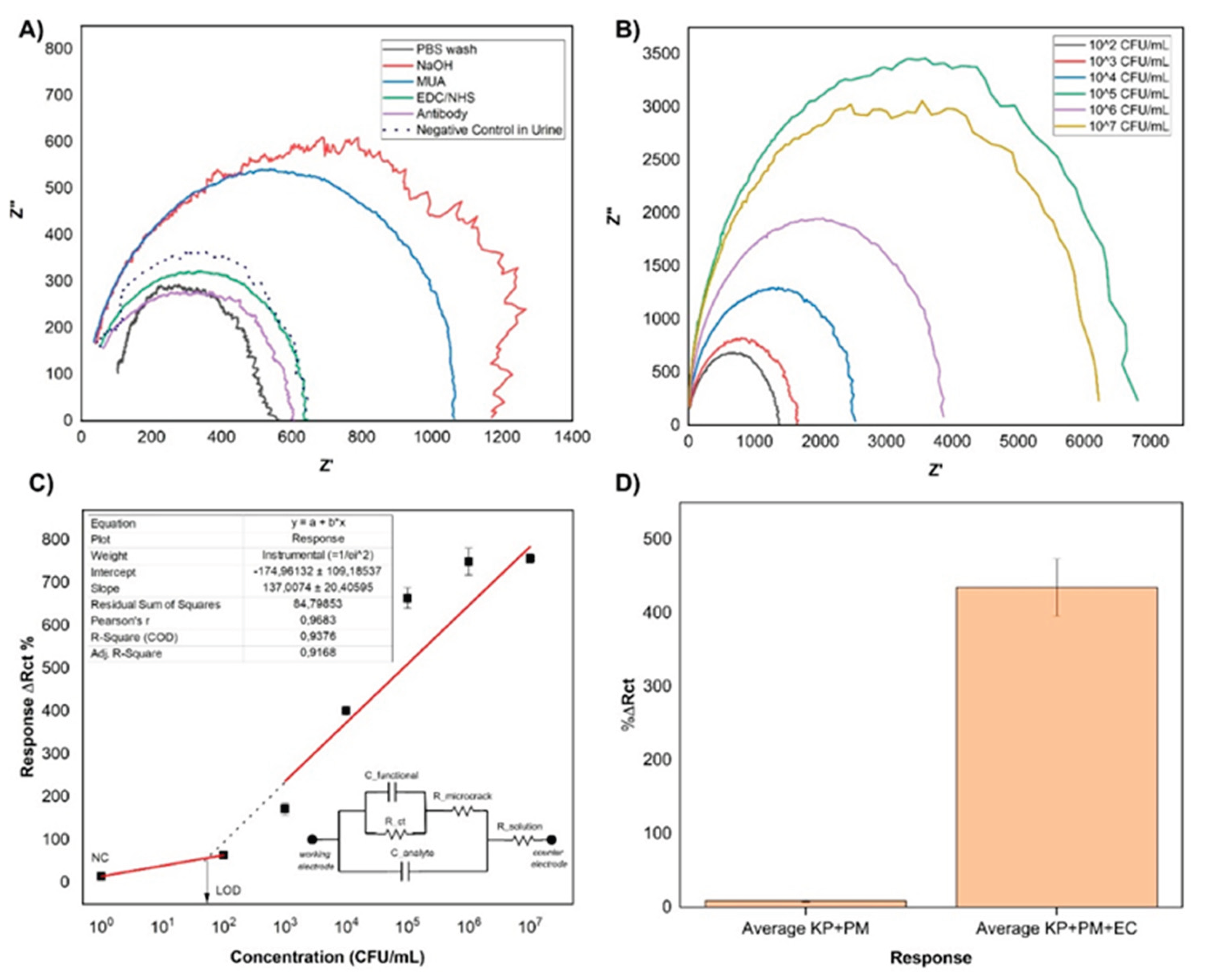
3.2. Analytical Performance of the NiTi Biosensor
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, T.; Liu, L.; Gou, G. Y.; Fang, Z.; Sun, J.; Chen, J.; Cheng, J.; Han, M.; Ma, T.; Liu, C.; Xue, N. Recent Advancements in Physiological, Biochemical, and Multimodal Sensors Based on Flexible Substrates: Strategies, Technologies, and Integrations. ACS Applied Materials & Interfaces CrossRef. 2023, 15(18), 21721–21745. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R. G.; Silva, A. P.; Nunes-Pereira, J. Current on-skin flexible sensors, materials, manufacturing approaches, and study trends for health monitoring: a review. ACS sensors CrossRef. 2024, 9(3), pp. 1104–1133. [Google Scholar] [CrossRef] [PubMed]
- Lawaniya, S. D.; Awasthi, A.; Kuznetsova, I.; Kolesov, V.; Awasthi, K. Polymer Hybrid based Nanomaterials for Next-Generation Sensing Technologies. Sensors and Actuators B: Chemical CrossRef. 2025, 138162. [Google Scholar] [CrossRef]
- Zhou, P.; Zhang, Z.; Mo, F.; Wang, Y. A review of functional hydrogels for flexible chemical sensors. Advanced Sensor Research CrossRef. 2024, 3(3), 2300021. [Google Scholar] [CrossRef]
- Xu, M.; Obodo, D.; Yadavalli, V. K. The design, fabrication, and applications of flexible biosensing devices. Biosensors and Bioelectronics CrossRef. 2019, 124, 96–114. [Google Scholar] [CrossRef]
- Gao, F.; Liu, C.; Zhang, L.; Liu, T.; Wang, Z.; Song, Z.; Cai, H.; Fang, Z.; Chen, J.; Wang, J.; Han, M.; Wang, J.; Lin, K.; Wang, R.; Li, M.; Mei, Q.; Ma, X.; Liang, S.; Gou, G.; Xue, N. Wearable and flexible electrochemical sensors for sweat analysis: a review. Microsystems & Nanoengineering CrossRef. 2023, 9(1), pp. 1–21. [Google Scholar] [CrossRef]
- Han, X.; Lin, X.; Sun, Y.; Huang, L.; Huo, F.; Xie, R. Advancements in Flexible Electronics Fabrication: Film Formation, Patterning, and Interface Optimization for Cutting-Edge Healthcare Monitoring Devices. ACS Applied Materials & Interfaces CrossRef. 2024, 16(41), 54976–55010. [Google Scholar]
- Duerig, T.; Pelton, A.; Stöckel, D. An overview of nitinol medical applications. Materials Science and Engineering A CrossRef. 1999, 273-275, 149–160. [Google Scholar] [CrossRef]
- Patel, S. K.; Behera, B.; Swain, B.; Roshan, R.; Sahoo, D.; Behera, A. A review on NiTi alloys for biomedical applications and their biocompatibility. Materials today: proceedings CrossRef. 2020, 33, pp. 5548–5551. [Google Scholar] [CrossRef]
- Gollerthan, S.; Young, M. L.; Neuking, K.; Ramamurty, U.; Eggeler, G. Direct physical evidence for the back-transformation of stress-induced martensite in the vicinity of cracks in pseudoelastic NiTi shape memory alloys. Acta materialia CrossRef. 2009, 57(19), 5892–5897. [Google Scholar] [CrossRef]
- Gao, A.; Zhang, X.; Peng, X.; Wu, H.; Bai, L.; Jin, W.; Wu, G.; Hang, R.; Chu, P. K. In situ synthesis of Ni (OH) 2/TiO2 composite film on NiTi alloy for non-enzymatic glucose sensing. Sensors and Actuators B: Chemical CrossRef. 2016, 232, 150–157. [Google Scholar] [CrossRef]
- Hang, R.; Liu, Y.; Gao, A.; Bai, L.; Huang, X.; Zhang, X.; Lin, N.; Tang, B.; Chu, P. K. Highly ordered Ni–Ti–O nanotubes for non-enzymatic glucose detection. Materials Science and Engineering: C CrossRef. 2015, 51, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Hui, Z.; Zhang, L.; Ren, G.; Sun, G.; Yu, H. D.; Huang, W. Green flexible electronics: natural materials, fabrication, and applications. Advanced Materials CrossRef. 2023, 35(28), 2211202. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Wang, G.; Yu, D.; Gan, X.; Tian, Q.; Guo, X. Towards “zero waste” extraction of nickel from scrap nickel-based superalloy using magnesium. Journal of Cleaner Production CrossRef. 2020, 262, 121275. [Google Scholar] [CrossRef]
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J. M. Molecular biosensors for electrochemical detection of infectious pathogens in liquid biopsies: current trends and challenges. Sensors CrossRef. 2017, 17(11), 2533, pp. 1–21. [Google Scholar] [CrossRef]
- Dospinescu, V. M.; Tiele, A.; Covington, J. A. Sniffing out urinary tract infection—Diagnosis based on volatile organic compounds and smell profile. Biosensors CrossRef. 2020, 10(8)(83), 2–28. [Google Scholar] [CrossRef]
- Otsuka, K.; Ren, X. Recent developments in the research of shape memory alloys. Intermetallics CrossRef. 1999, 7(5), 511–528. [Google Scholar] [CrossRef]
- Gall, K.; Sehitoglu, H. The role of texture in tension–compression asymmetry in polycrystalline NiTi. International Journal of Plasticity CrossRef. 1999, 15(1), 69–92. [Google Scholar] [CrossRef]
- Chen, B.; Li, L.; Liu, L.; Cao, J. Molecular simulation of adsorption properties of thiol-functionalized titanium dioxide (TiO2) nanostructure for heavy metal ions removal from aqueous solution. Journal of Molecular Liquids CrossRef. 2022, 346, 118281. [Google Scholar]
- Sánchez-Bodón, J.; Andrade del Olmo, J.; Alonso, J. M.; Moreno-Benítez, I.; Vilas-Vilela, J. L.; Pérez-Álvarez, L. Bioactive coatings on titanium: a review on hydroxylation, self-assembled monolayers (SAMs) and surface modification strategies. Polymers CrossRef. 2021, 14(1)(165), 1–30. [Google Scholar] [CrossRef]
- Fontanesi, C.; Tassinari, F.; Parenti, F.; Cohen, H.; Mondal, P. C.; Kiran, V.; Giglia, A.; Pasquali, L.; Naaman, R. New one-step thiol functionalization procedure for Ni by self-assembled monolayers. Langmuir CrossRef. 2015, 31(11), 3546–3552. [Google Scholar] [CrossRef] [PubMed]
- Yeyin, T.; Xiaoya, L.; Peiming, H.; Shusen, P.; Yongcun, M. Protection of nickel by self-assembled monolayers prepared in an aqueous self-emulsifying solution of a novel amphipathic organothiol. RSC advances CrossRef. 2023, 13(7), 4331–4339. [Google Scholar] [CrossRef] [PubMed]
- Mohajeri, M.; Case, R.; Haghgouyan, B.; Lagoudas, D. C.; Castaneda, H. Loading influence on the corrosion assessment during stress-induced martensite reorientation in nickel-titanium SMA. Smart Materials and Structures CrossRef. 2020, 29(3), 035013. Pp. 1–12. [Google Scholar] [CrossRef]
- Instruments, Gamry. Complex impedance in Corrosion. In Basics of electrochemical impedance spectroscop; 2007; Volume 1, pp. 1–30. [Google Scholar]
- Buck, R. P.; Lindner, E. Recommendations for nomenclature of ionselective electrodes (IUPAC Recommendations 1994). Pure and Applied Chemistry CrossRef. 1994, 66(12), 2527–2536. [Google Scholar] [CrossRef]
- Wu, Y.; Ojha, A.; Patriarca, L.; Sehitoglu, H. Fatigue crack growth fundamentals in shape memory alloys. Shape Memory and Superelasticity CrossRef. 2015, 1(1), 18–40. [Google Scholar] [CrossRef]
- Perumal, G.; Selvam, K. T.; Swayne, M.; McCarthy, E.; Babu, A.; Dzhurinskiy, D.; Brabazon, D. Exploring the role of volume energy density in altering microstructure and corrosion behavior of nitinol alloys produced by laser powder bed fusion. Scientific Reports CrossRef. 2025, 15(1)(2055), 1–14. [Google Scholar] [CrossRef]
- Racek, J.; Šittner, P.; Heller, L.; Pilch, J.; Petrenec, M.; Sedlák, P. Corrosion of NiTi wires with cracked oxide layer. Journal of materials engineering and performance CrossRef. 2014, 23(7), pp. 2659–2668. [Google Scholar] [CrossRef]
- Robertson, S. W.; Mehta, A.; Pelton, A. R.; Ritchie, R. O. Evolution of crack-tip transformation zones in superelastic Nitinol subjected to in situ fatigue: A fracture mechanics and synchrotron X-ray microdiffraction analysis. Acta Materialia CrossRef. 2007, 55(18), 6198–6207. [Google Scholar] [CrossRef]
- Soysaldı, F.; Dincyurek Ekici, D.; Soylu, M. Ç.; Mutlugun, E. Electrochemical and Optical Multi-Detection of Escherichia coli Through Magneto-Optic Nanoparticles: A Pencil-on-Paper Biosensor. Biosensors CrossRef. 2024, 14(12)(603), 1–17. [Google Scholar] [CrossRef]
- Keser, K.; Soylu, M. Ç. Simple, Specific, and Ultra-Sensitive Arsenic Detection in Real Drinking Water Samples Using an Impedimetric Green Sensor with Dimercaprol-Doped Solid Electrolyte. Electroanalysis CrossRef. 2025, 37(1), e202400253. [Google Scholar] [CrossRef]
- Zhang, S.; Pang, X.; Wang, Y.; Gao, K. Corrosion behavior of steel with different microstructures under various elastic loading conditions. Corrosion science CrossRef. 2013, 75, 293–299. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, Y.; Jia, C.; Chang, H.; Zhang, W.; Bai, Y.; Li, S.; Zhang, L. C.; Yuan, W. Corrosion behavior of laser powder bed fusion additive manufacturing produced TiNi alloy by micro-arc oxidation. npj Materials Degradation CrossRef. 2024, 8(1)(13), 1–13. [Google Scholar] [CrossRef]
- Sun, T.; Wang, M.; Lee, W. C. Surface characteristics, properties and in vitro biological assessment of a NiTi shape memory alloy after high temperature heat treatment or surface H2O2-oxidation: a comparative study. Materials Chemistry and Physics CrossRef. 2011, 130(1-2), 45–58. [Google Scholar] [CrossRef]
- Chu, C. L.; Hu, T.; Zhou, J.; Pu, Y. P.; Yin, L. H.; Dong, Y. S.; Lin, P. H.; Chung, J. C. Y.; Chu, P. K. Effects of H2O2 pretreatment on surface characteristics and bioactivity of NaOH-treated NiTi shape memory alloy. Transactions of Nonferrous Metals Society of China CrossRef. 2006, 16(6), 1295–1300. [Google Scholar] [CrossRef]
- Love, J. C.; Estroff, L. A.; Kriebel, J. K.; Nuzzo, R. G.; Whitesides, G. M. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chemical reviews CrossRef. 2005, 105(4), 1103–1170. [Google Scholar] [CrossRef]
- Mani, G.; Porter, D.; Grove, K.; Collins, S.; Ornberg, A.; Shulfer, R. Surface finishing of N itinol for implantable medical devices: A review. Journal of Biomedical Materials Research Part B: Applied Biomaterials CrossRef. 2022, 110(12), 2763–2778. [Google Scholar] [CrossRef]
- Sławski, S.; Kciuk, M.; Klein, W. Change in electrical resistance of SMA (NiTi) wires during cyclic stretching. Sensors CrossRef. 2022, 22(9)(3584), 1–19. [Google Scholar] [CrossRef]
- Perinetti, G.; Contardo, L.; Ceschi, M.; Antoniolli, F.; Franchi, L.; Baccetti, T.; Di Lenarda, R. Surface corrosion and fracture resistance of two nickel–titanium-based archwires induced by fluoride, pH, and thermocycling. An in vitro comparative study. The European Journal of Orthodontics CrossRef. 2012, 34(1), 1–9. [Google Scholar] [CrossRef]
- Ghazal, A. R. A.; Hajeer, M. Y.; Al-Sabbagh, R.; Alghoraibi, I.; Aldiry, A. An evaluation of two types of nickel-titanium wires in terms of micromorphology and nickel ions’ release following oral environment exposure. In Progress in orthodontics; CrossRef; PubMed, 2015; Volume 16(1), 9, pp. 1–8. [Google Scholar]
- Waitz, T.; Spišák, D.; Hafner, J.; Karnthaler, H. P. Size-dependent martensitic transformation path causing atomic-scale twinning of nanocrystalline NiTi shape memory alloys. Europhysics Letters CrossRef. 2005, 71(1)(98), 98–103. [Google Scholar] [CrossRef]
- Fu, Y. Q.; Zhang, S.; Wu, M. J.; Huang, W. M.; Du, H. J.; Luo, J. K.; Flewitt, A. J.; Milne, W. I. On the lower thickness boundary of sputtered TiNi films for shape memory application. Thin Solid Films CrossRef. 2006, 515(1), pp. 80–86. [Google Scholar] [CrossRef]
- Waitz, T.; Antretter, T.; Fischer, F. D.; Karnthaler, H. P. Size effects on martensitic phase transformations in nanocrystalline NiTi shape memory alloys. Materials Science and Technology CrossRef. 2008, 24(8), 934–940. [Google Scholar] [CrossRef]
- Shoute, L. C.; Abdelrasoul, G. N.; Ma, Y.; Duarte, P. A.; Edwards, C.; Zhuo, R.; Zeng, J.; Feng, Y.; Charlton, C. L.; Kanji, J. N.; Babiuk, S.; Chen, J. Label-free impedimetric immunosensor for point-of-care detection of COVID-19 antibodies. Microsystems & Nanoengineering CrossRef. 2023, 9(1)(3), 1–16. [Google Scholar]
- Bard, A. J.; Faulkner, L. R.; White, H. S. Electrochemical methods: fundamentals and applications.; John Wiley & Sons, 2022. [Google Scholar]
- Zhang, H.; Hu, H.; Li, Y.; Wang, J.; Ma, L. A ferrocene-based hydrogel as flexible electrochemical biosensor for oxidative stress detection and antioxidation treatment. Biosensors and Bioelectronics CrossRef. 2024, 248, 115997. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Lee, S. N.; Shin, M. K.; Kim, H. W.; Choi, H. K.; Lee, T.; Choi, J. W. Flexible electrochemical glucose biosensor based on GOx/gold/MoS2/gold nanofilm on the polymer electrode. Biosensors and Bioelectronics CrossRef. 2019, 140(111343), 1–7. [Google Scholar] [CrossRef]
- You, Z.; Qiu, Q.; Chen, H.; Feng, Y.; Wang, X.; Wang, Y.; Ying, Y. Laser-induced noble metal nanoparticle-graphene composites enabled flexible biosensor for pathogen detection. Biosensors and Bioelectronics CrossRef. 2020, 150, 111896. [Google Scholar] [CrossRef]
- Xu, M.; Yadavalli, V. K. Flexible biosensors for the impedimetric detection of protein targets using silk-conductive polymer biocomposites. Acs Sensors CrossRef. 2019, 4(4), 1040–1047. [Google Scholar] [CrossRef]
- Lamanna, L.; Rizzi, F.; Bhethanabotla, V. R.; De Vittorio, M. Conformable surface acoustic wave biosensor for E-coli fabricated on PEN plastic film. Biosensors and Bioelectronics CrossRef. 2020, 163(112164), 1–10. [Google Scholar] [CrossRef]
- Tian, B.; Zhang, J.; Zhou, S.; Liu, C.; Wang, Q.; Liu, Y.; Lu, M.; Sun, G.; Wang, C.; Gu, B. Silica interlayer-protected colorimetric-fluorescent dual-signal microbial tag mediate lateral flow immunoassay for flexible and ultrasensitive detection of sepsis biomarkers. Chemical Engineering Journal CrossRef. 2025, 163557. [Google Scholar] [CrossRef]
- Jampasa, S.; Sangthong, N.; Ozer, T.; Traiopop, S.; Ngamrojanavanich, N.; Panphut, W.; Chailapakul, O.; Kaneta, T.; Wonsawat, W. Silver-decorated nanobeads for high-performance and flexible analysis of Shigella dysenteriae utilizing a paper-based aptasensor. Bioelectrochemistry CrossRef. 2025, 109140. [Google Scholar] [CrossRef]
- Wang, R.; Zhai, Q.; An, T.; Gong, S.; Cheng, W. Stretchable gold fiber-based wearable textile electrochemical biosensor for lactate monitoring in sweat. Talanta CrossRef. 2021, 222(121484), 1–8. [Google Scholar] [CrossRef]
- Lim, T.; Zhang, H. Multilayer carbon nanotube/gold nanoparticle composites on gallium-based liquid metals for electrochemical biosensing. ACS Applied Nano Materials CrossRef. 2021, 4(11), 12690–12701. [Google Scholar] [CrossRef]
- Pan, T. M.; Chen, C. H.; Weng, W. C. Rapid and label-free detection of BNP and NT-proBNP in human serum using Ti-doped MoTex film-based extended-gate FET biosensors for heart failure diagnosis. Sensors and Actuators B: Chemical CrossRef. 2025, 138922. [Google Scholar] [CrossRef]
- Gaikwad, P. N.; Desai, T. R.; Ghosh, S.; Gurnani, C. Flexible Nanostructured NiS-Based Electrochemical Biosensor for Simultaneous Detection of DNA Nucleobases. ACS omega CrossRef. 2024, 10(3), 2561–2574. [Google Scholar] [CrossRef]
| Sensor Material | Flexibility Type | LOD | Linear Range | Detection Time (min) | Matrix | POCT Suitability | Ref |
| Ferrocene-based PVA/SA/PEI-Fc hydrogel | Polymeric (Hydrogel) | Not reported | Real time | 0-120 µM | PBS; cell culture media (L929, RAW 264.7) | Yes | [46] |
| PDMS/PET–Au/MoS₂–GOx nanofilm | Polymeric (PET) | 0.10 nM | 10–500 nM | 30 min | Glucose in PBS | Yes (Enzymatic) | [47] |
| AuNPs-LIG interdigitated electrode (flexible) | Polyimide substrate | 10² CFU/mL | 10²–10⁸ CFU/mL | 30 min | E. coli in PBS | No (Not target) | [48] |
| Silk fibroin flexible substrate+ PEDOT:PSS+anti-VEGF | Polymeric (organic) | 22-25 pg/mL | 1 pg/mL–10 ng/mL | 15 min | PBS, serum, artificial urine | Yes | [49] |
| AlN thin film on PEN substrate (SAW) | Polymeric (PEN) | 6.5x105 CFU/mL | 106-108 CFU/mL | 90 min | E. coli in water | Moderate (High LOD) | [50] |
| AuNP-SiO₂-QDs nanocomposite on LFIA strip | Polymeric LFIA substrate | IL-6: 4.7 pg /mL; PCT: 29.5 pg /mL | 0.1–100 ng /mL | 15 min | Human plasma (clinical) | Yes (clinically validated) | [51] |
| Ag-decorated magnetic nanobeads on paper-based aptasensor | Paper substrate | DPV: 90; EIS: 8.09 CFU/mL | DPV: 10²–10⁸; EIS: 10¹–10⁹ CFU/mL | 40 min | Food | Yes (on-site NFC-integrated) | [52] |
| Stretchable gold fiber textile | Elastomeric textile | 0.137 mM | 0-30 mM | Real time | Lactate in artificial sweat | Yes (Enzymatic) | [53] |
| CNT/PDDA + AuNP multilayer on Ga-based liquid metal | Intrinsically soft liquid metal core (ultrasoft) | 23 nM | 25 nM–1 µM | Real time | PBS / ACSF (in vitro neurochemical buffer) | Not target | [54] |
| Ti-doped MoTeₓ film EGFET | Thin-film hybrid | 5x10-3 pg/mL | 10-2-105 pg/mL | Real time | BNP in serum | Yes (clinically validated) | [55] |
| NiS/ Ni-foam | Metallic foam | 159 μM (A), 147.6μM (G), 16.8 μM (T), 45.9 μM (C) | 200–1000 μM (A,G); 50–500 μM (T,C) | Real time | Calf thymus DNA | Yes (portable) | [56] |
| Superelastic NiTi alloy | Intrinsic solid-state microcracks | 53 CFU/mL | 103-107 CFU/mL | 45 min | E. coli in sterile human urine | Yes (preliminary) | This work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





