Submitted:
04 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
Basal stem rot (BSR), caused by Ganoderma boninense, is a major threat to oil palm plantations, leading to severe yield losses and significant economic impact. Early detection of BSR remains challenging because of the delayed onset of symptoms, while monoculture practices further exacerbate disease prevalence. G. boninense, a white rot fungus, invades root and vascular tissues of oil palm, impairing water and nutrient transport, ultimately causing plant decay and death. This study aimed to isolate, characterize, and identify bacterial isolates with both anti-Ganoderma and plant growth-promoting (PGP) abilities from a commercial biofertilizer, CRPO, specifically formulated for oil palm cultivations. Two isolates, K3 (Margalitia shackletonii) and K8 (Bacillus subtilis) displayed strong antagonistic activity against G. boninense, with percentage inhibition of radial growth (PIRG) values exceeding 89%. For PGP traits, K3 demonstrated broad potential, including phosphate and potassium solubilization, nitrogen fixation, 1-aminocyclopropane-1-carboxylate (ACC) deaminase activity, siderophore and high indole-3-acetic acid production. In contrast, K8 displayed positive results only for nitrogen fixation and ACC deaminase production. The dual functions of these isolates in suppressing G. boninense while enhancing plant growth confirms their presence as biocontrol agents. These advance sustainable BSR management strategies by reducing reliance on chemical inputs and strengthening oil palm resilience.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Isolation of Bacteria
2.2. Bacterial Antagonistic Activities Against G. boninense In-Vitro:
2.2.1. Dual Culture Assay
2.2.2. Volatile Organic Compound (VOC) Assay
2.3. Plant Growth-Promoting Assay
2.3.1. Phosphate Solubilization Assay
2.3.2. Potassium Solubilization Assay
2.3.3. Nitrogen Fixation Assay
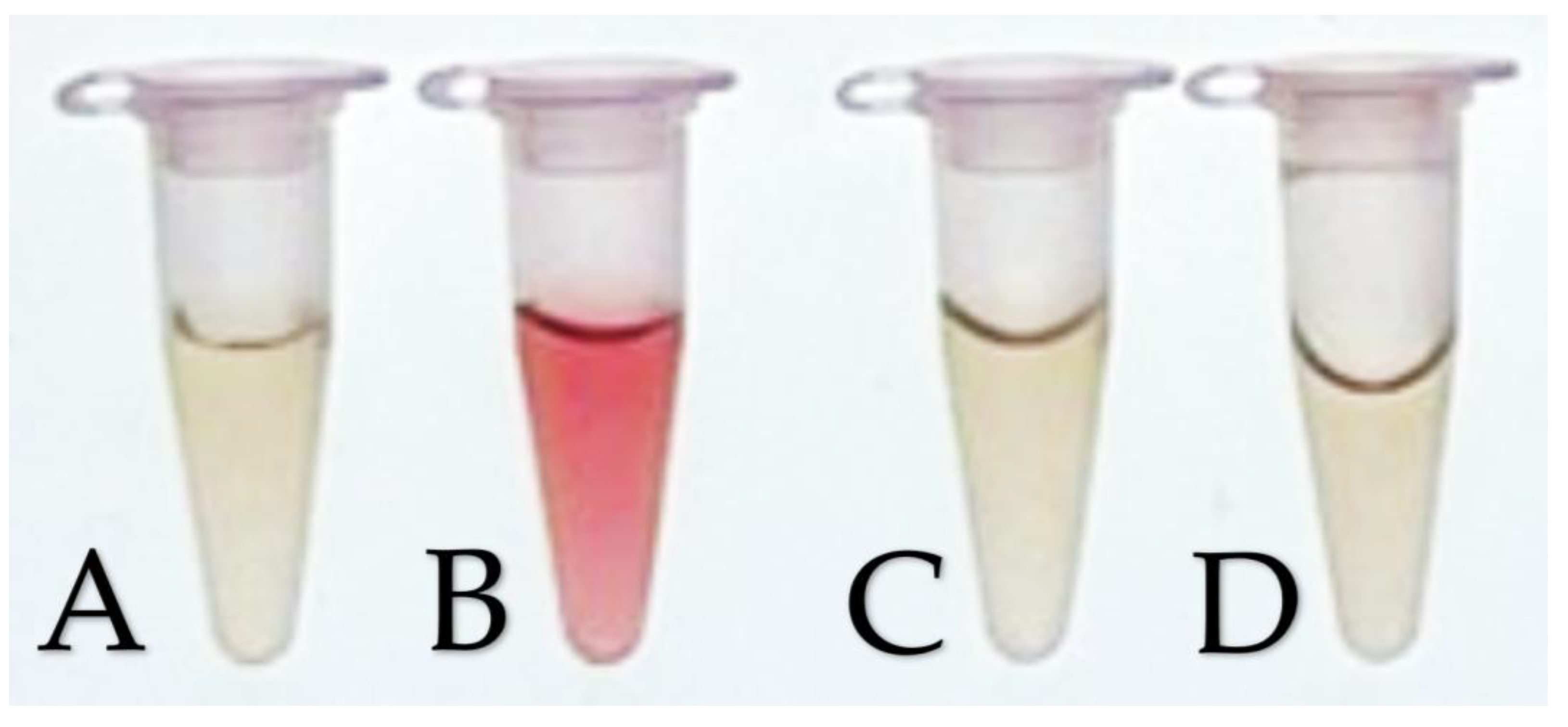
2.3.4. Siderophore Detection Assay
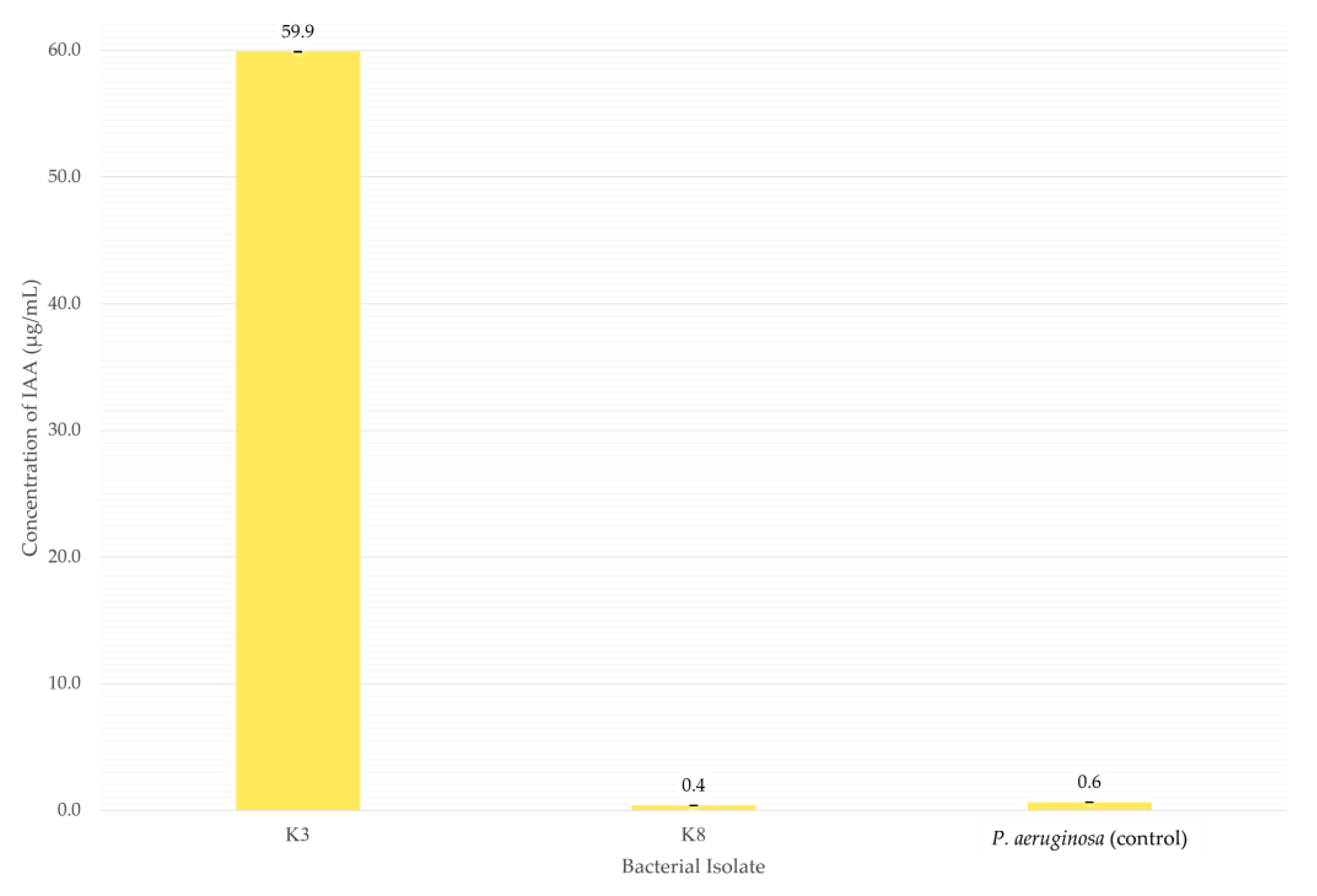
2.3.5. Quantitative Analysis of IAA Production
2.3.6. Quantitative Analysis of ACC Deaminase Production
2.3.7. Identification of Bacterial Isolates by 16S rDNA Sequencing
3. Results
3.1. Bacterial Isolation and Morphological Study
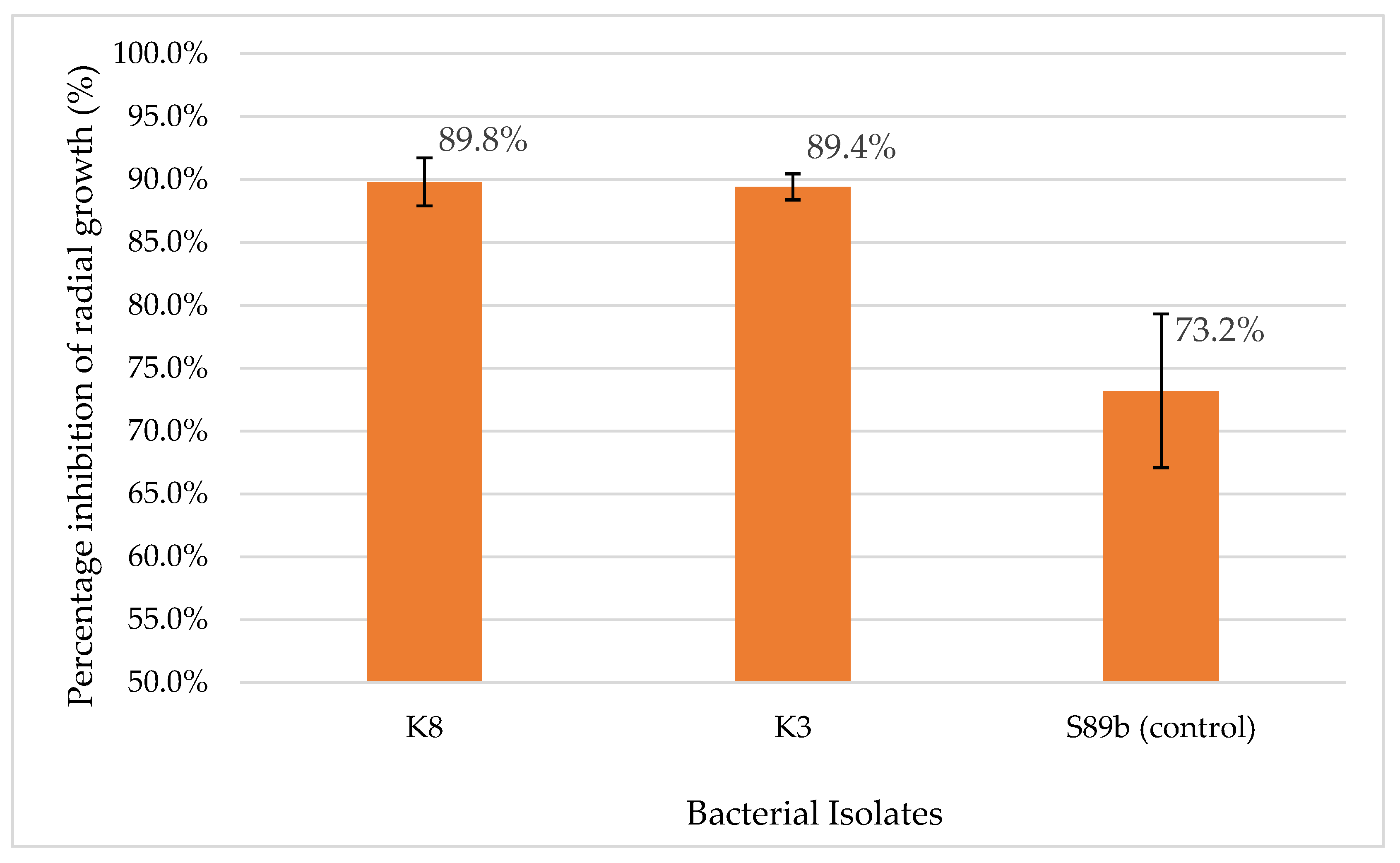
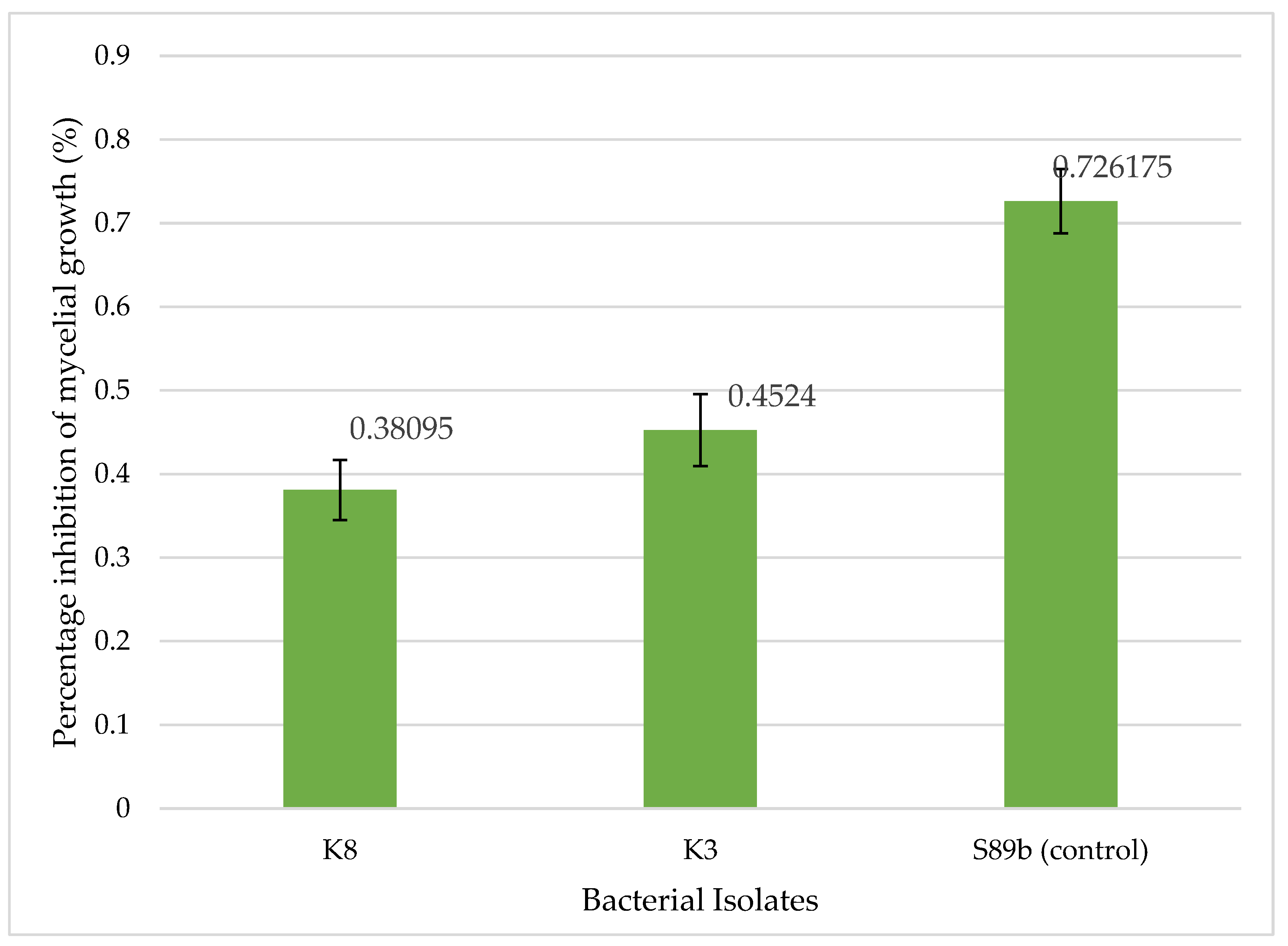
3.2. Bacterial Antagonistic Activities Against G. boninense
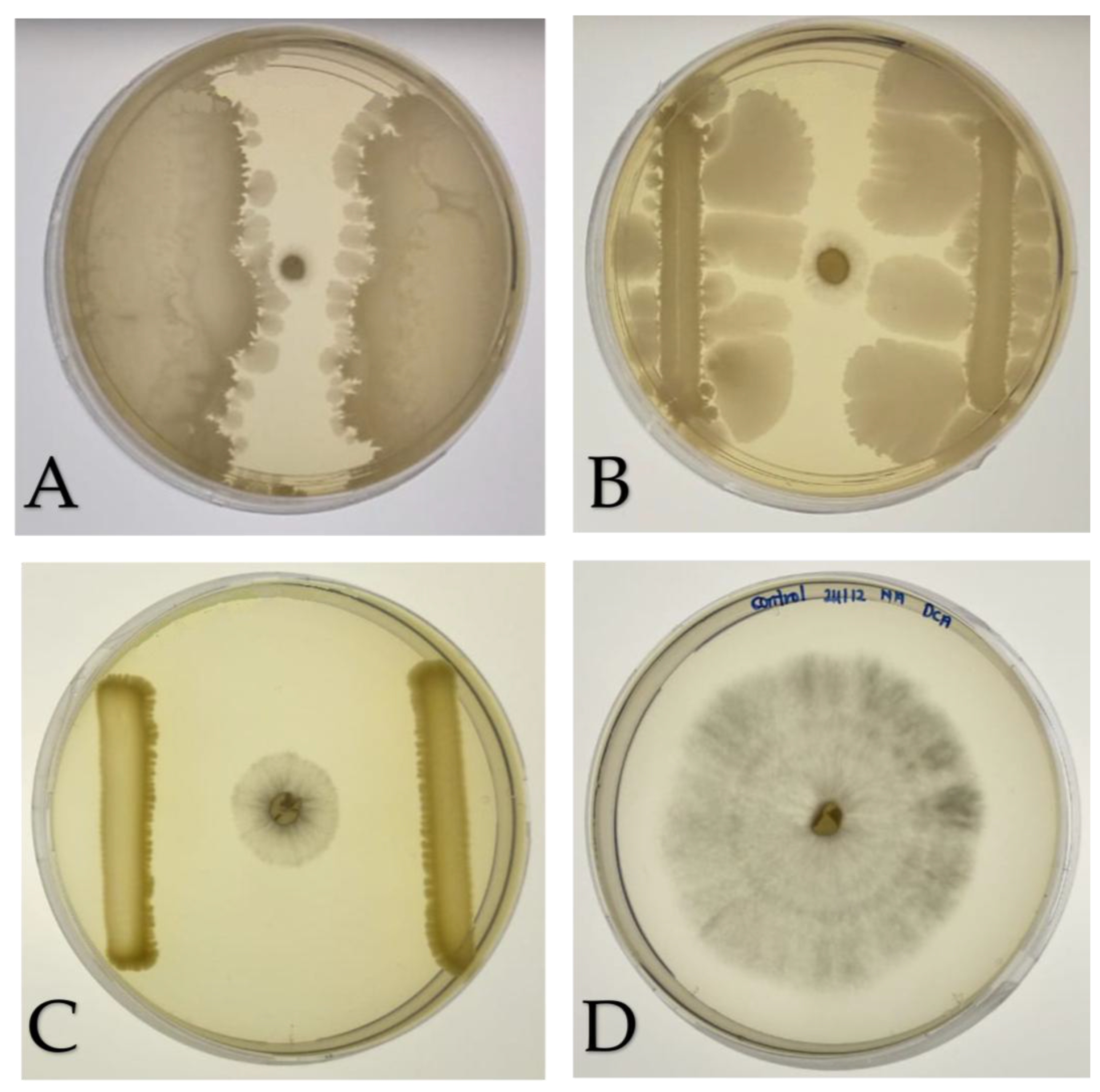
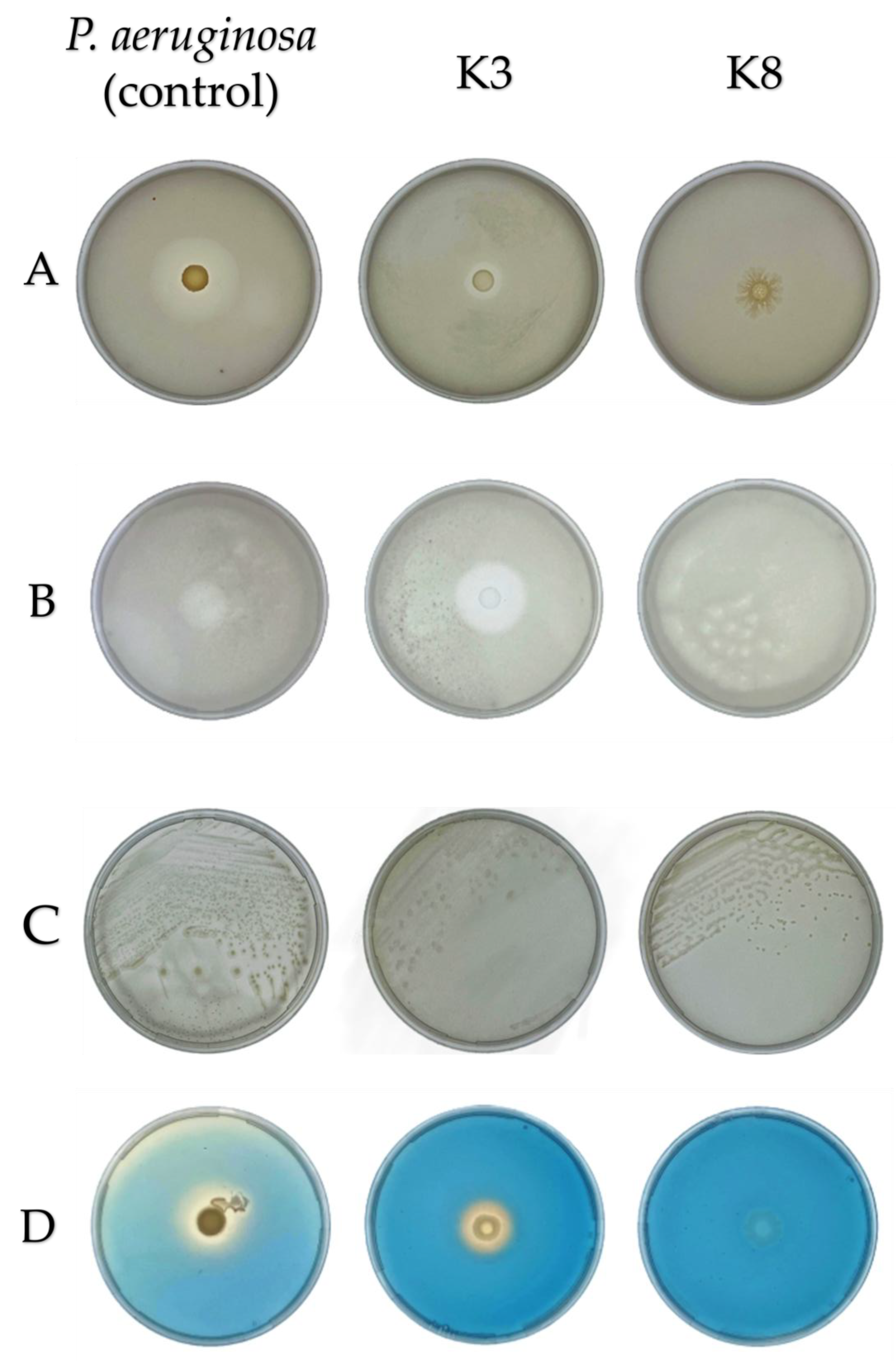
3.2.1. Dual Culture Assay
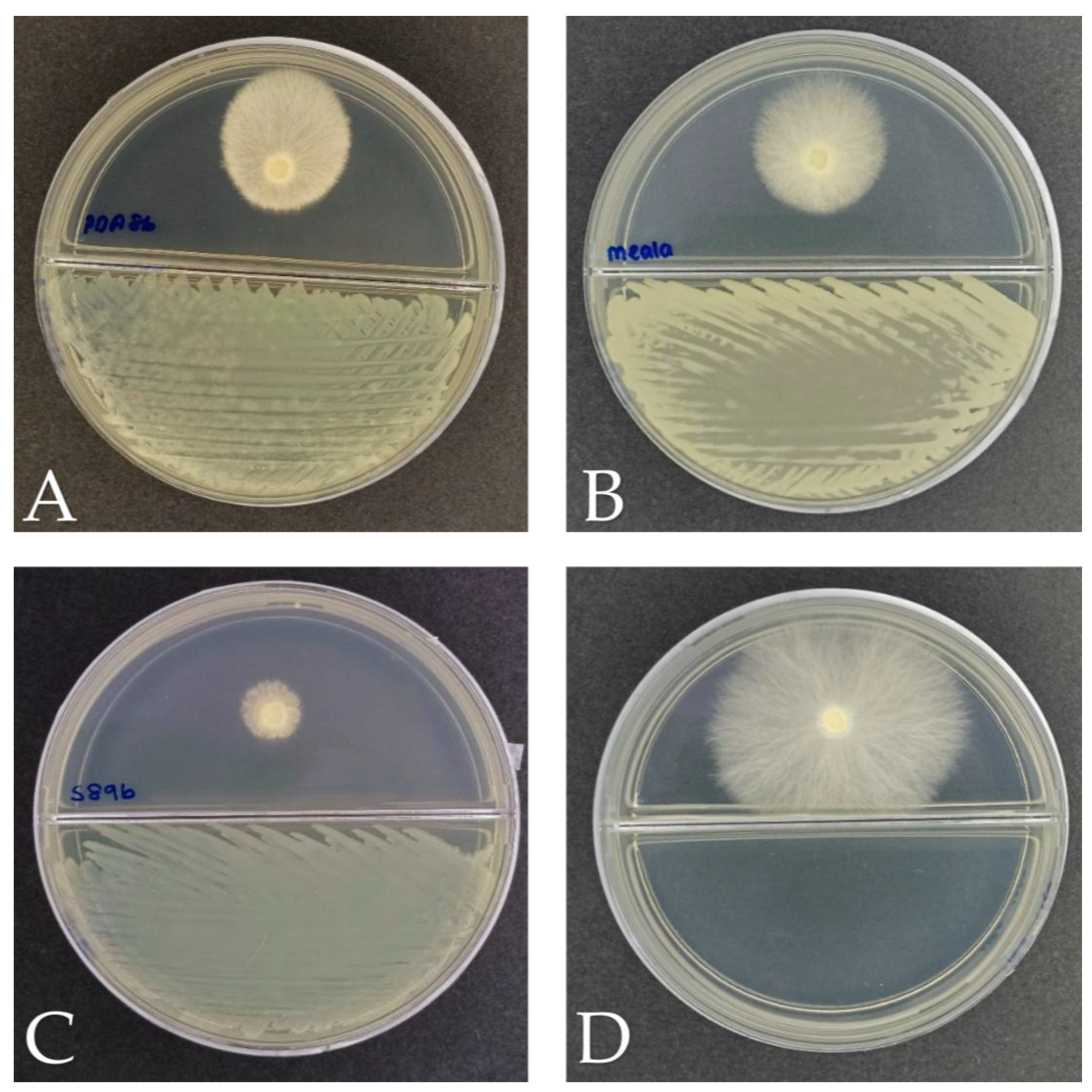
3.2.2. Volatile Organic Compound (VOC) Assay
3.3. PGP Traits of Bacterial Isolates
3.4. 16S rRNA Gene Sequencing and Taxonomic Identification
4. Discussion
4.1. Isolation of Bacteria from CRPO Biofertilizer
4.3. Bacterial Antagonistic Activities Against G. boninense
4.3.1. Dual Culture Assay
4.3.2. Volatile Organic Compound (VOC) Assay
4.4. Functional Characterization of K3 (M. shackletonii) and K8 (B. subtilis) PGP Traits
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sariah, M.; Hussin, M.Z.; Miller, R.N.G.; Holderness, M. Pathogenicity of Ganoderma boninense Tested by Inoculation of Oil Palm Seedlings. Plant Pathol. 1994, 43, 507–510. [Google Scholar] [CrossRef]
- Jazuli, N.A.; Kamu, A.; Chong, K.P.; Gabda, D.; Hassan, A.; Abu Seman, I.; Ho, C.M. A Review of Factors Affecting Ganoderma Basal Stem Rot Disease Progress in Oil Palm. Plants (Basel) 2022, 11, 2462. [Google Scholar] [CrossRef] [PubMed]
- Rees, R.W.; Flood, J.; Hasan, Y.; Potter, U.; Cooper, R.M. Basal Stem Rot of Oil Palm (Elaeis guineensis); Mode of Root Infection and Lower Stem Invasion by Ganoderma boninense. Plant Pathol. 2009, 58, 982–989. [Google Scholar] [CrossRef]
- Rees, R.W.; Flood, J.; Hasan, Y.; Cooper, R.M. Effects of Inoculum Potential, Shading and Soil Temperature on Root Infection of Oil Palm Seedlings by the Basal Stem Rot Pathogen Ganoderma boninense. Plant Pathol. 2007, 56, 862–870. [Google Scholar] [CrossRef]
- Alexander, A.; Sipaut, C.S.; Dayou, J.; Chong, K.P. Oil palm root colonisation by Ganoderma boninense: An Insight Study Using Scanning Electron Microscopy. J. Oil Palm Res. 2017, 29, 262–266. [Google Scholar] [CrossRef]
- Markom, M.A.; Shakaff, A.Y.M.; Adom, A.H.; Ahmad, M.N.; Hidayat, Wahyu; Abdullah, A.H.; Ahmad; Fikri, N. Intelligent Electronic Nose System for Basal Stem Rot Disease Detection. Comput. Electron. Agric. 2009, 66, 140–146. [Google Scholar] [CrossRef]
- Siddiqui, Y.; Surendran, A.; Paterson, R.R.M.; Ali, A.; Ahmad, K. Current Strategies and Perspectives in Detection and Control of Basal Stem Rot of Oil Palm. Saudi J. Biol. Sci. 2021, 28, 2840–2849. [Google Scholar] [CrossRef]
- Ying, W.K.; Khim, P.C. Ganoderma boninense: General Characteristics of Pathogenicity and Methods of Control. Front. Plant Sci. 2023, 14, 1156869. [Google Scholar] [CrossRef]
- Musa, H. Antagonistic Potential of Trichoderma Species against Ganoderma Disease of Oil Palm. Niger. J. Agric. Food Environ. 2017, 13, 60–67. [Google Scholar]
- Susanto, A.; Sudharto, P.S.; Purba, R.Y. Enhancing Biological Control of Basal Stem Rot Disease (Ganoderma boninense) in Oil Palm Plantations. Mycopathologia 2005, 159, 153–157. [Google Scholar] [CrossRef]
- Nur, R.R.; Maizatul, S.M.; Idris, A.S.; Madihah, A.Z.; Nasyaruddin, M. The Potential of Endophytic Bacteria as a Biological Control Agent for Ganoderma disease in Oil Palm. Sains Malaysiana 2016, 45, 401–409. [Google Scholar]
- Sneha, S.; Anitha, B.; Anjum Sahair, R.; Raghu, N; Gopenath, T.S.; Chandrashekrappa, G.K.; Basalingappa, K.M. Biofertilizer for Crop Production and Soil Fertility. Acad. J. Agric. Res. 2018, 6, 299–306. [Google Scholar] [CrossRef]
- Kumar, R.; Sahu, Y.K.; Kumawat, N. Role of Biofertilizers in Agriculture. Pop. Kheti 2017, 5(4), 63–66. [Google Scholar]
- Schütz, L.; Gattinger, A.; Meier, M.; Muller, A.; Boller, T.; Mader, P.; Mathimaran, N. Improving Crop Yield and Nutrient Use Efficiency via Biofertilization—A Global Meta-analysis. Front. Plant Sci. 2018, 8, 2204. [Google Scholar] [CrossRef] [PubMed]
- Pirttilä, A.M.; Tabas, H.M.P.; Baruah, N.; Koskimäki, J.J. Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms 2021, 9, 817. [Google Scholar] [CrossRef]
- Daniel, A.I.; Fadaka, A.O.; Gokul, A.; Bakare, O.O.; Aina, O.; Fisher, S.; Burt, A.F.; Mavumengwana, V.; Keyster, M.; Klein, A. Biofertilizer: The Future of Food Security and Food Safety. Microorganisms 2022, 10, 1220. [Google Scholar] [CrossRef]
- Kumar, S.; Diksha; Sindhu, S.S.; Kumar, R. Biofertilizers: An Ecofriendly Technology for Nutrient Recycling and Environmental Sustainability. Curr. Res. Microb. Sci. 2022, 3, 100094. [Google Scholar] [CrossRef]
- El-Saadony, M.T.; Saad, A.M.; Soliman, S.M.; Salem, H.M.; Ahmed, A.I.; Mahmood, M.; El-Tahan, A.M.; Ebrahim, A.A.M.; Abd El-Mageed, T.A.; Negm, S.H.; Selim, S.; Babalghith, A.O.; Elrys, A.S.; El-Tarabily, K.A.; Abu.Qamar, S.F. Plant Growth-Promoting Microorganisms as Biocontrol Agents of Plant Diseases: Mechanisms, Challenges, and Future Perspectives. Front. Plant Sci. 2022, 13, 923880. [Google Scholar] [CrossRef]
- Ngalimat, M.S.; Mohd Hata, E.; Zulperi, D.; Ismail, S.I.; Ismail, M.R.; Mohd Zainudin, N.A.I.; Saidi, N.B.; Yusof, M.T. Plant Growth-Promoting Bacteria as an Emerging Tool to Manage Bacterial Rice Pathogens. Microorganisms 2021, 9, 682. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Jeon, M.S.; Kim, J.Y.; Lee, S.H. Effects of an Auxin-Producing Symbiotic Bacterium on Cell Growth of the Microalga Haematococcus pluvialis: Elevation of Cell Density and Prolongation of Exponential Stage. Algal Res. 2019, 41, 101547. [Google Scholar] [CrossRef]
- Zhao, X.; Zhou, J.; Tian, R.; Liu, Y. Microbial Volatile Organic Compounds: Antifungal Mechanisms, Applications, and Challenges. Front. Microbiol. 2022, 13, 922450. [Google Scholar] [CrossRef]
- Almeida, O.A.C.; de Araujo, N.O.; Mulato, A.T.N.; Persinoti, G.F.; Sforc, M.L.; Calderan-Rodrigues, M.J.; Oliveira, J.Vd.C. Bacterial Volatile Organic Compounds (VOCs) Promote Growth and Induce Metabolic Changes in Rice. Front. Plant Sci. 2023, 13, 1056082. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Miranda, S.; Cabirol, N.; George-Téllez, R.; Zamudio-Rivera, L.S.; Fernández, F. O-CAS, a Fast and Universal Method for Siderophore Detection. J. Microbiol. Methods 2007, 70, 127–131. [Google Scholar] [CrossRef]
- Zhu, L.; Huang, J.; Lu, X.; Zhou, C. Development of Plant Systemic Resistance by Beneficial Rhizobacteria: Recognition, Initiation, Elicitation, and Regulation. Front. Plant Sci. 2022, 13, 952397. [Google Scholar] [CrossRef]
- Toral, L.; Rodríguez, M.; Martínez-Checa, F.; Montaño, A.; Cortés-Delgado, A.; Smolinska, A.; Llamas, I.; Sampedro. Identification of Volatile Organic Compounds in Extremophilic Bacteria and Their Effective Use in Biocontrol of Postharvest Fungal Phytopathogens. Front. Microbiol. 2021, 12, 773092. [Google Scholar] [CrossRef]
- Li, Q.; Wu, L.; Hao, J.; Luo, L.; Cao, Y.; Li, J. Biofumigation on Post-Harvest Diseases of Fruits Using a New Volatile-Producing Fungus of Ceratocystis fimbriata. PLoS ONE 2015, 10, e0132009. [Google Scholar] [CrossRef]
- Bhattacharyya, C.; Banerjee, S.; Acharya, U.; Mitra, A.; Mallick, I.; Haldar, A.; Haldar, S.; Ghosh, A.; Ghosh, A. Evaluation of Plant Growth Promotion Properties and Induction of Antioxidative Defence Mechanism by Tea Rhizobacteria of Darjeeling, India. Sci. Rep. 2020, 10, 72439. [Google Scholar] [CrossRef]
- Sudewi, S.; Ala, A.; Patandjengi, B.; Farid, M. Isolation of Phosphate Solubilizing Bacteria from The Rhizosphere of Local Aromatic Rice in Bada Valley Central Sulawesi, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2020, 575, 012017. [Google Scholar] [CrossRef]
- Adwani, M.S.; Pillai, T.S.; Isokar, S.; Rokade, G. Evaluation and Characterization of Potassium Solubilizing Bacteria. Int. J. Adv. Biochem. Res. 2024, 8(5), 245–248. https://www.biochemjournal.com/special-issue/2024.v8.i5S.d.1174. [CrossRef]
- Shomi, F.Y.; Uddin, M.B.; Zerin, T. Isolation and Characterization of Nitrogen-Fixing Bacteria from Soil Sample in Dhaka, Bangladesh. Stamford J. Microbiol. 2021, 11, 11–13. [Google Scholar] [CrossRef]
- Gordon, S.A.; Weber, R.P. Colorimetric Estimation of Indoleacetic Acid. Plant Physiol. 1951, 26, 192–195. [Google Scholar] [CrossRef]
- Li, Z.; Chang, S.; Lin, L.; Li, Y.; An, Q. A Colorimetric Assay of 1-Aminocyclopropane-1-Carboxylate (ACC) Based on Ninhydrin Reaction for Rapid Screening of Bacteria Containing ACC Deaminase. Lett. Appl. Microbiol. 2011, 53, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.M.; Geoffroy, V.A.; Baida, N.; Gardan, L.; Izard, D.; Lemanceau, P.; Achouak, W.; Palleroni, N.J. Siderophore Typing, a Powerful Tool for the Identification of Fluorescent and Nonfluorescent Pseudomonads. Applied and Environmental Microbiology 2002, 68, 2745–2753. [Google Scholar] [CrossRef] [PubMed]
- Micromaster Laboratories Private Limited Pikovskaya’s Broth (Medium) (DM1841). 2018.
- Sasirekha, B.; Shivakumar, S.; Sullia, S.B. Statistical Optimization for Improved Indole-3-Acetic Acid (IAA) Production by Pseudomonas aeruginosa and Demonstration of Enhanced Plant Growth Promotion. Journal of Soil Science and Plant Nutrition 2012, 12, 863–873. [Google Scholar] [CrossRef]
- Roychowdury, R.; Tasneem, F.Q.; Pritam, M.; Madhumita, R. Isolation and Characterization of a Pseudomonas aeruginosa Strain PGP for Plant Growth Promotion. Proceedings of the National Academy of Sciences, India – Section B: Biological Sciences 2017, 89, 535–538. [Google Scholar] [CrossRef]
- Saw, G.S. Personal communication, INO Nature Sdn. Bhd.: Raub, Pahang, Malaysia, 2020.
- Pramanik, P.; Goswami, A.J.; Ghosh, S.; Kalita, C. An Indigenous Strain of Potassium-solubilizing Bacteria Bacillus pseudomycoides Enhanced Potassium Uptake in Tea Plants by Increasing Potassium Availability in the Mica Waste-Treated Soil of North-east India. J. Appl. Microbiol. 2018, 126, 215–222. [Google Scholar] [CrossRef]
- Luo, L.; Zhao, C.; Wang, E.; Raza, A.; Yin, C. Bacillus amyloliquefaciens as an Excellent Agent for Biofertilizer and Biocontrol in Agriculture: An Overview for Its Mechanisms. Microbiol. Res. 2022, 259, 127016. [Google Scholar] [CrossRef]
- Logan, N.A.; Lebbe, L.; Verhelst, A.; Goris, J.; Forsyth, G.; Rodríguez-Díaz, M.; Heyndrickx, M.; De Vos, P. Bacillus Shackletonii Sp. Nov., from Volcanic Soil on Candlemas Island, South Sandwich Archipelago. International Journal of Systematic and Evolutionary Microbiology 2004, 54, 373–376. [Google Scholar] [CrossRef]
- Gupta, R.S.; Patel, S.; Saini, N.; Chen, S. Robust Demarcation of 17 Distinct Bacillus Species Clades, Proposed as Novel Bacillaceae Genera, by Phylogenomics and Comparative Genomic Analyses: Description of Robertmurraya kyonggiensis Sp. Nov. And Proposal for an Emended Genus Bacillus Limiting It Only to the Members of the Subtilis and Cereus Clades of Species. International Journal of Systematic and Evolutionary Microbiology 2020, 70, 5753–5798. [Google Scholar] [CrossRef]
- Tor, X.Y.; Toh, W.K.; Loh, P.C.; Wong, H.L. Isolation of Bacteria with Plant Growth-Promoting Activities from a Foliar Biofertilizer. Malays. J. Microbiol. 2022, 18, 211365. [Google Scholar] [CrossRef]
- Ghazy, N.; El-Nahrawy, S. Siderophore Production by Bacillus subtilis MF497446 and Pseudomonas koreensis MG209738 and Their Efficacy in Controlling Cephalosporium maydis in Maize Plant. Arch. Microbiol. 2020, 203, 1195–1209. [Google Scholar] [CrossRef]
- Anggita, S.A.; Munif, A.; Nawangsih, A.A.; Tryono, R. The Endophytic Bacteria of Oil Palm and Areca Nut are Beneficial as Antagonist of Ganoderma boninense and Potential as Plant Growth Promoter. IOP Conf. Ser. Earth Environ. Sci. 2020, 457, 012055. [Google Scholar] [CrossRef]
- Teixeira, G.M.; Mosela, M.; Nicoletto, M.L.A.; Ribeiro, R.A.; Hungria, M.; Youssef, K.; Higashi, A.K.; Mian, S.; Ferreira, A.S.; Gonçalves, L.S.A.; Pereira, U.P.; de Oliveira, A.G. Genomic Insights into the Antifungal Activity and Plant Growth-promoting Ability in Bacillus velezensis CMRP 4490. Front. Microbiol. 2021, 11, 618415. [Google Scholar] [CrossRef] [PubMed]
- Sondang, Y.; Anty, K.; Siregar, R. Isolation and Identification of Effective Microorganisms from Water Hyacinth Biofertilizer. IOP Conf. Ser. Earth Environ. Sci. 2021, 709, 012064. [Google Scholar] [CrossRef]
- Wang, K.; Qin, Z.; Wu, S.; Zhao, P.; Zhen, C.; Gao, H. Antifungal Mechanism of Volatile Organic Compounds Produced by Bacillus subtilis CF-3 on Colletotrichum gloeosporioides Assessed Using Omics Technology. J. Agric. Food Chem. 2021, 69, 5267–5278. [Google Scholar] [CrossRef]
- Paul, G.K.; Mahmud, S.; Dutta, A.K.; et al. Volatile Compounds of Bacillus pseudomycoides Induce Growth and Drought Tolerance in Wheat (Triticum aestivum L.). Sci. Rep. 2022, 12, 22354. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Cai, H.; Qin, S.; et al. Bacillus pinisoli sp. nov., Isolated from Soil of a Decayed Pine Tree. Curr. Microbiol. 2023, 80, 55. [Google Scholar] [CrossRef]
- Yara Malaysia. Iron Deficiency—Oil Palm. Yara Malaysia; 2018.
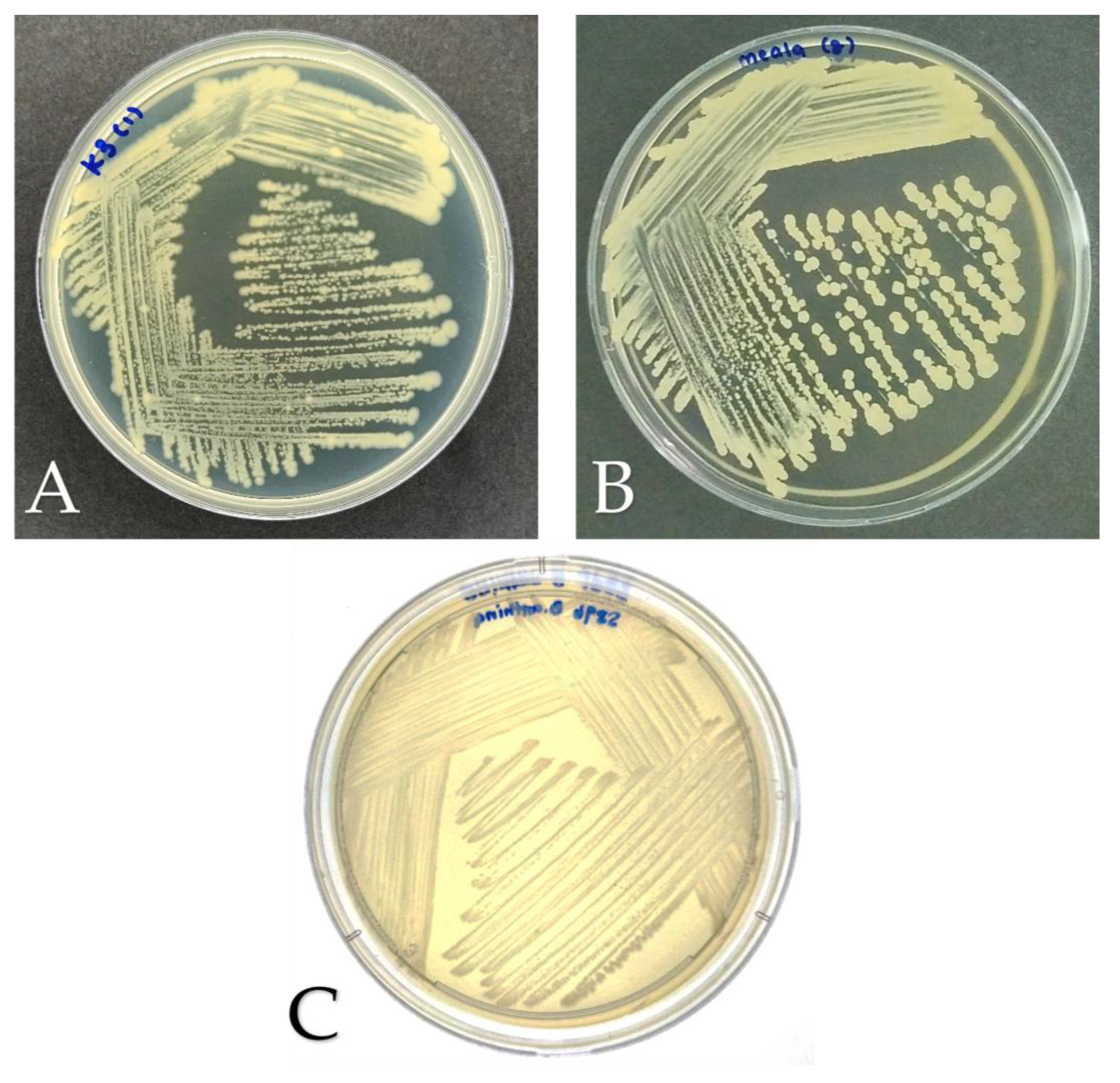
| Bacteria | Colony Morphology | ||||||
| Gram staining | Shape | Surface | Texture | Color | Elevation | Margin | |
| K3 | Positive | Bacillus | Dry | Rough | White | Raised | Entire |
| K8 | Positive | Bacillus | Dry | Rough | White | Flat | Undulate |
| S89b (control) | Negative | Bacillus | Wet | Smooth | Yellow | Raised | Entire |
| Bacteria | Plant Growth-Promoting Properties | |||||
| Phosphate solubilization | Potassium solubilization | Nitrogen fixation | Siderophore production | IAA production | ACC deaminase production | |
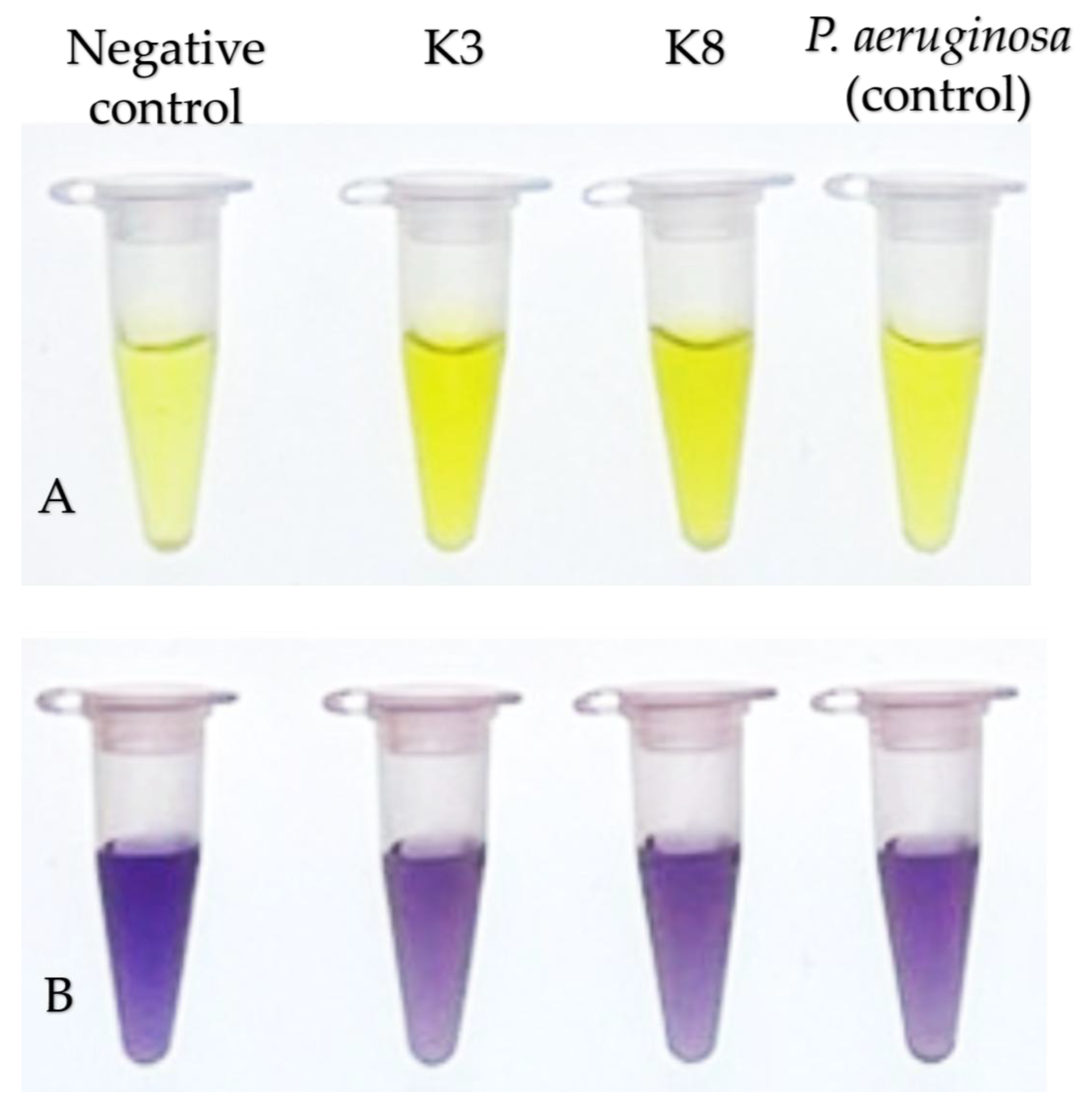
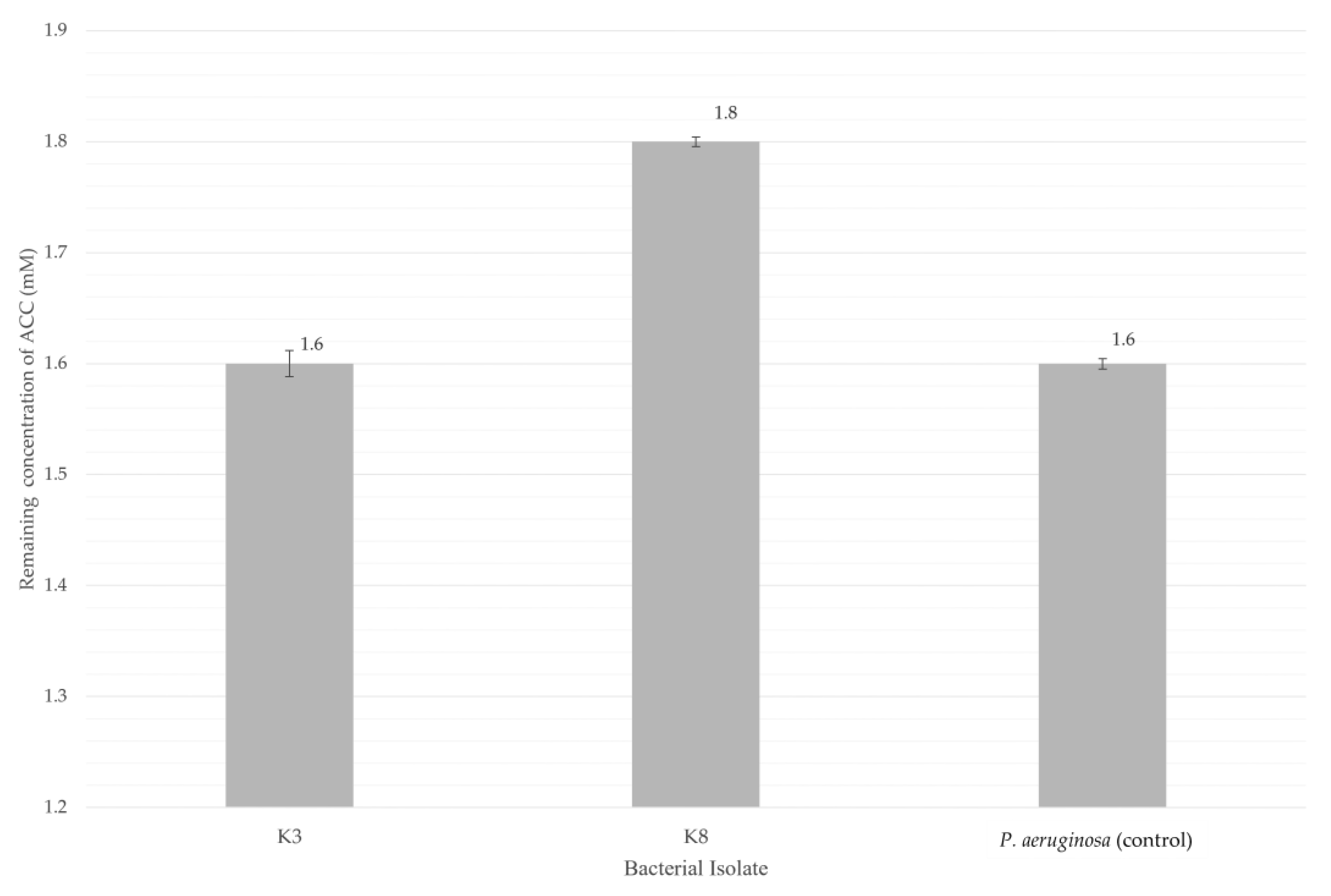
| K3 | Positive | Positive | Positive | Positive | +++ | Positive |
| K8 | Negative | Negative | Positive | Negative | - | Positive |
| P. aeruginosa (control) | Positive | Positive | Positive | Positive | - | Positive |
| Isolate | BLASTn identity | Query length | Query coverage (%) | E-value | Identity (%) | Accession number |
| K3 | Margalitia shackletonii | 1473 | 100.0% | 0.0 | 99.5 | OQ552639.1 |
| K8 | Bacillus subtilis | 1399 | 100.0% | 0.0 | 99.7 | OP798061.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




