Submitted:
13 November 2025
Posted:
17 November 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Space/Flight Hazards
2.1. Impact of Microgravity on Human Physiology and Health
2.1.1. Effects of Microgravity on the Cardiovascular System
2.1.2. Effects of Microgravity on the Hematological System
2.1.3. Effects of Microgravity on the Musculoskeletal System
2.1.4. Effects of Microgravity on the Neurologic System
2.1.5. Effects of Microgravity on the Gastrointestinal System and Gut Microbiome
2.2. Impact of Radiation on Human Physiology and Health
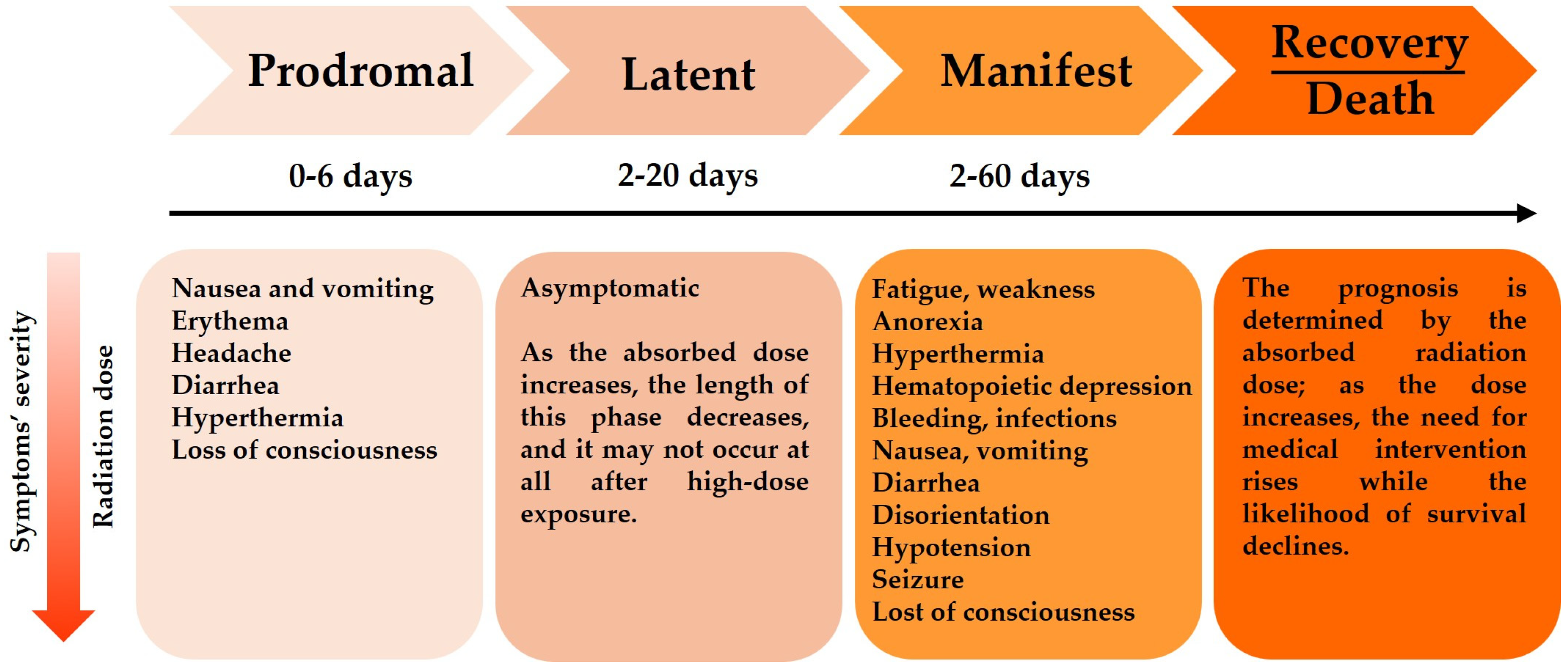
2.2.1. Acute Radiations Syndromes
2.2.2. Effects of Space Radiation on the Cardiovascular System
2.2.3. Carcinogenesis Induced by Space Radiation
2.2.4. Effects of Space Radiation on the Central Nervous System
2.3. Other Risk for Human Health Induced by Space Exposome
2.3.1. Immunological Dysfunctions Following Spaceflights
2.3.2. Neuro-Behavioural Alterations During Spaceflights
2.3.3. Dermatologic Alterations During Spaceflights
3. Pharmacological Interventions in Space
3.1. Medical Care in Spaceflight
| Category | API (Active Pharmaceutical Ingredients) | Main Uses in Space |
| Motion sickness | Scopolamine; Promethazine; Meclizine; Dimenhydrinate | Prevention and treatment of space motion sickness (nausea, vomiting, dizziness) due to adaptation to microgravity |
| Pain management | Acetaminophen (Paracetamol); Ibuprofen; Aspirin; Tramadol; Oxycodone | Relief of muscle pain, headache, joint or exercise-related pain, and minor acute or chronic pain |
| Sleep aids/Alertness | Zolpidem; Melatonin; Diphenhydramine; Modafinil; Caffeine | Regulation of disturbed circadian rhythms; sleep promotion; alertness maintenance during long shifts or after disrupted sleep |
| Respiratory/allergy | Loratadine; Cetirizine; Pseudoephedrine; Fluticasone; Albuterol (Salbutamol) | Management of allergies, nasal congestion, respiratory irritation, and cough in the closed spacecraft environment |
| Gastrointestinal | Omeprazole; Ranitidine; Loperamide; Ondansetron; Metoclopramide | Treatment of nausea, reflux, diarrhea, constipation, and other digestive disturbances linked to microgravity and space diet |
| Anti-infectives | Amoxicillin; Ciprofloxacin; Azithromycin; Mupirocin; Clotrimazole | Prevention and treatment of bacterial, skin, or urinary infections during space missions |
| Anti-inflammatory / Hormonal | Prednisone; Hydrocortisone; Dexamethasone; Naproxen | Management of acute inflammation, allergic reactions, edema; modulation of immune response in-flight |
| Chronic conditions | Levothyroxine; Insulin; Amlodipine; Metoprolol; Sertraline | Management of preexisting chronic conditions (hypertension, diabetes, hypothyroidism, anxiety/depression) during long-duration missions |
3.2. Stability and Degradation of Pharmaceutical Compounds in Space
3.3. Pharmacokinetics and Pharmacodynamics in Altered Physiology
3.4. In-Situ Pharmaceutical Manufacturing
3.5. Regulatory and Operational Challenges
4. Role of Medicinal Plants in Space Pharmacy
4.1. Therapeutic Potential of Phytochemicals in Space Medicine
4.1.1. Use of Medicinal Plants as Alternative Medication for Sleep Disorders
4.1.2. Use of Medicinal Plants as Alternative Medication for Pain
4.1.3. Use of Medicinal Plants as Alternative Medication for Space Motion Sickness
| Ailment | Medicinal plant | Main active ingredients | Pharmacological effects | Clinical studies |
| Sleep disturbance | Valeriana officinalis L. | Sesquiterpenes (valerenic acid), valepotriates, alkaloids [342,343] | Hypnotic, antioxidant, antimicrobial, anti-inflammatory, sedative, anxiolytic, spasmolytic, anticonvulsant, cytoprotective, neuroprotective activity [343] | [344,345,346,347,348,349,350,351] |
| Crocus sativus | Crocin, safranal and picrocrocin [352] | Hypnotic, antioxidant, anti-inflammatory, anxiolytic, antidepressive, antiepileptogenic, neuroprotective activity [353,354] | [355,356,357,358] | |
| Lavandula angustifolis Mill | Linalool, linalyl acetate [359] | Hypnotic, analgesic, stress relieving, anxiolytic, anti-inflammatory activity [360,361] | [362,363,364,365,366,367,368,369,370] | |
| Melissa officinalis L. | Volatile compounds, triterpenes, phenolic acids, and flavonoids [371] | Antioxidant, anti-inflammatory, hypnotic, antidepressive, neuroprotective, nootropic activity [372,373] | [374,375,376,377,378,379] | |
| Pain | Capsinum annum L. | Capsaicin, carotenoids [380] | Analgesic, antioxidant, anti-inflammatory, antifungal, antimicrobial, gastroprotective, antihyperlipidemic, immunomodulatory activity [380] | [381,382,383,384,385,386] |
| Curcuma longa L. | Curcumin, demethoxycurcumi, and bisdemethoxycurcumin [387] | Analgesic, antioxidant, anti-inflammatory, antimicrobial, anti-diabetic, hepatoprotective activity [388,389] | [390,391,392,393] | |
| Zingiber officinale Roscoe | Phenolic compounds (gingerols, shogaols, paradols, zingerone), terpenes (zingiberene, α-curcumene, β-sesquiphellandrene) [394] | Analgesic, antiarthritic, anti-inflammatory, antioxidant, gastroprotective, hepatoprotective activity [395] | [396,397,398,399] | |
| Willow bark | Salicin, flavonoids, tannins proanthocyanidins [400] | Analgesic, antiarthritic, anti-inflammatory, antimicrobial activity [400] | [401,402,403,404] | |
| Harpagophytum procubens (Burch.) DC. | Iridoid glycosides (harpagoside, harpagide, procumbide, 8-O-p-Coumaroylharpagide) [405] | Analgesic, antioxidant, anti-inflammatory, antimicrobial, anti-diabetic activity [406] | [407,408,409,410,411,412] | |
| Boswellia serrata Roxb | Boswellic acid [413] | Analgesic, antiarthritic, anti-inflammatory, antioxidant, anticancer, neuroprotective activity [413] | [414,415,416,417,418] | |
| SMS | Zingiber officinale Roscoe | Phenolic compounds (gingerols, shogaols, paradols, zingerone), terpenes (zingiberene, α-curcumene, β-sesquiphellandrene) [394] | Analgesic, antiarthritic, anti-inflammatory, antioxidant, gastroprotective, hepatoprotective activity [395] | [419,420,421,422,423] |
| Skin diseases | Aloe vera (L.) Burm. f. | Polysaccharides (acemannan), anthraquinones, enzymes, vitamins, minerals [424] | Antioxidant, wound-healing modulatory, immunomodulatory, anti-inflammatory, antimicrobial, gastroprotective [424,425] | [426,427,428,429,430,431] |
| Curcuma longa L. | Curcumin, demethoxycurcumi, and bisdemethoxycurcumin [387] | Analgesic, antioxidant, anti-inflammatory, antimicrobial, anti-diabetic, hepatoprotective activity [388,389] | [432,433,434,435] | |
| Ailment | Medicinal plant | Main active ingredients | Clinical studies | |
| Skin diseases | Calendula officinalis L. | Triterpenoids, flavonoids, saponins, carotenoids, and essential oils [436] | Wound-healing modulatory, anti-inflammatory, antioxidant, antimicrobial, anti-fungal, anti-cancer and analgesic activity [436] | [437,438,439,440,441] |
| Camellia sinensis (L. Kuntze | Polyphenols (epigallocatechin gallate), purine alkaloids [442] | Antioxidant, anticancer, antidiabetic, neuroprotective, immunomodulatory activity [442,443] | [444,445,446,447] | |
| Hypericum perforatum L. | Naphthodianthrones (hypericin), phloroglucinols (hyperforin), flavonoids (rutin, quercetin, hyperoside) [448,449] | Wound-healing modulatory, antioxidant, anti-inflammatory, antimicrobial, anticancer and antidepressant [448,450] | [451,452,453,454] | |
| Stress | Panax ginseng Meyer | Triterpene saponins (ginsenosides), polysaccharides, peptides, alkaloids, polyacetylenes, phenolic compounds [455] | Anti-inflammatory anti-fatigue, antioxidant, immunomodulatory, nootropic, neuro-protective cardioprotective activity [456] | [457,458,459,460,461,462,463] |
| Rhodiola rosea L. | Phenylpropanoids (rosavins), phenylethanoid derivatives (salidroside and tyrosol), flavonoids, monoterpenes, triterpenes, phenolic acids [464,465] | Anti-fatigue, antioxidant, anti-inflammatory, cardioprotective, neuroprotective, anxiolytic, antidepressant and nootropic activity [465]. | [466,467,468,469,470,471,472,473] | |
| Withania somnifera (L.) Dunal | Steroidal lactones (withanolides), alkaloids, sitoindosides, flavonoids, saponins [474] | Anti-inflammatory, antioxidant, anxiolytic, immunomodulatory, neuroprotective, antitumoral and anti-fatigue [475,476,477] | [478,479,480,481,482,483,484,485,486,487,488,489] |
4.1.4. Use of Medicinal Plants as Alternative Medication for Skin Diseases
4.1.5. Potential Use of Adaptogens to Increase Resilience
4.2. Phyto-Biomanufacturing: Synthetic Pharmacognosy for In Situ Production of Active Compounds
4.3. Psychological and Environmental Benefits for Astronauts
5. Challenges and Future Perspectives
- Beyond traditional claims, the therapeutic use of plant extracts requires rigorous clinical studies to establish both efficacy and safety [534,535]. It is important to note that clinical studies conducted on Earth typically involve either the general population or specific subpopulations of diseased individuals [535]. In contrast, astronauts represent a highly specialized population, and due to the physiological alterations associated with spaceflight, the effectiveness of plant-derived therapies remains to be determined [536]. Future preliminary testing in astronauts during spaceflight are then welcome.
- Studies of plant extracts—both preclinical and clinical—have often shown inconsistent findings in efficacy, dosing, and side effects. These inconsistencies largely stem from variations in plant matrix, cultivation practices, environmental conditions and extraction techniques [537]. Standardization of the entire production process is thus critical to ensure batch-to-batch consistency in terms of active compounds concentrations and reliable pharmacological outcomes, on Earth and ever more so in space.
-
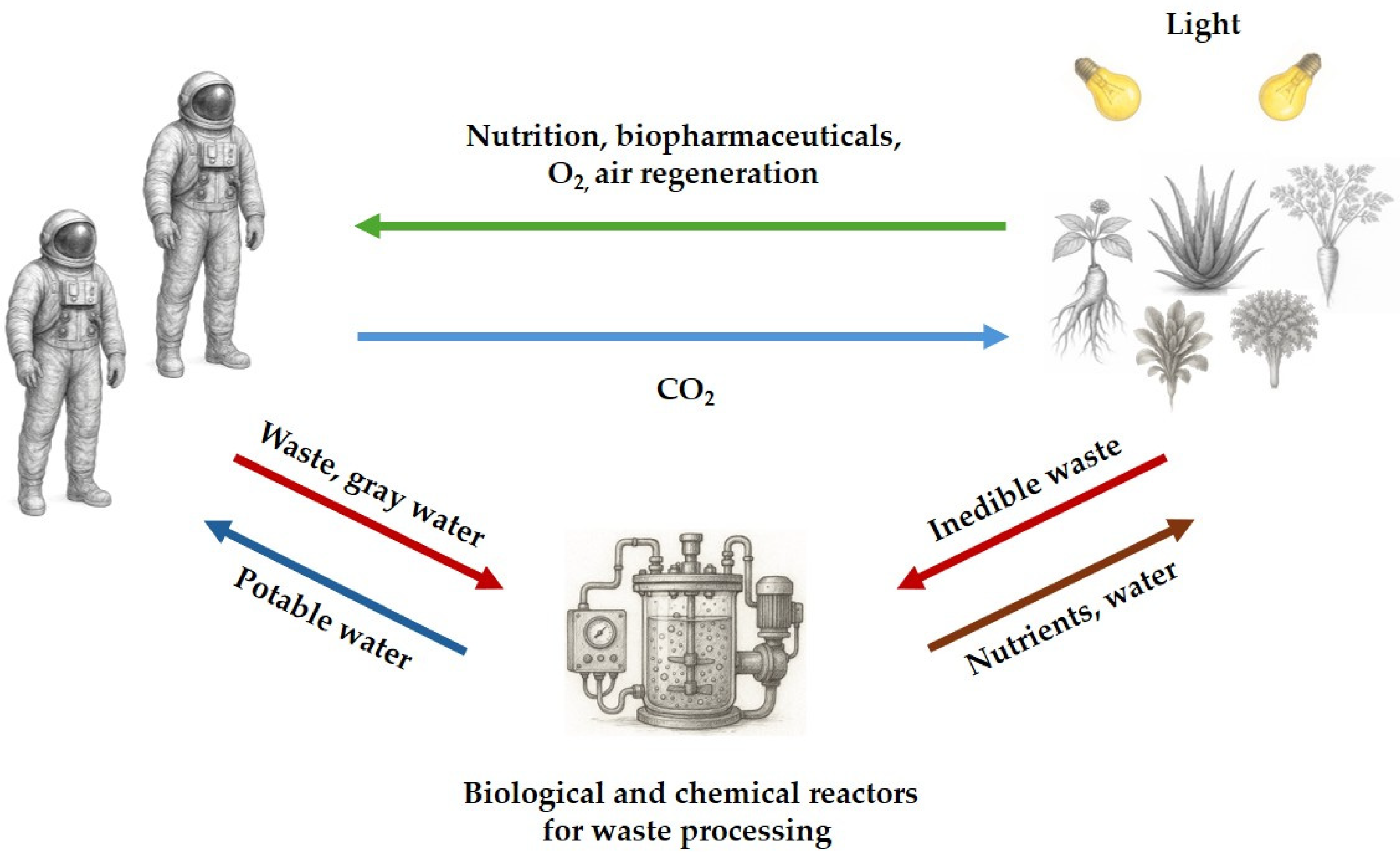
There is also the strategic decision of whether to rely on Earth-manufactured plant preparations or develop the capacity for on-demand production in space. It may mainly depend on the target of the mission and its duration. In short-duration spaceflights or during the early phases of extended missions, the use of Earth-prepared formulations appears to be the more practical and preferred option. It is important to note that, similar to conventional pharmaceuticals [311], plant extracts are susceptible to degradation. However, the stability of plant-derived compounds in space environments remains largely uncharacterized. To fill this gap, future studies should be finalized to verify physical and chemical stability of plant extracts directly in space [538] or in ground-based spaceflight analogues [539]. In the meantime, medicinal extracts may benefit from current strategies under investigation to improve and prolong drug stability in space, such as: the use or radioprotective packaging materials (i.e. high-density polyethylene composites) [7,311,540]; the storage of extracts at low temperatures (≤80°C), since it has been found that cold preserve pharmaceuticals from radiation-induced damages [541,542]. Moreover, it has been demonstrated that some excipients and antioxidants provide protective effects to medicines exposed to radiation [311]; it is then conceivable that the presence of antioxidant-active constituents in medicinal plant extracts mitigates the detrimental influence of space radiation.At variance, in the context of extended space exploration and off-Earth settlements, the autonomous and continuous production of fresh plant material constitutes an asset of considerable strategic importance. Predictably, the magnitude of the challenge is proportional to the importance of the outcome. Within this framework, the possibility of growing plants in the space environment for human alimentation is the most studied aspect, specifically in terms of feasibility [14,15,543]. Physical and storage constraints in both spacecraft and planetary colonies, as well as the costs of transporting materials into space, pose critical problems in space agriculture. Consequently, closed-loop cultivation systems are being designed and studied that can maximize plant biomass yields while minimizing dedicated space and optimizing resources (energy, water, and nutrients) through the almost complete recycling of waste [14,15]. Collectively referred to as BLSS, these systems pursue the ambitious goal of reproducing Earth-like biogeochemical cycles by fostering mutualistic interactions among humans, plants, and microbes in space [544]. Consequently, integrating medicinal plants into BLSS represents a promising avenue for achieving long-term sustainability and crew health on future missions. In other words, cultivating edible vegetables alongside medicinal plants into BLSS could provide a sustainable source of both nutrition and natural therapeutics. However, further progress in understanding the changes in plant physiology induced by the spatial exposome is essential [16,545,546].While growing plants in space is possible, albeit complicated, further challenges remain. Not all medicinal plants are suitable for space farming, at least in this pioneering phase. As an example, fast-growing and compact herbaceous plants are likely easier to cultivate and thus preferable to tree species. However, the greatest gap in knowledge concerns the effects of the space environment on the chemical composition of medicinal plants. Experimental evidence indicates that the exposure of several plant seeds to space environment can induce genetic modifications, leading to alterations in the chemical composition of the plants that develop from them [547,548,549,550,551]. It should be emphasized that in these studies, seed germination and plant cultivation were carried out on Earth once the seeds returned from space. The mutagenic effect of the space environment is so strong that it has given rise to a new technique called space mutation breeding, aimed at artificially improving crops [552]. The influence of extraterrestrial conditions on plant physiology and chemistry is not necessarily adverse but requires it to be fully investigated and understood. To put it differently, future studies will have to investigate the phytochemistry of medicinal plants that have completed their entire life cycle in space, which does not necessarily overlap with that observed on Earth. This will allow the accurate reassessment of the efficacy-to-safety ratio of the plant extracts produced in space.Not least of all, the entire production chain of medicinal plant extracts—including biomass harvesting and processing, solvent extraction, purification, chemical analysis, and storage— constitutes an exceptionally challenging process to implement in the space environment, whether aboard spacecraft or in extraterrestrial habitats. It requires rethinking the entire process to adapt to the different restrictions imposed by space environmental conditions, such as limited space and microgravity, while maintaining efficiency, reliability and reproducibility. Ideally, the process should be fully automated and include an appropriate waste management component aimed at minimizing waste production. This problem can be circumvented at least in part and where possible by using medicinal plants as fresh functional foods (food enriched of biologically active constituents) to supplement the diet of human in space [327].
- From a regulatory perspective, space agencies, product regulators and international health organizations need to develop standards for the safe use of space-grown medicinal products. It will therefore be critical to develop guidelines for herbal product production and classification, biosafety characterization, dosage validation, interaction with conventional therapies, and determination of stability.
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SSP | Space Soviet Program |
| NASA | National Aeronautics and Space Administration |
| ESA | European Space Agency |
| CSA | Canadian Space Agency |
| JAXA | Japan Aerospace eXploration Agency |
| CMSA | China Manned Space Agency |
| LEO | Low Earth Orbit |
| SANS | Spaceflight-Associated Neuro-ocular Syndrome |
| SMS | Space Motion Sickness |
| µG | Microgravity |
| SR | Space Radiation |
| SPEs | Solar Particle Events |
| GCRs | Galactic Cosmic Rays |
| TPR | Trapped Particle Radiation |
| HZE | high (H), atomic number (Z), and energy (E) |
| LET | Linear Energy Transfer |
| ARSs | Acute Radiation Syndrome |
| CNS | Central Nervous System |
| PK | PharmacoKinetic |
| PD | PharmacoDynamic |
| ISS | International Space Station |
| APIs | Active Pharmaceutical Ingredients |
| PBPK | Physiologically-Based Pharmacokinetic |
| ISPM | In-Situ Pharmaceutical Manufacturing |
| USP | United State Pharmacopeia |
| EMA | European Medicines Agency |
| WHO | World Health Organization |
| NSAIDs | Non-Steroidal Anti-Inflammatory Drugs |
| G-CSF | Granulocyte Colony-Stimulating Factor |
| BLSS | Bioregenerative Life Support Systems |
References
- Wu, J. History of Human Space Exploration. Introduction to Space Science 2021, 9–30. [CrossRef]
- Melamed, A.; Rao, A.; de Rohan Willner, O.; Kreps, S. Going to Outer Space with New Space: The Rise and Consequences of Evolving Public-Private Partnerships. Space Policy 2024, 68, 101626. [CrossRef]
- Paikowsky, D. What Is New Space? The Changing Ecosystem of Global Space Activity. New Space 2017, 5, 84–88. [CrossRef]
- Krittanawong, C.; Singh, N.K.; Scheuring, R.A.; Urquieta, E.; Bershad, E.M.; Macaulay, T.R.; Kaplin, S.; Dunn, C.; Kry, S.F.; Russomano, T.; et al. Human Health during Space Travel: State-of-the-Art Review. Cells 2022, 12, 40. [CrossRef]
- Hart, D.A. Homo Sapiens—A Species Not Designed for Space Flight: Health Risks in Low Earth Orbit and Beyond, Including Potential Risks When Traveling beyond the Geomagnetic Field of Earth. Life (Basel) 2023, 13, 757. [CrossRef]
- Smith, L. Space Station and Spacecraft Environmental Conditions and Human Mental Health: Specific Recommendations and Guidelines. Life Sci Space Res (Amst) 2024, 40, 126–134. [CrossRef]
- Seoane-Viaño, I.; Ong, J.J.; Basit, A.W.; Goyanes, A. To Infinity and beyond: Strategies for Fabricating Medicines in Outer Space. International Journal of Pharmaceutics: X 2022, 4, 100121. [CrossRef]
- Aziz, S.; Raza, M.A.; Noreen, M.; Iqbal, M.Z.; Raza, S.M. Astropharmacy: Roles of Pharmacist in Space. Innov Pharm 2022, 13, 10.24926/iip.v13i3.4956. [CrossRef]
- Sawyers, L.; Anderson, C.; Boyd, M.J.; Hessel, V.; Wotring, V.; Williams, P.M.; Toh, L.S. Astropharmacy: Pushing the Boundaries of the Pharmacists’ Role for Sustainable Space Exploration. Research in Social and Administrative Pharmacy 2022, 18, 3612–3621. [CrossRef]
- Aksoyalp, Z.Ş.; Temel, A.; Karpuz, M. Pharmacological Innovations in Space: Challenges and Future Perspectives. Pharm Res 2024, 41, 2095–2120. [CrossRef]
- Pachiyappan, J.K.; Patel, M.; Roychowdhury, P.; Nizam, I.; Seenivasan, R.; Sudhakar, S.; Jeyaprakash, M.R.; Karri, V.V.S.R.; Venkatesan, J.; Mehta, P.; et al. A Review of the Physiological Effects of Microgravity and Innovative Formulation for Space Travelers. J Pharmacokinet Pharmacodyn 2024, 51, 605–620. [CrossRef]
- d’Ischia, M.; Manini, P.; Moracci, M.; Saladino, R.; Ball, V.; Thissen, H.; Evans, R.A.; Puzzarini, C.; Barone, V. Astrochemistry and Astrobiology: Materials Science in Wonderland? International Journal of Molecular Sciences 2019, 20, 4079. [CrossRef]
- Clément, G. Fundamentals of Space Medicine; Springer, 2025;
- Mortimer, J.C.; Gilliham, M. SpaceHort: Redesigning Plants to Support Space Exploration and on-Earth Sustainability. Current Opinion in Biotechnology 2022, 73, 246–252. [CrossRef]
- Nguyen, M.T.P.; Knowling, M.; Tran, N.N.; Burgess, A.; Fisk, I.; Watt, M.; Escribà-Gelonch, M.; This, H.; Culton, J.; Hessel, V. Space Farming: Horticulture Systems on Spacecraft and Outlook to Planetary Space Exploration. Plant Physiology and Biochemistry 2023, 194, 708–721. [CrossRef]
- Maffei, M.E.; Balestrini, R.; Costantino, P.; Lanfranco, L.; Morgante, M.; Battistelli, A.; Del Bianco, M. The Physiology of Plants in the Context of Space Exploration. Commun Biol 2024, 7, 1311. [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [CrossRef]
- Adamopoulos, K.; Koutsouris, D.; Zaravinos, A.; Lambrou, G.I. Gravitational Influence on Human Living Systems and the Evolution of Species on Earth. Molecules 2021, 26, 2784. [CrossRef]
- Barratt, MichaelR. Physical and Bioenvironmental Aspects of Human Space Flight. In Principles of clinical medicine for space flight; Michael R. Barratt, Ellen S. Baker, & Sam L. Pool. Springer, 2019; pp. 3–37.
- Pollock, R.D.; Hodkinson, P.D.; Smith, T.G. Oh G: The x, y and z of Human Physiological Responses to Acceleration. Experimental Physiology 2021, 106, 2367–2384. [CrossRef]
- Giacinto, O.; Lusini, M.; Sammartini, E.; Minati, A.; Mastroianni, C.; Nenna, A.; Pascarella, G.; Sammartini, D.; Carassiti, M.; Miraldi, F.; et al. Cardiovascular Effects of Cosmic Radiation and Microgravity. J Clin Med 2024, 13, 520. [CrossRef]
- Han, H.; Jia, H.; Wang, Y.-F.; Song, J.-P. Cardiovascular Adaptations and Pathological Changes Induced by Spaceflight: From Cellular Mechanisms to Organ-Level Impacts. Military Med Res 2024, 11, 68. [CrossRef]
- Hariom, S.K.; Nelson, E.J.R. Cardiovascular Adaptations in Microgravity Conditions. Life Sciences in Space Research 2024, 42, 64–71. [CrossRef]
- Hussain, I.; Ullah, R.; Simran, B.F.N.U.; Kaur, P.; Kumar, M.; Raj, R.; Faraz, M.; Mehmoodi, A.; Malik, J. Cardiovascular Effects of Long-duration Space Flight. Health Sci Rep 2024, 7, e2305. [CrossRef]
- Mircea, A.A.; Pistritu, D.V.; Fortner, A.; Tanca, A.; Liehn, E.A.; Bucur, O. Space Travel: The Radiation and Microgravity Effects on the Cardiovascular System. Int J Mol Sci 2024, 25, 11812. [CrossRef]
- Siddiqui, R.; Qaisar, R.; Al-Dahash, K.; Altelly, A.H.; Elmoselhi, A.B.; Khan, N.A. Cardiovascular Changes under the Microgravity Environment and the Gut Microbiome. Life Sciences in Space Research 2024, 40, 89–96. [CrossRef]
- Azariah, J.; Terranova, U. Microgravity and Cardiovascular Health in Astronauts: A Narrative Review. Health Science Reports 2025, 8, e70316. [CrossRef]
- Hargens, A.R.; Richardson, S. Cardiovascular Adaptations, Fluid Shifts, and Countermeasures Related to Space Flight. Respiratory Physiology & Neurobiology 2009, 169, S30–S33. [CrossRef]
- Hinghofer-Szalkay, H. Gravity, the Hydrostatic Indifference Concept and the Cardiovascular System. Eur J Appl Physiol 2011, 111, 163–174. [CrossRef]
- Thornton, W.; Hoffler, G.W.; Rummel, J. Anthropometric Changes and Fluid Shifts.; 1977.
- Leach, C.S.; Alfrey, C.P.; Suki, W.N.; Leonard, J.I.; Rambaut, P.C.; Inners, L.D.; Smith, S.M.; Lane, H.W.; Krauhs, J.M. Regulation of Body Fluid Compartments during Short-Term Spaceflight. Journal of Applied Physiology 1996, 81, 105–116. [CrossRef]
- Watenpaugh, D.E. Fluid Volume Control during Short-Term Space Flight and Implications for Human Performance. J Exp Biol 2001, 204, 3209–3215. [CrossRef]
- Shykoff, B.E.; Farhi, L.E.; Olszowka, A.J.; Pendergast, D.R.; Rokitka, M.A.; Eisenhardt, C.G.; Morin, R.A. Cardiovascular Response to Submaximal Exercise in Sustained Microgravity. Journal of Applied Physiology 1996. [CrossRef]
- Norsk, P.; Damgaard, M.; Petersen, L.; Gybel, M.; Pump, B.; Gabrielsen, A.; Christensen, N.J. Vasorelaxation in Space. Hypertension 2006, 47, 69–73. [CrossRef]
- Norsk, P.; Asmar, A.; Damgaard, M.; Christensen, N.J. Fluid Shifts, Vasodilatation and Ambulatory Blood Pressure Reduction during Long Duration Spaceflight. The Journal of Physiology 2015, 593, 573–584. [CrossRef]
- Perhonen, M.A.; Franco, F.; Lane, L.D.; Buckey, J.C.; Blomqvist, C.G.; Zerwekh, J.E.; Peshock, R.M.; Weatherall, P.T.; Levine, B.D. Cardiac Atrophy after Bed Rest and Spaceflight. Journal of Applied Physiology 2001, 91, 645–653. [CrossRef]
- MacNamara, J.P.; Dias, K.A.; Sarma, S.; Lee, S.M.C.; Martin, D.; Romeijn, M.; Zaha, V.G.; Levine, B.D. Cardiac Effects of Repeated Weightlessness During Extreme Duration Swimming Compared With Spaceflight. Circulation 2021, 143, 1533–1535. [CrossRef]
- Summers, R.L.; Martin, D.S.; Meck, J.V.; Coleman, T.G. Mechanism of Spaceflight-Induced Changes in Left Ventricular Mass. The American Journal of Cardiology 2005, 95, 1128–1130. [CrossRef]
- Fraser, K.S.; Greaves, D.K.; Shoemaker, J.K.; Blaber, A.P.; Hughson, R.L. Heart Rate and Daily Physical Activity with Long-Duration Habitation of the International Space Station. Aviat Space Environ Med 2012, 83, 577–584. [CrossRef]
- May, C.; Borowski, A.; Martin, D.; Popovic, Z.; Negishi, K.; Hussan, J.R.; Gladding, P.; Hunter, P.; Iskovitz, I.; Kassemi, M.; et al. AFFECT OF MICROGRAVITY ON CARDIAC SHAPE: COMPARISON OF PRE- AND IN-FLIGHT DATA TO MATHEMATICAL MODELING. Journal of the American College of Cardiology 2014, 63, A1096. [CrossRef]
- van Loon, L.M.; Steins, A.; Schulte, K.-M.; Gruen, R.; Tucker, E.M. Computational Modeling of Orthostatic Intolerance for Travel to Mars. NPJ Microgravity 2022, 8, 34. [CrossRef]
- Tank, J.; Jordan, J. Mighty Hearts in Space. J Physiol 2015, 593, 485–486. [CrossRef]
- Purdy, R.E.; Kahwaji, C.I. Vascular Adaptation to Microgravity: Extending the Hypothesis. Journal of Applied Physiology 2002, 93, 1181–1182. [CrossRef]
- Meck, J.V.; Waters, W.W.; Ziegler, M.G.; deBlock, H.F.; Mills, P.J.; Robertson, D.; Huang, P.L. Mechanisms of Postspaceflight Orthostatic Hypotension: Low A1-Adrenergic Receptor Responses before Flight and Central Autonomic Dysregulation Postflight. American Journal of Physiology-Heart and Circulatory Physiology 2004, 286, H1486–H1495. [CrossRef]
- Pecaut, M.J.; Mao, X.W.; Bellinger, D.L.; Jonscher, K.R.; Stodieck, L.S.; Ferguson, V.L.; Bateman, T.A.; Mohney, R.P.; Gridley, D.S. Is Spaceflight-Induced Immune Dysfunction Linked to Systemic Changes in Metabolism? PLOS ONE 2017, 12, e0174174. [CrossRef]
- Meck, J.V.; Reyes, C.J.; Perez, S.A.; Goldberger, A.L.; Ziegler, M.G. Marked Exacerbation of Orthostatic Intolerance After Long- vs. Short-Duration Spaceflight in Veteran Astronauts. Biopsychosocial Science and Medicine 2001, 63, 865.
- Johnston, R.S.; Dietlein, L.F.; Berry, C.A.; James F. Parker, J.; West, V.; Jones, W.L. Biomedical Results of Apollo 1975.
- Johnston, R.S.; Dietlein, L.F. Biomedical Results from Skylab 1977.
- Baisden, D.L.; Jones, M.M. Cardiac Dysrhythmia Analysis on Flights STS-1. STS-61C. 1988.
- Anzai, T.; Frey, M.A.; Nogami, A. Cardiac Arrhythmias during Long-Duration Spaceflights. Journal of Arrhythmia 2014, 30, 139–149. [CrossRef]
- Khine, H.W.; Steding-Ehrenborg, K.; Hastings, J.L.; Kowal, J.; Daniels, J.D.; Page, R.L.; Goldberger, J.J.; Ng, J.; Adams-Huet, B.; Bungo, M.W.; et al. Effects of Prolonged Spaceflight on Atrial Size, Atrial Electrophysiology, and Risk of Atrial Fibrillation. Circulation: Arrhythmia and Electrophysiology 2018, 11, e005959. [CrossRef]
- D’Aunno, D.S.; Dougherty, A.H.; DeBlock, H.F.; Meck, J.V. Effect of Short- and Long-Duration Spaceflight on QTc Intervals in Healthy Astronauts. The American Journal of Cardiology 2003, 91, 494–497. [CrossRef]
- Marshall-Goebel, K.; Laurie, S.S.; Alferova, I.V.; Arbeille, P.; Auñón-Chancellor, S.M.; Ebert, D.J.; Lee, S.M.C.; Macias, B.R.; Martin, D.S.; Pattarini, J.M.; et al. Assessment of Jugular Venous Blood Flow Stasis and Thrombosis During Spaceflight. JAMA Network Open 2019, 2, e1915011. [CrossRef]
- Auñón-Chancellor, S.M.; Pattarini, J.M.; Moll, S.; Sargsyan, A. Venous Thrombosis during Spaceflight. New England Journal of Medicine 2020, 382, 89–90. [CrossRef]
- Pavela, J.; Sargsyan, A.; Bedi, D.; Everson, A.; Charvat, J.; Mason, S.; Johansen, B.; Marshall-Goebel, K.; Mercaldo, S.; Shah, R.; et al. Surveillance for Jugular Venous Thrombosis in Astronauts. Vasc Med 2022, 27, 365–372. [CrossRef]
- Elahi, M.M.; Witt, A.N.; Pryzdial, E.L.G.; McBeth, P.B. Thrombotic Triad in Microgravity. Thrombosis Research 2024, 233, 82–87. [CrossRef]
- Lansiaux, E.; Jain, N.; Yatin Chodnekar, S.; Siddiq, A.; Ibrahim, M.; Yèche, M.; Kantane, I. Understanding the Complexities of Space Anaemia in Extended Space Missions: Revelations from Microgravitational Odyssey. Front. Physiol. 2024, 15. [CrossRef]
- Alfrey, C.P.; Udden, M.M.; Huntoon, C.L.; Driscoll, T. Destruction of Newly Released Red Blood Cells in Space Flight. Medicine & Science in Sports & Exercise 1996, 28, 42.
- Smith, S.M. Red Blood Cell and Iron Metabolism during Space Flight. Nutrition 2002, 18, 864–866. [CrossRef]
- Trudel, G.; Shafer, J.; Laneuville, O.; Ramsay, T. Characterizing the Effect of Exposure to Microgravity on Anemia: More Space Is Worse. American Journal of Hematology 2020, 95, 267–273. [CrossRef]
- Trudel, G.; Shahin, N.; Ramsay, T.; Laneuville, O.; Louati, H. Hemolysis Contributes to Anemia during Long-Duration Space Flight. Nat Med 2022, 28, 59–62. [CrossRef]
- Agrawal, S.; Kumar, S.; Ingole, V.; Acharya, S.; Wanjari, A.; Bawankule, S.; Raisinghani, N. Does Anemia Affects Cognitive Functions in Neurologically Intact Adult Patients: Two Year Cross Sectional Study at Rural Tertiary Care Hospital. J Family Med Prim Care 2019, 8, 3005–3008. [CrossRef]
- Moosavi, D.; Wolovsky, D.; Depompeis, A.; Uher, D.; Lennington, D.; Bodden, R.; Garber, C.E. The Effects of Spaceflight Microgravity on the Musculoskeletal System of Humans and Animals, with an Emphasis on Exercise as a Countermeasure: A Systematic Scoping Review. Physiol Res 2021, 70, 119–151. [CrossRef]
- Man, J.; Graham, T.; Squires-Donelly, G.; Laslett, A.L. The Effects of Microgravity on Bone Structure and Function. NPJ Microgravity 2022, 8, 9. [CrossRef]
- Akima, H.; Kawakami, Y.; Kubo, K.; Sekiguchi, C.; Ohshima, H.; Miyamoto, A.; Fukunaga, T. Effect of Short-Duration Spaceflight on Thigh and Leg Muscle Volume. Med Sci Sports Exerc 2000, 32, 1743–1747. [CrossRef]
- Riley, D.A.; Bain, J.L.W.; Thompson, J.L.; Fitts, R.H.; Widrick, J.J.; Trappe, S.W.; Trappe, T.A.; Costill, D.L. Decreased Thin Filament Density and Length in Human Atrophic Soleus Muscle Fibers after Spaceflight. Journal of Applied Physiology 2000, 88, 567–572. [CrossRef]
- Antonutto, G.; Capelli, C.; Giradis, M.; Zamparo, P.; di Prampero, P.E. Effects of Microgravity on Muscular Explosive Power of the Lower Limbs in Humans. Acta Astronautica 1995, 36, 473–478. [CrossRef]
- Antonutto, G.; Bodem, F.; Zamparo, P.; di Prampero, P.E. Maximal Power and EMG of Lower Limbs after 21 Days Spaceflight in One Astronaut. J Gravit Physiol 1998, 5, P63-66.
- Bailey, J.F.; Miller, S.L.; Khieu, K.; O’Neill, C.W.; Healey, R.M.; Coughlin, D.G.; Sayson, J.V.; Chang, D.G.; Hargens, A.R.; Lotz, J.C. From the International Space Station to the Clinic: How Prolonged Unloading May Disrupt Lumbar Spine Stability. Spine J 2018, 18, 7–14. [CrossRef]
- Belavy, D.L.; Adams, M.; Brisby, H.; Cagnie, B.; Danneels, L.; Fairbank, J.; Hargens, A.R.; Judex, S.; Scheuring, R.A.; Sovelius, R.; et al. Disc Herniations in Astronauts: What Causes Them, and What Does It Tell Us about Herniation on Earth? Eur Spine J 2016, 25, 144–154. [CrossRef]
- Chang, D.; Healey, R.; Snyder, A.; Sayson, J.; Macias, B.; Coughlin, D.; Bailey, J.; Parazynski, S.; Lotz, J.; Hargens, A. Lumbar Spine Paraspinal Muscle and Intervertebral Disc Height Changes in Astronauts after Long-Duration Spaceflight on the International Space Station. Spine (Phila Pa 1976) 2016, 41, 1917–1924. [CrossRef]
- Puglia, I.; Balsamo, M.; Vukich, M.; Zolesi, V. Long-Term Microgravity Effects on Isometric Handgrip and Precision Pinch Force with Visual and Proprioceptive Feedback. International Journal of Aerospace Engineering 2018, 2018, 1952630. [CrossRef]
- Kenkre, J.S.; Bassett, J.H. The Bone Remodelling Cycle. 2018. [CrossRef]
- Collet, P.; Uebelhart, D.; Vico, L.; Moro, L.; Hartmann, D.; Roth, M.; Alexandre, C. Effects of 1- and 6-Month Spaceflight on Bone Mass and Biochemistry in Two Humans. Bone 1997, 20, 547–551. [CrossRef]
- Caillot-Augusseau, A.; Lafage-Proust, M.-H.; Soler, C.; Pernod, J.; Dubois, F.; Alexandre, C. Bone Formation and Resorption Biological Markers in Cosmonauts during and after a 180-Day Space Flight (Euromir 95). Clinical Chemistry 1998, 44, 578–585. [CrossRef]
- Vico, L.; Collet, P.; Guignandon, A.; Lafage-Proust, M.-H.; Thomas, T.; Rehailia, M.; Alexandre, C. Effects of Long-Term Microgravity Exposure on Cancellous and Cortical Weight-Bearing Bones of Cosmonauts. The Lancet 2000, 355, 1607–1611. [CrossRef]
- Lang, T.; LeBlanc, A.; Evans, H.; Lu, Y.; Genant, H.; Yu, A. Cortical and Trabecular Bone Mineral Loss From the Spine and Hip in Long-Duration Spaceflight. Journal of Bone and Mineral Research 2004, 19, 1006–1012. [CrossRef]
- Sibonga, J.D.; Evans, H.J.; Sung, H.G.; Spector, E.R.; Lang, T.F.; Oganov, V.S.; Bakulin, A.V.; Shackelford, L.C.; LeBlanc, A.D. Recovery of Spaceflight-Induced Bone Loss: Bone Mineral Density after Long-Duration Missions as Fitted with an Exponential Function. Bone 2007, 41, 973–978. [CrossRef]
- Dana Carpenter, R.; LeBlanc, A.D.; Evans, H.; Sibonga, J.D.; Lang, T.F. Long-Term Changes in the Density and Structure of the Human Hip and Spine after Long-Duration Spaceflight. Acta Astronautica 2010, 67, 71–81. [CrossRef]
- Burkhart, K.; Allaire, B.; Bouxsein, M.L. Negative Effects of Long-Duration Spaceflight on Paraspinal Muscle Morphology. Spine 2019, 44, 879. [CrossRef]
- Stein, T.P. Weight, Muscle and Bone Loss during Space Flight: Another Perspective. Eur J Appl Physiol 2013, 113, 2171–2181. [CrossRef]
- Seidler, R.D.; Mao, X.W.; Tays, G.D.; Wang, T.; Zu Eulenburg, P. Effects of Spaceflight on the Brain. Lancet Neurol 2024, 23, 826–835. [CrossRef]
- Wuyts, F.L.; Deblieck, C.; Vandevoorde, C.; Durante, M. Brains in Space: Impact of Microgravity and Cosmic Radiation on the CNS during Space Exploration. Nat. Rev. Neurosci. 2025, 26, 354–371. [CrossRef]
- Roberts, D.R.; Asemani, D.; Nietert, P.J.; Eckert, M.A.; Inglesby, D.C.; Bloomberg, J.J.; George, M.S.; Brown, T.R. Prolonged Microgravity Affects Human Brain Structure and Function. AJNR Am J Neuroradiol 2019, 40, 1878–1885. [CrossRef]
- Van Ombergen, A.; Jillings, S.; Jeurissen, B.; Tomilovskaya, E.; Rumshiskaya, A.; Litvinova, L.; Nosikova, I.; Pechenkova, E.; Rukavishnikov, I.; Manko, O.; et al. Brain Ventricular Volume Changes Induced by Long-Duration Spaceflight. Proceedings of the National Academy of Sciences 2019, 116, 10531–10536. [CrossRef]
- Kramer, L.A.; Hasan, K.M.; Stenger, M.B.; Sargsyan, A.; Laurie, S.S.; Otto, C.; Ploutz-Snyder, R.J.; Marshall-Goebel, K.; Riascos, R.F.; Macias, B.R. Intracranial Effects of Microgravity: A Prospective Longitudinal MRI Study. Radiology 2020, 295, 640–648. [CrossRef]
- Hupfeld, K.E.; McGregor, H.R.; Lee, J.K.; Beltran, N.E.; Kofman, I.S.; De Dios, Y.E.; Reuter-Lorenz, P.A.; Riascos, R.F.; Pasternak, O.; Wood, S.J.; et al. The Impact of 6 and 12 Months in Space on Human Brain Structure and Intracranial Fluid Shifts. Cereb Cortex Commun 2020, 1, tgaa023. [CrossRef]
- McGregor, H.R.; Hupfeld, K.E.; Pasternak, O.; Beltran, N.E.; De Dios, Y.E.; Bloomberg, J.J.; Wood, S.J.; Mulavara, A.P.; Riascos, R.F.; Reuter-Lorenz, P.A.; et al. Impacts of Spaceflight Experience on Human Brain Structure. Sci Rep 2023, 13, 7878. [CrossRef]
- Koppelmans, V.; Bloomberg, J.J.; Mulavara, A.P.; Seidler, R.D. Brain Structural Plasticity with Spaceflight. npj Microgravity 2016, 2, 1–8. [CrossRef]
- Galdamez, L.A.; Mader, T.H.; Ong, J.; Kadipasaoglu, C.M.; Lee, A.G. A Multifactorial, Evidence-Based Analysis of Pathophysiology in Spaceflight Associated Neuro-Ocular Syndrome (SANS). Eye 2025, 39, 700–709. [CrossRef]
- Mader, T.H.; Gibson, C.R.; Pass, A.F.; Kramer, L.A.; Lee, A.G.; Fogarty, J.; Tarver, W.J.; Dervay, J.P.; Hamilton, D.R.; Sargsyan, A.; et al. Optic Disc Edema, Globe Flattening, Choroidal Folds, and Hyperopic Shifts Observed in Astronauts after Long-Duration Space Flight. Ophthalmology 2011, 118, 2058–2069. [CrossRef]
- Mader, T.H.; Gibson, C.R.; Barratt, M.R.; Miller, N.R.; Subramanian, P.S.; Killer, H.E.; Tarver, W.J.; Sargsyan, A.E.; Garcia, K.; Hart, S.F.; et al. Persistent Globe Flattening in Astronauts Following Long-Duration Spaceflight. Neuroophthalmology 45, 29–35. [CrossRef]
- Buckey, J.C.; Lan, M.; Lewis, L.D. Space Travel-Associated Motion Sickness and Its Treatment. British Journal of Clinical Pharmacology n/a. [CrossRef]
- Mergner, T.; Rosemeier, T. Interaction of Vestibular, Somatosensory and Visual Signals for Postural Control and Motion Perception under Terrestrial and Microgravity Conditions—a Conceptual Model. Brain Research Reviews 1998, 28, 118–135. [CrossRef]
- Khalid, A.; Prusty, P.P.; Arshad, I.; Gustafson, H.E.; Jalaly, I.; Nockels, K.; Bentley, B.L.; Goel, R.; Ferrè, E.R. Pharmacological and Non-Pharmacological Countermeasures to Space Motion Sickness: A Systematic Review. Front Neural Circuits 2023, 17, 1150233. [CrossRef]
- Young, L.R.; Oman, C.M.; Watt, D.G.; Money, K.E.; Lichtenberg, B.K. Spatial Orientation in Weightlessness and Readaptation to Earth’s Gravity. Science 1984, 225, 205–208. [CrossRef]
- Souvestre, P.A.; Landrock, C.K.; Blaber, A.P. Reducing Incapacitating Symptoms during Space Flight: Is Postural Deficiency Syndrome an Applicable Model? Hippokratia 2008, 12, 41–48.
- Harris, L.R.; Jenkin, M.; Jenkin, H.; Zacher, J.E.; Dyde, R.T. The Effect of Long-Term Exposure to Microgravity on the Perception of Upright. npj Microgravity 2017, 3, 1–9. [CrossRef]
- Kornilova, L.N.; Grigorova, V.; Bodo, G. Vestibular Function and Sensory Interaction in Space Flight. J Vestib Res 1993, 3, 219–230.
- Lowrey, C.R.; Perry, S.D.; Strzalkowski, N.D.J.; Williams, D.R.; Wood, S.J.; Bent, L.R. Selective Skin Sensitivity Changes and Sensory Reweighting Following Short-Duration Space Flight. Journal of Applied Physiology 2014, 116, 683–692. [CrossRef]
- Carriot, J.; Jamali, M.; Cullen, K.E. Rapid Adaptation of Multisensory Integration in Vestibular Pathways. Front. Syst. Neurosci. 2015, 9. [CrossRef]
- Clément, G.; Wood, S.J.; Paloski, W.H.; Reschke, M.F. Changes in Gain of Horizontal Vestibulo-Ocular Reflex during Spaceflight. J Vestib Res 29, 241–251. [CrossRef]
- Glukhikh, D.O.; Naumov, I.A.; Schoenmaekers, C.; Kornilova, L.N.; Wuyts, F.L. The Role of Different Afferent Systems in the Modulation of the Otolith-Ocular Reflex After Long-Term Space Flights. Front. Physiol. 2022, 13. [CrossRef]
- Hupfeld, K.E.; McGregor, H.R.; Koppelmans, V.; Beltran, N.E.; Kofman, I.S.; De Dios, Y.E.; Riascos, R.F.; Reuter-Lorenz, P.A.; Wood, S.J.; Bloomberg, J.J.; et al. Brain and Behavioral Evidence for Reweighting of Vestibular Inputs with Long-Duration Spaceflight. Cereb Cortex 2021, 32, 755–769. [CrossRef]
- Demertzi, A.; Van Ombergen, A.; Tomilovskaya, E.; Jeurissen, B.; Pechenkova, E.; Di Perri, C.; Litvinova, L.; Amico, E.; Rumshiskaya, A.; Rukavishnikov, I.; et al. Cortical Reorganization in an Astronaut’s Brain after Long-Duration Spaceflight. Brain Struct Funct 2016, 221, 2873–2876. [CrossRef]
- Pechenkova, E.; Nosikova, I.; Rumshiskaya, A.; Litvinova, L.; Rukavishnikov, I.; Mershina, E.; Sinitsyn, V.; Van Ombergen, A.; Jeurissen, B.; Jillings, S.; et al. Alterations of Functional Brain Connectivity After Long-Duration Spaceflight as Revealed by fMRI. Front. Physiol. 2019, 10. [CrossRef]
- Jillings, S.; Van Ombergen, A.; Tomilovskaya, E.; Rumshiskaya, A.; Litvinova, L.; Nosikova, I.; Pechenkova, E.; Rukavishnikov, I.; Kozlovskaya, I.B.; Manko, O.; et al. Macro- and Microstructural Changes in Cosmonauts’ Brains after Long-Duration Spaceflight. Science Advances 2020, 6, eaaz9488. [CrossRef]
- Jillings, S.; Pechenkova, E.; Tomilovskaya, E.; Rukavishnikov, I.; Jeurissen, B.; Van Ombergen, A.; Nosikova, I.; Rumshiskaya, A.; Litvinova, L.; Annen, J.; et al. Prolonged Microgravity Induces Reversible and Persistent Changes on Human Cerebral Connectivity. Commun Biol 2023, 6, 1–11. [CrossRef]
- Mulavara, A.P.; Feiveson, A.H.; Fiedler, J.; Cohen, H.; Peters, B.T.; Miller, C.; Brady, R.; Bloomberg, J.J. Locomotor Function after Long-Duration Space Flight: Effects and Motor Learning during Recovery. Exp Brain Res 2010, 202, 649–659. [CrossRef]
- Cohen, H.S.; Kimball, K.T.; Mulavara, A.P.; Bloomberg, J.J.; Paloski, W.H. Posturography and Locomotor Tests of Dynamic Balance after Long-Duration Spaceflight. J Vestib Res 2012, 22, 191–196. [CrossRef]
- Wood, S.J.; Paloski, W.H.; Clark, J.B. Assessing Sensorimotor Function Following ISS with Computerized Dynamic Posturography. Aerospace Medicine and Human Performance 2015, 86, A45–A53. [CrossRef]
- Shishkin, N.; Kitov, V.; Sayenko, D.; Tomilovskaya, E. Sensory Organization of Postural Control after Long Term Space Flight. Front. Neural Circuits 2023, 17. [CrossRef]
- Moore, S.T.; Dilda, V.; Morris, T.R.; Yungher, D.A.; MacDougall, H.G.; Wood, S.J. Long-Duration Spaceflight Adversely Affects Post-Landing Operator Proficiency. Sci Rep 2019, 9, 2677. [CrossRef]
- Tays, G.D.; Hupfeld, K.E.; McGregor, H.R.; Salazar, A.P.; De Dios, Y.E.; Beltran, N.E.; Reuter-Lorenz, P.A.; Kofman, I.S.; Wood, S.J.; Bloomberg, J.J.; et al. The Effects of Long Duration Spaceflight on Sensorimotor Control and Cognition. Front. Neural Circuits 2021, 15. [CrossRef]
- Crucian, B.; Babiak-Vazquez, A.; Johnston, S.; Pierson, D.L.; Ott, C.M.; Sams, C. Incidence of Clinical Symptoms during Long-Duration Orbital Spaceflight. Int J Gen Med 2016, 9, 383–391. [CrossRef]
- Lynch, S.V.; Pedersen, O. The Human Intestinal Microbiome in Health and Disease. N Engl J Med 2016, 375, 2369–2379. [CrossRef]
- Siddiqui, R.; Akbar, N.; Khan, N.A. Gut Microbiome and Human Health under the Space Environment. J Appl Microbiol 2021, 130, 14–24. [CrossRef]
- Alauzet, C.; Cunat, L.; Wack, M.; Lanfumey, L.; Legrand-Frossi, C.; Lozniewski, A.; Agrinier, N.; Cailliez-Grimal, C.; Frippiat, J.-P. Impact of a Model Used to Simulate Chronic Socio-Environmental Stressors Encountered during Spaceflight on Murine Intestinal Microbiota. Int J Mol Sci 2020, 21, 7863. [CrossRef]
- Garrett-Bakelman, F.E.; Darshi, M.; Green, S.J.; Gur, R.C.; Lin, L.; Macias, B.R.; McKenna, M.J.; Meydan, C.; Mishra, T.; Nasrini, J.; et al. The NASA Twins Study: A Multidimensional Analysis of a Year-Long Human Spaceflight. Science 2019, 364, eaau8650. [CrossRef]
- Voorhies, A.A.; Mark Ott, C.; Mehta, S.; Pierson, D.L.; Crucian, B.E.; Feiveson, A.; Oubre, C.M.; Torralba, M.; Moncera, K.; Zhang, Y.; et al. Study of the Impact of Long-Duration Space Missions at the International Space Station on the Astronaut Microbiome. Sci Rep 2019, 9, 9911. [CrossRef]
- Borovsky, J.E.; Valdivia, J.A. The Earth’s Magnetosphere: A Systems Science Overview and Assessment. Surv Geophys 2018, 39, 817–859. [CrossRef]
- Christoudias, T.; Kirkby, J.; Stolzenburg, D.; Pozzer, A.; Sommer, E.; Brasseur, G.P.; Kulmala, M.; Lelieveld, J. Earth’s Atmosphere Protects the Biosphere from Nearby Supernovae. Commun Earth Environ 2024, 5, 1–8. [CrossRef]
- Takahashi, A.; Ikeda, H.; Yoshida, Y. Role of High-Linear Energy Transfer Radiobiology in Space Radiation Exposure Risks. Int J Part Ther 2018, 5, 151–159. [CrossRef]
- Restier-Verlet, J.; El-Nachef, L.; Ferlazzo, M.L.; Al-Choboq, J.; Granzotto, A.; Bouchet, A.; Foray, N. Radiation on Earth or in Space: What Does It Change? International Journal of Molecular Sciences 2021, 22, 3739. [CrossRef]
- Fogtman, A.; Baatout, S.; Baselet, B.; Berger, T.; Hellweg, C.E.; Jiggens, P.; La Tessa, C.; Narici, L.; Nieminen, P.; Sabatier, L.; et al. Towards Sustainable Human Space Exploration—Priorities for Radiation Research to Quantify and Mitigate Radiation Risks. NPJ Microgravity 2023, 9, 8. [CrossRef]
- Patel, Z.S.; Brunstetter, T.J.; Tarver, W.J.; Whitmire, A.M.; Zwart, S.R.; Smith, S.M.; Huff, J.L. Red Risks for a Journey to the Red Planet: The Highest Priority Human Health Risks for a Mission to Mars. npj Microgravity 2020, 6, 1–13. [CrossRef]
- Simonsen, L.C.; Slaba, T.C.; Guida, P.; Rusek, A. NASA’s First Ground-Based Galactic Cosmic Ray Simulator: Enabling a New Era in Space Radiobiology Research. PLOS Biology 2020, 18, e3000669. [CrossRef]
- Schwadron, N.A.; Cooper, J.F.; Desai, M.; Downs, C.; Gorby, M.; Jordan, A.P.; Joyce, C.J.; Kozarev, K.; Linker, J.A.; Mikíc, Z.; et al. Particle Radiation Sources, Propagation and Interactions in Deep Space, at Earth, the Moon, Mars, and Beyond: Examples of Radiation Interactions and Effects. Space Sci Rev 2017, 212, 1069–1106. [CrossRef]
- Nelson, G.A. Space Radiation and Human Exposures, A Primer. rare 2016, 185, 349–358. [CrossRef]
- Reames, D.V. Solar Energetic Particles: A Paradigm Shift. Reviews of Geophysics 1995, 33, 585–589. [CrossRef]
- Mewaldt, R.A. Elemental Composition and Energy Spectra of Galactic Cosmic Rays. 1988.
- ACES 2015.
- Li, W.; Hudson, M. k. Earth’s Van Allen Radiation Belts: From Discovery to the Van Allen Probes Era. Journal of Geophysical Research: Space Physics 2019, 124, 8319–8351. [CrossRef]
- Sources and Effects of Ionizing Radiation: UNSCEAR 2008 Report to the General Assembly with Scientific Annexes. Volume 2, Scientific Annexes C, D and E 2011.
- Zeitlin, C.; Hassler, D.M.; Cucinotta, F.A.; Ehresmann, B.; Wimmer-Schweingruber, R.F.; Brinza, D.E.; Kang, S.; Weigle, G.; Böttcher, S.; Böhm, E.; et al. Measurements of Energetic Particle Radiation in Transit to Mars on the Mars Science Laboratory. Science 2013, 340, 1080–1084. [CrossRef]
- Lin, E.C. Radiation Risk From Medical Imaging. Mayo Clin Proc 2010, 85, 1142–1146. [CrossRef]
- Boscolo, D.; Durante, M. Dose Limits and Countermeasures for Mitigating Radiation Risk in Moon and Mars Exploration. Physics 2022, 4, 172–184. [CrossRef]
- Bahadori, A.A. Space Radiation Protection in the Modern Era: New Approaches to Familiar Challenges. Radiation Physics and Chemistry 2024, 221, 111764. [CrossRef]
- Straume, T. Space Radiation Effects on Crew During and After Deep Space Missions. Curr Pathobiol Rep 2018, 6, 167–175. [CrossRef]
- Danho, S.; Thorgrimson, J.; Saary, J. Effects of Space Radiation on Mammalian Cells. In Handbook of Space Pharmaceuticals; Springer, Cham, 2022; pp. 371–388 ISBN 978-3-030-05526-4.
- Hu, S.; Barzilla, J.; Semones, E. Acute Radiation Risk Assessment and Mitigation Strategies in Near Future Exploration Spaceflights. Life Sciences in Space Research 2019, 24. [CrossRef]
- Werneth, C.M.; Huff, J.L. Chapter 2 - The Space Radiation Environment and Human Health Risks. In Precision Medicine for Long and Safe Permanence of Humans in Space; Krittanawong, C., Ed.; Academic Press, 2025; pp. 11–44 ISBN 978-0-443-22259-7.
- Arnautou, P.; Garnier, G.; Maillot, J.; Konopacki, J.; Brachet, M.; Bonnin, A.; Amabile, J.-C.; Malfuson, J.-V. Management of Acute Radiation Syndrome. Transfus Clin Biol 2024, 31, 253–259. [CrossRef]
- Linet, M.S.; Kazzi, Z.; Paulson, J.A.; COUNCIL ON ENVIRONMENTAL HEALTH; Lowry, J.A.; Ahdoot, S.; Baum, C.R.; Bernstein, A.S.; Bole, A.; Byron, L.G.; et al. Pediatric Considerations Before, During, and After Radiological or Nuclear Emergencies. Pediatrics 2018, 142, e20183001. [CrossRef]
- Wu, H.; Huff, J.L.; Casey, R.; Kim, M.-H.; Cucinotta, F.A. Risk of Acute Radiation Syndromes Due to Solar Particle Events.
- NASA Spaceflight Human-System Standard Volume 1, Crew Health | Standards Available online: https://standards.nasa.gov/standard/NASA/NASA-STD-3001_VOL_1 (accessed on 16 June 2025).
- Delp, M.D.; Charvat, J.M.; Limoli, C.L.; Globus, R.K.; Ghosh, P. Apollo Lunar Astronauts Show Higher Cardiovascular Disease Mortality: Possible Deep Space Radiation Effects on the Vascular Endothelium. Sci Rep 2016, 6, 29901. [CrossRef]
- Barravecchia, I.; De Cesari, C.; Forcato, M.; Scebba, F.; Pyankova, O.V.; Bridger, J.M.; Foster, H.A.; Signore, G.; Borghini, A.; Andreassi, M.; et al. Microgravity and Space Radiation Inhibit Autophagy in Human Capillary Endothelial Cells, through Either Opposite or Synergistic Effects on Specific Molecular Pathways. Cell Mol Life Sci 2021, 79, 28. [CrossRef]
- Hughson, R.L.; Helm, A.; Durante, M. Heart in Space: Effect of the Extraterrestrial Environment on the Cardiovascular System. Nat Rev Cardiol 2018, 15, 167–180. [CrossRef]
- Carr, Z.A.; Land, C.E.; Kleinerman, R.A.; Weinstock, R.W.; Stovall, M.; Griem, M.L.; Mabuchi, K. Coronary Heart Disease after Radiotherapy for Peptic Ulcer Disease. International Journal of Radiation Oncology*Biology*Physics 2005, 61, 842–850. [CrossRef]
- Shimizu, Y.; Kodama, K.; Nishi, N.; Kasagi, F.; Suyama, A.; Soda, M.; Grant, E.J.; Sugiyama, H.; Sakata, R.; Moriwaki, H.; et al. Radiation Exposure and Circulatory Disease Risk: Hiroshima and Nagasaki Atomic Bomb Survivor Data, 1950-2003. BMJ 2010, 340, b5349. [CrossRef]
- Sasaki, H.; Wong, F.L.; Yamada, M.; Kodama, K. The Effects of Aging and Radiation Exposure on Blood Pressure Levels of Atomic Bomb Survivors. Journal of Clinical Epidemiology 2002, 55, 974–981. [CrossRef]
- Shrestha, S.; Bates, J.E.; Liu, Q.; Smith, S.A.; Oeffinger, K.C.; Chow, E.J.; Gupta, A.C.; Owens, C.A.; Constine, L.S.; Hoppe, B.S.; et al. Radiation Therapy Related Cardiac Disease Risk in Childhood Cancer Survivors: Updated Dosimetry Analysis from the Childhood Cancer Survivor Study. Radiotherapy and Oncology 2021, 163, 199–208. [CrossRef]
- Wilson, J.; Jun Hua, C.; Aziminia, N.; Manisty, C. Imaging of the Acute and Chronic Cardiovascular Complications of Radiation Therapy. Circ Cardiovasc Imaging 2025, 18, e017454. [CrossRef]
- Yu, T.; Parks, B.W.; Yu, S.; Srivastava, R.; Gupta, K.; Wu, X.; Khaled, S.; Chang, P.Y.; Kabarowski, J.H.; Kucik, D.F. Iron-Ion Radiation Accelerates Atherosclerosis in Apolipoprotein E-Deficient Mice. Radiat Res 2011, 175, 766–773. [CrossRef]
- Soucy, K.G.; Lim, H.K.; Kim, J.H.; Oh, Y.; Attarzadeh, D.O.; Sevinc, B.; Kuo, M.M.; Shoukas, A.A.; Vazquez, M.E.; Berkowitz, D.E. HZE 56Fe-Ion Irradiation Induces Endothelial Dysfunction in Rat Aorta: Role of Xanthine Oxidase. Radiat Res 2011, 176, 474–485. [CrossRef]
- White, C.R.; Yu, T.; Gupta, K.; Babitz, S.K.; Black, L.L.; Kabarowski, J.H.; Kucik, D.F. Early Changes in Vascular Reactivity in Response to 56Fe Irradiation in ApoE-/- Mice. Acta Astronautica 2015, 108, 40–45. [CrossRef]
- Yan, X.; Sasi, S.P.; Gee, H.; Lee, J.; Yang, Y.; Mehrzad, R.; Onufrak, J.; Song, J.; Enderling, H.; Agarwal, A.; et al. Cardiovascular Risks Associated with Low Dose Ionizing Particle Radiation. PLoS ONE 2014, 9, e110269. [CrossRef]
- Lenarczyk, M.; Kronenberg, A.; Mäder, M.; Komorowski, R.; Hopewell, J.W.; Baker, J.E. Exposure to Multiple Ion Beams, Broadly Representative of Galactic Cosmic Rays, Causes Perivascular Cardiac Fibrosis in Mature Male Rats. PLOS ONE 2023, 18, e0283877. [CrossRef]
- Nemec-Bakk, A.S.; Sridharan, V.; Desai, P.; Landes, R.D.; Hart, B.; Allen, A.R.; Boerma, M. Effects of Simulated 5-Ion Galactic Cosmic Radiation on Function and Structure of the Mouse Heart. Life 2023, 13, 795. [CrossRef]
- Ali, Y.F.; Cucinotta, F.A.; Ning-Ang, L.; Zhou, G. Cancer Risk of Low Dose Ionizing Radiation. Front. Phys. 2020, 8. [CrossRef]
- Ozasa, K. Epidemiological Research on Radiation-Induced Cancer in Atomic Bomb Survivors. J Radiat Res 2016, 57, i112–i117. [CrossRef]
- Cardis, E.; Hatch, M. The Chernobyl Accident — an Epidemiological Perspective. Clin Oncol (R Coll Radiol) 2011, 23, 251–260. [CrossRef]
- Richardson, D.B.; Cardis, E.; Daniels, R.D.; Gillies, M.; O’Hagan, J.A.; Hamra, G.B.; Haylock, R.; Laurier, D.; Leuraud, K.; Moissonnier, M.; et al. Risk of Cancer from Occupational Exposure to Ionising Radiation: Retrospective Cohort Study of Workers in France, the United Kingdom, and the United States (INWORKS). 2015. [CrossRef]
- Reisz, J.A.; Bansal, N.; Qian, J.; Zhao, W.; Furdui, C.M. Effects of Ionizing Radiation on Biological Molecules—Mechanisms of Damage and Emerging Methods of Detection. Antioxid Redox Signal 2014, 21, 260–292. [CrossRef]
- Kumar, K.; Kumar, S.; Datta, K.; Fornace, A.J.; Suman, S. High-LET-Radiation-Induced Persistent DNA Damage Response Signaling and Gastrointestinal Cancer Development. Curr Oncol 2023, 30, 5497–5514. [CrossRef]
- Danforth, J.M.; Provencher, L.; Goodarzi, A.A. Chromatin and the Cellular Response to Particle Radiation-Induced Oxidative and Clustered DNA Damage. Front. Cell Dev. Biol. 2022, 10. [CrossRef]
- Guo, Z.; Zhou, G.; Hu, W. Carcinogenesis Induced by Space Radiation: A Systematic Review. Neoplasia 2022, 32, 100828. [CrossRef]
- Muhsen, I.N.; Zubair, A.C.; Niederwieser, T.; Hashmi, S.K. Space Exploration and Cancer: The Risks of Deeper Space Adventures. Leukemia 2024, 38, 1872–1875. [CrossRef]
- Reynolds, R.; Little, M.P.; Day, S.; Charvat, J.; Blattnig, S.; Huff, J.; Patel, Z.S. Cancer Incidence and Mortality in the USA Astronaut Corps, 1959–2017. Occup Environ Med 2021, 78, 869–875. [CrossRef]
- Elgart, S.R.; Little, M.P.; Chappell, L.J.; Milder, C.M.; Shavers, M.R.; Huff, J.L.; Patel, Z.S. Radiation Exposure and Mortality from Cardiovascular Disease and Cancer in Early NASA Astronauts. Sci Rep 2018, 8, 8480. [CrossRef]
- Reynolds, R.J.; Day, S.M.; Reynolds, R.J.; Day, S.M. The Mortality of Space Explorers. In Into Space - A Journey of How Humans Adapt and Live in Microgravity; IntechOpen, 2018 ISBN 978-1-78923-221-9.
- Reynolds, R.J.; Day, S.M. Mortality of US Astronauts: Comparisons with Professional Athletes. Occup Environ Med 2019, 76, 114–117. [CrossRef]
- Miura, K.; Olsen, C.M.; Rea, S.; Marsden, J.; Green, A.C. Do Airline Pilots and Cabin Crew Have Raised Risks of Melanoma and Other Skin Cancers? Systematic Review and Meta-analysis. British Journal of Dermatology 2019, 181, 55–64. [CrossRef]
- Sanlorenzo, M.; Wehner, M.R.; Linos, E.; Kornak, J.; Kainz, W.; Posch, C.; Vujic, I.; Johnston, K.; Gho, D.; Monico, G.; et al. The Risk of Melanoma in Airline Pilots and Cabin Crew: A Meta-Analysis. JAMA Dermatology 2015, 151, 51–58. [CrossRef]
- Beheshti, A.; McDonald, J.T.; Hada, M.; Takahashi, A.; Mason, C.E.; Mognato, M. Genomic Changes Driven by Radiation-Induced DNA Damage and Microgravity in Human Cells. International Journal of Molecular Sciences 2021, 22, 10507. [CrossRef]
- Han, Z.-B.; Suzuki, H.; Suzuki, F.; Suzuki, M.; Furusawa, Y.; Kato, T.; Ikenaga, M. Relative Biological Effectiveness of Accelerated Heavy Ions for Induction of Morphological Transformation in Syrian Hamster Embryo Cells. Journal of Radiation Research 1998, 39, 193–201. [CrossRef]
- Wang, M.; Hada, M.; Huff, J.; Pluth, J.M.; Anderson, J.; O’ Neill, P.; Cucinotta, F.A. Heavy Ions Can Enhance TGFβ Mediated Epithelial to Mesenchymal Transition. Journal of Radiation Research 2012, 53, 51–57. [CrossRef]
- Ding, L.-H.; Park, S.; Xie, Y.; Girard, L.; Minna, J.D.; Story, M.D. Elucidation of Changes in Molecular Signalling Leading to Increased Cellular Transformation in Oncogenically Progressed Human Bronchial Epithelial Cells Exposed to Radiations of Increasing LET. Mutagenesis 2015, 30, 685–694. [CrossRef]
- Kennedy, E.M.; Powell, D.R.; Li, Z.; Bell, J.S.K.; Barwick, B.G.; Feng, H.; McCrary, M.R.; Dwivedi, B.; Kowalski, J.; Dynan, W.S.; et al. Galactic Cosmic Radiation Induces Persistent Epigenome Alterations Relevant to Human Lung Cancer. Sci Rep 2018, 8, 6709. [CrossRef]
- Rodman, C.; Almeida-Porada, G.; George, S.; Moon, J.; Soker, S.; Pardee, T.; Beaty, M.; Guida, P.; Sajuthi, S.; Langefeld, C.; et al. In Vitro and in Vivo Assessment of Direct Effects of Simulated Solar and Galactic Cosmic Radiation on Human Hematopoietic Stem/Progenitor Cells. Leukemia 2017, 31, 1398–1407. [CrossRef]
- Shao, C.; Aoki, M.; Furusawa, Y. Bystander Effect on Cell Growth Stimulation in Neoplastic HSGc Cells Induced by Heavy-Ion Irradiation. Radiat Environ Biophys 2003, 42, 183–187. [CrossRef]
- Buonanno, M.; Toledo, S.M. de; Azzam, E.I. Increased Frequency of Spontaneous Neoplastic Transformation in Progeny of Bystander Cells from Cultures Exposed to Densely Ionizing Radiation. PLOS ONE 2011, 6, e21540. [CrossRef]
- Guracho, A.N.; Strigari, L.; GAlA, G.D.; Paolani, G.; Santoro, M.; Strolin, S.; Bartoloni, A. Space Radiation-Induced Bystander Effect in Estimating the Carcinogenic Risk Due to Galactic Cosmic Rays. J. Mech. Med. Biol. 2023, 23, 2340023. [CrossRef]
- Peng, Y.; Brown, N.; Finnon, R.; Warner, C.L.; Liu, X.; Genik, P.C.; Callan, M.A.; Ray, F.A.; Borak, T.B.; Badie, C.; et al. Radiation Leukemogenesis in Mice: Loss of PU.1 on Chromosome 2 in CBA and C57BL/6 Mice after Irradiation with 1 GeV/Nucleon 56Fe Ions, X Rays or γ Rays. Part I. Experimental Observations. rare 2009, 171, 474–483. [CrossRef]
- Weil, M.M.; Bedford, J.S.; Bielefeldt-Ohmann, H.; Ray, F.A.; Genik, P.C.; Ehrhart, E.J.; Fallgren, C.M.; Hailu, F.; Battaglia, C.L.R.; Charles, B.; et al. Incidence of Acute Myeloid Leukemia and Hepatocellular Carcinoma in Mice Irradiated with 1 GeV/Nucleon 56Fe Ions. rare 2009, 172, 213–219. [CrossRef]
- Weil, M.M.; Ray, F.A.; Genik, P.C.; Yu, Y.; McCarthy, M.; Fallgren, C.M.; Ullrich, R.L. Effects of 28Si Ions, 56Fe Ions, and Protons on the Induction of Murine Acute Myeloid Leukemia and Hepatocellular Carcinoma. PLOS ONE 2014, 9, e104819. [CrossRef]
- Wang, X.; Iii, A.B.F.; Wang, P.; Zhang, X.; Wang, H.; Wang, Y. Relative Effectiveness at 1 Gy after Acute and Fractionated Exposures of Heavy Ions with Different Linear Energy Transfer for Lung Tumorigenesis. rare 2015, 183, 233–239. [CrossRef]
- Finkelstein, S.R.; Patel, R.; Deland, K.; Mercer, J.; Starr, B.; Zhu, D.; Min, H.; Reinsvold, M.; Campos, L.D.S.; Williams, N.T.; et al. 56Fe-Ion Exposure Increases the Incidence of Lung and Brain Tumors at a Similar Rate in Male and Female Mice. rare 2024, 202, 734–744. [CrossRef]
- Luitel, K.; Siteni, S.; Barron, S.; Shay, J.W. Simulated Galactic Cosmic Radiation-Induced Cancer Progression in Mice. Life Sciences in Space Research 2024, 41, 43–51. [CrossRef]
- Watanabe, H.; Ogiu, T.; Nishizaki, M.; Fujimoto, N.; Kido, S.; Ishimura, Y.; Shiraki, K.; Kuramoto, K.; Hirata, S.; Shoji, S.; et al. Induction of Ovarian Tumors by Heavy Ion Irradiation in B6C3F1 Mice. Oncol Rep 1998, 5, 1377–1380. [CrossRef]
- Mishra, B.; Lawson, G.W.; Ripperdan, R.; Ortiz, L.; Luderer, U. Charged-Iron-Particles Found in Galactic Cosmic Rays Are Potent Inducers of Epithelial Ovarian Tumors. Radiat Res 2018, 190, 142–150. [CrossRef]
- Nia, A.M.; Shavkunov, A.; Ullrich, R.L.; Emmett, M.R. 137Cs γ Ray and 28Si Irradiation Induced Murine Hepatocellular Carcinoma Lipid Changes in Liver Assessed by MALDI-MSI Combined with Spatial Shrunken Centroid Clustering Algorithm: A Pilot Study. ACS Omega 2020, 5, 25164–25174. [CrossRef]
- Trani, D.; Datta, K.; Doiron, K.; Kallakury, B.; Fornace, A.J. Enhanced Intestinal Tumor Multiplicity and Grade in Vivo after HZE Exposure: Mouse Models for Space Radiation Risk Estimates. Radiat Environ Biophys 2010, 49, 389–396. [CrossRef]
- Datta, K.; Suman, S.; Kumar, S.; Fornace, A.J. Colorectal Carcinogenesis, Radiation Quality, and the Ubiquitin-Proteasome Pathway. J Cancer 2016, 7, 174–183. [CrossRef]
- Suman, S.; Kumar, S.; Fornace, A.J.; Datta, K. Decreased RXRα Is Associated with Increased β-Catenin/TCF4 in 56Fe-Induced Intestinal Tumors. Front. Oncol. 2015, 5. [CrossRef]
- Suman, S.; Kumar, S.; Moon, B.-H.; Strawn, S.J.; Thakor, H.; Fan, Z.; Shay, J.W.; Fornace, A.J.; Datta, K. Relative Biological Effectiveness of Energetic Heavy Ions for Intestinal Tumorigenesis Shows Male Preponderance and Radiation Type and Energy Dependence in APC1638N/+ Mice. International Journal of Radiation Oncology, Biology, Physics 2016, 95, 131–138. [CrossRef]
- Imaoka, T.; Nishimura, M.; Kakinuma, S.; Hatano, Y.; Ohmachi, Y.; Yoshinaga, S.; Kawano, A.; Maekawa, A.; Shimada, Y. High Relative Biologic Effectiveness of Carbon Ion Radiation on Induction of Rat Mammary Carcinoma and Its Lack of H-Ras and Tp53 Mutations. International Journal of Radiation Oncology, Biology, Physics 2007, 69, 194–203. [CrossRef]
- Illa-Bochaca, I.; Ouyang, H.; Tang, J.; Sebastiano, C.; Mao, J.-H.; Costes, S.V.; Demaria, S.; Barcellos-Hoff, M.H. Densely Ionizing Radiation Acts via the Microenvironment to Promote Aggressive Trp53-Null Mammary Carcinomas. Cancer Research 2014, 74, 7137–7148. [CrossRef]
- Hada, M.; Ikeda, H.; Rhone, J.R.; Beitman, A.J.; Plante, I.; Souda, H.; Yoshida, Y.; Held, K.D.; Fujiwara, K.; Saganti, P.B.; et al. Increased Chromosome Aberrations in Cells Exposed Simultaneously to Simulated Microgravity and Radiation. Int J Mol Sci 2018, 20, 43. [CrossRef]
- Yamanouchi, S.; Rhone, J.; Mao, J.-H.; Fujiwara, K.; Saganti, P.B.; Takahashi, A.; Hada, M. Simultaneous Exposure of Cultured Human Lymphoblastic Cells to Simulated Microgravity and Radiation Increases Chromosome Aberrations. Life (Basel) 2020, 10, 187. [CrossRef]
- Narici, L. Light Flashes and Other Sensory Illusions Perceived in Space Travel and on Ground, Including Proton and Heavy Ion Therapies. Z Med Phys 2023, 34, 44–63. [CrossRef]
- Pinsky, L.S.; Osborne, W.Z.; Bailey, J.V.; Benson, R.E.; Thompson, L.F. Light Flashes Observed by Astronauts on Apollo 11 through Apollo 17. Science 1974, 183, 957–959. [CrossRef]
- Pinsky, L.S.; Osborne, W.Z.; Hoffman, R.A.; Bailey, J.V. Light Flashes Observed by Astronauts on Skylab 4. Science 1975, 188, 928–930. [CrossRef]
- Avdeev, S.; Bidoli, V.; Casolino, M.; De Grandis, E.; Furano, G.; Morselli, A.; Narici, L.; De Pascale, M.P.; Picozza, P.; Reali, E.; et al. Eye Light Flashes on the Mir Space Station. Acta Astronaut 2002, 50, 511–525. [CrossRef]
- Casolino, M.; Bidoli, V.; Morselli, A.; Narici, L.; De Pascale, M.P.; Picozza, P.; Reali, E.; Sparvoli, R.; Mazzenga, G.; Ricci, M.; et al. Dual Origins of Light Flashes Seen in Space. Nature 2003, 422, 680–680. [CrossRef]
- Mesny, E.; Jacob, J.; Noël, G.; Bernier, M.-O.; Ricard, D. Specific Radiosensitivity of Brain Structures (Areas or Regions) and Cognitive Impairment after Focal or Whole Brain Radiotherapy: A Review. Cancer/Radiothérapie 2025, 29, 104625. [CrossRef]
- Surendran, H.P.; Sah, S.K.; Veeralakshmanan, P.; Nair, P.; Ashok, H.P.; Unnikrishnan, M.K.; Kalavagunta, S.; Sasidharan, A.; Chandran, D.; Poornachary, N.M.; et al. Efficacy of Hippocampal Avoidance Whole Brain Radiotherapy to Preserve the Cognitive Functions among Brain Metastasis Patients: Systematic Review and Meta-Analysis. Neurology India 2025, 73, 429. [CrossRef]
- Azizova, T.V.; Bannikova, M.V.; Grigoryeva, E.S.; Rybkina, V.L.; Hamada, N. Occupational Exposure to Chronic Ionizing Radiation Increases Risk of Parkinson’s Disease Incidence in Russian Mayak Workers. International Journal of Epidemiology 2020, 49, 435–447. [CrossRef]
- Dauer, L.T.; Walsh, L.; Mumma, M.T.; Cohen, S.S.; Golden, A.P.; Howard, S.C.; Roemer, G.E.; Boice, J.D. Moon, Mars and Minds: Evaluating Parkinson’s Disease Mortality among U.S. Radiation Workers and Veterans in the Million Person Study of Low-Dose Effects. Z Med Phys 2023, 34, 100–110. [CrossRef]
- Kiffer, F.; Boerma, M.; Allen, A. Behavioral Effects of Space Radiation: A Comprehensive Review of Animal Studies. Life Sci Space Res (Amst) 2019, 21, 1–21. [CrossRef]
- Britten, R.A.; Davis, L.K.; Johnson, A.M.; Keeney, S.; Siegel, A.; Sanford, L.D.; Singletary, S.J.; Lonart, G. Low (20 cGy) Doses of 1 GeV/u (56)Fe--Particle Radiation Lead to a Persistent Reduction in the Spatial Learning Ability of Rats. Radiat Res 2012, 177, 146–151. [CrossRef]
- Haley, G.E.; Yeiser, L.; Olsen, R.H.J.; Davis, M.J.; Johnson, L.A.; Raber, J. Early Effects of Whole-Body 56Fe Irradiation on Hippocampal Function in C57BL/6J Mice. Radiat Res 2013, 179, 590–596. [CrossRef]
- Rabin, B.M.; Poulose, S.M.; Carrihill-Knoll, K.L.; Ramirez, F.; Bielinski, D.F.; Heroux, N.; Shukitt-Hale, B. Acute Effects of Exposure to (56)Fe and (16)O Particles on Learning and Memory. Radiat Res 2015, 184, 143–150. [CrossRef]
- Carr, H.; Alexander, T.C.; Groves, T.; Kiffer, F.; Wang, J.; Price, E.; Boerma, M.; Allen, A.R. Early Effects of 16O Radiation on Neuronal Morphology and Cognition in a Murine Model. Life Sci Space Res (Amst) 2018, 17, 63–73. [CrossRef]
- Howe, A.; Kiffer, F.; Alexander, T.C.; Sridharan, V.; Wang, J.; Ntagwabira, F.; Rodriguez, A.; Boerma, M.; Allen, A.R. Long-Term Changes in Cognition and Physiology after Low-Dose 16O Irradiation. Int J Mol Sci 2019, 20, 188. [CrossRef]
- Hassler, D.M.; Zeitlin, C.; Wimmer-Schweingruber, R.F.; Ehresmann, B.; Rafkin, S.; Eigenbrode, J.L.; Brinza, D.E.; Weigle, G.; Böttcher, S.; Böhm, E.; et al. Mars’ Surface Radiation Environment Measured with the Mars Science Laboratory’s Curiosity Rover. Science 2014, 343, 1244797. [CrossRef]
- Liston, A.; Humblet-Baron, S.; Duffy, D.; Goris, A. Human Immune Diversity: From Evolution to Modernity. Nat Immunol 2021, 22, 1479–1489. [CrossRef]
- Marchal, S.; Choukér, A.; Bereiter-Hahn, J.; Kraus, A.; Grimm, D.; Krüger, M. Challenges for the Human Immune System after Leaving Earth. NPJ Microgravity 2024, 10, 106. [CrossRef]
- Wadhwa, A.; Moreno-Villanueva, M.; Crucian, B.; Wu, H. Synergistic Interplay between Radiation and Microgravity in Spaceflight-Related Immunological Health Risks. Immun Ageing 2024, 21, 50. [CrossRef]
- Konstantinova, I.V.; Rykova, M.; Meshkov, D.; Peres, C.; Husson, D.; Schmitt, D.A. Natural Killer Cells after ALTAIR Mission. Acta Astronaut 1995, 36, 713–718. [CrossRef]
- Mills, P.J.; Meck, J.V.; Waters, W.W.; D’Aunno, D.; Ziegler, M.G. Peripheral Leukocyte Subpopulations and Catecholamine Levels in Astronauts as a Function of Mission Duration. Biopsychosocial Science and Medicine 2001, 63, 886.
- Stowe, R.P.; Sams, C.F.; Mehta, S.K.; Kaur, I.; Jones, M.L.; Feeback, D.L.; Pierson, D.L. Leukocyte Subsets and Neutrophil Function after Short-Term Spaceflight. J Leukoc Biol 1999, 65, 179–186. [CrossRef]
- Stowe, R.P.; Sams, C.F.; Pierson, D.L. Effects of Mission Duration on Neuroimmune Responses in Astronauts. Aviat Space Environ Med 2003, 74, 1281–1284.
- Stowe, R.P.; Sams, C.F.; Pierson, D.L. Adrenocortical and Immune Responses Following Short- and Long-Duration Spaceflight. Aviat Space Environ Med 2011, 82, 627–634. [CrossRef]
- Kaur, I.; Simons, E.R.; Castro, V.A.; Mark Ott, C.; Pierson, D.L. Changes in Neutrophil Functions in Astronauts. Brain Behav Immun 2004, 18, 443–450. [CrossRef]
- Kaur, I.; Simons, E.R.; Castro, V.A.; Ott, C.M.; Pierson, D.L. Changes in Monocyte Functions of Astronauts. Brain Behav Immun 2005, 19, 547–554. [CrossRef]
- Crucian, B.; Stowe, R.P.; Mehta, S.; Quiriarte, H.; Pierson, D.; Sams, C. Alterations in Adaptive Immunity Persist during Long-Duration Spaceflight. npj Microgravity 2015, 1, 15013. [CrossRef]
- Bigley, A.B.; Agha, N.H.; Baker, F.L.; Spielmann, G.; Kunz, H.E.; Mylabathula, P.L.; Rooney, B.V.; Laughlin, M.S.; Mehta, S.K.; Pierson, D.L.; et al. NK Cell Function Is Impaired during Long-Duration Spaceflight. Journal of Applied Physiology 2019, 126, 842–853. [CrossRef]
- Irina, V.; Konstantinova, M.D. Immune Resistance of Man in Space Flights. Acta Astronautica 1991, 23, 123–127. [CrossRef]
- Crucian, B.E.; Cubbage, M.L.; Sams, C.F. Altered Cytokine Production by Specific Human Peripheral Blood Cell Subsets Immediately Following Space Flight. Journal of Interferon & Cytokine Research 2000, 20, 547–556. [CrossRef]
- Crucian, B.E.; Zwart, S.R.; Mehta, S.; Uchakin, P.; Quiriarte, H.D.; Pierson, D.; Sams, C.F.; Smith, S.M. Plasma Cytokine Concentrations Indicate That In Vivo Hormonal Regulation of Immunity Is Altered During Long-Duration Spaceflight. J Interferon Cytokine Res 2014, 34, 778–786. [CrossRef]
- Mehta, S.K.; Crucian, B.E.; Stowe, R.P.; Simpson, R.J.; Ott, C.M.; Sams, C.F.; Pierson, D.L. Reactivation of Latent Viruses Is Associated with Increased Plasma Cytokines in Astronauts. Cytokine 2013, 61, 205–209. [CrossRef]
- Buchheim, J.-I.; Matzel, S.; Rykova, M.; Vassilieva, G.; Ponomarev, S.; Nichiporuk, I.; Hörl, M.; Moser, D.; Biere, K.; Feuerecker, M.; et al. Stress Related Shift Toward Inflammaging in Cosmonauts After Long-Duration Space Flight. Front Physiol 2019, 10, 85. [CrossRef]
- Gertz, M.L.; Chin, C.R.; Tomoiaga, D.; MacKay, M.; Chang, C.; Butler, D.; Afshinnekoo, E.; Bezdan, D.; Schmidt, M.A.; Mozsary, C.; et al. Multi-Omic, Single-Cell, and Biochemical Profiles of Astronauts Guide Pharmacological Strategies for Returning to Gravity. Cell Rep 2020, 33, 108429. [CrossRef]
- Krieger, S.S.; Zwart, S.R.; Mehta, S.; Wu, H.; Simpson, R.J.; Smith, S.M.; Crucian, B. Alterations in Saliva and Plasma Cytokine Concentrations During Long-Duration Spaceflight. Front Immunol 2021, 12, 725748. [CrossRef]
- Buchheim, J.-I.; Ghislin, S.; Ouzren, N.; Albuisson, E.; Vanet, A.; Matzel, S.; Ponomarev, S.; Rykova, M.; Choukér, A.; Frippiat, J.-P. Plasticity of the Human IgM Repertoire in Response to Long-Term Spaceflight. The FASEB Journal 2020, 34, 16144–16162. [CrossRef]
- Rooney, B.V.; Crucian, B.E.; Pierson, D.L.; Laudenslager, M.L.; Mehta, S.K. Herpes Virus Reactivation in Astronauts During Spaceflight and Its Application on Earth. Front Microbiol 2019, 10, 16. [CrossRef]
- Agha, N.H.; Baker, F.L.; Kunz, H.E.; Spielmann, G.; Mylabathula, P.L.; Rooney, B.V.; Mehta, S.K.; Pierson, D.L.; Laughlin, M.S.; Markofski, M.M.; et al. Salivary Antimicrobial Proteins and Stress Biomarkers Are Elevated during a 6-Month Mission to the International Space Station. J Appl Physiol (1985) 2020, 128, 264–275. [CrossRef]
- Fulop, T.; Larbi, A.; Dupuis, G.; Le Page, A.; Frost, E.H.; Cohen, A.A.; Witkowski, J.M.; Franceschi, C. Immunosenescence and Inflamm-Aging As Two Sides of the Same Coin: Friends or Foes? Front. Immunol. 2018, 8. [CrossRef]
- Mermel, L.A. Infection Prevention and Control during Prolonged Human Space Travel. Clin Infect Dis 2013, 56, 123–130. [CrossRef]
- Stahn, A.C.; Werner, A.; Opatz, O.; Maggioni, M.A.; Steinach, M.; von Ahlefeld, V.W.; Moore, A.; Crucian, B.E.; Smith, S.M.; Zwart, S.R.; et al. Increased Core Body Temperature in Astronauts during Long-Duration Space Missions. Sci Rep 2017, 7, 16180. [CrossRef]
- Payne, D.A.; Mehta, S.K.; Tyring, S.K.; Stowe, R.P.; Pierson, D.L. Incidence of Epstein-Barr Virus in Astronaut Saliva during Spaceflight. Aviat Space Environ Med 1999, 70, 1211–1213.
- Cohrs, R.J.; Mehta, S.K.; Schmid, D.S.; Gilden, D.H.; Pierson, D.L. Asymptomatic Reactivation and Shed of Infectious Varicella Zoster Virus in Astronauts. J Med Virol 2008, 80, 1116–1122. [CrossRef]
- Mehta, S.K.; Stowe, R.P.; Feiveson, A.H.; Tyring, S.K.; Pierson, D.L. Reactivation and Shedding of Cytomegalovirus in Astronauts during Spaceflight. The Journal of Infectious Diseases 2000, 182, 1761–1764. [CrossRef]
- Mehta, S.K.; Cohrs, R.J.; Forghani, B.; Zerbe, G.; Gilden, D.H.; Pierson, D.L. Stress-Induced Subclinical Reactivation of Varicella Zoster Virus in Astronauts. Journal of Medical Virology 2004, 72, 174–179. [CrossRef]
- Mehta, S.K.; Laudenslager, M.L.; Stowe, R.P.; Crucian, B.E.; Sams, C.F.; Pierson, D.L. Multiple Latent Viruses Reactivate in Astronauts during Space Shuttle Missions. Brain Behav Immun 2014, 41, 210–217. [CrossRef]
- Mehta, S.K.; Laudenslager, M.L.; Stowe, R.P.; Crucian, B.E.; Feiveson, A.H.; Sams, C.F.; Pierson, D.L. Latent Virus Reactivation in Astronauts on the International Space Station. npj Microgravity 2017, 3, 11. [CrossRef]
- Pierson, D.L.; Stowe, R.P.; Phillips, T.M.; Lugg, D.J.; Mehta, S.K. Epstein-Barr Virus Shedding by Astronauts during Space Flight. Brain Behav Immun 2005, 19, 235–242. [CrossRef]
- McDonagh, F.; Cormican, M.; Morris, D.; Burke, L.; Singh, N.K.; Venkateswaran, K.; Miliotis, G. Medical Astro-Microbiology: Current Role and Future Challenges. J Indian Inst Sci 2023, 1–26. [CrossRef]
- Klaus, D.; Simske, S.; Todd, P.; Stodieck, L. Investigation of Space Flight Effects on Escherichia Coli and a Proposed Model of Underlying Physical Mechanisms. Microbiology 1997, 143, 449–455. [CrossRef]
- Kacena, M.A.; Merrell, G.A.; Manfredi, B.; Smith, E.E.; Klaus, D.M.; Todd, P. Bacterial Growth in Space Flight: Logistic Growth Curve Parameters for Escherichia Coli and Bacillus Subtilis. Appl Microbiol Biotechnol 1999, 51, 229–234. [CrossRef]
- Wilson, J.W.; Ott, C.M.; zu Bentrup, K.H.; Ramamurthy, R.; Quick, L.; Porwollik, S.; Cheng, P.; McClelland, M.; Tsaprailis, G.; Radabaugh, T.; et al. Space Flight Alters Bacterial Gene Expression and Virulence and Reveals a Role for Global Regulator Hfq. Proc Natl Acad Sci U S A 2007, 104, 16299–16304. [CrossRef]
- Crabbé, A.; Schurr, M.J.; Monsieurs, P.; Morici, L.; Schurr, J.; Wilson, J.W.; Ott, C.M.; Tsaprailis, G.; Pierson, D.L.; Stefanyshyn-Piper, H.; et al. Transcriptional and Proteomic Responses of Pseudomonas Aeruginosa PAO1 to Spaceflight Conditions Involve Hfq Regulation and Reveal a Role for Oxygen. Appl Environ Microbiol 2011, 77, 1221–1230. [CrossRef]
- Gilbert, R.; Torres, M.; Clemens, R.; Hateley, S.; Hosamani, R.; Wade, W.; Bhattacharya, S. Spaceflight and Simulated Microgravity Conditions Increase Virulence of Serratia Marcescens in the Drosophila Melanogaster Infection Model. npj Microgravity 2020, 6, 4. [CrossRef]
- Schiwon, K.; Arends, K.; Rogowski, K.M.; Fürch, S.; Prescha, K.; Sakinc, T.; Van Houdt, R.; Werner, G.; Grohmann, E. Comparison of Antibiotic Resistance, Biofilm Formation and Conjugative Transfer of Staphylococcus and Enterococcus Isolates from International Space Station and Antarctic Research Station Concordia. Microb Ecol 2013, 65, 638–651. [CrossRef]
- Singh, N.K.; Bezdan, D.; Checinska Sielaff, A.; Wheeler, K.; Mason, C.E.; Venkateswaran, K. Multi-Drug Resistant Enterobacter Bugandensis Species Isolated from the International Space Station and Comparative Genomic Analyses with Human Pathogenic Strains. BMC Microbiology 2018, 18, 175. [CrossRef]
- Urbaniak, C.; Sielaff, A.C.; Frey, K.G.; Allen, J.E.; Singh, N.; Jaing, C.; Wheeler, K.; Venkateswaran, K. Detection of Antimicrobial Resistance Genes Associated with the International Space Station Environmental Surfaces. Sci Rep 2018, 8, 814. [CrossRef]
- Zhang, B.; Bai, P.; Zhao, X.; Yu, Y.; Zhang, X.; Li, D.; Liu, C. Increased Growth Rate and Amikacin Resistance of Salmonella Enteritidis after One-month Spaceflight on China’s Shenzhou-11 Spacecraft. Microbiologyopen 2019, 8, e00833. [CrossRef]
- Knox, B.P.; Blachowicz, A.; Palmer, J.M.; Romsdahl, J.; Huttenlocher, A.; Wang, C.C.C.; Keller, N.P.; Venkateswaran, K. Characterization of Aspergillus Fumigatus Isolates from Air and Surfaces of the International Space Station. mSphere 2016, 1, e00227-16. [CrossRef]
- Singh, N.K.; Blachowicz, A.; Checinska, A.; Wang, C.; Venkateswaran, K. Draft Genome Sequences of Two Aspergillus Fumigatus Strains, Isolated from the International Space Station. Genome Announc 2016, 4, e00553-16. [CrossRef]
- Urbaniak, C.; van Dam, P.; Zaborin, A.; Zaborina, O.; Gilbert, J.A.; Torok, T.; Wang, C.C.C.; Venkateswaran, K. Genomic Characterization and Virulence Potential of Two Fusarium Oxysporum Isolates Cultured from the International Space Station. mSystems 2019, 4, e00345-18. [CrossRef]
- Nielsen, S.; White, K.; Preiss, K.; Peart, D.; Gianoulias, K.; Juel, R.; Sutton, J.; McKinney, J.; Bender, J.; Pinc, G.; et al. Growth and Antifungal Resistance of the Pathogenic Yeast, Candida Albicans, in the Microgravity Environment of the International Space Station: An Aggregate of Multiple Flight Experiences. Life (Basel) 2021, 11, 283. [CrossRef]
- Arone, A.; Ivaldi, T.; Loganovsky, K.; Palermo, S.; Parra, E.; Flamini, W.; Marazziti, D. The Burden of Space Exploration on the Mental Health of Astronauts: A Narrative Review. Clin Neuropsychiatry 18, 237–246. [CrossRef]
- Oluwafemi, F.A.; Abdelbaki, R.; Lai, J.C.-Y.; Mora-Almanza, J.G.; Afolayan, E.M. A Review of Astronaut Mental Health in Manned Missions: Potential Interventions for Cognitive and Mental Health Challenges. Life Sci Space Res (Amst) 2021, 28, 26–31. [CrossRef]
- Yin, Y.; Liu, J.; Fan, Q.; Zhao, S.; Wu, X.; Wang, J.; Liu, Y.; Li, Y.; Lu, W. Long-Term Spaceflight Composite Stress Induces Depression and Cognitive Impairment in Astronauts—Insights from Neuroplasticity. Transl Psychiatry 2023, 13, 342. [CrossRef]
- Wu, B.; Wang, Y.; Wu, X.; Liu, D.; Xu, D.; Wang, F. On-Orbit Sleep Problems of Astronauts and Countermeasures. Mil Med Res 2018, 5, 17. [CrossRef]
- Flynn-Evans, E.; Gregory, K.; Arsintescu, L.; Whitmire, A. Evidence Report: Performance Decrements and Adverse Health Outcomes Resulting from Sleep-Loss, Circadian Desynchronization, and Work-Overload.; 2016;
- Gundel, A.; Polyakov, V.V.; Zulley, J. The Alteration of Human Sleep and Circadian Rhythms during Spaceflight. J Sleep Res 1997, 6, 1–8. [CrossRef]
- Monk, T.H.; Buysse, D.J.; Billy, B.D.; Kennedy, K.S.; Willrich, L.M. Sleep and Circadian Rhythms in Four Orbiting Astronauts. J Biol Rhythms 1998, 13, 188–201. [CrossRef]
- Monk, T.H.; Kennedy, K.S.; Rose, L.R.; Linenger, J.M. Decreased Human Circadian Pacemaker Influence After 100 Days in Space: A Case Study. Biopsychosocial Science and Medicine 2001, 63, 881.
- Dijk, D.-J.; Neri, D.F.; Wyatt, J.K.; Ronda, J.M.; Riel, E.; Ritz-De Cecco, A.; Hughes, R.J.; Elliott, A.R.; Prisk, G.K.; West, J.B.; et al. Sleep, Performance, Circadian Rhythms, and Light-Dark Cycles during Two Space Shuttle Flights. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 2001, 281, R1647–R1664. [CrossRef]
- Whitmire, A.M.; Leveton, L.B.; Barger, L.; Brainard, G.; Dinges, D.F.; Klerman, E.; Shea, C. Risk of Performance Errors Due to Sleep Loss, Circadian Desynchronization, Fatigue, and Work Overload.; Houston: NASA Behavioral Health and performance program, Johnson Space Center; 2009;
- Stuster, J. Behavioral Issues Associated With Long Duration Space Expeditions: Review and Analysis of Astronaut Journals (Experiment 01-E104 Final Report); ANACAPA SCIENCES, INC. Santa Barbara, California 93101, 2010;
- Guo, J.-H.; Qu, W.-M.; Chen, S.-G.; Chen, X.-P.; Lv, K.; Huang, Z.-L.; Wu, Y.-L. Keeping the Right Time in Space: Importance of Circadian Clock and Sleep for Physiology and Performance of Astronauts. Mil Med Res 2014, 1, 23. [CrossRef]
- Gonfalone, A. Sleep on Manned Space Flights: Zero Gravity Reduces Sleep Duration. Pathophysiology 2016, 23, 259–263. [CrossRef]
- Kanas, N.; Manzey, D. Space Psychology and Psychiatry; Segundo and Dordrecht: Microcosm Press and Springer, 2008;
- Barger, L.K.; Flynn-Evans, E.E.; Kubey, A.; Walsh, L.; Ronda, J.M.; Wang, W.; Wright, K.P.; Czeisler, C.A. Prevalence of Sleep Deficiency and Hypnotic Use Among Astronauts Before, During and After Spaceflight: An Observational Study. Lancet Neurol 2014, 13, 904–912. [CrossRef]
- Watson, N.F.; Badr, M.S.; Belenky, G.; Bliwise, D.L.; Buxton, O.M.; Buysse, D.; Dinges, D.F.; Gangwisch, J.; Grandner, M.A.; Kushida, C.; et al. Recommended Amount of Sleep for a Healthy Adult: A Joint Consensus Statement of the American Academy of Sleep Medicine and Sleep Research Society. J Clin Sleep Med 2015, 11, 591–592. [CrossRef]
- Gundel, A.; Nalishiti, V.; Reucher, E.; Vejvoda, M.; Zulley, J. Sleep and Circadian Rhythm during a Short Space Mission. Clin Investig 1993, 71, 718–724. [CrossRef]
- Santy, P.A.; Kapanka, H.; Davis, J.R.; Stewart, D.F. Analysis of Sleep on Shuttle Missions. Aviat Space Environ Med 1988, 59, 1094–1097.
- Zong, H.; Fei, Y.; Liu, N. Circadian Disruption and Sleep Disorders in Astronauts: A Review of Multi-Disciplinary Interventions for Long-Duration Space Missions. Int J Mol Sci 2025, 26, 5179. [CrossRef]
- Tafforin, C. The Mars-500 Crew in Daily Life Activities: An Ethological Study. Acta Astronautica 2013, 91, 69–76. [CrossRef]
- Tafforin, C.; Vinokhodova, A.; Chekalina, A.; Gushin, V. Correlation of Etho-Social and Psycho-Social Data from “Mars-500” Interplanetary Simulation. Acta Astronautica 2015, 111, 19–28. [CrossRef]
- Palinkas, L.A.; Suedfeld, P. Psychosocial Issues in Isolated and Confined Extreme Environments. Neuroscience & Biobehavioral Reviews 2021, 126, 413–429. [CrossRef]
- Sandal, G.M. Crew Tension during a Space Station Simulation. Environ Behav 2001, 33, 134–150. [CrossRef]
- Kraft, N.O.; Lyons, T.J.; Binder, H. Intercultural Crew Issues in Long-Duration Spaceflight. Aviat Space Environ Med 2003, 74, 575–578.
- Slobodian, R. Space Psychology: Psychosocial Challenges of Living in Space - Isolation and Culture. 2012.
- Landon, L.B.; Rokholt, C.; Slack, K.J.; Pecena, Y. Selecting Astronauts for Long-Duration Exploration Missions: Considerations for Team Performance and Functioning. REACH 2017, 5, 33–56. [CrossRef]
- Landon, L.B.; Slack, K.J.; Barrett, J.D. Teamwork and Collaboration in Long-Duration Space Missions: Going to Extremes. Am Psychol 2018, 73, 563–575. [CrossRef]
- Bell, S.T.; Brown, S.G.; Mitchell, T. What We Know About Team Dynamics for Long-Distance Space Missions: A Systematic Review of Analog Research. Front. Psychol. 2019, 10. [CrossRef]
- Lefèvre-Utile, A.; Braun, C.; Haftek, M.; Aubin, F. Five Functional Aspects of the Epidermal Barrier. International Journal of Molecular Sciences 2021, 22, 11676. [CrossRef]
- Nguyen, C.N.; Urquieta, E. Contemporary Review of Dermatologic Conditions in Space Flight and Future Implications for Long-Duration Exploration Missions. Life Sci Space Res (Amst) 2023, 36, 147–156. [CrossRef]
- Toback, A.C.; Kohn, S.R. Manifesto of Space Medicine: The next Dermatologic Frontier. J Am Acad Dermatol 1989, 20, 489–495. [CrossRef]
- König, K.; Weinigel, M.; Pietruszka, A.; Bückle, R.; Gerlach, N.; Heinrich, U. Multiphoton Tomography of Astronauts.; March 1 2015; Vol. 9329, p. 93290Q.
- Crucian, B.; Johnston, S.; Mehta, S.; Stowe, R.; Uchakin, P.; Quiriarte, H.; Pierson, D.; Laudenslager, M.L.; Sams, C. A Case of Persistent Skin Rash and Rhinitis with Immune System Dysregulation Onboard the International Space Station. J Allergy Clin Immunol Pract 2016, 4, 759-762.e8. [CrossRef]
- Braun, N.; Binder, S.; Grosch, H.; Theek, C.; Ülker, J.; Tronnier, H.; Heinrich, U. Current Data on Effects of Long-Term Missions on the International Space Station on Skin Physiological Parameters. Skin Pharmacol Physiol 2019, 32, 43–51. [CrossRef]
- Gontcharov, I.B.; Kovachevich, I.V.; Pool, S.L.; Navinkov, O.L.; Barratt, M.R.; Bogomolov, V.V.; House, N. In-Flight Medical Incidents in the NASA-Mir Program. Aviat Space Environ Med 2005, 76, 692–696.
- Law, J.; Gilmore, S.; Kelly, S. Postflight Rash and Skin Sensitivity Following a Year-Long Spaceflight Mission. Aerospace Medicine and Human Performance 2020, 91, 604–607. [CrossRef]
- Mehta, S.K.; Szpara, M.L.; Rooney, B.V.; Diak, D.M.; Shipley, M.M.; Renner, D.W.; Krieger, S.S.; Nelman-Gonzalez, M.A.; Zwart, S.R.; Smith, S.M.; et al. Dermatitis during Spaceflight Associated with HSV-1 Reactivation. Viruses 2022, 14, 789. [CrossRef]
- Rutherford, A.; Glass, D.A.; Savory, S. Dermatology in Orbit: Anticipating Skin Care Requirements in the Space Age. J Am Acad Dermatol 2022, 87, 1223–1224. [CrossRef]
- Wotring, V.E. Medication Use by U.S. Crewmembers on the International Space Station. FASEB J 2015, 29, 4417–4423. [CrossRef]
- Barrat, M.R.; Baker, E.S.; Pool, S.L. Principles of Clinical Medicine for Space Flight | SpringerLink Available online: https://link.springer.com/book/10.1007/978-1-4939-9889-0 (accessed on 18 July 2025).
- Tronnier, H.; Wiebusch, M.; Heinrich, U. Change in Skin Physiological Parameters in Space--Report on and Results of the First Study on Man. Skin Pharmacol Physiol 2008, 21, 283–292. [CrossRef]
- Venugopalan, S.K.; Sri R, S.; N, H.; T, P.; G, U.M.; S, A.S.; Sharon, E.; Singh, A. Challenges and Countermeasures for Ensuring Health and Drug Stability During Long-Term Space Missions. Curr Drug Discov Technol 2025. [CrossRef]
- Blue, R.S.; Bayuse, T.M.; Daniels, V.R.; Wotring, V.E.; Suresh, R.; Mulcahy, R.A.; Antonsen, E.L. Supplying a Pharmacy for NASA Exploration Spaceflight: Challenges and Current Understanding. NPJ Microgravity 2019, 5, 14. [CrossRef]
- Putcha, L.; Berens, K.L.; Marshburn, T.H.; Ortega, H.J.; Billica, R.D. Pharmaceutical Use by U.S. Astronauts on Space Shuttle Missions. Aviat Space Environ Med 1999, 70, 705–708.
- Flynn-Evans, E.E.; Barger, L.K.; Kubey, A.A.; Sullivan, J.P.; Czeisler, C.A. Circadian Misalignment Affects Sleep and Medication Use before and during Spaceflight. npj Microgravity 2016, 2, 15019. [CrossRef]
- Mendt, S.; Gunga, H.-C.; Felsenberg, D.; Belavy, D.L.; Steinach, M.; Stahn, A.C. Regular Exercise Counteracts Circadian Shifts in Core Body Temperature during Long-Duration Bed Rest. npj Microgravity 2021, 7, 1. [CrossRef]
- Du, B.; Daniels, V.R.; Vaksman, Z.; Boyd, J.L.; Crady, C.; Putcha, L. Evaluation of Physical and Chemical Changes in Pharmaceuticals Flown on Space Missions. AAPS J 2011, 13, 299–308. [CrossRef]
- Mehta, P.; Bhayani, D. Impact of Space Environment on Stability of Medicines: Challenges and Prospects. Journal of Pharmaceutical and Biomedical Analysis 2017, 136, 111–119. [CrossRef]
- Simon, Á.; Smarandache, A.; Iancu, V.; Pascu, M.L. Stability of Antimicrobial Drug Molecules in Different Gravitational and Radiation Conditions in View of Applications during Outer Space Missions. Molecules 2021, 26, 2221. [CrossRef]
- Reichard, J.F.; Phelps, S.E.; Lehnhardt, K.R.; Young, M.; Easter, B.D. The Effect of Long-Term Spaceflight on Drug Potency and the Risk of Medication Failure. NPJ Microgravity 2023, 9, 35. [CrossRef]
- Dello Russo, C.; Bandiera, T.; Monici, M.; Surdo, L.; Yip, V.L.M.; Wotring, V.; Morbidelli, L. Physiological Adaptations Affecting Drug Pharmacokinetics in Space: What Do We Really Know? A Critical Review of the Literature. Br J Pharmacol 2022, 179, 2538–2557. [CrossRef]
- Kast, J.; Yu, Y.; Seubert, C.N.; Wotring, V.E.; Derendorf, H. Drugs in Space: Pharmacokinetics and Pharmacodynamics in Astronauts. Eur J Pharm Sci 2017, 109S, S2–S8. [CrossRef]
- Chou, W.-C.; Lin, Z. Machine Learning and Artificial Intelligence in Physiologically Based Pharmacokinetic Modeling. Toxicol Sci 2022, 191, 1–14. [CrossRef]
- Katakam, P.; Bhavaraju, M.L.R.; Narayana, T.V.; Bhandari, K.; Sriram, N.; Sisinty, V.S.; Adiki, S.K. Navigating the Challenges of 3D Printing Personalized Medicine in Space Explorations: A Comprehensive Review. CRT 2024, 41. [CrossRef]
- Zhang, T.; Liu, D.; Zhang, Y.; Chen, L.; Zhang, W.; Sun, T. Biomedical Engineering Utilizing Living Photosynthetic Cyanobacteria and Microalgae: Current Status and Future Prospects. Materials Today Bio 2024, 27, 101154. [CrossRef]
- Shi, Y.; Jiao, Q.; Hu, X.; Jia, T. Recent Advances in Plant-Based Vaccines: From Molecular Farming Innovations to Global Health Applications. Biotechnology Journal 2025, 20, e70120. [CrossRef]
- Schillberg, S.; Finnern, R. Plant Molecular Farming for the Production of Valuable Proteins – Critical Evaluation of Achievements and Future Challenges. Journal of Plant Physiology 2021, 258–259, 153359. [CrossRef]
- Agarwalla, A.; Ahmed, W.; Al-Marzouqi, A.H.; Zaneldin, E.; Rizvi, T.A.; Khan, M. Advancements in Synthetic Polymers for 3D Bioprinting Materials, Applications, and Future Prospects. International Journal of Polymeric Materials and Polymeric Biomaterials 0, 1–39. [CrossRef]
- Ciofani, G.; Bandiera, T.; Corsini, A.; Crescenzi, M.; De Vittorio, M.; Mari, S.; Martinelli, E.; Monici, M.; Piccirillo, S.; Narici, M.; et al. Pharmaceutical and Biomedical Challenges for Crew Autonomy in Health Preservation during Future Exploration Missions. Commun Med (Lond) 2025, 5, 418. [CrossRef]
- Barchetti, K.; Derobertmasure, A.; Boutouyrie, P.; Sestili, P. Redefining Space Pharmacology: Bridging Knowledge Gaps in Drug Efficacy and Safety for Deep Space Missions. Frontiers in Space Technologies 2024, 5, 1456614. [CrossRef]
- Tran, Q.D.; Tran, V.; Toh, L.S.; Williams, P.M.; Tran, N.N.; Hessel, V. Space Medicines for Space Health. ACS Med Chem Lett 2022, 13, 1231–1247. [CrossRef]
- Dakkumadugula, A.; Pankaj, L.; Alqahtani, A.S.; Ullah, R.; Ercisli, S.; Murugan, R. Space Nutrition and the Biochemical Changes Caused in Astronauts Health Due to Space Flight: A Review. Food Chem X 2023, 20, 100875. [CrossRef]
- Sonali, L.; Drisya Raj, M.P.; Pavithra, R.; Kanimozhi, N.V.; Suneetha, C.; Roopa Shri, B.; Sukumar, M. Functional Foods for Astronauts: Enhancing Health and Performance in Microgravity and Extreme Environments. Space Habitation 2025, 1, 100005. [CrossRef]
- Clemente-Villalba, J.; Cerdá-Bernad, D. Functional Food as a Nutritional Countermeasure to Health Risks from Microgravity and Space Radiation in Long-Term Spaceflights: A Review. Applied Sciences 2025, 15, 9220. [CrossRef]
- Mokhtari, M.; Reinsch, S.S.; Barcenilla, B.B.; Ziyaei, K.; Barker, R.J. Space-Driven ROS in Cells: A Hidden Danger to Astronaut Health and Food Safety. npj Microgravity 2025, 11, 52. [CrossRef]
- Wani, A.; Prabhakar, B.; Shende, P. Strategic Aspects of Space Medicine: A Journey from Conventional to Futuristic Requisites. Space: Science & Technology 2024, 4, 0123. [CrossRef]
- Kapoor, P.; Yadav, R.B.; Agrawal, N.; Gaur, S.; Arora, R. Long Duration Space Missions: Challenges and Prospects in Sustaining Humans in Space. Life Sciences in Space Research 2025, 47, 14–31. [CrossRef]
- Shanmugaraj, B.; I. Bulaon, C.J.; Phoolcharoen, W. Plant Molecular Farming: A Viable Platform for Recombinant Biopharmaceutical Production. Plants 2020, 9, 842. [CrossRef]
- De Martinis, D.; Hitzeroth, I.I.; Matsuda, R.; Soto Pérez, N.; Benvenuto, E. Editorial: Engineering the Plant Biofactory for the Production of Biologics and Small-Molecule Medicines-Volume 2. Front Plant Sci 2022, 13, 942746. [CrossRef]
- Buyel, J.F. Plant Molecular Farming – Integration and Exploitation of Side Streams to Achieve Sustainable Biomanufacturing. Front Plant Sci 2019, 9, 1893. [CrossRef]
- Onofri, S.; Moeller, R.; Billi, D.; Balsamo, M.; Becker, A.; Benvenuto, E.; Cassaro, A.; Catanzaro, I.; Cockell, C.S.; Desiderio, A.; et al. Synthetic Biology for Space Exploration. NPJ Microgravity 2025, 11, 41. [CrossRef]
- Lee, M.-S.; Lee, J.; Park, B.-J.; Miyazaki, Y. Interaction with Indoor Plants May Reduce Psychological and Physiological Stress by Suppressing Autonomic Nervous System Activity in Young Adults: A Randomized Crossover Study. J Physiol Anthropol 2015, 34, 21. [CrossRef]
- Han, K.-T.; Ruan, L.-W.; Liao, L.-S. Effects of Indoor Plants on Human Functions: A Systematic Review with Meta-Analyses. Int J Environ Res Public Health 2022, 19, 7454. [CrossRef]
- Hosseini, A.; Mobasheri, L.; Rakhshandeh, H.; Rahimi, V.B.; Najafi, Z.; Askari, V.R. Edible Herbal Medicines as an Alternative to Common Medication for Sleep Disorders: A Review Article. Curr Neuropharmacol 2024, 22, 1205–1232. [CrossRef]
- Ghasemzadeh Rahbardar, M.; Hosseinzadeh, H. Therapeutic Potential of Hypnotic Herbal Medicines: A Comprehensive Review. Phytother Res 2024, 38, 3037–3059. [CrossRef]
- Salame, A.; Mathew, S.; Bhanu, C.; Bazo-Alvarez, J.C.; Bhamra, S.K.; Heinrich, M.; Walters, K.; Frost, R. Over-the-Counter Products for Insomnia in Adults: A Scoping Review of Randomised Controlled Trials. Sleep Medicine 2025, 129, 219–237. [CrossRef]
- Jahromi, B.; Pirvulescu, I.; Candido, K.D.; Knezevic, N.N. Herbal Medicine for Pain Management: Efficacy and Drug Interactions. Pharmaceutics 2021, 13, 251. [CrossRef]
- Hasan, Md.K.; Zanzabil, K.Z.; Ara, I.; Rahman, T.; Kieu, A.; Östlundh, L.; Junaidi, S.; Khan, M.A. Herbal Therapies for Pain Management: A Scoping Review of the Current Evidence. Phytochem Rev 2024, 23, 1065–1116. [CrossRef]
- Patočka, J.; Jakl, J. Biomedically Relevant Chemical Constituents of Valeriana Officinalis. Journal of Applied Biomedicine 2010, 8, 11–18. [CrossRef]
- Nandhini, S.; Narayanan, K.; Ilango, K. VALERIANA OFFICINALIS: A REVIEW OF ITS TRADITIONAL USES, PHYTOCHEMISTRY AND PHARMACOLOGY. Asian Journal of Pharmaceutical and Clinical Research 2018, 11, 36. [CrossRef]
- Dorn, M. [Efficacy and tolerability of Baldrian versus oxazepam in non-organic and non-psychiatric insomniacs: a randomised, double-blind, clinical, comparative study]. Forsch Komplementarmed Klass Naturheilkd 2000, 7, 79–84. [CrossRef]
- Ziegler, G.; Ploch, M.; Miettinen-Baumann, A.; Collet, W. Efficacy and Tolerability of Valerian Extract LI 156 Compared with Oxazepam in the Treatment of Non-Organic Insomnia--a Randomized, Double-Blind, Comparative Clinical Study. Eur J Med Res 2002, 7, 480–486.
- Koetter, U.; Schrader, E.; Käufeler, R.; Brattström, A. A Randomized, Double Blind, Placebo-Controlled, Prospective Clinical Study to Demonstrate Clinical Efficacy of a Fixed Valerian Hops Extract Combination (Ze 91019) in Patients Suffering from Non-Organic Sleep Disorder. Phytotherapy Research 2007, 21, 847–851. [CrossRef]
- Maroo, N.; Hazra, A.; Das, T. Efficacy and Safety of a Polyherbal Sedative-Hypnotic Formulation NSF-3 in Primary Insomnia in Comparison to Zolpidem: A Randomized Controlled Trial. Indian J Pharmacol 2013, 45, 34–39. [CrossRef]
- Zare Elmi, H.K.; Gholami, M.; Saki, M.; Ebrahimzadeh, F. Efficacy of Valerian Extract on Sleep Quality after Coronary Artery Bypass Graft Surgery: A Triple-Blind Randomized Controlled Trial. Chin J Integr Med 2021, 27, 7–15. [CrossRef]
- Hajizadeh, I.; Jamshidi, M.; Kazemi, M.; Kargar, H.; Sadeghi, T. Comparison the Effect of Valerian and Gabapentin on RLS and Sleep Quality in Hemodialysis Patients: A Randomized Clinical Trial. Therapeutic Apheresis and Dialysis 2023, 27, 621–628. [CrossRef]
- Chandra Shekhar, H.; Joshua, L.; Thomas, J.V. Standardized Extract of Valeriana Officinalis Improves Overall Sleep Quality in Human Subjects with Sleep Complaints: A Randomized, Double-Blind, Placebo-Controlled, Clinical Study. Adv Ther 2024, 41, 246–261. [CrossRef]
- Schicktanz, N.; Gerhards, C.; Schlitt, T.; Aerni, A.; Müggler, E.; de Quervain, D.; Papassotiropoulos, A.; Boonen, G.; Drewe, J.; Butterweck, V. Effects of a Valerian-Hops Extract Combination (Ze 91019) on Sleep Duration and Daytime Cognitive and Psychological Parameters in Occasional Insomnia: A Randomized Controlled Feasibility Trial. Brain Behav 2025, 15, e70600. [CrossRef]
- Alavizadeh, S.H.; Hosseinzadeh, H. Bioactivity Assessment and Toxicity of Crocin: A Comprehensive Review. Food and Chemical Toxicology 2014, 64, 65–80. [CrossRef]
- Bej, E.; Volpe, A.R.; Cesare, P.; Cimini, A.; d’Angelo, M.; Castelli, V. Therapeutic Potential of Saffron in Brain Disorders: From Bench to Bedside. Phytother Res 2024, 38, 2482–2495. [CrossRef]
- Ghasemzadeh Rahbardar, M.; Hosseinzadeh, H. A Review of How the Saffron (Crocus Sativus) Petal and Its Main Constituents Interact with the Nrf2 and NF-κB Signaling Pathways. Naunyn-Schmiedeberg’s Arch Pharmacol 2023, 396, 1879–1909. [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Drummond, P.D. An Investigation into an Evening Intake of a Saffron Extract (Affron®) on Sleep Quality, Cortisol, and Melatonin Concentrations in Adults with Poor Sleep: A Randomised, Double-Blind, Placebo-Controlled, Multi-Dose Study. Sleep Medicine 2021, 86, 7–18. [CrossRef]
- Pachikian, B.D.; Copine, S.; Suchareau, M.; Deldicque, L. Effects of Saffron Extract on Sleep Quality: A Randomized Double-Blind Controlled Clinical Trial. Nutrients 2021, 13, 1473. [CrossRef]
- Tajaddini, A.; Roshanravan, N.; Mobasseri, M.; Aeinehchi, A.; Sefid-Mooye Azar, P.; Hadi, A.; Ostadrahimi, A. Saffron Improves Life and Sleep Quality, Glycaemic Status, Lipid Profile and Liver Function in Diabetic Patients: A Double-Blind, Placebo-Controlled, Randomised Clinical Trial. International Journal of Clinical Practice 2021, 75, e14334. [CrossRef]
- Schuster, J.; Mundhenke, C.; Nordsieck, H.; Pouchieu, C.; Pourtau, L.; Hahn, A. Effect of a Saffron Extract on Sleep Quality in Adults with Moderate Insomnia: A Decentralized, Randomized, Double-Blind, Placebo-Controlled Trial. Sleep Med X 2025, 10, 100147. [CrossRef]
- Dobros, N.; Zawada, K.D.; Paradowska, K. Phytochemical Profiling, Antioxidant and Anti-Inflammatory Activity of Plants Belonging to the Lavandula Genus. Molecules 2022, 28, 256. [CrossRef]
- Soares, G.A.B. e; Bhattacharya, T.; Chakrabarti, T.; Tagde, P.; Cavalu, S. Exploring Pharmacological Mechanisms of Essential Oils on the Central Nervous System. Plants (Basel) 2021, 11, 21. [CrossRef]
- de Melo Alves Silva, L.C.; de Oliveira Mendes, F. de C.; de Castro Teixeira, F.; de Lima Fernandes, T.E.; Barros Ribeiro, K.R.; da Silva Leal, K.C.; Dantas, D.V.; Neves Dantas, R.A. Use of Lavandula Angustifolia Essential Oil as a Complementary Therapy in Adult Health Care: A Scoping Review. Heliyon 2023, 9, e15446. [CrossRef]
- Kasper, S.; Gastpar, M.; Müller, W.E.; Volz, H.-P.; Möller, H.-J.; Dienel, A.; Schläfke, S. Silexan, an Orally Administered Lavandula Oil Preparation, Is Effective in the Treatment of “subsyndromal” Anxiety Disorder: A Randomized, Double-Blind, Placebo Controlled Trial. Int Clin Psychopharmacol 2010, 25, 277–287. [CrossRef]
- Woelk, H.; Schläfke, S. A Multi-Center, Double-Blind, Randomised Study of the Lavender Oil Preparation Silexan in Comparison to Lorazepam for Generalized Anxiety Disorder. Phytomedicine 2010, 17, 94–99. [CrossRef]
- Möller, H.-J.; Volz, H.-P.; Dienel, A.; Schläfke, S.; Kasper, S. Efficacy of Silexan in Subthreshold Anxiety: Meta-Analysis of Randomised, Placebo-Controlled Trials. Eur Arch Psychiatry Clin Neurosci 2019, 269, 183–193. [CrossRef]
- Genç, F.; Karadağ, S.; Kılıç Akça, N.; Tan, M.; Cerit, D. The Effect of Aromatherapy on Sleep Quality and Fatigue Level of the Elderly: A Randomized Controlled Study. Holist Nurs Pract 2020, 34, 155–162. [CrossRef]
- Seifritz, E.; Kasper, S.; Möller, H.-J.; Volz, H.-P.; Müller, W.E.; Eckert, A.; Hatzinger, M. Effect of Anxiolytic Drug Silexan on Sleep – a Narrative Review. The World Journal of Biological Psychiatry 2022, 23, 493–500. [CrossRef]
- Kavuran, E.; Yurttaş, A. The Effect of Aromatherapy with Lavender Essential Oil on the Sleep and Fatigue Level of Patients with Multiple Sclerosis in Turkey: A Randomized Controlled Trial. Nigerian Journal of Clinical Practice 2024, 27, 635. [CrossRef]
- Yin, X.-J.; Lin, G.-P.; Wu, X.-Y.; Huang, R.; Xu, C.-J.; Yao, M.-Y. Effects of Lavender Essential Oil Inhalation Aromatherapy on Depression and Sleep Quality in Stroke Patients: A Single-Blind Randomized Controlled Trial. Complementary Therapies in Clinical Practice 2024, 55, 101828. [CrossRef]
- Celik, S.; Nazik, E. The Effect of Aromatherapy Applied to Pregnant Women on Sleep Quality and Fatigue Level: A Randomized Clinical Trial. EXPLORE 2025, 21, 103157. [CrossRef]
- Yildirim, D.; Harman Ozdogan, M.; Erdal, S.; Selcuk, S.; Guneri, A.; Simsek, E.B.; Can, T.B.; Gunduz, H.; Kuni, A. The Efficacy of Lavender Oil on Fatigue and Sleep Quality in Patients with Hematological Malignancy Receiving Chemotherapy: A Single-Blind Randomized Controlled Trial. Support Care Cancer 2025, 33, 79. [CrossRef]
- Petrisor, G.; Motelica, L.; Craciun, L.N.; Oprea, O.C.; Ficai, D.; Ficai, A. Melissa Officinalis: Composition, Pharmacological Effects and Derived Release Systems—A Review. Int J Mol Sci 2022, 23, 3591. [CrossRef]
- Draginic, N.; Andjic, M.; Jeremic, J.; Zivkovic, V.; Kocovic, A.; Tomovic, M.; Bozin, B.; Kladar, N.; Bolevich, S.; Jakovljevic, V.; et al. Anti-Inflammatory and Antioxidant Effects of Melissa Officinalis Extracts: A Comparative Study. Iran J Pharm Res 2022, 21, e126561. [CrossRef]
- Awlqadr, F.H.; Altemimi, A.B.; Qadir, S.A.; Mohammed, O.A.; Saeed, M.N.; Hesarinejad, M.A.; Lakhssassi, N. Bioactive Compounds, Medicinal Benefits, and Contemporary Extraction Methods for Lemon Balm (Melissa Officinalis). Food Science & Nutrition 2025, 13, e70864. [CrossRef]
- Cases, J.; Ibarra, A.; Feuillère, N.; Roller, M.; Sukkar, S.G. Pilot Trial of Melissa Officinalis L. Leaf Extract in the Treatment of Volunteers Suffering from Mild-to-Moderate Anxiety Disorders and Sleep Disturbances. Med J Nutrition Metab 2011, 4, 211–218. [CrossRef]
- Aliakbari, F. The Effectiveness of Melissa Officinalis on Sleep Problem in Patients with Chronic Heart Failure. Journal of Pharmaceutical Negative Results 2018, 9, 55–58.
- Haybar, H.; Javid, A.Z.; Haghighizadeh, M.H.; Valizadeh, E.; Mohaghegh, S.M.; Mohammadzadeh, A. The Effects of Melissa Officinalis Supplementation on Depression, Anxiety, Stress, and Sleep Disorder in Patients with Chronic Stable Angina. Clinical Nutrition ESPEN 2018, 26, 47–52. [CrossRef]
- Heydari, N.; Dehghani, M.; Emamghoreishi, M.; Akbarzadeh, M. Effect of Melissa Officinalis Capsule on the Mental Health of Female Adolescents with Premenstrual Syndrome: A Clinical Trial Study. International Journal of Adolescent Medicine and Health 2019, 31. [CrossRef]
- Soltanpour, A.; Alijaniha, F.; Naseri, M.; Kazemnejad, A.; Heidari, M.R. Effects of Melissa Officinalis on Anxiety and Sleep Quality in Patients Undergoing Coronary Artery Bypass Surgery: A Double-Blind Randomized Placebo Controlled Trial. European Journal of Integrative Medicine 2019, 28, 27–32. [CrossRef]
- Di Pierro, F.; Sisti, D.; Rocchi, M.; Belli, A.; Bertuccioli, A.; Cazzaniga, M.; Palazzi, C.M.; Tanda, M.L.; Zerbinati, N. Effects of Melissa Officinalis Phytosome on Sleep Quality: Results of a Prospective, Double-Blind, Placebo-Controlled, and Cross-Over Study. Nutrients 2024, 16, 4199. [CrossRef]
- Mandal, S.K.; Rath, S.K.; Logesh, R.; Mishra, S.K.; Devkota, H.P.; Das, N. Capsicum Annuum L. and Its Bioactive Constituents: A Critical Review of a Traditional Culinary Spice in Terms of Its Modern Pharmacological Potentials with Toxicological Issues. Phytotherapy Research 2023, 37, 965–1002. [CrossRef]
- Keitel, W.; Frerick, H.; Kuhn, U.; Schmidt, U.; Kuhlmann, M.; Bredehorst, A. Capsicum Pain Plaster in Chronic Non-Specific Low Back Pain. Arzneimittelforschung 2001, 51, 896–903. [CrossRef]
- Frerick, H.; Keitel, W.; Kuhn, U.; Schmidt, S.; Bredehorst, A.; Kuhlmann, M. Topical Treatment of Chronic Low Back Pain with a Capsicum Plaster. Pain 2003, 106, 59–64. [CrossRef]
- Kim, K.S.; Kim, D.W.; Yu, Y.K. The Effect of Capsicum Plaster in Pain after Inguinal Hernia Repair in Children. Pediatric Anesthesia 2006, 16, 1036–1041. [CrossRef]
- Kim, K.S.; Nam, Y.M. The Analgesic Effects of Capsicum Plaster at the Zusanli Point after Abdominal Hysterectomy. Anesth Analg 2006, 103, 709–713. [CrossRef]
- Kim, K.S.; Kim, K.N.; Hwang, K.G.; Park, C.J. Capsicum Plaster at the Hegu Point Reduces Postoperative Analgesic Requirement after Orthognathic Surgery. Anesth Analg 2009, 108, 992–996. [CrossRef]
- Chrubasik, S.; Weiser, T.; Beime, B. Effectiveness and safety of topical capsaicin cream in the treatment of chronic soft tissue pain. Phytotherapy Research 2010, 24, 1877–1885. [CrossRef]
- Zhang, H.A.; Kitts, D.D. Turmeric and Its Bioactive Constituents Trigger Cell Signaling Mechanisms That Protect against Diabetes and Cardiovascular Diseases. Mol Cell Biochem 2021, 476, 3785–3814. [CrossRef]
- Zhang, P.; Liu, H.; Yu, Y.; Peng, S.; Zhu, S. Role of Curcuma Longae Rhizoma in Medical Applications: Research Challenges and Opportunities. Front Pharmacol 2024, 15, 1430284. [CrossRef]
- Tian, W.-W.; Liu, L.; Chen, P.; Yu, D.-M.; Li, Q.-M.; Hua, H.; Zhao, J.-N. Curcuma Longa (Turmeric): From Traditional Applications to Modern Plant Medicine Research Hotspots. Chin Med 2025, 20, 76. [CrossRef]
- Appelboom, T.; Maes, N.; Albert, A. A New Curcuma Extract (Flexofytol®) in Osteoarthritis: Results from a Belgian Real-Life Experience. Open Rheumatol J 2014, 8, 77–81. [CrossRef]
- Wang, Z.; Jones, G.; Winzenberg, T.; Cai, G.; Laslett, L.L.; Aitken, D.; Hopper, I.; Singh, A.; Jones, R.; Fripp, J.; et al. Effectiveness of Curcuma Longa Extract for the Treatment of Symptoms and Effusion–Synovitis of Knee Osteoarthritis. Ann Intern Med 2020, 173, 861–869. [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Jackson-Michel, S.; Fairchild, T. An Investigation into the Effects of a Curcumin Extract (Curcugen®) on Osteoarthritis Pain of the Knee: A Randomised, Double-Blind, Placebo-Controlled Study. Nutrients 2022, 14, 41. [CrossRef]
- Singhal, S.; Hasan, N.; Nirmal, K.; Chawla, R.; Chawla, S.; Kalra, B.S.; Dhal, A. Bioavailable Turmeric Extract for Knee Osteoarthritis: A Randomized, Non-Inferiority Trial versus Paracetamol. Trials 2021, 22, 105. [CrossRef]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber Officinale Roscoe). Foods 2019, 8, 185. [CrossRef]
- Verma, P.K.; Singh, B.; Sharma, P.; Tukra, S.; Aït-Kaddour, A.; Bhat, Z.F. Mechanistic Advances and Therapeutic Applications of Zingiber Officinale Roscoe. Food Chemistry Advances 2025, 8, 101060. [CrossRef]
- Altman, R.D.; Marcussen, K.C. Effects of a Ginger Extract on Knee Pain in Patients with Osteoarthritis. Arthritis & Rheumatism 2001, 44, 2531–2538. [CrossRef]
- Martins, L.B.; Rodrigues, A.M. dos S.; Rodrigues, D.F.; dos Santos, L.C.; Teixeira, A.L.; Ferreira, A.V.M. Double-Blind Placebo-Controlled Randomized Clinical Trial of Ginger (Zingiber Officinale Rosc.) Addition in Migraine Acute Treatment. Cephalalgia 2019, 39, 68–76. [CrossRef]
- Wilson, P.B. A Randomized Double-Blind Trial of Ginger Root for Reducing Muscle Soreness and Improving Physical Performance Recovery Among Experienced Recreational Distance Runners. J Diet Suppl 2020, 17, 121–132. [CrossRef]
- Broeckel, J.; Estes, L.; Leonard, M.; Dickerson, B.L.; Gonzalez, D.E.; Purpura, M.; Jäger, R.; Sowinski, R.J.; Rasmussen, C.J.; Kreider, R.B. Effects of Ginger Supplementation on Markers of Inflammation and Functional Capacity in Individuals with Mild to Moderate Joint Pain. Nutrients 2025, 17, 2365. [CrossRef]
- Tawfeek, N.; Mahmoud, M.F.; Hamdan, D.I.; Sobeh, M.; Farrag, N.; Wink, M.; El-Shazly, A.M. Phytochemistry, Pharmacology and Medicinal Uses of Plants of the Genus Salix: An Updated Review. Front Pharmacol 2021, 12, 593856. [CrossRef]
- Chrubasik, S.; Eisenberg, E.; Balan, E.; Weinberger, T.; Luzzati, R.; Conradt, C. Treatment of Low Back Pain Exacerbations with Willow Bark Extract: A Randomized Double-Blind Study. The American Journal of Medicine 2000, 109, 9–14. [CrossRef]
- Chrubasik, S.; Künzel, O.; Black, A.; Conradt, C.; Kerschbaumer, F. Potential Economic Impact of Using a Proprietary Willow Bark Extract in Outpatient Treatment of Low Back Pain:An Open Non-Randomized Study. Phytomedicine 2001, 8, 241–251. [CrossRef]
- Chrubasik, S.; Künzel, O.; Model, A.; Conradt, C.; Black, A. Treatment of Low Back Pain with a Herbal or Synthetic Anti-rheumatic: A Randomized Controlled Study. Willow Bark Extract for Low Back Pain. Rheumatology (Oxford) 2001, 40, 1388–1393. [CrossRef]
- Uehleke, B.; Müller, J.; Stange, R.; Kelber, O.; Melzer, J. Willow Bark Extract STW 33-I in the Long-Term Treatment of Outpatients with Rheumatic Pain Mainly Osteoarthritis or Back Pain. Phytomedicine 2013, 20, 980–984. [CrossRef]
- Gxaba, N.; Manganyi, M.C. The Fight against Infection and Pain: Devil’s Claw (Harpagophytum Procumbens) a Rich Source of Anti-Inflammatory Activity: 2011–2022. Molecules 2022, 27, 3637. [CrossRef]
- Mncwangi, N.; Chen, W.; Vermaak, I.; Viljoen, A.M.; Gericke, N. Devil’s Claw—A Review of the Ethnobotany, Phytochemistry and Biological Activity of Harpagophytum Procumbens. Journal of Ethnopharmacology 2012, 143, 755–771. [CrossRef]
- Chantre, P.; Cappelaere, A.; Leblan, D.; Guedon, D.; Vandermander, J.; Fournie, B. Efficacy and Tolerance of Harpagophytum Procumbens versus Diacerhein in Treatment of Osteoarthritis. Phytomedicine 2000, 7, 177–183. [CrossRef]
- Leblan, D.; Chantre, P.; Fournié, B. Harpagophytum Procumbens in the Treatment of Knee and Hip Osteoarthritis. Four-Month Results of a Prospective, Multicenter, Double-Blind Trial versus Diacerhein. Joint Bone Spine 2000, 67, 462–467.
- Laudahn, D.; Walper, A. Efficacy and Tolerance of Harpagophytum Extract LI 174 in Patients with Chronic Non-Radicular Back Pain. Phytotherapy Research 2001, 15, 621–624. [CrossRef]
- Chrubasik, S.; Model, A.; Black, A.; Pollak, S. A Randomized Double-blind Pilot Study Comparing Doloteffin® and Vioxx® in the Treatment of Low Back Pain. Rheumatology (Oxford) 2003, 42, 141–148. [CrossRef]
- Wegener, T.; Lüpke, N.-P. Treatment of Patients with Arthrosis of Hip or Knee with an Aqueous Extract of Devil’s Claw (Harpagophytum Procumbens DC.). Phytotherapy Research 2003, 17, 1165–1172. [CrossRef]
- Warnock, M.; McBean, D.; Suter, A.; Tan, J.; Whittaker, P. Effectiveness and Safety of Devil’s Claw Tablets in Patients with General Rheumatic Disorders. Phytotherapy Research 2007, 21, 1228–1233. [CrossRef]
- Iram, F.; Khan, S.A.; Husain, A. Phytochemistry and Potential Therapeutic Actions of Boswellic Acids: A Mini-Review. Asian Pacific Journal of Tropical Biomedicine 2017, 7, 513–523. [CrossRef]
- Kimmatkar, N.; Thawani, V.; Hingorani, L.; Khiyani, R. Efficacy and Tolerability of Boswellia Serrata Extract in Treatment of Osteoarthritis of Knee – A Randomized Double Blind Placebo Controlled Trial. Phytomedicine 2003, 10, 3–7. [CrossRef]
- Sengupta, K.; Krishnaraju, A.V.; Vishal, A.A.; Mishra, A.; Trimurtulu, G.; Sarma, K.V.; Raychaudhuri, S.K.; Raychaudhuri, S.P. Comparative Efficacy and Tolerability of 5-Loxin® and Aflapin® Against Osteoarthritis of the Knee: A Double Blind, Randomized, Placebo Controlled Clinical Study. Int J Med Sci 2010, 7, 366–377. [CrossRef]
- Kulkarni, P.D.; Damle, N.D.; Singh, S.; Yadav, K.S.; Ghante, M.R.; Bhaskar, V.H.; Hingorani, L.; Gota, V.S. Double-Blind Trial of Solid Lipid Boswellia Serrata Particles (SLBSP) vs. Standardized Boswellia Serrata Gum Extract (BSE) for Osteoarthritis of Knee. Drug Metab Pers Ther 2020, 35, /j/dmdi.2020.35.issue-2/dmpt-2020-0104/dmpt-2020-0104.xml. [CrossRef]
- Karlapudi, V.; Sunkara, K.B.; Konda, P.R.; Sarma, K.V.; Rokkam, M.P. Efficacy and Safety of Aflapin®, a Novel Boswellia Serrata Extract, in the Treatment of Osteoarthritis of the Knee: A Short-Term 30-Day Randomized, Double-Blind, Placebo-Controlled Clinical Study. J Am Nutr Assoc 2023, 42, 159–168. [CrossRef]
- Mohsenzadeh, A.; Karimifar, M.; Soltani, R.; Hajhashemi, V. Evaluation of the Effectiveness of Topical Oily Solution Containing Frankincense Extract in the Treatment of Knee Osteoarthritis: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. BMC Res Notes 2023, 16, 28. [CrossRef]
- Mowrey, DanielB.; Clayson, DennisE. MOTION SICKNESS, GINGER, AND PSYCHOPHYSICS. The Lancet 1982, 319, 655–657. [CrossRef]
- Grøntved, A.; Brask, T.; Kambskard, J.; Hentzer, E. Ginger Root against Seasickness. A Controlled Trial on the Open Sea. Acta Otolaryngol 1988, 105, 45–49. [CrossRef]
- Lien, H.-C.; Sun, W.M.; Chen, Y.-H.; Kim, H.; Hasler, W.; Owyang, C. Effects of Ginger on Motion Sickness and Gastric Slow-Wave Dysrhythmias Induced by Circular Vection. American Journal of Physiology-Gastrointestinal and Liver Physiology 2003, 284, G481–G489. [CrossRef]
- Nunes, C.P.; Rodrigues, C. de C.; Cardoso, C.A.F.; Cytrynbaum, N.; Kaufman, R.; Rzetelna, H.; Goldwasser, G.; Santos, A.; Oliveira, L.; Geller, M. Clinical Evaluation of the Use of Ginger Extract in the Preventive Management of Motion Sickness. Current Therapeutic Research 2020, 92, 100591. [CrossRef]
- Nunes, C.P.; Rodrigues, C.; Suchmacher, M.; Esteves, C.R.; Gonçalves, K.; Rzetelna, H.; Rodrigues, R.V.; de Vasconcelos, L.R.; Mezitis, S.G.E.; Rabelo, H.; et al. A Combination of Gamma-Aminobutyric Acid, Glutamic Acid, Calcium, Thiamine, Pyridoxine, and Cyanocobalamin vs Ginger Extract in the Management of Chronic Motion Sickness: A Clinical Evaluation. Curr Ther Res Clin Exp 2023, 99, 100719. [CrossRef]
- Matei, C.E.; Visan, A.I.; Cristescu, R. Aloe Vera Polysaccharides as Therapeutic Agents: Benefits Versus Side Effects in Biomedical Applications. Polysaccharides 2025, 6, 36. [CrossRef]
- Brandão, M.L.; Reis, P.R.M.; Araújo, L.A. de; Araújo, A.C.V.; Santos, M.H. de A.S.; Miguel, M.P. Evaluation of Wound Healing Treated with Latex Derived from Rubber Trees and Aloe Vera Extract in Rats. Acta Cir Bras 2016, 31, 570–577. [CrossRef]
- West, D.P.; Zhu, Y.F. Evaluation of Aloe Vera Gel Gloves in the Treatment of Dry Skin Associated with Occupational Exposure. American Journal of Infection Control 2003, 31, 40–42. [CrossRef]
- Reuter, J.; Jocher, A.; Stump, J.; Grossjohann, B.; Franke, G.; Schempp, C.M. Investigation of the Anti-Inflammatory Potential of Aloe Vera Gel (97.5%) in the Ultraviolet Erythema Test. Skin Pharmacol Physiol 2008, 21, 106–110. [CrossRef]
- Tanaka, M.; Yamamoto, Y.; Misawa, E.; Nabeshima, K.; Saito, M.; Yamauchi, K.; Abe, F.; Furukawa, F. Effects of Aloe Sterol Supplementation on Skin Elasticity, Hydration, and Collagen Score: A 12-Week Double-Blind, Randomized, Controlled Trial. Skin Pharmacol Physiol 2017, 29, 309–317. [CrossRef]
- Kaminaka, C.; Yamamoto, Y.; Sakata, M.; Hamamoto, C.; Misawa, E.; Nabeshima, K.; Saito, M.; Tanaka, M.; Abe, F.; Jinnin, M. Effects of Low-dose Aloe Sterol Supplementation on Skin Moisture, Collagen Score and Objective or Subjective Symptoms: 12-week, Double-blind, Randomized Controlled Trial. J Dermatol 2020, 47, 998–1006. [CrossRef]
- Tungkasamit, T.; Chakrabandhu, S.; Samakgarn, V.; Kunawongkrit, N.; Jirawatwarakul, N.; Chumachote, A.; Chitapanarux, I. Reduction in Severity of Radiation-Induced Dermatitis in Head and Neck Cancer Patients Treated with Topical Aloe Vera Gel: A Randomized Multicenter Double-Blind Placebo-Controlled Trial. European Journal of Oncology Nursing 2022, 59. [CrossRef]
- Jimenez-Garcia, C.; Perula-de Torres, L.A.; Villegas-Becerril, E.; Muñoz-Gavilan, J.J.; Espinosa-Calvo, M.; Montes-Redondo, G.; Romero-Rodriguez, E. Efficacy of an Aloe Vera, Chamomile, and Thyme Cosmetic Cream for the Prophylaxis and Treatment of Mild Dermatitis Induced by Radiation Therapy in Breast Cancer Patients: A Controlled Clinical Trial (Alantel Trials). Trials 2024, 25, 84. [CrossRef]
- Bahraini, P.; Rajabi, M.; Mansouri, P.; Sarafian, G.; Chalangari, R.; Azizian, Z. Turmeric Tonic as a Treatment in Scalp Psoriasis: A Randomized Placebo-Control Clinical Trial. Journal of Cosmetic Dermatology 2018, 17, 461–466. [CrossRef]
- Asada, K.; Ohara, T.; Muroyama, K.; Yamamoto, Y.; Murosaki, S. Effects of Hot Water Extract of Curcuma Longa on Human Epidermal Keratinocytes in Vitro and Skin Conditions in Healthy Participants: A Randomized, Double-Blind, Placebo-Controlled Trial. Journal of Cosmetic Dermatology 2019, 18, 1866–1874. [CrossRef]
- Jalalmanesh, S.; Mansouri, P.; Rajabi, M.; Monji, F. Therapeutic Effects of Turmeric Topical Cream in Vitiligo: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Journal of Cosmetic Dermatology 2022, 21, 4454–4461. [CrossRef]
- Thanawala, S.; Shah, R.; Alluri, K.V.; Bhupathiraju, K.; Salvi, A. Efficacy and Safety of an Oral Low-Dose Water-Dispersible Turmeric Extract Capsule on Facial Skin Health in Healthy Women: A Randomized, Double-Blind, Placebo-Controlled Trial. J Cosmet Dermatol 2025, 24, e70462. [CrossRef]
- Sapkota, B.; Kunwar, P. A Review on Traditional Uses, Phytochemistry and Pharmacological Activities of Calendula Officinalis Linn. Natural Product Communications 2024, 19, 1934578X241259021. [CrossRef]
- Akhtar, N.; Zaman, S.U.; Khan, B.A.; Amir, M.N.; Ebrahimzadeh, M.A. Calendula Extract: Effects on Mechanical Parameters of Human Skin. Acta Pol Pharm 2011, 68, 693–701.
- Sharp, L.; Finnilä, K.; Johansson, H.; Abrahamsson, M.; Hatschek, T.; Bergenmar, M. No Differences between Calendula Cream and Aqueous Cream in the Prevention of Acute Radiation Skin Reactions – Results from a Randomised Blinded Trial. European Journal of Oncology Nursing 2013, 17, 429–435. [CrossRef]
- Sharifi-Heris, Z.; Farahani, L.A.; Haghani, H.; Abdoli-Oskouee, S.; Hasanpoor–Azghady, S.B. Comparison the Effects of Topical Application of Olive and Calendula Ointments on Children’s Diaper Dermatitis: A Triple-Blind Randomized Clinical Trial. Dermatologic Therapy 2018, 31, e12731. [CrossRef]
- Siddiquee, S.; McGee, M.A.; Vincent, A.D.; Giles, E.; Clothier, R.; Carruthers, S.; Penniment, M. Efficacy of Topical Calendula Officinalis on Prevalence of Radiation-Induced Dermatitis: A Randomised Controlled Trial. Australas J Dermatol 2021, 62, e35–e40. [CrossRef]
- Giostri, G.S.; Novak, E.M.; Buzzi, M.; Guarita-Souza, L.C. Treatment of Acute Wounds in Hand with Calendula Officinalis L.: A Randomized Trial. Tissue Barriers 10, 1994822. [CrossRef]
- Zhao, T.; Li, C.; Wang, S.; Song, X. Green Tea (Camellia Sinensis): A Review of Its Phytochemistry, Pharmacology, and Toxicology. Molecules 2022, 27, 3909. [CrossRef]
- Naveed, M.; BiBi, J.; Kamboh, A.A.; Suheryani, I.; Kakar, I.; Fazlani, S.A.; FangFang, X.; kalhoro, S.A.; Yunjuan, L.; Kakar, M.U.; et al. Pharmacological Values and Therapeutic Properties of Black Tea (Camellia Sinensis): A Comprehensive Overview. Biomedicine & Pharmacotherapy 2018, 100, 521–531. [CrossRef]
- Chiu, A.E.; Chan, J.L.; Kern, D.G.; Kohler, S.; Rehmus, W.E.; Kimball, A.B. Double-Blinded, Placebo-Controlled Trial of Green Tea Extracts in the Clinical and Histologic Appearance of Photoaging Skin. Dermatol Surg 2005, 31, 855–860; discussion 860. [CrossRef]
- Mahmood, T.; Akhtar, N.; Khan, B.A.; Khan, H.M.S.; Saeed, T. Outcomes of 3% Green Tea Emulsion on Skin Sebum Production in Male Volunteers. Bosn J Basic Med Sci 2010, 10, 260–264. [CrossRef]
- Mahmood, T.; Akhtar, N.; Khan, B.A.; Shoaib Khan, H.M.; Saeed, T. Changes in Skin Mechanical Properties after Long-Term Application of Cream Containing Green Tea Extract. Aging Clin Exp Res 2011, 23, 333–336. [CrossRef]
- Rhodes, L.E.; Darby, G.; Massey, K.A.; Clarke, K.A.; Dew, T.P.; Farrar, M.D.; Bennett, S.; Watson, R.E.B.; Williamson, G.; Nicolaou, A. Oral Green Tea Catechin Metabolites Are Incorporated into Human Skin and Protect against UV Radiation-Induced Cutaneous Inflammation in Association with Reduced Production of pro-Inflammatory Eicosanoid 12-Hydroxyeicosatetraenoic Acid. British Journal of Nutrition 2013, 110, 891–900. [CrossRef]
- Zhang, R.; Ji, Y.; Zhang, X.; Kennelly, E.J.; Long, C. Ethnopharmacology of Hypericum Species in China: A Comprehensive Review on Ethnobotany, Phytochemistry and Pharmacology. Journal of Ethnopharmacology 2020, 254, 112686. [CrossRef]
- Alahmad, A.; Alghoraibi, I.; Zein, R.; Kraft, S.; Dräger, G.; Walter, J.-G.; Scheper, T. Identification of Major Constituents of Hypericum Perforatum L. Extracts in Syria by Development of a Rapid, Simple, and Reproducible HPLC-ESI-Q-TOF MS Analysis and Their Antioxidant Activities. ACS Omega 2022, 7, 13475–13493. [CrossRef]
- Wölfle, U.; Seelinger, G.; Schempp, C.M. Topical Application of St. Johnʼs Wort (Hypericum Perforatum). Planta Med 2014, 80, 109–120. [CrossRef]
- Schempp, C.M.; Windeck, T.; Hezel, S.; Simon, J.C. Topical Treatment of Atopic Dermatitis with St. John’s Wort Cream--a Randomized, Placebo Controlled, Double Blind Half-Side Comparison. Phytomedicine 2003, 10 Suppl 4, 31–37. [CrossRef]
- Kacerovská, D.; Pizinger, K.; Majer, F.; Šmíd, F. Photodynamic Therapy of Nonmelanoma Skin Cancer with Topical Hypericum Perforatum Extract—A Pilot Study. Photochemistry and Photobiology 2008, 84, 779–785. [CrossRef]
- Samadi, S.; Khadivzadeh, T.; Emami, A.; Moosavi, N.S.; Tafaghodi, M.; Behnam, H.R. The Effect of Hypericum Perforatum on the Wound Healing and Scar of Cesarean. The Journal of Alternative and Complementary Medicine 2010, 16, 113–117. [CrossRef]
- Clewell, A.; Barnes, M.; Endres, J.R.; Ahmed, M.; Ghambeer, D.K.S. Efficacy and Tolerability Assessment of a Topical Formulation Containing Copper Sulfate and Hypericum Perforatum on Patients with Herpes Skin Lesions: A Comparative, Randomized Controlled Trial. J Drugs Dermatol 2012, 11, 209–215.
- Liu, H.; Lu, X.; Hu, Y.; Fan, X. Chemical Constituents of Panax Ginseng and Panax Notoginseng Explain Why They Differ in Therapeutic Efficacy. Pharmacological Research 2020, 161, 105263. [CrossRef]
- Sutopo, N.C.; Qomaladewi, N.P.; Lee, H.W.; Lee, M.S.; Kim, J.H.; Cho, J.Y. Comprehensive Understanding and Underlying Molecular Mechanisms of the Adaptogenic Effects of Panax Ginseng. Journal of Ginseng Research 2025, 49, 356–365. [CrossRef]
- Kim, S.H.; Park, K.S.; Chang, M.J.; Sung, J.H. Effects of Panax Ginseng Extract on Exercise-Induced Oxidative Stress. J Sports Med Phys Fitness 2005, 45, 178–182.
- Kim, H.-G.; Cho, J.-H.; Yoo, S.-R.; Lee, J.-S.; Han, J.-M.; Lee, N.-H.; Ahn, Y.-C.; Son, C.-G. Antifatigue Effects of Panax Ginseng C.A. Meyer: A Randomised, Double-Blind, Placebo-Controlled Trial. PLoS One 2013, 8, e61271. [CrossRef]
- Reay, J.L.; Kennedy, D.O.; Scholey, A.B. Single Doses of Panax Ginseng (G115) Reduce Blood Glucose Levels and Improve Cognitive Performance during Sustained Mental Activity. J Psychopharmacol 2005, 19, 357–365. [CrossRef]
- Reay, J.L.; Kennedy, D.O.; Scholey, A.B. Effects of Panax Ginseng, Consumed with and without Glucose, on Blood Glucose Levels and Cognitive Performance during Sustained “mentally Demanding” Tasks. J Psychopharmacol 2006, 20, 771–781. [CrossRef]
- Reay, J.L.; Scholey, A.B.; Kennedy, D.O. Panax Ginseng (G115) Improves Aspects of Working Memory Performance and Subjective Ratings of Calmness in Healthy Young Adults. Human Psychopharmacology: Clinical and Experimental 2010, 25, 462–471. [CrossRef]
- Flanagan, S.D.; DuPont, W.H.; Caldwell, L.K.; Hardesty, V.H.; Barnhart, E.C.; Beeler, M.K.; Post, E.M.; Volek, J.S.; Kraemer, W.J. The Effects of a Korean Ginseng, GINST15, on Hypo-Pituitary-Adrenal and Oxidative Activity Induced by Intense Work Stress. J Med Food 2018, 21, 104–112. [CrossRef]
- Dormal, V.; Jonniaux, L.; Buchet, M.; Simar, L.; Copine, S.; Deldicque, L. Effect of Hydroponically Grown Red Panax Ginseng on Perceived Stress Level, Emotional Processing, and Cognitive Functions in Moderately Stressed Adults: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients 2025, 17, 955. [CrossRef]
- Panossian, A.; Wikman, G.; Sarris, J. Rosenroot (Rhodiola Rosea): Traditional Use, Chemical Composition, Pharmacology and Clinical Efficacy. Phytomedicine 2010, 17, 481–493. [CrossRef]
- Ivanova Stojcheva, E.; Quintela, J.C. The Effectiveness of Rhodiola Rosea L. Preparations in Alleviating Various Aspects of Life-Stress Symptoms and Stress-Induced Conditions—Encouraging Clinical Evidence. Molecules 2022, 27, 3902. [CrossRef]
- Darbinyan, V.; Kteyan, A.; Panossian, A.; Gabrielian, E.; Wikman, G.; Wagner, H. Rhodiola Rosea in Stress Induced Fatigue — A Double Blind Cross-over Study of a Standardized Extract SHR-5 with a Repeated Low-Dose Regimen on the Mental Performance of Healthy Physicians during Night Duty. Phytomedicine 2000, 7, 365–371. [CrossRef]
- Bystritsky, A.; Kerwin, L.; Feusner, J.D. A Pilot Study of Rhodiola Rosea (Rhodax) for Generalized Anxiety Disorder (GAD). J Altern Complement Med 2008, 14, 175–180. [CrossRef]
- Olsson, E.M.G.; Schéele, B. von; Panossian, A.G. A Randomised, Double-Blind, Placebo-Controlled, Parallel-Group Study of the Standardised Extract SHR-5 of the Roots of Rhodiola Rosea in the Treatment of Subjects with Stress-Related Fatigue. Planta Medica 2008, 75, 105–112. [CrossRef]
- Ross, S.M. Rhodiola Rosea (SHR-5), Part I: A Proprietary Root Extract of Rhodiola Rosea Is Found to Be Effective in the Treatment of Stress-Related Fatigue. Holist Nurs Pract 2014, 28, 149–154. [CrossRef]
- Cropley, M.; Banks, A.P.; Boyle, J. The Effects of Rhodiola Rosea L. Extract on Anxiety, Stress, Cognition and Other Mood Symptoms. Phytotherapy Research 2015, 29, 1934–1939. [CrossRef]
- Mao, J.J.; Xie, S.X.; Zee, J.; Soeller, I.; Li, Q.S.; Rockwell, K.; Amsterdam, J.D. Rhodiola Rosea versus Sertraline for Major Depressive Disorder: A Randomized Placebo-Controlled Trial. Phytomedicine 2015, 22, 394–399. [CrossRef]
- Ballmann, C.G.; Maze, S.B.; Wells, A.C.; Marshall, M.M.; Rogers, R.R. Effects of Short-Term Rhodiola Rosea (Golden Root Extract) Supplementation on Anaerobic Exercise Performance. J Sports Sci 2019, 37, 998–1003. [CrossRef]
- Marcos-Frutos, D.; Leban, Ž.; Li, Z.; Zhang, X.; Lara, P.M.; Alix-Fages, C.; Jiménez-Martínez, P.; Zebboudji, N.; Caillet, A.; Redondo, B.; et al. The Impact of Rhodiola Rosea Extract on Strength Performance in Alternative Bench-Press and Bench-Pull Exercises Under Resting and Mental Fatigue Conditions: A Randomized, Triple-Blinded, Placebo-Controlled, Crossover Trial. Nutrients 2025, 17, 940. [CrossRef]
- Saleem, S.; Muhammad, G.; Hussain, M.A.; Altaf, M.; Bukhari, S.N.A. Withania Somnifera L.: Insights into the Phytochemical Profile, Therapeutic Potential, Clinical Trials, and Future Prospective. Iran J Basic Med Sci 2020, 23, 1501–1526. [CrossRef]
- Orrù, A.; Casu, M.A.; Tambaro, S.; Marchese, G.; Casu, G.; Ruiu, S. Withania Somnifera (L.) Dunal Root Extract Alleviates Formalin-Induced Nociception in Mice: Involvement of the Opioidergic System. Behav Pharmacol 2016, 27, 57–68. [CrossRef]
- Orrù, A.; Marchese, G.; Ruiu, S. Alkaloids in Withania Somnifera (L.) Dunal Root Extract Contribute to Its Anti-Inflammatory Activity. Pharmacology 2023, 108, 301–307. [CrossRef]
- Khalid, M.U.; Sultan, M.T.; Baig, I.; Abbas, A.; Noman, A.M.; Zinedine, A.; Bartkiene, E.; Rocha, J.M. A Comprehensive Review on the Bioactivity and Pharmacological Attributes of Withania Somnifera. Natural Product Research 0, 1–15. [CrossRef]
- Chandrasekhar, K.; Kapoor, J.; Anishetty, S. A Prospective, Randomized Double-Blind, Placebo-Controlled Study of Safety and Efficacy of a High-Concentration Full-Spectrum Extract of Ashwagandha Root in Reducing Stress and Anxiety in Adults. Indian J Psychol Med 2012, 34, 255–262. [CrossRef]
- Biswal, B.M.; Sulaiman, S.A.; Ismail, H.C.; Zakaria, H.; Musa, K.I. Effect of Withania Somnifera (Ashwagandha) on the Development of Chemotherapy-Induced Fatigue and Quality of Life in Breast Cancer Patients. Integr Cancer Ther 2013, 12, 312–322. [CrossRef]
- Wankhede, S.; Langade, D.; Joshi, K.; Sinha, S.R.; Bhattacharyya, S. Examining the Effect of Withania Somnifera Supplementation on Muscle Strength and Recovery: A Randomized Controlled Trial. J Int Soc Sports Nutr 2015, 12, 43. [CrossRef]
- Choudhary, D.; Bhattacharyya, S.; Bose, S. Efficacy and Safety of Ashwagandha (Withania Somnifera (L.) Dunal) Root Extract in Improving Memory and Cognitive Functions. Journal of Dietary Supplements 2017, 14, 599–612. [CrossRef]
- Ziegenfuss, T.N.; Kedia, A.W.; Sandrock, J.E.; Raub, B.J.; Kerksick, C.M.; Lopez, H.L. Effects of an Aqueous Extract of Withania Somnifera on Strength Training Adaptations and Recovery: The STAR Trial. Nutrients 2018, 10, 1807. [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Malvi, H.; Kodgule, R. An Investigation into the Stress-Relieving and Pharmacological Actions of an Ashwagandha (Withania Somnifera) Extract. Medicine (Baltimore) 2019, 98, e17186. [CrossRef]
- Lopresti, A.L.; Drummond, P.D.; Smith, S.J. A Randomized, Double-Blind, Placebo-Controlled, Crossover Study Examining the Hormonal and Vitality Effects of Ashwagandha (Withania Somnifera) in Aging, Overweight Males. Am J Mens Health 2019, 13, 1557988319835985. [CrossRef]
- Smith, S.J.; Lopresti, A.L.; Fairchild, T.J. Exploring the Efficacy and Safety of a Novel Standardized Ashwagandha (Withania Somnifera) Root Extract (Witholytin®) in Adults Experiencing High Stress and Fatigue in a Randomized, Double-Blind, Placebo-Controlled Trial. J Psychopharmacol 2023, 37, 1091–1104. [CrossRef]
- Leonard, M.; Dickerson, B.; Estes, L.; Gonzalez, D.E.; Jenkins, V.; Johnson, S.; Xing, D.; Yoo, C.; Ko, J.; Purpura, M.; et al. Acute and Repeated Ashwagandha Supplementation Improves Markers of Cognitive Function and Mood. Nutrients 2024, 16, 1813. [CrossRef]
- Pandit, S.; Srivastav, A.K.; Sur, T.K.; Chaudhuri, S.; Wang, Y.; Biswas, T.K. Effects of Withania Somnifera Extract in Chronically Stressed Adults: A Randomized Controlled Trial. Nutrients 2024, 16, 1293. [CrossRef]
- Verma, N.; Gupta, S.K.; Patil, S.; Tiwari, S.; Mishra, A.K. Effects of Ashwagandha ( Withania Somnifera) Standardized Root Extract on Physical Endurance and VO 2max in Healthy Adults Performing Resistance Training: An Eight-Week, Prospective, Randomized, Double-Blind, Placebo-Controlled Study. F1000Res 2024, 12, 335. [CrossRef]
- Coope, O.C.; Reales Salguero, A.; Spurr, T.; Páez Calvente, A.; Domenech Farre, A.; Jordán Fisas, E.; Lloyd, B.; Gooderick, J.; Abad Sangrà, M.; Roman-Viñas, B. Effects of Root Extract of Ashwagandha (Withania Somnifera) on Perception of Recovery and Muscle Strength in Female Athletes. Eur J Sport Sci 2025, 25, e12265. [CrossRef]
- Holtmann, S.; Clarke, A.H.; Scherer, H.; Höhn, M. The Anti-Motion Sickness Mechanism of Ginger. A Comparative Study with Placebo and Dimenhydrinate. Acta Otolaryngol 1989, 108, 168–174. [CrossRef]
- Yew, Y.W.; Kuan, A.H.Y.; Ge, L.; Yap, C.W.; Heng, B.H. Psychosocial Impact of Skin Diseases: A Population-Based Study. PLoS One 2020, 15, e0244765. [CrossRef]
- Farkas, Á.; Farkas, G. Effects of Spaceflight on Human Skin. Skin Pharmacol Physiol 2021, 34, 239–245. [CrossRef]
- Tozzo, P.; Delicati, A.; Caenazzo, L. Skin Microbial Changes during Space Flights: A Systematic Review. Life (Basel) 2022, 12, 1498. [CrossRef]
- Nazerke Bolatkyzy Medicinal Plants for Skin Disorders: Phytochemistry and Pharmacological Insights.
- Israyilova, A.; Peykova, T.Z.; Kittleson, B.; Sprowl, P.C.; Mohammed, T.O.; Quave, C.L. From Plant to Patient: A Historical Perspective and Review of Selected Medicinal Plants in Dermatology. JID Innov 2024, 5, 100321. [CrossRef]
- Dawid-Pać, R. Medicinal Plants Used in Treatment of Inflammatory Skin Diseases. Postepy Dermatol Alergol 2013, 30, 170–177. [CrossRef]
- Ahuja, A.; Gupta, J.; Gupta, R. Miracles of Herbal Phytomedicines in Treatment of Skin Disorders: Natural Healthcare Perspective. Infectious Disorders - Drug Targets 21, 328–338. [CrossRef]
- Esmaealzadeh, N.; Iranpanah, A.; Sarris, J.; Rahimi, R. A Literature Review of the Studies Concerning Selected Plant-Derived Adaptogens and Their General Function in Body with a Focus on Animal Studies. Phytomedicine 2022, 105, 154354. [CrossRef]
- Kos, G.; Czarnek, K.; Sadok, I.; Krzyszczak-Turczyn, A.; Kubica, P.; Fila, K.; Emre, G.; Tatarczak-Michalewska, M.; Latalska, M.; Blicharska, E.; et al. Eleutherococcus Senticosus (Acanthopanax Senticosus): An Important Adaptogenic Plant. Molecules 2025, 30, 2512. [CrossRef]
- Yang, K.; Qiu, J.; Huang, Z.; Yu, Z.; Wang, W.; Hu, H.; You, Y. A Comprehensive Review of Ethnopharmacology, Phytochemistry, Pharmacology, and Pharmacokinetics of Schisandra Chinensis (Turcz.) Baill. and Schisandra Sphenanthera Rehd. et Wils. Journal of Ethnopharmacology 2022, 284, 114759. [CrossRef]
- Obembe, O.O.; Popoola, J.O.; Leelavathi, S.; Reddy, S.V. Advances in Plant Molecular Farming. Biotechnol Adv 2011, 29, 210–222. [CrossRef]
- Chaudhary, S.; Ali, Z.; Mahfouz, M. Molecular Farming for Sustainable Production of Clinical-Grade Antimicrobial Peptides. Plant Biotechnol J 2024, 22, 2282–2300. [CrossRef]
- McNulty, M.J.; Xiong, Y.M.; Yates, K.; Karuppanan, K.; Hilzinger, J.M.; Berliner, A.J.; Delzio, J.; Arkin, A.P.; Lane, N.E.; Nandi, S.; et al. Molecular Pharming to Support Human Life on the Moon, Mars, and Beyond. Crit Rev Biotechnol 2021, 41, 849–864. [CrossRef]
- Eidenberger, L.; Kogelmann, B.; Steinkellner, H. Plant-Based Biopharmaceutical Engineering. Nat Rev Bioeng 2023, 1, 426–439. [CrossRef]
- Lam, K.S.; Gustavson, D.R.; Pirnik, D.L.; Pack, E.; Bulanhagui, C.; Mamber, S.W.; Forenza, S.; Stodieck, L.S.; Klaus, D.M. The Effect of Space Flight on the Production of Actinomycin D by Streptomyces Plicatus. J Ind Microbiol Biotechnol 2002, 29, 299–302. [CrossRef]
- Benoit, M.R.; Li, W.; Stodieck, L.S.; Lam, K.S.; Winther, C.L.; Roane, T.M.; Klaus, D.M. Microbial Antibiotic Production Aboard the International Space Station. Appl Microbiol Biotechnol 2006, 70, 403–411. [CrossRef]
- Zhu, X.; Liu, X.; Liu, T.; Wang, Y.; Ahmed, N.; Li, Z.; Jiang, H. Synthetic Biology of Plant Natural Products: From Pathway Elucidation to Engineered Biosynthesis in Plant Cells. Plant Commun 2021, 2, 100229. [CrossRef]
- Douglas Kinghorn, A. Pharmacognosy in the 21st Century. J Pharm Pharmacol 2001, 53, 135–148. [CrossRef]
- Daduang, R.; Suwanchaikasem, P.; Rattanapisit, K.; Vitayathikornnasak, S.; Srisangsung, T.; Bulaon, C.J.I.; Phoolcharoen, W. LC-MS Determination of Nicotiana Benthamiana Host Plant Proteins in the Drug Products of Recombinant Plant-Produced Pembrolizumab. Sci Rep 2025, 15, 25635. [CrossRef]
- Wang, Y.; Demirer, G.S. Synthetic Biology for Plant Genetic Engineering and Molecular Farming. Trends Biotechnol 2023, 41, 1182–1198. [CrossRef]
- Frick, S.; Kramell, R.; Kutchan, T.M. Metabolic Engineering with a Morphine Biosynthetic P450 in Opium Poppy Surpasses Breeding. Metab Eng 2007, 9, 169–176. [CrossRef]
- Liu, B.; Wang, H.; Du, Z.; Li, G.; Ye, H. Metabolic Engineering of Artemisinin Biosynthesis in Artemisia Annua L. Plant Cell Rep 2011, 30, 689–694. [CrossRef]
- Trujillo, E.; Angulo, C. Perspectives on the Use of the CRISPR System in Plants to Improve Recombinant Therapeutic Protein Production. J Biotechnol 2025, 405, 111–123. [CrossRef]
- Ebrahimi, V.; Hashemi, A. CRISPR-Based Gene Editing in Plants: Focus on Reagents and Their Delivery Tools. Bioimpacts 2025, 15, 30019. [CrossRef]
- Baeshen, N.A.; Baeshen, M.N.; Sheikh, A.; Bora, R.S.; Ahmed, M.M.M.; Ramadan, H.A.I.; Saini, K.S.; Redwan, E.M. Cell Factories for Insulin Production. Microb Cell Fact 2014, 13, 141. [CrossRef]
- Holland, T.; Buyel, J.F. Bioreactor-Based Production of Glycoproteins in Plant Cell Suspension Cultures. Methods Mol Biol 2018, 1674, 129–146. [CrossRef]
- Frigerio, R.; Marusic, C.; Villani, M.E.; Lico, C.; Capodicasa, C.; Andreano, E.; Paciello, I.; Rappuoli, R.; Salzano, A.M.; Scaloni, A.; et al. Production of Two SARS-CoV-2 Neutralizing Antibodies with Different Potencies in Nicotiana Benthamiana. Front Plant Sci 2022, 13, 956741. [CrossRef]
- Verdú-Navarro, F.; Moreno-Cid, J.A.; Weiss, J.; Egea-Cortines, M. The Advent of Plant Cells in Bioreactors. Front Plant Sci 2023, 14, 1310405. [CrossRef]
- Motolinía-Alcántara, E.A.; Castillo-Araiza, C.O.; Rodríguez-Monroy, M.; Román-Guerrero, A.; Cruz-Sosa, F. Engineering Considerations to Produce Bioactive Compounds from Plant Cell Suspension Culture in Bioreactors. Plants (Basel) 2021, 10, 2762. [CrossRef]
- Walther, I. Space Bioreactors and Their Applications. Adv Space Biol Med 2002, 8, 197–213. [CrossRef]
- Yeğen, E. An inquiry of space architecture: design considerations and design process. Master Thesis, Middle East Technical University, 2019.
- Jeong, J.-E.; Park, S.-A. Physiological and Psychological Effects of Visual Stimulation with Green Plant Types. Int J Environ Res Public Health 2021, 18, 12932. [CrossRef]
- Paniccià, M.; Acito, M.; Grappasonni, I. How Outdoor and Indoor Green Spaces Affect Human Health: A Literature Review. Ann Ig 2025, 37, 333–349. [CrossRef]
- Gaekwad, J.S.; Sal Moslehian, A.; Roös, P.B.; Walker, A. A Meta-Analysis of Emotional Evidence for the Biophilia Hypothesis and Implications for Biophilic Design. Front Psychol 2022, 13, 750245. [CrossRef]
- Schlacht, I.L.; Kolrep, H.; Daniel, S.; Musso, G. Impact of Plants in Isolation: The EDEN-ISS Human Factors Investigation in Antarctica. In Proceedings of the Advances in Human Factors of Transportation; Springer, Cham, 2020; pp. 794–806.
- Liu, T.; He, L.; Yu, W.; Freudenreich, T.; Lin, X. Effect of Green Plants on Individuals’ Mental Stress during the COVID-19 Pandemic: A Preliminary Study. Int J Environ Res Public Health 2022, 19, 13541. [CrossRef]
- Li, Z.; Liu, H.; Zhang, W.; Liu, H. Psychophysiological and Cognitive Effects of Strawberry Plants on People in Isolated Environments. J Zhejiang Univ Sci B 2020, 21, 53–63. [CrossRef]
- Manzoor, S.; Rakha, A.; Rasheed, H.; Bhat, Z.F.; Khan, M.S.A.; Abdi, G.; Aadil, R.M. A Comprehensive Review on Anxiolytic Effect of Lavandula Angustifolia Mill. in Clinical Studies. Food Sci Nutr 2025, 13, e70993. [CrossRef]
- Luo, R.; Lun, X.; Gao, R.; Wang, L.; Yang, Y.; Su, X.; Habibullah-Al-Mamun, M.; Xu, X.; Li, H.; Li, J. A Review of Biogenic Volatile Organic Compounds from Plants: Research Progress and Future Prospects. Toxics 2025, 13, 364. [CrossRef]
- Antonelli, M.; Donelli, D.; Barbieri, G.; Valussi, M.; Maggini, V.; Firenzuoli, F. Forest Volatile Organic Compounds and Their Effects on Human Health: A State-of-the-Art Review. Int J Environ Res Public Health 2020, 17, 6506. [CrossRef]
- Doerr, D.F.; Convertino, V.A.; Blue, J.; Wheeler, R.M.; Knott, W.M. Interaction between Exercising Humans and Growing Plants in a Closed Ecological Life Support System. Acta Astronaut 1995, 36, 601–605. [CrossRef]
- Zabel, P.; Bamsey, M.; Schubert, D.; Tajmar, M. Review and Analysis of over 40 Years of Space Plant Growth Systems. Life Sci Space Res (Amst) 2016, 10, 1–16. [CrossRef]
- Galston, A.W. Photosynthesis as a Basis for Life Support on Earth and in Space: Photosynthesis and Transpiration in Enclosed Spaces. Bioscience 1992, 42, 490–493.
- Thornfeldt, C.R. Therapeutic Herbs Confirmed by Evidence-Based Medicine. Clin Dermatol 2018, 36, 289–298. [CrossRef]
- Koonrungsesomboon, N.; Sakuludomkan, C.; Na Takuathung, M.; Klinjan, P.; Sawong, S.; Perera, P.K. Study Design of Herbal Medicine Clinical Trials: A Descriptive Analysis of Published Studies Investigating the Effects of Herbal Medicinal Products on Human Participants. BMC Complement Med Ther 2024, 24, 391. [CrossRef]
- Ullah, H.; Dacrema, M.; Buccato, D.G.; Fayed, M.A.A.; De Lellis, L.F.; Morone, M.V.; Di Minno, A.; Baldi, A.; Daglia, M. A Narrative Review on Plant Extracts for Metabolic Syndrome: Efficacy, Safety, and Technological Advances. Nutrients 2025, 17, 877. [CrossRef]
- Palit, P.; Mandal, S.C. Climate Change, Geographical Location, and Other Allied Triggering Factors Modulate the Standardization and Characterization of Traditional Medicinal Plants: A Challenge and Prospect for Phyto-Drug Development. In Evidence Based Validation of Traditional Medicines; Springer, Singapore, 2021; pp. 359–369 ISBN 978-981-15-8127-4.
- Persaud, T.; Pathak, Y.V. Evaluation of Physical and Chemical Changes in Pharmaceuticals Flown on Space Missions. In Handbook of Space Pharmaceuticals; Springer, Cham, 2022; pp. 179–207 ISBN 978-3-030-05526-4.
- Plante, I.; Daniels, V.; Young, M.; Gaza, R.; Wu, H.; Reichard, J.F. The Long-Term Stability of Solid-State Oral Pharmaceuticals Exposed to Simulated Intravehicular Space Radiation. npj Microgravity 2025, 11, 17. [CrossRef]
- Shin, J.W.; Lee, J.-W.; Yu, S.; Baek, B.K.; Hong, J.P.; Seo, Y.; Kim, W.N.; Hong, S.M.; Koo, C.M. Polyethylene/Boron-Containing Composites for Radiation Shielding. Thermochimica Acta 2014, 585, 5–9. [CrossRef]
- Moyne, P.; Botella, A.; Peyrouset, A.; Rey, L. Sterilization of Injectable Drugs Solutions by Irradiation. Radiation Physics and Chemistry - RADIAT PHYS CHEM 2002, 63, 703–704. [CrossRef]
- Meents, A.; Gutmann, S.; Wagner, A.; Schulze-Briese, C. Origin and Temperature Dependence of Radiation Damage in Biological Samples at Cryogenic Temperatures. Proc Natl Acad Sci U S A 2010, 107, 1094–1099. [CrossRef]
- Oluwafemi, F.A.; De La Torre, A.; Afolayan, E.M.; Olalekan-Ajayi, B.M.; Dhital, B.; Mora-Almanza, J.G.; Potrivitu, G.; Creech, J.; Rivolta, A. Space Food and Nutrition in a Long Term Manned Mission. Advances in Astronautics Science and Technology 2018, 1, 1–21. [CrossRef]
- Lasseur, C. Melissa: The European Project of a Closed Life Support System. 2008.
- Nie, H.; Zhou, W.; Zheng, Z.; Deng, Y.; Zhang, W.; Zhang, M.; Jiang, Z.; Zheng, H.; Yuan, L.; Yang, J.; et al. Exploring Plant Responses to Altered Gravity for Advancing Space Agriculture. Plant Commun 2025, 6, 101370. [CrossRef]
- Hughes, A.M.; Kiss, J.Z. -Omics Studies of Plant Biology in Spaceflight: A Critical Review of Recent Experiments. Front. Astron. Space Sci. 2022, 9. [CrossRef]
- Gao, W.; Li, K.; Yan, S.; Gao, X.; Hu, L. Effects of Space Flight on DNA Mutation and Secondary Metabolites of Licorice (Glycyrrhiza Uralensis Fisch.). Sci China C Life Sci 2009, 52, 977–981. [CrossRef]
- Dong, Y.-Y.; Gao, W.-Y.; Zhang, J.-Z.; Zuo, B.-M.; Huang, L.-Q. Quantification of Four Active Ingredients and Fingerprint Analysis of Licorice (Glycyrrhiza Uralensis Fisch.) after Spaceflight by HPLC–DAD. Res Chem Intermed 2012, 38, 1719–1731. [CrossRef]
- Zhang, J.; Gao, W.; Yan, S.; Zhao, Y. Effects of Space Flight on the Chemical Constituents and Anti-Inflammatory Activity of Licorice (Glycyrrhiza Uralensis Fisch). Iran J Pharm Res 2012, 11, 601–609.
- Xia, P.; Li, Q.; Liang, Z.; Zhang, X.; Yan, K. Spaceflight Breeding Could Improve the Volatile Constituents of Andrographis Paniculata. Industrial Crops and Products 2021, 171, 113967. [CrossRef]
- Kharchenko, V.; Golubkina, N.; Skrypnik, L.; Murariu, O.C.; Vecchietti, L.; Caruso, G. The Effect of One-Year Seed Spaceflight Storage on Yield, Biochemical and Mineral Characteristics of Mature Leafy Vegetables Belonging to Brassicaceae, Apiaceae and Asteraceae Families. Horticulturae 2023, 9, 1073. [CrossRef]
- Mohanta, T.K.; Mishra, A.K.; Mohanta, Y.K.; Al-Harrasi, A. Space Breeding: The Next-Generation Crops. Front Plant Sci 2021, 12, 771985. [CrossRef]
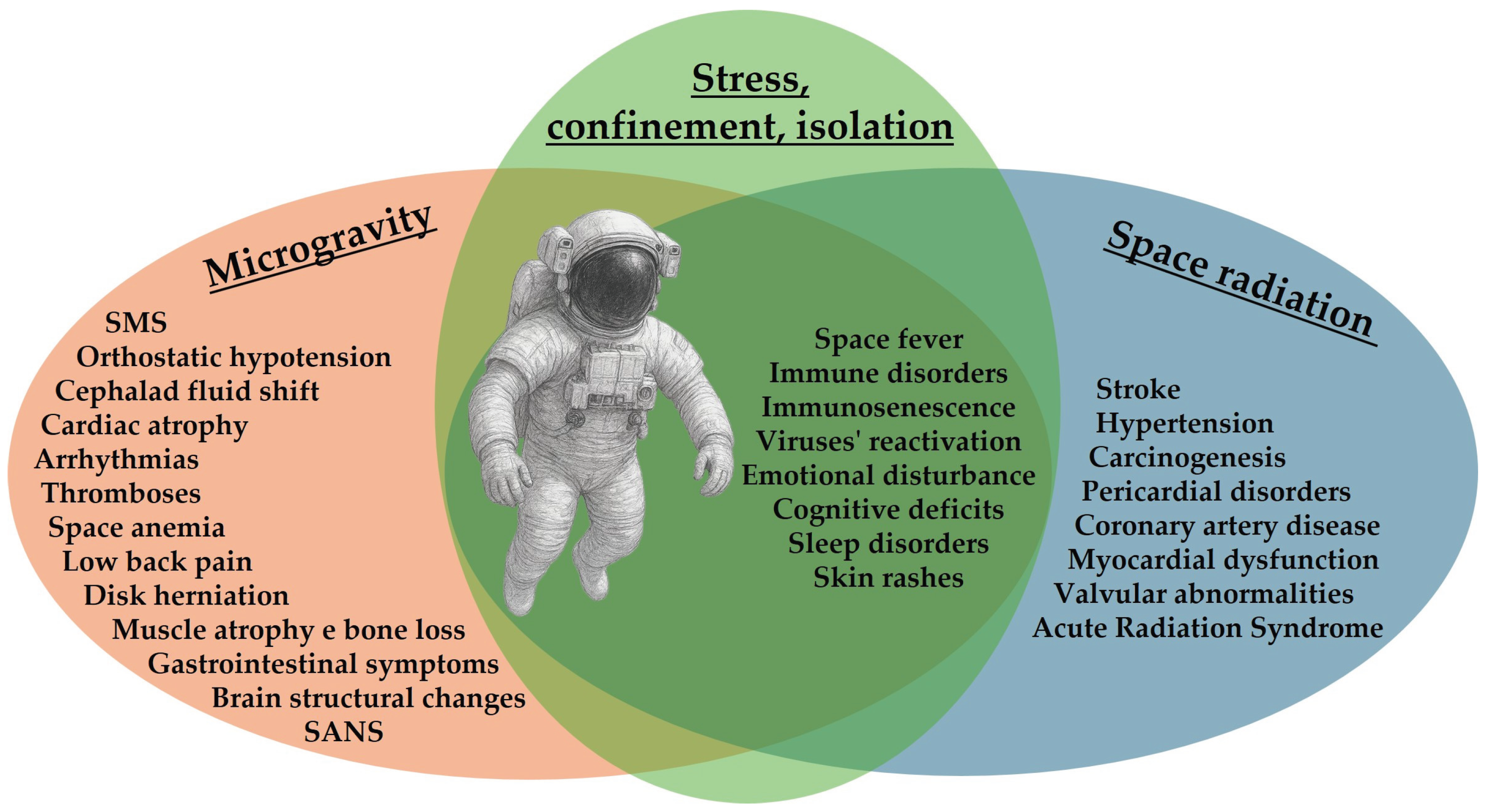
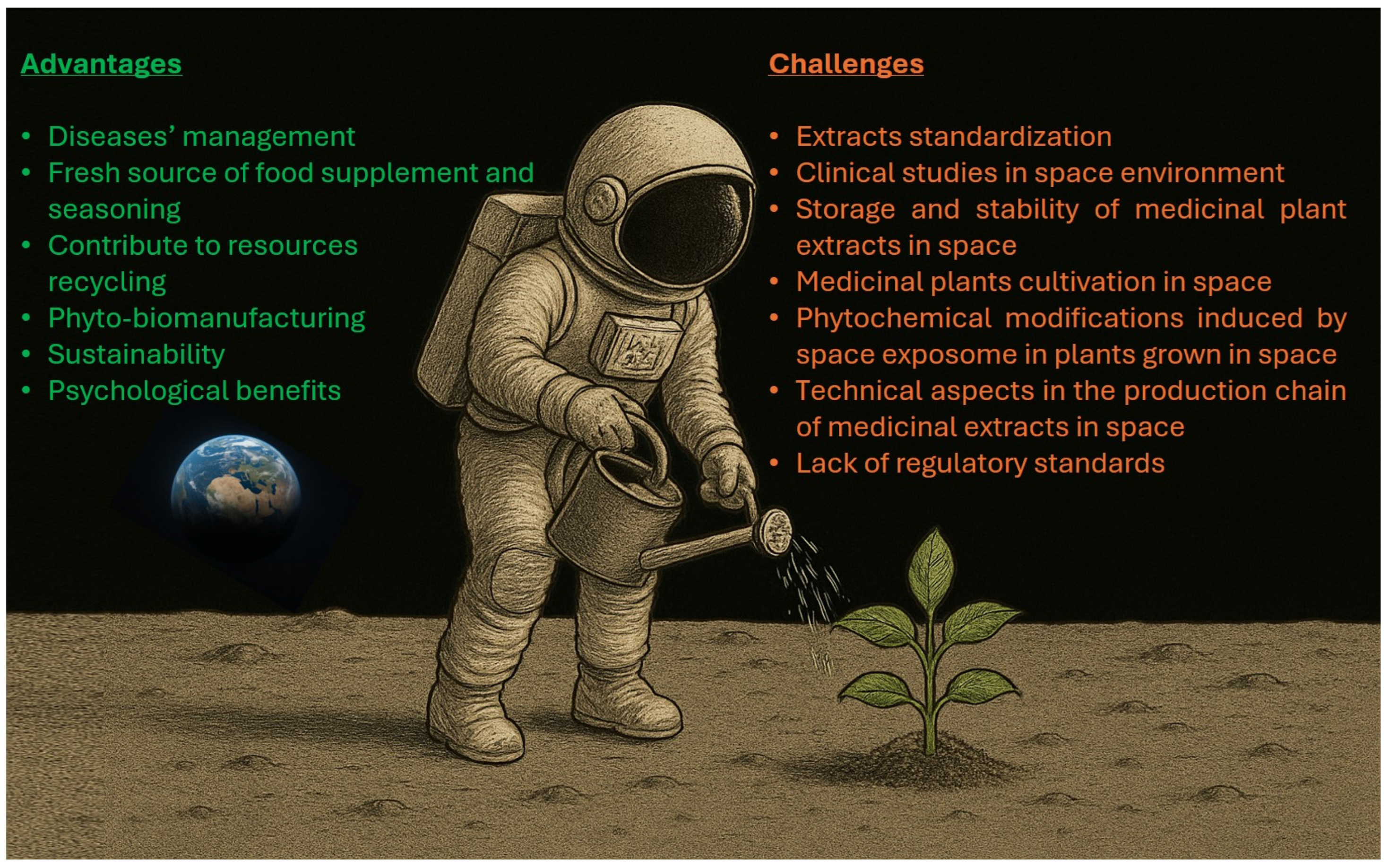
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




