2. Materials and Methods
2.1. Study Design
In this observational, single-center, open-label study, patients of both sexes with a diagnosis of knee osteoarthritis and referred to the pain room of the Clinical Pharmacology and Pharmacovigilance Unit of the “R Dulbecco” University Hospital of Catanzaro from January 2025 to September 2025, were enrolled. During this period, treatment with the fixed combination of a new nutraceutical (1 tablet daily for 3 months; Diaco Biofarmaceutici, Trieste, Italy) was added to their common therapy. At the beginning of the study, patients were asked not to change their usual dietary habits or any other medications used for their comorbidities.
2.2. Ethical Considerations
To ensure participant privacy, each subject was assigned a numerical identification code generated by a physician who was not involved in the study. All participants were fully informed about the study’s purpose and procedures and provided written informed consent prior to enrollment. The study was conducted in accordance with the principles of Good Clinical Practice and the Declaration of Helsinki, with full compliance with Italian privacy regulations. Ethical approval was obtained from the Institutional Ethics Committee (Authorization No. 120/2018; Clinical trial registration: NCT05509075).
2.3. Inclusion and Exclusion Criteria
Patients were enrolled according to the following inclusion criteria:
Age ≥ 18 years, of either sex.
Diagnosis of knee osteoarthritis (OA).
Ability to comply with the study protocol and provision of written informed consent.
Exclusion criteria included:
a) Age < 18 years.
b) Pregnancy or breastfeeding, or women of childbearing potential not using adequate contraception.
c) Known allergy or hypersensitivity to the study treatment or rescue medications.
d) Advanced-stage malignancies.
e) Moderate to severe renal impairment (glomerular filtration rate < 30 mL/min).
f) Severe hepatic or cardiac dysfunction.
g) Severe asthma.
h) History of drug or alcohol abuse.
i) Any condition or comorbidity that, in the investigator’s judgment, could pose a risk to the participant or interfere with the evaluation of efficacy and safety.
j) Participation in another clinical trial or receipt of an investigational drug within 30 days before screening.
2.4. Experimental Protocol
Clinical data were collected at two main time points: at enrollment (T0, prior to treatment initiation) and at the end of the study (T3, three months after T0). All patients presented with chronic pain and were receiving NSAIDs as needed; therefore, baseline data at T0 served as the control condition.
Questionnaires were administered by the study’s medical staff. Given the open-label design, patient confidentiality was maintained through the assignment of unique numerical codes by a physician not involved in the study. This ensured privacy while enabling accurate data analysis in compliance with ethical research standards.
At both T0 and T3, detailed medical history was collected, physical examinations were performed, and standardized questionnaires were completed. Pain intensity and functional status were assessed using the Numeric Rating Scale (NRS) and the Knee Injury and Osteoarthritis Outcome Score (KOOS). Throughout the study, any systemic or local adverse drug reactions (ADRs) were monitored and evaluated using the Naranjo scale to determine causality.
Prior to enrolling in this study, all patients had received systemic treatment with NSAIDs, without achieving any clinical improvement. Consequently, the pretreatment period (T0) was used as the control for comparison with follow-up assessments.
2.4.1. Questionnaires
Validated instruments were employed, consistent with previous studies:
36-Item Short Form Health Survey (SF-36): Assesses health-related quality of life across eight domains. Higher scores indicate better perceived health status, whereas lower scores reflect poorer quality of life.<sup>17,18</sup>
Zung Self-Rating Anxiety Scale (Zung SAS): A 20-item questionnaire assessing anxiety, categorized as normal (0–44), moderate (45–59), or severe (60–80).<sup>19</sup>
Zung Self-Rating Depression Scale (Zung SDS): A 20-item instrument measuring depressive symptoms, classified as normal (20–49), mild (50–59), moderate (60–69), or severe (70–80).<sup>20</sup>
Adverse Drug Reaction Probability Scale (Naranjo Scale): A 10-item tool used to standardize the assessment of ADR causality. Scores classify ADRs as doubtful (≤0), possible (1–4), probable (5–8), or definite (≥9).<sup>21,22</sup>
Knee Injury and Osteoarthritis Outcome Score (KOOS): Evaluates knee pain, symptoms (e.g., swelling, range of motion), activities of daily living, sport/recreation function, and knee-related quality of life. Scores range from 0 (extreme problems) to 100 (no problems).
2.4.2. Clinical Tests
Clinical assessments were performed according to established protocols:
Timed Up and Go (TUG) Test: Evaluates functional mobility and fall risk by timing how long it takes for a patient to rise from a chair, walk three meters, turn, return, and sit down. Shorter times indicate better mobility, whereas times ≥13.5 seconds indicate increased fall risk.
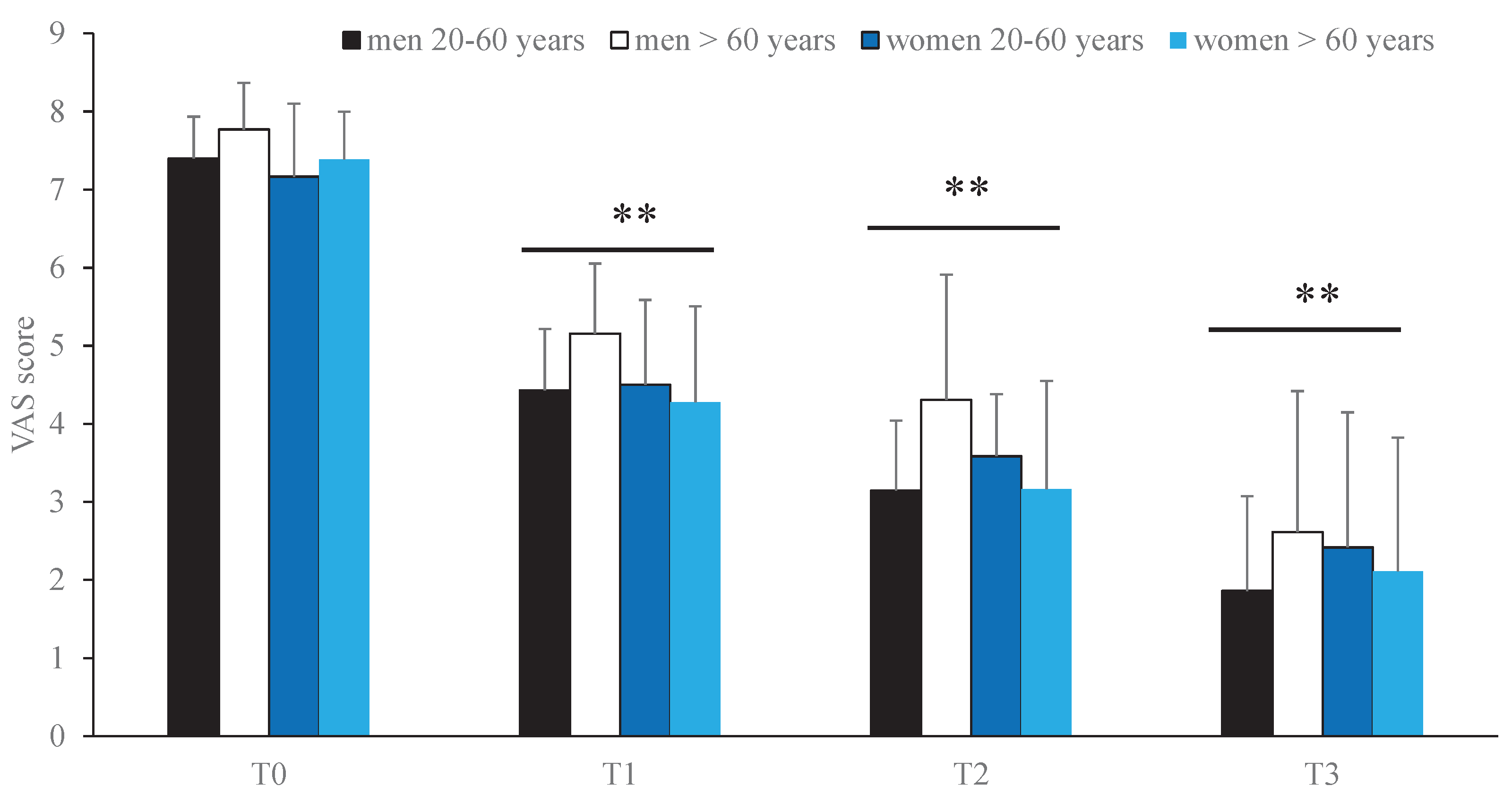
Visual Analogue Scale (VAS): Measures pain intensity on a 10-cm line, where 0 represents “no pain” and 10 represents “worst imaginable pain.” The onset of pain during the TUG test was also recorded.
2.5. Efficacy End Points
Primary endpoint:
Statistically significant improvement (p < 0.05) in KOOS scores at follow-up visits (T1–T3) compared with baseline (T0).
Secondary endpoints:
Statistically significant improvement (p < 0.05) in VAS scores at T1–T3 compared with T0.
Statistically significant improvement (p < 0.05) in overall SF-36 scores at T1–T3 compared with T0.
Statistically significant changes (p < 0.05) in mood disorder scores (Zung SAS and SDS) between T1–T3 and T0.
2.6. Safety End-Points
ADRs related to the nutraceutical treatment were recorded throughout the study using the Naranjo scale. ADRs leading to participant withdrawal were also documented.
2.7. Nutraceutical Formulation
Each tablet contained a fixed combination of chondroitin sulfate, α-lipoic acid, astaxanthin, lycopene, escin, omega-3 fatty acids, eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) (Diaco Biofarmaceutici, Trieste, Italy;
Table 1).
2.8. Statistical Analysis
Continuous variables with a Gaussian distribution were described as mean ± standard deviation (SD), whereas categorical variables were summarized as counts and percentages. The normality of continuous variables was assessed using the Shapiro–Wilk test. Comparisons of continuous variables were performed using the Student’s t-test or one-way analysis of variance (ANOVA), as appropriate. Categorical variables were compared using the chi-square test.
Multivariate analyses were adjusted for potential confounders, including age, sex, smoking status, alcohol consumption, physical activity, educational level, body mass index (BMI), comorbidities, and concomitant drug use. Correlations between continuous variables were assessed using Pearson’s correlation coefficient.
For repeated measures, the nonparametric Friedman test was used, followed by post hoc pairwise comparisons with the Wilcoxon signed-rank test. Data are presented as mean ± SD unless otherwise specified. Statistical significance was set at p < 0.05. All analyses were conducted using SPSS software, version 22.0 (IBM Corp., Armonk, NY, USA).
Because no prior data were available for this novel compound, a formal power calculation could not be performed; therefore, this investigation was designed as a pilot study.