Materials and Methods
This study examined the biophysical response of populations of the protozoan entamoeba gingivalis at pH values of 5.8 and 7.1 in the aquatic environment.
As demonstrated in [
4,
5,
6,
17,
18], the biophysical response of an amoeba population to an external electromagnetic field (EMF) can be recorded as a change in the aquatic resistance, with the entire biophysical response lasting approximately 1 hour. Thus, the biophysical response of an
entamoeba gingivalis population exposed to an EMF is a time-dependent process consisting of a constant value, a decrease in the constant value, and a smaller increase in the aquatic resistance. It will be interesting to determine how changes in the aquatic pH influence the biophysical radioresponse parameters of
entamoeba gingivalis, which will allow us to generalize the obtained results. The experiments used populations of
entamoeba gingivalis with concentrations of 1000 pcs/liter and 4000 pcs/liter during the population growth slowdown phase. The concentration of protozoan individuals was recorded before and after each experiment by measuring it with a guaranteed accuracy of 5% in a Goryaev chamber and, as measurements showed, remained virtually constant throughout the entire controlled period of the experiment.
The entamoeba gingivalis population is the only known model organism for studying the biophysical response of living organisms to external electromagnetic fields (EMFs) in the form of real-time changes in aquatic resistance. The advantage of the entamoeba gingivalis population in model experiments lies in the wide range of conditions under which it develops and thrives, including normal conditions. The entamoeba gingivalis population is characterized by rapid growth and metabolism, a distinct advantage over other simple microorganisms. Furthermore, an important indicator is the population’s resilience to environmental changes and the possible presence of other protozoans, which increases the reliability of the experiments. Another important characteristic is the similarity of metabolic processes in the entamoeba gingivalis population to those of higher organisms.
The temperature of the aquatic environment of the entamoeba gingivalis population was 22 °C in all experiments and was determined by the air temperature in the laboratory, which was automatically maintained using an air conditioner. Atmospheric pressure was 734 mmHg. During all experiments, deviations from the specified values did not exceed 1%.
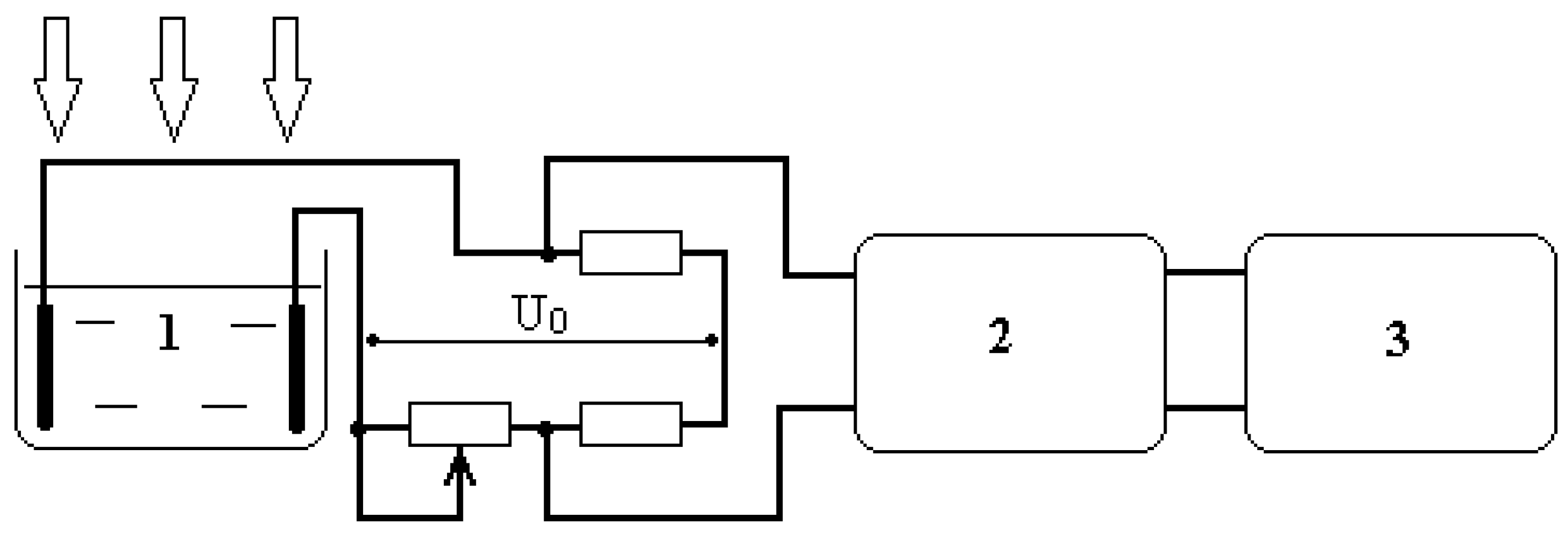
The experimental setup diagram is shown in
Figure 1 and corresponds to the setup described in [
17,
18]. Water cuvette 1 is used as a leg of the Wien-Robinson bridge circuit. The constant resistance values were 10 kOhm, while the variable resistance value was 15 kOhm. A change in the resistance of the aqueous medium in the cuvette resulted in an imbalance voltage in the bridge circuit, which was converted by amplifier 2 and recorded as a time diagram on graph plotter 3, which graphically recorded the voltage change during a single plotter sweep duration of 70 minutes. The time constant of the measuring circuit was 5 seconds, which, with a setup sensitivity of 5 10
-2%/cm, only allowed for the recording of processes of a fairly long duration. Voltage U
0 = 20 V.
The cuvette was irradiated with centimeter-wave (2.1 GHz) and millimeter-wave (42.25 GHz) non-thermal electromagnetic fields with an EMF power flux of 5 mW/cm2. The following radiation sources were used:
The experiments were accompanied by precise measurement of the water temperature in the cuvette with an accuracy of 0.01 °C, which revealed that in all cases, the change in the aqueous medium’s temperature in the cuvette did not exceed 0.02 °C during the entire plotter sweep time.
Studies conducted with water at pH values of 5.8 and 7.1 did not reveal any long-term effects from exposure to EMF. Preliminary experiments showed that at protozoan concentrations of up to 100 pcs/liter, no biophysical effects in the form of significant changes in the aqueous medium’s resistance were observed [
4,
5,
6,
17,
18].
Biophysical response of the entamoeba gingivalis population to external electromagnetic fields in aquatic environments at different pH values
The developed experimental methodology was hypothetically based on the possibility of recording the response of the “water—protozoan population” system in the form of changes in the parameters of the aquatic environment under external influence of EMF [
4,
5,
6,
17,
18]. Indeed, according to the concept of population homeostasis [
19], intrapopulation relationships of protozoans must ensure population unity through the population’s action on water as its habitat. Then, the effect of EMF on a protozoan population should lead to changes in the parameters of the aquatic environment due to the mutual influence of system “water—protozoan population”.
In this part, experimental studies are conducted on the biophysical response of a population of the entamoeba gingivalis under external influence of centimeter (2.1 GHz) and millimeter (42.25 GHz) non-thermal electromagnetic radiation with an EMF power flux of 5 mW/cm2. The exposure duration in all experiments was 90 seconds. The pH valuesof the aquatic habitat of entamoeba gingivalis populations were 5.8 and 7.1.
For each experiment, a series of four measurements was conducted. Average aquatic resistance values and their standard deviations were calculated using direct measurement data. Excel 2024 built-in capabilities were used to calculate average resistance curves.
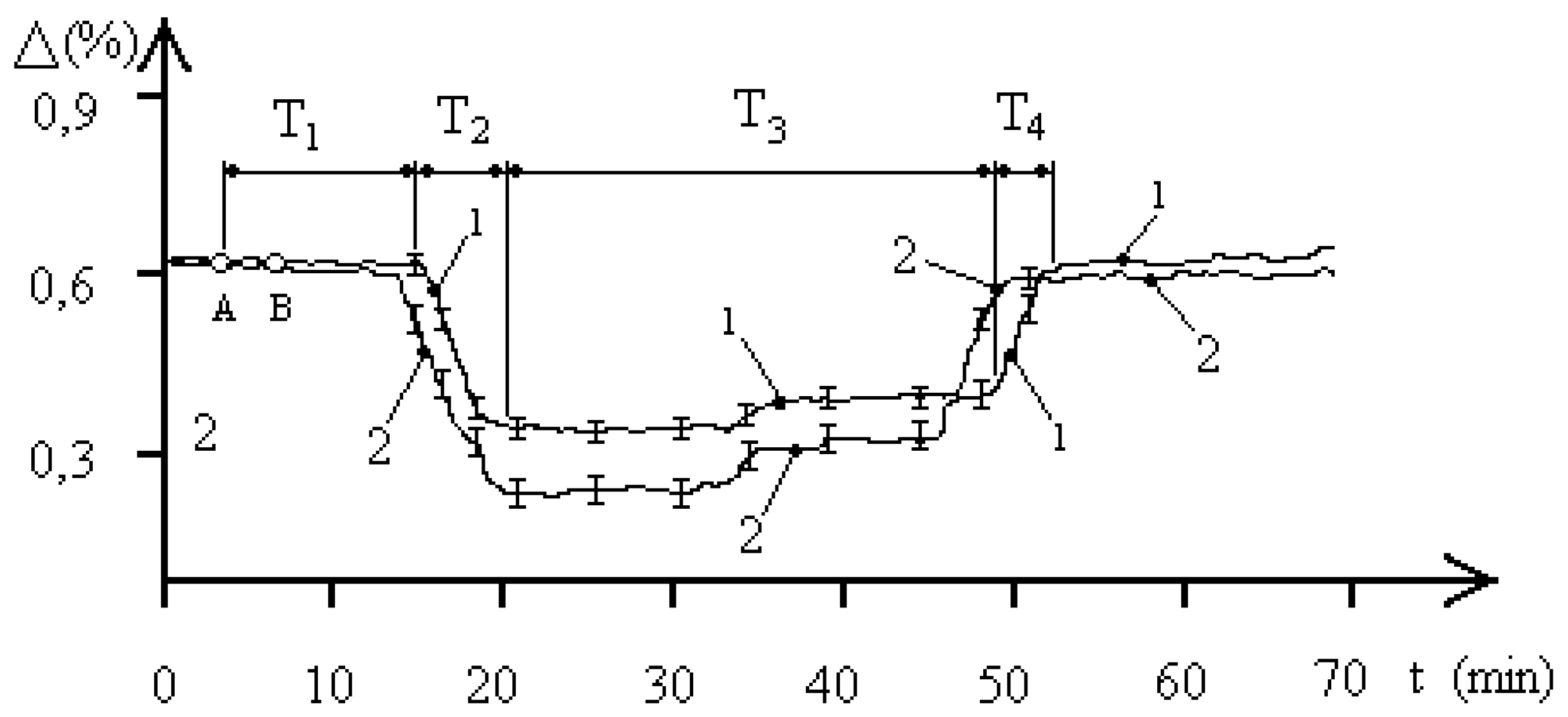
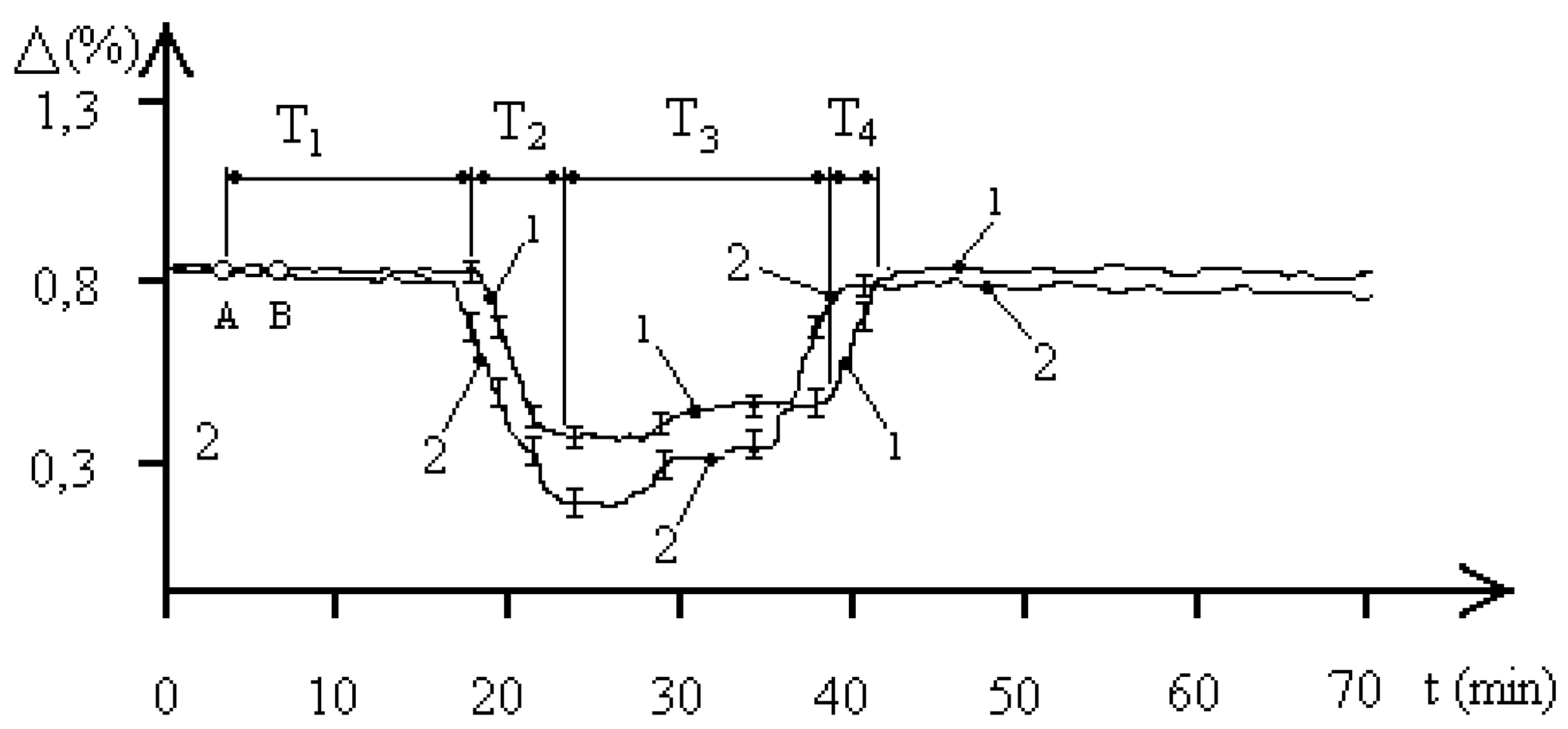
Figure 2 shows the average biophysical response curves for a population of the
entamoeba gingivalis exposed to 2,1 GHz electromagnetic radiation at a concentration of 1000 pcs/liter. The EMF exposure occurred between points A and B. The exposure time in all experiments was 90 seconds. The biophysical response curves were obtained for pH values of 5.8 and 7.1 in the aquatic environment of the
entamoeba gingivalis population. Curve 1 corresponds to a pH of 5.8, and curve 2 to a pH of 7.1. For simplicity, the characteristic biophysical response times T
1–T
2 of the “water—
entamoeba gingivalis population” system are shown for curve 1. The figure shows that the pH of the aquatic environment significantly influences the dynamics of processes occurring in the studied “water–
entamoeba gingivalis population” system. An increase in pH leads to changes in both the characteristic biophysical response times of the population and the maximum changes in aquatic resistance.
Let’s consider
Figure 2, which demonstrates changes in aquatic resistance of a population exposed to EMF, in more detail. The course of the curves indicates the biophysical nature of the response of the
entamoeba gingivalis population. Let’s look at curve 1. Initially, the studied system “water—
entamoeba gingivalis population” is characterized by a stable FS
1 with unchanged resistance for a time of T
1 = 12 minutes. After this, the aquatic resistance begins to decrease to the next stable FS
2. The time of decreasing aquatic resistance is T
2 = 6 minutes. The system remains in the second stable FS
2 of minimum aquatic resistance for T
3 = 28 minutes. Then, over a time of T
4 = 4 minutes, the aquatic resistance increases almost to the initial value, and the system transitions to the initial stable FS
1. It is worth noting that the stable FS
2 of the “water—
entamoeba gingivalis population” system with reduced aquatic resistance has a two-level nature. At a time of 33 minutes, as shown in
Figure 2, the value of the change in the resistance of the aquatic environment changes by 0.03%, which may indicate the characteristics of the group behavior of individuals of the
entamoeba gingivalis population at a minimum value of water resistance. The time from the beginning of phase T
3 to the point of transition to the second stable level of PS
2 is approximately equal to the time T
1, which indicates that the value of these times is determined by the duration of the exchange of information between individuals of the population, which is confirmed by the concept of population homeostasis of a protozoan population based on the integrity and stability of the population in the external environment [
18].
Let’s consider curve 2 in
Figure 2. Variations in the pH of the aquatic environment of an
entamoeba gingivalis population at a concentration of 1000 pcs/liter lead to a significant change in the qualitative picture of the biophysical response to EMF exposure with a frequency of 2.1 GHz. The experiments demonstrate four distinct phases of the biophysical response:
T1 is the phase of constant aquatic resistance;
T2 is the phase of decreasing aquatic resistance;
T3 is the phase of steadily reduced aquatic resistance;
T4 is the phase of increasing aquatic resistance to the initial level.
The duration of the biophysical response phases in each case ranges from a few minutes to tens of minutes. Interfunctional transitions in the “water—entamoeba gingivalis population” system (PS1–PS2–PS1) necessarily indicate the biological nature of the processes occurring in the studied system, as the duration of the processes recorded in the system exceeds the period of the external signal by ten orders of magnitude.
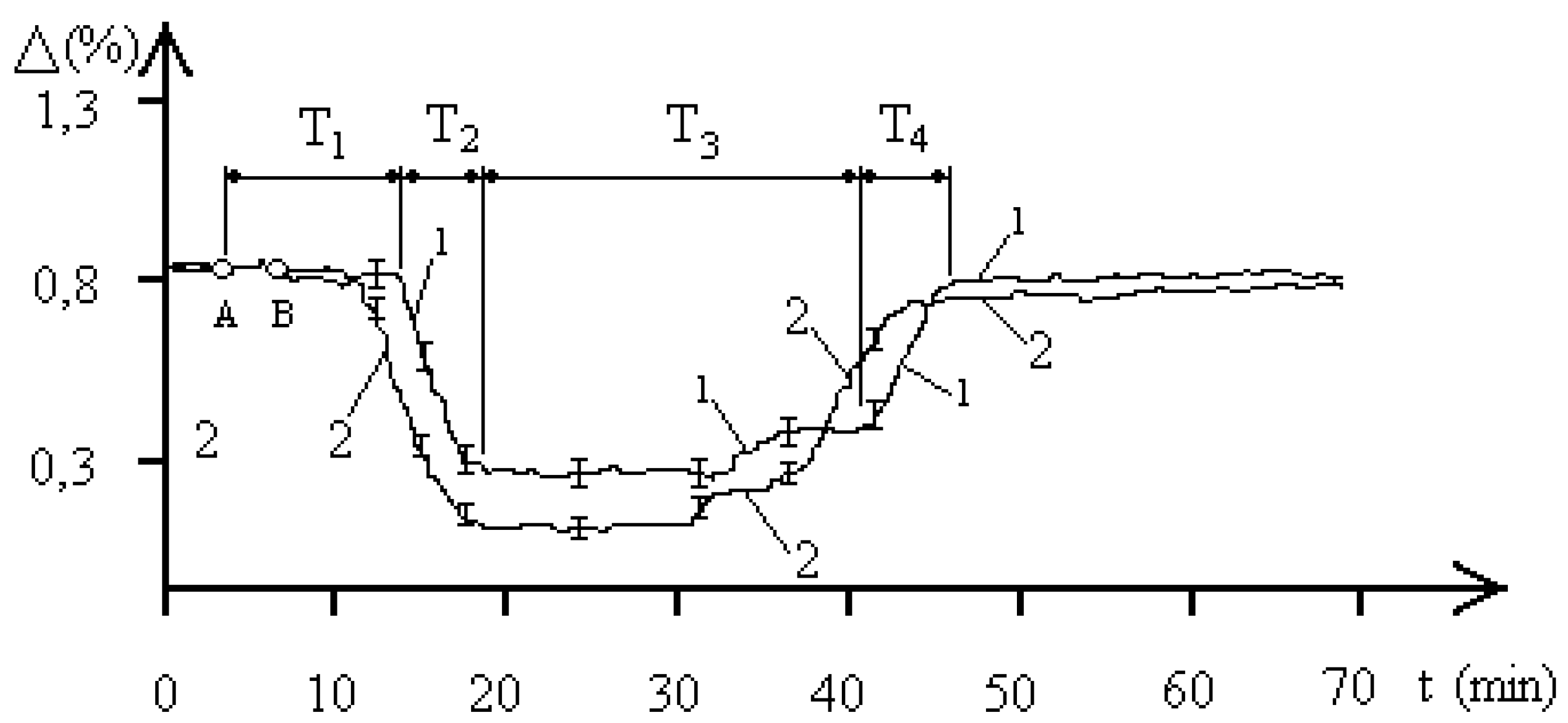
Let us refer to
Figure 3, reflecting the dynamics of the resistance of the aquatic environment of the
entamoeba gingivalis population with a concentration of 4000 pcs/liter under the influence of an EMF with a frequency of 2.1 GHz. The EMF exposure also occurred between points A and B and lasted 90 seconds. The biophysical response curves were also obtained for two pH values of the aquatic environment of the
entamoeba gingivalis population. Curve 1 corresponds to a pH of 5.8, curve 2 was obtained at a pH of 7.1. The temporal dynamics of changes in the resistance of the aquatic environment demonstrates clearly defined four phases of the biophysical response of the
entamoeba gingivalis population: T
1, T
2, T
3, T
4, which indicates the population transition PS
1–PS
2–PS
1. The nature of the processes in the “water—
entamoeba gingivalis population” system at a concentration of individuals of the population of 4000 pcs/liter. The results of exposure to 2.1 GHz EMF are qualitatively similar to those presented in
Figure 2 for external exposure to 2.1 GHz EMF on a population of
entamoeba gingivalis at a concentration of 1000 pcs/liter. Changes in the duration of the biophysical response phases affect both the overall duration of the radiobiological response of
entamoeba gingivalis and the duration of the determining time intervals of the process. The maximum value of the decrease in aquatic resistance also changed. In general, the biological response of the “water—
entamoeba gingivalis population” system at a population concentration of 4000 pcs/liter when exposed to 2.1 GHz EMF is, on average, more intense than at a population concentration of 1000 pcs/liter. The experiments also recorded a two-level nature of the stable FS
2 population of
entamoeba gingivalis with a minimum value of aquatic resistance replacing each other over a time interval of 32–33 minutes of the experimental time scale.
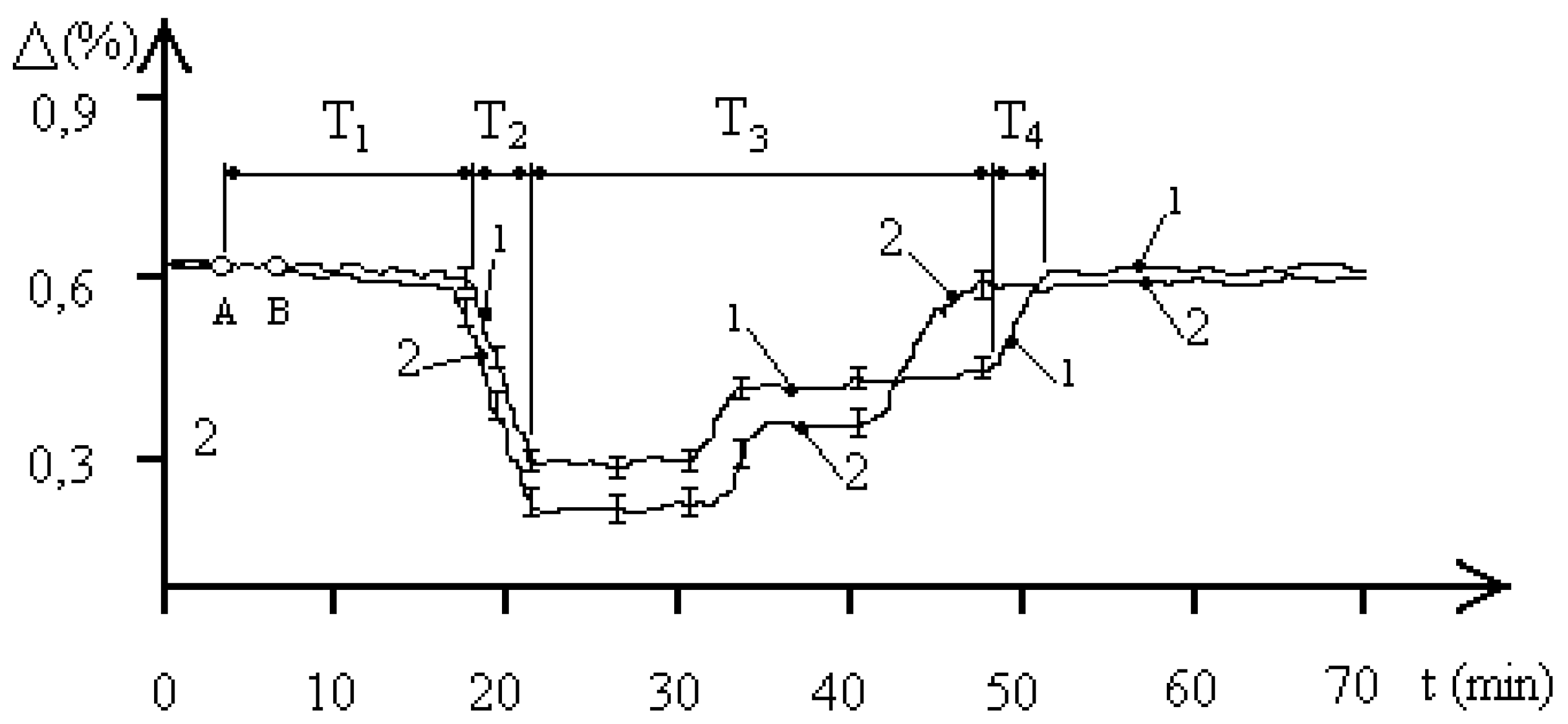
Let us consider
Figure 4. The figure shows the curves of the time dynamics of changes in the resistance of the aquatic environment in the “water—
entamoeba gingivalis population” system at a concentration of 1000 pcs/ liter under the influence of an EMF with a frequency of 42.25 GHz at two pH values of the aquatic environment. Curve 1 was obtained at pH = 5.8, curve 2 at pH = 7.1. The dynamics of the biophysical response process reveals 4 main phases: T
1, T
2, T
3, T
4, which qualitatively determine the time intervals of processes similar to those considered in
Figure 2 and
Figure 3. Such dynamics of processes indicates the transition FS
1–FS
2–FS
1, which is qualitatively similar to the processes recorded in experiments under the influence of EMF in the SM range. Of the main features of the identified biophysical response, the most interesting is the fact of a shorter time interval of stable FS
2 of the “water—population of
entamoeba gingivalis” system at the minimum value of the resistance of the aquatic environment and a more significant difference in the magnitude of stable levels in this state than in
Figure 2. It is also worth noting that an increase in the frequency of the external EMF significantly increases the first time interval T
1 of FS
2, indicating the time of information exchange between individuals of the population under MM external influence.
Let us turn to
Figure 5, which demonstrates the temporal dynamics of the biophysical response in the “water—
entamoeba gingivalis population” system at a population concentration of 4000 pcs/liter. The impact in this case is produced by an external EMF with a frequency of 42.25 GHz. The experiment was conducted for two pH values Δ(%) of the aquatic environment. Curve 1 was obtained at pH = 5.8, curve 2 at pH = 7.1. Although the system dynamics demonstrate the transition FS
1–FS
2–FS
1, the time interval of the change in the aquatic environment T
2 in this case is distinguished by its smallest value with practically the most significant information exchange time T
1. Moreover, the difference in the resistance of the aquatic environment FS
1 and FS
2 is not as significant as at a population concentration of 1000 pcs/liter.