Submitted:
08 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
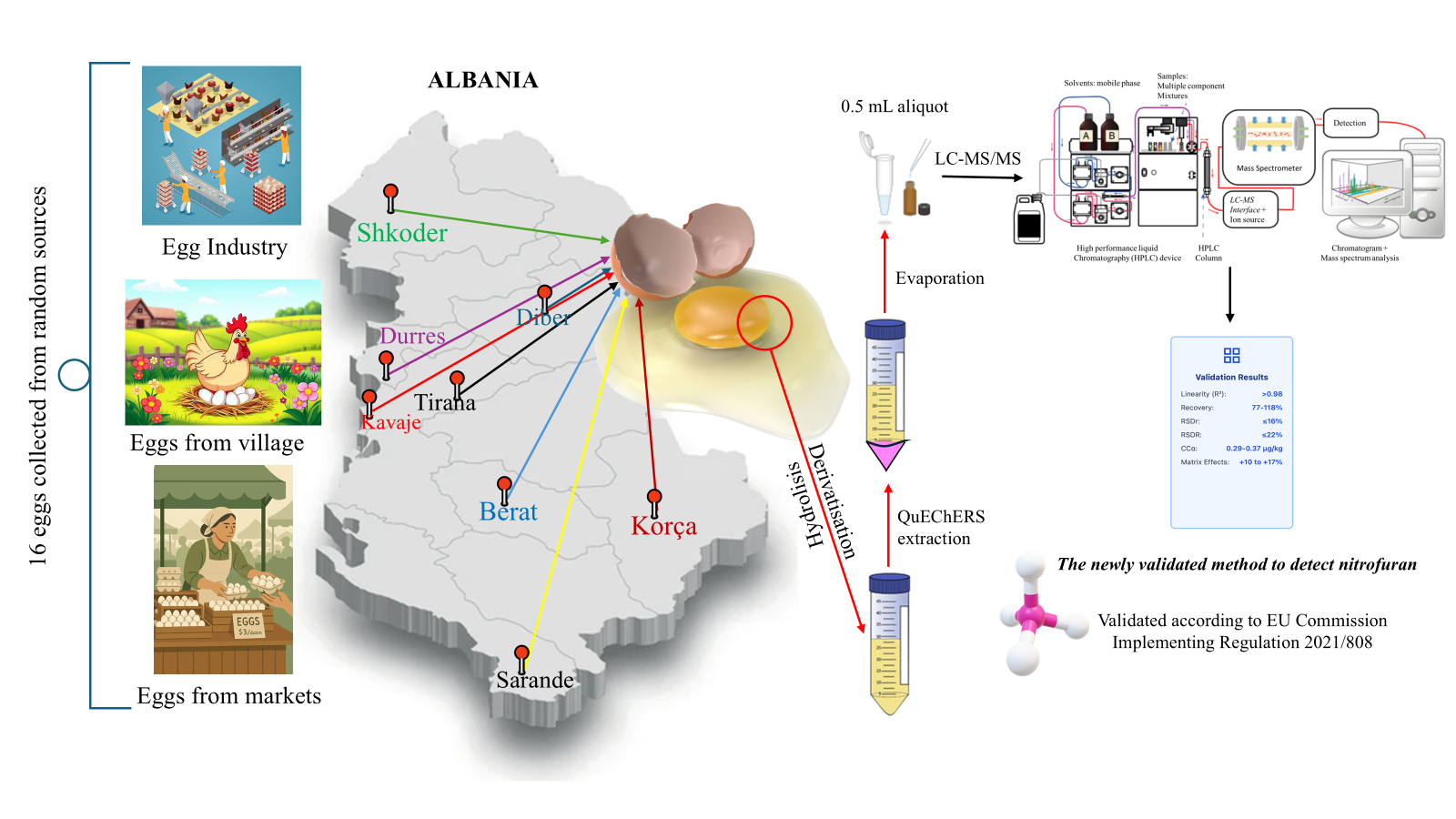
Material and Methods
Chemicals and Reagents
Standards
Standard Preparation
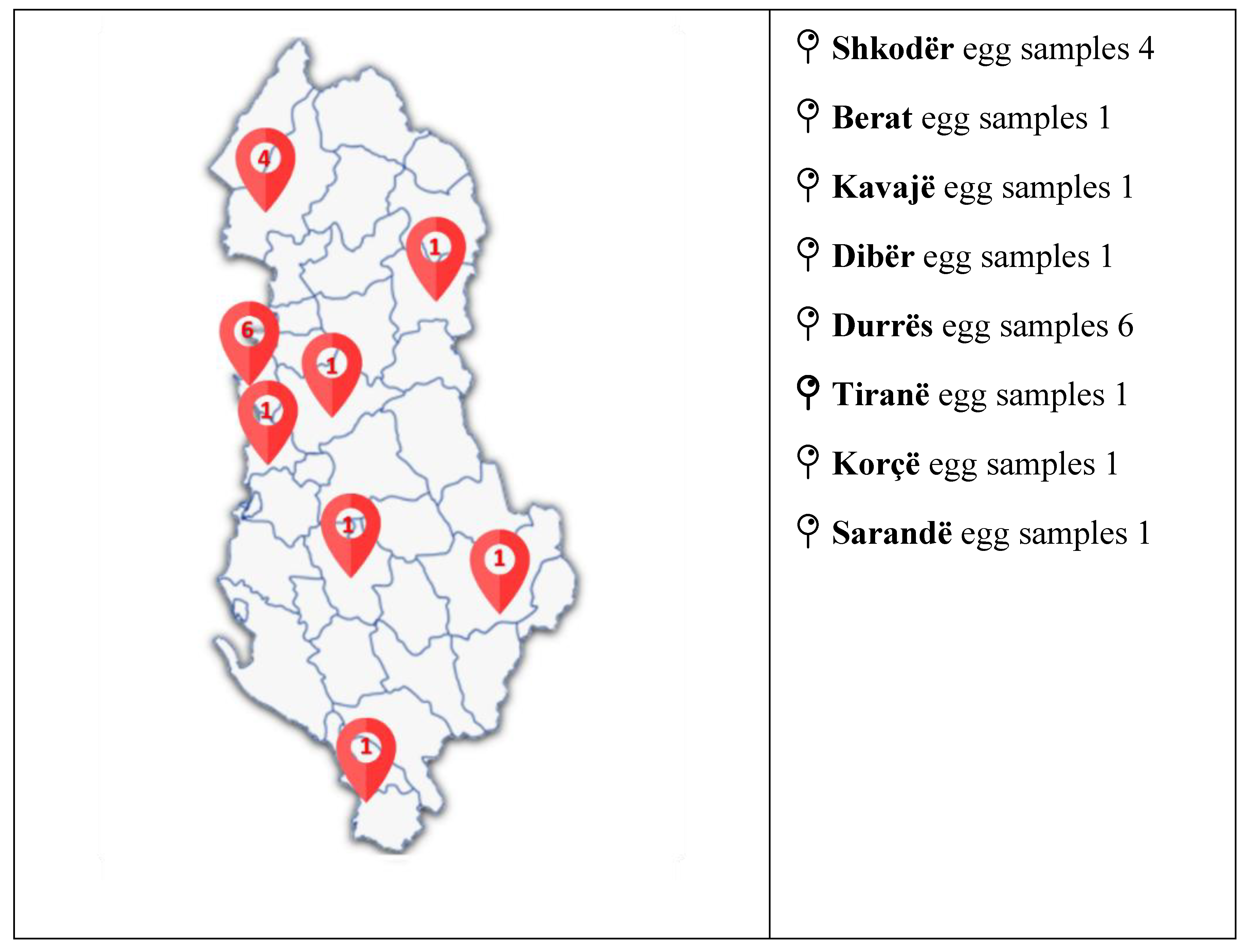
Sample Preparation

Instrument
LC-MS/MS Conditions
Method Validation
Identification
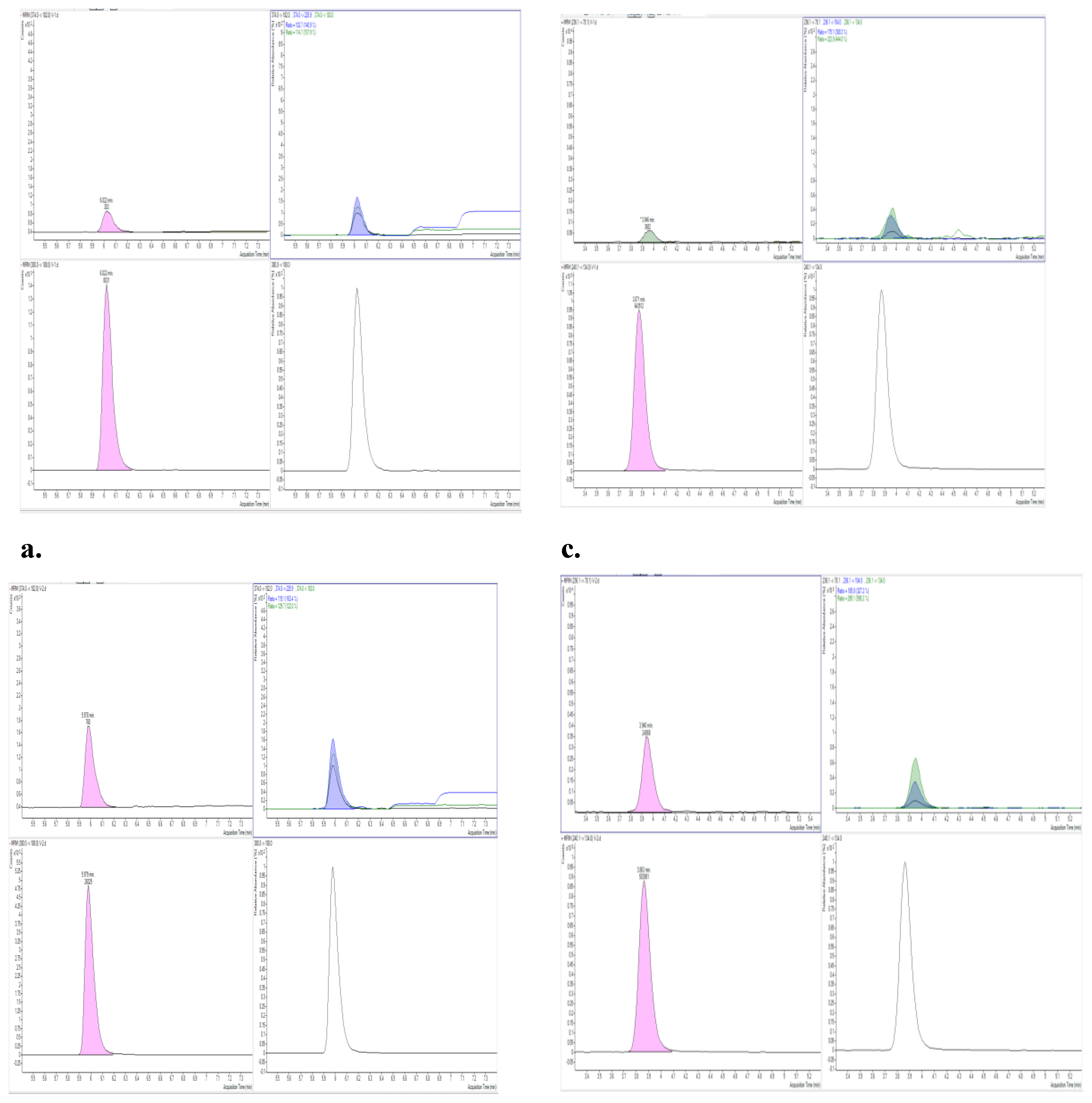
Selectivity and Specificity
Matrix Effects
Trueness, Precision, and Decision Limit (CCα)
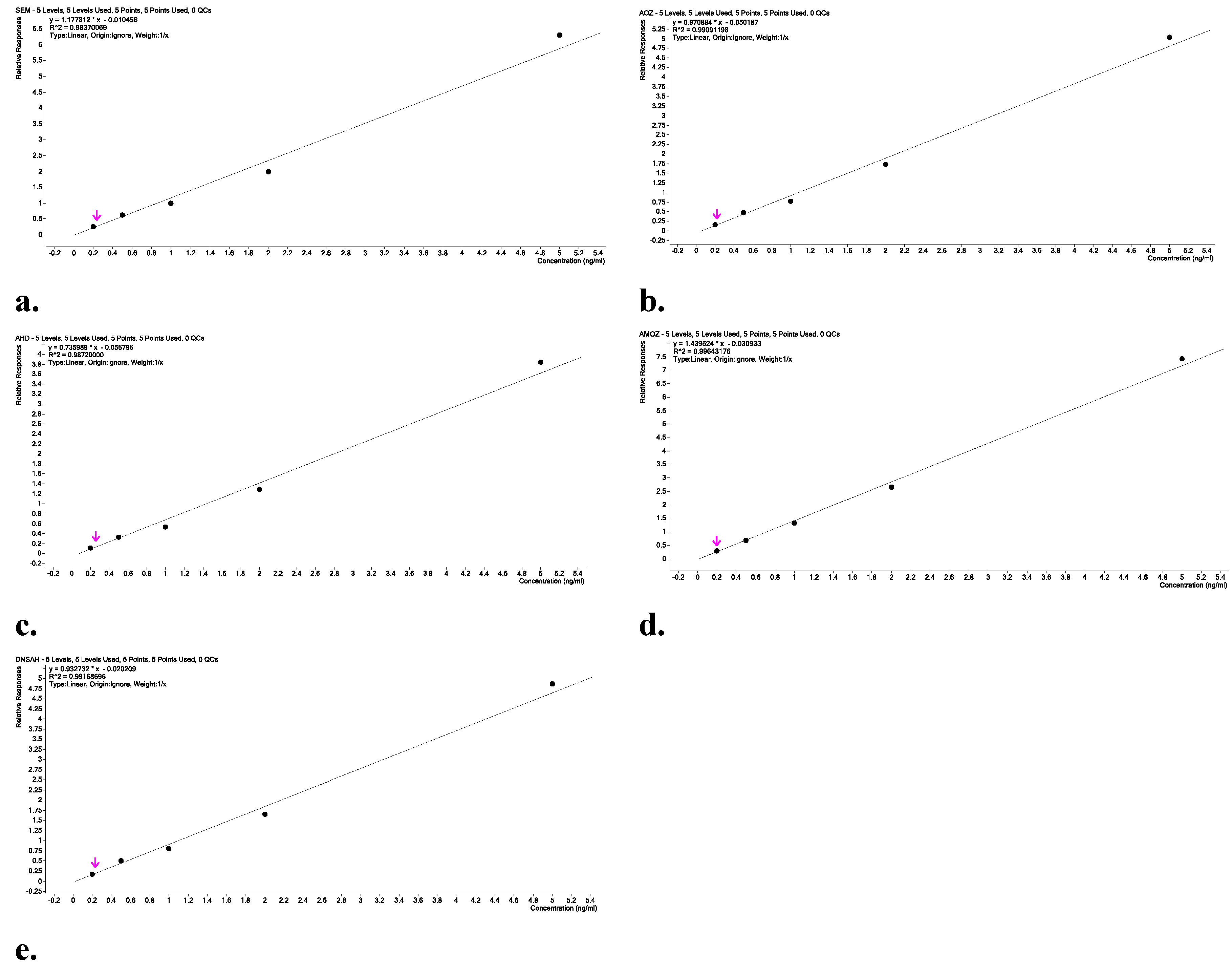
Linearity
Stability and Roughness
Results and Discussion
Chromatographic Performance and Mass Spectrometric Detection
Matrix Effect
Recovery and Precision
Sensitivity and Decision Limits
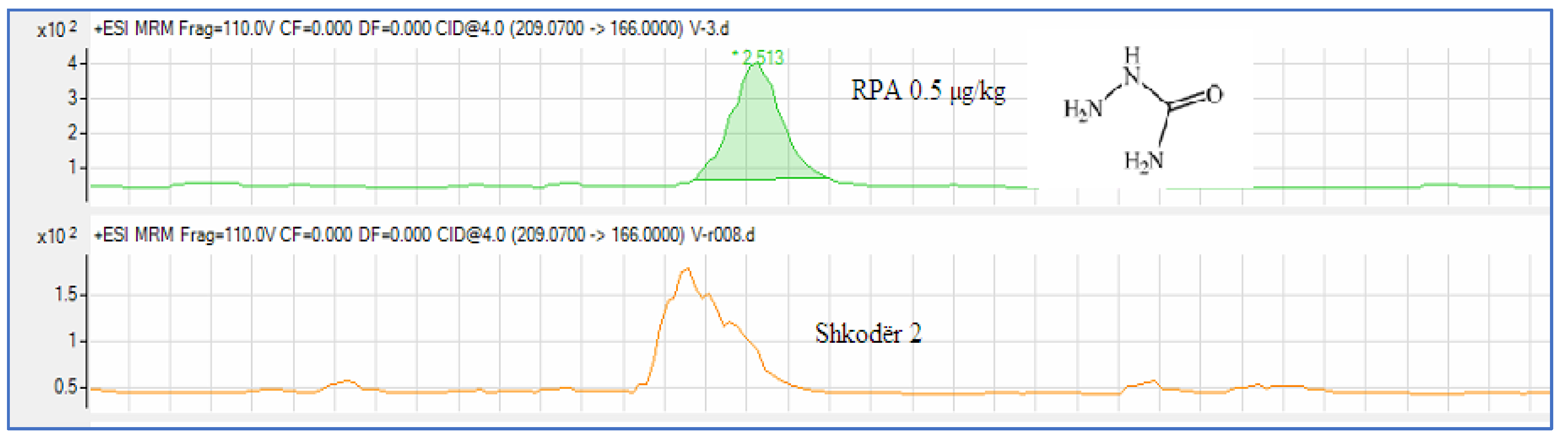
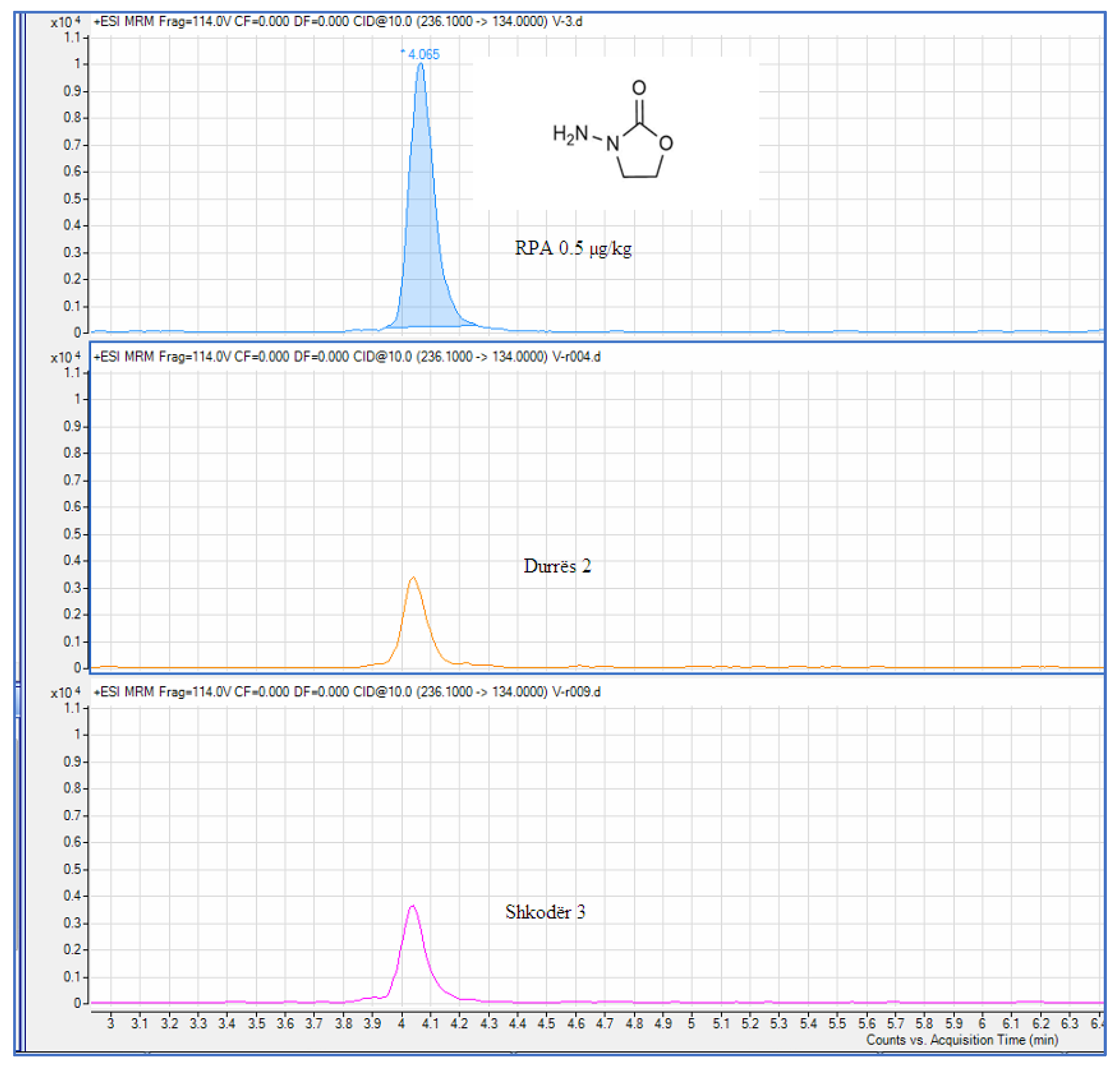
Analysis of a Real Sample
| Origin |
SEM (µg/kg) |
AOZ (µg/kg) |
RPA (µg/kg) |
|
| 1 | Shkodër 2 | 0,240 | 0,5 | |
| 2 | Durrës 2 | 0,227 | 0.5 | |
| 3 | Shkodër 3 | 0,271 | 0.5 |
Stability Data
| Day 0 | 4 Months | 6 Months | WLR | |||
| Analyte | Measured concentration (µg/kg) | Measured concentration (µg/kg) | Loss (%) | Measured concentration (µg/kg) | Loss (%) | |
| SEM | 0,893 | 0,808 | 10% | 0,682 | 32% | 16,20% |
| AHD | 0,943 | 0,856 | 9% | 0,780 | 22% | 10,47% |
| AOZ | 0,893 | 0,886 | 1% | 0,835 | 16% | 8,78% |
| AMOZ | 0,893 | 0,849 | 5% | 0,687 | 31% | 12,47% |
| DNSAH | 0,866 | 0,868 | 0% | 0,861 | 14% | 6,65% |
| AOZ incurred | 1,305 | 1,308 | 0% | 1,306 | 0% | N/A |
Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Artun YIBAR, Bülent OKUTAN, Saime GÜZEL. Effects of Boiling on Nitrofuran AOZ Residues in Commercial Eggs. Kafkas Universitesi Veteriner Fakultesi Dergisi 2013, 19 (6): 1023-1028. [CrossRef]
- Barbosa, J., Moura, S., & Barbosa, J. Analysis of nitrofuran residues in meat products: Development and validation of a confirmatory method using LC–MS/MS. Analytica Chimica Acta, 2007, 586(1-2), 359–365.
- César Aquiles Lázaro de la Torre, Juan Espinoza Blanco, Joab Trajano Silva, Vânia Margaret Flosi Paschoalin, Carlos Adam Conte Júnior. Chromatographic detection of nitrofurans in foods of animal origin. FOOD SAFETY. 2014, p. 1-9. [CrossRef]
- Commission Implementing Regulation (EU) 2021/808, laying down rules for the performance of analytical methods and the interpretation of results in the official control of residues of pharmacologically active substances in food of animal origin. Official Journal of the European Union, 22 March 2021, L172, 9.
- Commission Regulation (EC) 1442/95. Official Journal of the European Communities 1995, L143, p. 26.
- Commission Regulation (EU) 2019/1871 of 7 November 2019 on reference points for action for non-allowed pharmacologically active substances present in food of animal origin and repealing Decision 2005/34/EC. Off J Eur Union. 2019; L289.
- Connely A., Nugget A., O'Keeffe M., Mulder P.P.J., van Rhijn J.A., Kovacsics L., Fodor A., McCracken R.J., Kennedy D.G.: Isolation of bound residues of nitrofuran drugs from tissue by solid-phase extraction with determination by liquid chromatography with UV and tandem mass spectrometric detection. Anal Chim Acta 2003, 483(1-2), pp. 91-98.
- De la Calle MB and Szilagyi S. Determination of semicarbazide in fresh egg and whole egg powder by liquid chromatography/tandem mass spectrometry: interlaboratory validation study. Journal of AOAC International, 2006, 89, 1664–1671. [CrossRef]
- Douny, C., Widart, J., De Pauw, E., Silvestre, F., Kestemont, P., Tu, H. T., Phuong, N. T., Maghuin-Rogister, G., & Scippo, M. L. Development of an analytical method to detect metabolites of nitrofurans: Application to the study of furazolidone elimination in Vietnamese black tiger shrimp (Penaeus monodon). Aquaculture, 2013, 376–379, 54–58.
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on nitrofurans and their metabolites in food. EFSA Journal 2015, 13(6), 4140.
- Fernando Ramos, Lúcia Santos, Jorge Barbosa. Chapter 43-Nitrofuran Veterinary Drug Residues in Chicken Eggs. Egg Innovation and Strategies for Improvements, Oxford: Academic Press; 2017, 457-464.
- Finzi, J. K., Donato, J.L., Sucupira, M., & De Nucci, G. Determination of nitrofuran metabolites in poultry muscle and eggs by LC-MS/MS. Journal of Chromatography B, 2005, 824(1), 30–35.
- Gemma Regan, Mary Moloney, Melissa Di Rocco, Padraig McLoughlin, Wesley Smyth, Steven Crooks, Christopher Elliott, Martin Danaher. Development and validation of a rapid LC–MS/MS method for the confirmatory analysis of the bound residues of eight nitrofuran drugs in meat using microwave reaction. Analytical and Bioanalytical Chemistry 2022, 414:1375–1388. [CrossRef]
- Guichard, P., Laurentie, M., & Hurtaud-Pessel, D., Verdon, E. Confirmation of five nitrofuran metabolites, including nifursol metabolite, in meat and aquaculture products by liquid chromatography-tandem mass spectrometry: Validation according to European Union Decision 2002/657/EC. Food Chemistry, 2021, 342, 128389. [CrossRef]
- Hoenicke K, Gatermann R, Hartig L, Mandix M, Otte S, Formation of semicarbazide (SEM) in food by hypochlorite treatment: is SEM a specific marker for nitrofurazone abuse. Food Addit Contam 2004, 21(6):526-37. [CrossRef]
- Hoogenboom, L. A. P., Tomassini, O., & Kuiper, H. A. Persistent residues of nitrofuran metabolites in animal tissues. Food Additives & Contaminants, 1991, 8(3), 623–632.
- Hoogenboom L. A. P., Berghmans M. C. J., Polman T. H. G., Parker R., Shaw I.C. Depletion of protein-bound furazolidone metabolites containing the 3-amino-2-oxazolidinone side-chain from liver, kidney, and muscle tissues from pigs. Food Additives and Contaminants 1992, 9: 623–630.
- Hoogenboom, L.A., van Bruchem, G.D., Sonne, K., Enninga, I.C., van Rhijn, J.A., Heskamp, H., Huveneers-Oorsprong, M.B., van der Hoeven, J.C., Kuiper, H.A. Absorption of a mutagenic metabolite released from protein-bound residues of furazolidone. Environmental Toxicology and Pharmacology, 2002, 11(3-4): 273-287. [CrossRef]
- Horne E., Cadogan A., O'Keeffe M., Hoogenboom L.A.P.: Analysis of protein-bound metabolites of furazolidone and furaltadone in pig liver by high-performance liquid chromatography and liquid chromatography mass spectrometry. Analyst 1996, 121, 1463-1469. [CrossRef]
- Ina Pasho, Kozeta Vaso, and Landi Dardha. Nitroimidazoles in Albanian honey samples by LC-MS/MS analysis. Food Additives and Contaminants: Part B Surveillance(Taylor & Francis). 2025, Volume 18, Issue 2, 143-154. [CrossRef]
- Jorge Barbosa, Andreia Freitas, José Luis Mourao, Maria Irene Noronha da Silveira, and Fernando Ramos, Determination of Furaltadone and Nifursol Residues in Poultry Eggs by Liquid Chromatography-Electrospray Ionization Tandem Mass Spectrometry Lisboa, Portugal. J. Agric. Food Chem. 2012, 60, 4227-4234. [CrossRef]
- K. M. Cooper; J. Le; C. Kane; D. G. Kennedy. Kinetics of semicarbazide and nitrofurazone in chicken eggs and egg powders. Food Additives & Contaminants. Part A - Chemistry, Analysis, Control, Exposure & Risk Assessment. 2008, 25:6, 684-692. [CrossRef]
- Kozeta VASO, Ilir AJDINI, Ina Pasho, Elmira Marku, Jonida CANAJ, Erinda PLLAHA, Suela TEQJA. Presence Of Nitrofurans In Eggs: Legislation, Regulatory Measures, And Analytical Detection Methods. 3rd International Conference on Trends in Advanced Research, ICCTAR, Konya, Turkey, April 04-05, 2025. Proceedings p.205-219.
- Leitner A., Zoller P., Linder W. Determination of the metabolites of nitrofuran antibiotics in animal tissue by high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr A 2001, 939, 49-59.
- Luciano Molognoni, Heitor Daguer, Rodrigo Barcellos Hoff. Chapter 12 - Analysis of nitrofurans residues in foods of animal origin. Food Toxicology and Forensics. 2021, p 379-419.
- M. Vass, K. Hruska, M. Franek. Nitrofuran antibiotics: a review on the application, prohibition, and residual analysis. Veterinární medicína, 2008, 53(9):469-500. [CrossRef]
- McCracken R.J., Blanchflower W.J., Rowan C., McCoy M.A., Kennedy D.G. Determination of furazolidone in porcine tissue using thermospray liquid chromatography-mass spectrometry and a study of the pharmacokinetics and stability of its residues. Analyst 1995, 120, 2347-2356. [CrossRef]
- McCracken, R. J., & Kennedy, D. G. Detection, accumulation, and distribution of nitrofuran residues in egg yolk, albumen, and shell. Food Additives & Contaminants, 1997, 14(5), 507–513. [CrossRef]
- McCraken R.J., Spence D.E., Floyd S.D., Kennedy D.G.: Evaluation of the residues of furazolidone and its metabolite, 3-amino-2-oxazolidone (AOZ), in eggs. Food Additives & Contaminants 2001, 18(11), 954-959.
- McCracken, R. J., & Kennedy, D. G. Detection, accumulation, and distribution of nitrofuran residues in egg yolk, albumen, and shell. Food Addit Contam. 2007, 24(1):26-33. [CrossRef]
- Moragues, F., Miralles, P., Igualada, C., Coscollà, C. Determination of nitrofuran metabolites and nifurpirinol in animal tissues and eggs by ultra-high performance liquid chromatography-tandem mass spectrometry validated according to Regulation (EU) 2021/808. Heliyon, 2024, 10(6), 1-10, e27889. [CrossRef]
- Mottier, P., Parisod, V., & Gremaud, E. Determination of nitrofuran residues in meat by LC–MS/MS. Journal of Chromatography B, 2003, 789(2), 313–322.
- Murielle Gaugain, Juliette Durot, Laurine Levé, Estelle Dubreil-Chéneau, Pierre Guichard, Sophie Bourcier, Eric Verdon, Dominique Hurtaud-Pessel. Multiclass Method to Analyze Banned Veterinary Drugs in Casings by LC-MS/MS: A Validation Study According to Regulation (EU) 2021/808. Journal of Agricultural and Food Chemistry. 2025, online p. 1-13. [CrossRef]
- Pietro Picconi, Priya Prabaharan, Jennifer L Auer, Stephanie Sandiford, Francesco Cascio, Madiha Chowdhury, Charlotte Hind, Matthew E. Wand, J Mark Sutton, Khondaker M. Rahman. Novel pyridyl nitrofuranyl isoxazolines show antibacterial activity against multiple drug-resistant Staphylococcus species. Bioorganic & Medicinal Chemistry. 2017, Volume 25, Issue 15, 1, 3971-3979. [CrossRef]
- Report of the 40th Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), Evaluation of certain veterinary drug residues in food. World Health Organization, Geneva, 1993, p. 32–42.
- Sepideh Rezaei, Sholeh Akbari, Farzad Rahmani, Sara Dabbaghi Varnousfaderani, Saeideh Gomroki, Emad Jafarzadeh. Nitrofurans as Potent Antibacterial Agents: A Systematic Review of Literature. International Journal of Advanced Biological and Biomedical Research. 2022, Volume 10, Issue 2, pp. 126-138.
- Steven J Lehotay , Maïwenn Le Floch, Alan R Lightfield, Pierrick Couëdor, Dominique Hurtaud-Pessel, Nicolás Michlig, Eric Verdon. Stability study of selected veterinary drug residues spiked into extracts from different food commodities. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2023, 40(9):1198-1217. [CrossRef]
- Szilagyi S and De la Calle B. Development and validation of an analytical method for the determination of semicarbazide in fresh egg and in egg powder based on the use of liquid chromatography tandem mass spectrometry. Analytica Chimica Acta, 2006, 572, 113–120. [CrossRef]
- Tomasz Śniegocki, Andrzej Posyniak, and Jan Żmudzki, Determination of Nitrofuran Metabolite Residues In Eggs by Liquid Chromatography-Mass Spectrometry, Bull Vet Inst Pulawy 2008, 52(3), 421-425.
- Vermeulen, A., Environment, human reproduction, menopause, and andropause. Environ Health Perspect. 1993, 101 Suppl 2: 91-100.
- Vroomen, L. H. M., Berghams, M. C. J., van Leeuwen, P., van der Struijs, T. B. D., de Vries, P. H. U., & Kuiper, H. A. Formation and persistence of nitrofuran metabolites in animal tissues. Food Additives & Contaminants, 1986, 3(3), 331–346.
- Zhang, Y., Zhang, Y., & Chen, Y. Application of LC–MS in the determination of nitrofurans in animal tissues. Journal of Chromatography A, 2009, 1216(46), 7974–7980.
| Name | IUPAC Name | Molecular formula | Metabolite | Structure | ||
| 1 | AOZ | 3-Amino-2-oxazolidinone | 3-amino-1,3-oxazolidin-2-one | C3H6N2O2 | Furazolidone |  |
| 2 | AMOZ | 3-Amino-5-morpholinomethyl-2-oxazolidinone | 3-amino-5-(morpholin-4-ylmethyl)-1,3-oxazolidin-2-one | C8H15N3O3 | Furaltadone | 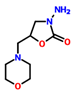 |
| 3 | AHD | 1-Amino hydantoin | 1-aminoimidazolidine-2,4-dione | C3H5N3O2 | Nitrofurantoine | 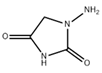 |
| 4 | SEM | Semicarbazide | Aminourea | CH5N3O3 | Nitrofurazone | 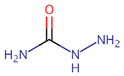 |
| 5 | DNSAH | 3,5-Dinitro-salicilik acid hydrazid | 2-hydroxy-3,5-dinitrobenzohydrazide | C7H6N4O6 | Nifursol | 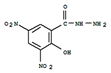 |
| Analyte | Precursor (m/z) | Product (m/z) | Fragmentor (V) | CE (V) | Polarity |
| NP-AHD | 249.1 | 133.9/103.9 | 140 | 8/24 | positive |
| NPA-AHD 13C3 | 252.1 | 133.9 | 140 | 8 | positive |
| NP-AMOZ | 335.1 | 291.1/262.1 | 76 | 6/14 | positive |
| NP-AMOZ-D5 | 340.1 | 296.1 | 80 | 8 | positive |
| NP-AOZ | 236.1 | 134/104 | 114 | 10/22 | positive |
| NP-AOZ-D4 | 240.1 | 134 | 140 | 8 | positive |
| NP-DNSAH | 374.0 | 225.9/183 | 140 | 24/28 | negative |
| NP-DNSAH 13C6 | 380.0 | 188 | 120 | 20 | negative |
| NP-SEM | 209.1 | 192/133.9 | 110 | 8/8 | positive |
| NP-SEM 13C 15N2 | 212.1 | 167.9 | 110 | 4 | positive |
| Parameters | WLR trueness (%) |
RSDr (%) |
RSDR (%) |
CCα (μg/kg) | ||||||
| Analyte | L1 | L2 | L3 | L 1 | L2 | L 3 | L 1 | L 2 | L 3 | |
| NP-AHD | 100 | 103 | 87 | 4,7 | 4,9 | 5,0 | 10,8 | 10,5 | 9,0 | 0.31 |
| NP-AOZ | 93 | 97 | 82 | 3,5 | 5,3 | 5,6 | 13,2 | 8,8 | 9,7 | 0.32 |
| NP-AMOZ | 98 | 98 | 86 | 9,6 | 7,7 | 16,1 | 21,6 | 12,5 | 17,3 | 0.37 |
| NP-DNSAH | 107 | 109 | 86 | 4,5 | 6,8 | 8,2 | 6,9 | 6,7 | 9,0 | 0.29 |
| NP-SEM | 100 | 103 | 87 | 7,0 | 6,5 | 7,5 | 12,5 | 16,2 | 10,1 | 0.32 |
| Analytes | AHD | AMOZ | AOZ |
| Maximum values (µg/kg) | 1,51 | 2,01 | 1,18 |
| Assigned values (µg/kg) | 1,05 | 1,39 | 0,82 |
| Minimum values (µg/kg) | 0,59 | 0,77 | 0,46 |
| Measured concentration (µg/kg) | 1,01 | 1,20 | 0,88 |
| Z-score | -0,17 | -0,62 | 0,34 |
| Measured concentration with Halo column (µg/kg) | 1,08 | 1,43 | 0,90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




