Submitted:
24 November 2025
Posted:
25 November 2025
You are already at the latest version
Abstract
Listeria monocytogenes (Lm) is a Gram-positive, facultative intracellular bacterium traditionally known as a foodborne pathogen but increasingly recognized as a promising vector for cancer immunotherapy. Its unique virulence factors—such as listeriolysin O (LLO), internalins, phospholipases, and ActA—enable robust activation of cell-mediated immunity, making Lm-based vaccines superior to conventional treatments and other bacterial vectors. This review discusses the mechanisms by which recombinant Lm induces antitumor responses, including antigen presentation via MHC I and II pathways, and outlines strategies to attenuate pathogenicity while preserving immunogenicity, such as gene deletions, partial restoration of PrfA, and killed-but-metabolically-active strains. Furthermore, we examine key clinical trials evaluating Lm-based vaccines in HPV-associated cancers, prostate cancer, and HER-2-positive malignancies, highlighting their safety, tolerability, and efficacy in improving survival outcomes. The versatility of Lm-based immunotherapies positions them as a novel and effective approach in oncology, with ongoing trials paving the way for future therapeutic applications.
Keywords:
Introduction
Mechanism of Recombinant Listeria monocytogenes (Lm) as an Immunotherapeutic Agent
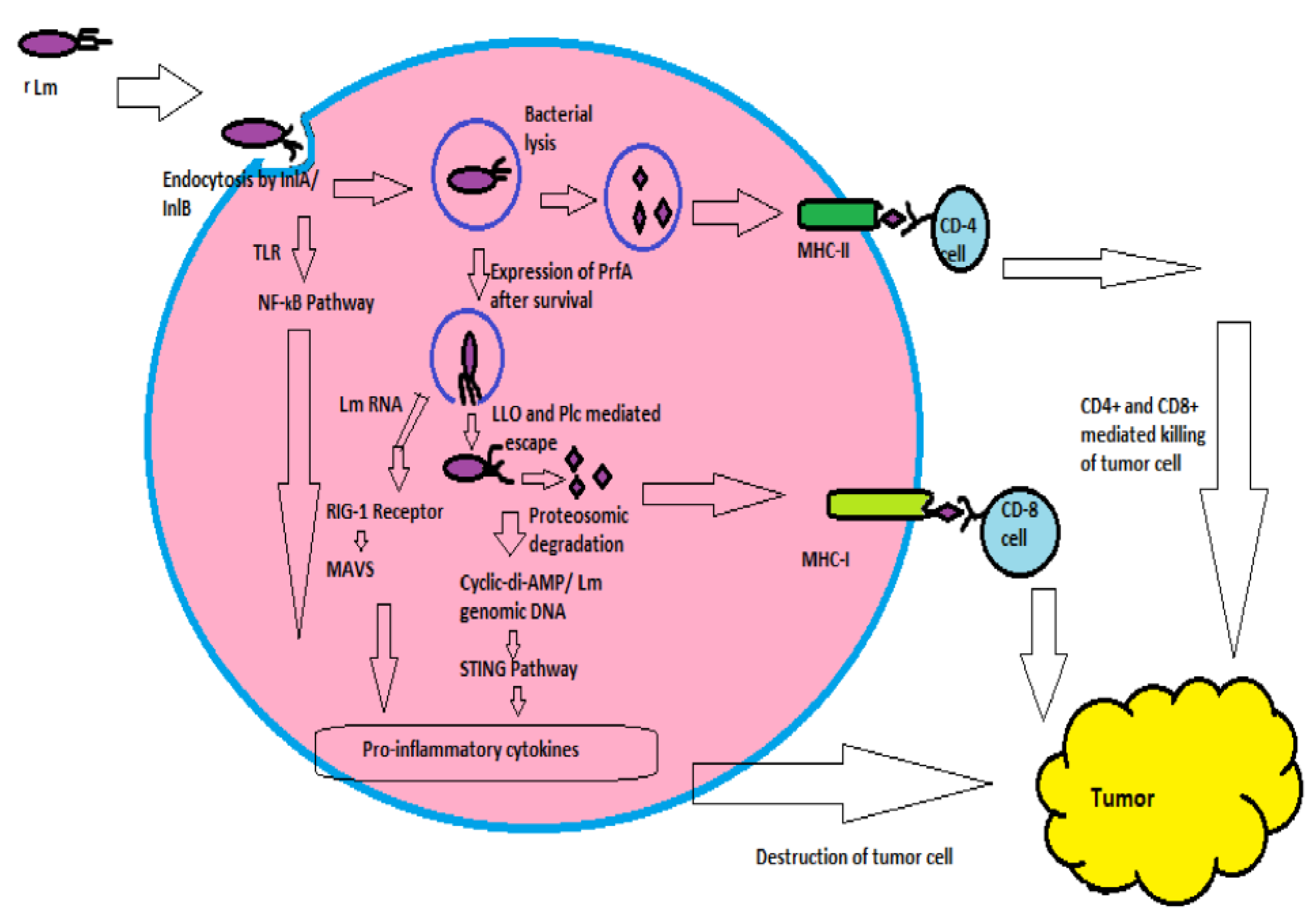
Activation Innate Immunity by Listeria monocytogenes (Lm)
-
Complete deletion of virulence factor:Listeria monocytogenes has a wide range of cellular tropism like hepatocytes, epithelial cells, endothelial cells, fibroblast, etc., mediated by InlB. (Vázquez-Boland et al, 2001) Deletion of InlB will limit the infection only to the phagocytic cells, which is the desired action. Furthermore, deleting ActA will limit the infection only into the cytoplasm and hinder the spread of the infection. The mutants lacking ActA are 1000-fold less virulent than the wild type, as their LD-50 is 1000-fold more or higher than the wild strains. (Starks et al, 2004)The strain that lacks both InlB and ActA is more selective and has lesser adverse effects than the wild type of Lm. Also this strain is widely used in different clinical trials, known as the live attenuated double deleted vaccine (LADD) produced by Aduro Biotech Company in the USA. Mutants lacking ActA and PlcB used in hepatocellular carcinoma in different clinical trials found highly immunogenic. (Chen et al, 2012)
- Partial restoration of virulence gene: As mentioned earlier, PrfA is the master transcription factor that encodes for the majority of the virulence factors. So deletion of PrfA would definitely make Listeria monocytogenes less pathogenic. However, the bacteria would be unable to escape from phagolysosome due to the absence of LLO and phospholipases, resulting in Lm less immunogenic. Partial restoration of PrfA by plasmid was the solution that was used in XFL-7 used in prostate cancer, and breast cancer was more immunogenic and attenuated than wild type. (Gunn et al, 2001; Singh et al, 2005; Shahabi et al, 2008)
- Killed but metabolically active strain: The above-mentioned attenuated vaccines can be reactivated in an immunocompromised host and might cause listeriosis. For example, ADXS11001 systemic listeriosis was observed in few patients. (Sacco et al, 2016) To minimal this adverse effect, heat-killed vaccines can be used. Nevertheless, these killed vaccines are poorly immunogenic as they cannot induce cytotoxic T-cell mediated immune response by escaping into the host cytoplasm.
- Truncated listeriolysin (t-LLO): In 2001, scientist George R. Gunn demonstrated that LLO could be used as an adjuvant. In his study, two groups of recombinant Listeria monocytogenes were produced as therapeutic vaccine. One of them was able to secret E7 (Human Papilloma Virus-16 associated cervical cancer protein) alone, and another group secreted E7 along with t-LLO (LLO deficient of hemolytic property). E7 was injected to the experimental mice and on day 7 there was 4-5mm palpable tumor was found on mice. A group of mice was treated with Lm-E7 another group was treated with Lm-LLO-E7. The result showed a 75% regression of tumor size in the second group, whereas there was no change in the tumor size in the first group. (Gunn et al, 2001) This study proved that t-LLO is responsible for escaping from phagolysosome and ultimately increases the cytotoxic T cell-mediated antitumor immunity by increased presentation of tumor antigen by MHC-I.
- ActA: In 2010, similar study was conducted by Laurence M. Wood by treating two different groups of mice; those were injected with E7 and treated one group with Lm-E7 and another group with Lm-ActA-E7. The tumor size dramatically shrunk in the second group after vaccination, as well there was little or no evidence of lung metastasis in the second group. Nevertheless, the first group showed gross lung metastasis. (Wood et al, 2010)
- 1.
- Safety trial with empty Lm vaccine: In 2002, Angelakopoulos conducted the first human trial with an empty Lm vaccine strain, where L. monocytogenes were deficient of ActA and PlcB. Here twenty healthy individuals were chosen, and 25ml of saline with the Lm vaccine escalating dose from 1×106 to 1×109 CFU was administered orally. Only two individuals had a transient increase in liver enzymes. Apart from that, no adverse effect was noted. Fifteen volunteers had detectable shedding of bacteria in the stool for four days. Lm based T-cell response was detected by the increased level of IFN-ү, but there no or was minimal level of the humoral immune response. (Angelakopoulos et al, 2002)
- 2.
- Trials with HPV-associated cancer: Human Papilloma Virus has been identified as a definite carcinogen for cervical, vulval, anal, oropharyngeal, penile, and vaginal cancer. In the year 2008, the total number of malignant cases was 12.7 million, among them 610,000 (5%) were due to HPV infection (mainly by the high-risk group 16, 18). (Forman et al, 2012) Viral oncoprotein E6 and E7 is responsible for forming the tumor by inhibition of tumor-suppressor gene p53 and RB (Roden and Stern, 2018) Axalimogene filolisbac clinical trial or AXAL or ADXS11-001 is an Lm-based vaccine that secrets E7 from HPV-16 along with tLLO.
- Phase I: In 2009, 15 individuals with invasive cervical cancer, which was unresponsive to traditional treatments were chosen to assess the safety of the ADXS11-001; the exclusion criteria was the previous history of listeriosis. They were divided into three groups, member of each group received 2 doses of ADXS11-001 vaccine intravenously every 21day either in 1×109 or 3.3×109 or 1×1010 CFU. The dose-limiting toxicity was found in those who received the highest dose; (most common was pyrexia, others were vomiting, nausea, tachycardia, headache, musculoskeletal pain) due to the innate immune response after vaccination. Two deaths were recorded during the study period, unrelated to the trial. (Maciag, Radulovic and Rothman, 2009)
- Phase II: After successfully completing the first trials, multiple phase II trials have been conducted in India and the USA. In 2010, a trial was conducted in India where 110 patients with invasive cervical cancer were chosen for the study. The study population were divided into different two groups; one group received three doses of 1×109 CFU of AXAL and another group received four doses of 1×109 CFU of AXAL along with Cisplatin (Chemotherapeutic agent). The overall survival rate was increased in both cases up to 1.5 to 2 folds. The ADXS11-001 was well-tolerated by the study population. However, the adverse effects were more in those who received cisplatin along with ADXS11-001. (Basu et al, 2018)
- Phase III (AIM2CERV): A phase III double-blinded, placebo-controlled randomized clinical trial was started in September 2016. Around 450 participants who have locally advanced cervical carcinoma and have already completed the cisplatin-based concurrent chemoradiotherapy and have a higher risk of recurrence. This is the only phase III trial Lm based vaccine till now and the estimated date of completion of the study is October 2024 (Clinical trial govt. identifier: NCT02853604)
- Trials with carcinoma of the Prostate: Prostate cancer is the second leading cause of death to the men in the USA after skin cancer and around 14% of men develop prostate cancer during their life time. (Jemal et al, 2007) Prostate-specific antigen (PSA) is widely expressed in adenocarcinoma of the prostate and it has been using as a tool for immunotherapeutic agents. (Cunha et al, 2006) In 2008, a study conducted with Lm based vaccine along with PSA (Lm-LLO-PSA) was injected into the mice with TPSA-23 tumor and the tumor showed 80% of regression from the initial. (Shahabi et al, 2008)
- 2.
- Trials with HER-2 associated cancer: Human epidermal growth factor-2 (HER-2) is a protooncogene or receptor tyrosine kinase. Overexpression of this gene responsible for several malignancies in the breast, salivary glands, bone, ovary, stomach, esophagus, etc. (Scotlandi et al, 2005; Omar, Yan and Salto-Tellez, 2015)
- 3.
- ADXS-NEO (Clinical trial govt. identifier NCT0365080): Advaxis has taken another project (Phase I) on the Lm vaccine known as ADXS-NEO, based on personalized medicine. It is a patient-specific immunotherapy for different malignancies. Following the histopathological examination, the neoepitope (MHC-bounded peptides resulting from tumor-associated mutations) (Leclerc et al, 2019) is identified from the primary or metastatic tumor. Later on, the neoepitope is integrated with LLO of live, attenuated L. monocytogenes. This vaccine is produced on the hypothesis of destruction of tumor cells by Listeria mediated cytotoxic T cells response and depletion of regulatory T-cells and myeloid-derived suppressor cells in the tumor microenvironment. (Wallecha, Singh and Malinina, 2013) As T-reg and myeloid-derived suppressor cells are responsible for tumor immunosuppression. (Plitas and Rudensky, 2020) The objective of this ongoing open-label, multicenter, non-randomized trial is to assess the safety, tolerability, clinical and immunological potency of ADXS-NEO as monotherapy or as combination therapy with immunological checkpoint inhibitor (anti PD-1 antibody) in different cancers (metastatic non-small cell carcinoma of the lung, squamous cell carcinoma of head and neck region, microsatellite stable colorectal cancer, urothelial cancer, melanoma etc.) (advaxis.com/neo-program)
Conclusions
Acknowledgement
References
- Angelakopoulos, H. et al. (2002) ‘Safety and shedding of an attenuated strain of Listeria monocytogenes with a deletion of actA/plcB in adult volunteers: a dose escalation study of oral inoculation’, Infection and immunity, 70(7), pp. 3592-3601.
- Basu, P. et al. (2018) ‘A Randomized Phase 2 Study of ADXS11-001 Listeria monocytogenes-Listeriolysin O Immunotherapy With or Without Cisplatin in Treatment of Advanced Cervical Cancer’, International journal of gynecological cancer : official journal of the International Gynecological Cancer Society, 28(4), pp. 764-772.
- Cunha, A.C. et al. (2006) ‘Tissue-specificity of prostate specific antigens: comparative analysis of transcript levels in prostate and non-prostatic tissues’, Cancer letters, 236(2), pp. 229-238.
- Denham, J.D. et al. (2018) ‘Two cases of disseminated infection following live organism anti-cancer vaccine administration in cancer patients’, International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases, 72, pp. 1-2.
- Farber, J.M. and Peterkin, P.I. (1991) ‘Listeria monocytogenes, a food-borne pathogen’, Microbiological reviews, 55(3), pp. 476-511.
- Flickinger, J.C.,Jr, Rodeck, U. and Snook, A.E. (2018) ‘Listeria monocytogenes as a Vector for Cancer Immunotherapy: Current Understanding and Progress’, Vaccines, 6(3), pp. 48. doi:.
- 10.3390/vaccines6030048.
- Forman, D. et al. (2012) ‘Global burden of human papillomavirus and related diseases’, Vaccine, 30 Suppl 5, pp. 12.
- Jemal, A. et al. (2007) ‘Cancer statistics, 2007’, CA: a cancer journal for clinicians, 57(1), pp. 43-66.
- Le, D.T. et al. (2012) ‘A live-attenuated Listeria vaccine (ANZ-100) and a live-attenuated Listeria vaccine expressing mesothelin (CRS-207) for advanced cancers: phase I studies of safety and immune induction’, Clinical cancer research : an official journal of the American Association for Cancer Research, 18(3), pp. 858-868.
- Leclerc, M. et al. (2019) ‘Recent Advances in Lung Cancer Immunotherapy: Input of T-Cell Epitopes Associated With Impaired Peptide Processing’, Frontiers in immunology, 10, pp. 1505.
- Maciag, P.C., Radulovic, S. and Rothman, J. (2009) ‘The first clinical use of a live-attenuated Listeria monocytogenes vaccine: a Phase I safety study of Lm-LLO-E7 in patients with advanced carcinoma of the cervix’, Vaccine, 27(30), pp. 3975-3983.
- Musser, M.L. et al. (2021) ‘Safety evaluation of the canine osteosarcoma vaccine, live Listeria vector’, Veterinary and comparative oncology, 19(1), pp. 92-98.
- Omar, N., Yan, B. and Salto-Tellez, M. (2015) ‘HER2: An emerging biomarker in non-breast and nongastric cancers’, Pathogenesis, 2(3), pp. 1-9.
- Plitas, G. and Rudensky, A.Y. (2020) ‘Regulatory T Cells in Cancer’, Annual Review of Cancer Biology, 4(1), pp. 459-477.
- Roden, R.B.S. and Stern, P.L. (2018) ‘Opportunities and challenges for human papillomavirus vaccination in cancer’, Nature reviews.Cancer, 18(4), pp. 240-254.
- Scotlandi, K. et al. (2005) ‘Prognostic and therapeutic relevance of HER2 expression in osteosarcoma and Ewing’s sarcoma’, European journal of cancer (Oxford, England : 1990), 41(9), pp. 1349-1361.
- Seavey, M.M. et al. (2009) ‘A novel human Her-2/neu chimeric molecule expressed by Listeria monocytogenes can elicit potent HLA-A2 restricted CD8-positive T cell responses and impact the growth and spread of Her-2/neu-positive breast tumors’, Clinical cancer research : an official journal of the American Association for Cancer Research, 15(3), pp. 924-932.
- Shahabi, V. et al. (2008) ‘Development of a Listeria monocytogenes based vaccine against prostate cancer’, Cancer immunology, immunotherapy : CII, 57(9), pp. 1301-1313.
- Stein, M.N. et al. (2020) ‘KEYNOTE-046 (Part B): Effects of ADXS-PSA in combination with pembrolizumab on survival in metastatic, castration-resistant prostate cancer patients with or without prior exposure to docetaxel’, JCO, 38(6), pp. 126.
- Wallecha, A., Singh, R. and Malinina, I. (2013) ‘Listeria monocytogenes (Lm)-LLO immunotherapies reduce the immunosuppressive activity of myeloid-derived suppressor cells and regulatory T cells in the tumor microenvironment’, Journal of immunotherapy (Hagerstown, Md.: 1997), 36(9), pp. 468476.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



