Submitted:
19 November 2025
Posted:
21 November 2025
You are already at the latest version
Abstract
Keywords:
Introduction
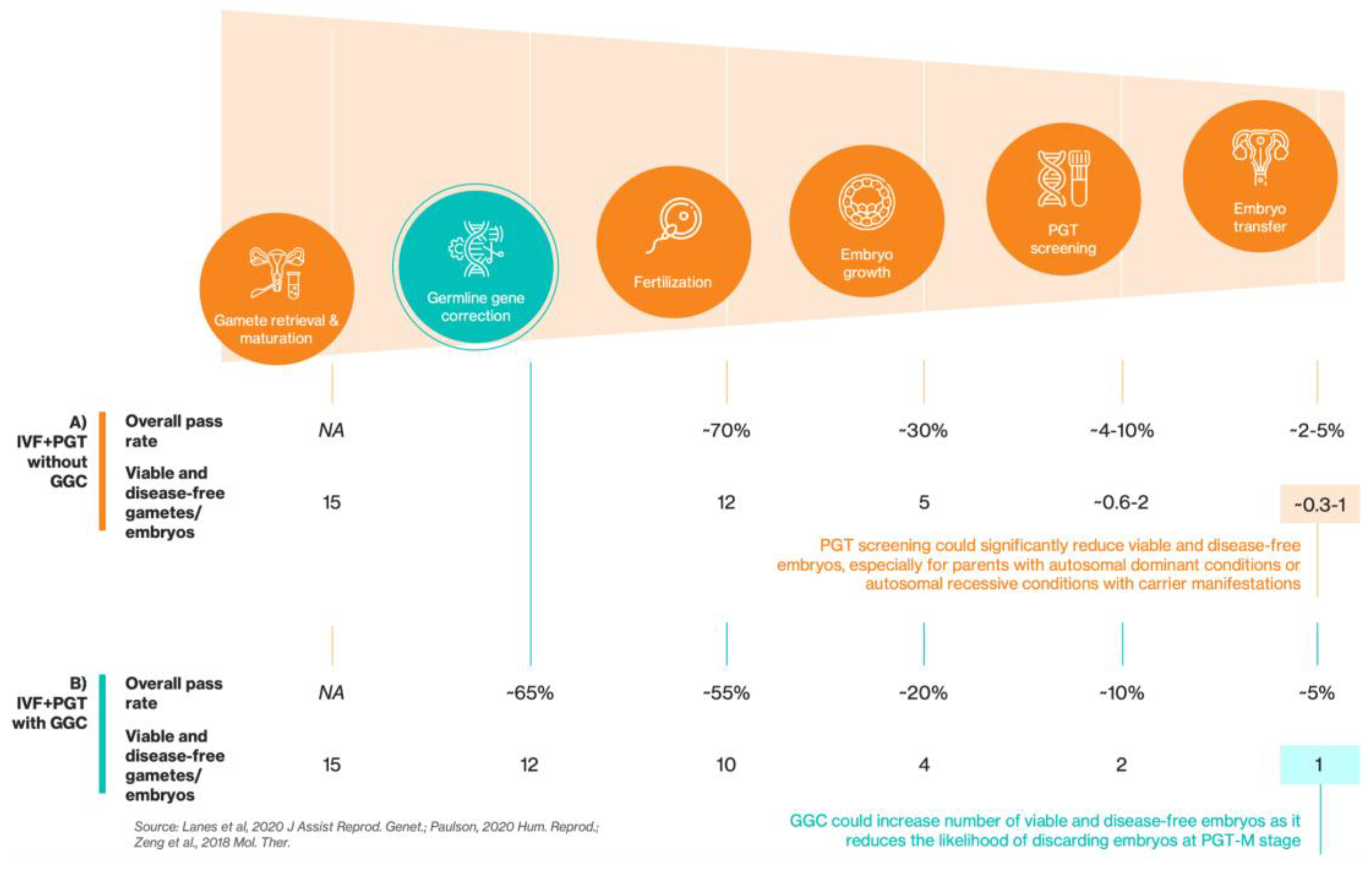
Results
Discussion
Materials & Methods
Model Overview and Key Assumptions
Calculation of Annual Births with Condition
Scenario 1: Conservative Floor (Current Access)
Scenario 2: Potential Ceiling
Data availability statement
Declaration of interest
Footnotes:
- a. In the USA, the newborn rate for sickle cell diseases (SCD) is about 1 in 365 African American births and about 1 in 16,300 Hispanic births[18]. There were 3.6M live births in the US in 2023, of which 25.3% were Hispanic (0.91M) and 13.9% were African American (0.50M)[27]. For African American births, ~1,425 of them are estimated to have SCD; for Hispanic births, ~56 of them are estimated to have SCD.
- b. Using the 2.6% average IVF usage rate in the USA[29], it is estimated that ~36 births with SCD were assisted by IVF. Assuming each birth was the result of 1 IVF cycle during which 18 oocytes were extracted. 25% of oocytes carry disease mutations for SCD and GGC was successful, ~4.5 oocytes would have been corrected. Given the approximately 80% attrition rate from oocyte retrieval to development of viable blastocysts, about 1 embryo per IVF cycle would have been SCD-free because the oocyte was corrected by GGC. In sum, a total of ~35 embryos would have been corrected per year in the US.
- c. Assuming IVF-GGC-PGT pipeline is available to all at-risk couples and GGC is 60-80% effective at creating on-target only changes (as shown in recent study [25,30]), the IVF-GGC-PGT pipeline has the potential to prevent SCD in 80% of the ~1,425 births with the condition each year in the US. Given ~1 embryo corrected per birth, the total embryos corrected are ~855-1,140.
- d. In the USA, Cystic Fibrosis (CF) affects 1 in every 4,000 newborns (via newborn screening)[20]. Given the 3.6M live births, ~900 births are expected to have CF.
- e. Using the 2.6% average IVF usage rate, it’s estimated that ~25 births were assisted by IVF. Using the same assumptions in b, about 1 embryo would have been corrected by GGC per IVF. Therefore, a total of ~25 embryos would have been corrected.
- f. Using the same assumptions in c, given ~1 embryo corrected per birth, the total embryos corrected are ~540-720.
- g. Marfan syndrome (MFS) occurs in 1 of 5,000 live births[24]. Given the 3.6M live births in the US, ~720 births are expected to have Marfan syndrome in the USA.
- h. Using the 2.6% average IVF usage rate in the USA, it is estimated that ~19 births with MFS were assisted by IVF. Assuming each birth was the result of 1 IVF cycle during which 18 oocytes were extracted, 50% of oocytes carry disease mutation for MFS and GGC was successful, ~9 oocytes would have been corrected. Given the ~80% IVF attrition rate (from extraction to embryo development), about 2 embryos per IVF cycle would have been MFS-free because the oocyte was corrected by GGC. In sum, a total of ~35 embryos would have been corrected.
- i. Using the same assumptions in c, given ~2 embryos corrected per birth and 60-80% GGC success rate, the total embryos corrected are ~865-1,150.
- j. In the USA, Huntington’s disease (HD) is diagnosed in 6.5 per 100,000 persons each year[28]. Because HD is not routinely screened in newborns, age-adjusted diagnostic frequency is a proxy available for measure of frequency of birth incidence. See limitation of this approach in the “Study limitation” section. Given the 3.6M live births in the US, ~235 births are expected to have HD.
- k. Using the 2.6% average IVF usage rate, it’s estimated that ~6 births with HD were assisted by IVF. Using the same assumptions in h, about 2 embryos per IVF cycle would have been HD-free because the oocyte was corrected by GGC. In sum, a total of ~10 embryos would have been corrected per year in the US.
- l. Using the same assumptions in c, given ~2 embryos corrected per birth and 60-80% GGC success rate, the total embryos corrected are ~280-375. While the number of embryos corrected is higher than the number of live births with Huntington’s disease, these disease-allele-free embryos could substantially enhance the chances of disease-free live birth per IVF cycle for at-risk couples.
References
- Marwaha, S., Knowles, J.W., and Ashley, E.A. (2022). A guide for the diagnosis of rare and undiagnosed disease: beyond the exome. Genome Med. 14, 23. [CrossRef]
- Fermaglich, L.J., and Miller, K.L. (2023). A comprehensive study of the rare diseases and conditions targeted by orphan drug designations and approvals over the forty years of the Orphan Drug Act. Orphanet J. Rare Dis. 18, 163. [CrossRef]
- Takeuchi, K. (2021). Pre-implantation genetic testing: Past, present, future. Reprod. Med. Biol. 20, 27–40. [CrossRef]
- Ran, F.A., Hsu, P.D., Wright, J., Agarwala, V., Scott, D.A., and Zhang, F. (2013). Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308.
- Gaudelli, N.M., Komor, A.C., Rees, H.A., Packer, M.S., Badran, A.H., Bryson, D.I., and Liu, D.R. (2017). Programmable base editing of A•T to G•C in genomic DNA without DNA cleavage. Nature 551, 464–471.
- Anzalone, A.V., Randolph, P.B., Davis, J.R., Sousa, A.A., Koblan, L.W., Levy, J.M., Chen, P.J., Wilson, C., Newby, G.A., Raguram, A., et al. (2019). Search-and-replace genome editing without double-strand breaks or donor DNA. Nature 576, 149–157. [CrossRef]
- Ghazal, S., and Patrizio, P. (2017). Embryo wastage rates remain high in assisted reproductive technology (ART): a look at the trends from 2004-2013 in the USA. J. Assist. Reprod. Genet. 34, 159–166. [CrossRef]
- Deltas, C., Savva, I., Voskarides, K., Papazachariou, L., and Pierides, A. (2015). Carriers of autosomal recessive Alport syndrome with thin basement membrane nephropathy presenting as focal segmental glomerulosclerosis in later life. Nephron 130, 271–280. [CrossRef]
- Hames, A., Khan, S., Gilliland, C., Goldman, L., Lo, H.W., Magda, K., and Keathley, J. (2023). Carriers of autosomal recessive conditions: are they really “unaffected?” J. Med. Genet. 61, 1–7.
- Paulson, R.J. (2020). Hidden in plain sight: the overstated benefits and underestimated losses of potential implantations associated with advertised PGT-A success rates. Hum. Reprod. 35, 490–493. [CrossRef]
- Bahadur, G., Homburg, R., Jayaprakasan, K., Raperport, C.J., Huirne, J.A.F., Acharya, S., Racich, P., Ahmed, A., Gudi, A., Govind, A., et al. (2023). Correlation of IVF outcomes and number of oocytes retrieved: a UK retrospective longitudinal observational study of 172 341 non-donor cycles. BMJ Open 13, e064711. [CrossRef]
- Gunnala, V., Irani, M., Melnick, A., Rosenwaks, Z., and Spandorfer, S. (2018). One thousand seventy-eight autologous IVF cycles in women 45 years and older: the largest single-center cohort to date. J. Assist. Reprod. Genet. 35, 435–440. [CrossRef]
- Bender, M.A., and Carlberg, K. (1993). Sickle cell disease. In GeneReviews(®) (University of Washington, Seattle).
- Ye, L., Zhao, D., Li, J., Wang, Y., Li, B., Yang, Y., Hou, X., Wang, H., Wei, Z., Liu, X., et al. (2024). Glycosylase-based base editors for efficient T-to-G and C-to-G editing in mammalian cells. Nat. Biotechnol. 42, 1538–1547. [CrossRef]
- Agasa, B., Bosunga, K., Opara, A., Tshilumba, K., Dupont, E., Vertongen, F., Cotton, F., and Gulbis, B. (2010). Prevalence of sickle cell disease in a northeastern region of the Democratic Republic of Congo: what impact on transfusion policy? Transfus. Med. 20, 62–65. [CrossRef]
- Gupta, K., Krishnamurti, L., and Jain, D. (2024). Sickle cell disease in India: the journey and hope for the future. Hematology Am. Soc. Hematol. Educ. Program 2024, 1–9. [CrossRef]
- GBD 2021 Sickle Cell Disease Collaborators (2023). Global, regional, and national prevalence and mortality burden of sickle cell disease, 2000-2021: a systematic analysis from the Global Burden of Disease Study 2021. Lancet Haematol. 10, e585–e599.
- CDC (2024). Data and Statistics on Sickle Cell Disease. Sickle Cell Disease (SCD). https://www.cdc.gov/sickle-cell/data/index.html.
- Chambers, G.M., Dyer, S., Zegers-Hochschild, F., de Mouzon, J., Ishihara, O., Banker, M., Mansour, R., Kupka, M.S., and Adamson, G.D. (2021). International Committee for Monitoring Assisted Reproductive Technologies world report: assisted reproductive technology, 2014. Hum. Reprod. 36, 2921–2934. [CrossRef]
- Scotet, V., L’Hostis, C., and Férec, C. (2020). The changing epidemiology of cystic fibrosis: Incidence, survival and impact of the CFTR gene discovery. Genes (Basel) 11, 589. [CrossRef]
- Sousa, A.A., Hemez, C., Lei, L., Traore, S., Kulhankova, K., Newby, G.A., Doman, J.L., Oye, K., Pandey, S., Karp, P.H., et al. (2025). Systematic optimization of prime editing for the efficient functional correction of CFTR F508del in human airway epithelial cells. Nat. Biomed. Eng. 9, 7–21. [CrossRef]
- Faquih, T.O., Aziz, N.A., Gardiner, S.L., Li-Gao, R., de Mutsert, R., Milaneschi, Y., Trompet, S., Jukema, J.W., Rosendaal, F.R., van Hylckama Vlieg, A., et al. (2023). Normal range CAG repeat size variations in the HTT gene are associated with an adverse lipoprotein profile partially mediated by body mass index. Hum. Mol. Genet. 32, 1741–1752. [CrossRef]
- Matuszek, Z., Arbab, M., Kesavan, M., Hsu, A., Roy, J.C.L., Zhao, J., Yu, T., Weisburd, B., Newby, G.A., Doherty, N.J., et al. (2025). Base editing of trinucleotide repeats that cause Huntington’s disease and Friedreich's ataxia reduces somatic repeat expansions in patient cells and in mice. Nat. Genet. 57, 1437–1451. [CrossRef]
- Marfan syndrome and congenital heart conditions (2009). https://www.aboutkidshealth.ca/marfan-syndrome-and-congenital-heart-conditions.
- Zeng, Y., Li, J., Li, G., Huang, S., Yu, W., Zhang, Y., Chen, D., Chen, J., Liu, J., and Huang, X. (2018). Correction of the Marfan syndrome pathogenic FBN1 mutation by base editing in human cells and heterozygous embryos. Mol. Ther. 26, 2631–2637. [CrossRef]
- Amato, P., Mikhalchenko, A., and Mitalipov, S. (2025). The case for germline gene correction: state of the science. Fertil. Steril. 124, 22–29. [CrossRef]
- CDC (2025). Births and Natality. https://www.cdc.gov/nchs/fastats/births.htm.
- Bruzelius, E., Scarpa, J., Zhao, Y., Basu, S., Faghmous, J.H., and Baum, A. (2019). Huntington’s disease in the United States: Variation by demographic and socioeconomic factors: HUNTINGTON'S DISEASE IN THE UNITED STATES. Mov. Disord. 34, 858–865.
- ASRM (2025). US IVF usage increases in 2023, leads to over 95,000 babies born. American Society for Reproductive Medicine (ASRM). https://www.asrm.org/news-and-events/asrm-news/press-releasesbulletins/us-ivf-usage-increases-in-2023-leads-to-over-95000-babies-born/.
- Marti-Gutierrez, N., Liang, D., Chen, T., Lee, Y., Ma, H., Koski, A., Mikhalchenko, A., Heitner, S.B., Kang, E., Amato, P., et al. (2020). Gene conversion in human embryos induced by gene editing. Fertil. Steril. 114, e35. [CrossRef]
| Condition | Mode of Inheritance | Births with Condition Each Year in US | Estimated Embryos Corrected Each Year in USA | |
|---|---|---|---|---|
| Conservative Floor | Potential Ceiling | |||
| Sickle Cell Disease | Autosomal Recessive | ~1,425 a | ~35 b | ~855-1,140 c |
| Cystic Fibrosis | Autosomal Recessive | ~900 d | ~25 e | ~540-720 f |
| Marfan Syndrome | Autosomal Dominant | ~720 g | ~35 h | ~865-1,150 i |
| Huntington’s Disease | Autosomal Dominant | ~235 j | ~10 k | ~280-375 l |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




