The findings of this study support the concept that SARS-CoV-2 infection may lead to persistent endocrine consequences, even several years after the acute episode. Although the respiratory impact of COVID-19 was initially the main focus, increasing evidence has demonstrated that the virus exerts complex systemic effects, involving multiple ACE2-expressing target organs, including the pancreas and thyroid gland [
7,
10]. These observations are consistent with the hypothesis that SARS-CoV-2 is not solely a respiratory pathogen, but also a disruptor of endocrine and metabolic homeostasis.
4.1. General Considerations on the Persistent Endocrine Implications of SARS-CoV-2 Infection
The present study provides a detailed assessment of endocrine status in previously hospitalized COVID-19 patients, evaluated four years after the acute episode. This represents one of the longest post-COVID endocrine follow-up investigations reported to date, allowing characterization of long-term endocrine sequelae well beyond the 6–24 month interval explored in most published studies [
5,
6].
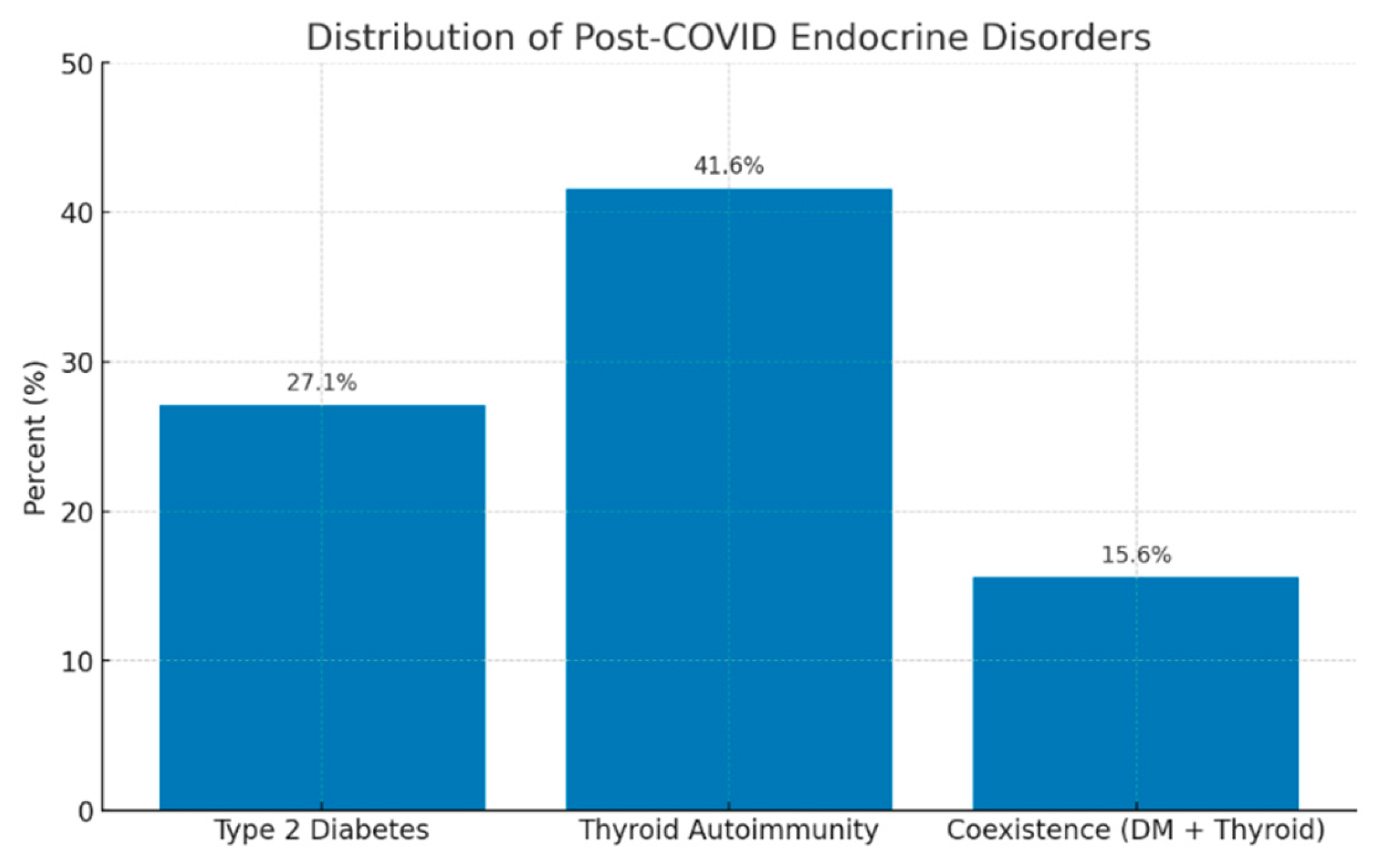
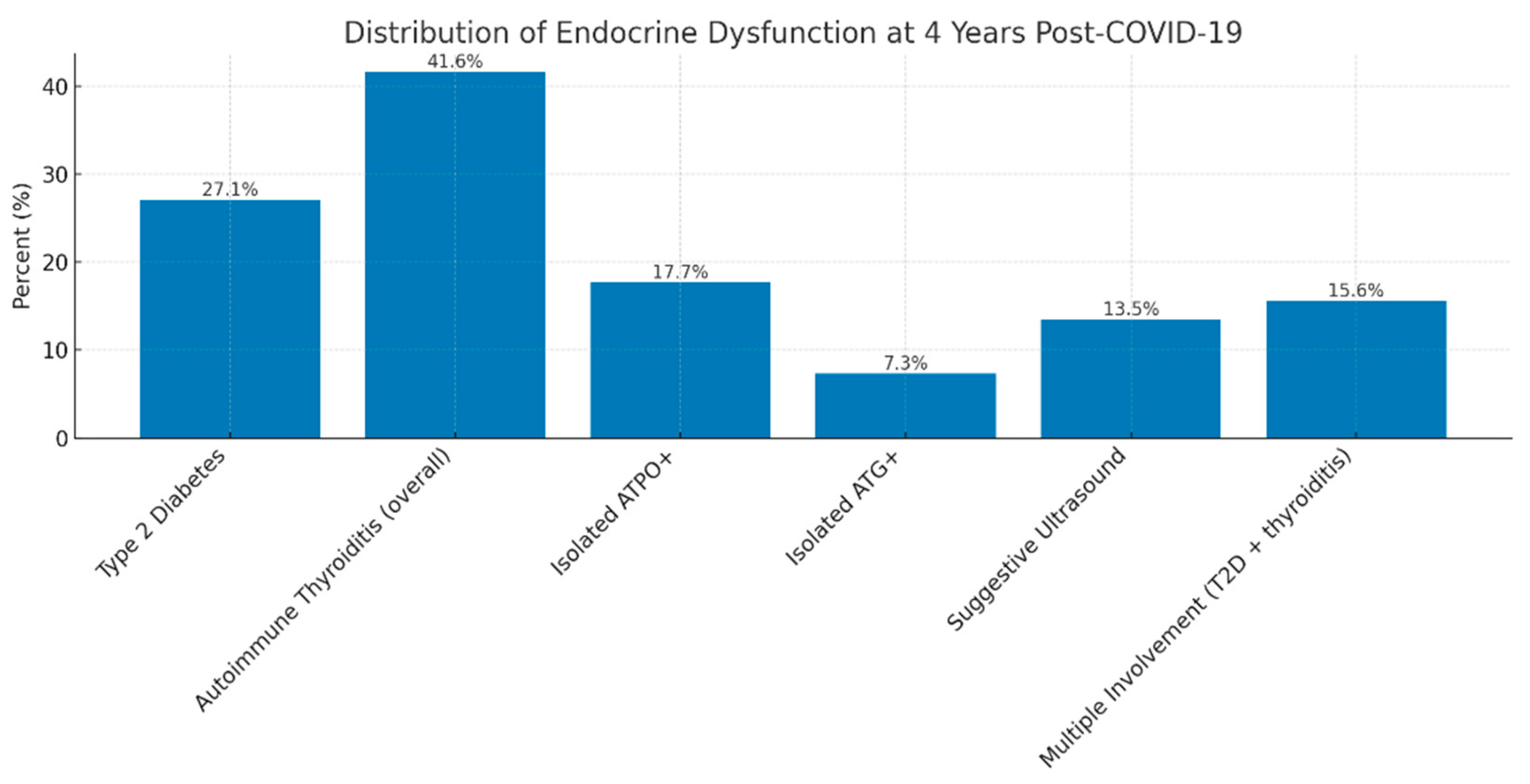
At the time of reassessment, 27% of patients without a prior history of diabetes met diagnostic criteria for type 2 diabetes mellitus. This proportion aligns with the increased risk of post-COVID diabetes documented in large population-based studies [
13,
26], but its persistence at four years post-infection suggests that metabolic disturbances may remain long after clinical recovery.
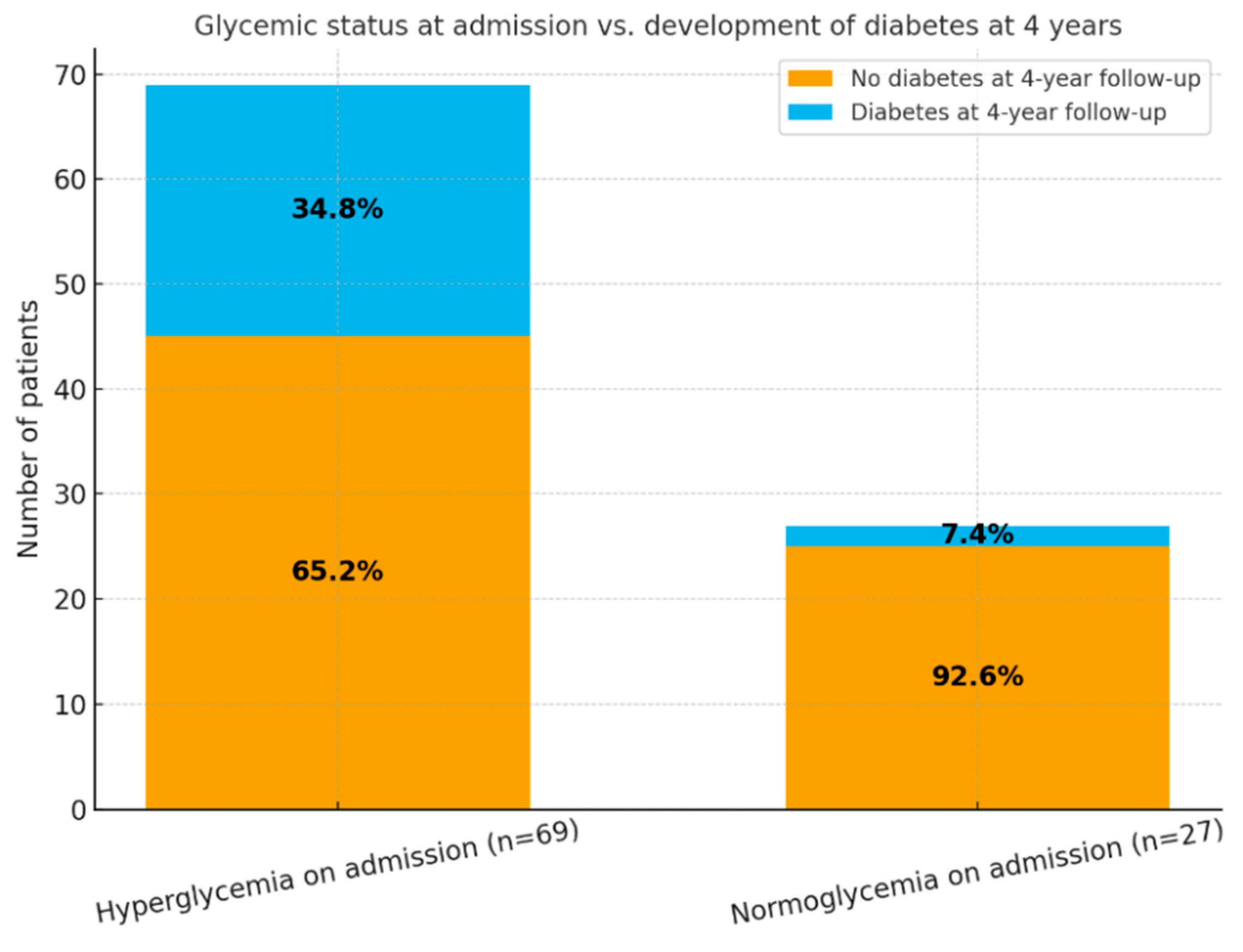
A particularly relevant finding is the association between acute-phase hyperglycemia and subsequent diabetes development. Among patients who presented with hyperglycemia at admission (70.8%), 22.1% developed diabetes by the 4-year follow-up, compared with only 7.4% of initially normoglycemic patients. These results support the hypothesis that COVID-19–related stress hyperglycemia reflects an underlying metabolic vulnerability, amplified by systemic inflammation and the host immune response to viral infection [
12,
16].
Thyroid analysis revealed a high prevalence of autoimmune markers in patients with no previous thyroid disease: 29.8% had elevated anti-TPO antibodies, 17.8% had anti-thyroglobulin antibodies (ATG), and 19% showed ultrasound features suggestive of autoimmune thyroiditis, including diffuse hypoechogenicity, parenchymal heterogeneity, or hypervascularization. These rates exceed those expected in the general population (≈10–15%) and are consistent with findings from Asian and European cohorts assessed 6–12 months post-COVID [
18,
21,
27].
The persistence of these abnormalities four years after infection suggests that thyroid autoimmunity may be a delayed or chronic process, even in the absence of overt symptoms. Notably, higher rates of ATPO positivity and structural thyroid changes were observed among patients who experienced moderate, severe, or critical COVID-19, reinforcing the link between systemic inflammatory burden and post-infectious thyroid autoimmunity [
20,
22].
Overall, the findings indicate that a substantial proportion of previously hospitalized COVID-19 patients exhibit persistent or newly developed metabolic dysfunction, thyroid autoimmunity, and, in some cases, structural thyroid alterations detectable on ultrasound at four years post-infection. These observations support the concept that SARS-CoV-2 may act as a trigger for long-lasting endocrine disturbances, mediated by systemic inflammation, oxidative stress, immune dysregulation, and possibly direct viral effects on endocrine tissues [
8,
9,
19].
The defining strength of this study lies in the unusually long interval between the initial COVID-19 infection and the time of evaluation, offering a rare perspective on the very long-term endocrine consequences of the disease. By jointly assessing thyroid function, autoimmune markers, and glucose metabolism, the study provides strong evidence that SARS-CoV-2 infection can leave detectable endocrine footprints even four years after the acute episode. These findings support the need for periodic monitoring of endocrine function in patients with a history of COVID-19, particularly those who experienced severe disease or hyperglycemia during hospitalization [
6,
28]. Implementing late post-infection metabolic and thyroid screening protocols may enable early detection and appropriate management of delayed endocrine complications.
4.2. New-Onset Diabetes Mellitus as a Metabolic Sequela of COVID-19
In our cohort, 27% of patients without a prior history of diabetes mellitus (DM) were diagnosed with type 2 diabetes at the 4-year post-infection follow-up. This proportion exceeds the incidence reported within the first 6–24 months after infection, but aligns in trend with findings from large population-based analyses. Wander et al. observed a significant increase in diabetes incidence among more than 2.8 million U.S. veterans with a history of COVID-19, reporting a relative excess risk of 40–60% and an excess burden of 13–15 new cases per 1000 persons at 12 months, including those who were not hospitalized [
29]. Subsequently, Xie and Al-Aly confirmed, in extensive cohorts from the U.S. Veterans Affairs Health System, a persistent elevation in diabetes risk at 12–24 months post-infection, with a gradient proportional to the severity of the acute illness [
13,
26]. European studies, including analyses based on German health records, have also documented incidence rate ratios ranging from 1.2 to 1.6 for new-onset diabetes following COVID-19, compared to acute respiratory infections of non-COVID origin [
30].
Several potential mechanisms may explain the association between SARS-CoV-2 infection and post-infectious diabetes mellitus. In the first place, direct viral injury to pancreatic β-cells appears to play a central role. SARS-CoV-2 can infect pancreatic islet cells through ACE2 and TMPRSS2 receptors, leading to cytopathic effects, impaired insulin secretion, and altered β-cell gene expression. These changes may reduce the functional β-cell reserve and accelerate the transition from prediabetes to overt diabetes [
8,
31,
32]. Then, another mechanism involves the systemic inflammatory response, or “cytokine storm”, characteristic of acute COVID-19, which can further disrupt glucose homeostasis. Elevated proinflammatory cytokines such as IL-6 and TNF-α interfere with insulin signaling pathways, particularly the insulin receptor substrate/phosphoinositide 3-kinase/protein kinase B (IRS/PI3K/AKT) pathway, leading to reduced glucose uptake and increased insulin resistance, promote lipolysis and hepatic gluconeogenesis, and decrease peripheral insulin sensitivity [
10,
33]. Persistent low-grade inflammation after recovery may maintain this dysglycemic state. Additionally, post-infectious insulin resistance and mitochondrial dysfunction represent another pathway linking COVID-19 to chronic hyperglycemia. Endothelial injury, oxidative stress, and mitochondrial abnormalities induced by SARS-CoV-2 can aggravate hepatic and muscular insulin resistance, impair glucose utilization, and blunt compensatory β-cell responses [
9,
34,
35]. Taken together, these mechanisms suggest that COVID-19 may exert long-lasting metabolic effects extending well beyond the acute phase of infection.
4.3. Admission Hyperglycemia as a Predictor of Long-Term Diabetes
In our cohort, 71.9% of patients exhibited hyperglycemia at hospital admission, and 33.3% of these individuals developed type 2 diabetes at the 4-year follow-up. This finding supports the hypothesis that stress-induced hyperglycemia serves as a marker of pre-existing metabolic vulnerability that becomes "unmasked" in the context of acute inflammation [
12,
16]. The persistence of systemic inflammation, alongside subsequent behavioral and metabolic factors, may further drive the transition to overt diabetes mellitus.
Our findings are consistent with those of Zhu et al., who demonstrated that hospitalized COVID-19 patients with blood glucose levels >140 mg/dL at admission had more than twice the risk of developing subsequent diabetes compared to normoglycemic patients [
36]. This reinforces the premise that stress-induced hyperglycemia represents not only a marker of acute disease severity, but also a metabolic predictor of long-term dysglycemia post-COVID [
37].
Stress hyperglycemia likely arises from a combination of mechanisms described earlier, compounded by excessive activation of the hypothalamic–pituitary–adrenal (HPA) axis, which elevates cortisol secretion and promotes hyperglycemia [
8,
10,
33]. Over time, these pathways may exceed the compensatory capacity of pancreatic β-cells, precipitating type 2 diabetes even in individuals previously considered normoglycemic. Chronic inflammation, oxidative stress, and mitochondrial dysfunction additionally contribute to sustained insulin resistance and progressive dysregulation of glucose homeostasis [
34,
35].
These findings underscore the clinical relevance of admission hyperglycemia as an early warning indicator [
37]. Patients presenting with elevated blood glucose during acute COVID-19 infection should undergo long-term metabolic monitoring, even if normoglycemia is restored at discharge. Recommended follow-up assessments include HbA1c, fasting plasma glucose, and lipid profile testing at 3–6 months post-discharge, and annually thereafter, to enable early detection of new-onset diabetes or prediabetes [
12]. Enhanced vigilance is particularly warranted in individuals with established cardiometabolic risk factors, such as older age, obesity, hypertension, dyslipidemia, or corticosteroid exposure during the acute illness. Implementing such strategies may facilitate timely lifestyle modifications and, where necessary, pharmacologic intervention to mitigate progression to overt diabetes mellitus.
4.4. COVID-19 and Thyroid Dysfunction: Long-Term Implications
The results of our study confirm that thyroid involvement represents one of the most frequent endocrine sequelae following COVID-19. Four years after the acute episode, nearly one-third of patients exhibited markers of thyroid autoimmunity, with 29.8% testing positive for anti-thyroid peroxidase (anti-TPO) antibodies and 17.8% for anti-thyroglobulin (anti-Tg) antibodies, while 19% showed ultrasonographic features suggestive of autoimmune thyroiditis. These rates are higher than those reported in the general European population (10–15%) and are comparable to findings from international post-COVID cohorts [
21,
27]. In an Italian study, Campi et al. reported a 15.7% prevalence of anti-TPO antibodies three months post-infection—approximately double that observed in a pre-pandemic control group (7.7%) [
27]. In a Hong Kong cohort, Lui et al. identified a 1.7% incidence at six months, particularly among patients treated with interferon beta-1b [
21]. Similar data have emerged from studies in China and Italy, where post-COVID thyroid autoimmunity prevalence ranged between 12% and 25% at 6–12 months of follow-up [
39,
40,
41]. By comparison, the higher values observed in our cohort may reflect persistent residual immune activation and a cumulative long-term inflammatory effect.
Several mechanisms may underlie the link between SARS-CoV-2 infection and the development of post-infectious autoimmune thyroiditis. One of the most plausible explanations involves the high expression of ACE2 and TMPRSS2 receptors in the thyroid gland. Transcriptomic and immunohistochemical analyses have demonstrated dense ACE2 expression in thyroid follicular epithelial cells, comparable to that observed in the lung, intestine, and kidney [
42,
43]. This expression pattern renders the thyroid susceptible to direct viral entry, allowing SARS-CoV-2 to induce local cytopathic damage through viral replication and destruction of follicular cells. As a result, intracellular thyroid antigens—particularly thyroglobulin (Tg) and thyroid peroxidase (TPO), are released into the extracellular environment, becoming targets for the immune system. This so-called
antigen spillage phenomenon may trigger secondary autoimmune responses, especially in genetically predisposed individuals carrying HLA-DR3 or HLA-DR5 haplotypes. Moreover, the local inflammatory process may upregulate adhesion and co-stimulatory molecules on follicular cells, effectively transforming them into non-professional antigen-presenting cells that perpetuate thyroid autoimmunity [
42,
43].
Direct infection–induced cytopathic injury is further amplified by the systemic inflammatory response characteristic of acute COVID-19, commonly referred to as the “cytokine storm.” Elevated circulating levels of IL-6, TNF-α, IFN-γ, and IL-1β can alter thyroid antigen expression and increase their immunogenicity. These cytokines also promote lymphocytic infiltration into thyroid tissue via chemokines such as CXCL10 and CCL2, while disrupting peripheral immune tolerance and activating autoreactive T and B lymphocytes [
44]. Persistent low-grade inflammation and elevated pro-inflammatory cytokines months or even years after infection may sustain subclinical immune activation, gradually promoting the transition to chronic autoimmune thyroiditis of the Hashimoto type [
44,
45].
Another key mechanism is molecular mimicry, a well-established concept in post-viral autoimmunity. Structural similarities have been identified between specific regions of the SARS-CoV-2 spike (S) and nucleocapsid (N) proteins and thyroid autoantigens such as thyroglobulin and thyroid peroxidase [
46,
47]. This molecular homology may elicit cross-reactive immune responses, whereby lymphocytes and antibodies initially directed against viral epitopes begin to recognize and attack thyroid structures. The persistent immune stimulation during acute infection amplifies this process, potentially initiating or exacerbating autoimmune thyroiditis even after viral clearance [
45,
46].
Finally, SARS-CoV-2 infection may reactivate latent or subclinical thyroid autoimmunity in genetically predisposed individuals. Host genetic factors may further modulate the risk of post-COVID thyroid autoimmunity. Previous studies have identified HLA-DR3 and HLA-DR5 alleles as major susceptibility markers for autoimmune thyroid diseases, including Hashimoto’s thyroiditis. The interplay between viral-induced immune activation, molecular mimicry, and genetic predisposition could therefore explain the persistence of thyroid autoantibodies observed in our cohort [
45,
47]. In such cases, the intense systemic inflammatory response and immune activation during acute COVID-19 may destabilize immune homeostasis, leading to loss of self-tolerance [
44,
45,
48]. This mechanism is more frequently described in women, reflecting sex-specific immunological dimorphism and potentially accounting for the higher incidence of autoimmune thyroiditis observed in our cohort.
Most longitudinal studies indicate that thyroid dysfunctions emerging during the acute phase of COVID-19 are transient. Lui et al. (
Thyroid, 2023) observed that 82.4% of patients with acute thyroid abnormalities returned to normal within 3–6 months. However, a subset of patients develop persistent autoimmunity, characterized by sustained elevation of anti-TPO and/or anti-Tg antibody titers at successive follow-up evaluations, even in the absence of overt clinical hypothyroidism [
21,
40,
48]. Our findings, obtained four years after infection, confirm the existence of this subset: nearly 30% of patients exhibit serological or ultrasonographic markers of autoimmune thyroiditis, suggesting a late post-viral autoimmune phase with slow evolution.The persistence of thyroid autoantibodies, even in the absence of overt hormonal dysfunction, may have significant clinical implications, increasing the risk of progressive hypothyroidism, fatigue, and subtle metabolic disturbances [
49].
Recent reviews published in
Frontiers in Endocrinology (2023) have highlighted that patients with severe COVID-19 are at an increased risk of developing thyroid dysfunction, including non-thyroidal illness syndrome during the acute phase and autoimmune thyroiditis during long-term follow-up. The persistence of elevated thyroid autoantibody titers has been reported particularly among subgroups of patients with marked inflammatory responses [
50]. Given these findings, periodic monitoring of thyroid function is recommended for individuals with a history of COVID-19, especially those who experienced moderate or severe disease with elevated inflammatory markers during the acute phase [
21,
41]. Re-evaluation should be performed 12–24 months after infection, even in the absence of clinical symptoms, and should include measurement of TSH, FT4, and FT3, assessment of anti-TPO and anti-Tg antibodies, and thyroid ultrasonography to allow early detection of structural or functional abnormalities. Post-COVID thyroid involvement should be interpreted within the broader context of SARS-CoV-2–induced endocrine disturbances, which may also encompass new-onset diabetes mellitus, insulin resistance, and hypothalamic–pituitary axis dysfunctions [
51]. These manifestations reflect the systemic impact of the virus on immune–endocrine homeostasis, underscoring the potential for chronic sequelae and the importance of long-term multidisciplinary follow-up [
6,
49,
51].
4.5. Endocrine Autoimmunity – A Potential Link in Post-COVID Syndrome (PASC)
Our study provides additional evidence supporting the hypothesis that endocrine autoimmunity may represent a key component of the complex spectrum of post-acute sequelae of COVID-19 (PASC). This syndrome is defined by the persistence or emergence of new symptoms more than 12 weeks after the acute infection, in the absence of alternative explanations, and encompasses a wide range of systemic manifestations, including chronic fatigue, cognitive impairment (“brain fog”), respiratory dysfunction, cardiovascular involvement, and metabolic disturbances [
52,
53].
In this context, the thyroid and metabolic dysfunctions observed four years after SARS-CoV-2 infection may be interpreted not merely as isolated sequelae, but as endocrine expressions of long COVID syndrome. Dysregulation of the hypothalamic–pituitary–peripheral axes, residual inflammation, and persistent autoimmunity may account for part of the nonspecific symptomatology observed in these patients, particularly fatigue, reduced physical performance, and mild cognitive disturbances [
54,
55].
Thyroid autoimmunity, identified in nearly one-third of re-evaluated patients, may play a central role in the pathophysiological mechanisms underlying PASC. Even in the absence of overt hypothyroidism, the presence of anti-TPO and anti-thyroglobulin antibodies may be associated with subclinical thyroid dysfunction, potentially impacting energy metabolism, thermoregulation, and neuromuscular function [
56]. Studies have shown that patients with early-stage autoimmune thyroiditis can experience symptoms such as chronic fatigue, concentration difficulties, and mood disturbances, features that closely resemble those described in long COVID [
56,
57].
Post-COVID metabolic alterations, including new-onset diabetes and persistent insulin resistance, may amplify systemic inflammation and contribute to mitochondrial dysfunction and reduced exercise capacity [
12,
58]. Chronic hyperglycemia activates proinflammatory pathways (NF-κB, IL-6, TNF-α) and induces oxidative stress, sustaining a state of low-grade inflammation characteristic of PASC [
59]. This supports an integrated pathogenic model in which acute SARS-CoV-2 infection acts as a trigger for endocrine autoimmunity and dysfunction, while residual inflammation and metabolic disturbances contribute to symptom persistence [
45]. Furthermore, potential correlations can be drawn between endocrine autoimmunity and persistent symptomatology. Chronic fatigue may reflect subclinical hypothyroidism or subtle alterations in energy metabolism, whereas cognitive disturbances (“brain fog”) may be associated with impaired neurotransmission and cerebral glucose metabolism. Exercise intolerance and muscle weakness could be exacerbated by insulin resistance and altered thyroid hormone activity, while autonomic symptoms (tachycardia, anxiety, heat intolerance) may arise from combined neuroendocrine and autoimmune dysregulation [
56,
58,
59]. Lui et al. (2021) reported that patients with post-COVID thyroid dysfunction more frequently experience fatigue and concentration difficulties, even in the absence of persistent abnormalities in TSH or FT4 levels [
56]. Similarly, recent metabolic studies have confirmed the role of chronic inflammation and insulin resistance in sustaining post-COVID symptoms [
58,
59].
Patients presenting with persistent fatigue, exercise intolerance, or cognitive disturbances should be evaluated for subclinical hypothyroidism and thyroid autoimmunity, while post-infectious glycemic monitoring may help prevent delayed diagnosis of post-COVID diabetes mellitus. Implementing a multidisciplinary follow-up protocol involving infectious disease specialists, endocrinologists, and rehabilitation experts could significantly improve functional outcomes and quality of life in these patients [
60]. Overall, these findings highlight the importance of recognizing endocrine autoimmunity as a potential driver of long-term systemic symptoms in post-COVID syndrome and underscore the need for integrated clinical management strategies.
4.6. Interaction Between Metabolic and Thyroid Axes and Risk Factors for Post-COVID Endocrine Sequelae
There is a close interrelationship between thyroid function and glucose metabolism, both systems being regulated through interconnected hormonal mechanisms. Thyroid hormones influence insulin sensitivity, hepatic glucose production, and basal metabolic rate, whereas insulin and metabolic status modulate the peripheral conversion of thyroxine (T4) to triiodothyronine (T3) [
60,
61]. Hypothyroidism, even in its subclinical form, leads to reduced insulin clearance, decreased expression of glucose transporters (GLUT-4), and increased insulin resistance at both muscular and hepatic levels [
62]. Conversely, hyperthyroidism enhances hepatic gluconeogenesis and protein catabolism, thereby promoting hyperglycemia [
59]. These bidirectional alterations explain the high prevalence of glucose intolerance and diabetes mellitus among patients with thyroid dysfunction [
59,
61]. In the post-COVID context, where autoimmune thyroid involvement and persistent insulin resistance may coexist, a vicious endocrine–metabolic cycle can develop. Systemic inflammation and oxidative stress induce both thyroid autoimmunity and impaired insulin sensitivity; in turn, chronic hyperglycemia and insulin resistance sustain low-grade inflammation, perpetuating thyroid injury [
58,
59].
In our cohort, a partial overlap was observed between patients with thyroid autoimmunity and those diagnosed with post-COVID diabetes, suggesting the presence of shared pathogenic mechanisms. This association aligns with evidence from previous studies showing that persistent systemic inflammation, a hallmark of post-acute COVID-19 syndrome, can sustain both thyroid autoimmunity and metabolic dysregulation through overlapping immune–endocrine pathways [
51]. Furthermore, recent comprehensive reviews have highlighted that COVID-19–related thyroid disorders often coexist with insulin resistance and other components of metabolic dysfunction, supporting a bidirectional link between autoimmune and metabolic sequelae [
55].
Risk factor analysis in our cohort indicates that the occurrence of post-COVID endocrine sequelae, both new-onset diabetes mellitus and thyroid autoimmunity, is not a random phenomenon but rather reflects the interplay between individual predisposition and the severity of systemic inflammation during the acute phase of infection. Female sex was associated with a higher prevalence of thyroid autoimmunity, confirming the increased immunological susceptibility of women to autoimmune disorders, including Hashimoto’s thyroiditis [
56].
Likewise, older patients (≥60 years) more frequently exhibited diabetes and thyroid dysfunction, which can be explained by immunosenescence, decreased β-cell reserve, and preexisting metabolic comorbidities [
6]. The severity of acute COVID-19 emerged as the strongest predictor of long-term endocrine disturbances, consistent with previous recommendations emphasizing the need for endocrine follow-up in patients with severe disease [
6]. Our results align with the findings of Xie and Al-Aly, who demonstrated a progressive increase in the risk of incident diabetes proportional to the severity of the initial infection [
13]. Regarding thyroid involvement, Campi et al. and Lui et al. described a similar association between the intensity of the acute inflammatory response (“cytokine storm”) and persistently elevated anti-TPO titers at 6–12 months after infection [
21,
55]. Moreover, obesity and dyslipidemia were correlated with an increased risk of post-COVID diabetes, reinforcing their role as preexisting metabolic vulnerability factors. These comorbidities amplify the inflammatory response and insulin resistance, creating a favorable background for chronic glycemic disturbances following severe infectious stress [
10,
58].
Overall, these findings support the hypothesis that severe forms of COVID-19, intense systemic inflammation, and adverse metabolic profiles act synergistically to determine long-term endocrine risk. Identifying these determinants enables the definition of a post-COVID endocrine risk profile, useful for guiding targeted screening strategies and implementing individualized endocrine follow-up protocols for high-risk patients [
62].
4.7. The Significance of the Cumulative Endocrine Burden After COVID-19
The high proportion of patients exhibiting endocrine dysfunctions four years after SARS-CoV-2 infection reflects the systemic and long-lasting impact of COVID-19 on metabolic and immune homeostasis. The finding that nearly one in two individuals presented with an endocrine abnormality, either metabolic or autoimmune, suggests that post-COVID syndrome displays a multisystemic behavior with a marked endocrine tropism. These results are consistent with international observations reporting an increased prevalence of new-onset diabetes and thyroid autoimmunity following SARS-CoV-2 infection [
26,
27]. Prospective studies have shown that systemic inflammation, oxidative stress, and activation of the hypothalamic–pituitary–adrenal axis can disrupt metabolic and immune regulation, triggering persistent autoimmune processes [
14,
53]. The concomitant presence of diabetes and thyroid autoimmunity in approximately 18% of patients supports the hypothesis of a post-COVID immunometabolic phenotype characterized by interrelated endocrine disturbances mediated through shared inflammatory mechanisms. This profile may resemble virus-induced polyautoimmune syndromes described after other infections (e.g., Epstein–Barr virus, cytomegalovirus), but with broader clinical expression due to the multisystemic tropism of SARS-CoV-2 [
45,
63].
From a clinical standpoint, these findings justify the inclusion of systematic endocrine evaluations in post-COVID follow-up protocols, not only to monitor isolated disturbances (such as thyroid or glucose abnormalities), but also to quantify the overall endocrine impact of infection. An integrated surveillance algorithm including fasting glucose, HbA1c, TSH, FT4, and anti-TPO/anti-Tg antibody testing could contribute to the early identification of such abnormalities and to reducing the long-term metabolic and functional burden.
4.8. Study Limitations
This study has several limitations that should be considered when interpreting the results. First, the relatively small sample size of patients re-evaluated (96 out of 1009 initially hospitalized individuals) may limit the statistical power and generalizability of the findings. Although the participation rate (≈9%) is comparable to that of other long-term post-pandemic cohorts, a potential selection bias cannot be excluded. Individuals who agreed to participate in follow-up may have been more health-conscious or symptomatic, which could have resulted in an overestimation of the true prevalence of post-COVID endocrine abnormalities. Second, the monocentric nature of the study limits the external validity of the results, as demographic, genetic, and therapeutic differences across regions could influence outcomes. The absence of a control group composed of individuals without prior SARS-CoV-2 infection precludes direct comparison of the prevalence of new-onset diabetes and thyroid autoimmunity with that in the general population. Nevertheless, indirect comparisons with published international data offer a valuable interpretive framework and reinforce the plausibility of the observed associations. Another limitation is the single follow-up point at four years post-infection, without intermediate assessments (e.g., at 6, 12, or 24 months). It remains uncertain whether the abnormalities detected represent early post-infectious changes that persisted over time or late-onset phenomena arising from chronic inflammatory and autoimmune processes. Additionally, the potential confounding effect of corticosteroid therapy used during severe forms of COVID-19 must be acknowledged. Glucocorticoids, such as dexamethasone, may induce transient hyperglycemia, exacerbate insulin resistance, and accelerate the progression from prediabetes to overt diabetes in susceptible individuals. Given the lack of detailed data on cumulative dose and duration, the potential confounding effect of corticosteroid exposure on the observed association between COVID-19 and diabetes mellitus cannot be entirely excluded [
36].
Furthermore, post-pandemic behavioral changes may represent additional risk factors for metabolic disturbances. Reduced physical activity, weight gain, sleep disruption, unhealthy dietary patterns, and chronic psychological stress may independently contribute to the development of insulin resistance and endocrine dysfunctions [
65].
Despite these limitations, the present study provides a distinctive long-term perspective on endocrine sequelae following SARS-CoV-2 infection, representing one of the longest follow-up periods reported to date. By integrating serological and ultrasonographic assessments of both metabolic and thyroid axes, this work contributes valuable evidence regarding the sustained impact of COVID-19 on endocrine homeostasis and highlights the need for continued multidisciplinary follow-up in affected individuals.
4.9. Future Directions
The results of this study outline several priority areas for future research. To precisely define the temporal trajectory of post-COVID endocrine dysfunctions, prospective studies with periodic assessments of glucose metabolism, thyroid function, and other endocrine axes (adrenal, gonadal) are needed. Expanding research to a multicenter level would allow for more representative findings and enable comparisons across regional, genetic, and therapeutic differences. The implementation of dedicated clinical registries would facilitate the systematic collection of data regarding the prevalence, types, and progression of endocrine disorders, support international comparisons, and inform public health policies aimed at the long-term monitoring of COVID-19 survivors. Based on the current observations, inclusion of fasting glucose, HbA1c, TSH, FT4, and anti-thyroid antibody testing in the standard follow-up protocol for post-COVID patients is warranted. A multidisciplinary approach involving specialists in infectious diseases, endocrinology, immunology, and rehabilitation could optimize early diagnosis and functional recovery in these patients.