Submitted:
11 December 2025
Posted:
11 December 2025
You are already at the latest version
Abstract
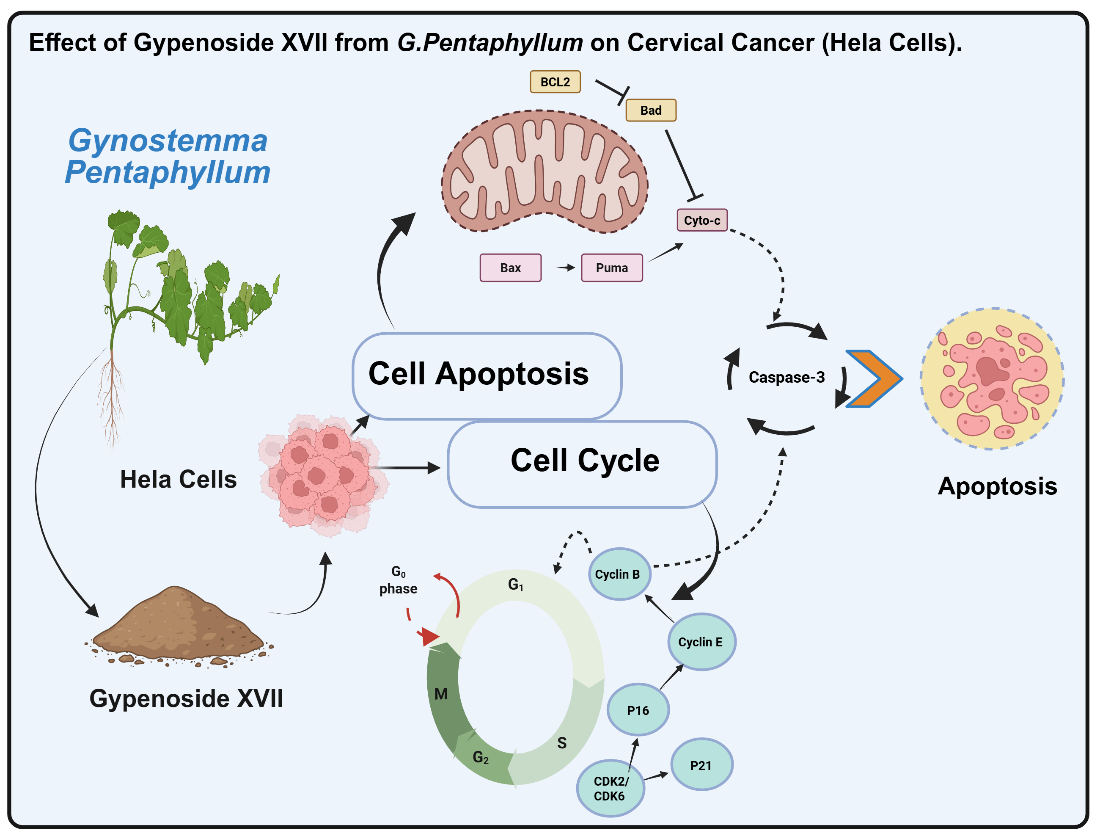
Keywords:
1. Introduction
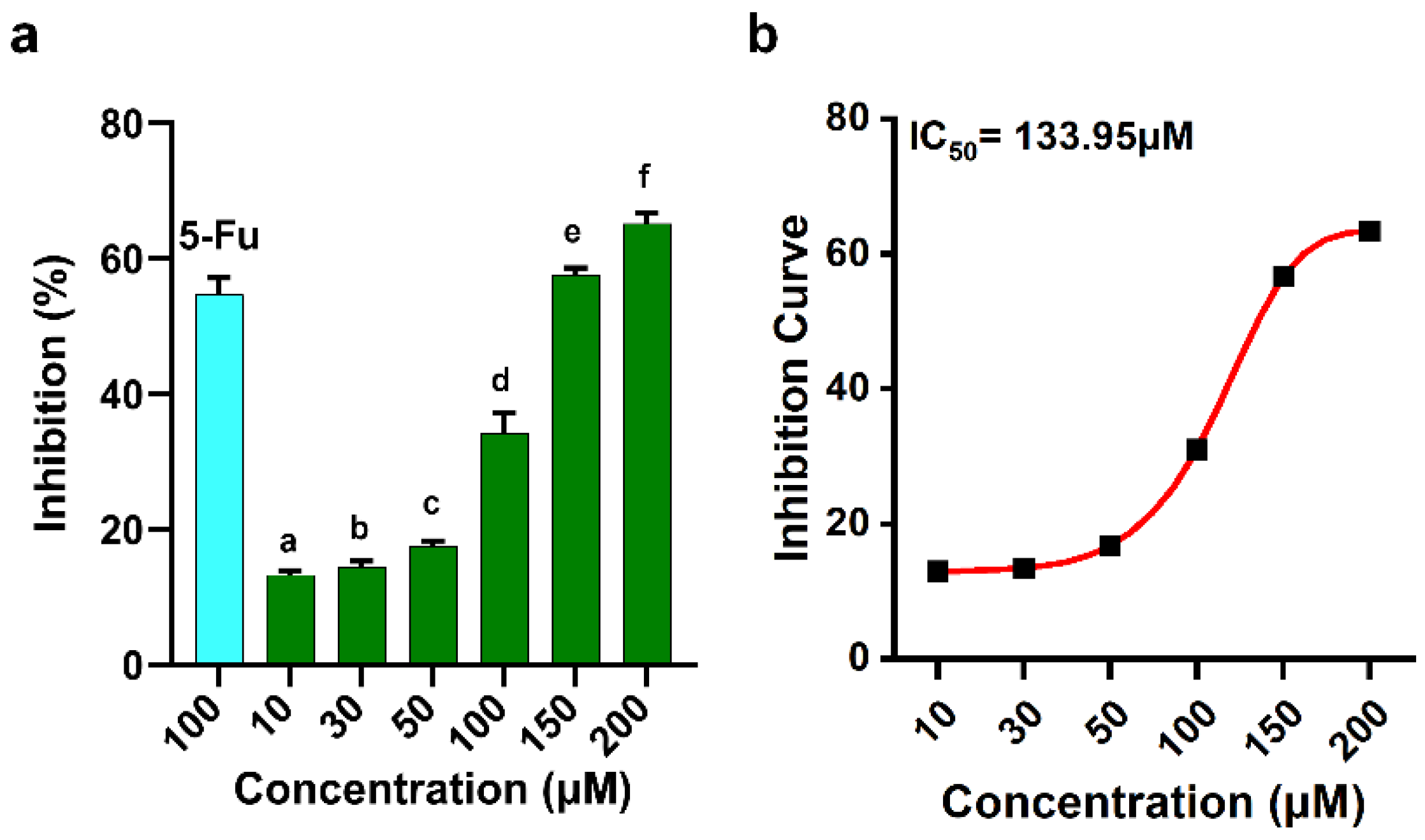
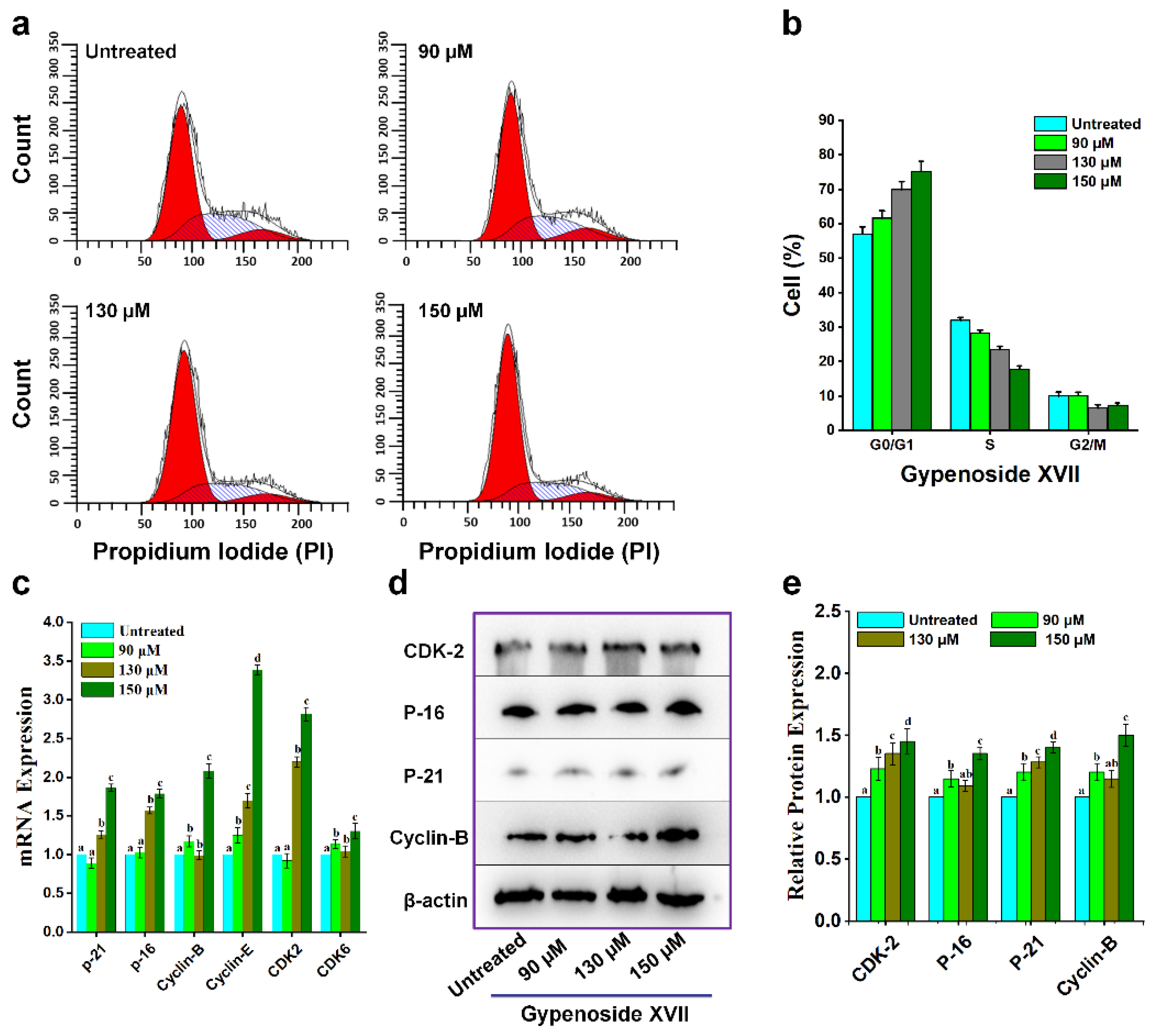
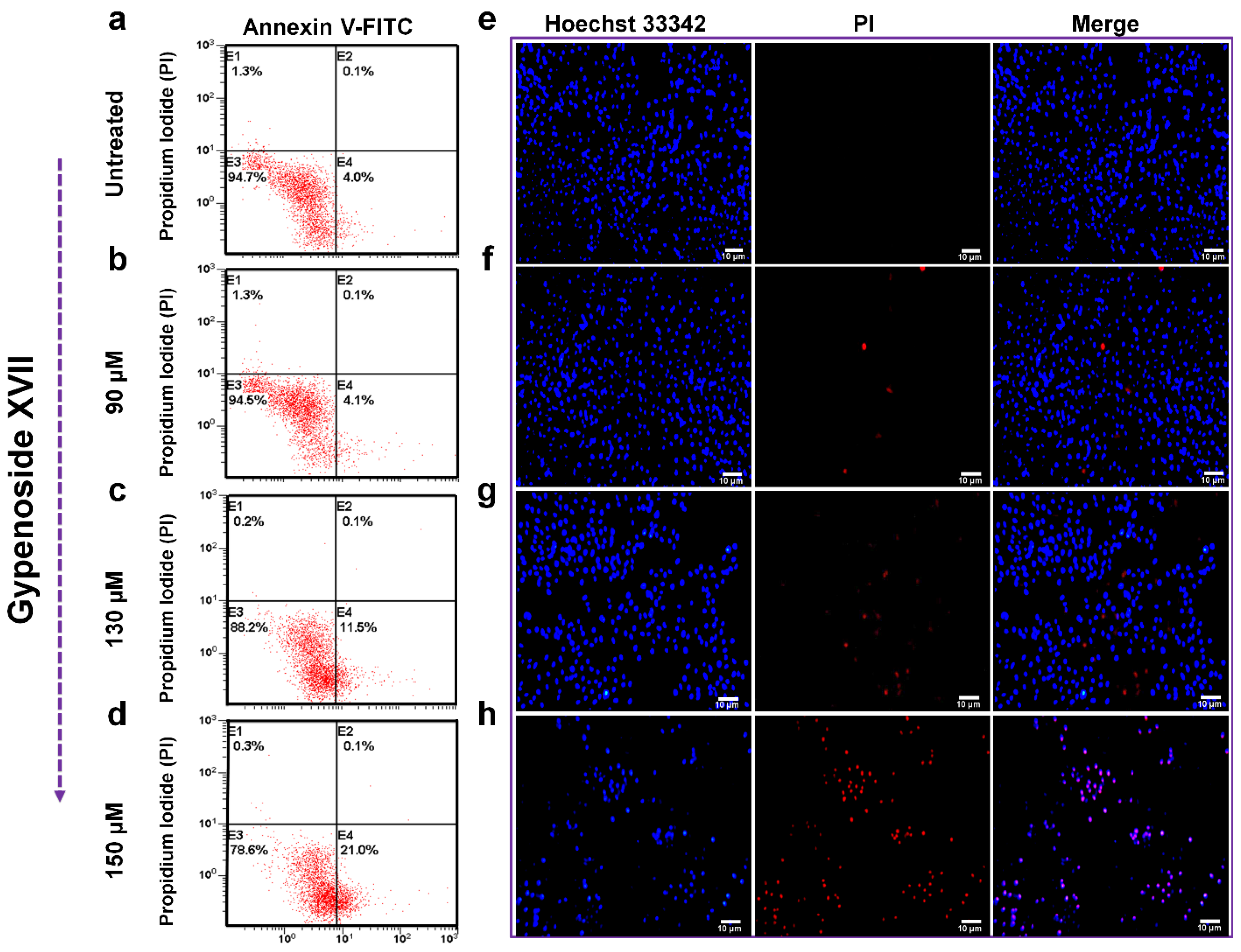
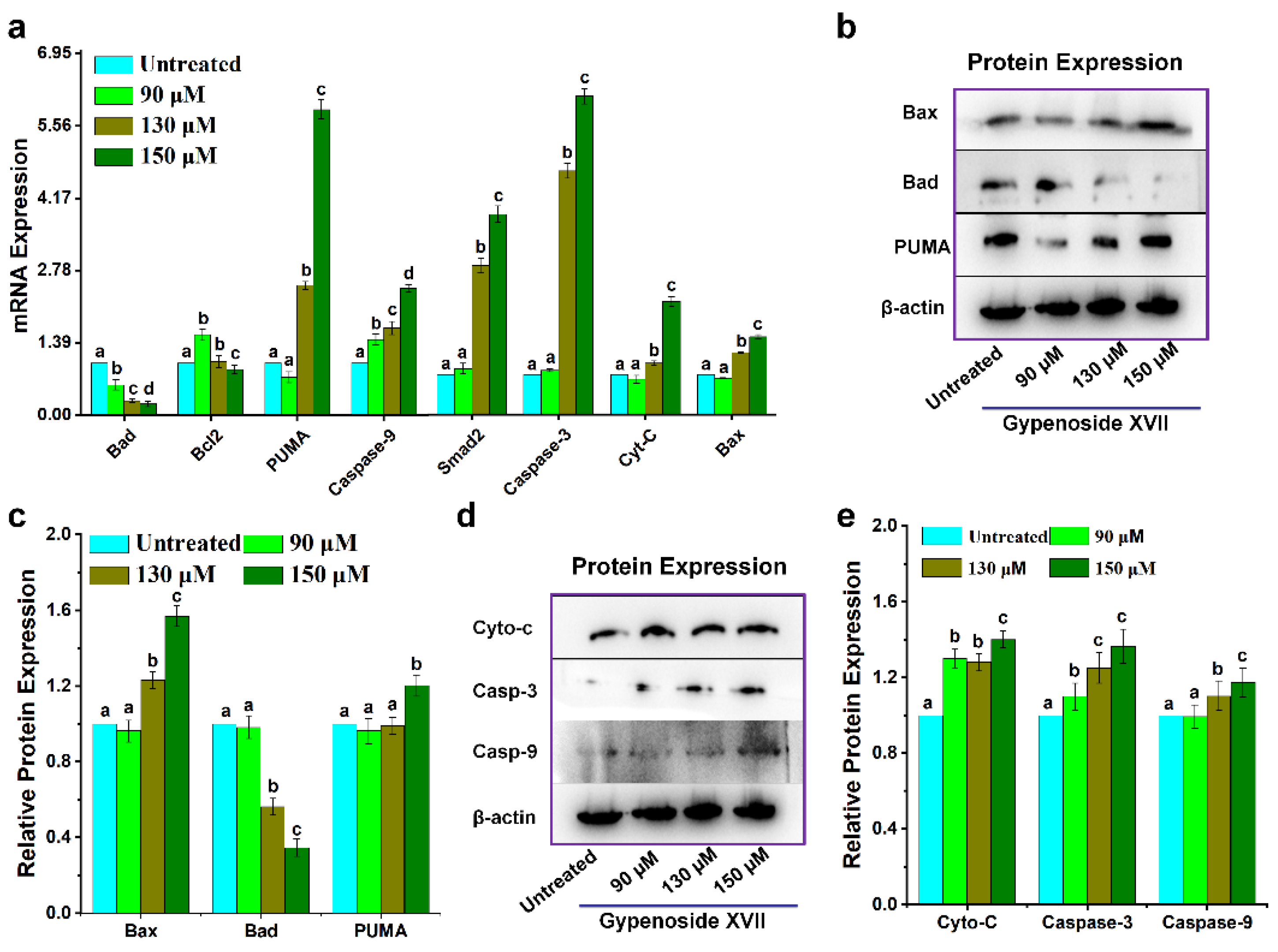
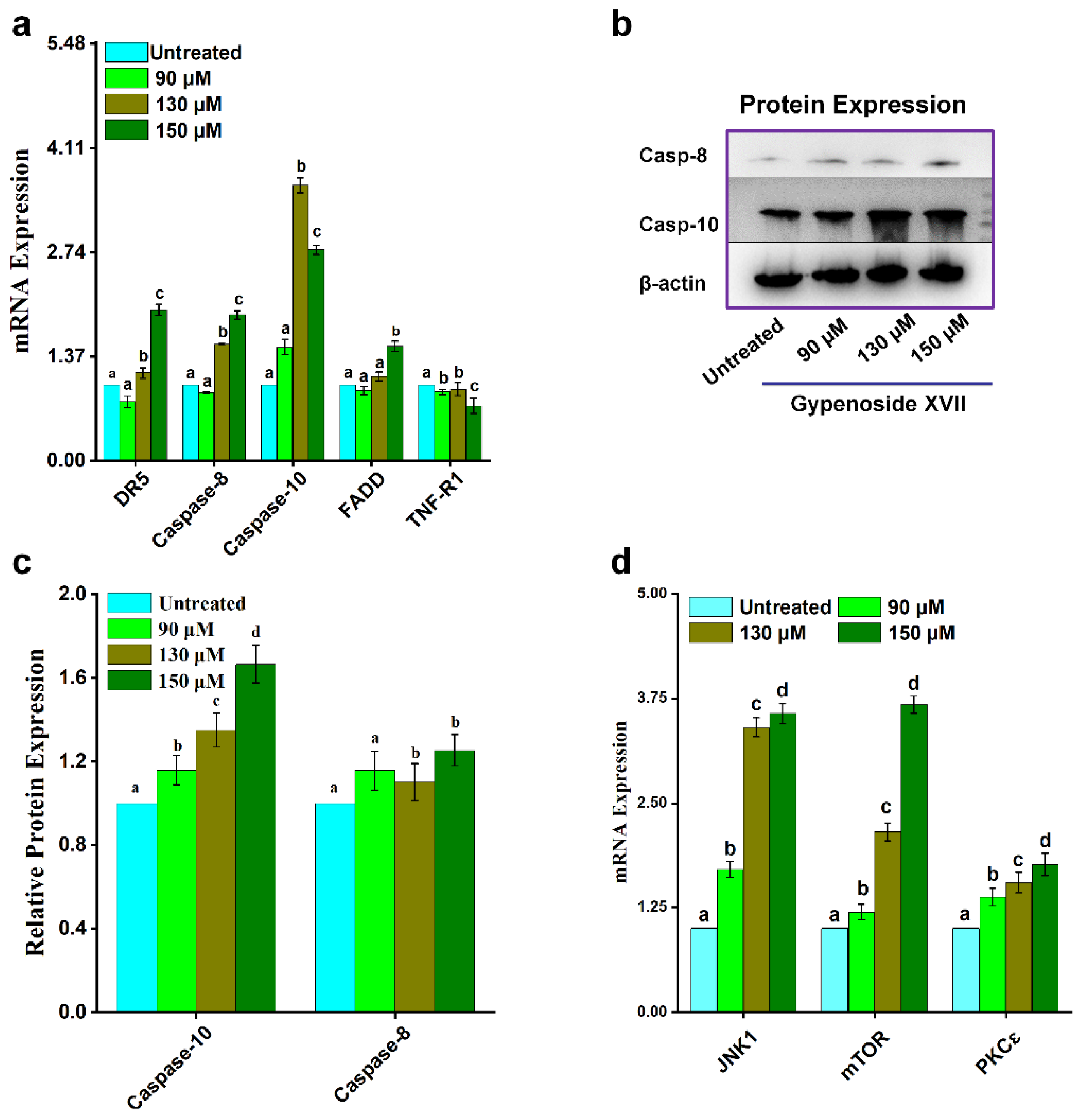
2. Results
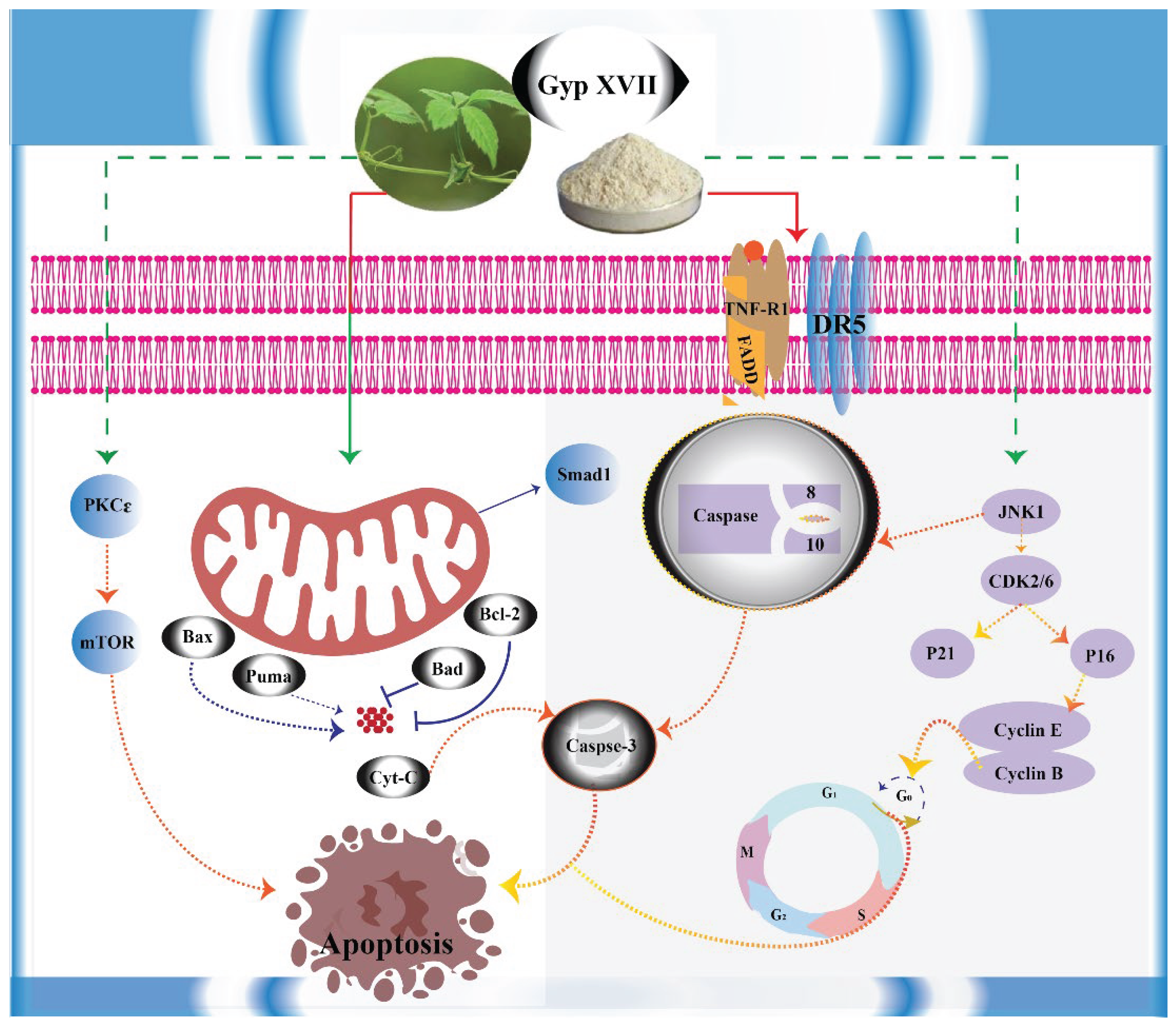
3. Discussion
4. Experimental Method
5. Conclusions
Author Contributions
Funding
Availability of data and materials
Acknowledgments
Conflict of interest
References
- American Cancer Society. (2022). Global cancer facts & figures 4th edition. In: ; 2022. https://www.cancer.org/research/cancer-facts-statistics/global-cancer-facts-and-figures.html.
- Bray, F;, Laversanne, M;, Sung, H;, Ferlay, J;, Siegel, R.L;, Soerjomataram, I;, Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74 (3):229-263. [CrossRef]
- Ma, Y.L;, Zhang, Y.S;, Zhang, F;, Zhang, Y.Y;, Thakur, K;, Zhang, J.G;, Wei, Z.J. Methyl protodioscin from Polygonatum sibiricum inhibits cervical cancer through cell cycle arrest and apoptosis induction. Food and Chemical Toxicology. 2019; 132:110655. [CrossRef]
- Sekar, P.K.C;, Thomas, S.M;, Veerabathiran, R. The future of cervical cancer prevention: advances in research and technology. Explor Med. 2024; 5(3):384-400.
- Ahmad, B;, Khan, S;, Nabi, G;, Gamallat, Y;, Su, P;, Jamalat, Y;, Duan, P;, Yao, L. Natural gypenosides: targeting cancer through different molecular pathways. Cancer Manag Res. 2019;11:2287-2297.
- Bokelmann, J.M. Gynostemma/Jiaogulan (Gynostemma Pentaphyllum). Elsevier eBooks; 2022.
- Quang, H.T;, Thi, P;, Sang, D.N;, Tram, T.T.N;, Huy, N.D;, Dung, T.Q;, The, Q.T.T. Effects of Plant Elicitors on Growth and Gypenosides Biosynthesis in Cell Culture of Giao co lam (Gynostemma pentaphyllum). Molecules. 2022; 27(9):2972. [CrossRef]
- Wang, J;, Yang, J.L;, Zhou, P.P;, Meng, X.H;, Shi, Y.P. Further new gypenosides from Jiaogulan (Gynostemma pentaphyllum). J Agric Food Chem. 2017; 65(29):5926-5934.
- Zhen-xing, WANG;, Jin-mei, YANG;, Zhi-bin, ZHANG;, Xiao-tian ZHU;, Ping XIANG;, Jian, SUN;, Xia-hong, HE. Chemical constituents and biological activity of Gynostemma pentaphyllum: A review. Journal of Southern Agriculture. 2023);54(6):1741-1752.
- Ji, X;, Shen, Y;, Guo, X. Isolation, structures, and bioactivities of the polysaccharides from Gynostemma pentaphyllum (Thunb.) Makino: A review. BioMed research international. 2018; (1):6285134.
- Su, C;, Li, N;, Ren, R;, Wang, Y;, Su, X;, Lu, F;, Ma, X. Progress in the medicinal value, bioactive compounds, and pharmacological activities of Gynostemma pentaphyllum. Molecules. 2021; 26(20):6249. [CrossRef]
- Ahmed, A;, Saleem, M.A;, Saeed, F;, Afzaal, M;, Imran, A;, Nadeem, M;, Ambreen, S;, Imran, M;, Hussain, M;, Al Jbawi, E. Gynostemma pentaphyllum an immortal herb with promising therapeutic potential: a comprehensive review on its phytochemistry and pharmacological perspective. Int J Food Prop. 2023;26(1):808-832.
- Anuradha, B;, Vaibhav, C. Gynostemma pentaphyllum extract market: Trends & growth analysis. 2024; 2032.
- Yuge, N;, Wei, Y;, Junli, L;, Wenbing, Y;, Liangli, Y. Characterization of a Novel Polysaccharide from Tetraploid Gynostemma pentaphyllum Makino. J Agric Food Chem 2013; 61(20):4882-4889.
- Deng, Q;, Yang, X. Protective effects of Gynostemma pentaphyllum polysaccharides on PC12 cells impaired by MPP(+). Int J Biol Macromol. 2014; 69:171-175. [CrossRef]
- Ahn, Y;, Lee, H.S;, Lee, S.H;, Joa, K.L;, Lim, C.Y;, Ahn, Y.J;, Hong, KB. Effects of gypenoside L-containing Gynostemma pentaphyllum extract on fatigue and physical performance: A double-blind, placebo-controlled, randomized trial. Phytotherapy Research. 2023;37(7):3069-3082.
- Li, Y;, Ouyang, Q;, Li, X;, Alolgal, R.N;, Fan, Y;, Sun, Y;, Ma, G. The role of Gynostemma pentaphyllum in regulating hyperlipidemia. The American Journal of Chinese Medicine. 2023;51(04):953-978. [CrossRef]
- Ren, D;, Zhao, Y;, Zheng, Q;, Alim, A;, Yang, X. Immunomodulatory effects of an acidic polysaccharide fraction from herbal Gynostemma pentaphyllum tea in RAW264. 7 cells. Food & Function. 2019;10(4):2186-2197.
- Zhang, Z;, Zhang, W;, Ji, Y.P;, Zhao, Y;, Wang, C.G;, Hu, J.F. Gynostemosides A-E, megastigmane glycosides from Gynostemma pentaphyllum. Phytochemistry. 2010; 71(5-6):693-700. [CrossRef]
- Liu, H.L;, Kao, T.H;, Chen, BH. Determination of carotenoids in the chinese medical herb Jiao-Gu-Lan (Gynostemma Pentaphyllum MAKINO) by liquid chromatography. Chromatographia. 2004;60(7):411-417.
- Huang, S.C;, Hung, C.F;, Wu, W.B;, Chen, BH. Determination of chlorophylls and their derivatives in Gynostemma pentaphyllum Makino by liquid chromatography-mass spectrometry. J Pharm Biomed Anal. 2008; 48(1):105-112.
- Wang, X.W;, Zhang, H.P;, Chen, F;, Wang, X;, Wen, W.Y. A new lignan from Gynostemma pentaphyllum. Chinese Chem Lett. 2009; 20(5):589-591.
- Yang, X;, Zhao, Y;, Yang, Y;, Ruan, Y. Isolation and characterization of immunostimulatory polysaccharide from an herb tea, Gynostemma pentaphyllum Makino. J Agric Food Chem. 2008;56(16):6905-6909. [CrossRef]
- Bai, M.S;, Gao, J.M;, Fan, C;, Yang, S.X;, Zhang, G;, Zheng, C.D. Bioactive dammarane-type triterpenoids derived from the acid hydrolysate of Gynostemma pentaphyllum saponins. Food Chem. 2010; 119(1):306-310.
- Yan, H;, Wang, X;, Wang, Y;, Wang, P;, Xiao, Y. Antiproliferation and anti-migration induced by gypenosides in human colon cancer SW620 and esophageal cancer Eca-109 cells. Hum Exp Toxicol. 2014; 33(5):522-533.
- Yin, Q;, Chen, H;, Ma, R.H;, Zhang, Y.Y;, Liu, M.M;, Thakur, K;, Wei, Z.J. Ginsenoside CK induces apoptosis of human cervical cancer HeLa cells by regulating autophagy and endoplasmic reticulum stress. Food & Function.2021; 12(12): 5301-5316.
- Tavakoli, F;, Jahanban-Esfahlan, R;, Seidi, K;, Jabbari, M;, Behzadi, R;, Pilehvar-Soltanahmadi, Y;, Zarghami, N. Effects of nano-encapsulated curcumin-chrysin on telomerase, MMPs and TIMPs gene expression in mouse B16F10 melanoma tumour model. Artificial cells, nanomedicine, and biotechnology. 2018; 46:75-86. [CrossRef]
- Van Meerloo, J;, Kaspers, G.J.L;, Cloos, J. Cell sensitivity assays: the MTT assay. Cancer cell Cult methods Protoc. 2011;237-245.
- Zhang, Y.Y;, Zhang, F;, Zhang, Y.S;, Thakur, K;, Zhang, J.G;, Liu, Y;, Kan, H;, Wei, Z.J.Mechanism of juglone-induced cell cycle arrest and apoptosis in Ishikawa human endometrial cancer cells. J Agric Food Chem. 2019; 67(26):7378-7389.
- Hussain, S.S;, Zhang, F;, Zhang, Y;, Thakur, K;, Naudhani, M;, Cespedes-Acuña, C.L;, Wei, Z.J.Stevenleaf from Gynostemma pentaphyllum inhibits human hepatoma cell (HepG2) through cell cycle arrest and apoptotic induction. Food Sci Hum Wellnes. 2020; 9(3):295-303.
- Darzynkiewicz, Z. Recent advances in cytometry. Academic Press. 2011.
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2024;256(3):183-194. [CrossRef]
- Thompson, C.B. Apoptosis in the pathogenesis and treatment of disease. Science. 1995; 267(5203):1456-1462.
- Leist, M;, Jäättelä, M. Four deaths and a funeral: from caspases to alternative mechanisms. Nat Rev Mol Cell Biol. 2001; 2(8):589-598. [CrossRef]
- Green, D.R;, Kroemer, G. The pathophysiology of mitochondrial cell death. Science. 2004;305(5684):626-629.
- Massagué, J. G1 cell-cycle control and cancer. Nature. 2004;432(7015):298-306.
- Karimian, A;, Ahmadi, Y;, Yousefi, B. Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage. DNA Repair. 2016; 42:63-71.
- Wang, J;, Liao, A.M;, Thakur, K;, Zhang, J.G;, Huang, J.H;, Wei, Z.J.Licochalcone B extracted from Glycyrrhiza uralensis Fisch induces apoptotic effects in human hepatoma cell HepG2. J Agric Food Chem. 2019; 67(12):3341-3353. [CrossRef]
- Serrano, M;, Hannon, G.J;, Beach, D. A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature. 1993; 366(6456):704-707.
- Ruas, M;, Peters, G.The p16INK4a/CDKN2A tumor suppressor and its relatives. Biochim Biophys Acta (BBA)-Reviews Cancer. 1998; 1378(2):F115-F177.
- Fan, J.P;, Kim, H.S;, Han, G.D. Induction of apoptosis by L-carnitine through regulation of two main pathways in Hepa1c1c 7 cells. Amino Acids. 2009; 36:365-372.
- McCoy, M.K;, Tansey, M.G. TNF signaling inhibition in the CNS: implications for normal brain function and neurodegenerative disease. J Neuroinflammation. 2008; 5:1-13. [CrossRef]
- Siddiqui, W.A;, Ahad, A;, Ahsan, H. The mystery of BCL2 family: Bcl-2 proteins and apoptosis: an update. Arch Toxicol. 2015;89:289-317.
- Pan, Z;, Zhang, X;, Yu, P;, Chen, X;, Lu, P;, Li, M;, Li, D. Cinobufagin induces cell cycle arrest at the G2/M phase and promotes apoptosis in malignant melanoma cells. Frontiers in Oncology. 2019; 9:853. [CrossRef]
- Sun, Y.S;, Thakur, K;, Hu, F;, Zhang, J.G;, Wei, Z.J. Icariside II inhibits tumorigenesis via inhibiting AKT/Cyclin E/CDK 2 pathway and activating mitochondria-dependent pathway. Pharmacol Res. 2020; 152:104616.
- Wang, J;, Zhang, Y.S;, Thakur, K;, Hussain, S.S;, Zhang, J.G;, Xiao, G.R;, Wei, Z.J. Licochalcone A from licorice root, an inhibitor of human hepatoma cell growth via induction of cell apoptosis and cell cycle arrest. Food Chem Toxicol. 2018;120:407-417. [CrossRef]
| Genes | Primers | Sequences | Genes | Primers | Sequences |
|---|---|---|---|---|---|
| Caspase-3 | Forward | TGGACTGTGGCATTGAGAA | FADD | Forward | GGGAAGAAGACCTGTGTGCA |
| Reverse | CAGGTGCTGTGGAGTATGCA | Reverse | ATTCTCAGTGACTCCCGCAC | ||
| p-21 | Forward | GCGGAACAAGGAGTCAGACA | Cyclin-E | Forward | GGATTATTGCACCATCCAGAGGCT |
| Reverse | GAACCAGGACACATGGGGAG | Reverse | CTTGTGTCGCCATATACCGGTCAA | ||
| p-16 | Forward | CTTCCTGGACACGCTGGT | Cyclin-B1 | Forward | CTGCTGGGTGTAGGTCCTTG |
| Reverse | ATCTATGCGGGCATGGTTACT | Reverse | TGCCATGTTGATCTTCGCCT | ||
| Cyt-c | Forward | AGGAGGTGGAGGCAAAGGTA | PKCɛ | Forward | TGCCCCACAAGTTCGGTATC |
| Reverse | ATATTTGCACAGTGAAACATAGGA | Reverse | GCCGCTGTTGGTGATTTTGT | ||
| TNF-R1 | Forward | CTCTCCCCTCCTCTCTGCTT | mTOR | Forward | TTATGGGCAGCAACGGACAT |
| Reverse | GGGTTGAGACTCGGGCATAG | Reverse | CTTCTCCCTGTAGTCCCGGA | ||
| Caspase-10 | Forward | CAGGGGCAGGAAGAGAACAG | JNK1 | Forward | ACATTGAGCAGAGCAGGCAT |
| Reverse | ACTAGGAAACGCTGCTCCAC | Reverse | GTCAGGAGCAGCACCATTCT | ||
| Caspase-8 | Forward | TATCCCGGATGGCTGACT | Bcl-2 | Forward | GGAGCGTCAACAGGGAGATG |
| Reverse | GACATCGCTCTCAGGCTC | Reverse | GATGCCGGTTCAGGTACTCAG | ||
| Smad1 | Forward | GGCCTCACGTCATCTACTGCC | Puma | Forward | ATGCCTGCCTCACCTTCATC |
| Reverse | GGGTTACGGAA-GCGTGGCAGCAT | Reverse | TCAGCCAAAATCTCCCACCC | ||
| CDK-6 | Forward | CGGGATCCACCATGGAGAAGGACGGCCTG | Bax | Forward | AGTAACATGGAGCTGCAGAGG |
| Reverse | CGGATCCATTGCTCAGGCTGTATTCAGCTCCGA | Reverse | ATGGTTCTGATCAGTTCCGG | ||
| CDK2 | Forward | CTTTGGAGTCCCTGTCCGTA | Bad | Forward | AGAGTTTGAGCCGAGTGAGC |
| Reverse | CGAAAGATCCGGAAGAGTTG | Reverse | CATCCCTTCGTCGTCCTCC | ||
| DR5 | Forward | CGTCCGCATAAATCAGCACG | β-actin | Forward | TGTGATGGTGGGAATGGGTCAG |
| Reverse | TCTGTCCCCGTTGTTCCATG | Reverse | TTTGATGTCACGCACGATTTCC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




