2. Materials and Methods
2.1. Study Design
This prospective longitudinal cohort pilot study was conducted at the Neonatal Intensive Care Unit of the V.I. Kulakov National Medical Research Center for Obstetrics, Gynecology, and Perinatology from June 2024 to February 2025. Preterm neonates (gestational age ⩽ 32 weeks) were enrolled; the study was approved by the local ethics committee (Protocol 04, April 18, 2024), and written informed consent was obtained from parents or legal guardians.
Eligible infants were admitted to the NICU with gestational age ⩽ 32 weeks; exclusion criteria included severe congenital malformations, chromosomal abnormalities, inherited metabolic disorders, hydrops fetalis. In addition, we collected stool samples in more mature neonates (GA>32 weeks) if they developed signs suspicious for NEC (one or more signs: pronounced abdominal distension, pain on abdominal palpation, frequent regurgitation, blood in the stool, lack of intestinal peristalsis). The samples were taken on the day these signs appeared . NEC diagnosis was based on the modified Bell’s criteria [
33] and assessed by two independent clinical experts (I.N., O.K.J.) before obtaining the results of SCFA testing.
Given real–world enrollment, the original NEC–centered plan was adapted to focus on
early–onset neonatal sepsis (EONS) versus absence of sepsis while retaining an exploratory NEC–risk subgroup. Early–onset neonatal sepsis was defined as the presence of at least two clinical symptoms and at least two laboratory signs in the presence of, or as a result of, suspected or proven infection (positive culture, microscopy, or polymerase chain reaction) within the first 72 h of life, according to the criteria listed in the expert report on neonatal and paediatric sepsis by the European Medicines Agency [
34]. Infants fulfilling these criteria comprised the EONS group; infants without clinical or laboratory evidence of sepsis during the observation windows comprised the non–EONS group. All cases of EONS were evaluated independently by two experts (I.N. and O.K.J.) before the results of SCFA testing became available. All neonates were followed up until discharge from the NICU. The small NEC–risk subgroup (Bell stage I during hospitalization) was analyzed descriptively for hypothesis generation and was not included in the between–group comparisons in
Table 1.
Fecal sampling followed predefined postnatal windows centered at approximately day of life (DoL) 3, 7, 14, 21, and 28 (hereafter TP1–TP5); when multiple samples occurred within a window, the specimen closest to the window’s median timing was selected for primary analyses.
The comparative analysis set comprised preterm infants: EONS () and non–sepsis (). Continuous variables are reported as median (Q1–Q3) and were compared using the Mann–Whitney U test; categorical variables are shown as n (%) and were compared using two–sided Fisher’s exact test, with considered significant.
Clinical and demographic characteristics are summarized in
Table 1. Groups did not differ in gestational age at birth (31 [28.5–32] vs. 31.2 [29.3–32] weeks;
) or sex distribution (
). Birth weight was lower in EONS (1121 [840–1329] vs. 1440 [1092–1679] g;
). Apgar scores were lower in EONS at 1 min (
) and 5 min (
). Surfactant treatment and invasive mechanical ventilation were more frequent in EONS (
and
, respectively). Length of NICU stay was longer in EONS (24 [15–35] vs. 11 [7–25] days;
). Lethal cases were observed only in the EONS group (4 cases, 22% vs. 0%;
). Other perinatal and feeding variables were comparable between groups (all
).
Notwithstanding this design, we additionally characterized, for descriptive and contextual purposes, a small ancillary cohort of infants who developed NEC (Bell stage II) with gestational age
weeks. These cases were reviewed under the same ethics approval and are presented here solely as a post hoc, hypothesis–generating extension; they were not included in the primary EONS vs. non–sepsis longitudinal analyses or in
Table 1.
2.2. Chemicals and Reagents
For the quantitative GC–MS analysis of SCFAs in feces, analytical-grade standards and reagents were obtained from reliable suppliers (Sigma-Aldrich, Saint Louis, MO, USA). The reference standards included acetic (AA, ≥99%), propionic (PA, ≥99%), butyric (BA, ≥99%), valeric (VA, ≥99%), isobutyric (iBA, ≥99%), isovaleric (iVA, ≥99%), 2-methylbutyric (2mBA, ≥99%), 2-methylvaleric (2mVA, ≥99%), 3-methylvaleric (3mVA, ≥99%), hexanoic (HA, ≥99%), and octanoic (OA, ≥99%) acids.
To improve quantification accuracy and correct for matrix effects, internal standards (IS) were used. AA-d4 (≥99.5%) served as the internal standard for acetic acid, while BA-1,2-13C2 (≥98%) was applied for other SCFAs, including PA, BA, VA, iBA, iVA, 2mBA, 2mVA, 3mVA, HA, and OA.
Milli-Q water was used for the preparation and dilution of calibration and working solutions, while 2.0 M hydrochloric acid (HCl) was employed for acidification prior to extraction. Methyl tert-butyl ether (MTBE) served as the solvent for liquid–liquid extraction (LLE) of SCFAs, ensuring efficient recovery and phase separation.
All reagents and consumables were handled in accordance with standard laboratory protocols and quality-control procedures to ensure the reliability and reproducibility of analytical results [
35,
36].
2.3. Sample Collection
Naturally-excreted stool samples were collected on days 3, 7, 14, 21, and 28 of life, preferably before antibiotic therapy or the morning feeding. Samples were placed in sterile containers with inert plastic inserts to prevent contact with the diaper surface and minimize volatile loss. All samples were immediately frozen at -80 °C until analysis.
In infants who developed acute intestinal symptoms during hospitalization (e.g., cases later evaluated for suspected or confirmed NEC), stool samples were additionally collected within 24 h after symptom onset according to unit protocol. Enteral feeding was discontinued at the time of symptom appearance; therefore, the feeding characteristics reported in the Results correspond to the last 24 h of enteral nutrition prior to clinical deterioration. These cases were not part of the longitudinal comparison and are presented descriptively.
2.4. Sample Preparation
A 50–100 mg aliquot of native stool was weighed into 1.6–2.0 mL Eppendorf tubes. Milli-Q water (1000 µL per 100 mg stool) was added, and the exact dilution factor was determined gravimetrically. After GC–MS analysis, SCFA concentrations (µM) were recalculated to µmol per g of stool. Samples were vortexed, sonicated for 20 min at room temperature, vortexed again for 10 min, and centrifuged (5000 rpm, RT). Then 100 µL of supernatant was transferred to a 0.5 mL tube, 5 µL of internal standard solution (d4-acetic acid 103 µM, 13C2-butyric acid 102 µM) and 5 µL of 2 M HCl were added, followed by 200 µL of methyl tert-butyl ether (MTBE). After vortexing (10 min) and centrifugation (15000 rpm, 5 min), the upper organic phase was transferred to an autosampler vial for GC–MS analysis.
The protocol was adapted from previously published methods, demonstrating the applicability of water as a surrogate matrix and MTBE extraction after acidification. Kim et al. (2022) [
36] established a methodological basis validated across several bio-matrices, whereas our work [
35] applied and tested it on clinical stool and plasma samples. In this study, the protocol was further modified for neonatal stool, to reduce matrix effects and improve reproducibility.
2.5. Calibration Standards and Quality Controls
Calibration standards were prepared in Milli-Q water, with sample preparation identical to that of the stool extracts, beginning with a 100 µL aqueous aliquot. Stock solutions of the analytes were prepared in acetonitrile at the following concentrations: AA at 106 µM; PA, BA, and iBA acids at 105 µM; and VA, iVA, 2mBA, 2mVA, 3mVA, HA, and OA at 104 µM. These stock standards were stored at and serially diluted with water to create working solutions. The internal standards (IS) consisted of d4-AA (104 µM, stored at ) and 13C2-BA (10 mg/mL, stored at ). A working internal standard (ISTD) solution containing 103 µM d4-AA and 102 µM 13C2-BA was prepared, with 5 µL added to each sample.
Seven-point calibration curves and four levels of quality-control (QC) standards were prepared in Milli-Q water, which was selected as a surrogate matrix based on previous validation data [
35,
36] and confirmed by standard-addition tests in this study. Calibration and QC samples underwent identical preparation steps to fecal extracts, starting from the stage of 100 µL aliquot acidification and MTBE extraction. For AA, calibration levels were 2, 4, 10, 20, 40, 100, and 160 µM, with an additional high-level point at 2000 µM when needed to account for elevated concentrations. For PA, BA and iBA, calibration levels were 0.2, 0.4, 1, 2, 4, 10, and 16 µM, with an optional extended level of 200 µM for high-concentration samples. For other acids (2mBA, iVA, VA, 2mVA, 3mVA, HA, OA), levels were 0.02, 0.04, 0.1, 0.2, 0.4, 1, and 1.6 µM, optionally extended to 20 µM.
Calibration curves were constructed by plotting the peak area ratio of each SCFA to its corresponding internal standard against the analyte concentration, followed by linear regression. The linearity for each SCFA was confirmed by a coefficient of determination (R2) exceeding 0.995. The limit of detection (LOD) was calculated as 3.3×SD/b, where SD is the standard deviation of the Y-intercept and b is the slope of the regression curve. The limit of quantification (LOQ) was defined as 3×LOD. For several samples, the quantification was additionally verified using the standard addition method with four spiked concentration levels.
Calibration curves were constructed by plotting the peak area ratio of each SCFA to its corresponding internal standard against the analyte concentration, followed by linear regression (
Table 2). The linearity for each SCFA was confirmed by a coefficient of determination (R
2) exceeding 0.995. The limit of detection (LOD) was calculated as 3.3×SD/b, where SD is the standard deviation of the Y-intercept and b is the slope of the regression curve. The limit of quantification (LOQ) was defined as 3×LOD. For several samples, the quantification was additionally verified using the standard addition method with four spiked concentration levels.
2.6. GC–MS Analysis
Analyses were performed using an Agilent 7890B/5977B GC–MS system equipped with an HP-FFAP capillary column (30 m × 0.25 mm × 0.25 µm). Helium was used as the carrier gas at a constant flow of 1.7 mL/min. The oven temperature was programmed as follows: initial temperature 60 ∘C (held for 1.5 min), ramped at 20 ∘C/min to 240 ∘C, and held for 7.5 min. Electron ionization (EI) at 70 eV was used with a solvent delay of 4 min. Injector, ion source, and quadrupole temperatures were maintained at 240 ∘C, 230 ∘C, and 150 ∘C, respectively.
A preliminary series of full-scan measurements was performed to determine retention times and identify characteristic fragment ions by spectral matching with the NIST17 library [
37]. Subsequently, ion registration was carried out in SIM (Single Ion Monitoring) mode for quantitative determination of individual analytes (
Table 2). Quantification was performed in Selected Ion Monitoring (SIM) mode by targeting characteristic fragment ions for each analyte. The ion at m/z 43 was monitored for isobutyric acid (iBA), while the ion at m/z 60 was used for the simultaneous detection of acetic, butyric, valeric, isovaleric, and 3-methylvaleric acids. Propionic, 2-methylbutyric, and 2-methylvaleric acids were quantified using the ion at m/z 74. For the IS, the ions m/z 46 and 63 were selected for the deuterated standard d
4-acetic acid, and the ion m/z 62 was selected for
13C
2-butyric acid. Compound identity was confirmed by retention time and spectral comparison with authentic standards (NIST17). Method performance (R
2 > 0.995, LOD 0.5–1 µM, accuracy ±10 %, CV < 15 %) and extract stability at
up to 14 days were consistent with published data [
35,
36].
2.7. Statistical Analysis
All statistical analyses were performed in Python 3.13 using the packages pandas (data handling), NumPy (numerical operations), SciPy (non-parametric tests), scikit-learn (multivariate modelling), and matplotlib/seaborn (visualisation).
Continuous variables are summarised as median (Q1; Q3), and categorical variables as n (%). Between-group comparisons for clinical characteristics were carried out using the Mann–Whitney U test for continuous variables and Fisher’s exact test for categorical variables. Statistical significance was defined as ; values with were interpreted as trends.
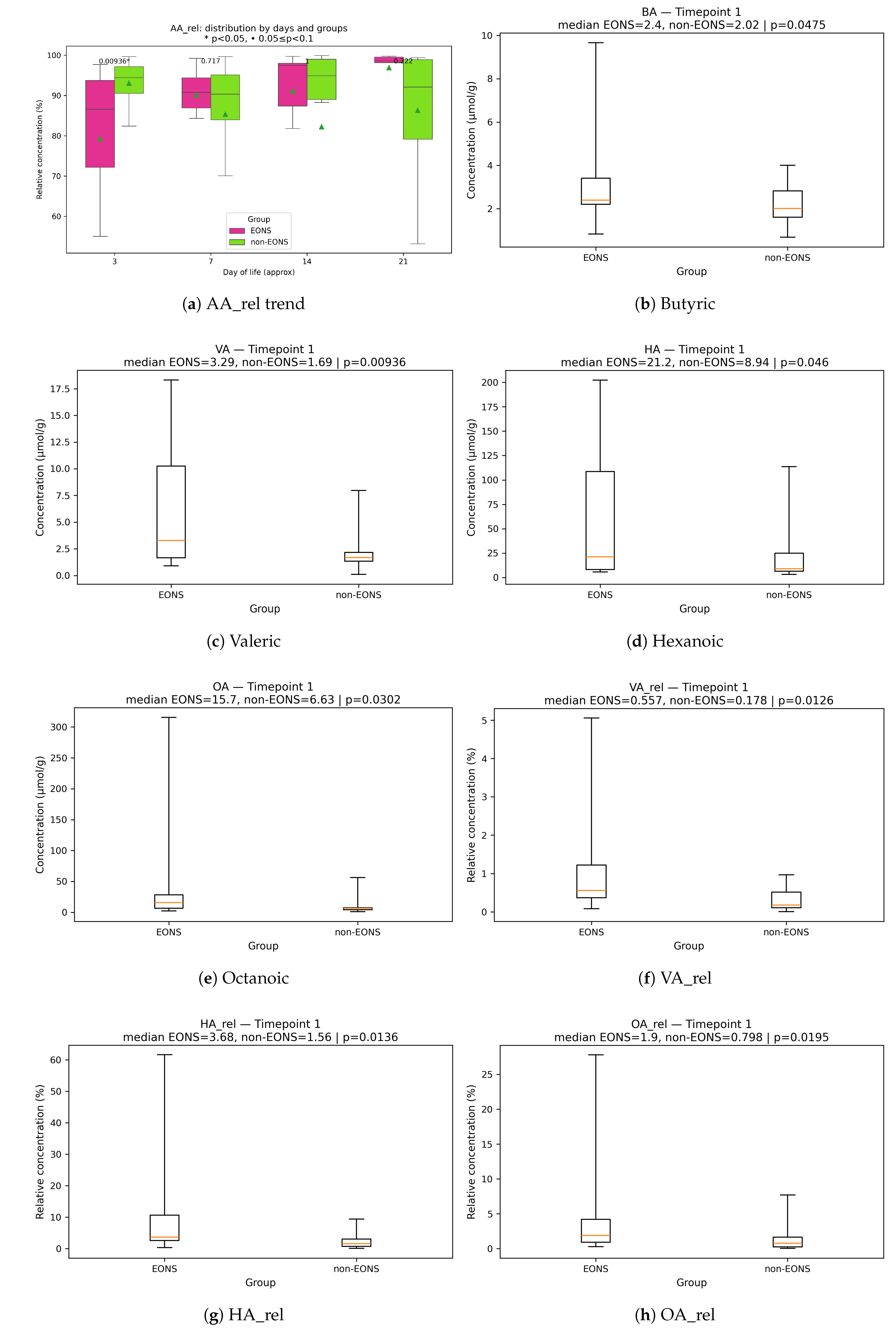
For fecal short-chain fatty acid (SCFA) analysis, both absolute concentrations and derived metrics were evaluated. Derived variables included:
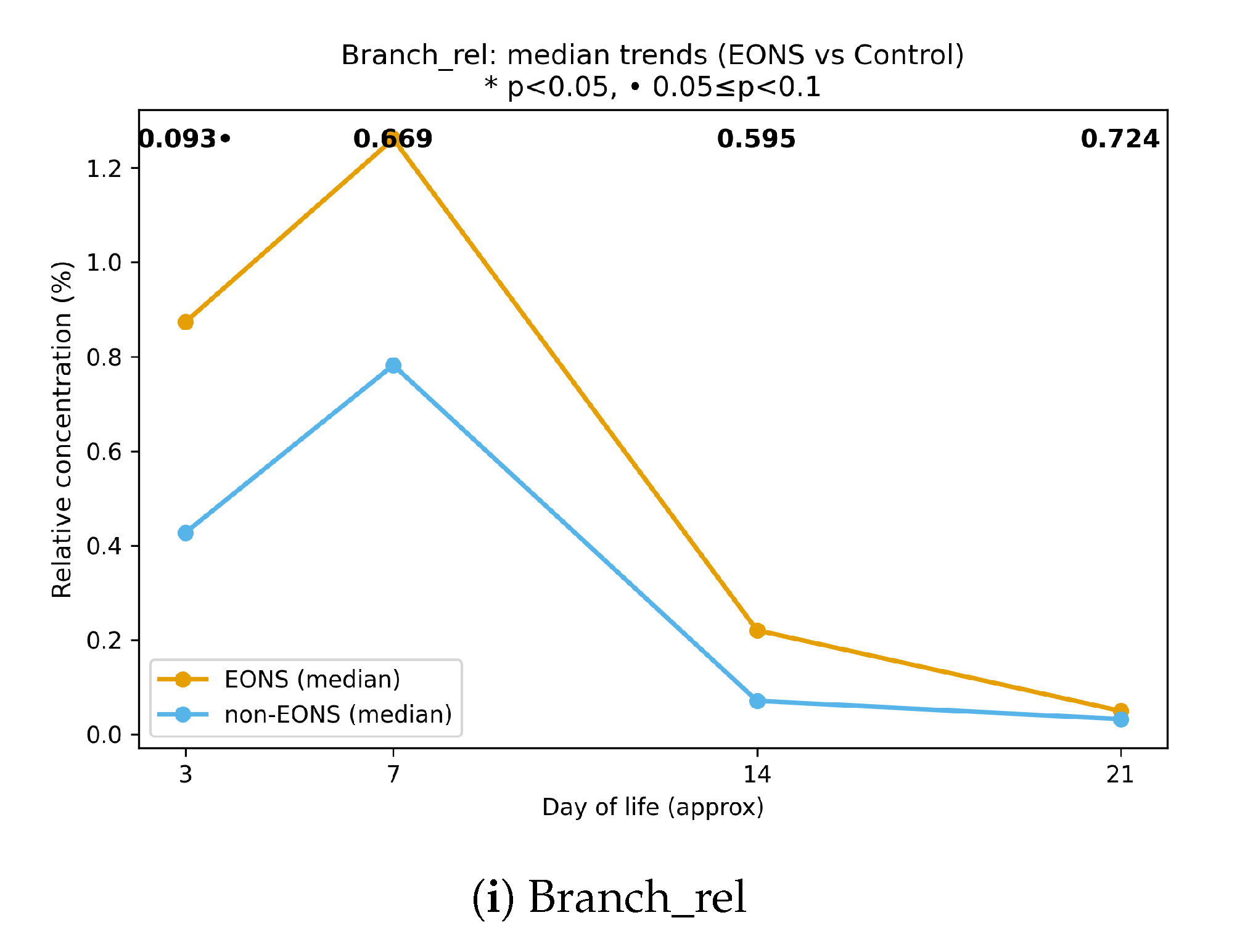
relative fractions, (percentage of total SCFAs);
composite branched-chain indices, and ;
pairwise ratios of metabolites, .
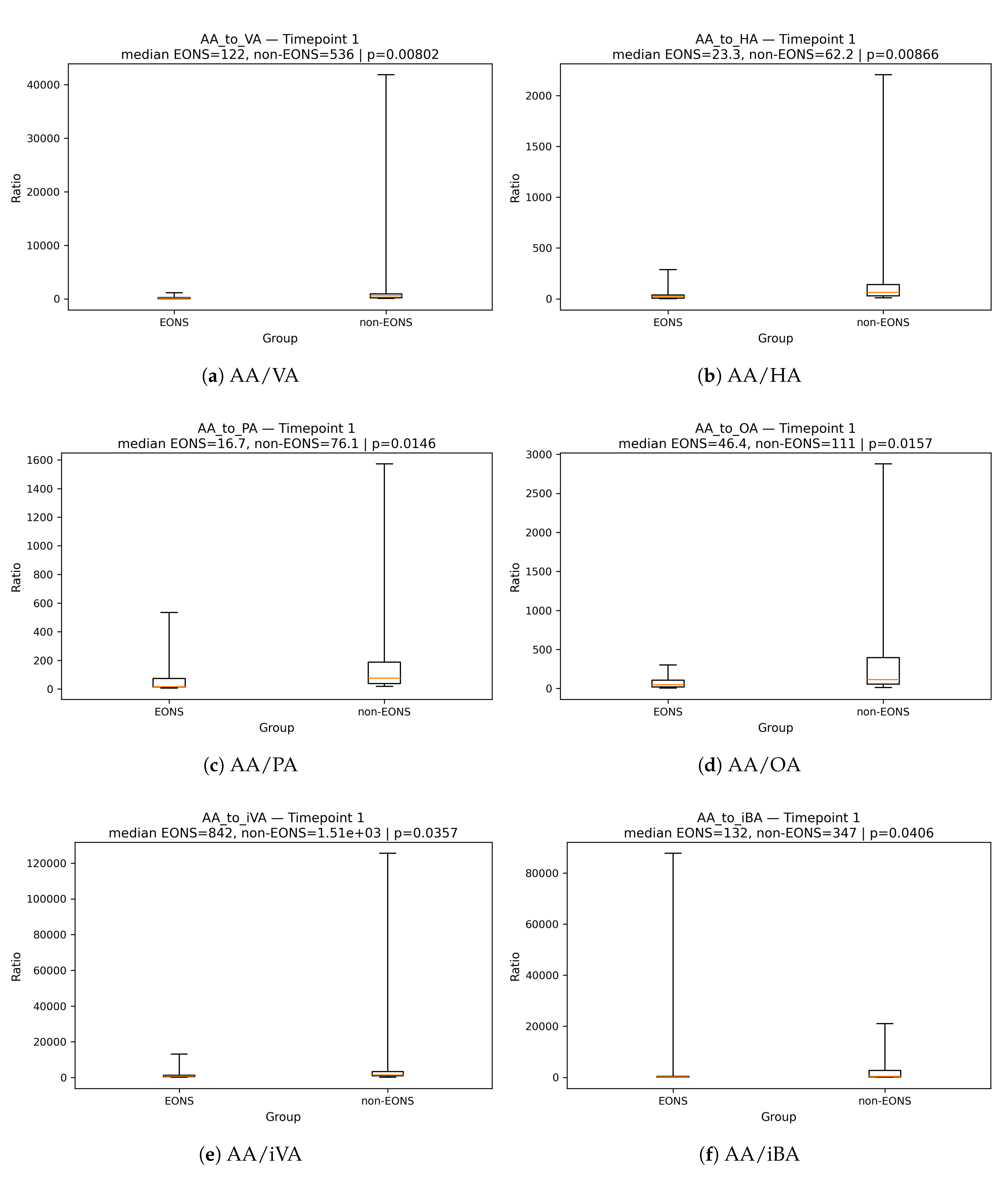
For each analyte, timepoint-specific differences between the EONS and non-sepsis groups were assessed at TP1–TP5 (approximately 3, 7, 14, 21, and 28 days of life) using the Mann–Whitney U test. Infants evaluated for suspected or confirmed NEC formed a small exploratory subgroup; their SCFA profiles were summarised descriptively and were not included in inferential testing.
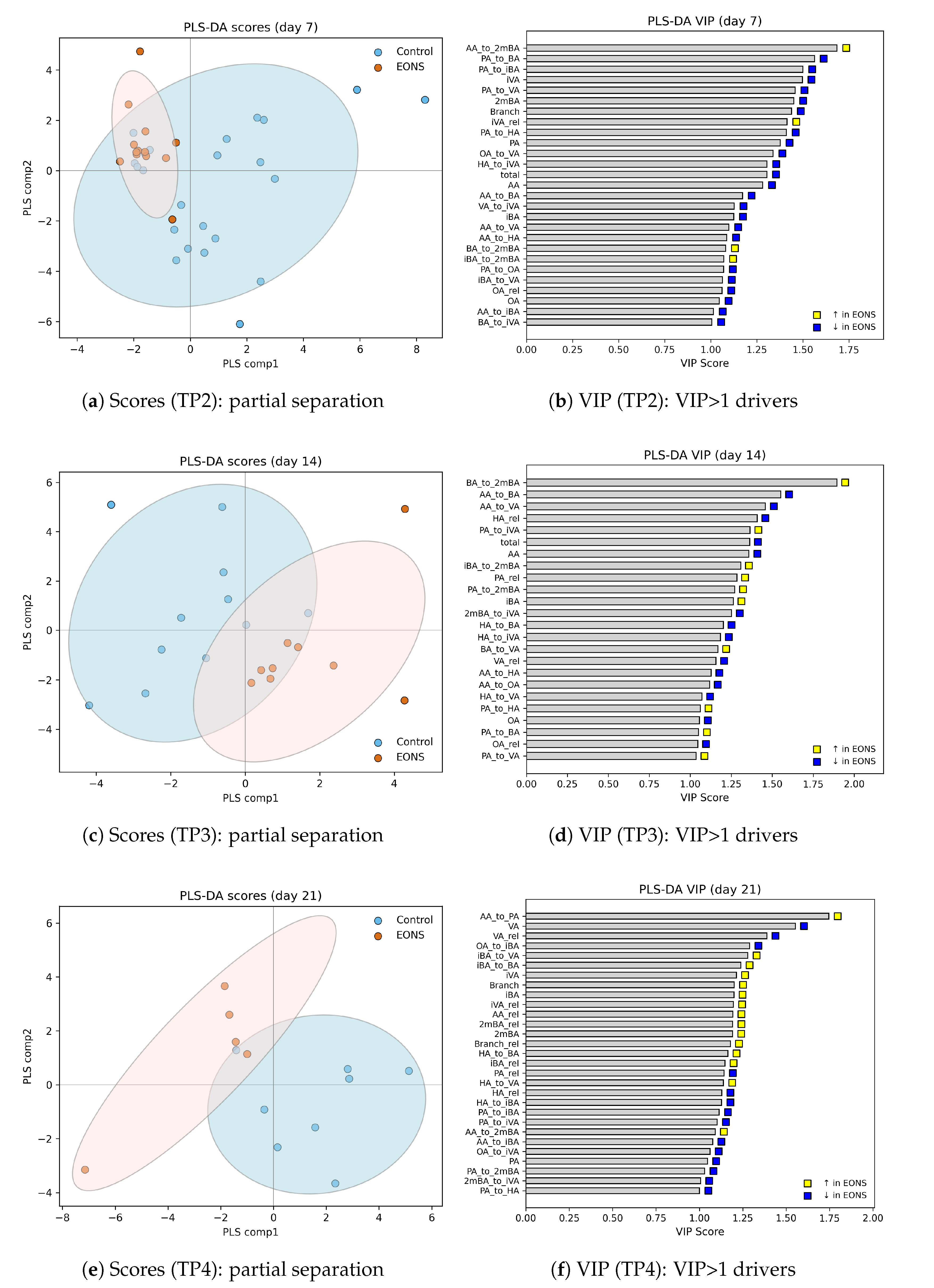
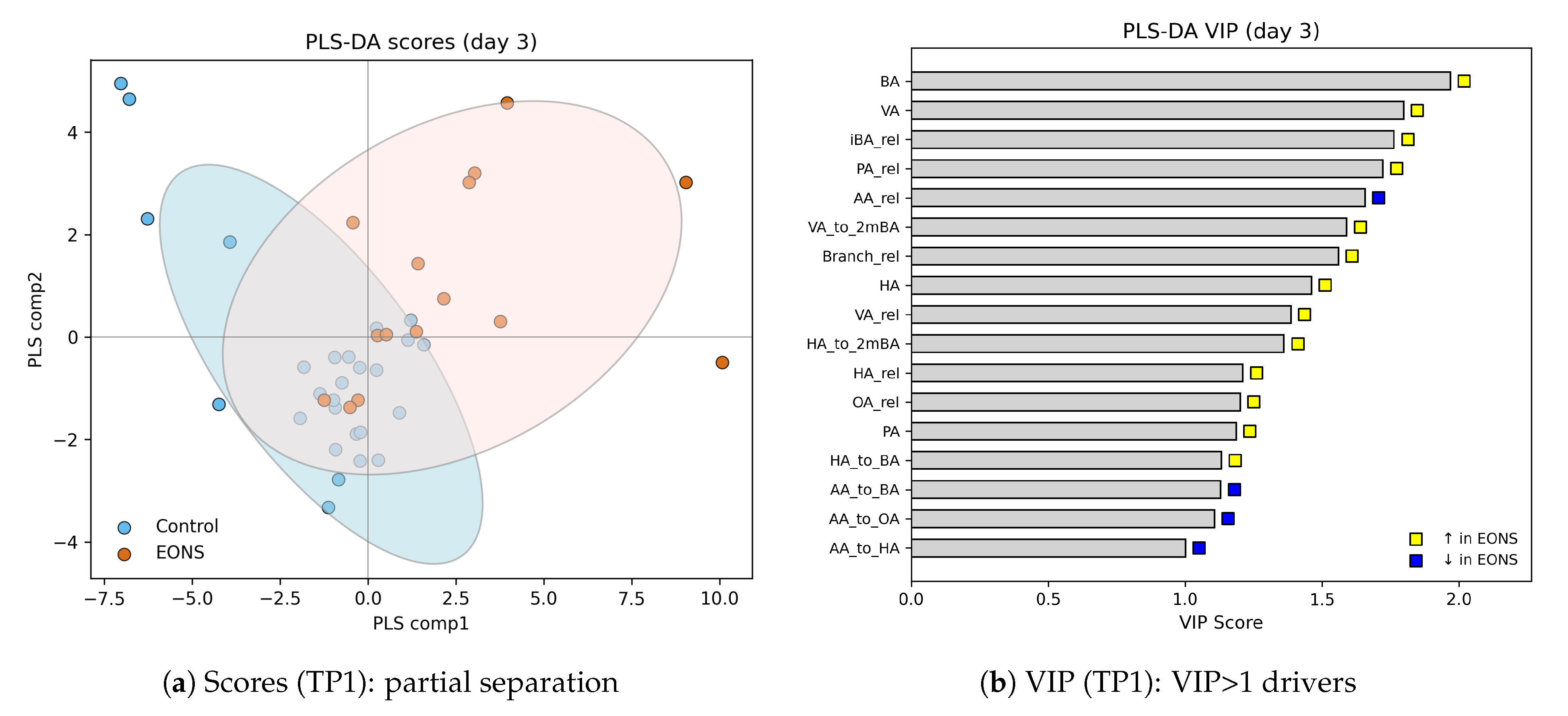
Multivariate analysis was performed using partial least squares–discriminant analysis (PLS–DA) implemented in scikit-learn. Variable Importance in Projection (VIP) scores were calculated for each metabolite and ratio; variables with VIP were considered informative contributors to group separation. Temporal patterns were visualised using boxplots, PLS–DA score plots, and median trend curves across days of life, with p-values annotated on timepoint-specific plots where appropriate.
2.8. Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the local ethics committee (2024, April 18, # 04). Informed consent was obtained from parents or legal guardians of all participants.