Submitted:
01 December 2025
Posted:
04 December 2025
You are already at the latest version
Abstract
Background: Human Immunodeficiency Virus type 1 (HIV-1) remains a major global health challenge. Despite advances in antiretroviral therapy, new prevention strategies are needed, particularly topical microbicides capable of blocking the earliest steps of viral entry. HIV-1 attachment relies on interactions with heparan sulfate proteoglycans on host cell surfaces; therefore, sulfated heparan-mimetic polymers have been explored as antiviral agents. In this context, sulfated chitosan microparticles are designed to mimic natural glycosaminoglycan receptors, acting as biomimetic decoys that prevent viral attachment and entry. Methods: Low molecular weight sulfated chitosan (LMW Ch-S) microparticles were synthesized and characterized (FTIR, SEM) following US Patent No. 11,246,839 B2 (Bucarey et al., 2022). Their antiviral activity was evaluated by incubating the microparticles with high-viral-load HIV-1–positive plasma to enable viral binding and removal by pull-down. The performance of the synthesized Ch-S microparticles was compared with established heparinoid controls, including soluble heparin and heparin microparticles. Results: The Ch-S microparticles exhibited stronger virus-binding and neutralizing capacity than all heparinoid comparators, achieving the highest overall reduction in viral load. Subsequent evaluation across multiple tested concentrations confirmed a consistent antiviral effect, indicating that the synthesized Ch-S microparticles maintain robust virus–particle interactions throughout the concentration range examined. Conclusions: These findings demonstrate that LMW Ch-S microparticles possess potent antiviral properties and outperform classical heparinoid materials, supporting their potential application as topical microbicides targeting early HIV-1 entry mechanism.
Keywords:
1. Introduction
2. Results
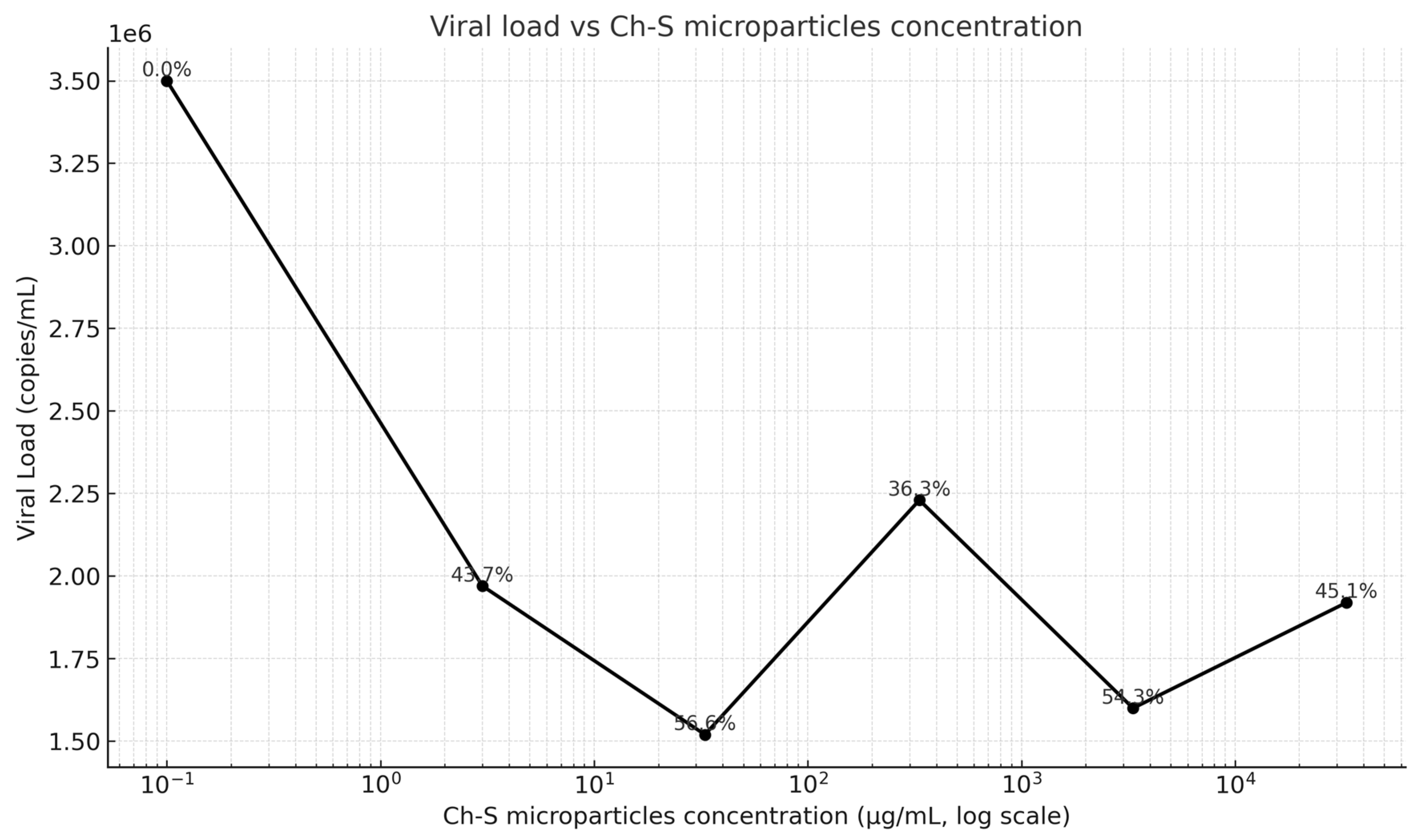
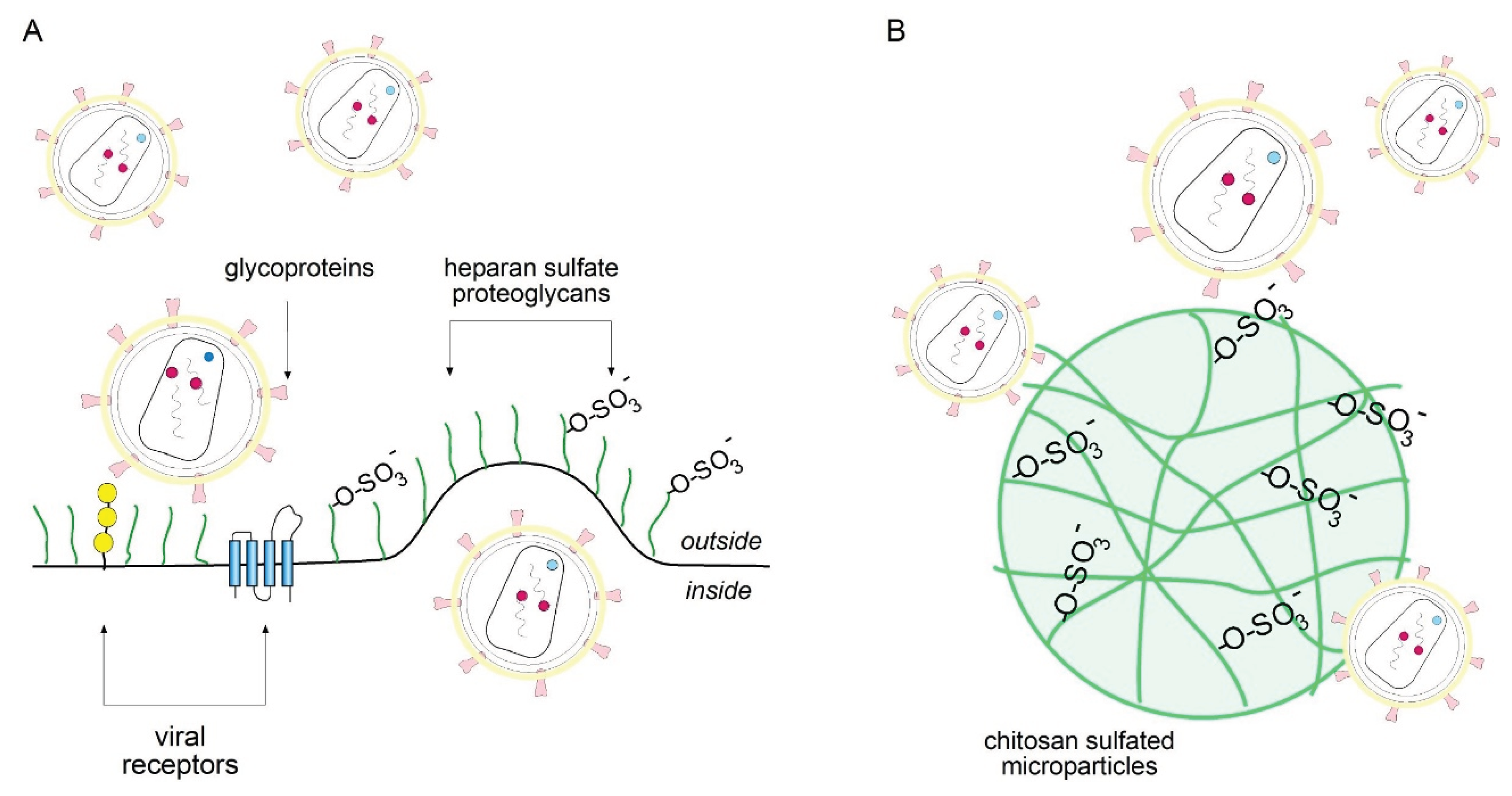
3. Discussion
4. Materials and Methods
4.1. Synthesis of Sulfated Chitosan (Ch-S) Microparticles
4.2. Morphostructural and Elemental Characterization
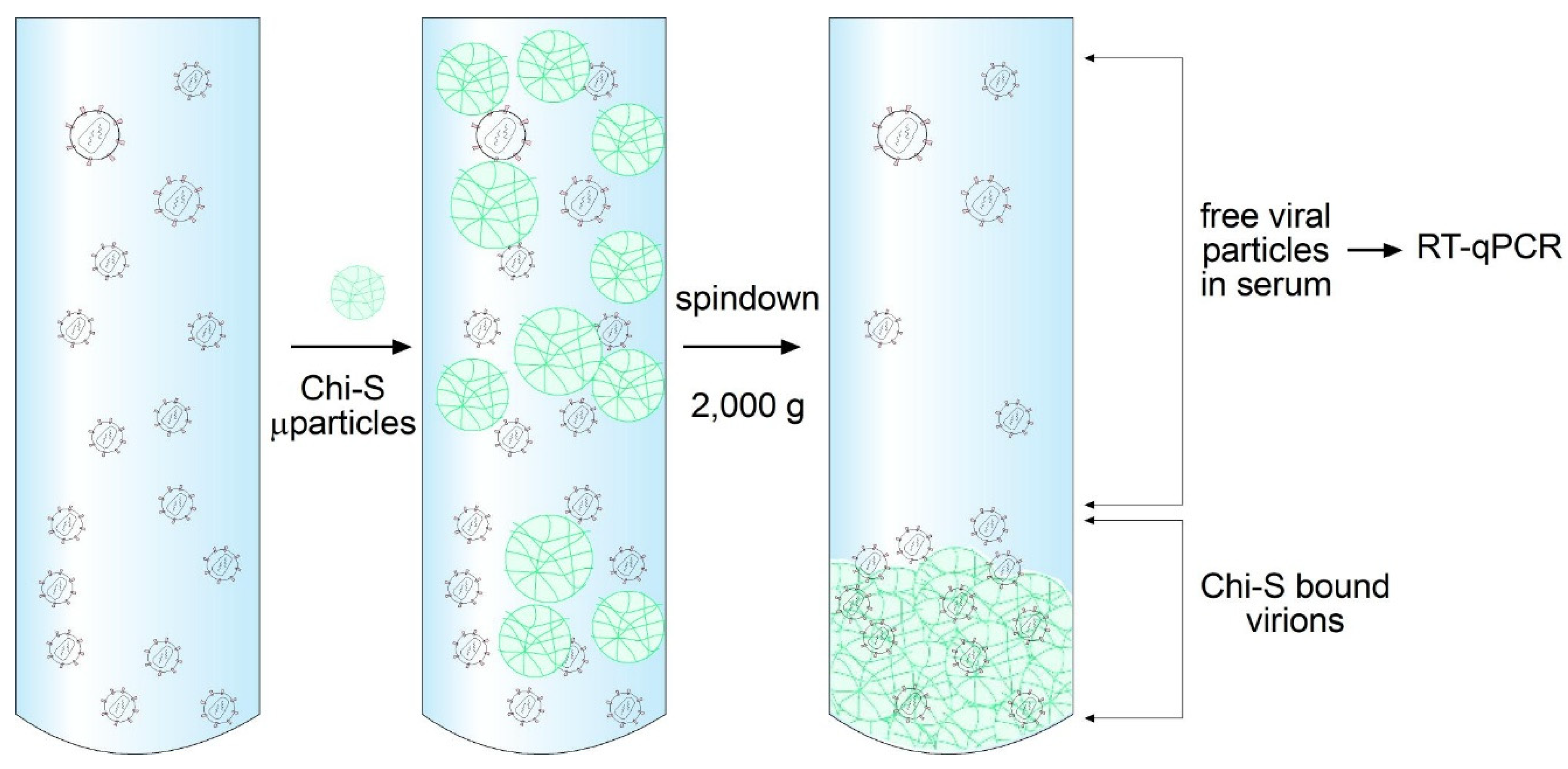
4.3. Virus Binding Assay
4.4. HIV-1 Viral Load Quantification Protocol
4.5. Dose-Response Assay
4.6. Reagents
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Global HIV & AIDS Statistics — Fact Sheet | UNAIDS. Available online: https://www.unaids.org/en/resources/fact-sheet (accessed on 27 November 2025).
- Pirrone, V.; Wigdahl, B.; Krebs, F.C. The Rise and Fall of Polyanionic Inhibitors of the Human Immunodeficiency Virus Type 1. Antiviral Res. 2011, 90, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Connell, B.J.; Lortat-Jacob, H. Human Immunodeficiency Virus and Heparan Sulfate: From Attachment to Entry Inhibition. Front. Immunol. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Harrop, H.A.; Rider, C.C. Heparin and Its Derivatives Bind to HIV-1 Recombinant Envelope Glycoproteins, Rather than to Recombinant HIV-1 Receptor, CD4. Glycobiology 1998, 8, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Mondor, I.; Ugolini, S.; Sattentau, Q.J. Human Immunodeficiency Virus Type 1 Attachment to HeLa CD4 Cells Is CD4 Independent and Gp120 Dependent and Requires Cell Surface Heparans. J. Virol. 1998, 72, 3623–3634. [Google Scholar] [CrossRef] [PubMed]
- Witvrouw, M.; De Clercq, E. Sulfated Polysaccharides Extracted from Sea Algae as Potential Antiviral Drugs. Gen. Pharmacol. 1997, 29, 497–511. [Google Scholar] [CrossRef] [PubMed]
- Aranaz, I.; Mengibar, M.; Harris, R.; Panos, I.; Miralles, B.; Acosta, N.; Galed, G.; Heras, A. Functional Characterization of Chitin and Chitosan. http://www.eurekaselect.com.
- Jiménez-Arriagada, D.; Hidalgo, A.A.; Neira, V.; Neira-Carrillo, A.; Bucarey, S.A. Low Molecular Weight Sulfated Chitosan Efficiently Reduces Infection Capacity of Porcine Circovirus Type 2 (PCV2) in PK15 Cells. Virol. J. 2022, 19, 52. [Google Scholar] [CrossRef] [PubMed]
- Soppimath, K.S.; Aminabhavi, T.M.; Kulkarni, A.R.; Rudzinski, W.E. Biodegradable Polymeric Nanoparticles as Drug Delivery Devices. J. Controlled Release 2001, 70, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, H.; Inazawa, K.; Ichiyama, K.; Ito, M.; Ikushima, N.; Shoji, T.; Katsuraya, K.; Uryu, T.; Yamamoto, N.; Juodawlkis, A.S.; et al. Sulfated Alkyl Oligosaccharides Inhibit Human Immunodeficiency Virus in Vitro and Provide Sustained Drug Levels in Mammals. Antivir. Chem. Chemother. 1995, 6, 271–280. [Google Scholar] [CrossRef]
- Montefiori, D.C. Measuring HIV Neutralization in a Luciferase Reporter Gene Assay. Methods Mol. Biol. Clifton NJ 2009, 485, 395–405. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-Based Nanoparticles: An Overview of Biomedical Applications. J. Control. Release Off. J. Control. Release Soc. 2012, 161, 505–522. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, U.; Li, J. Interactions between Heparan Sulfate and Proteins-Design and Functional Implications. Int. Rev. Cell Mol. Biol. 2009, 276, 105–159. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.; Wiegand, A.P.; Maldarelli, F.; Bazmi, H.; Mican, J.M.; Polis, M.; Dewar, R.L.; Planta, A.; Liu, S.; Metcalf, J.A.; et al. New Real-Time Reverse Transcriptase-Initiated PCR Assay with Single-Copy Sensitivity for Human Immunodeficiency Virus Type 1 RNA in Plasma. J. Clin. Microbiol. 2003, 41, 4531–4536. [Google Scholar] [CrossRef] [PubMed]
- Connell, B.J.; Lortat-Jacob, H. Human Immunodeficiency Virus and Heparan Sulfate: From Attachment to Entry Inhibition. Front. Immunol. 2013, 4, 385. [Google Scholar] [CrossRef] [PubMed]
- Connell, B.J.; Baleux, F.; Coic, Y.-M.; Clayette, P.; Bonnaffé, D.; Lortat-Jacob, H. A Synthetic Heparan Sulfate-Mimetic Peptide Conjugated to a Mini CD4 Displays Very High Anti- HIV-1 Activity Independently of Coreceptor Usage. Chem Biol 2012, 19, 131–139. [Google Scholar] [CrossRef] [PubMed]
- De Pasquale, V.; Quiccione, M.S.; Tafuri, S.; Avallone, L.; Pavone, L.M. Heparan Sulfate Proteoglycans in Viral Infection and Treatment: A Special Focus on SARS-CoV-2. Int. J. Mol. Sci. 2021, 22, 6574. [Google Scholar] [CrossRef] [PubMed]
- Cagno, V.; Tseligka, E.D.; Jones, S.T.; Tapparel, C. Heparan Sulfate Proteoglycans and Viral Attachment: True Receptors or Adaptation Bias? Viruses 2019, 11, 596. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Vogan, E.M.; Gong, H.; Skehel, J.J.; Wiley, D.C.; Harrison, S.C. Structure of an Unliganded Simian Immunodeficiency Virus Gp120 Core. Nature 2005, 433, 834–841. [Google Scholar] [CrossRef] [PubMed]
- D’Cruz, O.J.; Uckun, F.M. Clinical Development of Microbicides for the Prevention of HIV Infection. Curr. Pharm. Des. 2004, 10, 315–336. [Google Scholar] [CrossRef] [PubMed]
- Cone, R.A. Barrier Properties of Mucus. Adv. Drug Deliv. Rev. 2009, 61, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Bucarey Sergio; Neira-Carrillo Andrónico; Neira Victor, S.A. US20210059951 Vaccine Treatment and Control Infectious against Viral Pathogens Utilizing Heparan Sulfate (HS) as Cellular Receptor. Available online: https://patentscope.wipo.int/search/en/detail.jsf?docId=US319556756&_cid=P22-MIJQD7-64114-1 (accessed on 29 November 2025).
- Bucarey, S.A.; Pujol, M.; Poblete, J.; Nuñez, I.; Tapia, C.V.; Neira-Carrillo, A.; Martinez, J.; Bassa, O. Chitosan Microparticles Loaded with Yeast-Derived PCV2 Virus-like Particles Elicit Antigen-Specific Cellular Immune Response in Mice after Oral Administration. Virol. J. 2014, 11, 149. [Google Scholar] [CrossRef] [PubMed]
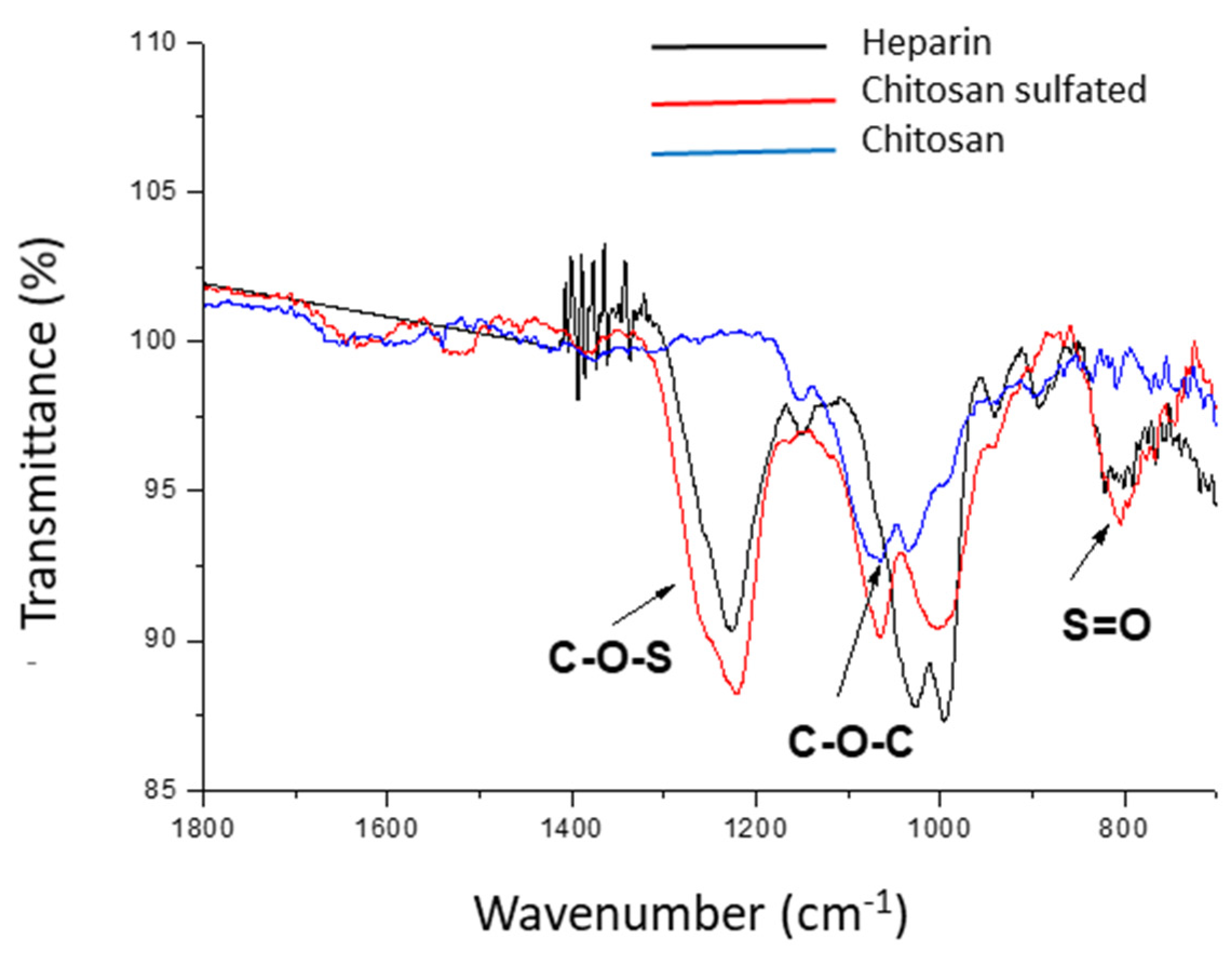

| Element | Concentration (%) |
| C | 41.35 |
| N | 9.68 |
| O | 44.4 |
| S | 4.56 |
| Total | 100 |
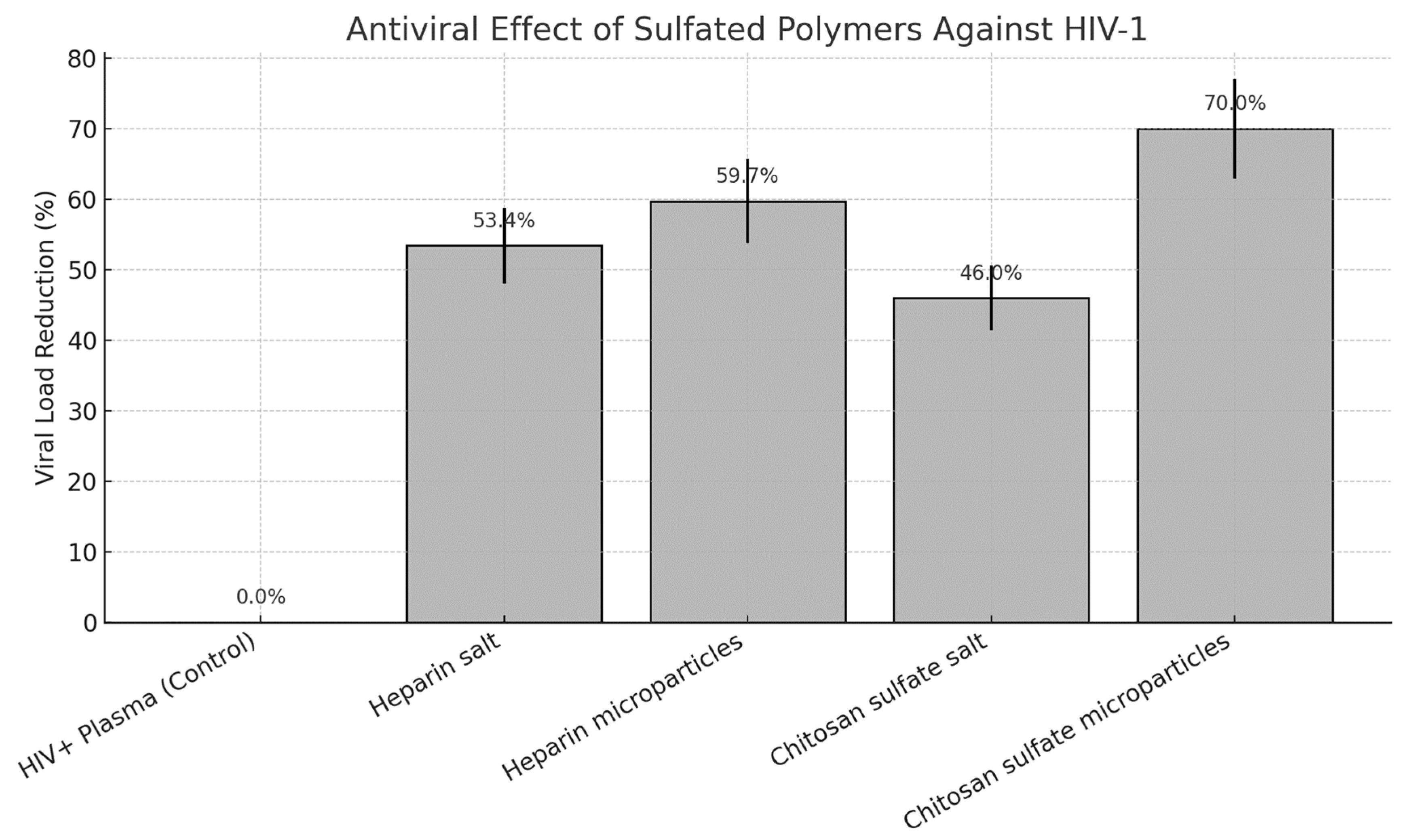
| Sample | Viral Load (copies/mL) | log10 | % Reduction ± SD |
| HIV+ Plasma | 3,500,000 | 6.54 | 0.00% ± 0.00 |
| Heparin salt | 1,630,000 | 6.21 | 53.43% ± 5.34 |
| Heparin microparticles | 1,410,000 | 6.15 | 59.71% ± 5.97 |
| Chitosan sulfate salt | 1,890,000 | 6.28 | 46.00% ± 4.60 |
| Chitosan sulfate microparticles | 1,050,000 | 6.02 | 70.00% ± 7.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




