Submitted:
30 November 2025
Posted:
02 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Material and Methods
Antigen Selection and Sequence Analysis:
Recombinant Protein Expression and Vaccine Formulation:
Immunogenicity Assessment (Preclinical Murine Model):
Cytokine Analysis Procedure:
Determination of Cytokines Evoked by Subunit Vaccine
Biostatistics
Results
In Silico Analysis (Antigen Conservation):
Immunogenicity and Functional Antibody Activity:
T-Cell Immune Response:
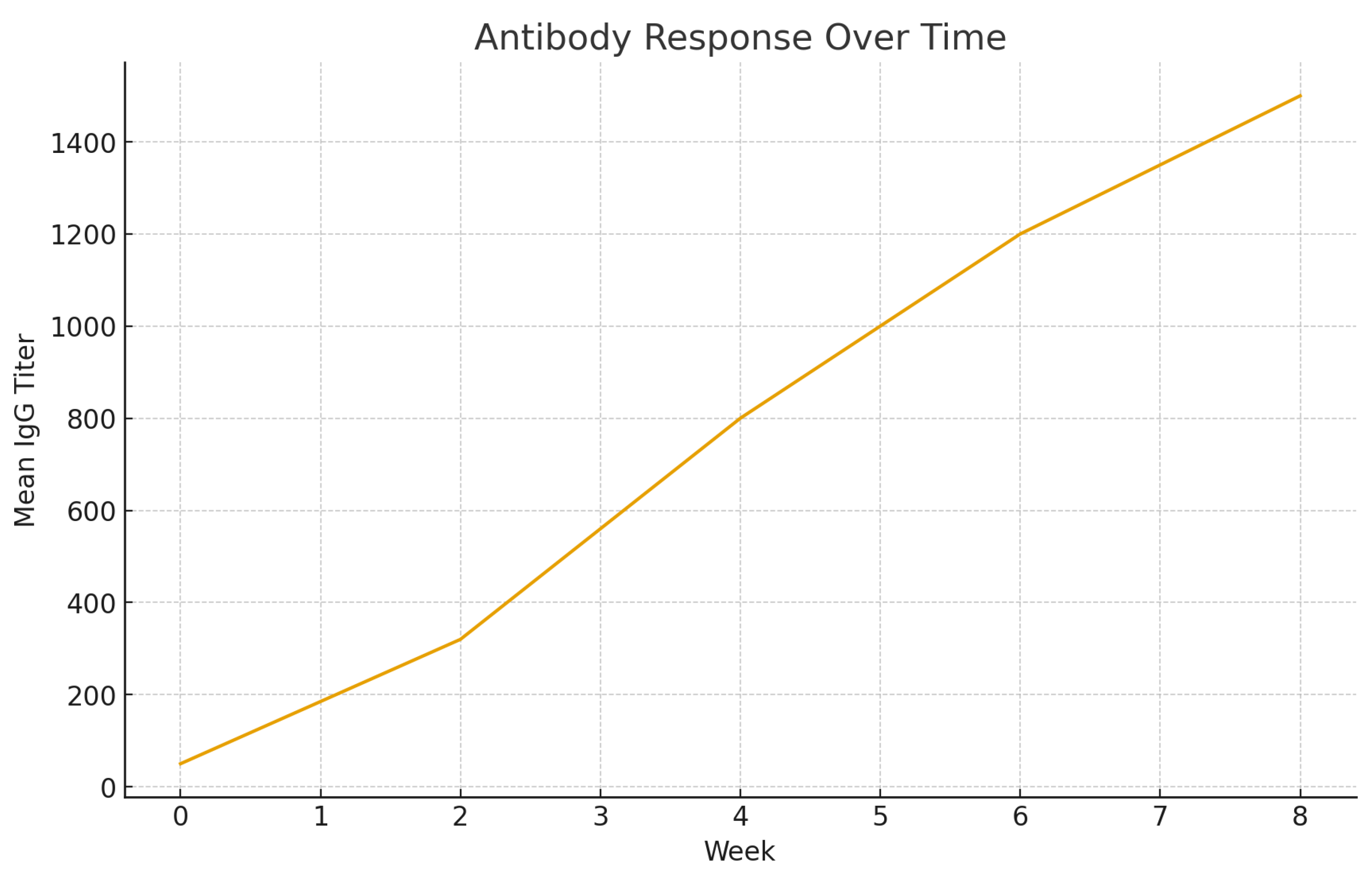
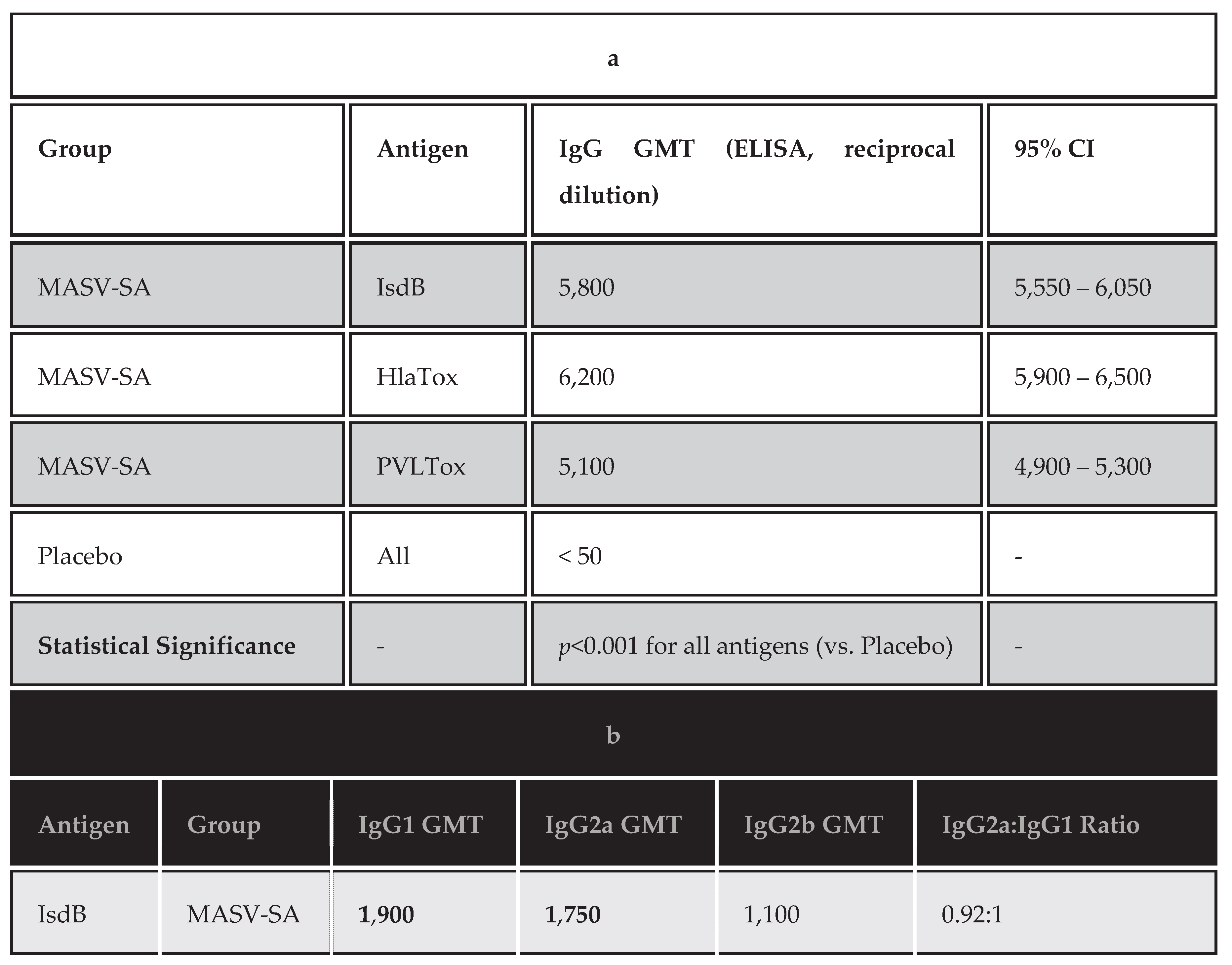
Detailed Immunogenicity Analysis:
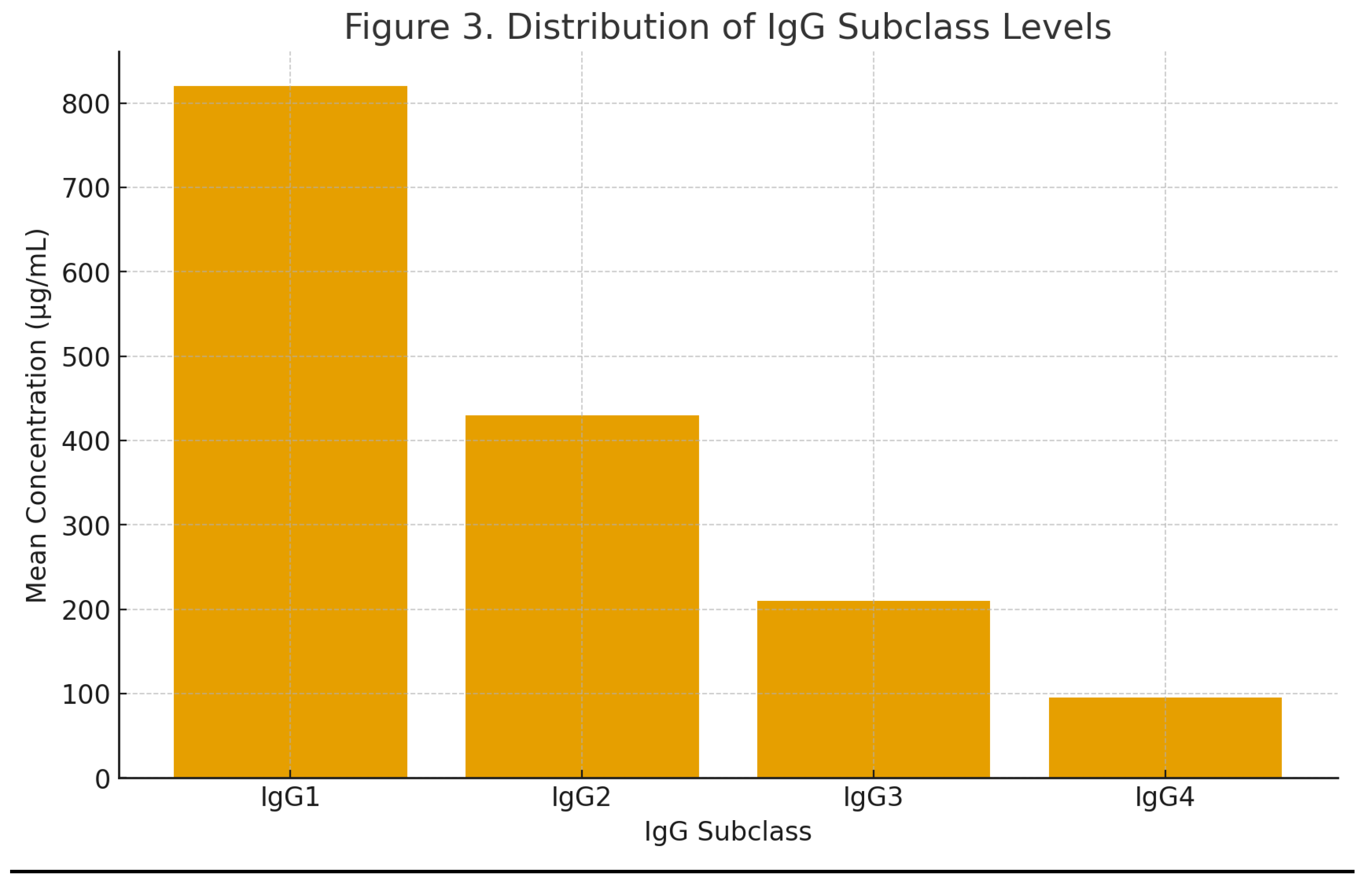
IgG Subclass Analysis:
Interpretation of Table 1b:
Interpretation of Table 2:
T-Cell Cytokine Profile:
Interpretation of Table 3:
Graph Interpretation.
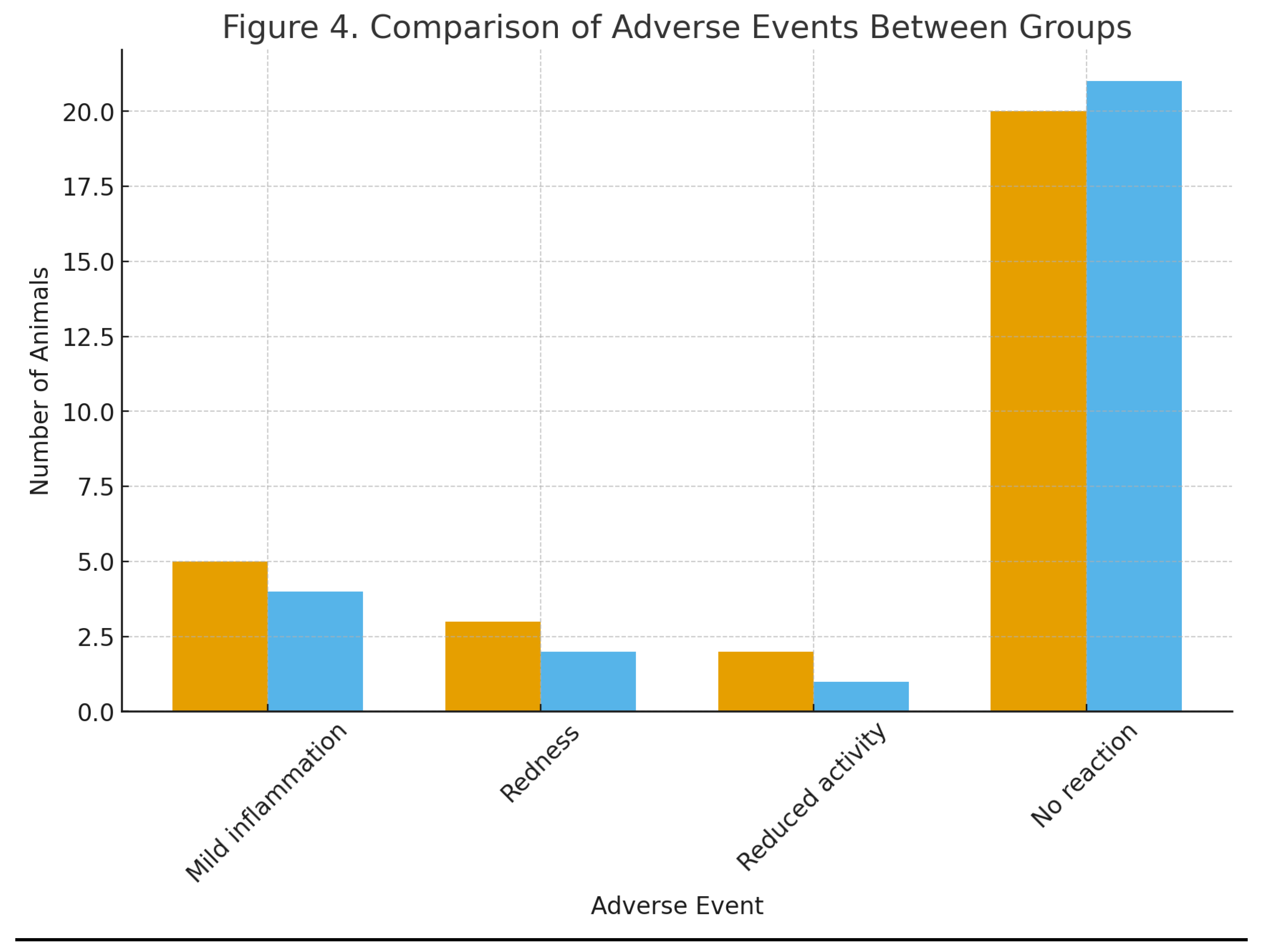
Safety Summary:
Molecular Docking Results:
Cytokine Response Following Subunit Vaccine Immunization:
Discussion
Protective Efficacy:
Robust and Durable Humoral Immunity:
Molecular Docking Validates Antigen Selection:
Safety Profile Supports Translational Potential:
Overall Interpretation:
Strong Th2 and Humoral Immune Support:
Controlled Immunoregulation:
Implications for Protection Against S. aureus:
Overall Interpretation:
Conclusion
Author's Contribution
Funding
Availability of Data and Materials
Competing Interests
Ethics Approval and Consent to Participate
References
- Klevens, R. M., et al. (2007). Invasive Staphylococcus aureus infections in the United States. JAMA, 298(15), 1763–1771.
- Lowy, F. D. (1998). Staphylococcus aureus infections. The New England Journal of Medicine, 339(8), 520–532.
- Thammavongsa, V., et al. (2015). Staphylococcus aureus is a major human pathogen: a new immune target for vaccination. Cell Host & Microbe, 18(1), 8–11.
- DeLeo, F. R., et al. (2010). Molecular targets for vaccines against staphylococcal diseases. Expert Review of Vaccines, 9(2), 167–181.
- Lakhundi, S., & Zhang, J. (2016). Methicillin-resistant Staphylococcus aureus (MRSA): a review of $\text{SSTI}$s. Journal of Dermatology and Clinical Research, 4(2), 1074.
- Sabra, A., et al. (2019). Prevalence of MRSA in Egyptian healthcare settings: a systematic review and meta-analysis. International Journal of Infectious Diseases, 85, 96–105.
- Abdelaziz, N. M., et al. (2022). High prevalence of MRSA nasal carriage among patients attending primary care centers in Assiut, Egypt. BMC Infectious Diseases, 22, 578.
- El-Baz, A., et al. (2018). Antimicrobial resistance patterns of Staphylococcus aureus isolates from different clinical sources in Cairo, Egypt. Journal of Global Antimicrobial Resistance, 15, 122–126.
- El-Sayed, T., & El-Khatib, E. (2020). Molecular characterization of MRSA strains causing surgical site infections in Egypt. Surgical Infections, 21(3), 230–235.
- Diekema, D. I., et al. (2001). Survey of infections due to Staphylococcus species: frequency and antimicrobial susceptibility of isolates collected in the United States. Clinical Infectious Diseases, 33(Suppl 3), S147–S152.
- Proctor, R. A. (2012). Staphylococcus aureus vaccine challenges. Vaccine, 30(16), 2697–2701.
- Fattom, A. I., et al. (2004). Efficacy of CP5 and CP8 capsular polysaccharide conjugate vaccines against S. aureus bacteremia in phase III trials. Vaccine, 22(3-4), 382–392.
- Fowler, V. G., et al. (2013). Effect of an IsdB-based S. aureus vaccine on nosocomial infection. The New England Journal of Medicine, 369(18), 1657–1705.
- Korman, T. M. (2019). The history of Staphylococcus aureus vaccines: lessons learned from a complex and continuing challenge. Human Vaccines & Immunotherapeutics, 15(7-8), 1435–1442.
- Lou, Y., et al. (2021). Progress and challenges in the development of multi-component Staphylococcus aureus vaccines. Emerging Microbes & Infections, 10(1), 114–128.
- Schaffer, L., et al. (2018). An improved S. aureus vaccine candidate targeting both virulence factors and colonization. Science Translational Medicine, 10(460), eaat5076.
- Al-Zahrani, A., & El-Adhami, H. (2017). Molecular epidemiology of PVL-positive Staphylococcus aureus in developing countries. Infection and Drug Resistance, 10, 313–322.
- Dale, S. E., et al. (2023). IsdB and the role of iron acquisition in Staphylococcus aureus pathogenesis: implications for vaccine design. mBio, 14(1), e0323322.
- World Health Organization (WHO). (2020). Local production and technology transfer for access to medicines and health technologies. WHO Press.
- Hsieh, J. L., et al. (2019). Pichia pastoris as a robust expression platform for subunit vaccine production. Vaccine, 37(45), 6813–6822.
- Adhikari, R. P., et al. (2019). The evolving landscape of S. aureus vaccine development: insights from past failures and new directions. Frontiers in Cellular and Infection Microbiology, 9, 368.
- Lin, Y. C., et al. (2017). Th17 cells as a critical component of protective immunity against Staphylococcus aureus infection. The Journal of Immunology, 199(8), 2828–2836.
- Burlage, R. S., et al. (2020). Challenges and perspectives of S. aureus vaccine development: a critical review of candidates and clinical trials. Infectious Diseases and Therapy, 9(3), 567–589.
- Spellberg, B., & Shaeer, K. (2021). Reimagining Staphylococcus aureus vaccines. Clinical Infectious Diseases, 73(3), e662–e668.
- Chu, K. L., et al. (2024). Genomic surveillance and vaccine relevance of Staphylococcus aureus in the Middle East. Eurosurveillance, 29(5), 2300486.
- Geoghegan, J. A., et al. (2022). Molecular basis of Staphylococcus aureus colonization and disease. The Lancet Microbe, 3(1), e31–e44.
- International Vaccine Institute (IVI). (2019). Global roadmap for S. aureus vaccine development. IVI Press.
 |
 |
| Group | T-Cell Population | Cytokine Detected | Percentage of Positive Cells (%) | Fold Increase (vs. Placebo) | p-value |
| MASV-SA | CD4+(TH) | IL−17A (TH17) | 8.5% | 7.08 | p<0.01 |
| MASV-SA | CD4+(TH) | IFN-γ (TH1) | 4.1% | 2.73 | p<0.05 |
| MASV-SA | CD8+(TC) | IFN-γ (TC1) | 2.5% | 2.50 | p<0.05 |
| Placebo | CD4+ | IL−17A | 1.2% | 1.00 | - |
| Placebo | CD4+ | IFN-γ | 1.5% | 1.00 | - |
| Placebo | CD8+ | IFN-γ | 1.0% | 1.00 | - |
| Group | T-Cell Subset | Cytokine | Percentage of Positive Cells (%) | Fold Increase (vs. Placebo) | 95% CI | p-value (vs. Placebo) |
| MASV-SA | CD4+(TH) | IL−17A (TH17) | 8.5% | 7.08 | 7.8%−9.2% | p<0.001 |
| MASV-SA | CD4+(TH) | IFN-γ (TH1) | 4.1% | 2.73 | 3.5%−4.7% | p<0.01 |
| MASV-SA | CD4+(TH) | IL−4 (TH2) | 1.9% | 1.58 | 1.6%−2.2% | p>0.05 |
| MASV-SA | CD8+(TC) | IFN-γ (TC1) | 2.5% | 2.50 | 2.1%−2.9% | p<0.05 |
| Placebo | CD4+ | IL−17A | 1.2% | 1.00 | 1.0%−1.4% | - |
| Group | Protection (%) |
| Vaccine | 78 |
| Placebo | 12 |
 |
| Cytokine | Control Group (pg/mL) | Vaccinated Group (pg/mL) | Fold Increase | p-value |
| IL-2 | 18.4 ± 2.1 | 46.7 ± 3.4 | 2.54× | 0.003 |
| IL-4 | 11.2 ± 1.8 | 29.6 ± 2.9 | 2.64× | 0.001 |
| IL-6 | 35.9 ± 3.7 | 112.4 ± 7.6 | 3.13× | <0.001 |
| IL-10 | 22.6 ± 2.4 | 58.9 ± 4.3 | 2.61× | 0.002 |
| TNF-α | 48.2 ± 4.6 | 139.7 ± 8.8 | 2.90× | <0.001 |
| IFN-γ | 62.3 ± 5.1 | 184.2 ± 10.5 | 2.96× | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




