Submitted:
09 December 2025
Posted:
11 December 2025
You are already at the latest version
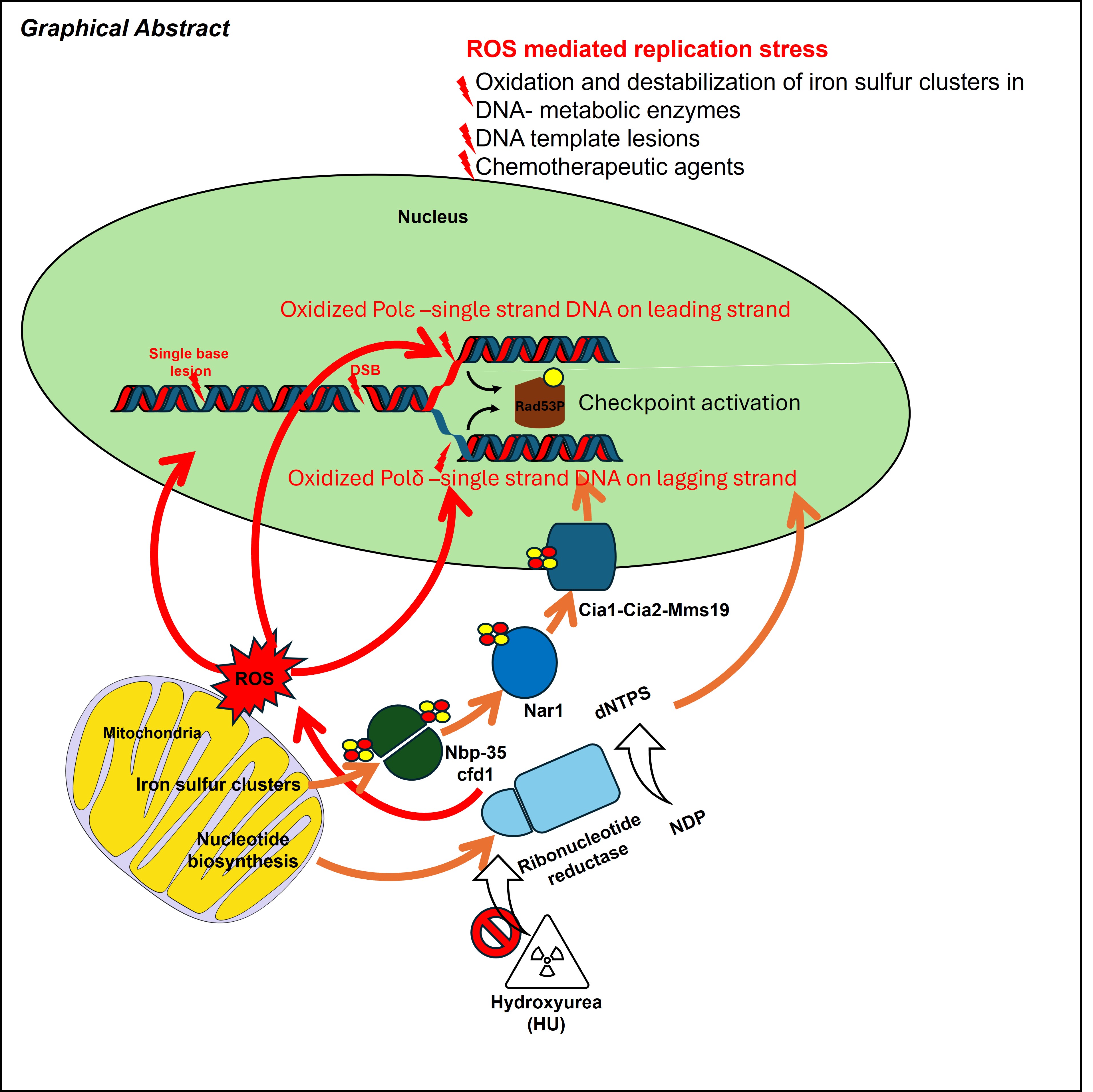
Abstract
Keywords:
1. Introduction
2. Molecular Components of Core Eukaryotic Replisome
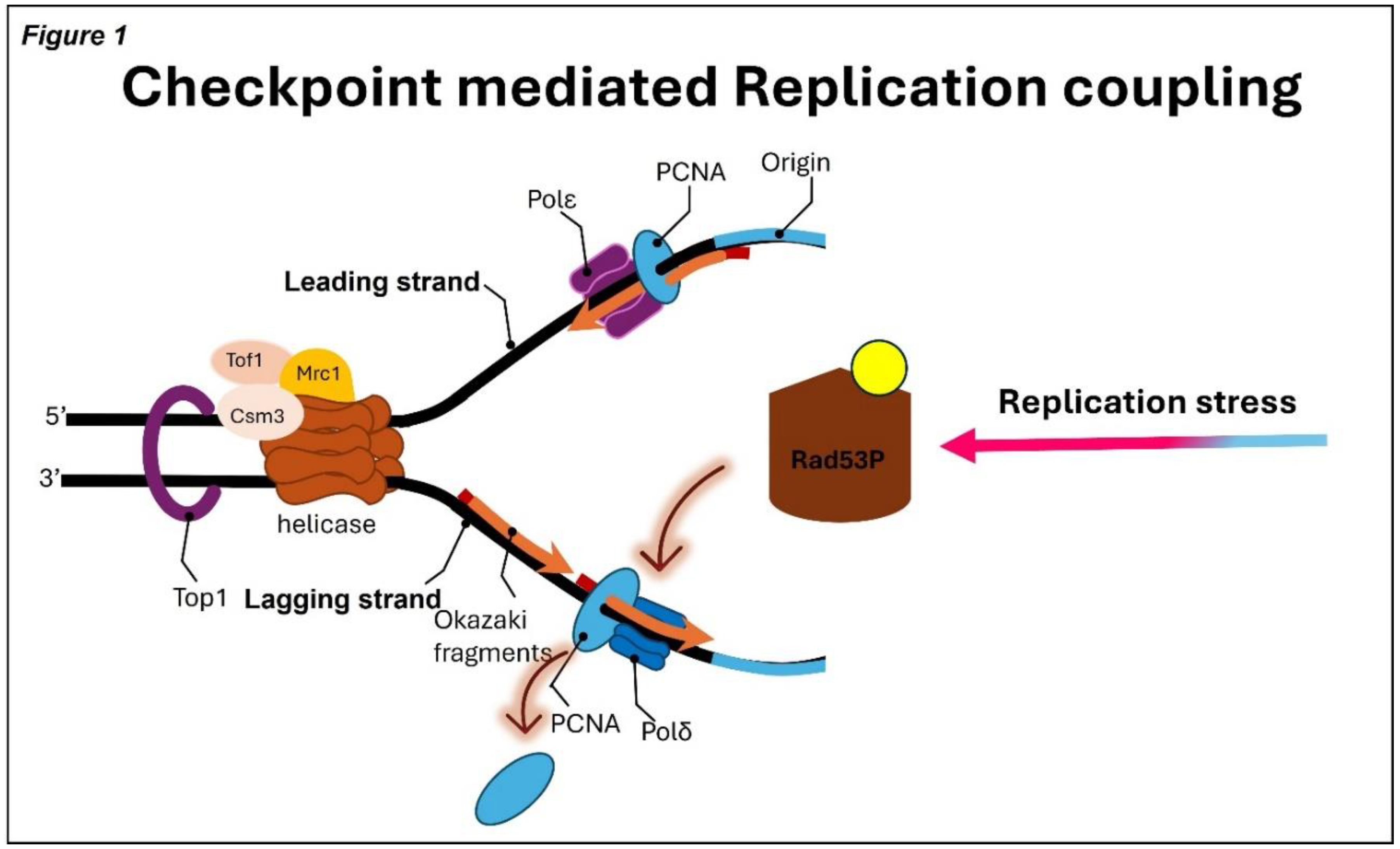
3. Replication Checkpoint Activation
4. HU Modulates Replication Fork Progression and Stability by ROS Independent Pathways (RNR Inhibition, Checkpoint Activations and Others)
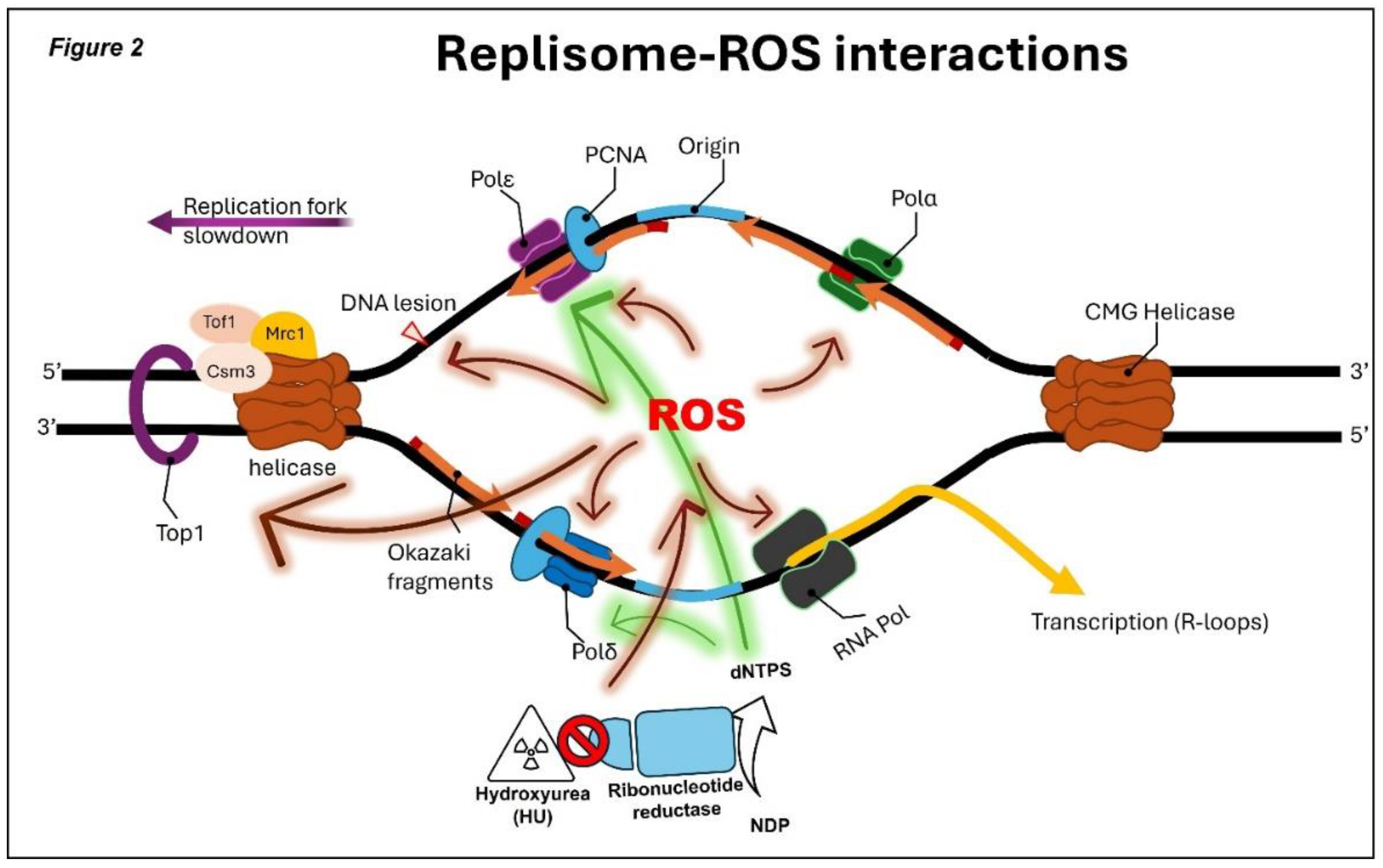
5. General Concepts of ROS and Their Impact on DNA Replication
6. HU Contributes to Replication Stress and Genome Instability Through ROS-dependent Pathways
7. Conclusions Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Alabert, C.; Bukowski-Wills, J.-C.; Lee, S.-B.; Kustatscher, G.; Nakamura, K.; Alves, F.D.L.; Menard, P.; Mejlvang, J.; Rappsilber, J.; Groth, A. Nascent chromatin capture proteomics determines chromatin dynamics during DNA replication and identifies unknown fork components. Nat. Cell Biol. 2014, 16, 281–291. [CrossRef]
- Zoghbi, H.Y.; Beaudet, A.L. Epigenetics and Human Disease. Cold Spring Harb. Perspect. Biol. 2016, 8, a019497. [CrossRef]
- Luo, M.; He, H.; Kelley, M.R.; Georgiadis, M.M. Redox Regulation of DNA Repair: Implications for Human Health and Cancer Therapeutic Development. Antioxidants Redox Signal. 2010, 12, 1247–1269. [CrossRef]
- Venkatachalam, G.; Surana, U.; Clément, M.-V. Replication stress-induced endogenous DNA damage drives cellular senescence induced by a sub-lethal oxidative stress. Nucleic Acids Res. 2017, 45, 10564–10582. [CrossRef]
- Fishbein, W. N., and Carbone, P. P. (1963) Hydroxyurea: Mechanism of Action, Science 142, 1069-1070.
- Kennedy, B.J. The evolution of hydroxyurea therapy in chronic myelogenous leukemia.. 1992, 19, 21–6.
- Shaw, A.E.; Mihelich, M.N.; Whitted, J.E.; Reitman, H.J.; Timmerman, A.J.; Tehseen, M.; Hamdan, S.M.; Schauer, G.D. Revised mechanism of hydroxyurea-induced cell cycle arrest and an improved alternative. Proc. Natl. Acad. Sci. 2024, 121. [CrossRef]
- Andrs, M.; Stoy, H.; Boleslavska, B.; Chappidi, N.; Kanagaraj, R.; Nascakova, Z.; Menon, S.; Rao, S.; Oravetzova, A.; Dobrovolna, J.; et al. Excessive reactive oxygen species induce transcription-dependent replication stress. Nat. Commun. 2023, 14, 1–15. [CrossRef]
- Huang, M.-E.; Facca, C.; Fatmi, Z.; Baïlle, D.; Bénakli, S.; Vernis, L. DNA replication inhibitor hydroxyurea alters Fe-S centers by producing reactive oxygen species in vivo. Sci. Rep. 2016, 6, 29361. [CrossRef]
- Koç, A.; Wheeler, L.J.; Mathews, C.K.; Merrill, G.F. Hydroxyurea Arrests DNA Replication by a Mechanism That Preserves Basal dNTP Pools. J. Biol. Chem. 2004, 279, 223–230. [CrossRef]
- Musialek, M. W., and Rybaczek, D. (2021) Hydroxyurea-The Good, the Bad and the Ugly, Genes (Basel) 12.
- Jenkyn-Bedford, M.; Jones, M.L.; Baris, Y.; Labib, K.P.M.; Cannone, G.; Yeeles, J.T.P.; Deegan, T.D. A conserved mechanism for regulating replisome disassembly in eukaryotes. Nature 2021, 600, 743–747. [CrossRef]
- Dang, H.Q.; Li, Z. The Cdc45·Mcm2–7·GINS Protein Complex in Trypanosomes Regulates DNA Replication and Interacts with Two Orc1-like Proteins in the Origin Recognition Complex. J. Biol. Chem. 2011, 286, 32424–32435. [CrossRef]
- Aria, V.; Yeeles, J.T. Mechanism of Bidirectional Leading-Strand Synthesis Establishment at Eukaryotic DNA Replication Origins. Mol. Cell 2019, 73, 199–211.e10. [CrossRef]
- Yuan, Z.; Georgescu, R.; Li, H.; O’dOnnell, M.E. Molecular choreography of primer synthesis by the eukaryotic Pol α-primase. Nat. Commun. 2023, 14, 1–14. [CrossRef]
- McElhinny, S.A.N.; Gordenin, D.A.; Stith, C.M.; Burgers, P.M.; Kunkel, T.A. Division of Labor at the Eukaryotic Replication Fork. Mol. Cell 2008, 30, 137–144. [CrossRef]
- Stillman, B. DNA Polymerases at the Replication Fork in Eukaryotes. Mol. Cell 2008, 30, 259–260. [CrossRef]
- Pursell, Z.F.; Isoz, I.; Lundström, E.-B.; Johansson, E.; Kunkel, T.A. Yeast DNA Polymerase ε Participates in Leading-Strand DNA Replication. Science 2007, 317, 127–130. [CrossRef]
- Yeeles, J.T.; Janska, A.; Early, A.; Diffley, J.F. How the Eukaryotic Replisome Achieves Rapid and Efficient DNA Replication. Mol. Cell 2017, 65, 105–116. [CrossRef]
- Keszthelyi, A.; Mansoubi, S.; Whale, A.; Houseley, J.; Baxter, J. The fork protection complex generates DNA topological stress–induced DNA damage while ensuring full and faithful genome duplication. Proc. Natl. Acad. Sci. 2024, 121. [CrossRef]
- Saxena, S.; Zou, L. Hallmarks of DNA replication stress. Mol. Cell 2022, 82, 2298–2314. [CrossRef]
- Byun, T.S.; Pacek, M.; Yee, M.-C.; Walter, J.C.; Cimprich, K.A. Functional uncoupling of MCM helicase and DNA polymerase activities activates the ATR-dependent checkpoint. Genes Dev. 2005, 19, 1040–1052. [CrossRef]
- Serra-Cardona, A.; Yu, C.; Zhang, X.; Hua, X.; Yao, Y.; Zhou, J.; Gan, H.; Zhang, Z. A mechanism for Rad53 to couple leading- and lagging-strand DNA synthesis under replication stress in budding yeast. Proc. Natl. Acad. Sci. 2021, 118. [CrossRef]
- Feng, W.; Collingwood, D.; Boeck, M.E.; Fox, L.A.; Alvino, G.M.; Fangman, W.L.; Raghuraman, M.K.; Brewer, B.J. Genomic mapping of single-stranded DNA in hydroxyurea-challenged yeasts identifies origins of replication. Nat. Cell Biol. 2006, 8, 148–155. [CrossRef]
- Fu, Y.V.; Yardimci, H.; Long, D.T.; Ho, T.V.; Guainazzi, A.; Bermudez, V.P.; Hurwitz, J.; Van Oijen, A.; Schärer, O.D.; Walter, J.C. Selective Bypass of a Lagging Strand Roadblock by the Eukaryotic Replicative DNA Helicase. Cell 2011, 146, 931–941. [CrossRef]
- Sparks, J.L.; Chistol, G.; Gao, A.O.; Räschle, M.; Larsen, N.B.; Mann, M.; Duxin, J.P.; Walter, J.C. The CMG Helicase Bypasses DNA-Protein Cross-Links to Facilitate Their Repair. Cell 2019, 176, 167–181.e21. [CrossRef]
- Maréchal, A.; Zou, L. RPA-coated single-stranded DNA as a platform for post-translational modifications in the DNA damage response. Cell Res. 2014, 25, 9–23. [CrossRef]
- Brush, G.S.; Morrow, D.M.; Hieter, P.; Kelly, T.J. The ATM homologue MEC1 is required for phosphorylation of replication protein A in yeast. Proc. Natl. Acad. Sci. 1996, 93, 15075–15080. [CrossRef]
- Clerici, M.; Trovesi, C.; Galbiati, A.; Lucchini, G.; Longhese, M.P. Mec1/ATR regulates the generation of single-stranded DNA that attenuates Tel1/ATM signaling at DNA ends. EMBO J. 2013, 33, 198–216. [CrossRef]
- Tannous, E.A.; Burgers, P.M. Novel insights into the mechanism of cell cycle kinases Mec1(ATR) and Tel1(ATM). Crit. Rev. Biochem. Mol. Biol. 2021, 56, 441–454. [CrossRef]
- Navadgi-Patil, V.M.; Burgers, P.M. Cell-cycle-specific activators of the Mec1/ATR checkpoint kinase. Biochem. Soc. Trans. 2011, 39, 600–605. [CrossRef]
- Zheng, F.; Georgescu, R.E.; Yao, N.Y.; O’donnell, M.E.; Li, H. DNA is loaded through the 9-1-1 DNA checkpoint clamp in the opposite direction of the PCNA clamp. Nat. Struct. Mol. Biol. 2022, 29, 376–385. [CrossRef]
- Navadgi-Patil, V.M.; Burgers, P.M. Yeast DNA Replication Protein Dpb11 Activates the Mec1/ATR Checkpoint Kinase. J. Biol. Chem. 2008, 283, 35853–35859. [CrossRef]
- Villa, M.; Bonetti, D.; Carraro, M.; Longhese, M.P. Rad9/53 BP 1 protects stalled replication forks from degradation in Mec1/ ATR -defective cells. Embo Rep. 2018, 19, 351–367. [CrossRef]
- Hodgson, B.; Calzada, A.; Labib, K. Mrc1 and Tof1 Regulate DNA Replication Forks in Different Ways during Normal S Phase. Mol. Biol. Cell 2007, 18, 3894–3902. [CrossRef]
- McClure, A.W.; Diffley, J.F. Rad53 checkpoint kinase regulation of DNA replication fork rate via Mrc1 phosphorylation. eLife 2021, 10. [CrossRef]
- Uto, K.; Inoue, D.; Shimuta, K.; Nakajo, N.; Sagata, N. Chk1, but not Chk2, inhibits Cdc25 phosphatases by a novel common mechanism. EMBO J. 2004, 23, 3386–3396. [CrossRef]
- Zur, A.; Brandeis, M. Securin degradation is mediated by fzy and fzr, and is required for complete chromatid separation but not for cytokinesis. EMBO J. 2001, 20, 792–801. [CrossRef]
- Zhao, X.; Muller, E.G.; Rothstein, R. A Suppressor of Two Essential Checkpoint Genes Identifies a Novel Protein that Negatively Affects dNTP Pools. Mol. Cell 1998, 2, 329–340. [CrossRef]
- Chen, S.-H.; Smolka, M.B.; Zhou, H. Mechanism of Dun1 Activation by Rad53 Phosphorylation in Saccharomyces cerevisiae. J. Biol. Chem. 2007, 282, 986–995. [CrossRef]
- Lopez-Mosqueda, J.; Maas, N.L.; Jonsson, Z.O.; DeFazio-Eli, L.G.; Wohlschlegel, J.; Toczyski, D.P. Damage-induced phosphorylation of Sld3 is important to block late origin firing. Nature 2010, 467, 479–483. [CrossRef]
- Zegerman, P.; Diffley, J.F.X. Checkpoint-dependent inhibition of DNA replication initiation by Sld3 and Dbf4 phosphorylation. Nature 2010, 467, 474–478. [CrossRef]
- Karri, S.; Dickinson, Q.; Li, Z.; Yu, C.; Zhang, Z. Strand-Specific Analysis of Proteins at Replicating DNA Strands by Enrichment and Sequencing of Protein-Associated Nascent DNA Method. J. Vis. Exp. 2025. [CrossRef]
- Yu, C.; Gan, H.; Zhang, Z. (2018) Strand-Specific Analysis of DNA Synthesis and Proteins Association with DNA Replication Forks in Budding Yeast, In Genome Instability: Methods and Protocols (Muzi-Falconi, M., and Brown, G. W., Eds.), pp 227-238, Springer New York, New York, NY.
- Thakar, T.; Leung, W.; Nicolae, C.M.; Clements, K.E.; Shen, B.; Bielinsky, A.-K.; Moldovan, G.-L. Ubiquitinated-PCNA protects replication forks from DNA2-mediated degradation by regulating Okazaki fragment maturation and chromatin assembly. Nat. Commun. 2020, 11, 1–14. [CrossRef]
- Serra-Cardona, A.; Hua, X.; McNutt, S.W.; Zhou, H.; Toda, T.; Jia, S.; Chu, F.; Zhang, Z. The PCNA–Pol δ complex couples lagging strand DNA synthesis to parental histone transfer for epigenetic inheritance. Sci. Adv. 2024, 10, eadn5175. [CrossRef]
- Lengronne, A.; Pasero, P.; Bensimon, A.; Schwob, E. Monitoring S phase progression globally and locally using BrdU incorporation in TK+ yeast strains. Nucleic Acids Res. 2001, 29, 1433–1442. [CrossRef]
- Raveendranathan, M.; Chattopadhyay, S.; Bolon, Y.; Haworth, J.; Clarke, D.J.; Bielinsky, A. Genome-wide replication profiles of S-phase checkpoint mutants reveal fragile sites in yeast. EMBO J. 2006, 25, 3627–3639. [CrossRef]
- Koren, A.; Soifer, I.; Barkai, N. MRC1-dependent scaling of the budding yeast DNA replication timing program. Genome Res. 2010, 20, 781–790. [CrossRef]
- Naylor, M.L.; Li, J.-M.; Osborn, A.J.; Elledge, S.J. Mrc1 phosphorylation in response to DNA replication stress is required for Mec1 accumulation at the stalled fork. Proc. Natl. Acad. Sci. 2009, 106, 12765–12770. [CrossRef]
- Osborn, A.J.; Elledge, S.J. Mrc1 is a replication fork component whose phosphorylation in response to DNA replication stress activates Rad53. Genes Dev. 2003, 17, 1755–1767. [CrossRef]
- Gan, H.; Yu, C.; Devbhandari, S.; Sharma, S.; Han, J.; Chabes, A.; Remus, D.; Zhang, Z. Checkpoint Kinase Rad53 Couples Leading- and Lagging-Strand DNA Synthesis under Replication Stress. Mol. Cell 2017, 68, 446–455.e3. [CrossRef]
- Yu, C., Gan, H., and Zhang, Z. (2017) Both DNA Polymerases delta and epsilon Contact Active and Stalled Replication Forks Differently, Mol Cell Biol 37.
- Hong, Y.; Boiti, A.; Vallone, D.; Foulkes, N.S. Reactive Oxygen Species Signaling and Oxidative Stress: Transcriptional Regulation and Evolution. Antioxidants 2024, 13, 312. [CrossRef]
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.A.; Sethi, G. Role of Reactive Oxygen Species in Cancer Progression: Molecular Mechanisms and Recent Advancements. Biomolecules 2019, 9, 735. [CrossRef]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [CrossRef]
- Weidinger, A.; Kozlov, A.V. Biological Activities of Reactive Oxygen and Nitrogen Species: Oxidative Stress versus Signal Transduction. Biomolecules 2015, 5, 472–484. [CrossRef]
- Zhou, X.; An, B.; Lin, Y.; Ni, Y.; Zhao, X.; Liang, X. Molecular mechanisms of ROS-modulated cancer chemoresistance and therapeutic strategies. Biomed. Pharmacother. 2023, 165, 115036. [CrossRef]
- Somyajit, K.; Gupta, R.; Sedlackova, H.; Neelsen, K.J.; Ochs, F.; Rask, M.-B.; Choudhary, C.; Lukas, J. Redox-sensitive alteration of replisome architecture safeguards genome integrity. Science 2017, 358, 797–802. [CrossRef]
- Heinke, L. Mitochondrial ROS drive cell cycle progression. Nat. Rev. Mol. Cell Biol. 2022, 23, 581–581. [CrossRef]
- Vernis, L.; Facca, C.; Delagoutte, E.; Soler, N.; Chanet, R.; Guiard, B.; Faye, G.; Baldacci, G. A Newly Identified Essential Complex, Dre2-Tah18, Controls Mitochondria Integrity and Cell Death after Oxidative Stress in Yeast. PLOS ONE 2009, 4, e4376. [CrossRef]
- Chiu, J.; Tactacan, C.M.; Tan, S.-X.; Lin, R.C.; Wouters, M.A.; Dawes, I.W. Cell Cycle Sensing of Oxidative Stress in Saccharomyces cerevisiae by Oxidation of a Specific Cysteine Residue in the Transcription Factor Swi6p. J. Biol. Chem. 2011, 286, 5204–5214. [CrossRef]
- Rudolph, J. Redox Regulation of the Cdc25 Phosphatases. Antioxidants Redox Signal. 2005, 7, 761–767. [CrossRef]
- Kirova, D.G.; Judasova, K.; Vorhauser, J.; Zerjatke, T.; Leung, J.K.; Glauche, I.; Mansfeld, J. A ROS-dependent mechanism promotes CDK2 phosphorylation to drive progression through S phase. Dev. Cell 2022, 57, 1712–1727.e9. [CrossRef]
| Yeast | Human | Function |
| Chl1 | CHLR1 | Helicase, sister chromatid cohesion, heterochromatin organization |
| Dna2 | DNA2 | Helicase/nuclease, DNA repair, Okazaki fragment maturation, telomere maintenance |
| Absent | FANCJ | Helicase, repair of DNA interstrand crosslinks |
| Absent | MUTYH | DNA glycosylase, base excision repair |
| Ntg2 | NTHL1 | DNA glycosylase, base excision repair |
| Pol1 | POLA | Catalytic subunit of polymerase α, DNA replication |
| Pol3 | POLD1 | Catalytic subunit of polymerase δ, DNA replication |
| Pol2 | POLE1 | Catalytic subunit of polymerase ε, DNA replication |
| Pri2 | PRIM2 | Subunit of DNA Primase, DNA synthesis and double strand break repair |
| Absent | RTEL1 | Helicase, regulation of telomere length, anti-recombinase |
| Rad3 | XPD | Helicase, nucleotide excision repair |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




