Submitted:
14 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Endpoints
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Derakhshan F, Reis-Filho JS. Pathogenesis of Triple-Negative Breast Cancer. Annu Rev Pathol. 2022 Jan 24;17:181-204. PMID: 35073169; PMCID: PMC9231507. [CrossRef]
- Li Y, Zhang H, Merkher Y, Chen L, Liu N, Leonov S, Chen Y. Recent advances in therapeutic strategies for triple-negative breast cancer. J Hematol Oncol. 2022 Aug 29;15(1):121. PMID: 36038913; PMCID: PMC9422136. [CrossRef]
- Harbeck N. Neoadjuvant and adjuvant treatment of patients with HER2-positive early breast cancer. Breast. 2022 Mar;62 Suppl 1(Suppl 1):S12-S16. Epub 2022 Jan 19. PMID: 35148934; PMCID: PMC9097807. [CrossRef]
- Korde LA, Somerfield MR, Carey LA, Crews JR, Denduluri N, Hwang ES, Khan SA, Loibl S, Morris EA, Perez A, Regan MM, Spears PA, Sudheendra PK, Symmans WF, Yung RL, Harvey BE, Hershman DL. Neoadjuvant Chemotherapy, Endocrine Therapy, and Targeted Therapy for Breast Cancer: ASCO Guideline. J Clin Oncol. 2021 May 1;39(13):1485-1505. Epub 2021 Jan 28. PMID: 33507815; PMCID: PMC8274745. [CrossRef]
- O’Rourke H, Hart C, De Boer RH. Current usage of pembrolizumab in triple negative breast cancer (TNBC). Expert Rev Anticancer Ther. 2024 May;24(5):253-261. Epub 2024 Apr 16. PMID: 38594892. [CrossRef]
- Leon-Ferre RA, Goetz MP. Advances in systemic therapies for triple negative breast cancer. BMJ. 2023 May 30;381:e071674. PMID: 37253507. [CrossRef]
- Pusztai L, Denkert C, O’Shaughnessy J, Cortes J, Dent R, McArthur H, Kümmel S, Bergh J, Park YH, Hui R, Harbeck N, Takahashi M, Untch M, Fasching PA, Cardoso F, Zhu Y, Pan W, Tryfonidis K, Schmid P. Event-free survival by residual cancer burden with pembrolizumab in early-stage TNBC: exploratory analysis from KEYNOTE-522. Ann Oncol. 2024 May;35(5):429-436. Epub 2024 Feb 17. PMID: 38369015. [CrossRef]
- Schmid P, Cortes J, Pusztai L, McArthur H, Kümmel S, Bergh J, Denkert C, Park YH, Hui R, Harbeck N, Takahashi M, Foukakis T, Fasching PA, Cardoso F, Untch M, Jia L, Karantza V, Zhao J, Aktan G, Dent R, O’Shaughnessy J; KEYNOTE-522 Investigators. Pembrolizumab for Early Triple-Negative Breast Cancer. N Engl J Med. 2020 Feb 27;382(9):810-821. PMID: 32101663. [CrossRef]
- Schmid P, Cortes J, Dent R, McArthur H, Pusztai L, Kümmel S, Denkert C, Park YH, Hui R, Harbeck N, Takahashi M, Im SA, Untch M, Fasching PA, Mouret-Reynier MA, Foukakis T, Ferreira M, Cardoso F, Zhou X, Karantza V, Tryfonidis K, Aktan G, O’Shaughnessy J; KEYNOTE-522 Investigators. Overall Survival with Pembrolizumab in Early-Stage Triple-Negative Breast Cancer. N Engl J Med. 2024 Nov 28;391(21):1981-1991. Epub 2024 Sep 15. PMID: 39282906. [CrossRef]
- Connors C, Valente SA, ElSherif A, Escobar P, Chichura A, Kopicky L, Roesch E, Ritner J, McIntire P, Wu Y, Tu C, Lang JE. Real-World Outcomes with the KEYNOTE-522 Regimen in Early-Stage Triple-Negative Breast Cancer. Ann Surg Oncol. 2025 Feb;32(2):912-921. Epub 2024 Oct 22. PMID: 39436619; PMCID: PMC11843215. [CrossRef]
- Downs-Canner S, Mittendorf EA. Preoperative Immunotherapy Combined with Chemotherapy for Triple-Negative Breast Cancer: Perspective on the KEYNOTE-522 Study. Ann Surg Oncol. 2023 Jun;30(6):3166-3169. Epub 2023 Mar 10. Erratum in: Ann Surg Oncol. 2023 Jun;30(6):3286. doi: 10.1245/s10434-023-13448-w. PMID: 36897418. [CrossRef]
- Caramelo O, Silva C, Caramelo F, Frutuoso C, Pinto L, Almeida-Santos T. Efficacy of different neoadjuvant treatment regimens in BRCA-mutated triple negative breast cancer: a systematic review and meta-analysis. Hered Cancer Clin Pract. 2022 Sep 9;20(1):34. PMID: 36085046; PMCID: PMC9463858. [CrossRef]
- Bonadio RC, Tarantino P, Testa L, Punie K, Pernas S, Barrios C, Curigliano G, Tolaney SM, Barroso-Sousa R. Management of patients with early-stage triple-negative breast cancer following pembrolizumab-based neoadjuvant therapy: What are the evidences? Cancer Treat Rev. 2022 Nov;110:102459. Epub 2022 Aug 18. PMID: 35998514. [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17(5), 405–424. https://doi.org/10.1038/gim.2015.30.
- Mezzanotte-Sharpe J, Hsu CY, Choi D, Sheffield H, Zelinskas S, Proskuriakova E, Montalvo M, Lee DS, Whisenant JG, Gaffney K, Thompson MS, Blenman K, Tawagi K, Symonds L, Santa-Maria C, Unni N, Quiroga D, Shyr Y, Kennedy LC. Adverse events in patients treated with neoadjuvant chemo/immunotherapy for triple negative breast cancer: results from seven academic medical centers. Breast Cancer Res Treat. 2025 Aug;213(1):71-80. Epub 2025 Jul 4. PMID: 40613977; PMCID: PMC12259778. [CrossRef]
- Andrade MO, Gutierres IG, Tavares MC, de Sousa IM, Balint FC, Marin Comini AC, Gouveia MC, Bines J, Madasi F, Ferreira RDP, Rosa DD, Santos CL, Assad-Suzuki D, de Souza ZS, de Araújo JAP, de Melo Gagliato D, Dos Anjos CH, Zucchetti BM, Ferrari A, de Brito ML, Cangussu R, Fernandes Monteiro MM, Hoff PM, Del Pilar Estevez-Diz M, Testa L, Barroso-Sousa R, Bonadio RC. Immune-related adverse events among patients with early-stage triple-negative breast cancer treated with pembrolizumab plus chemotherapy: real-world data from the Neo-Real/GBECAM 0123 study. Breast. 2025 Oct;83:104473. Epub 2025 Apr 10. PMID: 40240201; PMCID: PMC12496205. [CrossRef]
- El Bairi K, Haynes HR, Blackley E, Fineberg S, Shear J, Turner S, de Freitas JR, Sur D, Amendola LC, Gharib M, Kallala A, Arun I, Azmoudeh-Ardalan F, Fujimoto L, Sua LF, Liu SW, Lien HC, Kirtani P, Balancin M, El Attar H, Guleria P, Yang W, Shash E, Chen IC, Bautista V, Do Prado Moura JF, Rapoport BL, Castaneda C, Spengler E, Acosta-Haab G, Frahm I, Sanchez J, Castillo M, Bouchmaa N, Md Zin RR, Shui R, Onyuma T, Yang W, Husain Z, Willard-Gallo K, Coosemans A, Perez EA, Provenzano E, Ericsson PG, Richardet E, Mehrotra R, Sarancone S, Ehinger A, Rimm DL, Bartlett JMS, Viale G, Denkert C, Hida AI, Sotiriou C, Loibl S, Hewitt SM, Badve S, Symmans WF, Kim RS, Pruneri G, Goel S, Francis PA, Inurrigarro G, Yamaguchi R, Garcia-Rivello H, Horlings H, Afqir S, Salgado R, Adams S, Kok M, Dieci MV, Michiels S, Demaria S, Loi S; International Immuno-Oncology Biomarker Working Group. The tale of TILs in breast cancer: A report from The International Immuno-Oncology Biomarker Working Group. NPJ Breast Cancer. 2021 Dec 1;7(1):150. PMID: 34853355; PMCID: PMC8636568. [CrossRef]
| Value | Variable |
| 48 (IQR 42–55) | Median age (years) |
| 24.6 (IQR 22.1–27.5) | Median BMI (kg/m²) |
| 152 (82.6%) | ECOG 0 |
| 32 (17.4%) | ECOG ≥ 1 |
| 37 (20.1%) | BRCA-mutated |
| 25 (13.6%) | • BRCA1 |
| 12 (6.5%) | • BRCA2 |
| 147 (79.9%) | BRCA wild-type |
| 28 (IQR 15–45) | Median TILs (%) |
| 82 (44.6%) | High TILs (≥ 30%) |
| 102 (55.4%) | Low TILs (< 30%) |
| 137 (74.5%) | ≥ 1 pregnancy |
| 86 (46.7%) | Comorbidities – any |
| 21 (11.4%) | • Thyroid disease |
| 12 (6.5%) | • Autoimmune/inflammatory |
| 5 (2.7%) | • Hypertension |
| 4 (2.2%) | • Gastrointestinal |
| 2 (1.1%) | • Hepatic disease |
| 1 (0.5%) | • Cardiovascular disease |
| 1 (0.5%) | • Respiratory disease |
| 1 (0.5%) | • Renal disease |
| 1 (0.5%) . |
• Psychiatric disorder |
| Variable | BRCA-mutated (n = 37) | BRCA wild-type (n = 147) | p-value |
| Median age, years (IQR) | 46 (41–52) | 49 (43–56) | 0.09 |
| Stage III, n (%) | 11 (29.7%) | 54 (36.7%) | 0.45 |
| Median BMI (kg/m²) | 24.3 (21.9–27.1) | 24.7 (22.2–27.6) | 0.68 |
| ECOG ≥1, n (%) | 5 (13.5%) | 27 (18.4%) | 0.49 |
| High TILs (≥30%), n (%) | 20 (54.1%) | 62 (42.2%) | 0.22 |
| Median Ki-67 (%) | 72 (60–80) | 68 (50–75) | 0.12 |
| Comorbidities – any, n (%) | 15 (40.5%) | 71 (48.3%) | 0.42 |
| Pregnancy ≥1, n (%) | 26 (70.3%) | 111 (75.5%) | 0.54 |
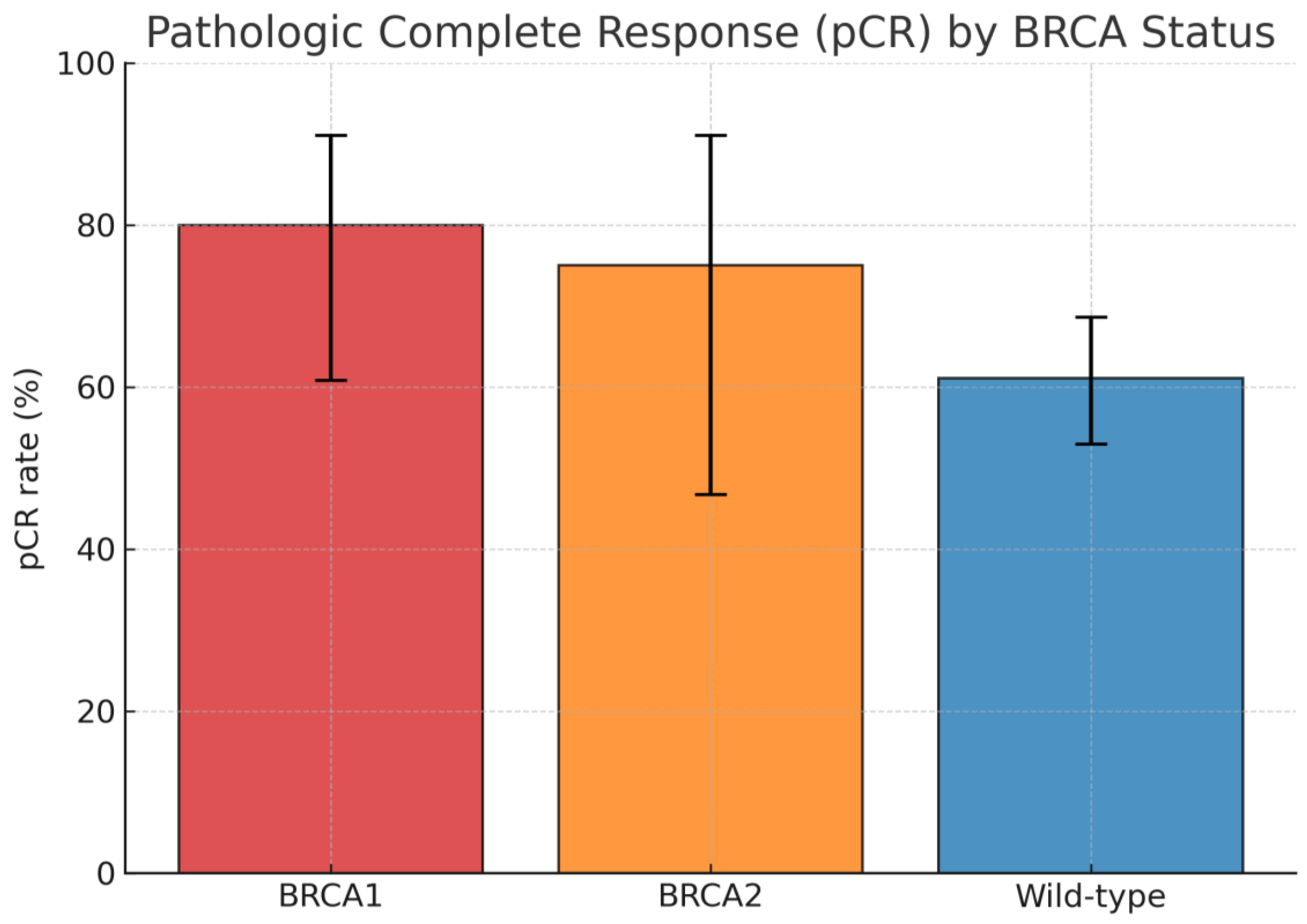
| Group | N | pCR n (%) | 95% Confidence Interval |
| BRCA1 | 25 | 20 (80.0%) | 60.9 – 91.1 |
| BRCA2 | 12 | 9 (75.0%) | 46.8 – 91.1 |
| Wild-type | 144 | 88 (61.1%) | 53.0 – 68.7 |
| BRCA1/2 (combined) | 37 | 29 (78.4%) | 62.8 – 88.6 |
| Variable | Univariate OR (95% CI) | p-value | Multivariate OR (95% CI) | p-value |
| BRCA mutation (mut vs wt) | 2.26 (1.00–5.12) | 0.049 | 2.17 (1.01–4.97) | 0.048 |
| High TILs (≥30%) | 1.98 (1.05–3.75) | 0.034 | 1.75 (0.91–3.41) | 0.09 |
| Stage II vs III | 1.64 (0.87–3.09) | 0.12 | 1.41 (0.73–2.86) | 0.22 |
| Age (continuous) | 0.98 (0.94–1.02) | 0.34 | — | — |
| ECOG ≥1 | 0.79 (0.35–1.78) | 0.57 | — | — |
| BMI (continuous) | 0.97 (0.89–1.04) | 0.39 | — | — |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




