Submitted:
09 December 2025
Posted:
10 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Ternary Eutectic of Molten Carbonates
3. Data Analysis
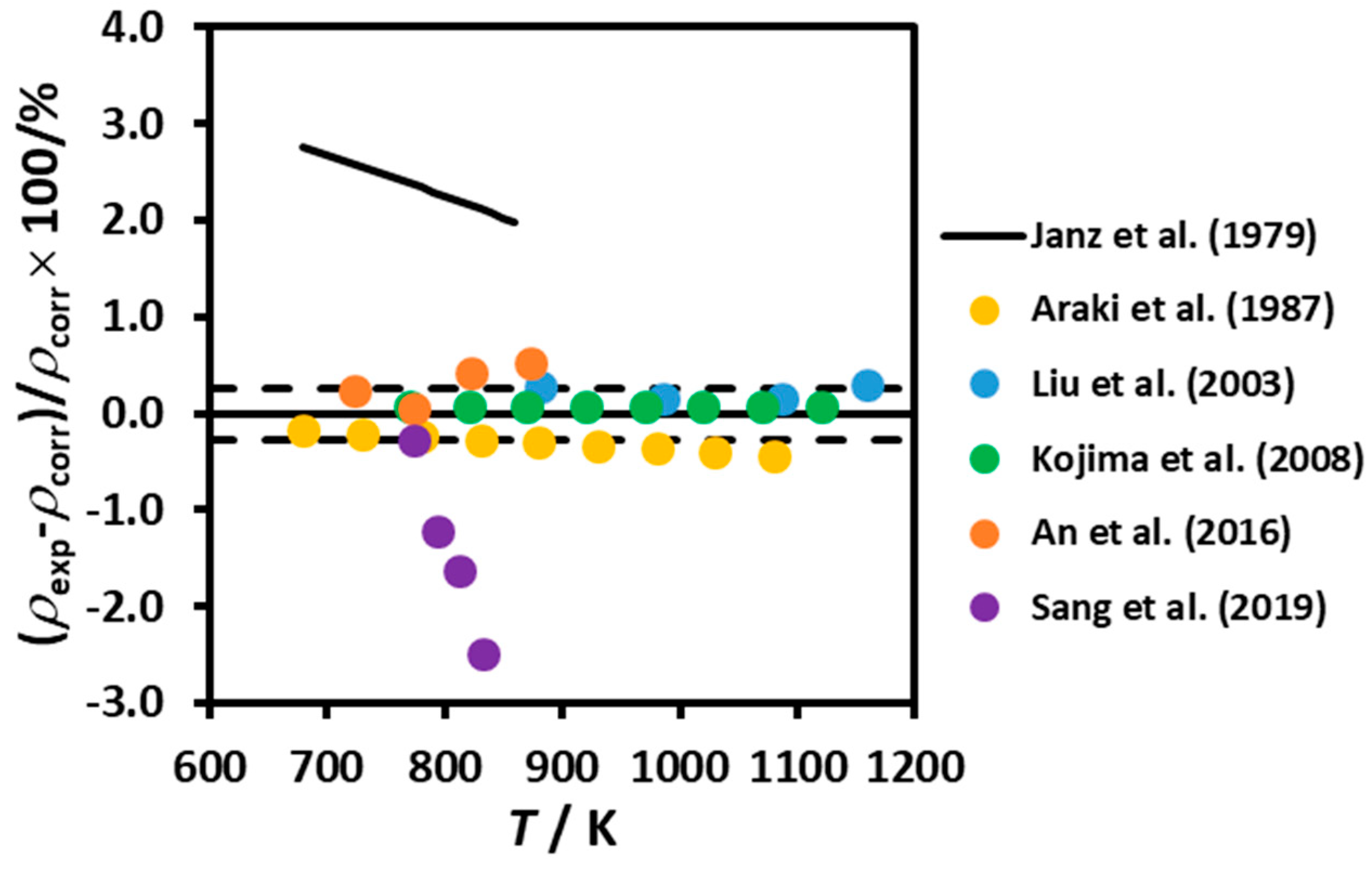
3.1. Density
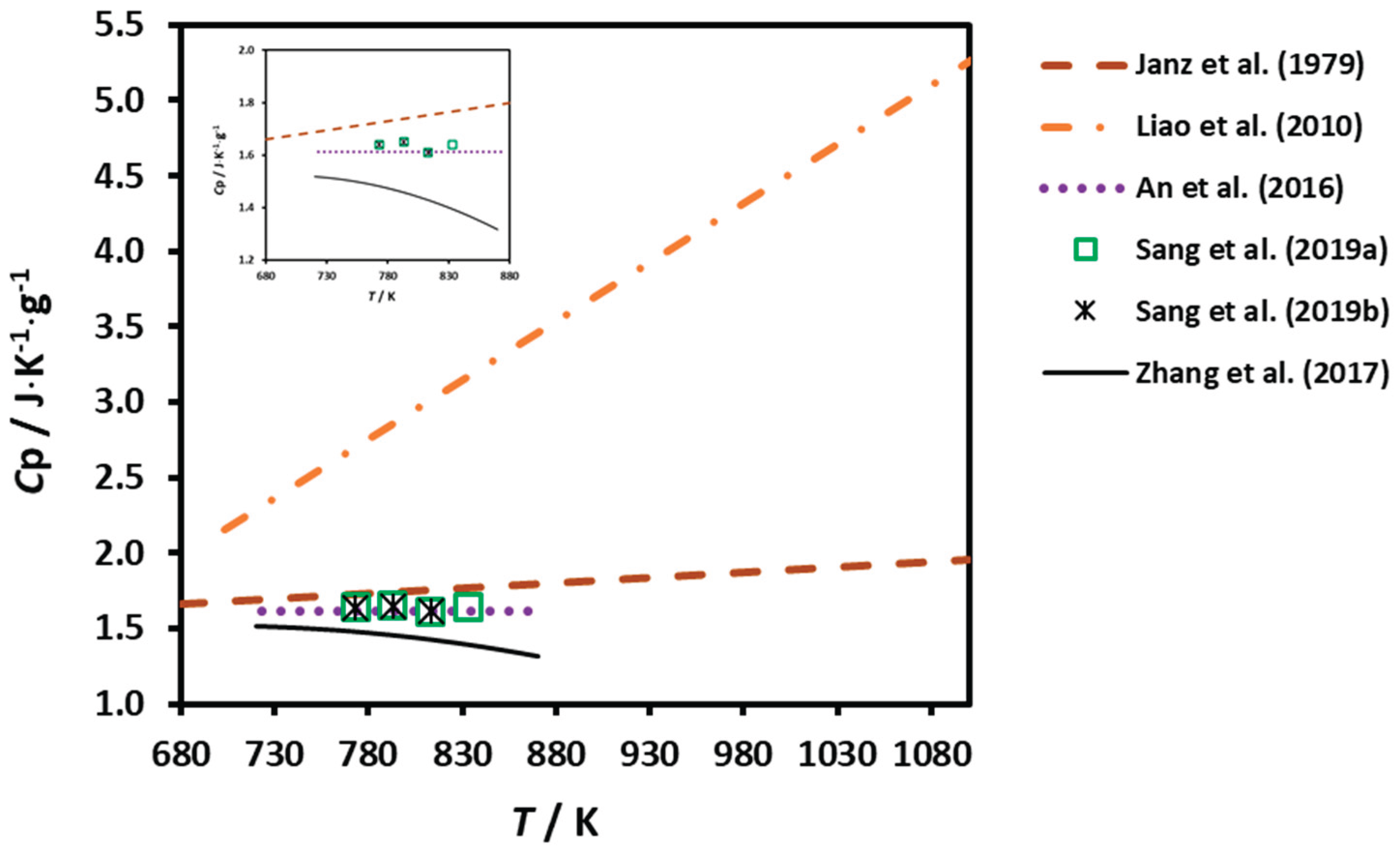
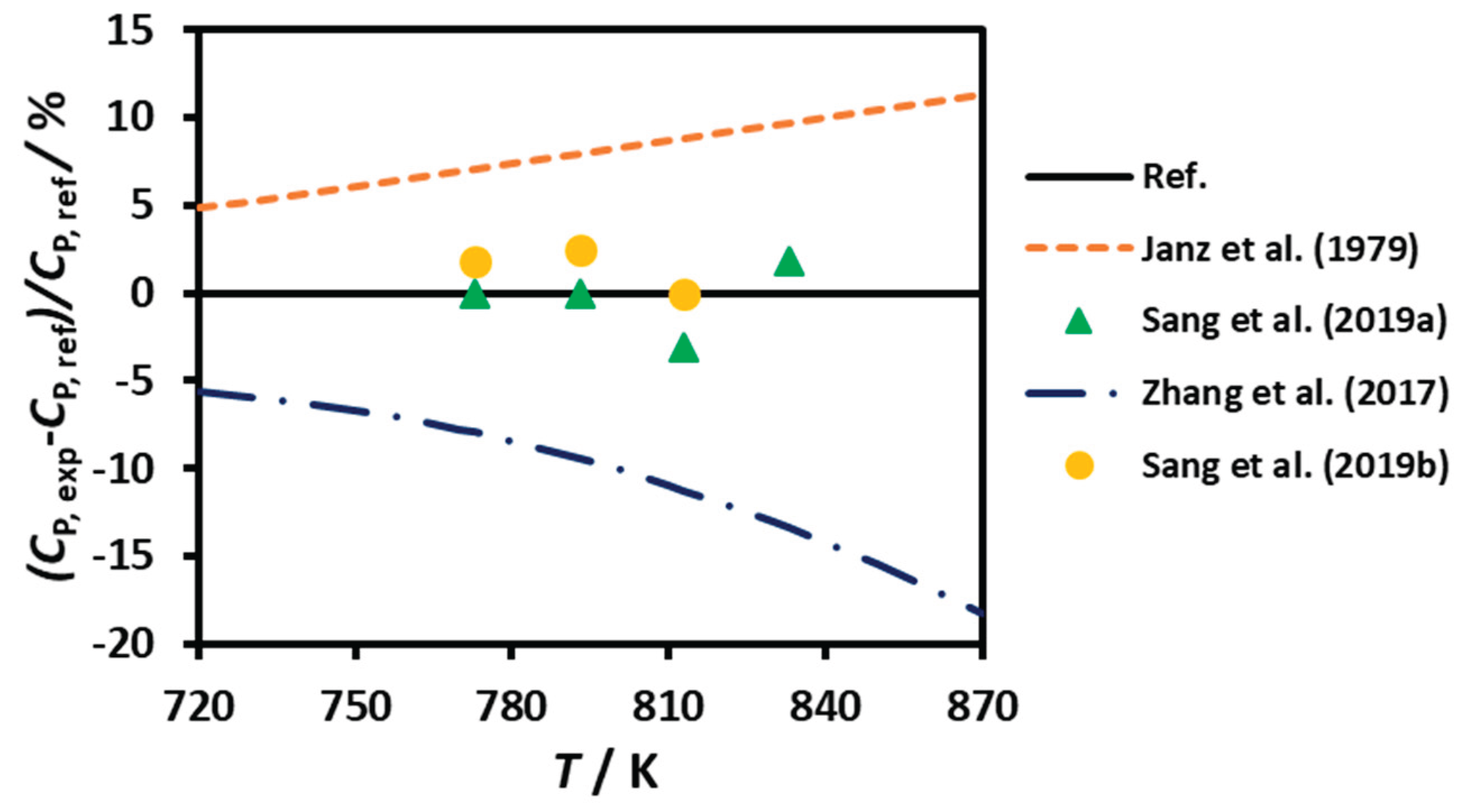
3.2. Heat Capacity
3.3. Thermal Conductivity
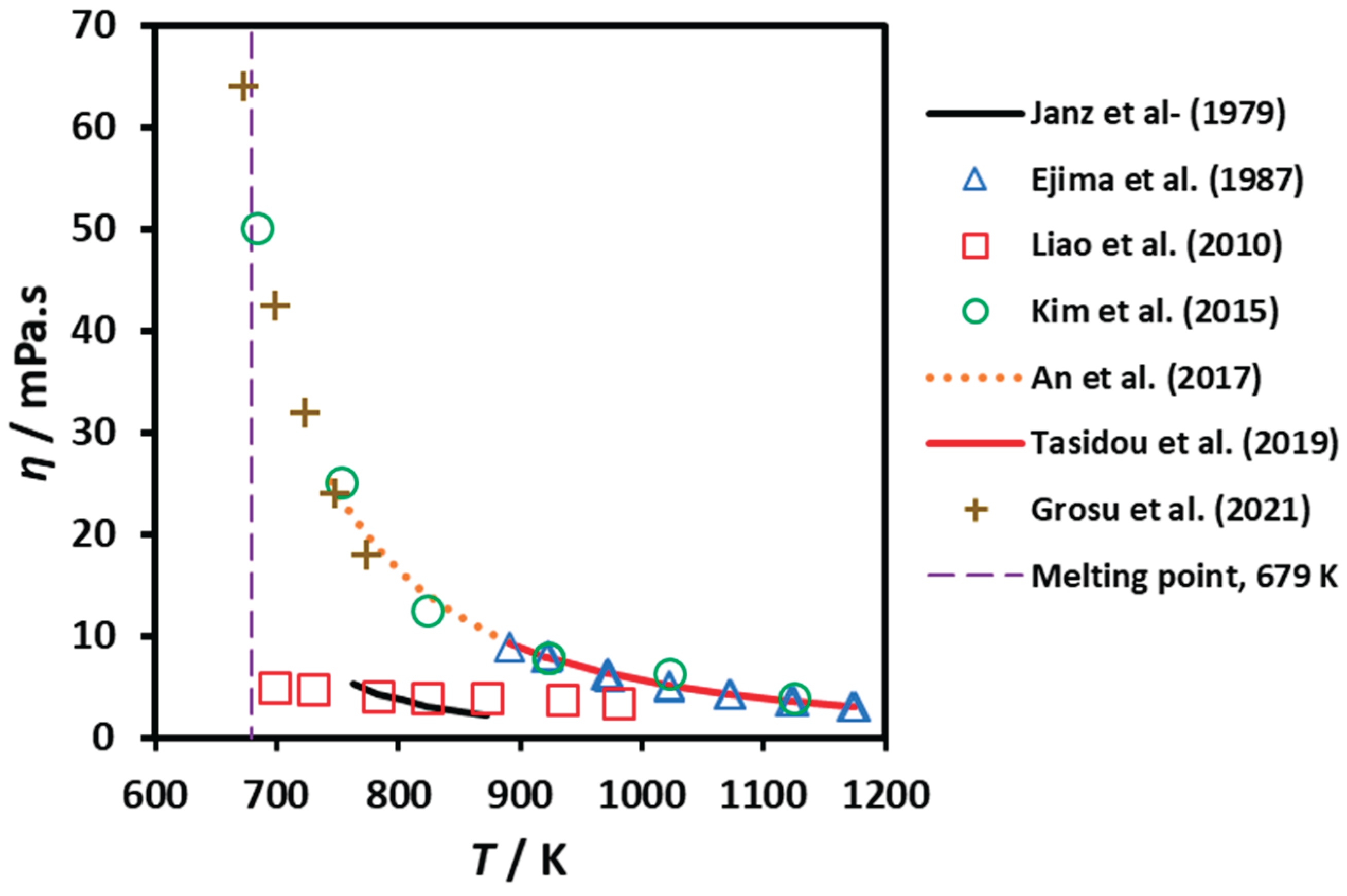
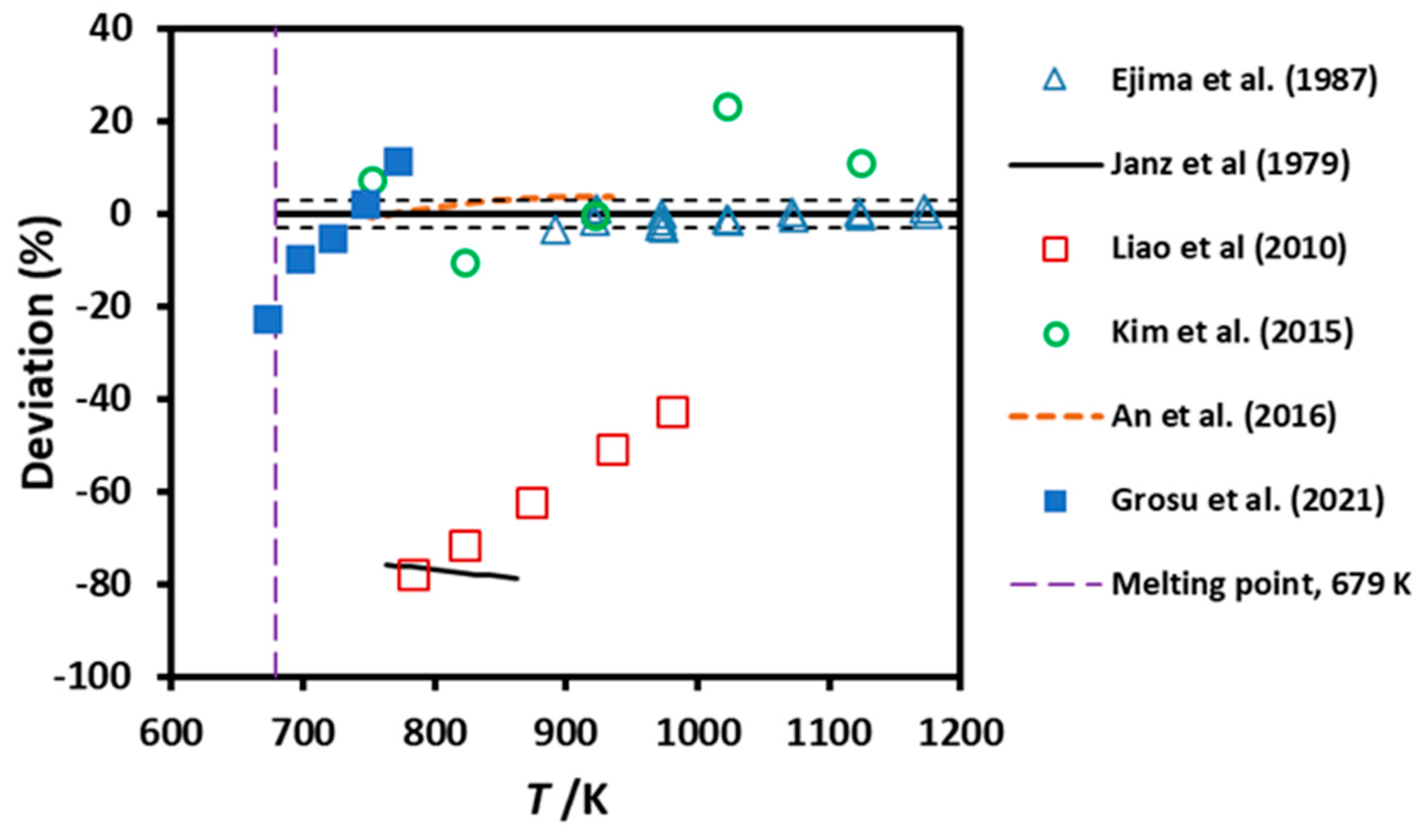
3.3. Viscosity
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CSP | Concentrated solar power |
| MSO | Molten salts oxidation |
| A | Archimedean |
| DBA | Double-bob Archimedean |
| MBP | Maximum bubble pressure |
| DC | Drop calorimetry |
| DSC | Differential scanning calorimetry |
| STA | Simultaneous thermal analysis |
| LF (Calc) | Laser flash, calculated from thermal diffusivity, heat capacity, and density |
| FRS (Calc) | Forced Rayleigh scattering, calculated from thermal diffusivity, heat capacity, and density |
| THW | Transient hot wire |
| SSCC | Steady state coaxial cylinders |
| OCY | Oscillating cylinder |
| RC | Rotating cylinder |
| RPP | Rheometer, parallel plate |
| NA | Not available |
References
- Aneke, M.; Wang, M. Energy storage technologies and real-life applications – a state of the art review. Appl Energy 2016, 179, 350-77. [CrossRef]
- Frangini, S.; Masi, A. Molten carbonates for advanced and sustainable energy applications: Part I. Revisiting molten carbonate properties from a sustainable viewpoint. Int J. Hydrogen Energy 2016, 41, 18739-18746. [CrossRef]
- Frangini, S.; Masi, A. Molten carbonates for advanced and sustainable energy applications: Part II. Review of recent literature. Int. J. Hydrogen Energy 2016, 41, 18971-18994. [CrossRef]
- Prieto, C.; Fereres, S.; Ruiz-Cabañas, F.J.; Rodriguez, A.; Montero, C. Carbonate molten salt solar thermal pilot facility: Plant design, commissioning and operation up to 700ºC. Renewable Energy 2020, 151, 528-541. [CrossRef]
- Nunes, V.M.B.; Lourenço, M.J.V.; Santos, F.J.V.; Nieto de Castro, C.A. Molten alkali carbonates as alternative engineering fluids for high temperature applications. Applied Energy 2019, 242, 1626-1633. [CrossRef]
- Tasidou, K.A.; Chliatzou, Ch.D.; Assael, M.J.; Antoniadis, K.D.; Mylona, S.K.; Huber, M.L.; Wakeham, W.A. Reference correlations for the viscosity of 13 inorganic molten salts. J. Phys. Chem. Ref. Data 2019, 48, 013101. [CrossRef]
- Tasidou, K.A.; Magnusson, J.M.; Munro, T.; Assael, M.J. Reference correlations for the viscosity of LiF-NaF-KF, LiF-BeF2, and Li2CO3-Na2CO3-K2CO3. J. Phys. Chem. Ref. Data 2019, 48, 043102.
- Ramires, M.L.V.; Nieto de Castro, C.A.; Nagasaka, Y.; Nagashima, A.; Assael M.J.; Wakeham, W.A. Standard reference data for the thermal conductivity of water. J. Phys. Chem. Ref. Data 1995, 24, 1377-1381. [CrossRef]
- Nieto de Castro, C.A.; Li, S.F.Y.; Nagashima, A.; Trengove, R.D.; Wakeham, W.A. Standard Reference Data for the Thermal Conductivity of Liquids. J. Phys. Chem. Ref. Data 1986, 15, 1073-1086. [CrossRef]
- Santos, F.J.V.; Nieto de Castro, C.A.; Dymond, J.H.; Dalaouti, N.K.; Assael, M.J.; Nagashima, A. Standard Reference Data for the Viscosity of Toluene. J. Phys. Chem. Ref. Data 2006, 35, 1-8. [CrossRef]
- Janz, G.J.; Allen, C.B.; Bansal, N.P.; Murphy, R.M.; Tomkins, R.P.T. Physical Properties Data Compilations. II. Molten Salts: Data on Single and Multicomponent Salt Systems, NSRDS-NBS 61, Part II, 1979.
- Araki, N.; Matsuura, M. Measurements of Thermophysical Properties of Molten Salts (Mixtures of Alkaline Carbonate Salts). Proceedings of the Eight Japan Symposium on Thermophysical Properties 1987, 1-4. [CrossRef]
- Liu, Q.; Lange, R.A. New density measurements on carbonate liquids and the partial molar volume of the CaCO3 component. Contrib. Mineral. Petrol. 2003, 146, 370-381. [CrossRef]
- Kojima, T.; Miyazaki, Y.; Nomura, K.; Tanimoto, K. Density, Surface Tension, and Electrical Conductivity of Ternary Molten Carbonate System Li2CO3–Na2CO3–K2CO3 and Methods for Their Estimation. J. Electrochem. Soc. 2008, 155(7), F150-F156.
- An, X.; Cheng, J.; Zhang, P.; Tang, Z.; Wang, J. Determination and evaluation of the thermophysical properties of an alkali carbonate eutectic molten salt. Farad. Disc. 2016, 190: 327-338. [CrossRef]
- Sang, L.; Ai, W.; Wu, Y.; Ma, C. Enhanced specific heat and thermal conductivity of ternary carbonate nanofluids with carbon nanotubes for solar power applications. Int. J. Energy Res. 2019, 1-10. [CrossRef]
- Kojima, T.; Yanagida, M.; Tanimoto, K.; Tamiya, Y.; Matsumoto, H.; Miyazaki, Y. The surface tension and the density of molten binary alkali carbonate systems. Electrochemistry (Tokyo, Jpn.) 1999, 67, 593-602. [CrossRef]
- Liao, M.; Wei, X.L.; Ding, J.; Hu, B.H.; Peng, Q. Preparation and experimental investigation for LNK carbonate molten salts. Acta Energ. Solar Sin. 2010, 31, 863-867.
- Zhang, Z.; Yuan, Y.; Zhang, N.; Sun, Q.; Cao, X.; Sun, L. Thermal properties enforcement of carbonate ternary via lithium fluoride: A heat transfer fluid for concentrating solar power systems. Renew. Energy 2017, 111, 523-531. [CrossRef]
- Sang, L.; Ai, W.; Liu, T.; Wu, Y.; Ma, C. Insights into the specific heat capacity enhancement of ternary carbonate nanofluids with SiO2 nanoparticles: the effect of change in the composition ratio. RSC Adv. 2019, 9, 5288-5294. [CrossRef]
- Otsubo, S.; Nagasaka, Y.; Nagashima, A. Experimental study on the forced Rayleigh scattering method using CO2 laser (3rd report, measurements of molten simple carbonates and their binary and ternary mixtures). Trans. Japan Soc. Mech. Engrs. Part B 1998, 64, 806-813. [CrossRef]
- Zhang, X.; Wicaksono, H.; Fujiwara, S.; Fujii, M. Accurate measurements of thermal conductivity and thermal diffusivity of molten carbonates. High Temp.-High Press. 2002, 34, 617-625. [CrossRef]
- Dokutovich, V.N.; Khokhlov, V.A.; Zakir’yanova, I.D. Thermal conductivity of composite materials: Alkali carbonate-based melts filled with fine α-Al2O3. Int. J. Heat Mass Transf. 2018, 119, 365-371. [CrossRef]
- Grosu, Y.; Anagnostopoulos, A.; Balakin, B.; Krupanek, J.; Navarro, M.E.; González-Fernández, L.; Ding, Y.; Faik, A. Nanofluids based on molten carbonate salts for high-temperature thermal energy storage: Thermophysical properties, stability, compatibility and life cycle analysis, Sol. Energy Mater. Sol. Cells. 2021, 220, 110838. [CrossRef]
- Nieto de Castro, C.A.; Lourenço, M.J.V. Towards the Correct Measurement of Thermal Conductivity of Ionic Melts and Nanofluids, Energies 2020, 13(1), 99.
- Lourenço, M.J.V.; Alves, M.; Serra, J.M.; Nieto de Castro, C.A.; Buschmann, M.H. The Thermal Conductivity of Near-Eutectic Galinstan (Ga68.4In21.5Sn10) Molten Alloy, Int. J. Thermophys. 2024, 45, 6. [CrossRef]
- Janz, G.J.; Saegusa, F. Molten carbonates as electrolytes: viscosity and transport properties. J. Electrochem. Soc. 1963, 110, 452-456. [CrossRef]
- Ejima, T.; Sato, Y.; Yamamura, T; Tamal, K.; Hasebe, M.; Bohn, M.S.; Janz, G.J. Viscosity of the Eutectic Li2CO3-Na2CO3-K2CO3 Melt. J. Chem. Eng. Data 1987, 32, 180-182.
- Ejima, T.; Sato, Y.; Yamamura, T.; Tamal, K.; Hasebe, M.; Bohn, M.S.; Janz, G.J. Viscosity Measurements: Molten Ternary Carbonate Eutectic. Proceedings of The Electrochemical Society, 1987, PV1987-7, 317-323. [CrossRef]
- Kim S.W.; Uematsu, K.; Toda, K.; Sato, M. Viscosity analysis of alkali metal carbonate molten salts at high temperature. J. Ceram. Soc. Jpn. 2015, 123, 355-358. [CrossRef]
- An, X; Cheng, J.; Su, T.; Zhang, P. Determination of thermal physical properties of alkali fluoride/carbonate eutectic molten salt AIP Conf. Proc. 2017, 1850, 070001.
- Starke, A.R.; Cardemil, J.M.; Bonini, V.R.B.: Escobar, R.; Castro-Quijada, M.; Videla, A. Assessing the performance of novel molten salt mixtures on CSP applications. Applied Energy 2024, 359, 122689. [CrossRef]


| Measuring Methoda | Declared Relative Uncertaintyb | Temperature Range /K | Purity of Samplec, /% | Number of Data Points/ Equation | Reference |
| A | 0.01 | 680 - 860 | NA | Equation | Janz et al. (1979) [11] |
| A | --- | 677 - 1081 | NA | Equation | Araki et al. (1987) [12] |
| DBA | 0.005 | 882 - 1158 | NA | #4 | Liu et al. (2003) [13] |
| MBP | 0.003 | 770 - 1163 | 99.5 | Equation | Kojima et al. (2008) [14] |
| A | --- | 723 - 893 | 99.9 | #4 | An et al. (2016) [15] |
| T/K | ρ /kg·m-3 | T/K | ρ /kg·m-3 | |
| 680 | 2085.1 | 950 | 1965.1 | |
| 710 | 2071.8 | 980 | 1951.8 | |
| 740 | 2058.5 | 1010 | 1938.4 | |
| 770 | 2045.1 | 1040 | 1925.1 | |
| 800 | 2031.8 | 1070 | 1911.7 | |
| 830 | 2018.5 | 1100 | 1898.4 | |
| 860 | 2005.1 | 1130 | 1885.1 | |
| 890 | 1991.8 | 1160 | 1871.7 | |
| 920 | 1978.4 | |||
| Measuring Methoda | Declared Relative Uncertaintyb |
Temperature Range /K |
Purity of Samplec /% |
Number of Data Points/ Equation | Reference |
| DC | 0.01 | 680 - 1100 | ---- | Equation | Janz et al. (1979) [11] |
| DSC | --- | 723 - 873 | 99.9 | Equation | An et al. (2016) [15] |
| STA | -- | 703 - 1023 | ---- | Equation | Liao et al. (2010) [18] |
| DSC | 0.01 | 723 - 873 | > 99 | Equation | Zhang et al. (2017) [19] |
| DSC | 0.03 | 773 - 833 | 99.7 | #4; #3 | Sang et al. (2019a, 2019b) [16,20] |
| Measuring Methoda | Declared Relative Uncertaintyb | Temperature Range /K | Purity of Samplec /% | Number of Data Points/ Equation | Reference |
| LF (Calc) | -- | 673 - 764 | NA | Equation | Araki et al. (1987) [12] |
| FRS (Calc) | -- | 680 - 1030 | NA | Equation | Otsubo et al. (1998) [21] |
| THW | 0.03 | 803 - 943 | NA | Plot | Zhang et al. (2002) [22] |
| LF (Calc) | -- | 723 - 873 | NA | #4 | An et al. (2016) [15] |
| SSCC | 0.03 | 670 - 930 | pure | Equation | Dokutovich et al. (2018) [23] |
| LF (Calc) | 0.03 | 773 - 833 | > 99 | #4 | Sang et al. (2019) [16] |
| LF (Calc) | 0.05 | 693 - 823 | NA | #4 | Grosu et al. (2021) [24] |
| Measuring Methoda | Declared Relative Uncertaintyb | Temperature Range /K | Purity of samplec /% | Number of Data Points/ Equation | Reference |
| OCY | 0.25 | 760 - 870 | NA | Equation | Janz et al. (1979) [11] |
| OCY | 0.03 | 890 - 1175 | 98.4 | #15 | Ejima et al. (1987) [28,29] |
| RC | NA | 700-970 | NA | Diagram | Liao et al. (2010) [18] |
| RC | 0.05 | 690 - 1120 | 99.8 | #6 | Kim et al. (2015) [30] |
| RC | NA | 740 - 920 | 99.9 | Equation | An et al. (2016) [15] |
| RC | NA | 740 - 890 | 99.9 | Equation | An et al. (2017) [31] |
| RPP | NA | 673 - 773 | 99 | Diagram | Grosu et al. (2021) [24] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




