Submitted:
25 November 2025
Posted:
26 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. PBMC Culture for mRNA-Sequencing
2.3. Cell Viability Testing
2.4. RNA-Seq and Analysis
2.5. Cytokine Activity Assays
2.6. DPPH Assay
2.7. Evaluation of Ferrous Iron Chelating Ability
3. Results
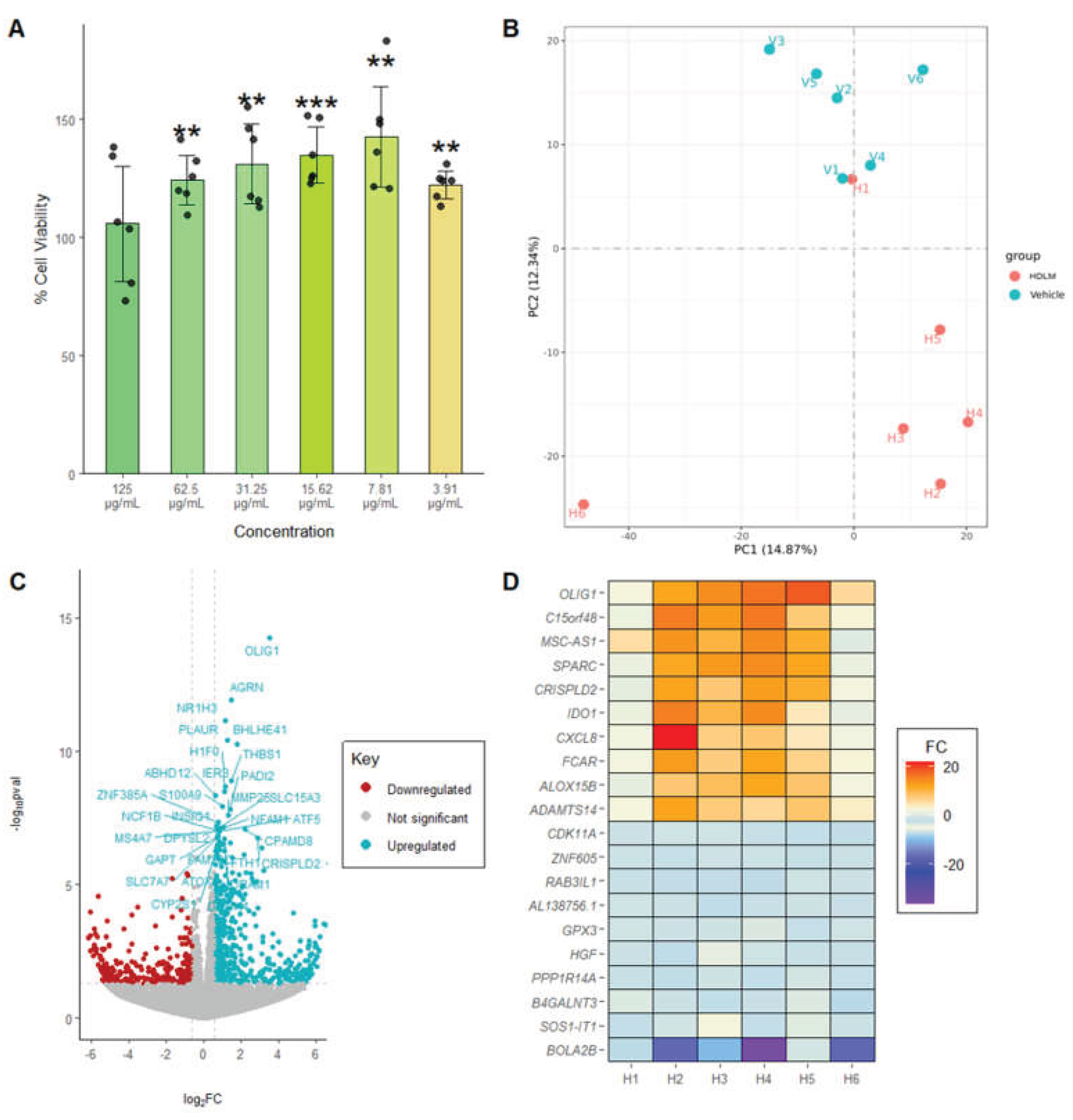
3.1. Cell Culture Optimization and Cell Viability Testing
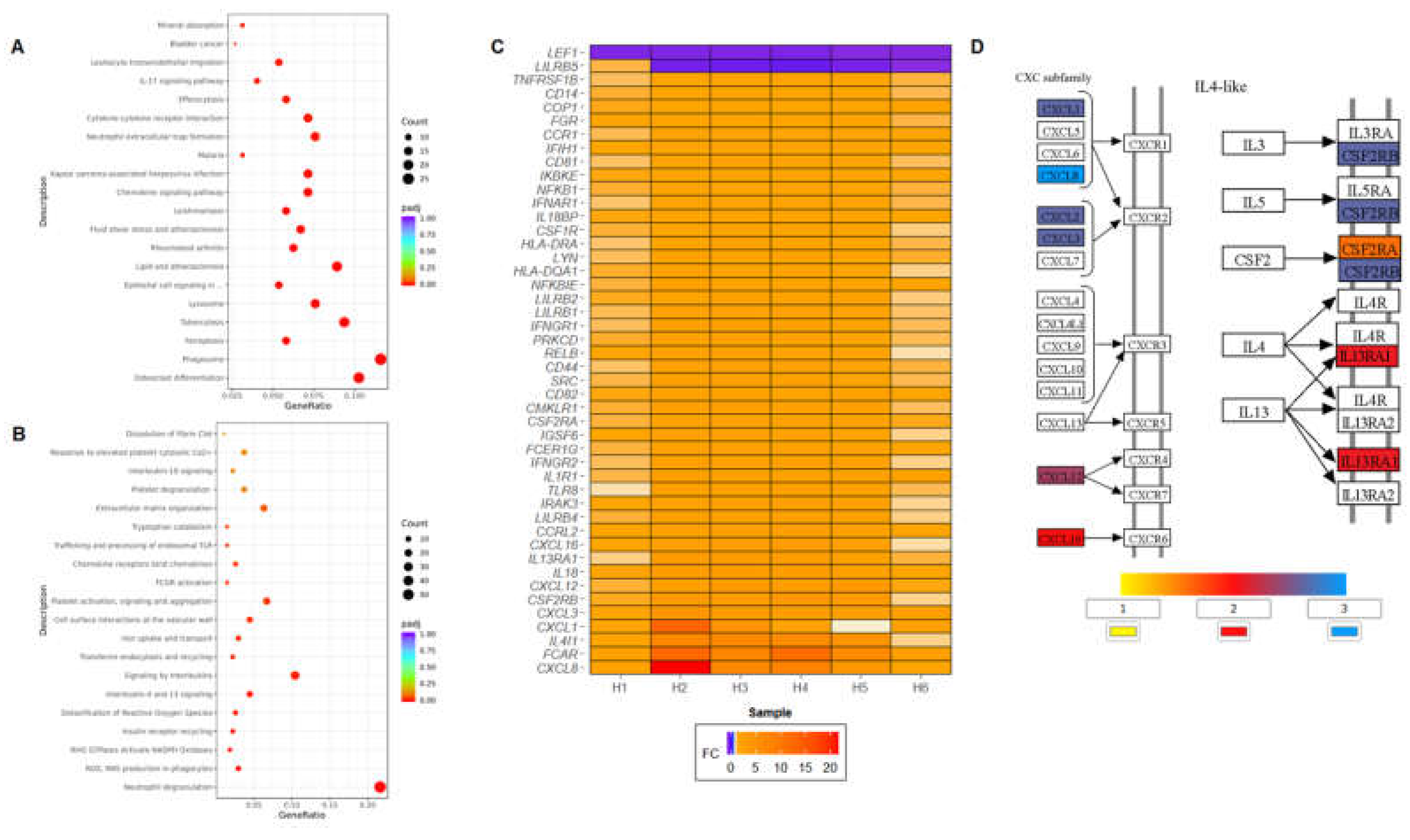
3.2. Transcriptomic Impact of HDLM on Human PBMCs
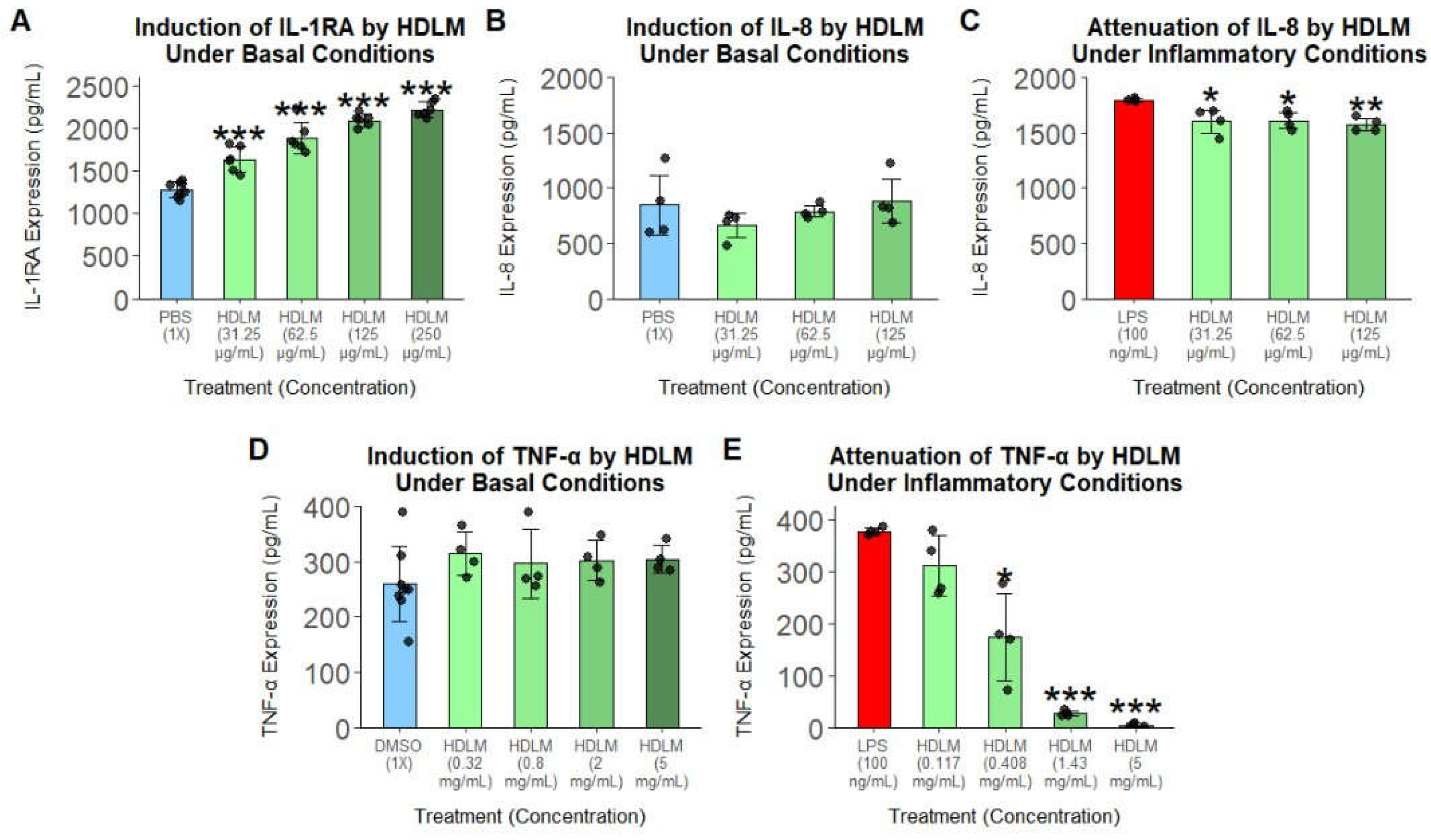
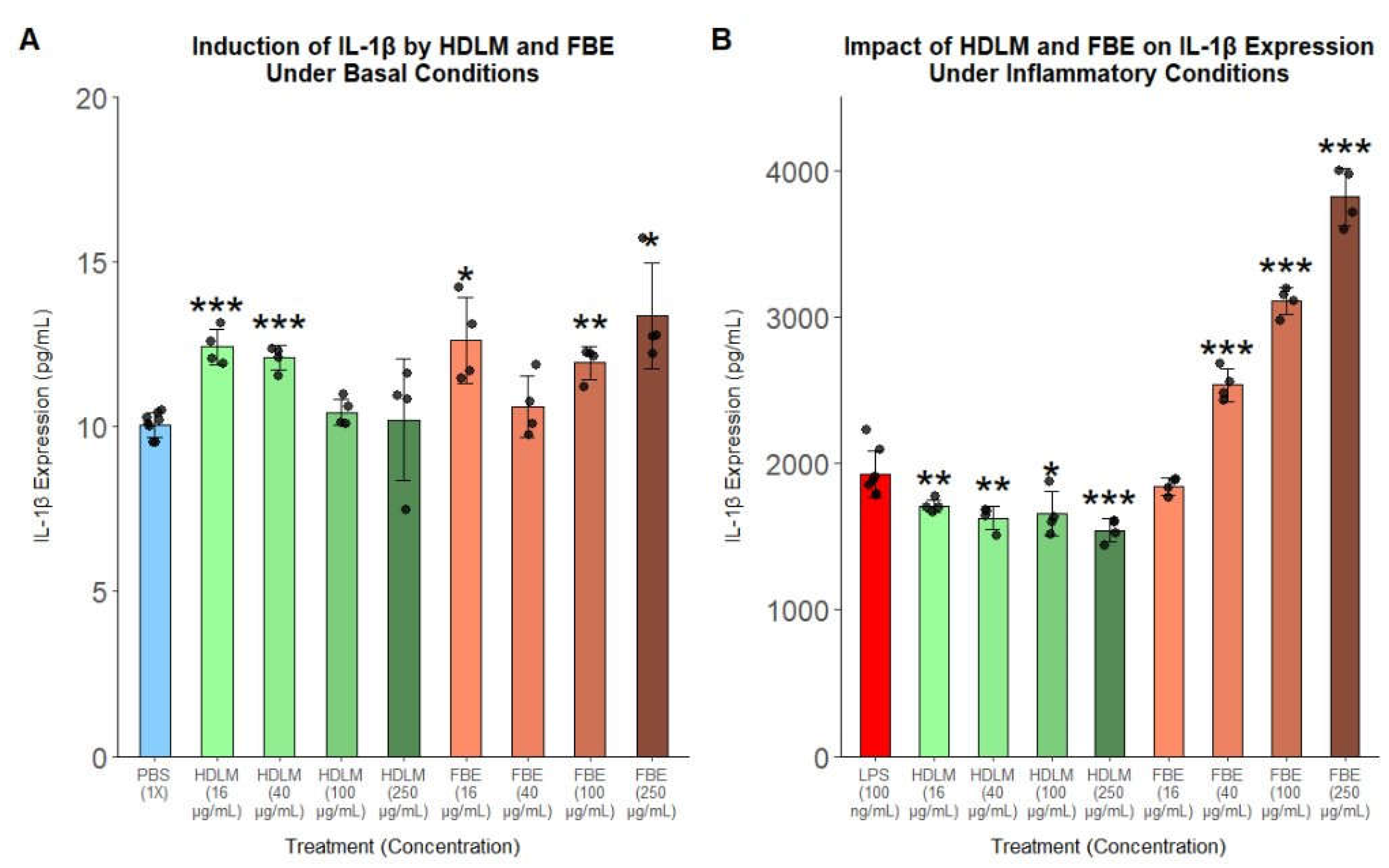
3.3. Cytokine Modulation Under Basal and Inflammatory Conditions
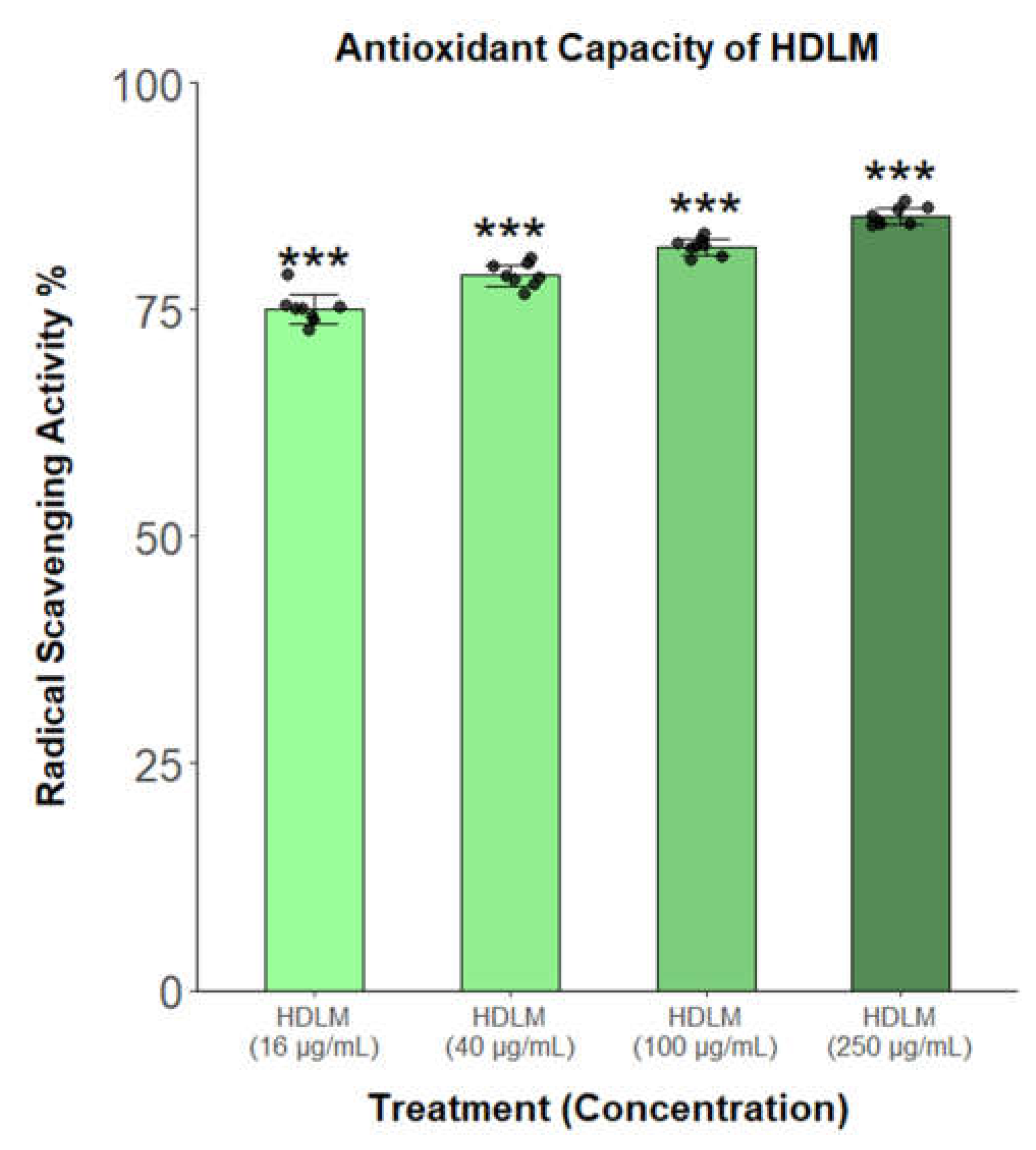
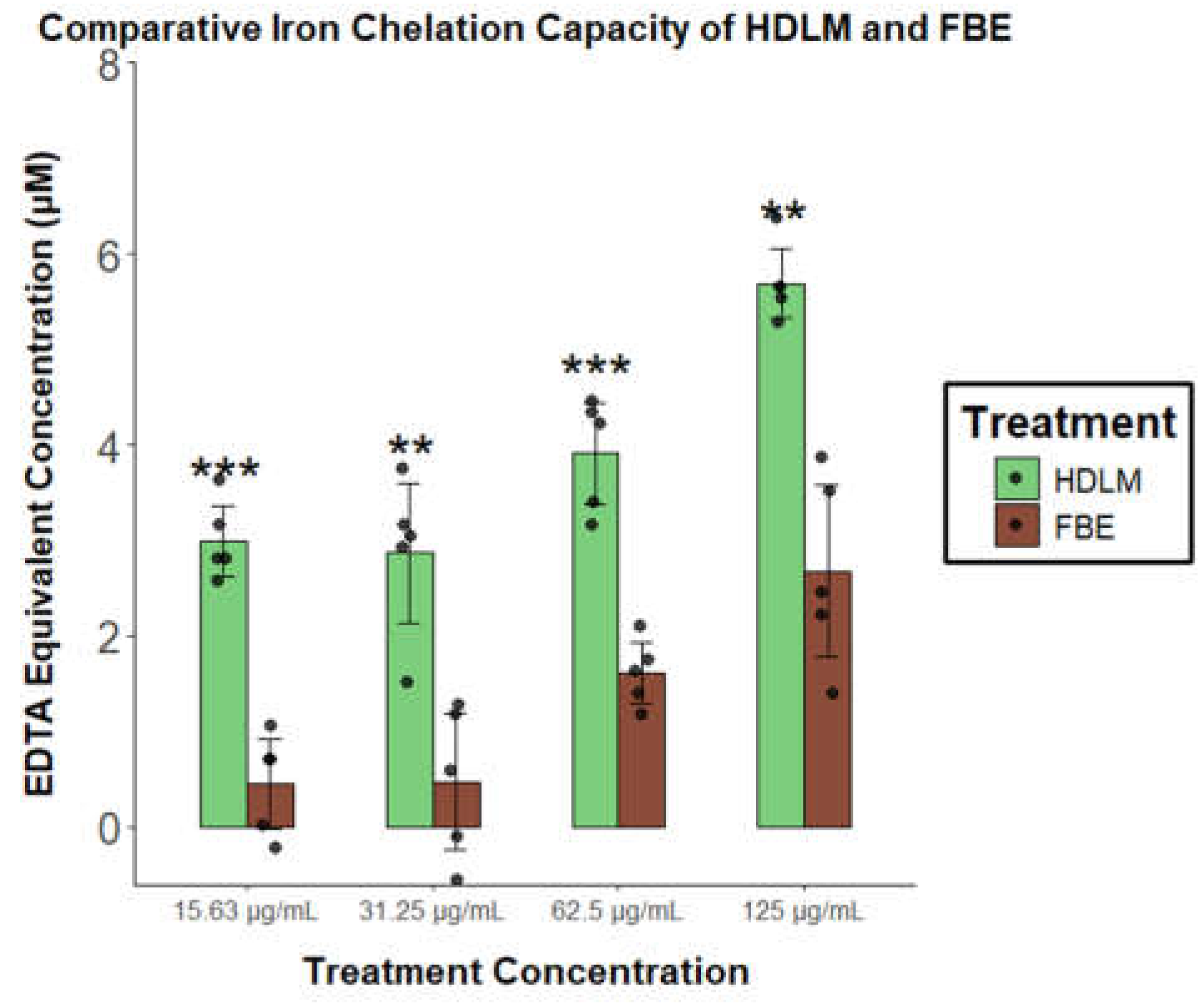
3.4. Antioxidant Capacity of HDLM and FBE
4. Discussion
4.1. HDLM-Elicited Activation and Pacification of Immune Responses
4.2. NF-κB Signaling and Adaptive Immune Responses
4.3. Inflammatory Response Triggered by FBE
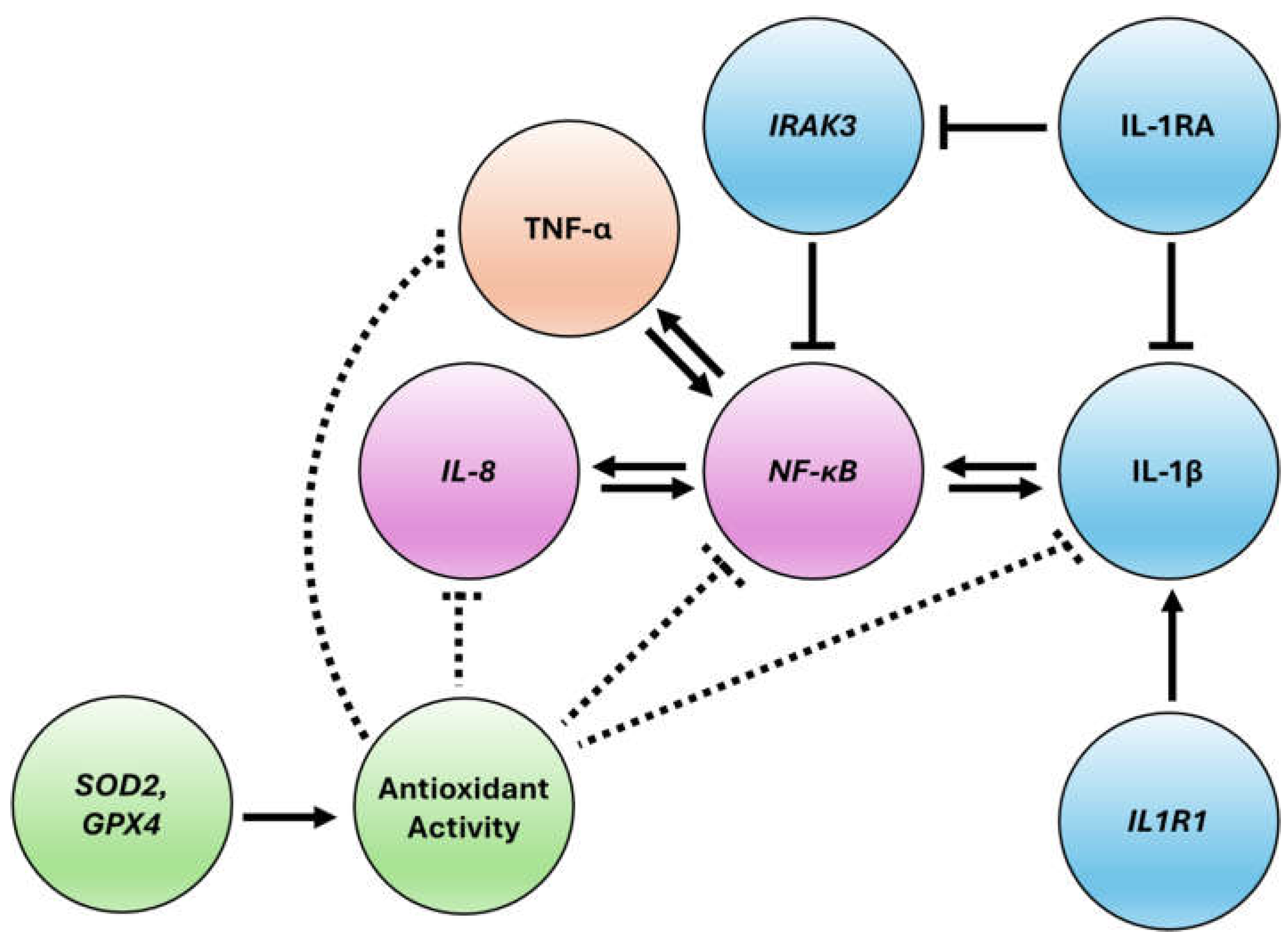
4.4. Stress Quenching Abilities of HDLM and FBE
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, I.-C.; Chang, H.-H.; Lin, C.-H.; Chen, W.-P.; Lu, T.-H.; Lee, L.-Y.; Chen, Y.-W.; Chen, Y.-P.; Chen, C.-C.; Lin, D.P.-C. Prevention of Early Alzheimer’s Disease by Erinacine A-Enriched Hericium erinaceus Mycelia Pilot Double-Blind Placebo-Controlled Study. Front Aging Neurosci 2020, 12, 155. [CrossRef]
- Mori, K.; Obara, Y.; Hirota, M.; Azumi, Y.; Kinugasa, S.; Inatomi, S.; Nakahata, N. Nerve Growth Factor-Inducing Activity of Hericium erinaceus in 1321N1 Human Astrocytoma Cells. Biol Pharm Bull 2008, 31, 1727–1732. [CrossRef]
- Ornish, D.; Madison, C.; Kivipelto, M.; Kemp, C.; McCulloch, C.E.; Galasko, D.; Artz, J.; Rentz, D.; Lin, J.; Norman, K.; et al. Effects of Intensive Lifestyle Changes on the Progression of Mild Cognitive Impairment or Early Dementia Due to Alzheimer’s Disease: A Randomized, Controlled Clinical Trial. Alz Res Therapy 2024, 16, 122. [CrossRef]
- Vigna, L.; Morelli, F.; Agnelli, G.M.; Napolitano, F.; Ratto, D.; Occhinegro, A.; Di Iorio, C.; Savino, E.; Girometta, C.; Brandalise, F.; et al. Hericium erinaceus Improves Mood and Sleep Disorders in Patients Affected by Overweight or Obesity: Could Circulating pro-BDNF and BDNF Be Potential Biomarkers? Evid Based Complement Alternat Med 2019, 2019, 7861297. [CrossRef]
- Chang, H.C.; Yang, H.-L.; Pan, J.-H.; Korivi, M.; Pan, J.-Y.; Hsieh, M.-C.; Chao, P.-M.; Huang, P.-J.; Tsai, C.-T.; Hseu, Y.-C. Hericium erinaceus Inhibits TNF-α-Induced Angiogenesis and ROS Generation through Suppression of MMP-9/NF-κB Signaling and Activation of Nrf2-Mediated Antioxidant Genes in Human EA.Hy926 Endothelial Cells. Oxidative Medicine and Cellular Longevity 2016, 2016, 8257238. [CrossRef]
- Contato, A.G.; Conte-Junior, C.A. Lion’s Mane Mushroom (Hericium erinaceus): A Neuroprotective Fungus with Antioxidant, Anti-Inflammatory, and Antimicrobial Potential—a Narrative Review. Nutrients 2025, 17, 1307. [CrossRef]
- Gao, Y.; Zheng, W.; Wang, M.; Xiao, X.; Gao, M.; Gao, Q.; Xu, D. Molecular Properties, Structure, and Antioxidant Activities of the Oligosaccharide Hep-2 Isolated from Cultured Mycelium of Hericium erinaceus. Journal of Food Biochemistry 2019, 43, e12985. [CrossRef]
- Kushairi, N.; Phan, C.W.; Sabaratnam, V.; David, P.; Naidu, M. Lion’s Mane Mushroom, Hericium erinaceus (Bull.: Fr.) Pers. Suppresses H2O2-Induced Oxidative Damage and LPS-Induced Inflammation in HT22 Hippocampal Neurons and BV2 Microglia. Antioxidants (Basel) 2019, 8, 261. [CrossRef]
- Wang, M.; Zhang, Y.; Xiao, X.; Xu, D.; Gao, Y.; Gao, Q. A Polysaccharide Isolated from Mycelia of the Lion’s Mane Medicinal Mushroom Hericium erinaceus (Agaricomycetes) Induced Apoptosis in Precancerous Human Gastric Cells. IJM 2017, 19. [CrossRef]
- Ren, Y.; Geng, Y.; Du, Y.; Li, W.; Lu, Z.-M.; Xu, H.-Y.; Xu, G.-H.; Shi, J.-S.; Xu, Z.-H. Polysaccharide of Hericium erinaceus Attenuates Colitis in C57BL/6 Mice via Regulation of Oxidative Stress, Inflammation-Related Signaling Pathways and Modulating the Composition of the Gut Microbiota. The Journal of Nutritional Biochemistry 2018, 57, 67–76. [CrossRef]
- Xie, X.-Q.; Geng, Y.; Guan, Q.; Ren, Y.; Guo, L.; Lv, Q.; Lu, Z.-M.; Shi, J.-S.; Xu, Z.-H. Influence of Short-Term Consumption of Hericium erinaceus on Serum Biochemical Markers and the Changes of the Gut Microbiota: A Pilot Study. Nutrients 2021, 13, 1008. [CrossRef]
- Friedman, M. Chemistry, Nutrition, and Health-Promoting Properties of Hericium erinaceus (Lion’s Mane) Mushroom Fruiting Bodies and Mycelia and Their Bioactive Compounds. J. Agric. Food Chem. 2015, 63, 7108–7123. [CrossRef]
- Kawagishi, H.; Shimada, A.; Shirai, R.; Okamoto, K.; Ojima, F.; Sakamoto, H.; Ishiguro, Y.; Furukawa, S. Erinacines A, B and C, Strong Stimulators of Nerve Growth Factor (NGF)-Synthesis, from the Mycelia of Hericium erinaceum. Tetrahedron Letters 1994, 35, 1569–1572. [CrossRef]
- Ma, B.-J.; Shen, J.-W.; Yu, H.-Y.; Ruan, Y.; Wu, T.-T.; Zhao, X. Hericenones and Erinacines: Stimulators of Nerve Growth Factor (NGF) Biosynthesis in Hericium erinaceus. Mycology 2010, 1, 92–98. [CrossRef]
- Doar, E.; Meyer, K.W.; Bair, Z.J.; Nally, R.; McNalley, S.; Davis, R.; Beathard, C. Influences of Substrate and Tissue Type on Erinacine Production and Biosynthetic Gene Expression in Hericium erinaceus. Fungal Biol Biotechnol 2025, 12, 4. [CrossRef]
- Kawagishi, H.; Zhuang, C. Compounds for Dementia from Hericium erinaceum. Drugs of the Future 2008, 33, 0149. [CrossRef]
- Shimbo, M.; Kawagishi, H.; Yokogoshi, H. Erinacine A Increases Catecholamine and Nerve Growth Factor Content in the Central Nervous System of Rats. Nutrition Research 2005, 25, 617–623. [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biology 2014, 15, 550. [CrossRef]
- Dinis, T.C.P.; Madeira, V.M.C.; Almeida, L.M. Action of Phenolic Derivatives (Acetaminophen, Salicylate, and 5-Aminosalicylate) as Inhibitors of Membrane Lipid Peroxidation and as Peroxyl Radical Scavengers. Archives of Biochemistry and Biophysics 1994, 315, 161–169. [CrossRef]
- Yusof, N.Bt.Md.; Hasan, M.H.; Armayni, U.A.; Ahmad, M.S.B.; Wahab, I.A. The Ferrous Iron Chelating Assay of Extracts. The Open Conference Proceedings Journal 2013, 4, 1555. [CrossRef]
- Cambier, S.; Gouwy, M.; Proost, P. The Chemokines CXCL8 and CXCL12: Molecular and Functional Properties, Role in Disease and Efforts towards Pharmacological Intervention. Cell Mol Immunol 2023, 20, 217–251. [CrossRef]
- Fialkow, L.; Wang, Y.; Downey, G.P. Reactive Oxygen and Nitrogen Species as Signaling Molecules Regulating Neutrophil Function. Free Radical Biology and Medicine 2007, 42, 153–164. [CrossRef]
- Hoffmann, E.; Dittrich-Breiholz, O.; Holtmann, H.; Kracht, M. Multiple Control of Interleukin-8 Gene Expression. J Leukoc Biol 2002, 72, 847–855.
- Vacchini, A.; Mortier, A.; Proost, P.; Locati, M.; Metzemaekers, M.; Borroni, E.M. Differential Effects of Posttranslational Modifications of CXCL8/Interleukin-8 on CXCR1 and CXCR2 Internalization and Signaling Properties. Int J Mol Sci 2018, 19, 3768. [CrossRef]
- Freen-van Heeren, J.J. Post-Transcriptional Control of T-Cell Cytokine Production: Implications for Cancer Therapy. Immunology 2021, 164, 57–72. [CrossRef]
- Martinez, F.O.; Sironi, M.; Vecchi, A.; Colotta, F.; Mantovani, A.; Locati, M. IL-8 Induces a Specific Transcriptional Profile in Human Neutrophils: Synergism with LPS for IL-1 Production. European Journal of Immunology 2004, 34, 2286–2292. [CrossRef]
- Luís, J.P.; Simões, C.J.V.; Brito, R.M.M. The Therapeutic Prospects of Targeting IL-1R1 for the Modulation of Neuroinflammation in Central Nervous System Disorders. International Journal of Molecular Sciences 2022, 23, 1731. [CrossRef]
- Arend, W.P. The Balance between IL-1 and IL-1Ra in Disease. Cytokine & Growth Factor Reviews 2002, 13, 323–340. [CrossRef]
- Kaplanski, G.; Farnarier, C.; Kaplanski, S.; Porat, R.; Shapiro, L.; Bongrand, P.; Dinarello, C.A. Interleukin-1 Induces Interleukin-8 Secretion from Endothelial Cells by a Juxtacrine Mechanism. Blood 1994, 84, 4242–4248. [CrossRef]
- Freihat, L.A.; Wheeler, J.I.; Wong, A.; Turek, I.; Manallack, D.T.; Irving, H.R. IRAK3 Modulates Downstream Innate Immune Signalling through Its Guanylate Cyclase Activity. Sci Rep 2019, 9, 15468. [CrossRef]
- Pereira, M.; Gazzinelli, R.T. Regulation of Innate Immune Signaling by IRAK Proteins. Front. Immunol. 2023, 14. [CrossRef]
- Dambuza, I.M.; Brown, G.D. C-Type Lectins in Immunity: Recent Developments. Current Opinion in Immunology 2015, 32, 21–27. [CrossRef]
- Geijtenbeek, T.B.H.; Gringhuis, S.I. C-Type Lectin Receptors in the Control of T Helper Cell Differentiation. Nat Rev Immunol 2016, 16, 433–448. [CrossRef]
- Yasuda, K.; Takeuchi, Y.; Hirota, K. The Pathogenicity of Th17 Cells in Autoimmune Diseases. Semin Immunopathol 2019, 41, 283–297. [CrossRef]
- Chen, G.-Y.; Tang, J.; Zheng, P.; Liu, Y. CD24 and Siglec-10 Selectively Repress Tissue Damage-Induced Immune Responses. Science 2009, 323, 1722–1725. [CrossRef]
- Wevers, B.A. C-Type Lectin Signaling in Dendritic Cells: Molecular Control of Antifungal Inflammation, 2014.
- Wevers, B.A.; Kaptein, T.M.; Zijlstra-Willems, E.M.; Theelen, B.; Boekhout, T.; Geijtenbeek, T.B.H.; Gringhuis, S.I. Fungal Engagement of the C-Type Lectin Mincle Suppresses Dectin-1-Induced Antifungal Immunity. Cell Host Microbe 2014, 15, 494–505. [CrossRef]
- Romagnani, S. IL4I1: Key Immunoregulator at a Crossroads of Divergent T-Cell Functions. Eur J Immunol 2016, 46, 2302–2305. [CrossRef]
- Zhang, Y.; Zhang, Y.; Gu, W.; Sun, B. Th1/Th2 Cell Differentiation and Molecular Signals. In T Helper Cell Differentiation and Their Function; Sun, B., Ed.; Springer Netherlands: Dordrecht, 2014; pp. 15–44 ISBN 978-94-017-9487-9.
- Downton, P.; Bagnall, J.S.; England, H.; Spiller, D.G.; Humphreys, N.E.; Jackson, D.A.; Paszek, P.; White, M.R.H.; Adamson, A.D. Overexpression of IκB⍺ Modulates NF-κB Activation of Inflammatory Target Gene Expression. Front. Mol. Biosci. 2023, 10. [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-κB Family of Transcription Factors and Its Regulation. Cold Spring Harb Perspect Biol 2009, 1, a000034. [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB Signaling in Inflammation. Sig Transduct Target Ther 2017, 2, 17023. [CrossRef]
- Manna, S.K.; Ramesh, G.T. Interleukin-8 Induces Nuclear Transcription Factor-κB through a TRAF6-Dependent Pathway. Journal of Biological Chemistry 2005, 280, 7010–7021. [CrossRef]
- Whitley, S.K.; Balasubramani, A.; Zindl, C.L.; Sen, R.; Shibata, Y.; Crawford, G.E.; Weathington, N.M.; Hatton, R.D.; Weaver, C.T. IL-1R Signaling Promotes STAT3 and NF-κB Factor Recruitment to Distal Cis-Regulatory Elements That Regulate Il17a/f Transcription. Journal of Biological Chemistry 2018, 293, 15790–15800. [CrossRef]
- Verstrepen, L.; Bekaert, T.; Chau, T.-L.; Tavernier, J.; Chariot, A.; Beyaert, R. TLR-4, IL-1R and TNF-R Signaling to NF-κB: Variations on a Common Theme. Cell. Mol. Life Sci. 2008, 65, 2964–2978. [CrossRef]
- Sun, S.-C. The Non-Canonical NF-κB Pathway in Immunity and Inflammation. Nat Rev Immunol 2017, 17, 545–558. [CrossRef]
- Li, C.; Deng, X.; Xie, X.; Liu, Y.; Friedmann Angeli, J.P.; Lai, L. Activation of Glutathione Peroxidase 4 as a Novel Anti-Inflammatory Strategy. Front. Pharmacol. 2018, 9. [CrossRef]
- Jin, J.; Hu, H.; Li, H.S.; Yu, J.; Xiao, Y.; Brittain, G.C.; Zou, Q.; Cheng, X.; Mallette, F.A.; Watowich, S.S.; et al. Noncanonical NF-κB Pathway Controls the Production of Type I Interferons in Antiviral Innate Immunity. Immunity 2014, 40, 342–354. [CrossRef]
- Sun, S.-C. The Noncanonical NF-κB Pathway. Immunological Reviews 2012, 246, 125–140. [CrossRef]
- De Silva, N.S.; Silva, K.; Anderson, M.M.; Bhagat, G.; Klein, U. Impairment of Mature B Cell Maintenance upon Combined Deletion of the Alternative NF-κB Transcription Factors RELB and NF-κB2 in B Cells. The Journal of Immunology 2016, 196, 2591–2601. [CrossRef]
- Cervantes, J.L.; Weinerman, B.; Basole, C.; Salazar, J.C. TLR8: The Forgotten Relative Revindicated. Cell Mol Immunol 2012, 9, 434–438. [CrossRef]
- Pestka, S.; Kotenko, S.V.; Muthukumaran, G.; Izotova, L.S.; Cook, J.R.; Garotta, G. The Interferon Gamma (IFN-γ) Receptor: A Paradigm for the Multichain Cytokine Receptor. Cytokine & Growth Factor Reviews 1997, 8, 189–206. [CrossRef]
- Dinarello, C.A.; Novick, D.; Puren, A.J.; Fantuzzi, G.; Shapiro, L.; Mühl, H.; Yoon, D.-Y.; Reznikov, L.L.; Kim, S.-H.; Rubinstein, M. Overview of Interleukin-18: More than an Interferon-γ Inducing Factor. J Leukoc Biol. 1998, 63, 658–664. [CrossRef]
- Nakamura, K.; Okamura, H.; Wada, M.; Nagata, K.; Tamura, T. Endotoxin-Induced Serum Factor That Stimulates Gamma Interferon Production. Infection and Immunity 1989, 57, 590–595. [CrossRef]
- Yoshimoto, T.; Okamura, H.; Tagawa, Y.-I.; Iwakura, Y.; Nakanishi, K. Interleukin 18 Together with Interleukin 12 Inhibits IgE Production by Induction of Interferon-γ Production from Activated B Cells. Proceedings of the National Academy of Sciences 1997, 94, 3948–3953. [CrossRef]
- Bian, F.; Yan, D.; Wu, X.; Yang, C. A Biological Perspective of TLR8 Signaling in Host Defense and Inflammation. Infectious Microbes & Diseases 2023, 5, 44. [CrossRef]
- Braga, A.C.S.; Carneiro, B.M.; Batista, M.N.; Akinaga, M.M.; Bittar, C.; Rahal, P. Heat Shock Proteins HSPB8 and DNAJC5B Have HCV Antiviral Activity. PLOS ONE 2017, 12, e0188467. [CrossRef]
- Dinarello, C.A.; Novick, D.; Kim, S.; Kaplanski, G. Interleukin-18 and IL-18 Binding Protein. Front Immunol 2013, 4, 289. [CrossRef]
- Elder, M.J.; Webster, S.J.; Chee, R.; Williams, D.L.; Hill Gaston, J.S.; Goodall, J.C. β-Glucan Size Controls Dectin-1-Mediated Immune Responses in Human Dendritic Cells by Regulating IL-1β Production. Front. Immunol. 2017, 8. [CrossRef]
- Murphy, E.J.; Rezoagli, E.; Major, I.; Rowan, N.J.; Laffey, J.G. β-Glucan Metabolic and Immunomodulatory Properties and Potential for Clinical Application. Journal of Fungi 2020, 6, 356. [CrossRef]
- Vannucci, L.; Krizan, J.; Sima, P.; Stakheev, D.; Caja, F.; Rajsiglova, L.; Horak, V.; Saieh, M. Immunostimulatory Properties and Antitumor Activities of Glucans (Review). Int J Oncol 2013, 43, 357–364. [CrossRef]
- Duan, L.; Rao, X.; Sigdel, K.R. Regulation of Inflammation in Autoimmune Disease. J Immunol Res 2019, 2019. [CrossRef]
- Singh, R.P.; Hasan, S.; Sharma, S.; Nagra, S.; Yamaguchi, D.T.; Wong, D.T.W.; Hahn, B.H.; Hossain, A. Th17 Cells in Inflammation and Autoimmunity. Autoimmunity Reviews 2014, 13, 1174–1181. [CrossRef]
- Alschuler, L.; Weil, A.; Horwitz, R.; Stamets, P.; Chiasson, A.M.; Crocker, R.; Maizes, V. Integrative Considerations during the COVID-19 Pandemic. EXPLORE 2020, 16, 354–356. [CrossRef]
- Potere, N.; Buono, M.G.D.; Caricchio, R.; Cremer, P.C.; Vecchié, A.; Porreca, E.; Gasperina, D.D.; Dentali, F.; Abbate, A.; Bonaventura, A. Interleukin-1 and the NLRP3 Inflammasome in COVID-19: Pathogenetic and Therapeutic Implications. eBioMedicine 2022, 85. [CrossRef]
- Ratajczak, M.Z.; Bujko, K.; Ciechanowicz, A.; Sielatycka, K.; Cymer, M.; Marlicz, W.; Kucia, M. SARS-CoV-2 Entry Receptor ACE2 Is Expressed on Very Small CD45− Precursors of Hematopoietic and Endothelial Cells and in Response to Virus Spike Protein Activates the Nlrp3 Inflammasome. Stem Cell Rev Rep 2021, 17, 266–277. [CrossRef]
- Kozłowska, E.; Agier, J.; Różalska, S.; Jurczak, M.; Góralczyk-Bińkowska, A.; Żelechowska, P. Fungal β-Glucans Shape Innate Immune Responses in Human Peripheral Blood Mononuclear Cells (PBMCs): An in Vitro Study on PRR Regulation, Cytokine Expression, and Oxidative Balance. Int J Mol Sci 2025, 26, 6458. [CrossRef]
- Mata-Martínez, P.; Bergón-Gutiérrez, M.; del Fresno, C. Dectin-1 Signaling Update: New Perspectives for Trained Immunity. Front. Immunol. 2022, 13. [CrossRef]
- Vuscan, P.; Kischkel, B.; Joosten, L.A.B.; Netea, M.G. Trained Immunity: General and Emerging Concepts. Immunological Reviews 2024, 323, 164–185. [CrossRef]
- Vetvicka, V.; Vannucci, L.; Sima, P. β-Glucan as a New Tool in Vaccine Development. Scandinavian Journal of Immunology 2020, 91, e12833. [CrossRef]
- Eliza, W.L.Y.; Fai, C.K.; Chung, L.P. Efficacy of Yun Zhi (Coriolus versicolor) on Survival in Cancer Patients: Systematic Review and Meta-Analysis. Recent Pat Inflamm Allergy Drug Discov 2012, 6, 78–87. [CrossRef]
- Quayle, K.; Coy, C.; Standish, L.; Lu, H. The TLR2 Agonist in Polysaccharide-K Is a Structurally Distinct Lipid Which Acts Synergistically with the Protein-Bound β-Glucan. J Nat Med 2015, 69, 198–208. [CrossRef]
- Benson, K.F.; Stamets, P.; Davis, R.; Nally, R.; Taylor, A.; Slater, S.; Jensen, G.S. The Mycelium of the Trametes Versicolor (Turkey Tail) Mushroom and Its Fermented Substrate Each Show Potent and Complementary Immune Activating Properties in Vitro. BMC Complementary and Alternative Medicine 2019, 19, 342. [CrossRef]
- Li, J.; Cao, F.; Yin, H.; Huang, Z.; Lin, Z.; Mao, N.; Sun, B.; Wang, G. Ferroptosis: Past, Present and Future. Cell Death Dis 2020, 11, 1–13. [CrossRef]
- Xie, Y.; Hou, W.; Song, X.; Yu, Y.; Huang, J.; Sun, X.; Kang, R.; Tang, D. Ferroptosis: Process and Function. Cell Death Differ 2016, 23, 369–379. [CrossRef]
- Zou, X.; Ratti, B.A.; O’Brien, J.G.; Lautenschlager, S.O.; Gius, D.R.; Bonini, M.G.; Zhu, Y. Manganese Superoxide Dismutase (SOD2): Is There a Center in the Universe of Mitochondrial Redox Signaling? J Bioenerg Biomembr 2017, 49, 325–333. [CrossRef]
- Takakura, Y.; Machida, M.; Terada, N.; Katsumi, Y.; Kawamura, S.; Horie, K.; Miyauchi, M.; Ishikawa, T.; Akiyama, N.; Seki, T.; et al. Mitochondrial Protein C15ORF48 Is a Stress-Independent Inducer of Autophagy That Regulates Oxidative Stress and Autoimmunity. Nat Commun 2024, 15, 953. [CrossRef]
- Zhang, J.; Zhu, Q.; Wang, X.; Yu, J.; Chen, X.; Wang, J.; Wang, X.; Xiao, J.; Wang, C.-C.; Wang, L. Secretory Kinase Fam20C Tunes Endoplasmic Reticulum Redox State via Phosphorylation of Ero1α. EMBO J 2018, 37, e98699. [CrossRef]
- Zhang, F.; Tao, Y.; Zhang, Z.; Guo, X.; An, P.; Shen, Y.; Wu, Q.; Yu, Y.; Wang, F. Metalloreductase Steap3 Coordinates the Regulation of Iron Homeostasis and Inflammatory Responses. Haematologica 2012, 97, 1826–1835. [CrossRef]
- Zhou, X.; An, Z.; Lei, H.; Liao, H.; Guo, X. Role of the Human Cytochrome B561 Family in Iron Metabolism and Tumors (Review). Oncol Lett 2025, 29, 111. [CrossRef]
- Hatori, Y.; Lutsenko, S. An Expanding Range of Functions for the Copper Chaperone/Antioxidant Protein Atox1. Antioxid Redox Signal 2013, 19, 945–957. [CrossRef]
- Yang, D.; Xiao, P.; Qiu, B.; Yu, H.-F.; Teng, C.-B. Copper Chaperone Antioxidant 1: Multiple Roles and a Potential Therapeutic Target. J Mol Med 2023, 101, 527–542. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



