Submitted:
10 December 2025
Posted:
12 December 2025
Read the latest preprint version here
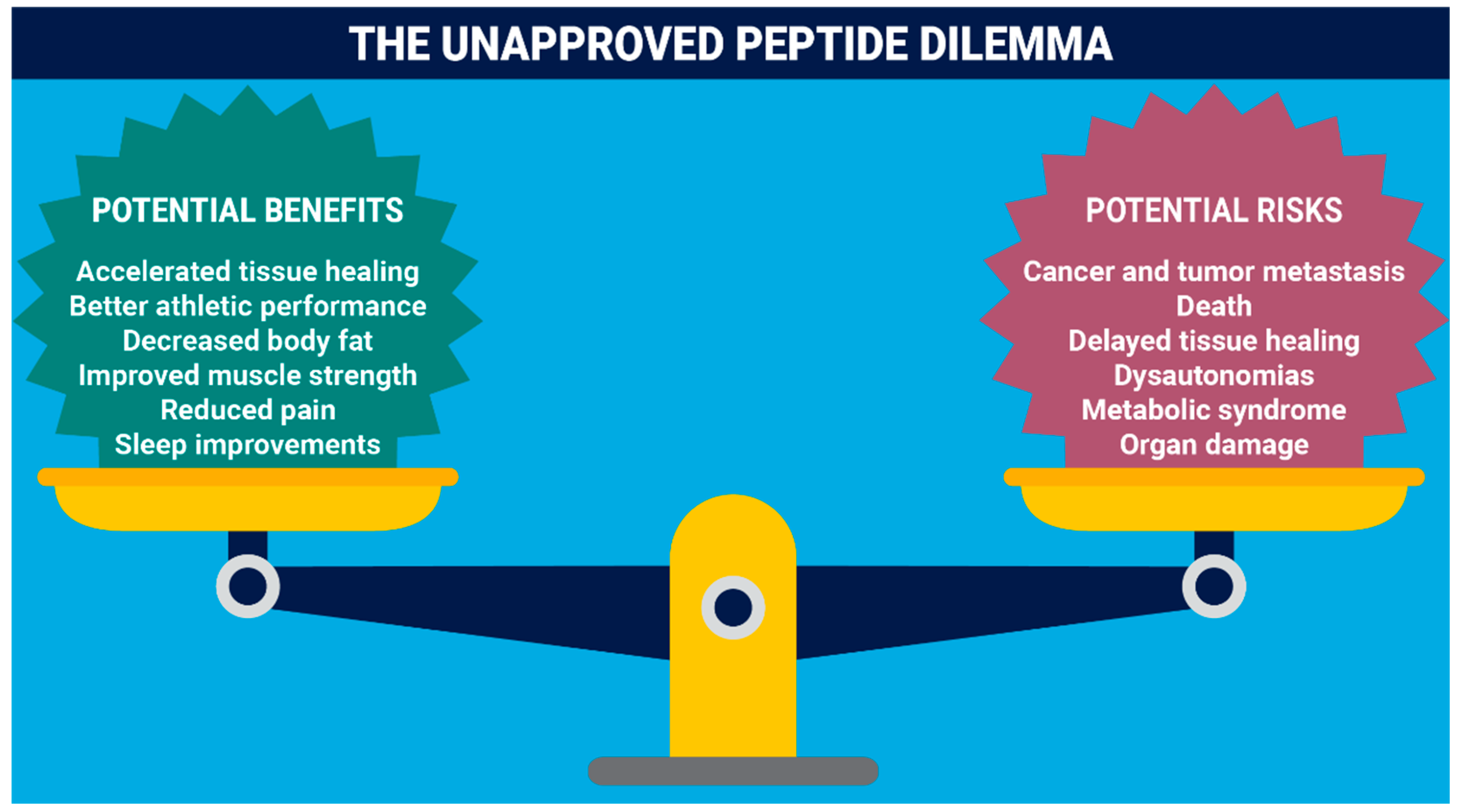
Abstract
Keywords:
1. Introduction
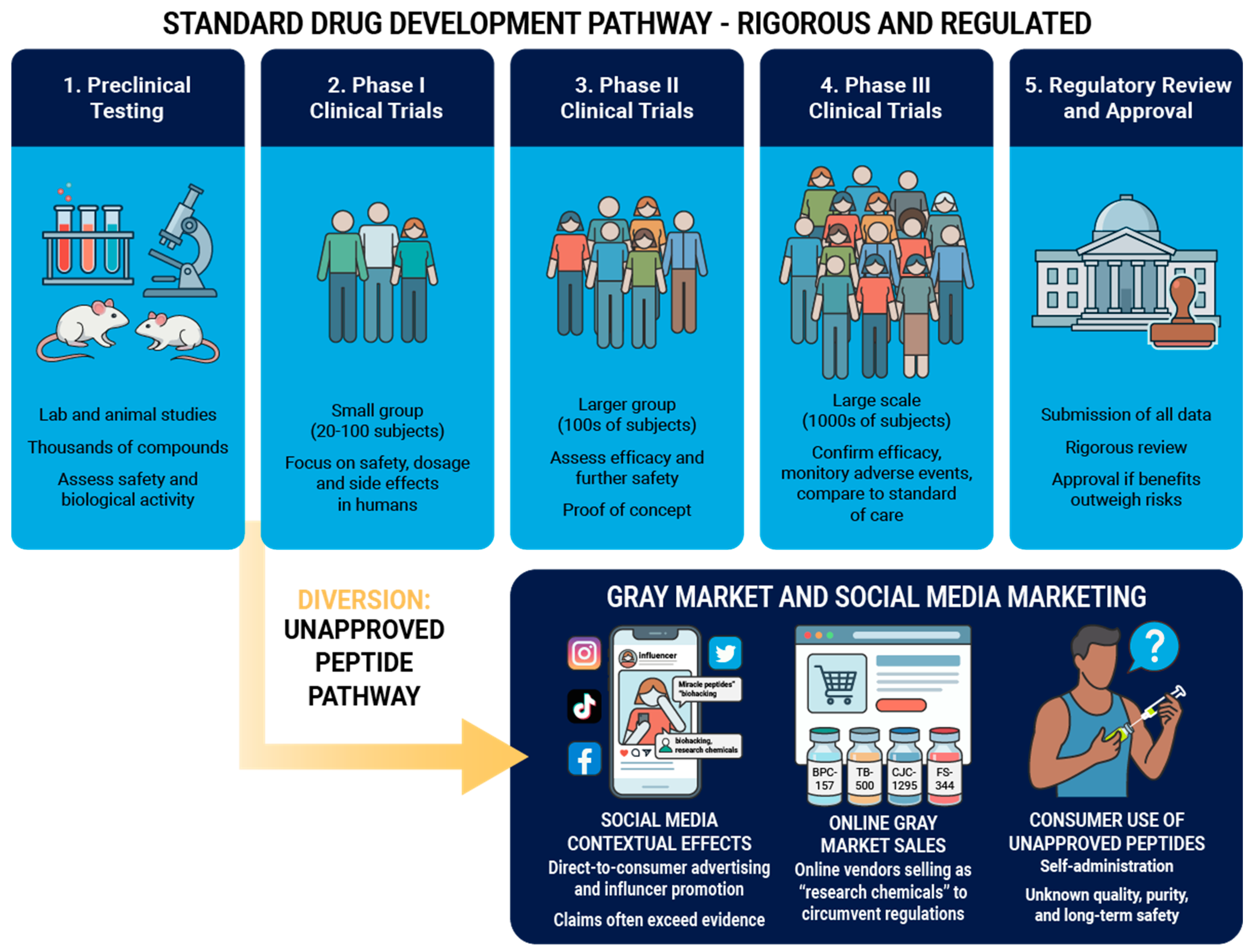
2. Peptide Synthesis, Manufacturing, and Compounding
2.1. Peptide Synthesis and Manufacturing
2.2. Compounding of Approved Peptides
3. Peptides with Potential Use in Sports Medicine
3.1. AOD-9604
3.2. BPC-157
3.3. CJC-1295
3.5. FS-344
3.6. GHK-Cu
3.7. Ipamorelin
3.8. MOTS-C
3.9. Sermorelin and Tesamorelin
3.10. SS-31
3.11. TB-500
4. The Placebo and Contextual Effects of Peptides
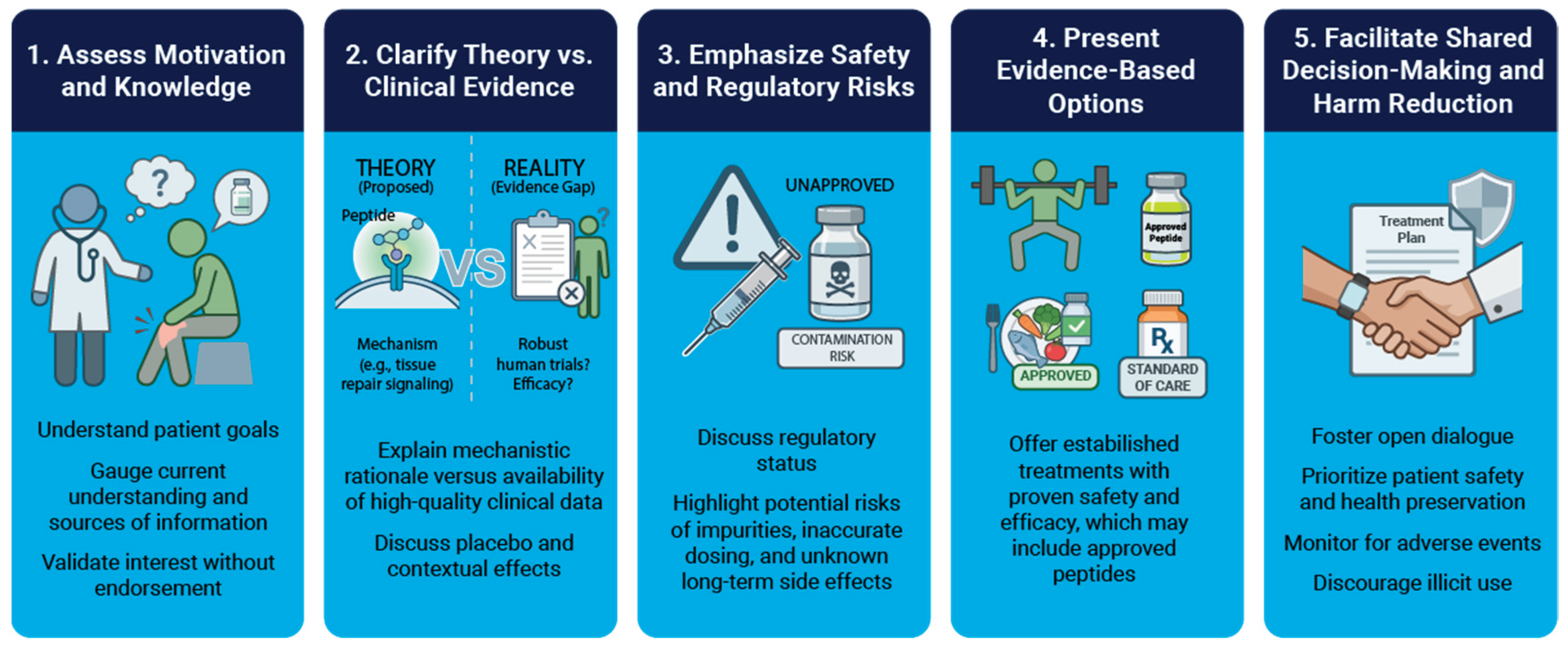
5. Discussing Peptide Therapy with Patients
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | Adeno-associated virus |
| AOD-9604 | Anti obesity drug 9604 |
| ARTG | Australian Register of Therapeutic Goods |
| BMAC | Bone marrow aspirate concentrate |
| BPC-157 | Body protection compound 157 |
| cGMP | Current good manufacturing practice |
| CMV | Cytomegalovirus |
| EMA | European Medicines Agency |
| ESWT | Extracorporeal shock wave therapy |
| FDA | U.S. Food and Drug Administration |
| FS-344 | Follistatin 344 |
| GHK-Cu | Glycyl L histidyl L lysine copper (copper peptide complex) |
| GHRH | Growth hormone releasing hormone |
| GHSR1 | Ghrelin/growth hormone secretagogue receptor 1 |
| GLP-1RA | Glucagon like peptide 1 receptor agonist |
| hGH | Human growth hormone |
| HIV | Human immunodeficiency virus |
| IGF-1 | Insulin like growth factor 1 |
| MOTS-c | Mitochondrial ORF of the 12S rRNA type c |
| PDE5 | Phosphodiesterase type 5 |
| PPP | Platelet poor plasma |
| PRP | Platelet rich plasma |
| PTH | Parathyroid hormone |
| SS-31 | Elamipretide |
| SVF | Stromovascular fraction (of adipose tissue) |
| TB-500 | Thymosin beta 4 fragment |
| TGA | Therapeutic Goods Administration (Australia) |
| USP | United States Pharmacopeia |
| VEGF | Vascular endothelial growth factor |
References
- Otvos L, Wade JD. Big peptide drugs in a small molecule world. Front Chem. 2023;11:1302169. [CrossRef]
- Wang L, Wang N, Zhang W, Cheng X, Yan Z, Shao G, et al. Therapeutic peptides: current applications and future directions. Signal Transduct Target Ther. 2022;7:48. [CrossRef]
- Bonnet A-L, Aboishava L, Mannstadt M. Advances in Parathyroid Hormone-based medicines. J Bone Miner Res. 2025;40:1195–206. [CrossRef]
- Vasireddi N, Hahamyan H, Salata MJ, Karns M, Calcei JG, Voos JE, et al. Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review. HSS J. 2025;21:485–95. [CrossRef]
- Turnock DL, Gibbs DN. Click, click, buy: The market for novel synthetic peptide hormones on mainstream e-commerce platforms in the UK. Perform Enhanc Heal. 2023;11:100251.
- Sharma A, Kumar A, Torre BG de la, Albericio F. Liquid-Phase Peptide Synthesis (LPPS): A Third Wave for the Preparation of Peptides. Chem Rev. 2022;122:13516–46. [CrossRef]
- Janvier S, Cheyns K, Canfyn M, Goscinny S, Spiegeleer BD, Vanhee C, et al. Impurity profiling of the most frequently encountered falsified polypeptide drugs on the Belgian market. Talanta. 2018;188:795–807. [CrossRef]
- D’Hondt M, Bracke N, Taevernier L, Gevaert B, Verbeke F, Wynendaele E, et al. Related impurities in peptide medicines. J Pharm Biomed Anal. 2014;101:2–30. [CrossRef]
- Erckes V, Streuli A, Rendueles LC, Krämer SD, Steuer C. Towards a Consensus for the Analysis and Exchange of TFA as a Counterion in Synthetic Peptides and Its Influence on Membrane Permeation. Pharmaceuticals. 2025;18:1163. [CrossRef]
- Cornish J, Callon KE, Lin CQ-X, Xiao CL, Mulvey TB, Cooper GJS, et al. Trifluoroacetate, a contaminant in purified proteins, inhibits proliferation of osteoblasts and chondrocytes. Am J Physiol-Endocrinol Metab. 1999;277:E779–83. [CrossRef]
- Janvier S, Sutter ED, Wynendaele E, Spiegeleer BD, Vanhee C, Deconinck E. Analysis of illegal peptide drugs via HILIC-DAD-MS. Talanta. 2017;174:562–71. [CrossRef]
- Høj LJ, Rasmussen BS, Dalsgaard PW, Linnet K. Analysis of seized peptide and protein-based doping agents using four complimentary methods: Liquid chromatography coupled with time of flight mass spectrometry, liquid chromatography–ultraviolet, Bradford, and immunoassays. Drug Test Anal. 2021;13:1457–63. [CrossRef]
- Sood N, Garg R. Global Rise of Compounded Weight-Loss Medicines: A Worrisome Trend. J Endocr Soc. 2025;9:bvaf084. [CrossRef]
- DiStefano MJ, Dardouri M, Moore GD, Saseen JJ, Nair KV. Compounded glucagon-like peptide-1 receptor agonists for weight loss: the direct-to-consumer market in Colorado. J Pharm Polic Pr. 2025;18:2441220. [CrossRef]
- Carvalho M, Almeida IF. The Role of Pharmaceutical Compounding in Promoting Medication Adherence. Pharmaceuticals. 2022;15:1091. [CrossRef]
- Yoffe A, Liu J, Smith G, Chisholm O. Regulatory Reform Outcomes and Accelerated Regulatory Pathways for New Prescription Medicines in Australia. Ther Innov Regul Sci. 2023;57:271–86. [CrossRef]
- Cox HD, Smeal SJ, Hughes CM, Cox JE, Eichner D. Detection and in vitro metabolism of AOD9604. Drug Test Anal. 2015;7:31–8. [CrossRef]
- Stier. Safety and Tolerability of the Hexadecapeptide AOD9604 in Humans. J Endocrinol Metab. 2013;
- Heffernan M, Summers RJ, Thorburn A, Ogru E, Gianello R, Jiang W-J, et al. The Effects of Human GH and Its Lipolytic Fragment (AOD9604) on Lipid Metabolism Following Chronic Treatment in Obese Mice andβ 3-AR Knock-Out Mice. Endocrinology. 2001;142:5182–9. [CrossRef]
- Valentino MA, Lin JE, Waldman SA. Central and Peripheral Molecular Targets for Antiobesity Pharmacotherapy. Clin Pharmacol Ther. 2010;87:652–62. [CrossRef]
- Kwon DR, Park GY. Effect of Intra-articular Injection of AOD9604 with or without Hyaluronic Acid in Rabbit Osteoarthritis Model. Ann Clin Lab Sci. 2015;45:426–32.
- Fedorowski A. Postural orthostatic tachycardia syndrome: clinical presentation, aetiology and management. J Intern Med. 2019;285:352–66. [CrossRef]
- Bhandarkar A, Bhat S, Kapoor N. Effect of GLP-1 receptor agonists on body composition. Curr Opin Endocrinol Diabetes Obes. 2025;32:279–85. [CrossRef]
- Márquez-Cruz M, Kammar-García A, Huerta-Cruz JC, Carrasco-Portugal M del C, Barranco-Garduño LM, Rodríguez-Silverio J, et al. Three- and six-month efficacy and safety of phentermine in a Mexican obese population. Int J Clin Pharmacol Ther. 2021;59:539–48. [CrossRef]
- Apovian CM, Aronne LJ, Bessesen DH, McDonnell ME, Murad MH, Pagotto U, et al. Pharmacological Management of Obesity: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metabolism. 2015;100:342–62.
- Andia I, Maffulli N. Biological Therapies in Regenerative Sports Medicine. Sports Med. 2017;47:807–28. [CrossRef]
- Józwiak M, Bauer M, Kamysz W, Kleczkowska P. Multifunctionality and Possible Medical Application of the BPC 157 Peptide—Literature and Patent Review. Pharmaceuticals. 2025;18:185. [CrossRef]
- Lee E, Padgett B. Intra-Articular Injection of BPC 157 for Multiple Types of Knee Pain. Altern Ther Heal Med. 2021;27:8–13.
- Yang Y, Cao Y. The impact of VEGF on cancer metastasis and systemic disease. Semin Cancer Biol. 2022;86:251–61. [CrossRef]
- Mead MP, Gumucio JP, Awan TM, Mendias CL, Sugg KB. Pathogenesis and management of tendinopathies in sports medicine. Transl Sports Med. 2018;1:5–13. [CrossRef]
- Praet SFE, Ong JH, Purdam C, Welvaert M, Lovell G, Dixon L, et al. Microvascular volume in symptomatic Achilles tendons is associated with VISA-A score. J Sci Med Sport. 2018;21:1185–91. [CrossRef]
- Chung B, Wiley JP. Extracorporeal Shockwave Therapy. Sports Med. 2002;32:851–65.
- Hudgens JL, Sugg KB, Grekin JA, Gumucio JP, Bedi A, Mendias CL. Platelet-Rich Plasma Activates Proinflammatory Signaling Pathways and Induces Oxidative Stress in Tendon Fibroblasts. Am J Sports Med. 2016;44:1931–40. [CrossRef]
- Malliaras P, Barton CJ, Reeves ND, Langberg H. Achilles and Patellar Tendinopathy Loading Programmes. Sports Med. 2013;43:267–86. [CrossRef]
- Mendias CL, Enselman ERS, Olszewski AM, Gumucio JP, Edon DL, Konnaris MA, et al. The Use of Recombinant Human Growth Hormone to Protect Against Muscle Weakness in Patients Undergoing Anterior Cruciate Ligament Reconstruction: A Pilot, Randomized Placebo-Controlled Trial. Am J Sports Med. 2020;48:1916–28. [CrossRef]
- Weber AE, Gallo MC, Bolia IK, Cleary EJ, Schroeder TE, Hatch GFR. Anabolic Androgenic Steroids in Orthopaedic Surgery: Current Concepts and Clinical Applications. Jaaos Global Res Rev. 2022;6:e21.00156. [CrossRef]
- Teichman SL, Neale A, Lawrence B, Gagnon C, Castaigne J-P, Frohman LA. Prolonged Stimulation of Growth Hormone (GH) and Insulin-Like Growth Factor I Secretion by CJC-1295, a Long-Acting Analog of GH-Releasing Hormone, in Healthy Adults. J Clin Endocrinol Metab. 2006;91:799–805. [CrossRef]
- A Study to Evaluate CJC 1295 in HIV Patients With Visceral Obesity. NCT00267527.
- GELATO MC, RITTMASTER RS, PESCOVITZ OH, NICOLETTI MC, NIXON WE, D’AGATA, R, et al. Growth Hormone Responses to Continuous Infusions of Growth Hormone-Releasing Hormone*. J Clin Endocrinol Metab. 1985;61:223–8. [CrossRef]
- Melmed S. Acromegaly pathogenesis and treatment. J Clin Investig. 2009;119:3189–202.
- Arora NK, Donath L, Owen PJ, Miller CT, Saueressig T, Winter F, et al. The Impact of Exercise Prescription Variables on Intervention Outcomes in Musculoskeletal Pain: An Umbrella Review of Systematic Reviews. Sports Med. 2024;54:711–25. [CrossRef]
- Rodino-Klapac LR, Haidet AM, Kota J, Handy C, Kaspar BK, Mendell JR. Inhibition of myostatin with emphasis on follistatin as a therapy for muscle disease. Muscle Nerve. 2009;39:283–96. [CrossRef]
- Dueweke JJ, Awan TM, Mendias CL. Regeneration of Skeletal Muscle After Eccentric Injury. J Sport Rehabil. 2017;26:171–9. [CrossRef]
- Haidet AM, Rizo L, Handy C, Umapathi P, Eagle A, Shilling C, et al. Long-term enhancement of skeletal muscle mass and strength by single gene administration of myostatin inhibitors. Proc Natl Acad Sci USA. 2008;105:4318–22. [CrossRef]
- Kota J, Handy CR, Haidet AM, Montgomery CL, Eagle A, Rodino-Klapac LR, et al. Follistatin Gene Delivery Enhances Muscle Growth and Strength in Nonhuman Primates. Sci Transl Med. 2009;1:6ra15. [CrossRef]
- Al-Zaidy SA, Sahenk Z, Rodino-Klapac LR, Kaspar B, Mendell JR. Follistatin Gene Therapy Improves Ambulation in Becker Muscular Dystrophy. J Neuromuscul Dis. 2015;2:185–92. [CrossRef]
- Phillips DJ, Jones KL, McGaw DJ, Groome NP, Smolich JJ, Pärsson H, et al. Release of Activin and Follistatin during Cardiovascular Procedures Is Largely due to Heparin Administration1. J Clin Endocrinol Metab. 2000;85:2411–5. [CrossRef]
- Bäck S, Dossat A, Parkkinen I, Koivula P, Airavaara M, Richie CT, et al. Neuronal Activation Stimulates Cytomegalovirus Promoter-Driven Transgene Expression. Mol Ther - Methods Clin Dev. 2019;14:180–8. [CrossRef]
- Pickart L, Margolina A. Skin Regenerative and Anti-Cancer Actions of Copper Peptides. Cosmetics. 2018;5:29. [CrossRef]
- Dou Y, Lee A, Zhu L, Morton J, Ladiges W. The potential of GHK as an anti-aging peptide. Aging Pathobiol Ther. 2020;2:58–61. [CrossRef]
- Maquart FX, Bellon G, Chaqour B, Wegrowski J, Patt LM, Trachy RE, et al. In vivo stimulation of connective tissue accumulation by the tripeptide-copper complex glycyl-L-histidyl-L-lysine-Cu2+ in rat experimental wounds. J Clin Investig. 1993;92:2368–76. [CrossRef]
- Ahnfelt-Rønne I, Nowak J, Olsen UB. Do growth hormone-releasing peptides act as ghrelin secretagogues? Endocrine. 2001;14:133–5. [CrossRef]
- Raun K, Hansen BS, Johansen NL, Thogersen H, Madsen K, Ankersen M, et al. Ipamorelin, the first selective growth hormone secretagogue. Eur J Endocrinol. 1998;139:552–61. [CrossRef]
- Johansen PB, Nowak J, Skjærbæk C, Flyvbjerg A, Andreassen TT, Wilken M, et al. Ipamorelin, a new growth-hormone-releasing peptide, induces longitudinal bone growth in rats. Growth Horm IGF Res. 1999;9:106–13. [CrossRef]
- Prinz P, Stengel A. Control of Food Intake by Gastrointestinal Peptides: Mechanisms of Action and Possible Modulation in the Treatment of Obesity. J Neurogastroenterol Motil. 2017;23:180–96. [CrossRef]
- Mohammadi EN, Louwies T, Pietra C, Northrup SR, Meerveld BG-V. Attenuation of Visceral and Somatic Nociception by Ghrelin Mimetics. J Exp Pharmacol. 2020;12:267–74. [CrossRef]
- Group O behalf of the I 201 S, Beck DE, Sweeney WB, McCarter MD. Prospective, randomized, controlled, proof-of-concept study of the Ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients. Int J Color Dis. 2014;29:1527–34.
- Maughan RJ, Burke LM, Dvorak J, Larson-Meyer DE, Peeling P, Phillips SM, et al. IOC consensus statement: dietary supplements and the high-performance athlete. Br J Sports Med. 2018;52:439–55. [CrossRef]
- Poher A-L, Tschöp MH, Müller TD. Ghrelin regulation of glucose metabolism. Peptides. 2018;100:236–42. [CrossRef]
- Wasko R, Jaskula M, Kotwicka M, Andrusiewicz M, Jankowska A, Liebert W, et al. The expression of ghrelin in somatotroph and other types of pituitary adenomas. Neuro Endocrinol Lett. 2008;29:929–38.
- Prakash A, Goa KL. Sermorelin. BioDrugs. 1999;12:139–57. [CrossRef]
- González-Sales M, Barrière O, Tremblay PO, Nekka F, Mamputu J-C, Boudreault S, et al. Population pharmacokinetic and pharmacodynamic analysis of tesamorelin in HIV-infected patients and healthy subjects. J Pharmacokinet Pharmacodyn. 2015;42:287–99. [CrossRef]
- Bilbao A, Spanagel R. Medical cannabinoids: a pharmacology-based systematic review and meta-analysis for all relevant medical indications. BMC Med. 2022;20:259. [CrossRef]
- Souza MJD, Strock NCA, Ricker EA, Koltun KJ, Barrack M, Joy E, et al. The Path Towards Progress: A Critical Review to Advance the Science of the Female and Male Athlete Triad and Relative Energy Deficiency in Sport. Sports Med. 2022;52:13–23. [CrossRef]
- Lee C, Zeng J, Drew BG, Sallam T, Martin-Montalvo A, Wan J, et al. The Mitochondrial-Derived Peptide MOTS-c Promotes Metabolic Homeostasis and Reduces Obesity and Insulin Resistance. Cell Metab. 2015;21:443–54. [CrossRef]
- Zheng Y, Wei Z, Wang T. MOTS-c: A promising mitochondrial-derived peptide for therapeutic exploitation. Front Endocrinol. 2023;14:1120533. [CrossRef]
- Lyons CL, Roche HM. Nutritional Modulation of AMPK-Impact upon Metabolic-Inflammation. Int J Mol Sci. 2018;19:3092. [CrossRef]
- Ishida J, Saitoh M, Ebner N, Springer J, Anker SD, Haehling S von. Growth hormone secretagogues: history, mechanism of action, and clinical development. JCSM Rapid Commun. 2020;3:25–37. [CrossRef]
- Heinemeier KM, Mackey AL, Doessing S, Hansen M, Bayer ML, Nielsen RH, et al. GH/IGF-I axis and matrix adaptation of the musculotendinous tissue to exercise in humans. Scand J Med Sci Sports. 2012;22:e1-7. [CrossRef]
- Disser NP, Sugg KB, Talarek JR, Sarver DC, Rourke BJ, Mendias CL. Insulin-like growth factor 1 signaling in tenocytes is required for adult tendon growth. The FASEB Journal. 2019;33:12680–95.
- Marshall L, Mölle M, Böschen G, Steiger A, Fehm HL, Born J. Greater efficacy of episodic than continuous growth hormone-releasing hormone (GHRH) administration in promoting slow-wave sleep (SWS). J Clin Endocrinol Metab. 1996;81:1009–13. [CrossRef]
- Bartke A. Growth Hormone and Aging: Updated Review. World J Men’s Heal. 2019;37:19–30. [CrossRef]
- Sinha DK, Balasubramanian A, Tatem AJ, Rivera-Mirabal J, Yu J, Kovac J, et al. Beyond the androgen receptor: the role of growth hormone secretagogues in the modern management of body composition in hypogonadal males. Transl Androl Urol. 2019;9:S149–59. [CrossRef]
- Black DS, O’Reilly GA, Olmstead R, Breen EC, Irwin MR. Mindfulness Meditation and Improvement in Sleep Quality and Daytime Impairment Among Older Adults With Sleep Disturbances: A Randomized Clinical Trial. JAMA Intern Med. 2015;175:494–501.
- Tung C, Varzideh F, Farroni E, Mone P, Kansakar U, Jankauskas SS, et al. Elamipretide: A Review of Its Structure, Mechanism of Action, and Therapeutic Potential. Int J Mol Sci. 2025;26:944. [CrossRef]
- Zhao C, Zhuang X, Gao J. Elamipretide: The first cardiolipin-directed mitochondrial therapeutic for Barth syndrome approved under accelerated approval. Drug Discov Ther. 2025;2025.01111. [CrossRef]
- Zhu Y, Wang H, Fang J, Dai W, Zhou J, Wang X, et al. SS-31 Provides Neuroprotection by Reversing Mitochondrial Dysfunction after Traumatic Brain Injury. Oxidative Med Cell Longev. 2018;2018:4783602. [CrossRef]
- Storoschuk KL, Moran-MacDonald A, Gibala MJ, Gurd BJ. Much Ado About Zone 2: A Narrative Review Assessing the Efficacy of Zone 2 Training for Improving Mitochondrial Capacity and Cardiorespiratory Fitness in the General Population. Sports Med. 2025;55:1611–24. [CrossRef]
- Brisswalter J, Louis J. Vitamin Supplementation Benefits in Master Athletes. Sports Med. 2014;44:311–8. [CrossRef]
- Goldstein AL, Kleinman HK. Advances in the basic and clinical applications of thymosin β4. Expert Opin Biol Ther. 2015;15:139–45.
- Freeman KW, Banyard J. B-thymosins in cancer: implications for the clinic. Futur Oncol. 2009;5:755–8. [CrossRef]
- Hutchings DC, Anderson SG, Caldwell JL, Trafford AW. Phosphodiesterase-5 inhibitors and the heart: compound cardioprotection? Heart. 2018;104:1244. [CrossRef]
- Sahay S, Kwak SY. The Efficacy of Adjuvant Hyperbaric Oxygen Therapy in Chronic Wound Management: A Narrative Review. Cureus. 2025;17:e92728. [CrossRef]
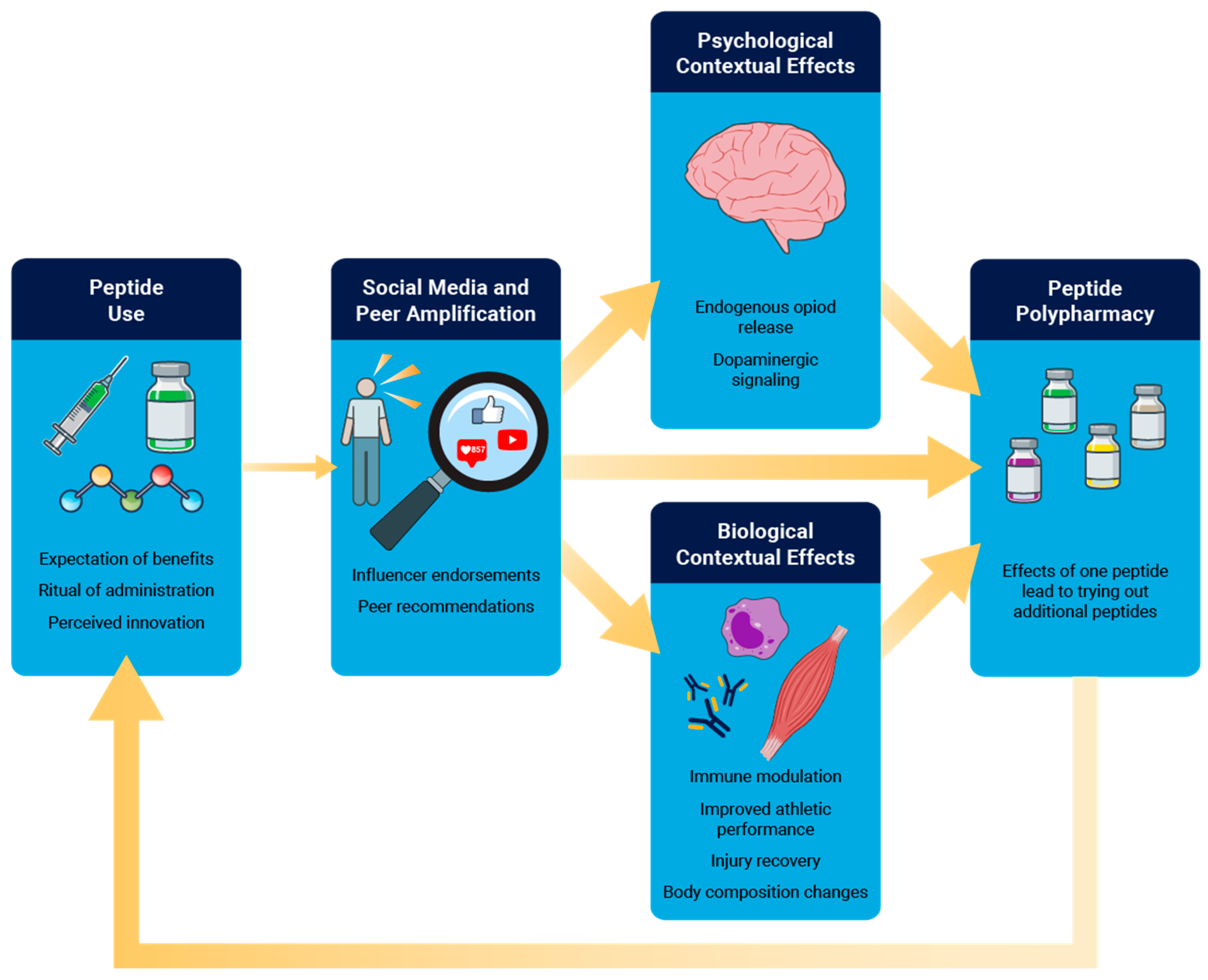
- Benedetti F. Placebo and the New Physiology of the Doctor-Patient Relationship. Physiol Rev. 2013;93:1207–46. [CrossRef]
- Benedetti F, Mayberg HS, Wager TD, Stohler CS, Zubieta J-K. Neurobiological Mechanisms of the Placebo Effect. J Neurosci. 2005;25:10390–402. [CrossRef]
- Smits RM, Veldhuijzen DS, Wulffraat NM, Evers AWM. The role of placebo effects in immune-related conditions: mechanisms and clinical considerations. Expert Rev Clin Immunol. 2018;14:761–70. [CrossRef]
- Lennep J (Hans) PA van, Trossèl F, Perez RSGM, Otten RHJ, Middendorp H van, Evers AWM, et al. Placebo effects in low back pain: A systematic review and meta-analysis of the literature. Eur J Pain. 2021;25:1876–97.
- Saueressig T, Owen PJ, Pedder H, Tagliaferri S, Kaczorowski S, Altrichter A, et al. The importance of context (placebo effects) in conservative interventions for musculoskeletal pain: A systematic review and meta-analysis of randomized controlled trials. Eur J Pain. 2024;28:675–704. [CrossRef]
- Valero F, González-Mohíno F, Salinero JJ. Belief That Caffeine Ingestion Improves Performance in a 6-Minute Time Trial Test without Affecting Pacing Strategy. Nutrients. 2024;16:327. [CrossRef]
- Pollo A, Carlino E, Benedetti F. The top-down influence of ergogenic placebos on muscle work and fatigue. Eur J Neurosci. 2008;28:379–88. [CrossRef]
- Carvalho C, Caetano JM, Cunha L, Rebouta P, Kaptchuk TJ, Kirsch I. Open-label placebo treatment in chronic low back pain. PAIN. 2016;157:2766–72. [CrossRef]
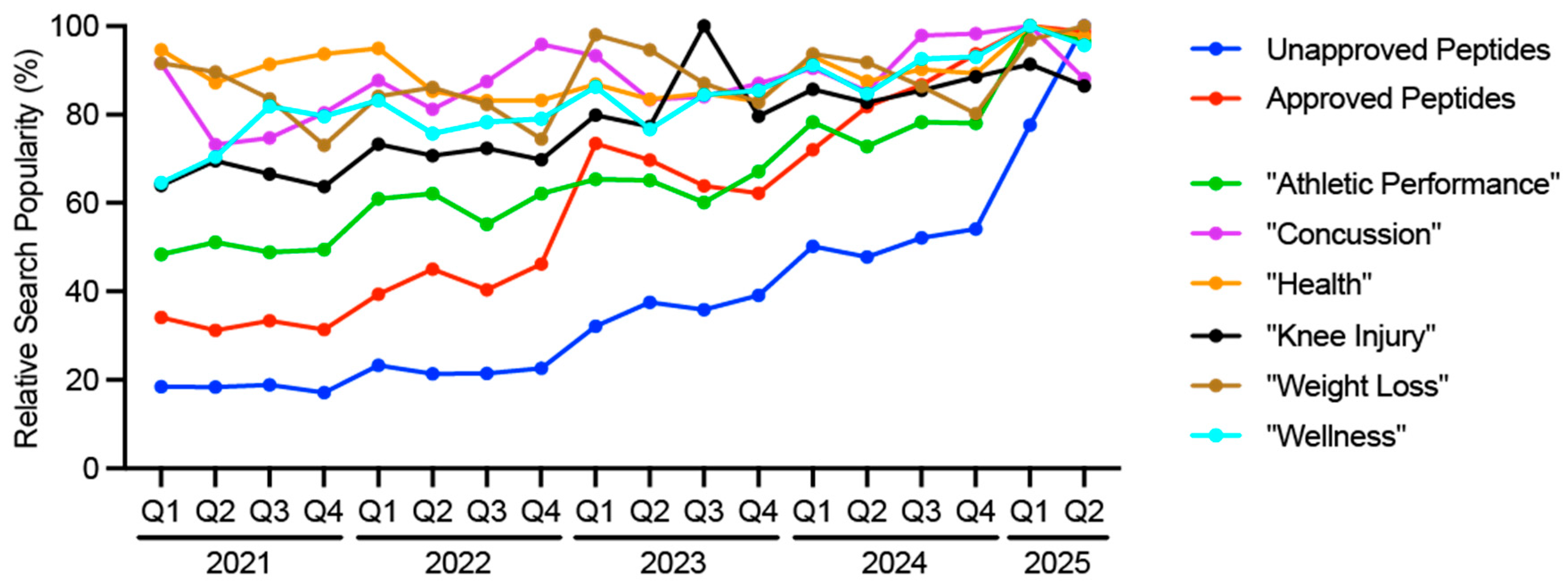
- González-Bailón S, Lelkes Y. Do social media undermine social cohesion? A critical review. Soc Issues Polic Rev. 2023;17:155–80. [CrossRef]
- Zamil DH, Ameri M, Fu S, Abughosh FM, Katta R. Skin, hair, and nail supplements advertised on Instagram. Bayl Univ Méd Cent Proc. 2023;36:38–40. [CrossRef]
- Ricke J-N, Seifert R. Disinformation on dietary supplements by German influencers on Instagram. Naunyn-Schmiedeberg’s Arch Pharmacol. 2025;398:5629–47. [CrossRef]
- Chou W-YS, Gaysynsky A, Trivedi N, Vanderpool RC. Using Social Media for Health: National Data from HINTS 2019. J Heal Commun. 2021;26:184–93. [CrossRef]
- Kraljevic S, Stambrook PJ, Pavelic K. Accelerating drug discovery. EMBO Rep. 2004;5:837–42.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



