Submitted:
02 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Foot PVT and Task

2.2. Participants and Experimental Protocol
2.3. Sleep Assessment
2.4. Recorded Data
| Event No. | Color | Evaluation | Interval [ms] | RT [ms] | Elapsed time [ms] |
| 1 | Yellow | T | 6445 | 1108 | 7562 |
| 2 | Blue | T | 6369 | 611 | 14566 |
| 3 | False start | F | 9227 | -3623 | 20170 |
| 4 | Red | F | 7427 | 1105 | 28723 |
| 4_Retry | Red | T | 0 | 1770 | 30493 |
| 5 | Yellow | T | 5062 | 909 | 36472 |
2.5. Statistical Analysis
3. Results
| Statistic (METs·h/week) | Younger (n=20) | Older (n=24) | Comment |
| Minimum | 0 | 0 | Both groups included participants with no regular exercise. |
| 25%tile | 0 | 5.3 | One quarter of younger adults did not exercise at all. |
| Median | 0 | 15.0 | Half of the younger adults had 0 METs·h/week, whereas older adults averaged 15. |
| 75%tile | 6.0 | 19.0 | Upper quartile of older adults exercised regularly. |
| Maximum | 20.5 | 98.0 | A few older adults were extremely active (outlier level). |
| Factor | Younger (n=20) | Older (n=24) | Test | p-value |
| Sleep1 | 47.5±8.6 | 49.1±9.0 | S | 0.532 |
| Sleep2 | 47.1±7.8 | 45.8±10.4 | S | 0.664 |
| Sleep3 | 50.6±10.1 | 49.7±9.9 | M | 0.930 |
| Sleep4 | 46.3±9.6 | 50.8±8.5 | S | 0.102 |
| Sleep5 | 47.0±7.6 | 51.2±11.8 | M | 0.194 |
| Height (cm) | 168±9 | 162±9 | S | 0.039 |
| PAL (METs・h/week) | 4.9±7.6 | 19.6±23.2 | M | 0.002 |
| Index | Younger (n=20) | older (n=24) | Test | p-value |
| RT mean (ms) | 700 ± 73 (666–734) |
818 ± 105 (773–863) |
S |
<0.001 |
| RT median (ms) | 675 ± 72 (641–709) |
796 ± 96 (754–837) |
S | <0.001 |
| RT SD (ms) | 122± 25 | 139± 53 | M | 0.283 |
| Skewness | 1.93± 0.99 | 1.41± 0.69 | S | 0.047 |
| Kurtosis | 7.20± 4.66 | 4.13± 3.24 | S | 0.014 |
| r (RT mean .vs. Skewness) | 0.054 | 0.109 | - | 0.822 Older:0.611) |
| r (RT mean .vs. Kurtosis) | 0.252 | -0.135 | - | 0.283 (Older:0.528) |
| Factor | Younger (n=20) | Older (n=24) | All (n=44) | |||
| r | p-value | r | p-value | r | p-value | |
| Sleep1 | -0.557 | 0.011 | -0.261 | 0.216 | -0.248 | 0.104 |
| Sleep2 | -0.223 | 0.346 | -0.184 | 0.391 | -0.200 | 0.194 |
| Sleep3 | 0.303 | 0.194 | 0.093 | 0.664 | 0.115 | 0.457 |
| Sleep4 | -0.363 | 0.115 | -0.138 | 0.523 | -0.041 | 0.791 |
| Sleep5 | -0.388 | 0.091 | -0.267 | 0.206 | -0.127 | 0.411 |
| Height | -0.593 | 0.006 | -0.279 | 0.188 | -0.478 | 0.001 |
| PAL (METs・h/week) | -0.269 | 0.252 | 0.231 | 0.277 | 0.325 | 0.032 |
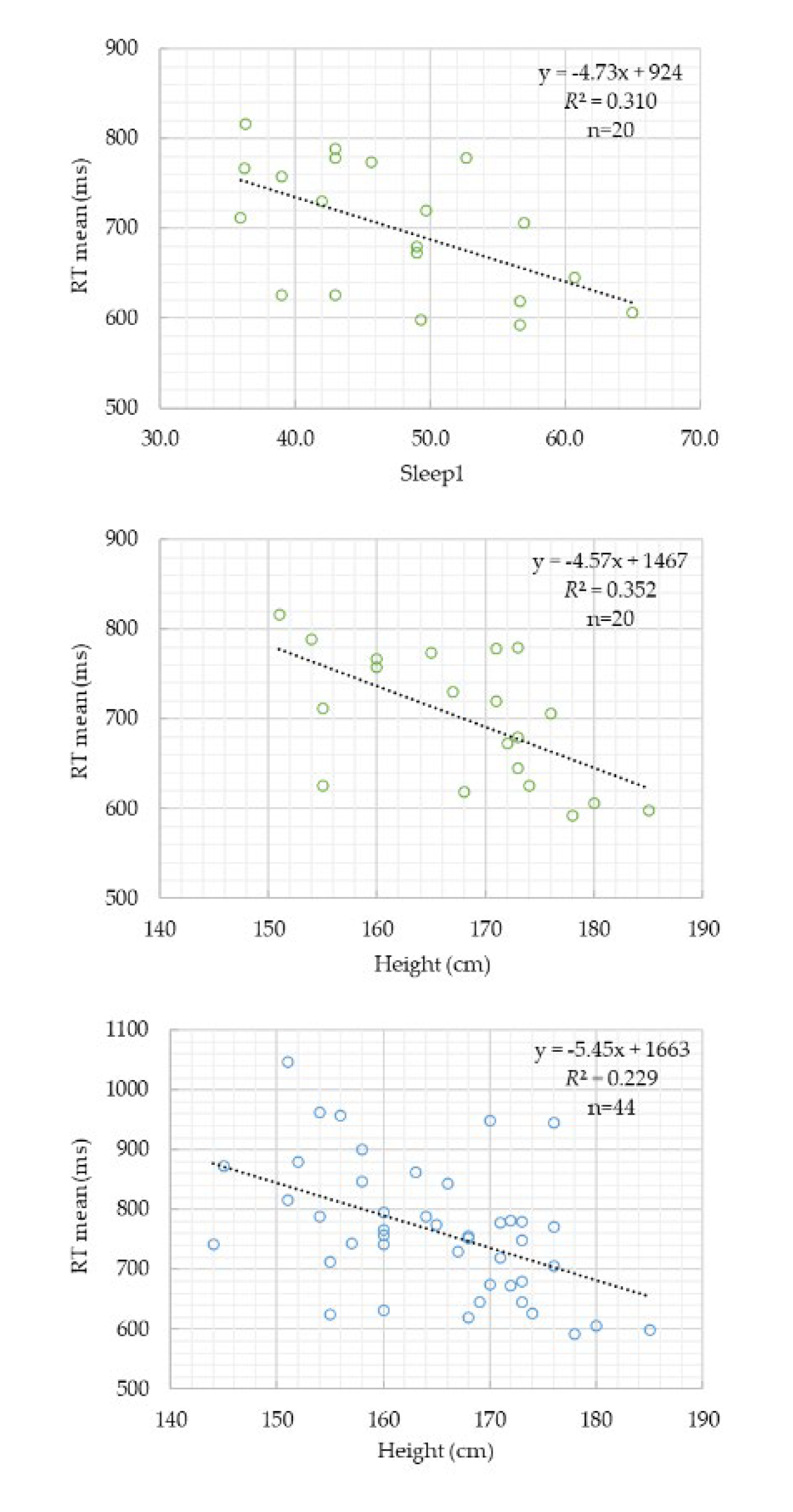
| Model | Predictor | B | SE | t | p | Adjusted R² | F(p) |
| (i) Younger | Constant | 1467.18 | 245.67 | 5.97 | 0.000 | 0.316 | 13.75 (0.006) |
| Height (cm) | -4.57 | 1.46 | -3.13 | 0.006 | |||
| (ii) Older | Constant | 818.15 | 21.81 | 37.52 | 0.000 | 0 | — |
| (iii) Combined |
Constant | 1258.94 | 255.71 | 4.92 | 0.000 | 0.372 | 13.75 (0.001) |
| Height (cm) | -3.89 | 1.45 | -2.69 | 0.010 | |||
| Age group | 95.12 | 27.66 | 3.44 | 0.001 |
| Category |
Young (n = 20) |
Older (n = 24) |
Category |
Young (n = 20) |
Older (n = 24) |
Correction time (ms) |
| Miss count | Count of participants | False start count | Count of participants | Mean±SD | ||
| 0 times | 11 | 16 | 0 times | 10 | 19 | Young: 666 ± 347 Older: 714 ± 225 |
| 1 time | 4 | 6 | 1 time | 2 | 2 | |
| 2 times | 4 | 1 | 2 times | 5 | 3 | |
| 8 times | 1 | 1 | 3–9 times | 3 | 0 | |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PVT | psychomotor vigilance test |
| Foot PVT | foot-response version of the PVT |
| RT | reaction time |
| PAL | physical activity level |
References
- Dinges, D.F.; Powell, J.W. Microcomputer analysis of performance on a portable, simple visual RT task during sustained opera tions. Behav. Res. Methods Instrum. Comput. 1985, 17, 652–655.
- Dinges, D.F.; Pack, F.; Williams, K.; Gillen, K.A.; Powell, J.W.; Ott, G.E.; Aptowicz, C.; Pack, A.I. Cumulative sleepiness, mood disturbance, and psychomotor vigilance performance decrements during a week of sleep restricted to 4–5 hours per night. Sleep 1997, 20, 267–277.
- Basner, M.; Moore, T.M.; Nasrini, J.; Gur, R.C.; Dinges, D.F. Response speed measurements on the psychomotor vigilance test: How precise is precise enough? Sleep 2021, 44, zsaa121.
- Chaisilprungraung, T.; Stekl, E.K.; Thomas, C.L.; Blanchard, M.E.; Hughes, J.D.; Balkin, T.J.; Doty, T.J. Quantifying the effects of sleep loss: Relative effect sizes of the psychomotor vigilance test, multiple sleep latency test, and maintenance of wakefulness test. Sleep Adv. 2022, 3, zpac034.3.
- Jones, M.J.; Dunican, I.C.; Murray, K.; Peeling, P.; Dawson, B.; Halson, S.; Miller, J.; Eastwood, P.R. The psychomotor vigilance test: A comparison of different test durations in elite athletes. J. Sports Sci. 2018, 36(18), 2033–2037.
- Basner M, Moore TM, Nasrini J, Gur RC, Dinges DF. Response speed measurements on the psychomotor vigilance test: how precise is precise enough? Sleep. 2021, 44, zsaa121.
- Mollicone DJ, Kan K, Coats S, Mott C, van Wollen M, Hatch A, Gallagher J, Williams S, Motzkin D. Use of the psychomotor vigilance test to aid in the selection of risk controls in an air medical transport operation. Sleep Adv. 2023, 4, zpad003.
- Antler CA, Yamazaki EM, Casale CE, Brieva TE, Goel N. The 3-Minute Psychomotor Vigilance Test Demonstrates Inadequate Convergent Validity Relative to the 10-Minute Psychomotor Vigilance Test Across Sleep Loss and Recovery. Front. Neurosci. 2022, 16, 815697.9.
- Thompson BJ, Shugart C, Dennison K, Louder TJ. Test-retest reliability of the 5-minute psychomotor vigilance task in working-aged females. J. Neurosci. Methods 2022, 365, 109379.
- Ferris M, Bowles KA, Bray M, Bosley E, Rajaratnam SMW, Wolkow AP. The impact of shift work schedules on PVT performance in naturalistic settings: a systematic review. Int. Arch Occup. Environ. Health 2021, 94, 1475-1494.
- Çolak M, Esin MN. Factors affecting the psychomotor vigilance of nurses working night shift. Int. Nurs. Rev. 2024, 71 ,84-93.
- Grant DA, Honn KA, Layton ME, Riedy SM, Van Dongen HPA. 3-minute smartphone-based and tablet-based psychomotor vigilance tests for the assessment of reduced alertness due to sleep deprivation. Behav. Res. Methods. 2017,49, 1020-1029.
- Reifman, J.; Ramakrishnan, S.; Liu, J.; Kapela, A.; Doty, T.J.; Balkin, T.J.; Kumar, K.; Khitrov, M.Y. 2B-Alert App: A Mobile Application for Real-Time Individualized Prediction of Alertness. J. Sleep Res. 2019, 28, e12725.
- Droździel, P.; Tarkowski, S.; Rybicka, I.; Wrona, R. Drivers’ Reaction Time Research in the Conditions in the Real Traffic. Open Eng. 2020, 10, 35-47.
- Poliak, M.; Svabova, L.; Benus, J.; Demirci, E. Driver Response Time and Age Impact on the Reaction Time of Drivers: A Driving Simulator Study among Professional-Truck Drivers. Mathematics 2022, 10, 1489.
- Bauder, M.; Paula, D.; Pfeilschifter, C.; Petermeier, F.; Kubjatko, T.; Riener, A.; Schweiger, H.-G. Influences of Vehicle Communication on Human Driving Reactions: A Simulator Study on Reaction Times and Behavior for Forensic Accident Analysis. Sensors 2024, 24, 4481.
- Ministry of Internal Affairs and Communications. Statistics Topics No.142. Available online: https://www.stat.go.jp/data/topics/pdf/topics142.pdf (accessed on 1 Nov 2025).
- CabinetOffice, Government of Japan. Available online: https://www8.cao.go.jp/koutu/taisaku/r02kou_haku/zenbun/genkyo/feature/feature_01_3.html (accessed on 1 November 2025).
- Tokyo Metropolitan Police Department. Preventing Traffic Accidents Involving Elderly Drivers. Tokyo Metropolitan Police Department 2024. Available online: https://www.keishicho.metro.tokyo.lg.jp/kotsu/jikoboshi/koreisha/koreijiko.html (accessed on 1 November 2025).
- Yoshida, Y.; Yuda, E.; Yokoyama, K. Design of the new Foot PVT for screening driving ability. Hardware 2025, 3, 3.
- Mota Albuquerque P, Ribeiro Franco CM, Sampaio Rocha-Filho PA. Assessing the impact of sleep restriction on the attention and executive functions of medical students: a prospective cohort study. Acta. Neurol. Belg. 2023, 123, 1421-1427.
- Chiaramonte, R.; Pavone, V.; Testa, G.; Pesce, I.; Scaturro, D.; Musumeci, G.; Mauro, G.L.; Vecchio, M. The Role of Physical Exercise and Rehabilitative Implications in the Process of Nerve Repair in Peripheral Neuropathies: A Systematic Review. Diagnostics 2023, 13, 364.
- Alonso, A.C.; Luna, N.M.; Mochizuki, L.; Barbieri, F.; Santos, S.; Greve, J.M. The influence of anthropometric factors on postural balance: The relationship between body composition and posturographic measurements in young adults. Clinics (Sao Paulo) 2012, 67, 1433–1441.
- Okamura, H.; Mihara, K.; Tsuda, A.; Morisaki, T.; Tanaka, Y.; Shoji, Y. Subjective Happiness Is Associated with Objectively Evaluated Sleep Efficiency and Heart Rate during Sleep: An Exploratory Study Using Non-Contact Sheet Sensors. Sustainability 2020, 12, 4630.
- Japan Organization of Better Sleep (JOBS). OSA Sleep Inventory MA version. Available online: https://www.jobs.gr.jp/osa_ma.html (accessed on 1 Nov 2025).
- Herrmann, S.D.; Willis, E.A.; Ainsworth, B.E.; Barreira, T.V.; Hastert, M.; Kracht, C.L.; Schuna, J.M. Jr; Cai, Z.; Quan, M.; Tudor-Locke, C.; Whitt-Glover, M.C.; Jacobs, D.R. Jr. 2024 Adult Compendium of Physical Activities: A Third Update of the Energy Costs of Human Activities. J. Sport Health Sci. 2024, 13, 6–12.
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R., Jr.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. 2011 Compendium of Physical Activities: A second update of codes and MET values. Medicine & Science in Sports & Exercise 2011, 43, 1575–1581.
- National Institutes of Biomedical Innovation, Health and Nutrition. Revised “List of Physical Activity METs (METs Table)”; Available online: https://www.nibn.go.jp/eiken/programs/2011mets.pdf (accessed on 1 November 2025).
- Osuka, Y.; Kojima, N.; Sugie, M.; Omura, T.; Motokawa, K.; Maruo, K.; Ono, R.; Aoyama, T.; Inoue, S.; Kim, H.; Sasai, H. Effects of a Home-Based Radio-Taiso Exercise Programme on Health-Related Quality of Life in Older Adults with Frailty: Protocol for an Assessor-Blind Randomized Controlled Trial. BMJ Open. 2022, 12, e063201.
- Doroudgar, S.; Chuang, H.M.; Perry, P.J.; Thomas, K.; Bohnert, K.; Canedo, J. Driving Performance Comparing Older versus Younger Drivers. Traffic Inj. Prev. 2017, 18, 41-46.
- Cooper, J.M.; Wheatley, C.L.; McCarty, M.M.; Motzkus, C.J.; Lopes, C.L.; Erickson, G.G.; Baucom, B.R.W.; Horrey, W.J.; Strayer, D.L. Age-Related Differences in the Cognitive, Visual, and Temporal Demands of In-Vehicle Information Systems. Front. Psychol. 2020, 11, 1154.
- Depestele, S.; Ross, V.; Verstraelen, S.; Brijs, K.; Brijs, T.; van Dun, K.; Meesen, R. The Impact of Cognitive Functioning on Driving Performance of Older Persons in Comparison to Younger Age Groups: A Systematic Review. Transp. Res. Part F Traffic Psychol. Behav. 2020, 73, 433-452.
- Robertsen, R.; Lorås, H.W.; Polman, R.; Simsekoglu, O.; Sigmundsson, H. Aging and Driving: A Comparison of Driving Performance Between Older and Younger Drivers in an On-Road Driving Test. SAGE Open 2022, 12, 2.
- Mouloua, M.; Rinalducci, E.; Smither, J.; Brill, J.C. Effect of Aging on Driving Performance. Proc. Hum. Factors Ergon. Soc. Annu. Meet. 2004, 48, 253-257.
- Greene, W.R.; Smith, R. Driving in the Geriatric Population. Clin. Geriatr. Med. 2019, 35, 127-131.
- Rivner, M.H.; Swift, T.R.; Malik, K. Influence of age and height on nerve conduction. Muscle Nerve 2001, 24, 1134-1141.
- Thakker, D.V.; Kariya, V.B. A Cross-Sectional Study of Comparison between Individual Height and Median Nerve Conduction Velocity. Sch. Int. J. Anat. Physiol. 2019, 2, 325-329.
- Tjolleng, A.; Yang, J.; Jung, K. Analysis of Leg Muscle Activities and Foot Angles while Pressing the Accelerator Pedal by Different Foot Postures. Appl. Sci. 2022, 12, 13025.
- Dinges, D.F.; Pack, F.; Williams, K.; Gillen, K.A.; Powell, J.W.; Ott, G.E.; Aptowicz, C.; Pack, A.I. Cumulative sleepiness, mood disturbance, and psychomotor vigilance performance decrements during a week of sleep restricted to 4-5 hours per night. Sleep 1997, 20(4), 267-277.
- Lim, J.; Dinges, D.F. Sleep deprivation and vigilant attention. Ann. N. Y. Acad. Sci. 2008, 1129, 305-322.
- Mattis, J.; Sehgal, A. Circadian Rhythms, Sleep, and Disorders of Aging. Trends Endocrinol. Metab. 2016, 27(4), 192-42.
- Schmidt, C.; Peigneux, P.; Cajochen, C. Age-related changes in sleep and circadian rhythms: Impact on cognitive performance and underlying neuroanatomical networks. Front. Neurol. 2012, 3, 118.
- Landry, G.J.; Best, J.R.; Liu-Ambrose, T. Measuring sleep quality in older adults: A comparison using subjective and objective methods. Front. Aging Neurosci. 2015, 7, 166.
- Yuda, E.; Yoshida, Y. Individual Differences in Sustained Attention: Effects of Age, Sex, and Time of Day Based on Psychomotor Vigilance Task Performance. Appl. Sci. 2025, 15, 5487.
- Voelcker-Rehage, C.; Niemann, C. Structural and functional brain changes related to different types of physical activity across the life span. Neurosci. Biobehav. Rev. 2013, 37, 2268–2295.
- Colcombe, S.; Kramer, A.F. Fitness effects on the cognitive function of older adults: A meta-analytic study. Psychol. Sci. 2003, 14, 125–130.
- Shi, B.; Mou, H.; Tian, S.; Meng, F.; Qiu, F. Effects of Acute Exercise on Cognitive Flexibility in Young Adults with Different Levels of Aerobic Fitness. Int. J. Environ. Res. Public Health 2022, 19, 9106.
- Sun, Q.; Xu, Z.; Lyu, D.; Xu, X.; Wang, L.; Yan, T.; Yan, J. Physical activity and cognitive difficulties in adolescents: A cross-sectional study of 13,677 participants. Complement. Ther. Clin. Pract. 2025, 59, 101965.
- Basner, M.; Dinges, D.F. Maximizing sensitivity of the psychomotor vigilance test (PVT) to sleep loss. Sleep 2011, 34, 581–591.
- Yamashita, A.; Rothlein, D.; Kucyi, A.; Valera, E.M.; Germine, L.; Wilmer, J.; DeGutis, J.; Esterman, M. Variable Rather Than Extreme Slow Reaction Times Distinguish Brain States during Sustained Attention. Sci. Rep. 2021, 11, 14883.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




