Submitted:
08 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Oxygen–Glucose Deprivation
2.3. EF5 Staining
2.4. Immunofluorescence Assay
2.5. Cell Viability Assays
2.5.1. MTT Assay
2.5.2. Neutral Red Assay
2.5.3. Annexin V/PI Flow Cytometry
2.6. ATP Content
2.7. Oxygen Consumption Rate Assessment
2.8. Western Blot
2.9. Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
2.10. Statistical Analyses
3. Results
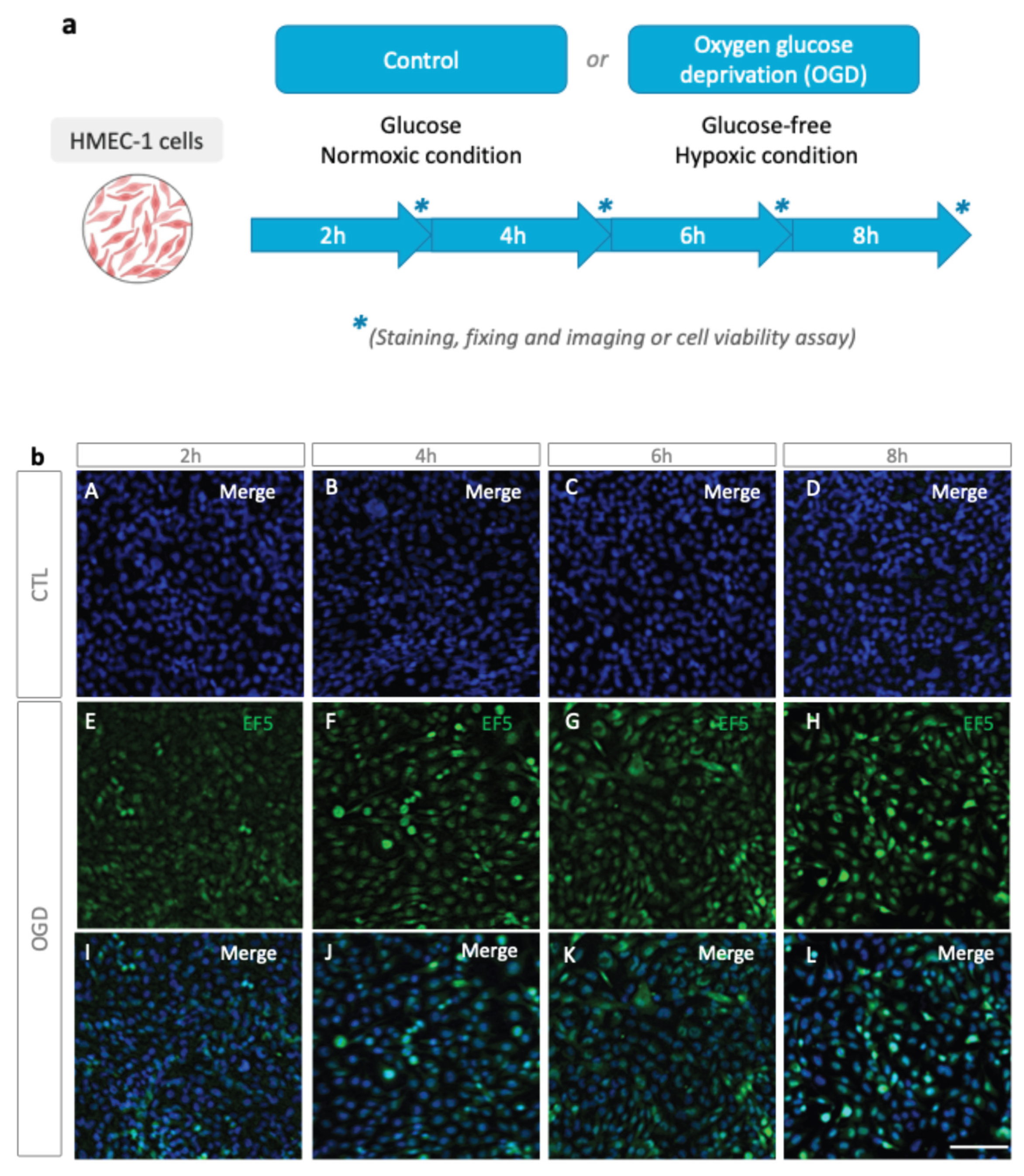
3.1. GasPak OGD Induces Hypoxia in HMEC-1 Cells
3.1.1. Establishing the In Vitro OGD Model
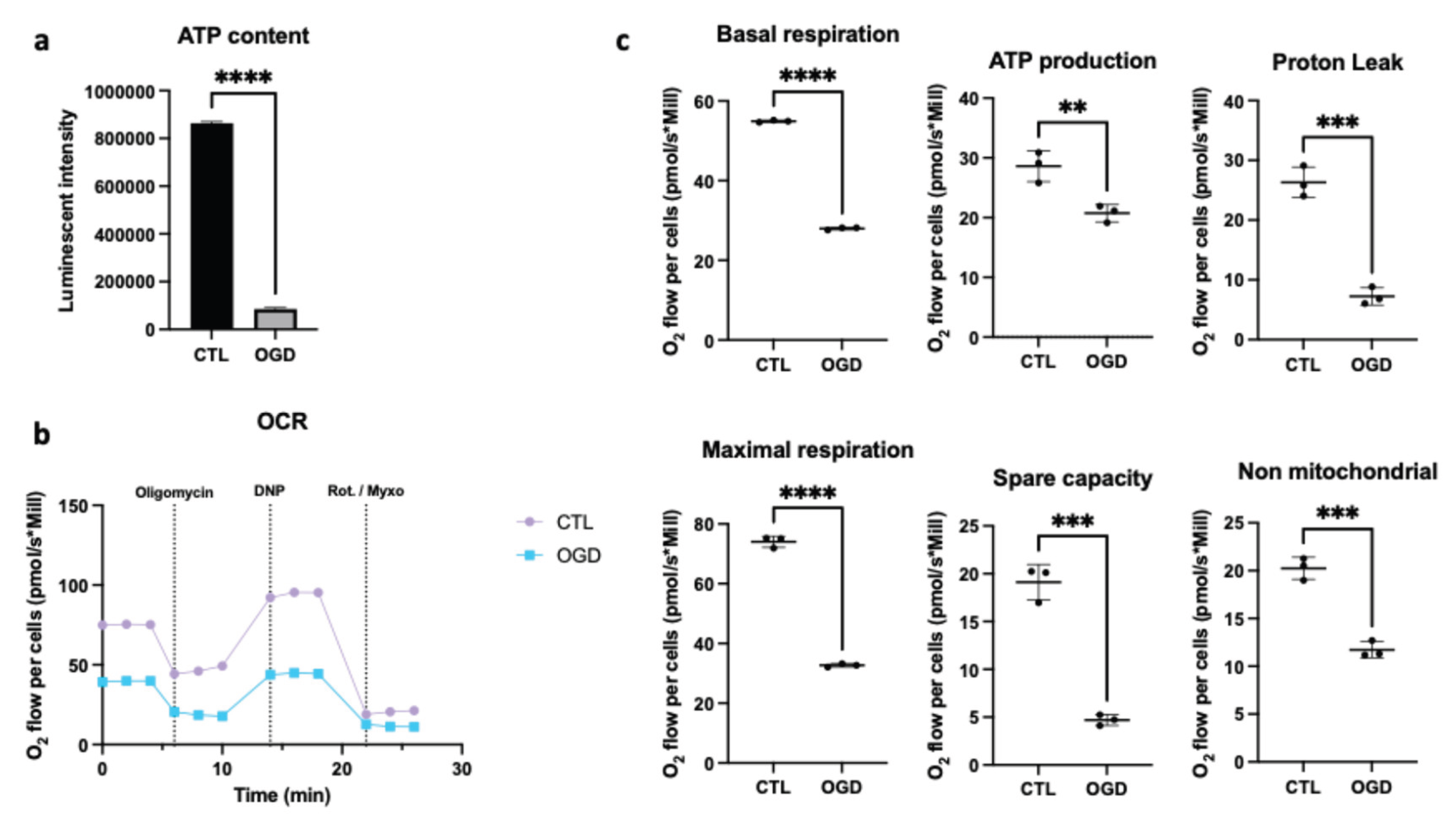
3.1.2. OGD Alter Mitochondria Function and Metabolic Plasticity in HMEC-1 Cells
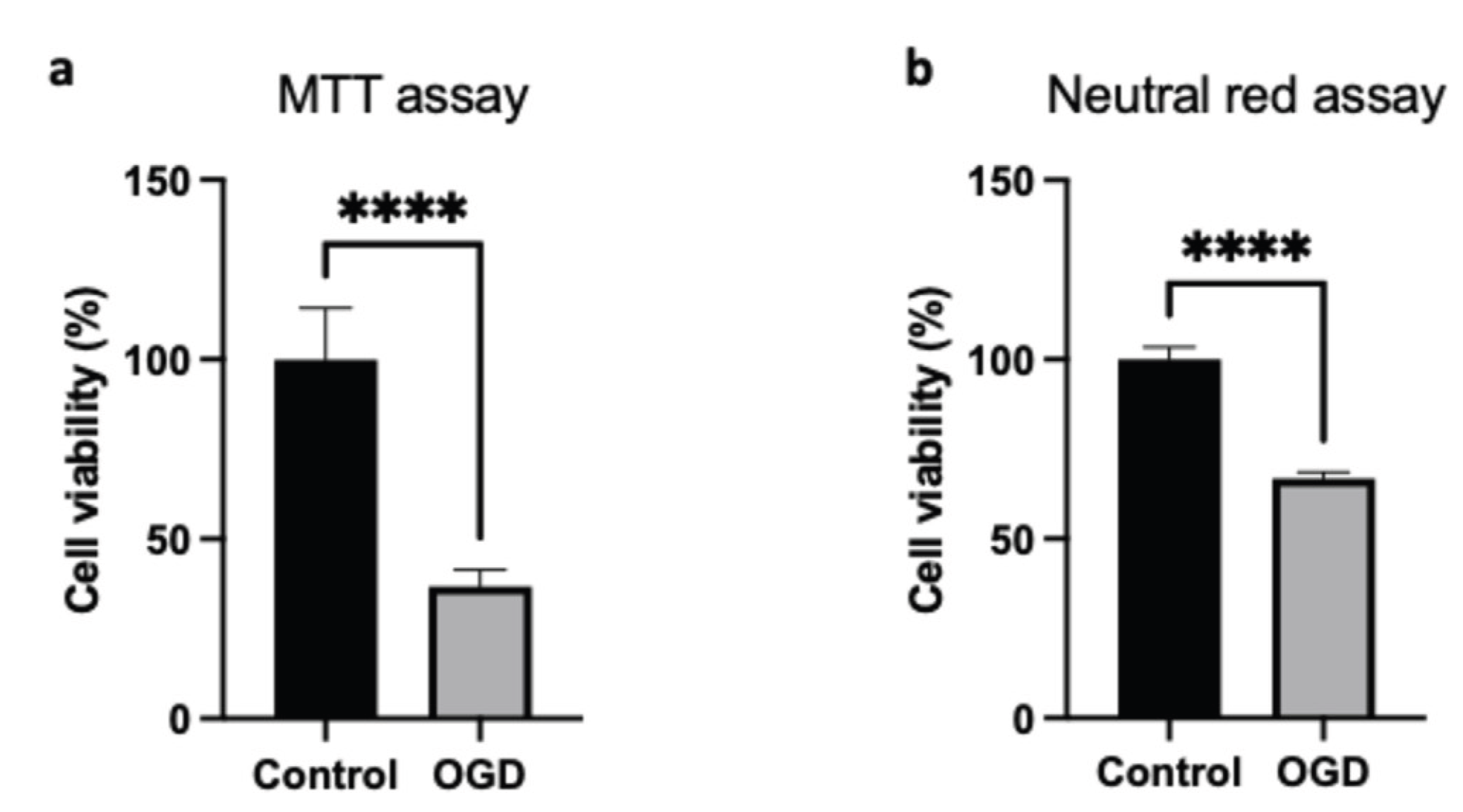
3.2. OGD Causes Cell Death in HMEC-1 Cells
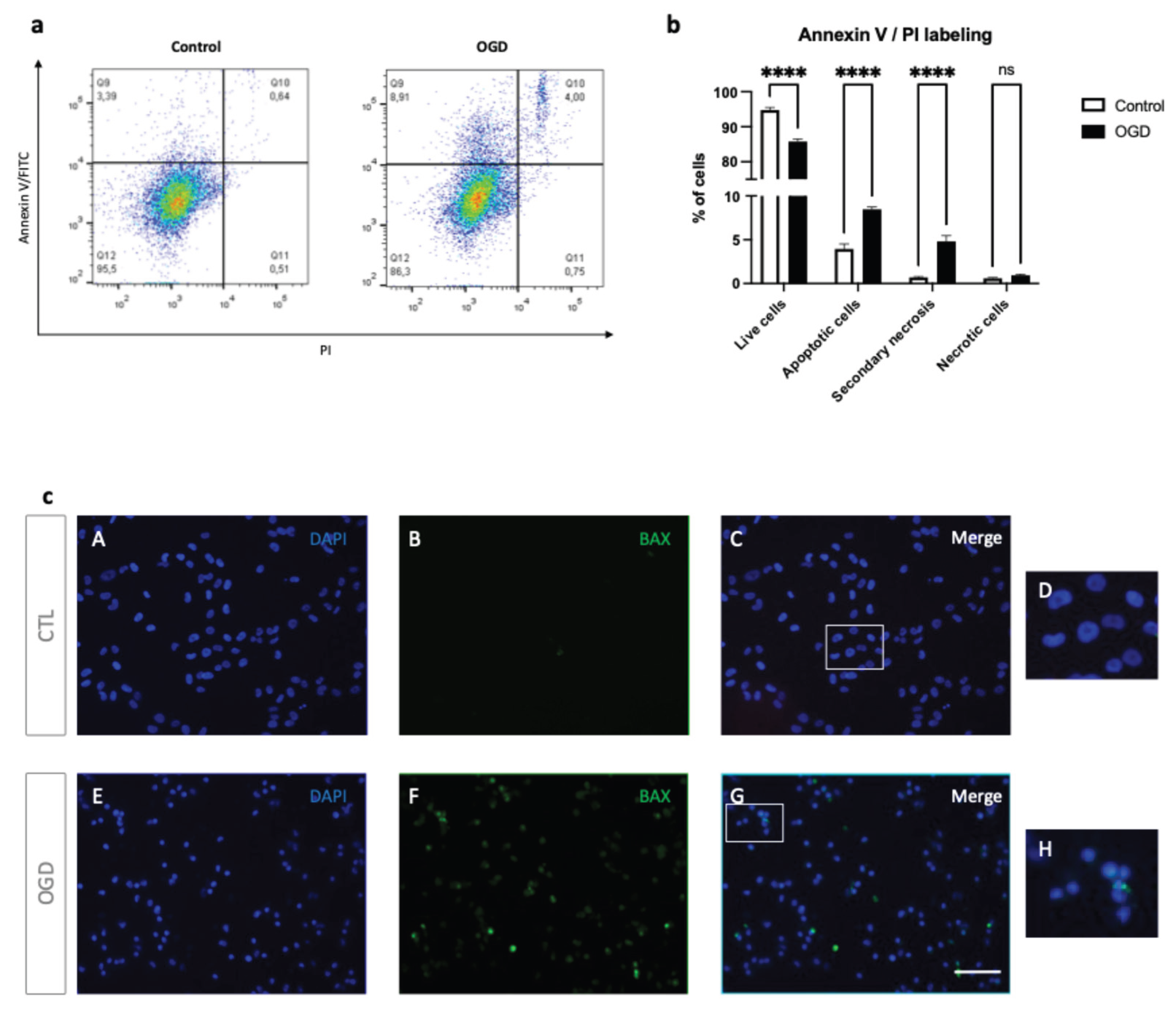
3.3. OGD Induces Apoptosis in HMEC-1 Cells
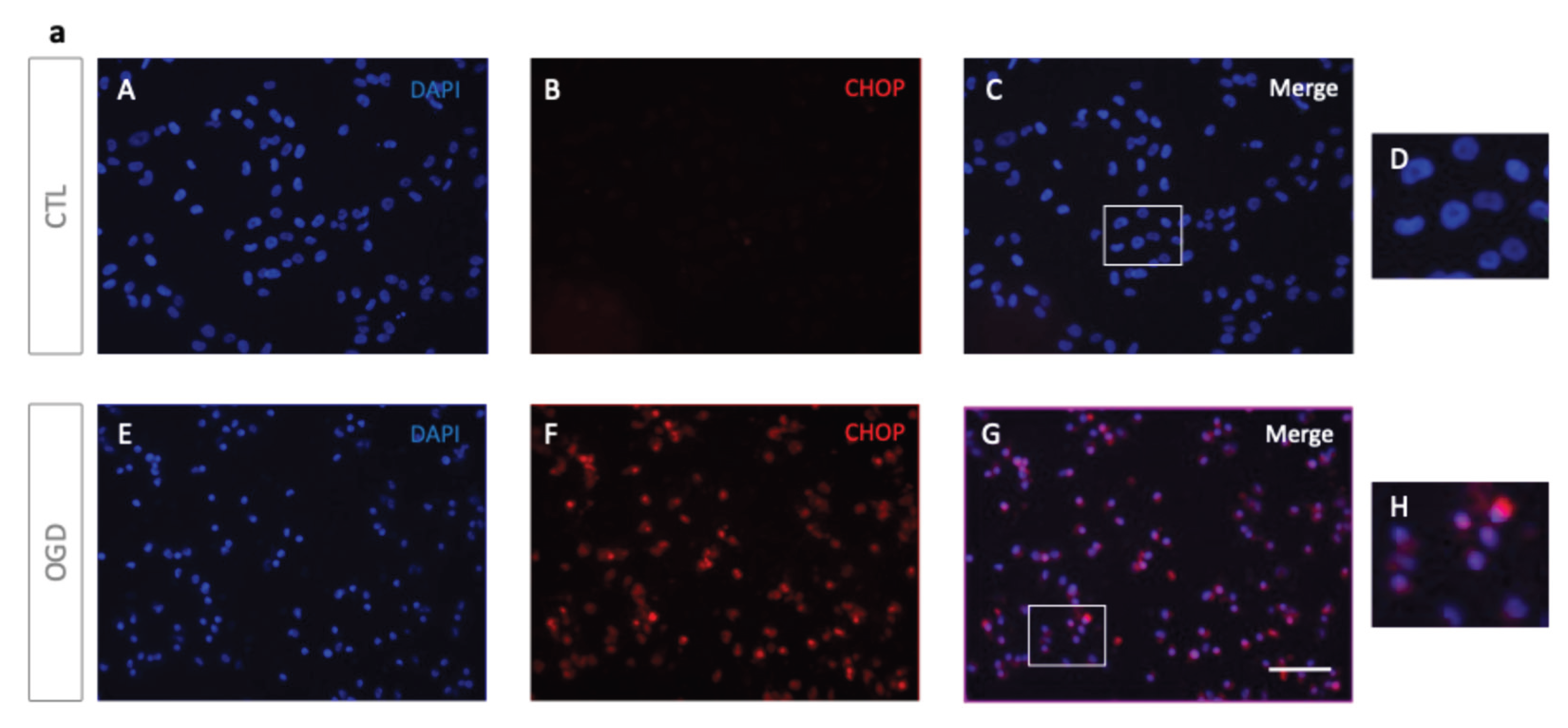
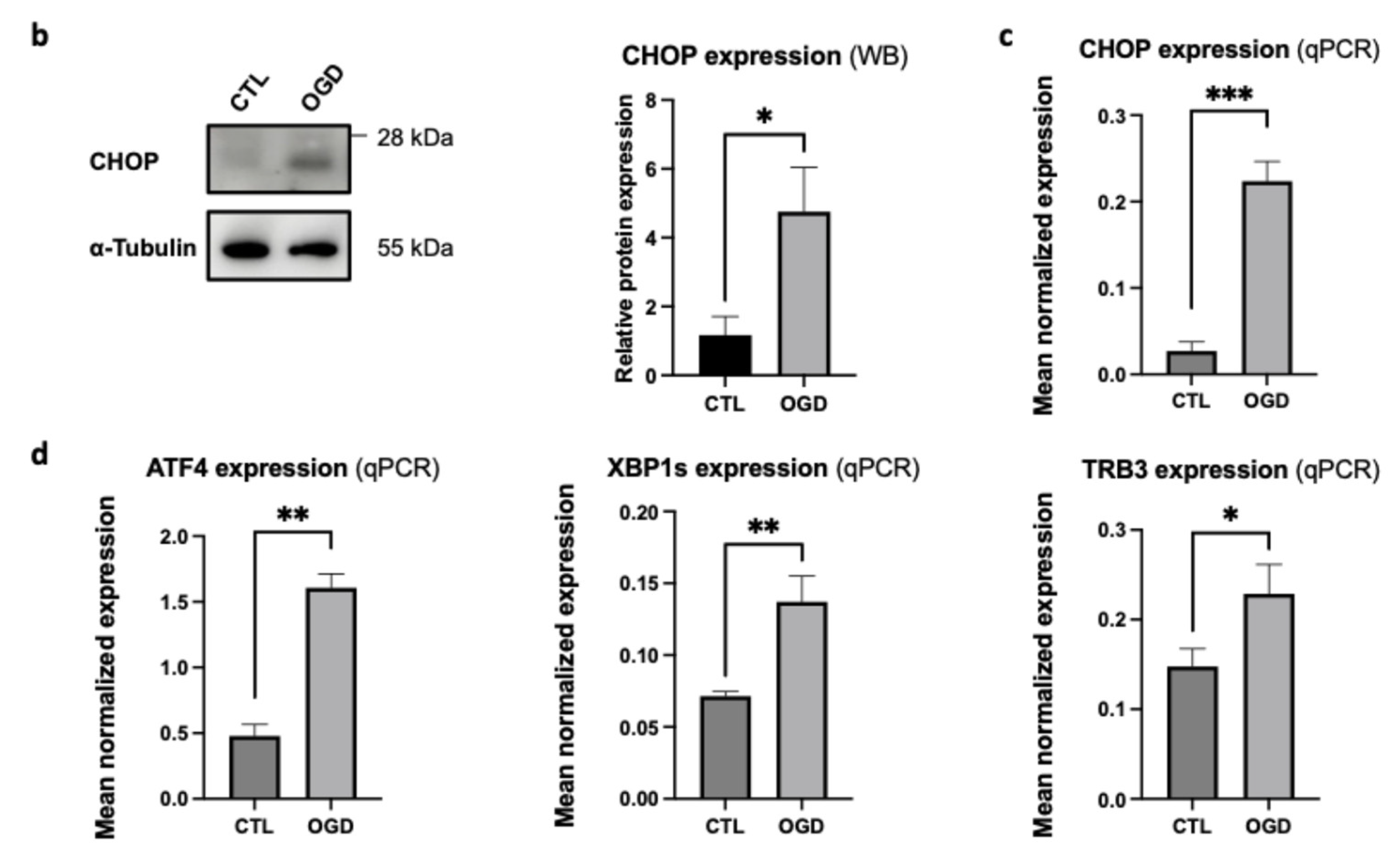
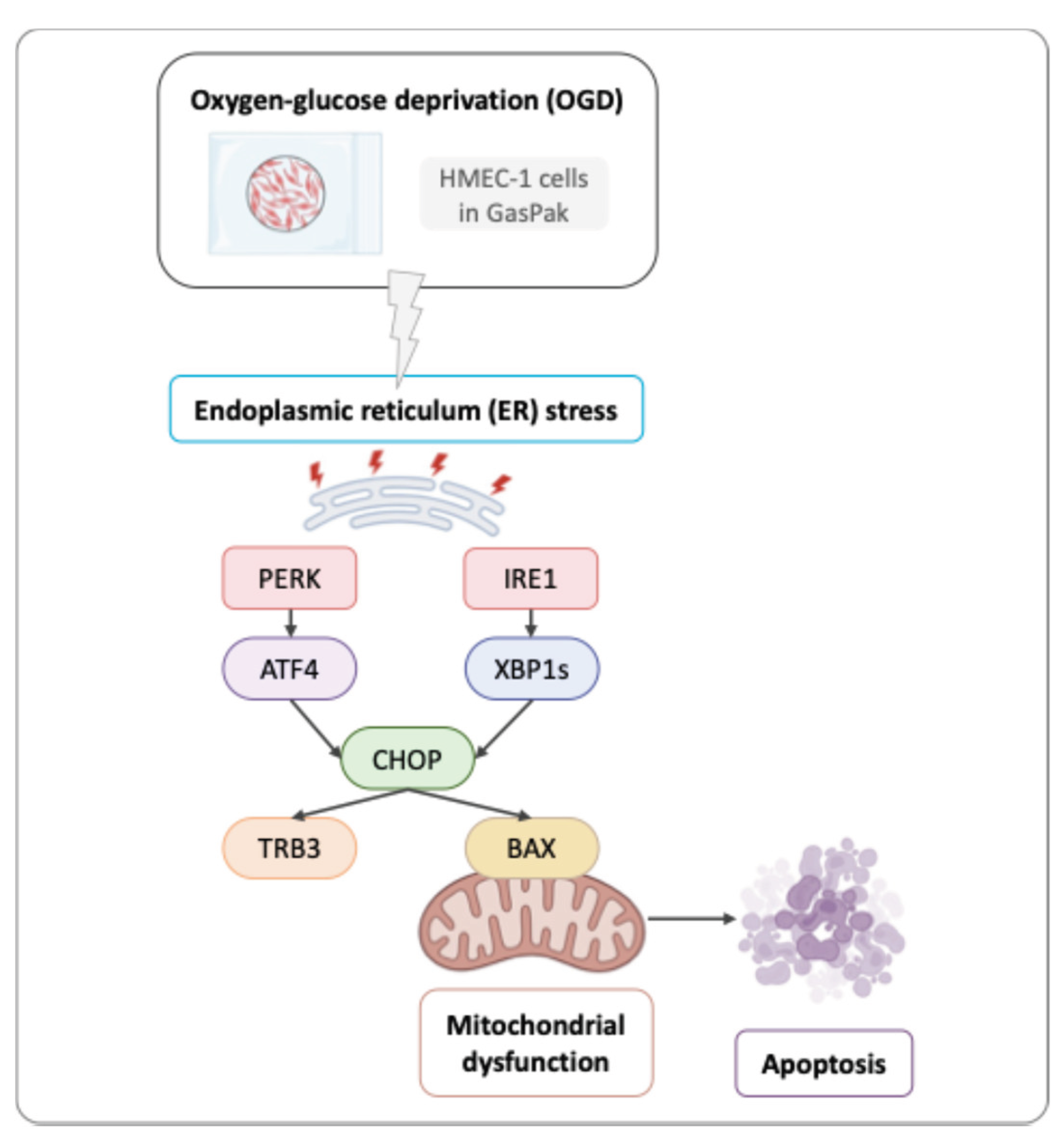
3.4. OGD Induces ER Stress and UPR Signaling in HMEC-1 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATF | activating transcription factor |
| BAX | Bcl-2–associated X |
| BBB | blood-brain barrier |
| CHOP | C/EBP homologous protein |
| ER | endoplasmic reticulum |
| GRP | glucose-regulated protein |
| HMEC-1 | human microvascular endothelial cells-1 |
| IRE1 | inositol-requiring enzyme 1 |
| MTT | 3-(4,5-dimethylthiazol-2-yl) -2: 5-diphenyl tetrazolium bromide |
| OCR | oxygen consumption rate |
| OGD | oxygen–glucose deprivation |
| PERK | protein kinase RNA (PKR)-like ER kinase |
| PI | propidium iodide |
| TRB3 | tribbles homolog 3 |
| UPR | unfolded protein response |
| WB | Western blot |
| XPB1s | spliced X-box binding protein 1 |
References
- Xu, C.; Bailly-Maitre, B.; Reed, J.C. Endoplasmic Reticulum Stress: Cell Life and Death Decisions. J Clin Invest 2005, 115, 2656–2664. [Google Scholar] [CrossRef]
- Szegezdi, E.; MacDonald, D.C.; Ní Chonghaile, T.; Gupta, S.; Samali, A. Bcl-2 Family on Guard at the ER. American Journal of Physiology-Cell Physiology 2009, 296, C941–C953. [Google Scholar] [CrossRef]
- Marciniak, S.J.; Yun, C.Y.; Oyadomari, S.; Novoa, I.; Zhang, Y.; Jungreis, R.; Nagata, K.; Harding, H.P.; Ron, D. CHOP Induces Death by Promoting Protein Synthesis and Oxidation in the Stressed Endoplasmic Reticulum. Genes Dev 2004, 18, 3066–3077. [Google Scholar] [CrossRef]
- Sommer, C.J. Ischemic Stroke: Experimental Models and Reality. Acta Neuropathol 2017, 133, 245–261. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in Cancer and Other Diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Keeley, T.P.; Mann, G.E. Defining Physiological Normoxia for Improved Translation of Cell Physiology to Animal Models and Humans. Physiological Reviews 2019, 99, 161–234. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Hilliard, G.; Ferguson, T.; Millhorn, D.E. Cobalt Inhibits the Interaction between Hypoxia-Inducible Factor-α and von Hippel-Lindau Protein by Direct Binding to Hypoxia-Inducible Factor-A*. Journal of Biological Chemistry 2003, 278, 15911–15916. [Google Scholar] [CrossRef] [PubMed]
- Hirsilä, M.; Koivunen, P.; Xu, L.; Seeley, T.; Kivirikko, K.I.; Myllyharju, J. Effect of Desferrioxamine and Metals on the Hydroxylases in the Oxygen Sensing Pathway. The FASEB Journal 2005, 19, 1308–1310. [Google Scholar] [CrossRef]
- Place, T.L.; Domann, F.E.; Case, A.J. Limitations of Oxygen Delivery to Cells in Culture: An Underappreciated Problem in Basic and Translational Research. Free Radic Biol Med 2017, 113, 311–322. [Google Scholar] [CrossRef]
- Mehta, G.; Mehta, K.; Sud, D.; Song, J.W.; Bersano-Begey, T.; Futai, N.; Heo, Y.S.; Mycek, M.-A.; Linderman, J.J.; Takayama, S. Quantitative Measurement and Control of Oxygen Levels in Microfluidic Poly(Dimethylsiloxane) Bioreactors during Cell Culture. Biomed Microdevices 2007, 9, 123–134. [Google Scholar] [CrossRef]
- Bhatia, S.N.; Ingber, D.E. Microfluidic Organs-on-Chips. Nat Biotechnol 2014, 32, 760–772. [Google Scholar] [CrossRef]
- Hirschhaeuser, F.; Menne, H.; Dittfeld, C.; West, J.; Mueller-Klieser, W.; Kunz-Schughart, L.A. Multicellular Tumor Spheroids: An Underestimated Tool Is Catching up Again. Journal of Biotechnology 2010, 148, 3–15. [Google Scholar] [CrossRef]
- Cui, H.; Han, W.; Yang, L.; Chang, Y. Expression of Hypoxia-Inducible Factor 1 Alpha and Oligodendrocyte Lineage Gene-1 in Cultured Brain Slices after Oxygen-Glucose Deprivation. Neural Regen Res 2013, 8, 328–337. [Google Scholar] [CrossRef]
- Singh, A.; Chen, R. The Duration of Oxygen and Glucose Deprivation (OGD) Determines the Effects of Subsequent Reperfusion on Rat Pheochromocytoma (PC12) Cells and Primary Cortical Neurons. International Journal of Molecular Sciences 2023, 24, 7106. [Google Scholar] [CrossRef]
- Chen, J.-L.; Duan, W.-J.; Luo, S.; Li, S.; Ma, X.-H.; Hou, B.-N.; Cheng, S.-Y.; Fang, S.-H.; Wang, Q.; Huang, S.-Q.; et al. Ferulic Acid Attenuates Brain Microvascular Endothelial Cells Damage Caused by Oxygen-Glucose Deprivation via Punctate-Mitochondria-Dependent Mitophagy. Brain Research 2017, 1666, 17–26. [Google Scholar] [CrossRef]
- Bouhamida, E.; Morciano, G.; Perrone, M.; Kahsay, A.E.; Della Sala, M.; Wieckowski, M.R.; Fiorica, F.; Pinton, P.; Giorgi, C.; Patergnani, S. The Interplay of Hypoxia Signaling on Mitochondrial Dysfunction and Inflammation in Cardiovascular Diseases and Cancer: From Molecular Mechanisms to Therapeutic Approaches. Biology (Basel) 2022, 11, 300. [Google Scholar] [CrossRef] [PubMed]
- Kalogeris, T.; Baines, C.P.; Krenz, M.; Korthuis, R.J. Cell Biology of Ischemia/Reperfusion Injury. Int Rev Cell Mol Biol 2012, 298, 229–317. [Google Scholar] [CrossRef]
- Hawkins, B.T.; Davis, T.P. The Blood-Brain Barrier/Neurovascular Unit in Health and Disease. Pharmacological Reviews 2005, 57, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Yetkin-Arik, B.; Vogels, I.M.C.; Nowak-Sliwinska, P.; Weiss, A.; Houtkooper, R.H.; Van Noorden, C.J.F.; Klaassen, I.; Schlingemann, R.O. The Role of Glycolysis and Mitochondrial Respiration in the Formation and Functioning of Endothelial Tip Cells during Angiogenesis. Sci Rep 2019, 9, 12608. [Google Scholar] [CrossRef] [PubMed]
- TeSlaa, T.; Teitell, M.A. Techniques to Monitor Glycolysis. Methods Enzymol 2014, 542, 91–114. [Google Scholar] [CrossRef]
- Li, J.; Yang, C.; Wang, Y. miR-126 Overexpression Attenuates Oxygen-Glucose Deprivation/Reperfusion Injury by Inhibiting Oxidative Stress and Inflammatory Response via the Activation of SIRT1/Nrf2 Signaling Pathway in Human Umbilical Vein Endothelial Cells. Mol Med Rep 2021, 23, 165. [Google Scholar] [CrossRef] [PubMed]
- Ohoka, N.; Yoshii, S.; Hattori, T.; Onozaki, K.; Hayashi, H. TRB3, a Novel ER Stress-inducible Gene, Is Induced via ATF4–CHOP Pathway and Is Involved in Cell Death. The EMBO Journal 2005, 24, 1243–1255. [Google Scholar] [CrossRef]
- Li, Y.; Guo, Y.; Tang, J.; Jiang, J.; Chen, Z. New Insights into the Roles of CHOP-Induced Apoptosis in ER Stress. ABBS 2014, 46, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Badiola, N.; Penas, C.; Miñano-Molina, A.; Barneda-Zahonero, B.; Fadó, R.; Sánchez-Opazo, G.; Comella, J.X.; Sabriá, J.; Zhu, C.; Blomgren, K.; et al. Induction of ER Stress in Response to Oxygen-Glucose Deprivation of Cortical Cultures Involves the Activation of the PERK and IRE-1 Pathways and of Caspase-12. Cell Death Dis 2011, 2, e149. [Google Scholar] [CrossRef] [PubMed]
- Vavilis, T.; Delivanoglou, N.; Aggelidou, E.; Stamoula, E.; Mellidis, K.; Kaidoglou, A.; Cheva, A.; Pourzitaki, C.; Chatzimeletiou, K.; Lazou, A.; et al. Oxygen–Glucose Deprivation (OGD) Modulates the Unfolded Protein Response (UPR) and Inflicts Autophagy in a PC12 Hypoxia Cell Line Model. Cell Mol Neurobiol 2015, 36, 701–712. [Google Scholar] [CrossRef]
- Ades, E.W.; Candal, F.J.; Swerlick, R.A.; George, V.G.; Summers, Susan.; Bosse, D.C.; Lawley, T.J. HMEC-1: Establishment of an Immortalized Human Microvascular Endothelial Cell Line. Journal of Investigative Dermatology 1992, 99, 683–690. [Google Scholar] [CrossRef]
- Camós, S.; Mallolas, J. Experimental Models for Assaying Microvascular Endothelial Cell Pathophysiology in Stroke. Molecules 2010, 15, 9104–9134. [Google Scholar] [CrossRef]
| Method | Principle | Advantages | Limitations | Typical O2 level | References |
| Hypoxia chambers / controlled incubators | O2, CO2, and humidity precisely regulated using premixed gases | Physiologically relevant; stable and reproductible | Expensive equipment; slow transition between normoxia / hypoxia | 0.1–5% | [5,6] |
| Chemical mimetics (CoCl2, DFO, DMOG) |
Stabilize HIF-1α by inhibiting prolyl hydroxylases or chelationg Fe2+ | Simple; inexpensive; no special equipment needed |
Off-target effects; do not reduce O2 directly |
Normoxia, but “hypoxia-mimicked” |
[7,8] |
| Oxygen scavenging systems | Enzymatic / chemical consumption of dissolved O2 | Rapid induction; small-scale; lowcost |
Hard to maintain stable levels; alters pH / nutrients | Variable, often < 1% |
[9] |
| Microfluidic devices / organ-on-chip | Controlled O2 gradients via microchannels and oxygen-impermeable membranes | High spatially temporal resolution; mimics in vivo gradients | Technically demanding; limited throughtput | Tunable (0–21%) |
[10,11] |
| Physical diffusion barriers | O2 diffusion limited by extracellular matrix or media depth | Models tumors-like hypoxic gradients | Poor control of exact O2 level; variable reproductibility | Typically < 5% in core regions |
[12] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




