3. Results
3.1. Case 1: The Dosing and Schedule Sufficient for Rapid Remission
Miss W, a 24-year-old registered nurse, was seen for her first psychiatric assessment on 10 November 2025 after abruptly resigning from her initial hospital post. Until that point she had never consulted mental-health services. The crisis began soon after she started working without close supervision; every routine comment from a senior triggered punishing self-criticism and a spiral of “rabbit-hole” rumination over whether she was fit for the profession. At home she stopped eating, lay awake dreading the next dawn, and made an emergency-department visit for what proved to be severe anxiety. On formal screening she scored 19 on the Patient Health Questionnaire-9 (PHQ-9) and 16 on the Generalised Anxiety Disorder-7 (GAD-7). The interview also uncovered childhood-onset inattentiveness suggestive of undiagnosed ADHD, but the dominant picture was an obsessional form of obsessive–compulsive disorder marked by relentless catastrophic thinking.
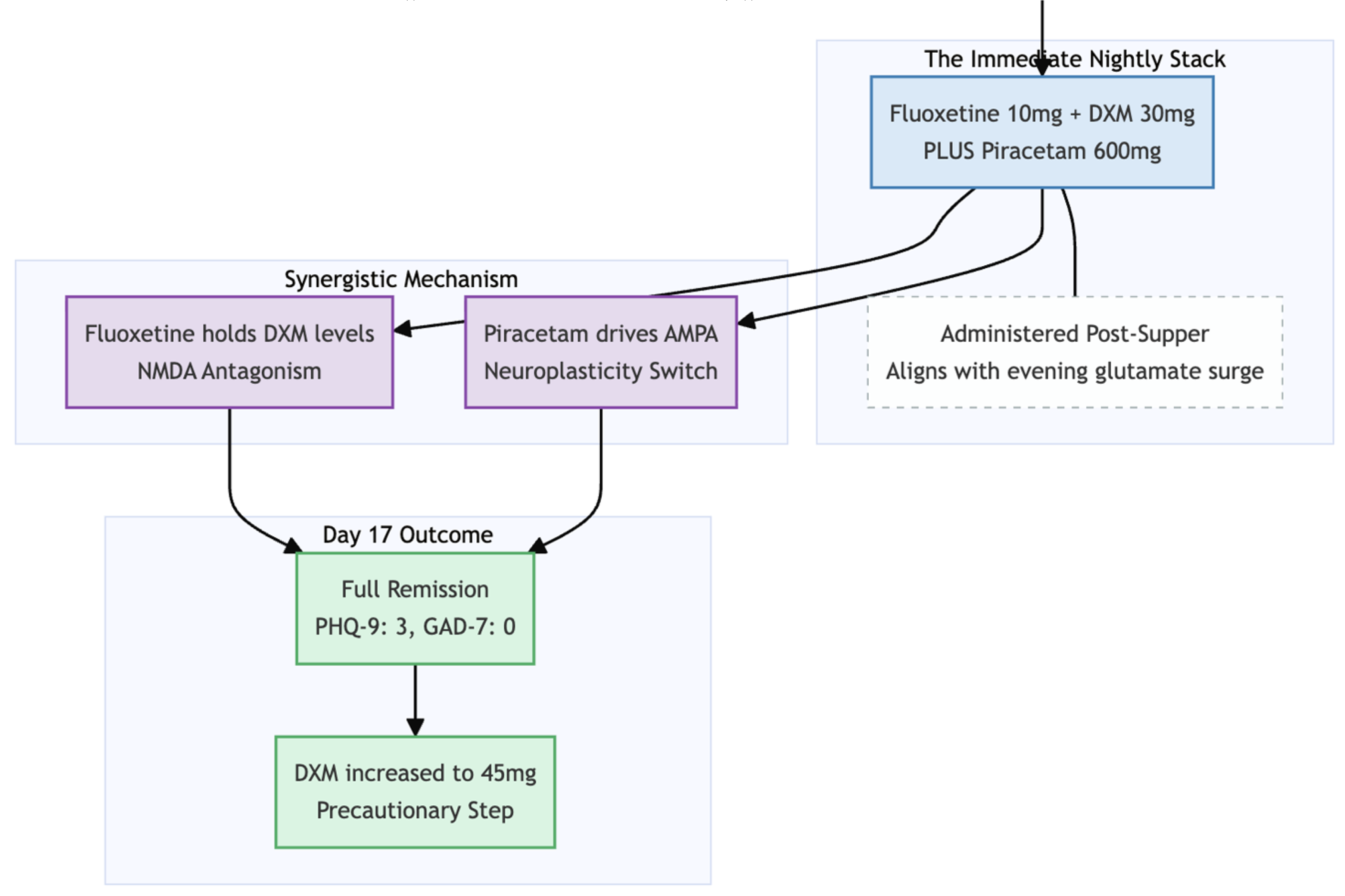
Because of the acuteness of her distress, we started an oral glutamatergic protocol at the very first visit. Each night she took fluoxetine 10 mg to inhibit CYP2D6, dextromethorphan (DXM) 30 mg in two 15-mg tablets, piracetam 600 mg, and risperidone 0.5 mg as a low-dose augmenting agent. All medication was scheduled after supper to reduce daytime activation.
Seventeen days later, on 27 November, she returned looking noticeably brighter and reported that the mental free-fall had simply stopped. She was free of restlessness and catastrophic ideation, eating normally, and had already picked up casual nursing shifts while planning a full return to public-sector work. Follow-up scores confirmed the change (PHQ-9 = 3, GAD-7 = 0). DXM was increased to 45 mg nightly to consolidate the response; the rest of the regimen was left unchanged. She denied side-effects throughout.
At the time of writing Miss W remains in full symptomatic and functional remission, working part-time without anxiety or rumination and preparing to rejoin her original hospital. The combination of fluoxetine, nightly DXM 45 mg, piracetam 600 mg, and risperidone 0.5 mg is well-tolerated and requires no additional support.
3.2. Lessons Learnt from Case 1
Miss W’s experience suggests that we may not need large doses or complicated schedules to unlock the glutamatergic “switch” in newly diagnosed obsessive-compulsive disorder (
Figure 1). Two details stand out: how little drug was required, and how decisively the bedtime schedule orked.
To begin with, a nightly capsule containing only 30 mg of dextromethorphan, held in circulation by 10 mg of fluoxetine, plus 600 mg of piracetam was enough to flatten scores from PHQ-9 19 to 3 and GAD-7 16 to 0 in just over two weeks. The patient’s catastrophic spirals, fear of sleep, and loss of appetite all melted away while she was still on that entry dose; the later step-up to 45 mg of dextromethorphan was precautionary rather than curative. This mirrors the pattern seen in Cheung’s growing clinical series, where treatment-naïve patients—unburdened by years of polypharmacy or ultrarapid CYP2D6 metabolism—often top out at the same 30-mg plateau [
12].
Equally important was timing. Giving fluoxetine, dextromethorphan, and piracetam together after supper lines the drug pulse up with the evening rise in cortical glutamate activity and the synaptic “housekeeping” that follows during early sleep. In Miss W, that single bedtime dose eliminated the sedativeness or dizziness sometimes reported when DXM is taken in the morning, and she began sleeping more deeply as the rumination lifted.
Taken together, the case adds weight to a simple, clinically friendly rule of thumb—start with 30 mg of fluoxetine-boosted dextromethorphan and 600 mg of piracetam at bedtime, and think about higher doses only if symptoms plateau. For young adults meeting their first psychiatrist, that may be all that is needed for a full and durable remission.
3.3. Case 2: When Nocte Dose Not Good Enough
Miss F is a 24-year-old woman who was referred on 18 October 2025 for escalating health-centred anxiety. According to the patient, the problem began several months earlier, soon after she changed jobs. She found herself preoccupied by the idea that she was harbouring a serious but undiagnosed illness. Hours were spent checking her pulse, palpating lymph-nodes, or searching the internet for rare disorders. Palpitations, diffuse muscle tension, and a sense of internal trembling accompanied these thoughts; nights were long, with prolonged sleep-onset latency followed by broken, non-refreshing sleep. Screening questionnaires reflected prominent anxiety (PHQ-9 = 5; GAD-7 = 16). A previous psychiatrist had prescribed duloxetine, but the dose, duration, and benefit were all unclear. On the basis of persistent intrusive health fears, compulsive bodily checking, and accompanying somatic arousal, we formulated the case as obsessive–compulsive disorder with a hypochondriacal theme.
At the first visit we focused on rapid symptomatic relief. Miss F began risperidone 0.5 mg and a tablet of flupentixol/melitracen (Deanxit) each night for ruminative drive and low mood. Alprazolam 0.25 mg and propranolol 10 mg were provided on an as-needed basis for panic surges, and lemborexant 2.5–5 mg was offered for insomnia.
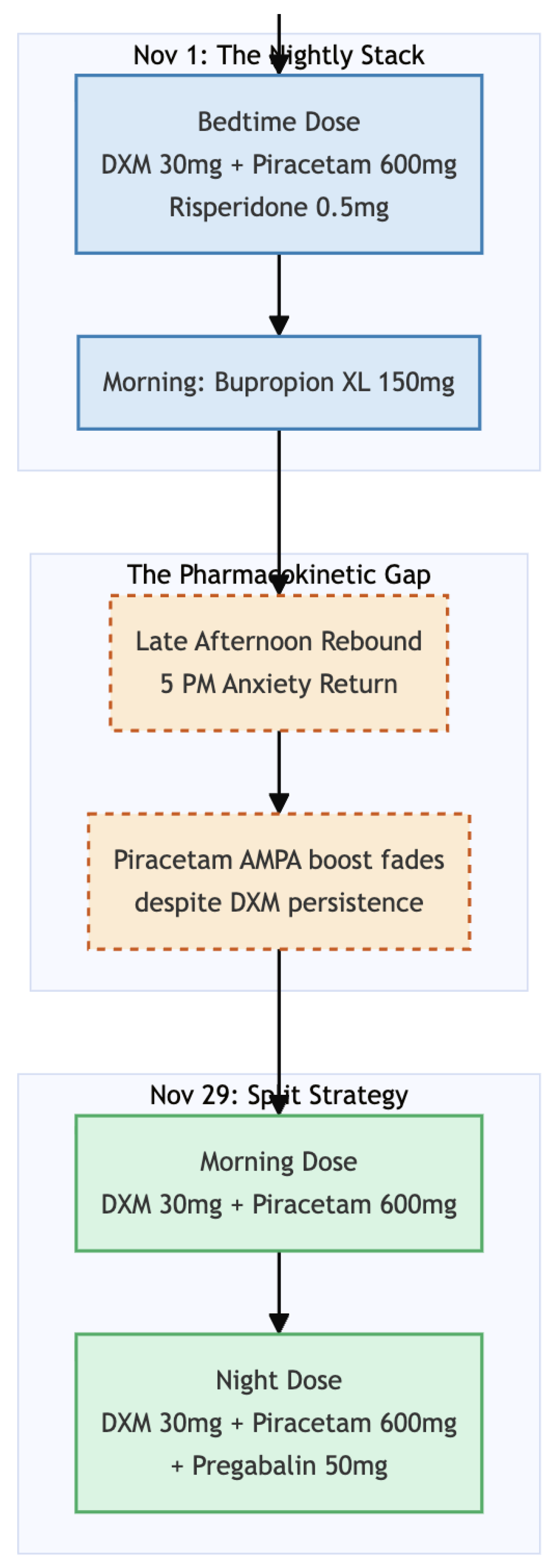
Two weeks later, on 1 November 2025, we introduced an oral glutamatergic protocol patterned after Cheung’s regimen. All glutamatergic agents were taken at bedtime: dextromethorphan 30 mg in two 15-mg tablets, piracetam 600 mg, and the existing risperidone 0.5 mg. Bupropion XL 150 mg was added each morning to improve daytime drive. Within a few nights the patient reported a clear reduction in catastrophic health thoughts; hypnotics were no longer required.
At follow-up on 29 November she described calm, productive mornings but noted that worry still crept in toward late afternoon. To extend coverage we divided the glutamatergic dose: dextromethorphan 30 mg and piracetam 600 mg were given on waking and repeated at night, bringing daily totals to 60 mg and 1 200 mg respectively. Pregabalin 50 mg was started at bedtime for residual somatic tension, while risperidone, bupropion, and the as-needed agents were continued unchanged. She now used alprazolam and propranolol only rarely, reported vivid yet non-disturbing dreams two or three times a week, and was making concrete plans for an overseas holiday in January 2026. Repeat screening showed PHQ-9 = 2 and GAD-7 = 9, consistent with her subjective sense of “much better, just a little worry in the evenings.”
3.4. Lessons Learnt from Case 2
Miss F’s progress throws light on a practical weakness of the “all-at-bedtime” version of the Cheung glutamatergic stack (
Figure 2). Taken once nightly, the combination of dextromethorphan 30 mg and piracetam 600 mg calmed her health worries through the night and well into the following morning, yet by late afternoon the same thoughts were slipping back in. She described the pattern clearly: “I’m fine until about five o’clock, then the what-ifs start again.” Screening numbers told the same story—depression had melted away (PHQ-9 = 2) but anxiety was still in the mild range (GAD-7 = 9).
Pharmacokinetics offer a neat explanation. Even with fluoxetine or bupropion on board, and dextromethorphan’s active levels may persist despite discontinuation of the regimen [
13], the AMPA boost from a single 600-mg dose of piracetam may fade sooner [
14]. Bedtime dosing therefore covers the biologically busy night-time window and the first half of the working day, but by late afternoon plasma concentrations and receptor occupancy are waning—just when fatigue and fewer distractions create a perfect stage for rumination to return.
The solution in Miss F’s case—splitting both glutamatergic agents into morning and evening doses without raising the daily total—proved immediately helpful. Morning stability was preserved, evening “escape” all but disappeared, and the need for rescue alprazolam or propranolol dropped to near zero.
In practice, then, single bedtime dosing remains a sensible starting point: it is simple, aligns with circadian glutamate peaks, and often improves sleep quality. Clinicians should, however, listen for reports of late-day rebound; when that pattern emerges, a twice-daily schedule can plug the gap quickly and without extra medication burden.
3.5. Case 3: Insights upon Dose Scheduling
Miss L is a 20-year-old woman in her second year of nursing school who was referred on 19 May 2025 for a sudden intensification of long-standing anxiety. During the week before the visit she had lived in a near-constant state of alarm, describing palpitations, queasy stomach, fine tremor, sweating, intermittent numbness of her forearms, and little interest in food. Sleep, however, remained intact. Several months earlier two different primary-care clinics had treated her as a straightforward case of depression and generalized anxiety, first with fluoxetine 20 mg, later with escitalopram 10 mg; neither agent brought durable relief. At presentation her Patient Health Questionnaire-9 (PHQ-9) score was 15 and her Generalised Anxiety Disorder-7 (GAD-7) score was 15, both in the moderately severe range. Careful questioning revealed intrusive doubt-laden thoughts and hours of mental checking, leading us to re-frame the problem as predominantly obsessional obsessive–compulsive disorder (OCD).
3.5.1. Early Management (May–June 2025)
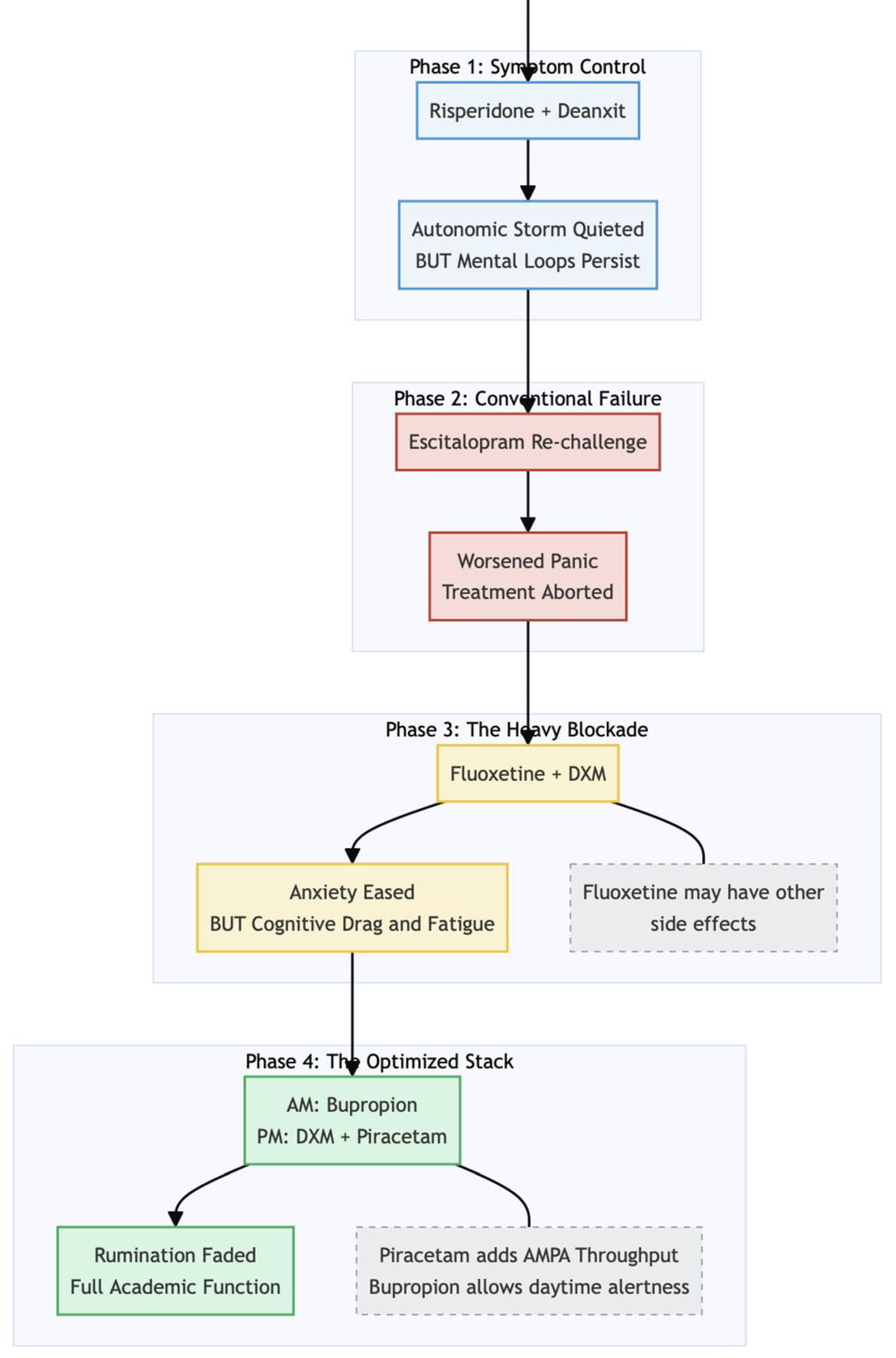
We opted for a symptom-control strategy while planning longer-term therapy. Low-dose risperidone 0.5 mg each night, propranolol 10 mg up to three times daily as needed, and one tablet of flupentixol/melitracen (Deanxit) at bedtime quickly quieted the autonomic storm; by the first week of June her PHQ-9 had fallen to 4 and her GAD-7 to 6.
3.5.2. Trials of Conventional Agents (Late June–July 2025)
Because obsessive rumination persisted, we re-introduced escitalopram at 5 mg and added pregabalin 50 mg nightly. Within ten days the selective-serotonin-reuptake inhibitor worsened panic, so it was withdrawn; pregabalin alone offered little more than sedation.
3.5.3. Shift to a Glutamatergic Approach (August–September 2025)
On 25 August we began an oral ketamine-class regimen first proposed by Cheung: fluoxetine 10 mg nightly to inhibit CYP2D6, plus dextromethorphan (DXM) 30 mg (two 15-mg tablets) at bedtime. One month later anxiety had eased but the patient now grappled with leaden fatigue, poor concentration, a globus sensation, and scant appetite (PHQ-9 = 13, GAD-7 = 11). To address the cognitive drag we discontinued fluoxetine and, on 6 September, completed the Cheung stack by adding piracetam 600 mg nightly. Bupropion XL 150 mg each morning replaced the fluoxetine to provide daylight activation, while risperidone 0.5 mg was retained for its anti-obsessional effect.
3.5.4. Clinical Course (September–October 2025)
Three weeks later Miss L reported that the relentless mental churning had dwindled to brief, manageable episodes. She denied side-effects, although her mother observed occasional daytime sluggishness that the patient herself did not notice. Screening scores on 26 September were PHQ-9 = 11 and GAD-7 = 9. At her most recent review on 24 October she felt “okay so far—much less ruminating,” was attending all classes, and was preparing for her November clinical practicum. Questionnaires had normalised (PHQ-9 = 8, GAD-7 = 6).
3.5.5. Current Medication Profile and Status (24 October 2025)
• Dextromethorphan 30 mg nightly
• Piracetam 600 mg nightly
• Risperidone 0.5 mg nightly
• Flupentixol/melitracen one tablet nightly
• Bupropion XL 150 mg each morning
• Alprazolam 0.25 mg as needed (rarely required)
Miss L is now functioning at her pre-morbid level, attending university without limitation and ready to enter clinical rotations. Obsessive rumination is minimal, somatic anxiety is quiescent, and no significant adverse effects have emerged from the nightly Cheung glutamatergic combination—DXM plus piracetam—used alongside low-dose adjuncts.
3.6. Lessons Learnt from Case 3
Miss L’s course offers a window into how an oral, ketamine-class strategy can be fine-tuned in day-to-day practice (
Figure 3). Two details seem to have driven the turnaround.
First, the night she added piracetam 600 mg marked the point where improvement moved from slow and uneven to steady and obvious. Up to that moment she was taking dextromethorphan (DXM) alone and still felt drained, jittery, and stuck in loops of worry. Within three weeks of adding piracetam her rumination had faded to background noise, her energy was back, and both PHQ-9 and GAD-7 scores nearly halved. The simple timeline argues that piracetam supplied the missing piece—extra AMPA throughput—needed to turn DXM’s NMDA blockade into real neuroplastic change. Similar “second-wind” responses after piracetam add-on are described throughout the original protocol paper [
11].
Second, the decision to keep the activating drug bupropion in the morning and reserve DXM plus piracetam for bedtime appears to have paid off. Bupropion does inhibit CYP2D6, but not as forcefully as fluoxetine, so giving it 12 hours apart from DXM reduces the risk of unpredictable peaks while still providing daytime drive. Cheung [
15] explicitly recommends this split schedule for students or shift-workers who need clear mornings without the buzz that can follow a one-time, all-in dose. This is because bupropion could inhibit CYP2D6 by turning down its gene transcription [
16]. Miss L never complained of insomnia or overshoot, yet reported sharper concentration during clinical skills labs—a small but practical endorsement of the timing strategy.