Submitted:
14 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
2.3. Treatment
2.4. Activity Evaluation
2.5. Statistics
3. Results
3.1. Patient Population
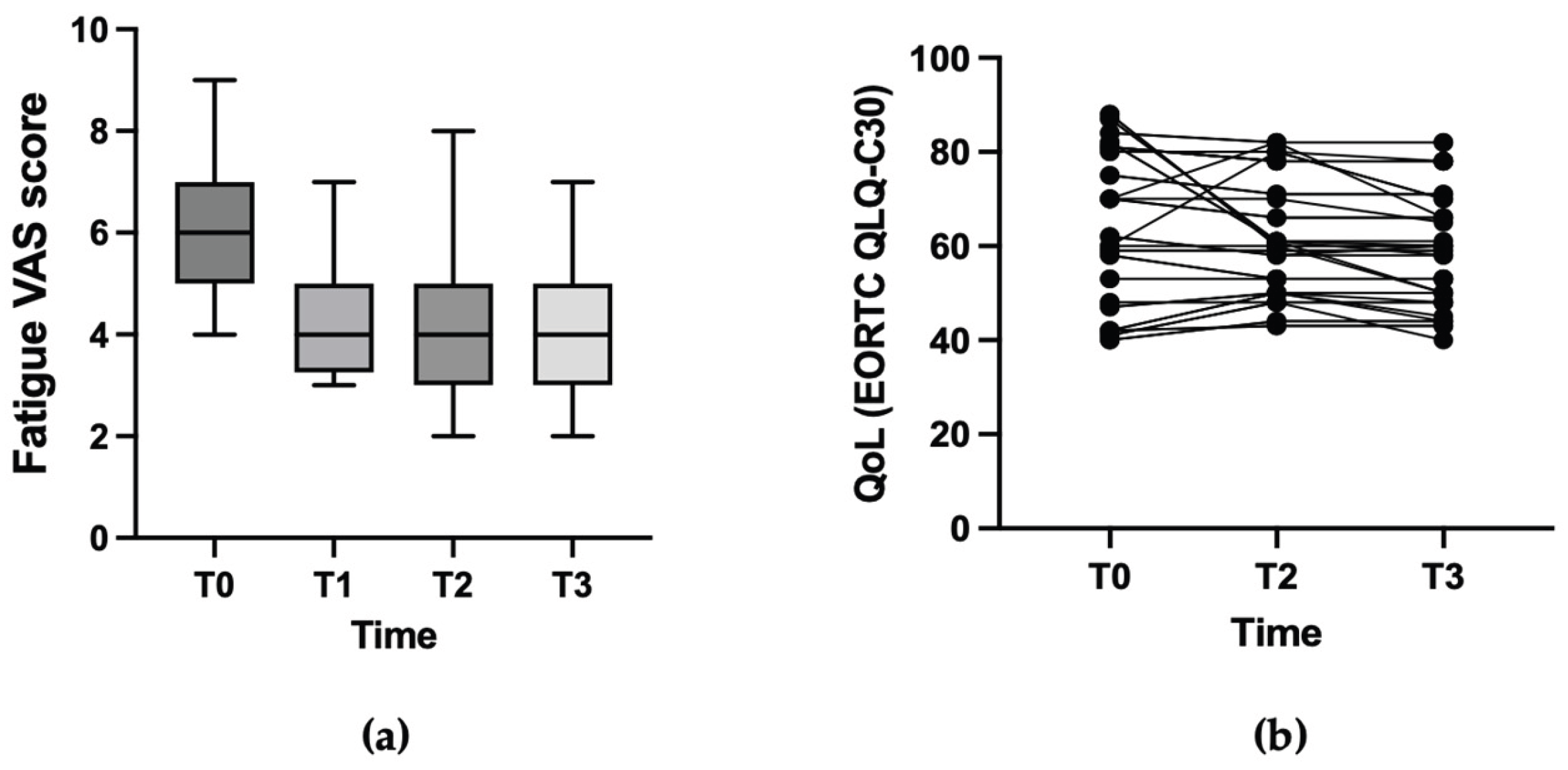
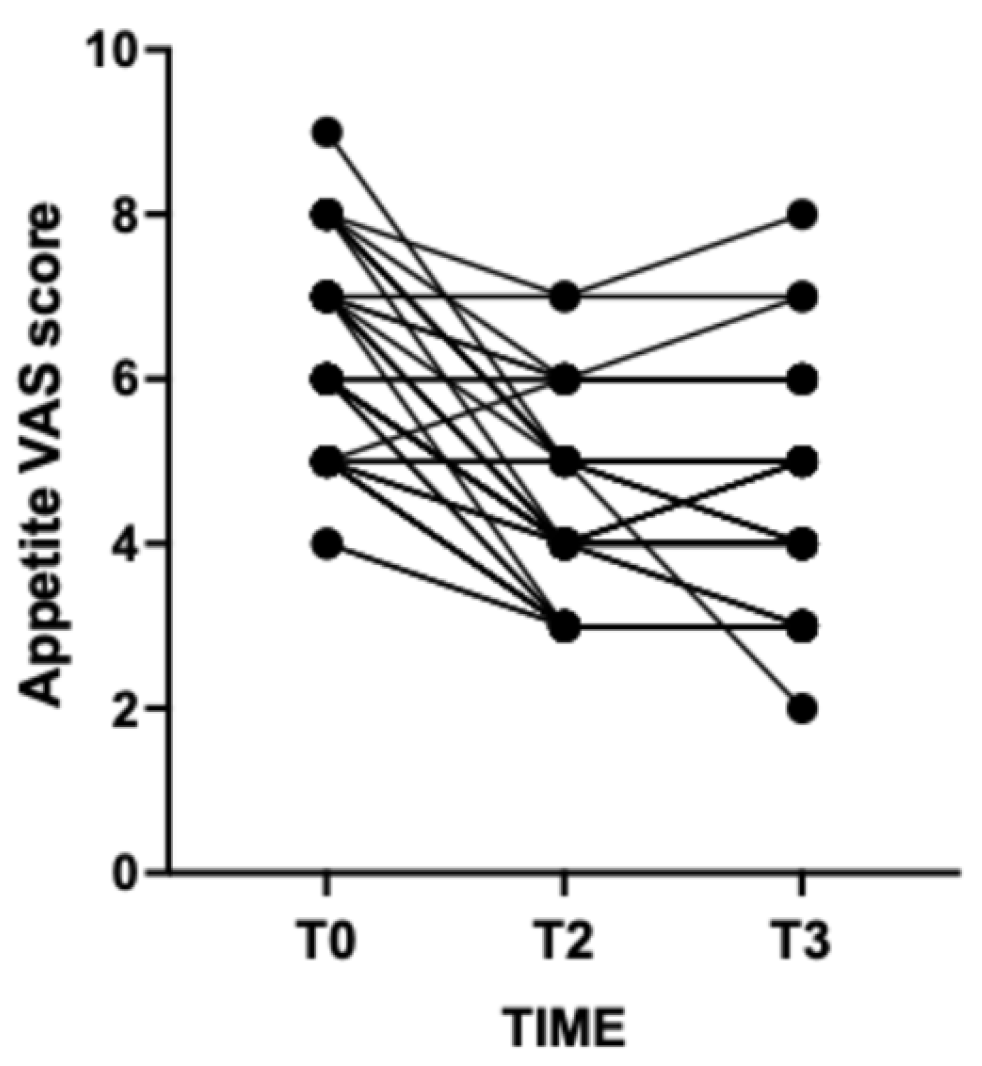
3.2. Activity of L-Arginine/Vitamin C
3.3. Safety
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CRF | Cancer-Related Fatigue |
| l-Arg | Levo-Arginine |
| HR-QoL | Health-Related Quality of Life |
| VAS | Visual Analog Scale |
References
- Curt, G.A.; Breitbart, W.; Cella, D.; Groopman, J.E.; Horning, S.J.; Itri, L.M.; Johnson, D.H.; Miaskowski, C.; Scherr, S,L,; Portenoy, R.K.; Vogelzang, N.J. Impact of cancer-related fatigue on the lives of patients: new findings from the Fatigue Coalition. Oncologist, 2000; 5, 353–360.
- Kang, Y.E.; Yoon, J.H.; Park, N.H.; Ahn, Y.C.; Lee, E.J.; Son, C.G. Prevalence of cancer-related fatigue based on severity: a systematic review and meta-analysis. Sci. Rep. 2023, 13, 12815. [Google Scholar] [CrossRef] [PubMed]
- NCCN Cancer-related fatigue version 2, 2025. www.nccn.org/guidelines.
- Stone, P.; Candelmi, D.E.; Kandola, K.; Montero, L.; Smetham, D.; Suleman, S.; Fernando, A.; Rojí, R. Management of Fatigue in Patients with Advanced Cancer. Curr. Treat. Options Oncol. 2023, 24, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, P.L.; Morales, J.S.; Emanuele, E.; Pareja-Galeano, H.; Lucia, A. Supplements with purported effects on muscle mass and strength. Eur. J. Nutr. 2019, 58, 2983–3008. [Google Scholar] [CrossRef] [PubMed]
- Ling, Z.N.; Jiang, Y.F.; Ru, N.; Lu, J,H,; Ding, B.; Wu, J.Amino acid metabolism in health and disease. Signal Transduct. Target Ther. 2023, 8, 345. [CrossRef] [PubMed]
- Rose, A,J. Amino Acid Nutrition and Metabolism in Health and Disease. Nutrients. 2019, 11, 2623. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.I.; La Bounty, P.M.; Roberts, M. he ergogenic potential of arginine. J. Int. Soc. Sports Nutr. 2004, 1, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Lind, D.S. Arginine and cancer. J. Nutr. 2004, 134 (10 Suppl), 2837S–2841S. [Google Scholar] [CrossRef] [PubMed]
- Szefel, J.; Danielak, A.; Kruszewski, W.J. Metabolic pathways of L-arginine and therapeutic consequences in tumors. Adv. Med. Sci. 2019, 64, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, P.C.; Quiceno, D.G.; Ochoa, A.C. L-arginine availability regulates T- lymphocyte cell-cycle progression. Blood, 2007; 109, 1568–73. [Google Scholar]
- Bronte, V.; Zanovello, P. Regulation of immune responses by L-arginine metabolism. Nat. Rev. Immunol, 2005; 5, 641–54. [Google Scholar]
- Geiger, R.; Rieckmann, J.C.; Wolf, T.; et al. L-Arginine Modulates T Cell Metabolism and Enhances Survival and Anti-tumor Activity. Cell, 2016; 167, 829–42. [Google Scholar]
- Jean-Pierre, P.; Figueroa-Moseley, C.D.; Kohli, S.; Fiscella, K., Palesh, O.G. Morrow GR. Assessment of cancer-related fatigue: implications for clinical diagnosis and treatment. Oncologist. 2007;12 Suppl 1:11-21.
- Di Meglio, A.; Charles, C.; Martin, E.; Havas, J.; Gbenou, A.; Flaysakier, J.D.; Martin, A.L.; Everhard, S.; Laas, E.; Chopin, N.; Vanlemmens, L.; Jouannaud, C.; Levy, C.; Rigal, O.; Fournier, M.; Soulie, P.; Scotte, F.; Pistilli, B.; Dumas, A.; Menvielle, G.; André, F.; Michiels, S.; Dauchy, S.; Vaz-Luis, I. Uptake of Recommendations for Posttreatment Cancer-Related Fatigue Among Breast Cancer Survivors. J. Natl. Compr. Canc. Netw, 2022; 7, 20(13). [Google Scholar]
- Schmidt, M.E.; Bergbold, S.; Hermann, S.; Steindorf, K. Knowledge, perceptions, and management of cancer-related fatigue: the patients’ perspective. Support. Care. Cancer. 2021, 29, 2063–2071. [Google Scholar] [CrossRef] [PubMed]
- Ragni, M.; Fornelli, C.; Nisoli, E.; Penna, F. Amino Acids in Cancer and Cachexia: An Integrated View. Cancers (Basel). 2022, 14, 5691. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.D.P.; Howell, S.L.; Teixeira, F.J. Pimentel GD. Dietary Amino Acids and Immunonutrition Supplementation in Cancer-Induced Skeletal Muscle Mass Depletion: A Mini-Review. Curr. Pharm. Des, 2020; 26, 970–978. [Google Scholar]
- Muscaritoli, M.; Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Hütterer, E.; Isenring, E.; Kaasa, S.; Krznaric, Z.; Laird, B.; Larsson, M.; Laviano, A.; Mühlebach, S.; Oldervoll, L.; Ravasco, P.; Solheim, T.S.; Strasser, F.; de van der Schueren, M.; Preiser, J.C.; Bischoff, S.C. ESPEN practical guideline: Clinical Nutrition in cancer. Clin. Nutr. 2021, 40, 2898–2913. [Google Scholar] [CrossRef] [PubMed]
- Paddon-Jones D, Børsheim E, Wolfe RR. Potential ergogenic effects of arginine and creatine supplementation. J Nutr. 2004, 134 (10 Suppl), 2888S-2894S; discussion 2895S.
- Buijs, N.; Luttikhold, J.; Houdijk, A.P.; van Leeuwen, P.A. The role of a disturbed arginine/NO metabolism in the onset of cancer cachexia: a working hypothesis. Curr. Med. Chem, 2012; 19, 5278-86. [Google Scholar]
- Fiorentino, G.; Coppola, A.; Izzo, R.; Annunziata, A.; Bernardo, M.; Lombardi, A.; Trimarco, V.; Santulli, G.; Trimarco, B. Effects of adding L-arginine orally to standard therapy in patients with COVID-19: A randomized, double-blind, placebo-controlled, parallel-group trial. Results of the first interim analysis. EClinicalMedicine, 2021; 40, 101125. [Google Scholar]
- Muralidharan, J.; Kashyap, S.P.; Jacob, M.; Ollapally, A.; Idiculla, J.; Raj, J.M.; Thomas, T.; Kurpad, A.V. The effect of l-arginine supplementation on amelioration of oxygen support in severe COVID-19 pneumonia. Clin. Nutr. ESPEN. 2022; 52, 431–435. [Google Scholar]
- Turcu-Stiolica, A.; Ionele, C.M.; Ungureanu, B.S.; Subtirelu, M.S. The Effects of Arginine-Based Supplements on Fatigue Levels following COVID-19 Infection: A Prospective Study in Romania. Healthcare (Basel). 2023, 11, 1477. [Google Scholar] [CrossRef] [PubMed]
- Arribas-López, E.; Zand, N.; Ojo, O.; Snowden, M.J.; Kochhar, T. The Effect of Amino Acids on Wound Healing: A Systematic Review and Meta-Analysis on Arginine and Glutamine. Nutrients. 2021, 13, 2498. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Li, N.; Zhang, J.; Fu, Q.; Qiu, Y.; Chen, Y. The Effect of immunonutrition in patients undergoing pancreaticoduodenectomy: a systematic review and meta-analysis. BMC. Cancer. 2023, 23, 351. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Vissers, M.C.; Cook, J.S. The effect of intravenous vitamin C on cancer- and chemotherapy-related fatigue and quality of life. Front. Oncol. 2014, 4, 283. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Li, H.; Chiu, Y.S.; Huang, C.C.; Chen, W.C. Supplementation of L-Arginine, L-Glutamine, Vitamin C, Vitamin E, Folic Acid, and Green Tea Extract Enhances Serum Nitric Oxide Content and Antifatigue Activity in Mice. Evid. Based. Complement. Alternat. Med. 2020, 2020, 2020, 8312647. [Google Scholar] [CrossRef] [PubMed]
- Chitapanarux, I.; Traisathit, P.; Chitapanarux, T.; Jiratrachu, R.; Chottaweesak, P.; Chakrabandhu, S.; Rasio, W.; Pisprasert, V.; Sripan, P. Arginine, glutamine, and fish oil supplementation in cancer patients treated with concurrent chemoradiotherapy: A randomized control study. Curr. Probl. Cancer. 2020, 44, 100482. [Google Scholar] [CrossRef] [PubMed]
- Saligan, L.N.; Lukkahatai, N.; Zhang, Z.J.; Cheung, C.W.; Wang, X.M. Altered Cd8+ T lymphocyte Response Triggered by Arginase 1: Implication for Fatigue Intensification during Localized Radiation Therapy in Prostate Cancer Patients. Neuropsychiatry (London). 2018, 8, 1249–1262. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Number | Percent |
|---|---|---|
| Patients | 44 | 100% |
| N. of centers | ||
| Median age and range (years). | 59 (28-79) | NA |
| Sex (male/female) | 20/24 | 45%/55% |
|
ECOG performance status (median, range) |
1 (1-2) |
NA |
|
Primary tumor Breast Colorectal Kidney Prostate Lung |
20 4 2 8 10 |
45% 9% 5% 18% 23% |
| Disease status Disease-free* Metastatic |
18 26 |
41% 59% |
| Anticancer therapy** Chemotherapy Chemoradiotherapy Chemoimmunotherapy TKI CDKI ARSI |
24 27 2 3 4 4 7 |
54.5% 4.5% 6.8% 9.1% 9.1% 16% |
|
Regimens Epirubicin/cyclophosphamide → paclitaxel + trastuzumab + pertuzumab Carboplatin/Paclitaxel Cisplatin/vinorelbine Pemetrexed/platinum salts Capecitabine/oxaliplatin Nivolumab + cabozantinib Osimertinib Docetaxel Enzalutamide + LHRH CDKI + aromatase inhibitor or fulvestrant |
10 3 4 4 4 2 2 4 7 4 |
23% 7% 9% 9% 9% 5% 5% 9% 16% 9% |
| Basal fatigue (VAS) | 6.3 (range 4-9) | |
| Basal HR-QoL (median) | 64.3 (range 40-88). | |
| Appetite VAS score (median) | 6.27 (range 4-9). | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




