1. Introduction
Hereditary transthyretin (ATTRv) amyloidosis is an autosomal dominant disease with a progressive and fatal course if left untreated. It is caused by pathogenic variants in the gene encoding transthyretin (TTR) [
1,
2]. Over 150 different pathogenic
TTR gene variants (
TTRv) have been described that can cause the TTR tetramer to become unstable, to dissociate into monomers that aggregate into insoluble amyloid fibrils deposited into the extracellular space of tissues and organs [
2,
3,
4]. The most common disease manifestations are autonomic neuropathy (ANP), peripheral neuropathy, and/or cardiomyopathy [
4,
5,
6]. Autonomic manifestations, such as orthostasis, vasomotor, gastrointestinal, bladder and pupillomotor dysfunction, often occur in the early stages of ATTRv amyloidosis. The neuropathy in ATTRv amyloidosis is characterized by early involvement of somatic thin myelinated Aδ and autonomic unmyelinated C fibers, resulting in a length-dependent, symmetrical syndrome of loss of temperature sensation, autonomic symptoms, and burning neuropathic pain. As the disease progresses, large myelinated fibers also become impaired [
7,
8,
9].
In the last decade, several disease-modifying treatments have become available for ATTRv amyloidosis. These treatments are most beneficial when initiated at an early disease stage [
6,
10,
11,
12]. Therefore, systematic and regular monitoring of asymptomatic
TTRv carriers to identify the earliest stages of the disease is recommended to improve the outcome.
Neurofilament light chain (NfL) is a neuron-specific cytoskeletal protein released into the blood and cerebrospinal fluid during axonal damage [
13]. Recent studies demonstrated that serum neurofilament light chain (sNfL) is released following axonal damage to thick myelinated Aβ nerve fibers, leading to polyneuropathy [
14,
15]. sNfL serves as a biomarker for polyneuropathy in
TTRv carriers and ATTRv amyloidosis patients and is indicative of early disease onset [
15]. Axonal damage to the thin myelinated Aδ and unmyelinated C nerve fibers causes ANP [
14]. Therefore, sNfL may be a biomarker not only for polyneuropathy but also for ANP. However, there currently is no evidence regarding the role of sNfL as an early marker in ANP [
15].
The autonomic nervous system comprises two divisions: the sympathetic and parasympathetic. Several tests are available for assessing autonomic nervous system function. The Ewing battery comprises several autonomic function tests and evaluates both sympathetic and parasympathetic cardiovascular autonomic dysfunction [
16,
17]. Iodine-123 labelled metaiodobenzylguanidine ([
123I]
mIBG) scintigraphy is a nuclear imaging method used to assess the sympathetic conducting system of the heart. [
123I]
mIBG is a chemically modified analogue of norepinephrine that normally accumulates in vesicles at sympathetic nerve endings near cardiomyocytes. [
123I]
mIBG scintigraphy reflects the sympathetic tone, making it useful for evaluating the sympathetic innervation in conditions such as cardiac amyloidosis [
18,
19,
20].
This study aims to evaluate whether sNfL can serve as a potential early biomarker for ANP by predicting the presence of ANP in TTRv carriers and ATTRv amyloidosis patients based on sNfL levels. To this end, ANP was evaluated by the Ewing battery, [123I]mIBG scintigraphy, and autonomic symptom assessment.
2. Methods
2.1. Study Participants
In a previous study conducted at the University Medical Center Groningen (UMCG) between November 2007 and October 2017, 40
TTRv carriers and ATTRv amyloidosis patients underwent a comprehensive assessment [
21]. This included Ewing battery, [
123I]
mIBG scintigraphy, nerve conduction studies (NCS), quantitative sensory testing (QST),
99mTechnetium-hydroxymethylene diphosphonate ([
99mTc]Tc-HDP) scintigraphy, and subcutaneous fat tissue aspirates. Four patients from the previous study were excluded due to the absence of serum samples for NfL measurement at the time of [
123I]
mIBG scintigraphy. Two additional patients were included in the current study cohort having undergone all previously mentioned tests between October 2017 and April 2018 for the purposes of routine patient care (
Figure S1).
Serum samples for NfL measurement and subcutaneous fat tissue aspirates within one year before or after [123I]mIBG scintigraphy were included. Levels of creatinine, N-terminal pro-brain-type natriuretic peptide (NT-proBNP) and troponin T at the time of sNfL measurement, as well as the medical history and results of physical examination, were retrieved from the electronic patient records.
In this study, TTRv carriers were defined as carriers of a pathogenic TTR-gene variant without symmetrical distal neuropathic symptoms, or signs of sensory loss and negative NCS and QST; no signs of ANP on Ewing battery and [123I]mIBG scintigraphy; no signs of cardiomyopathy on [99mTc]Tc-HDP scintigraphy; and no amyloid deposits in the subcutaneous fat tissue aspirate. ATTRv amyloidosis patients were defined as carriers of a pathogenic TTR-gene variant with symmetrical distal neuropathic symptoms, or signs of sensory loss that had to be confirmed by NCS or QST, and/or ANP confirmed by Ewing battery and/or [123I]mIBG scintigraphy, and/or signs of cardiomyopathy on [99mTc]Tc-HDP scintigraphy showing any grade of cardiac tracer uptake (i.e., Perugini grade ≥1), and/or a positive subcutaneous fat tissue aspirate (i.e., Congo red score ≥1).
2.2. Ewing Battery
The Ewing battery was used to detect cardiac ANP and included heart rate variability during deep breathing, heart rate response to Valsalva maneuver, and heart rate response to standing up to assess parasympathetic function, as well as blood pressure response to the isometric handgrip test and standing up to evaluate sympathetic function. Parasympathetic dysfunction was diagnosed if at least two parasympathetic function tests are abnormal, and sympathetic dysfunction was diagnosed if one or both of the sympathetic function tests are abnormal [
17,
22].
2.3. [123. I]mIBG Scintigraphy
[
123I]
mIBG scintigraphy was performed as described [
23]. Briefly, after blockage of thyroid uptake by oral administration of iodine potassium iodide, patients were intravenously injected with 185 MBq [
123I]
mIBG. Planar images of the thorax were acquired at fifteen minutes and at four hours after radiotracer injection to quantify the early and late heart-to-mediastinum ratio (HMR) [
23]. HMR was determined by the counts in a manually drawn region of interest (ROI) along the contour of the left ventricle, divided by the counts in a fixed rectangular ROI in the upper mediastinum (carefully leaving the thyroid region out). The cardiac wash-out rate (WR) was defined as a change in percentage of the activity ratio, calculated as follows:
[
18,
20,
23,
24,
25]. Either late HMR <2.0 or WR >20% were considered as abnormal [
123I]
mIBG parameters, and hence suggestive for impaired sympathetic cardiac innervation [
26,
27,
28].
2.4. Patient-Reported Autonomic Symptoms
Symptoms as described in the Composite Autonomic Symptom Score (COMPASS) 31 were used to assess the presence of ANP symptoms [
29].
2.5. Autonomic Neuropathy
According to expert panel consensus in diabetes mellitus patients, the Ewing battery is the gold standard for assessment of ANP. Parasympathetic dysfunction is diagnosed if at least two parasympathetic function tests are abnormal, and sympathetic dysfunction is diagnosed if one or both of the sympathetic function tests are abnormal. We chose not to subcategorize autonomic dysfunction into sympathetic and parasympathetic components to maintain maximum statistical power for analysis. Therefore, Ewing battery results were reported as either normal or abnormal (
ANP consensus definition: definition A) [
17,
22]. As there is no validated consensus definition for ANP in ATTRv amyloidosis patients, we used a modified definition for ANP (
ANP modified definition: definition B), which requires abnormal results in at least two of the following three assessments: Ewing battery, [
123I]
mIBG scintigraphy, and/or the presence of autonomic symptoms as assessed by the COMPASS-31 questionnaire (
Figure S1) [
29].
2.6. Peripheral Neuropathy
QST was performed to detect small fiber neuropathy (SFN) [
12,
30]. NCS were used to detect large fiber neuropathy [
12,
31]. Peripheral neuropathy, including both small and large fiber neuropathy, was defined as symmetrical distal neuropathic symptoms, or signs of sensory loss and had to be confirmed by NCS or QST. The severity of the peripheral neuropathy was graded according to the polyneuropathy disability (PND) score. The PND score indicates how walking is affected: 0: No disturbance; I: Sensory disturbance; II: Walking difficulties, not requiring support or care; IIIa: Need of one stick or one crutch for ambulation; IIIb: Need of two sticks or two crutches for ambulation; IV: Patient confined to a bed or wheelchair [
32].
2.7. Blood Sample Collection and NfL Measurement
Blood samples were drawn by venipuncture at the outpatient clinic of the UMCG, centrifuged at 2700 rpm for 10 minutes at room temperature and were stored within one hour at –20 °C and subsequently stored at –80 °C within six months. Measurements of NfL in serum were performed using the NF-light Kit (Quanterix) on a single molecule array (Simoa HDX) technology (Quanterix Corp., Billerica, MA USA) [
33]. The measurements were performed at the Neurochemistry Laboratory of the Amsterdam University Medical Center by certified technicians who were blinded to clinical information.
2.8. Statistical Analysis
The Kolmogorov-Smirnoff test was used to assess the normality of the data. Patient characteristics were described as number (n), mean±standard deviation (SD) when normally distributed, and median [interquartile range (IQR)] when not normally distributed. Groups were compared using the unpaired t-test, Mann-Whitney U test or Chi-square test (for trend), where appropriate. Univariable correlations were assessed by Spearman’s rho correlation coefficient.
Logistic regression analysis was performed to assess whether sNfL levels predicted ANP. sNfL levels were first converted into a dichotomous variable (normal versus abnormal for age), based on normative data from Vermunt et al. [
34]. In univariable logistic regression, all relevant clinical variables were tested individually with enter inclusion of the variables. Variables that were significant in univariable logistic regression analysis were included in the multivariable logistic regression model using enter inclusion. A p-value <0.05 was considered statistically significant.
IBM SPSS Statistics version 28.0 (IBM Corp., Armonk, New York, USA) was used for statistical analysis. GraphPad Prism 9 (GraphPad Software, La Jolla, California, USA) was used to generate graphs.
2.9. Ethics Approval
All procedures were in compliance with the Declaration of Helsinki. The study was approved by the institutional review board of the UMCG (Registration number 201800204 and 201900860). According to the local Dutch regulations for retrospective observational studies, formal informed consent was not required.
3. Results
3.1. Characteristics of TTRv Carriers and ATTRv Amyloidosis Patients
A total of 38 individuals (19 male and 19 female) were included of which 10 were
TTRv carriers and 28 ATTRv amyloidosis patients. The overall mean age at the time of sNfL measurement was 50±14 years. Most frequent (45%) was the Val30Met (p.Val50Met) genotype. See
Table S1 for a specification of genotypes. Peripheral neuropathy was diagnosed in 17 individuals. Twenty individuals had PND score 0, including 9
TTRv carriers and 11 ATTRv amyloidosis patients without peripheral neuropathy, but with autonomic neuropathy, cardiomyopathy and/or confirmed amyloid deposits in fat tissue biopsy. Cardiomyopathy was diagnosed in 14 individuals. Autonomic symptoms listed on the COMPASS-31 questionnaire were reported by 14 individuals [
29], abnormal results on the Ewing battery by 15 individuals, and abnormal findings on [
123I]
mIBG scintigraphy by 15 individuals. There was a negative correlation between sNfL and late HMR (r=-0.62, p<0.001) and a positive correlation between sNfL and WR (r=0.64, p<0.001). ANP was diagnosed in 15 individuals according to definition A [
22] and in 14 individuals according to definition B. A total of 16 individuals received treatment, either with diflunisal (n=10) or tafamidis (n=6) at the time of blood collection (
Table 1).
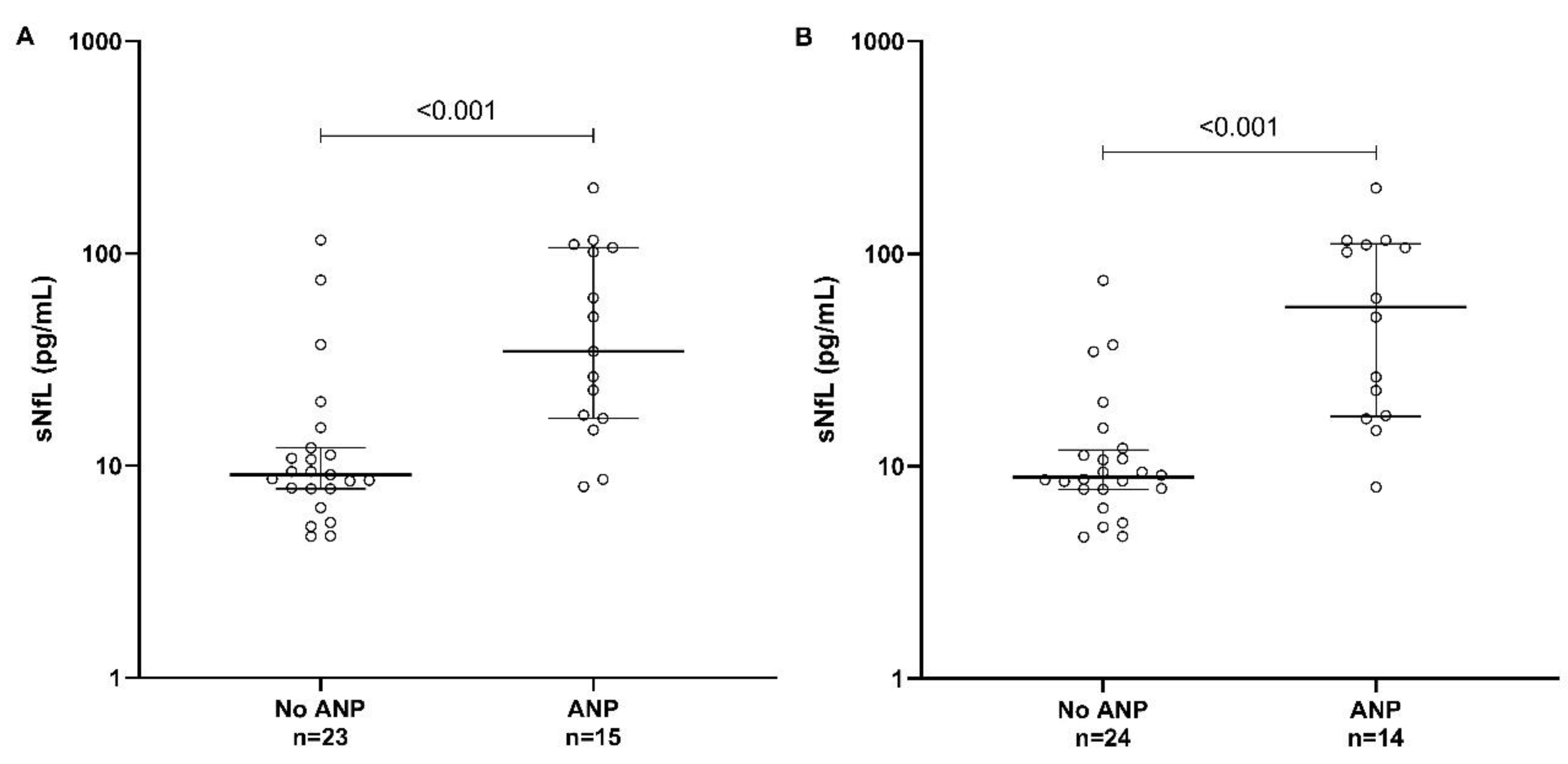
3.2. sNfL in Relation to Autonomic Neuropathy
According to ANP definition A [
22], the median sNfL level was higher in individuals with ANP (n=15) compared to those without ANP (n=23), 34.6 pg/mL [16.7–106.5] and 9.1 pg/mL [7.8–12.1] (p<0.001), respectively (
Table 2 and
Figure 1A).
According to ANP definition B, the median sNfL level was higher in individuals with ANP (n=14) compared to those without ANP (n=24), 56.0 pg/mL [17.3–110.1] and 8.9 pg/mL [7.8–11.7] (p<0.001), respectively (
Table 2 and
Figure 1B).
Table 2 presents the median sNfL levels based on results from the Ewing battery, [
123I]
mIBG scintigraphy, and assessment of autonomic symptoms.
3.3. sNfL Levels in Patients with Autonomic Neuropathy and No Peripheral Neuropathy
Median sNfL levels in individuals with only ANP (definition A n=4; and definition B n=3) did not significantly differ compared to those without any form of neuropathy (definition A n=17; definition B n=18) (
Figure S2A,B).
3.4. Univariable Logistic Regression Analysis
Univariable logistic regression analysis showed that peripheral neuropathy had the highest explained variance for age-adjusted sNfL status based on the Nagelkerke R
2 (
Table 3). The variables sex, autonomic symptoms, Ewing battery, [
123I]
mIBG scintigraphy, ANP definition A, ANP definition B, and peripheral neuropathy were all significantly associated with age-adjusted sNfL status (p<0.05) (
Table 3).
3.5. Multivariable Logistic Regression Analysis
Variables that were significant in univariable logistic regression were included in the multivariable logistic regression model.
In
Model A, the dependent variable age-adjusted sNfL status and the independent variables sex, ANP definition A [
22], and peripheral neuropathy were included (
Table 4A). Among these, only peripheral neuropathy (odds ratio (OR) 25.03 95% confidence interval (CI) 2.33–269.26) was identified as independent predictor for age-adjusted sNfL status (p<0.05). The Nagelkerke R
2 for peripheral neuropathy improved from 0.57 in univariable logistic regression analysis to 0.64 in this model.
In
Model B, ANP definition B was used instead of ANP definition A. The other included variables remained the same (
Table 4B). Again, only peripheral neuropathy (OR 24.24 95% CI 2.23–263.89) was identified as independent predictor for age-adjusted sNfL status (p<0.05). The Nagelkerke R
2 for peripheral neuropathy improved from 0.57 in univariable logistic regression analysis to 0.64 in this model.
4. Discussion
This study shows that (1) sNfL levels are increased in ATTRv amyloidosis patients with ANP according to two different definitions; (2) sNfL levels in patients with ANP are increased predominantly because of the concurrent presence of peripheral neuropathy in these patients. Therefore, sNfL is not a useful early biomarker to detect ANP in pathogenic TTRv carriers and ATTRv amyloidosis patients. This emphasizes the continued necessity of current screening and diagnostic methods for the detection of ANP.
NfL is released into the blood following peripheral neuronal damage, particularly to thick myelinated Aβ nerve fibers [
14,
15]. Axonal damage to the somatic thin myelinated Aδ and autonomic unmyelinated C nerve fibers may also result in the release of NfL [
14]. Therefore, we hypothesized that NfL could serve as an early biomarker for ANP. Indeed, we observed increased sNfL levels in individuals with ANP compared to those without ANP (
Figure 1A,B). To further investigate the relationship between sNfL and ANP, we performed logistic regression analysis to assess whether ANP could be predicted based on age-adjusted sNfL status. Multivariable logistic regression analysis revealed that peripheral neuropathy in this study was the sole independent predictor of age-adjusted sNfL status. Individuals with peripheral neuropathy had approximately 25 times higher odds of having an abnormal age-adjusted sNfL status compared to those without peripheral neuropathy (
Table 4A,B). In line with the results of logistic regression analysis, we observed that the median sNfL levels did not significantly differ between individuals with only ANP (definition A n=4; definition B n=3) compared to individuals without any form of neuropathy (definition A n=17; definition B n=18) (
Figure S2A,B). Based on these findings, sNfL is not a reliable early biomarker for ANP in
TTRv carriers and ATTRv amyloidosis patients.
Few studies have investigated the role of sNfL in ANP, limiting direct comparisons. Luigetti et al. investigated correlations between sNfL and systemic disease parameters in patients with ATTRv amyloidosis [
35]. They used the questionnaire-based compound autonomic dysfunction test (CADT) and Sudoscan to assess autonomic dysfunction. Significant correlations were reported between NfL levels and Sudoscan parameters, but not CADT scores. However, they did not correct for the presence of polyneuropathy [
35]. Similarly, Russo
et al. observed a significant correlation between sNfL and disease severity in ATTRv amyloidosis patients, including autonomic dysfunction as assessed by the CADT [
36]. However, this study also lacked adjustment for factors known to elevate sNfL levels, such as large fiber neuropathy. Another study in patients with type II diabetes mellitus with or without diabetic neuropathy assessed heart rate variability measures and sNfL levels [
37]. After adjusting for glycemic control, they found significant correlations between sNfL and the standard deviation of the R-R intervals and the root mean square differences of successive R-R intervals. This suggests that sNfL could play a role in identifying individuals at risk of cardiac ANP. However, as with the aforementioned studies, they did not adjust for the presence of peripheral neuropathy [
37].
Taken together, our findings and those from the aforementioned studies indicate a potential association between sNfL and autonomic dysfunction [
35,
36,
37]. However, the impact of confounding factors, particularly co-existing peripheral neuropathy, had not been thoroughly investigated. Based on our data, we conclude that sNfL does not serve as an early biomarker for isolated ANP in ATTRv amyloidosis patients. As ANP is a form of SFN that affects the thin myelinated Aδ and unmyelinated C nerve fibers, we reviewed the literature on SFN and NfL [
14]. Galosi
et al. assessed sNfL levels and SFN in ATTRv amyloidosis [
38]. Consistent with previous studies [
39,
40,
41], they found that intraepidermal nerve fiber density (IENFD) and thermal thresholds measured by QST are often impaired early, even in asymptomatic
TTRv carriers. Furthermore, they observed a significant negative correlation between sNfL and distal IENFD, and sNfL levels significantly correlated with QST measures indicating small fiber impairment. However, they did not account for the presence of polyneuropathy in their analysis, nor did they compare sNfL levels between asymptomatic
TTRv carriers with normal versus abnormal IENFD and QST results [
38]. In another study, sNfL levels were investigated in a cohort of patients with SFN of various aetiologies and they showed that sNfL levels did not correlate with any parameters of small nerve fiber function [
42]. Both studies suggested that either the NfL content of small intraepidermal nerve fibers is too low, or that the axonal damage in SFN is generally too subtle to detect altered sNfL levels [
38,
42]. Our finding that sNfL levels are not an independent predictor of ANP is consistent with previous studies showing no correlation between sNfL levels and SFN parameters [
38,
42].
Our study does not completely rule out the possibility that sNfL reflects the presence of ANP. However, if it does, its release in the context of peripheral SFN and ANP is likely overshadowed by the contribution of large fiber neuropathy. In clinical practice, isolated ANP is a rare phenomenon. In our cohort, the number of cases presenting with exclusively ANP was limited, as most had peripheral neuropathy as well. The incidence of isolated ANP in ATTRv amyloidosis is not well described in the literature. Data from the Transthyretin Amyloidosis Outcomes Survey showed that ANP was the first presenting symptom category in 37.3% of Val30Met (p.Val50Met) and 12.5% of non-Val30Met (non-p.Val50Met) patients [
43]. However, it was not reported whether these cases involved exclusively ANP. Therefore, the clinical relevance of a biomarker for the early detection of isolated ANP in ATTRv amyloidosis remains unclear. Previous studies in small patient groups have shown that sNfL can be useful in detecting neuropathy even in a presymptomatic stage [
44,
45].
To date, no other biomarkers have been identified for ANP in ATTRv amyloidosis. However, glial fibrillary acidic protein (GFAP), expressed in the central, peripheral, and enteric nervous system, may serve as a potential biomarker for the detection of ANP [
46]. A study by Plantone et al. found that serum GFAP levels were elevated in both asymptomatic
TTRv carriers and symptomatic ATTRv amyloidosis patients, reflecting early disease activity [
47]. As GFAP is expressed not only in the central nervous system astrocytes but also in non-myelinating Schwann cells and enteric glial cells [
46], its elevation may originate from subclinical peripheral or enteric nervous system involvement. Therefore, GFAP may represent a promising biomarker for peripheral SFN and ANP in ATTRv amyloidosis. However, until such data will become available, current diagnostic methods remain necessary for evaluating ANP.
4.1. Limitations
Clear limitations of this study are its retrospective design and the small number of TTRv carriers and ATTRv amyloidosis patients with isolated ANP that could be included. A longitudinal study of a large cohort of asymptomatic TTRv carriers who eventually develop symptoms of peripheral SFN/ANP is likely a more appropriate approach for evaluating the clinical utility of sNfL than logistic regression analysis. Furthermore, this study was limited to ANP. Although an assessment of SFN in relation to sNfL would also have been valuable, such analysis was not feasible due to insufficient data from QST. Furthermore, IENFD assessment via skin biopsy is not routinely performed at our center.
Lastly, ANP was defined based on the results of the Ewing battery, [123I]mIBG scintigraphy, and COMPASS-31 questionnaire. The Ewing battery and [123I]mIBG scintigraphy primarily assess cardiovascular ANP, whereas the COMPASS-31 provides a broader evaluation of autonomic function. Consequently, ANP definition A and B mainly reflect cardiovascular ANP. Including additional objective tests for non-cardiovascular autonomic function (e.g., Sudoscan, and quantitative sudomotor axon reflex test (QSART)) would have provided a more comprehensive assessment of ANP. Unfortunately, there were too few Sudoscan data and no QSART data available for analysis.
4.2. Further Considerations
Although sNfL has shown to be a useful biomarker for the early detection and monitoring of peripheral neuropathy in ATTRv amyloidosis [
15], this study and other studies suggest that sNfL is not sensitive enough to detect peripheral SFN and ANP [
38,
42]. Consequenly, ANP still needs to be assessed by tools such as the Ewing battery, [
123I]
mIBG scintigraphy, Sudoscan, and/or the COMPASS-31 questionnaire. Of these, [
123I]
mIBG scintigraphy is particularly promising for the early detection of cardiac ANP[
21]. It has shown to be more sensitive than bone scintigraphy, with the ability to detect cardiac sympathetic denervation prior to the emergence of structural heart abnormalities, clinical symptoms, or positive findings on other imaging modalities [
21,
24,
48,
49]. For assessing sudomotor function, the measurement of feet electrochemical skin conductance using the Sudoscan has shown to be a sensitive and non-invasive method for detecting early autonomic dysfunction in ATTRv amyloidosis patients [
39,
50].
For peripheral SFN, QST and skin biopsies to assess IENFD remain essential. Notably, IENFD has found to be positive in asymptomatic
TTRv carriers, which highlights its importance in the early detection of ATTRv amyloidosis [
38,
39].
The biomarker GFAP shows potential as non-invasive candidate for the early detection of not only peripheral polyneuropathy, but also ANP [
47]. However, further research is needed to clarify the precise role of GFAP in the early diagnosis of ANP and SFN.
5. Conclusion
Based on this study population, sNfL was not a useful early biomarker to detect ANP in pathogenic TTRv carriers and ATTRv amyloidosis patients. We observed that abnormal age-adjusted sNfL status was associated with the presence of ANP but it was not an independent predictor of age-adjusted sNfL status. Instead, peripheral neuropathy emerged as the primary contributor to the abnormal sNfL status observed in these individuals.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org. Table S1 Genotype distribution. Figure S1 Study inclusion overview. Figure S2 sNfL levels in patients with autonomic neuropathy compared to those without any form of neuropathy.
Author Contributions
Conceptualization, M.B., P.A.V.D.Z., B.P.C.H., H.L.A.N. and R.H.J.A.S.; Methodology, M.B., A.F.B., H.S.A.T., J.B., P.A.V.D.Z., B.P.C.H., F.L., G.D., H.L.A.N. and R.H.J.A.S.; Software, M.B.; Validation, H.L.A.N.; Formal Analysis, M.B.; Investigation, M.B.; Resources, J.B. and W.N.; Data Curation, M.B., H.S.A.T., F.L., G.D. and H.L.A.N.; Writing—Original Draft Preparation, M.B.; Writing—Review & Editing, A.F.B., H.S.A.T, J.B., C.E.T, P.A.V.D.Z., R.O.B.G., B.P.C.H., F.L., G.D., W.N., H.L.A.N. and, R.H.J.A.S.; Visualization, M.B.; Supervision, H.L.A.N. and R.H.J.A.S.; Project Administration, M.B.
Funding
This article concerns a non-funded study.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of interest statement
Milou Berends: reimbursement of travelling costs by Pfizer and Alnylam. Anne Floor Brunger: nothing to declare. Hendrea S.A. Tingen: reimbursement of travelling costs by Pfizer and Alnylam. Johan Bijzet: nothing to declare. Charlotte E. Teunissen: supported by the European Commission (Marie Curie International Training Network, grant agreement No 860197 (MIRIADE) and TAME, Innovative Medicines Initiatives 3TR (Horizon 2020, grant no 831434) EPND (IMI 2 Joint Undertaking (JU), grant No. 101034344) and JPND (bPRIDE, CCAD), European Partnership on Metrology, co-financed from the European Union’s Horizon Europe Research and Innovation Programme and by the Participating States (22HLT07 NEuroBioStand), CANTATE project funded by the Alzheimer Drug Discovery Foundation, Alzheimer Association, Michael J Fox Foundation, Health Holland, the Dutch Research Council (ZonMW), Alzheimer Drug Discovery Foundation, The Selfridges Group Foundation, Alzheimer Netherlands; Recipient of ABOARD, which is a public-private partnership receiving funding from ZonMW (#73305095007) and Health~Holland, Topsector Life Sciences & Health (PPP-allowance; #LSHM20106); Recipient of TAP-dementia, a ZonMw funded project (#10510032120003) in the context of the Dutch National Dementia Strategy; Research contracts with Acumen, ADx Neurosciences, AC-Immune, Alamar, Aribio, Axon Neurosciences, Beckman-Coulter, BioConnect, Bioorchestra, Brainstorm Therapeutics, Celgene, Cognition Therapeutics, EIP Pharma, Eisai, Eli Lilly, Fujirebio, Instant Nano Biosensors, Novo Nordisk, Olink, PeopleBio, Quanterix, Roche, Toyama, Vivoryon; Editor in chief of Alzheimer Research and Therapy, and serves on editorial boards of Alzheimer’s and Dementia, Molecular Neurodegeneration, Neurobiology of Aging, Neurology: Neuroimmunology & Neuroinflammation, Medidact Neurologie/Springer, and serves on committee to define guidelines for Cognitive disturbances, and one for acute Neurology in the Netherlands; Consultancy/speaker contracts for Aribio, Biogen, Beckman-Coulter, Cognition Therapeutics, Eli Lilly, Merck, Novo Nordisk, Olink, Roche and Veravas. Paul A. van der Zwaag: received consultancy fees from Pfizer and Alnylam. Reinold O.B. Gans: noting to declare. Bouke P.C. Hazenberg: speaker fee and reimbursement of travelling costs by Pfizer. Fiete Lange: nothing to declare. Gea Drost: nothing to declare. Walter Noordzij: received consultancy fees from Bayer and Sirtex. Hans L.A. Nienhuis: received consultancy fees from Pfizer, Alnylam and Novartis. Riemer H.J.A. Slart: received independent grant support of Pfizer and Siemens Healthineers and is associate editor in the European Journal of Nuclear Medicine and Molecular Imaging.
Acknowledgments
The authors have no acknowledgements to share.
References
- Benson MD, Kincaid JC. The molecular biology and clinical features of amyloid neuropathy. Muscle Nerve 2007, 36, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Sekijima, Y. Transthyretin (ATTR) amyloidosis: Clinical spectrum, molecular pathogenesis and disease-modifying treatments. J Neurol Neurosurg Psychiatry 2015, 86, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- Landrum MJ, Lee JM, Benson M, et al. ClinVar: Improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 2018, 46, D1062–D1067. [Google Scholar] [CrossRef]
- Samuelsson K, Jovanovic A, Egervall K, et al. Hereditary transthyretin amyloidosis in Sweden: Comparisons between a non-endemic and an endemic region. Amyloid 2022, 29, 220–227. [Google Scholar] [CrossRef]
- Ueda, M. Transthyretin: Its function and amyloid formation. Neurochem Int. 2022, 155, 1–7. [Google Scholar] [CrossRef]
- Conceição I, González-Duarte A, Obici L, et al. Red-flag symptom clusters in transthyretin familial amyloid polyneuropathy. J Peripher Nerv Syst. 2016, 21, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Loavenbruck AJ, Singer W, Mauermann ML, et al. Transthyretin amyloid neuropathy has earlier neural involvement but better prognosis than primary amyloid counterpart: an answer to the paradox? Ann Neurol. 2016, 80, 401–411. [Google Scholar] [CrossRef]
- Ando Y, Suhr OB. Autonomic dysfunction in familial amyloidotic polyneuropathy (FAP). Amyloid 1998, 5, 288–300. [Google Scholar] [CrossRef]
- Adams D, Koike H, Slama M, et al. Hereditary transthyretin amyloidosis: a model of medical progress for a fatal disease. Nat Rev Neurol. 2019, 15, 387–404. [Google Scholar] [CrossRef]
- Obici L, Kuks JB, Buades J, et al. Recommendations for presymptomatic genetic testing and management of individuals at risk for hereditary transthyretin amyloidosis. Recommendations for presymptomatic genetic testing and management of individuals at risk for hereditary transthyretin amyloidosis. Curr Opin Neurol. 2016, 29 (Suppl. S1), S27–S35. [Google Scholar]
- Adams D, Ando Y, Beirão JM, et al. Expert consensus recommendations to improve diagnosis of ATTR amyloidosis with polyneuropathy. J Neurol. 2021, 268, 2109–2122. [Google Scholar] [CrossRef]
- Adams D, Suhr OB, Hund E, et al. First european consensus for diagnosis, management, and treatment of transthyretin familial amyloid polyneuropathy. Curr Opin Neurol. 2016, 29, S14–S26. [Google Scholar] [CrossRef]
- Khalil M, Teunissen CE, Otto M, et al. Neurofilaments as biomarkers in neurological disorders. Nat Rev Neurol. 2018, 14, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Glatte P, Buchmann SJ, Hijazi MM, et al. Architecture of the Cutaneous Autonomic Nervous System. Front Neurol. 2019, 10, 970. [Google Scholar] [CrossRef] [PubMed]
- Berends M, Nienhuis HLA, Adams D, et al. Neurofilament Light Chains in Systemic Amyloidosis: A Systematic Review. Int J Mol Sci. 2024, 25, 3770. [Google Scholar] [CrossRef]
- Stranieri A, Abawajy J, Kelarev A, et al. An approach for Ewing test selection to support the clinical assessment of cardiac autonomic neuropathy. Artif Intell Med. 2013, 58, 185–193. [Google Scholar] [CrossRef]
- Ewing DJ, Clarke F. Diagnosis and management of diabetic autonomic neuropathy. Br Med J (Clin Res Ed) 1982, 285, 916–918. [Google Scholar] [CrossRef] [PubMed]
- Slart RHJA, Glaudemans AWJM, Hazenberg BPC, et al. Imaging cardiac innervation in amyloidosis. J Nucl Cardiol. 2019, 26, 174–187. [Google Scholar] [CrossRef]
- Gimelli A, Aimo A, Vergaro G, et al. Cardiac sympathetic denervation in wild-type transthyretin amyloidosis. Amyloid 2020, 27, 237–243. [Google Scholar] [CrossRef]
- Delahaye N, Dinanian S, Slama MS, et al. Cardiac sympathetic denervation in familial amyloid polyneuropathy assessed by iodine-123 metaiodobenzylguanidine scintigraphy and heart rate variability. Eur J Nucl Med. 1999, 26, 416–424. [Google Scholar] [CrossRef]
- Jonker DL, Hazenberg BPC, Nienhuis HLA, et al. Imaging cardiac innervation in hereditary transthyretin (ATTRm) amyloidosis: A marker for neuropathy or cardiomyopathy in case of heart failure? J Nucl Cardiol. 2020, 27, 1774–1784. [Google Scholar] [CrossRef]
- Tesfaye S, Boulton AJM, Dyck PJ, et al. Diabetic neuropathies: Update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care 2010, 33, 2285–2293. [Google Scholar] [CrossRef]
- Noordzij W, Glaudemans AWJM, Van Rheenen RWJ, et al. 123I-Labelled metaiodobenzylguanidine for the evaluation of cardiac sympathetic denervation in early stage amyloidosis. Eur J Nucl Med Mol Imaging 2012, 39, 1609–1617. [Google Scholar] [CrossRef]
- Tanaka M, Hongo M, Kinoshita O, et al. Iodine-123 Metaiodobenzylguanidine Scintigraphic Assessment of Myocardial Sympathetic Innervation in Patients with Familial Amyloid Polyneuropathy. J Am Coll Cardiol. 1997, 29, 168–174. [Google Scholar] [CrossRef]
- Mendes Sousa M, Cardoso I, Fernandes R, et al. Deposition of Transthyretin in Early Stages of Familial Amyloidotic Polyneuropathy Evidence for Toxicity of Nonfibrillar Aggregates. Am J Pathol. 2001, 159, 1993–2000. [Google Scholar] [CrossRef] [PubMed]
- Inoue Y, Abe Y, Kikuchi K, et al. Correction of collimator-dependent differences in the heart-to-mediastinum ratio in 123I-metaiodobenzylguanidine cardiac sympathetic imaging: Determination of conversion equations using point-source imaging. J Nucl Cardiol. 2017, 24, 1725–1736. [Google Scholar] [CrossRef]
- Flotats A, Carrió I, Agostini D, et al. Proposal for standardization of 123I-metaiodobenzylguanidine (MIBG) cardiac sympathetic imaging by the EANM Cardiovascular Committee and the European Council of Nuclear Cardiology. Eur J Nucl Med Mol Imaging 2010, 37, 1802–1812. [Google Scholar] [CrossRef]
- Nakajima K, Okuda K, Yoshimura M, et al. Multicenter cross-calibration of I-123 metaiodobenzylguanidine heart-to-mediastinum ratios to overcome camera-collimator variations. J Nucl Cardiol. 2014, 21, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Sletten DM, Suarez GA, Low PA, et al. COMPASS 31: A refined and abbreviated composite autonomic symptom score. Mayo Clin Proc. 2012, 87, 1196–1201. [Google Scholar] [CrossRef]
- Dyck PJ, Dyck; P J B, Kennedy; W R, et al. Limitations of quantitative sensory testing when patients are biased toward a bad outcome. Neurology 1998, 50, 1213. [Google Scholar] [CrossRef] [PubMed]
- PJ Dyck, JL Davies, WJ Litchy, et al. Longitudinal assessment of diabetic polyneuropathy using a composite score in the Rochester Diabetic Neuropathy Study cohort. Neurology 1997, 49, 229–239. [Google Scholar] [CrossRef]
- Ando Y, Coelho T, Berk JL, et al. Guideline of transthyretin-related hereditary amyloidosis for clinicians. Orphanet J Rare Dis. 2013, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Van Lierop ZYGJ, Verberk IMW, Van Uffelen KWJ, et al. Pre-analytical stability of serum biomarkers for neurological disease: Neurofilament-light, glial fibrillary acidic protein and contactin-1. Clin Chem Lab Med. 2022, 60, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Vermunt L, Otte M, Verberk IMW, et al. Age- and disease-specific reference values for neurofilament light presented in an online interactive support interface. Ann Clin Transl Neurol. 2022, 9, 1832–1837. [Google Scholar] [CrossRef]
- Luigetti M, Di Paolantonio A, Guglielmino V, et al. Neurofilament light chain as a disease severity biomarker in ATTRv: data from a single-centre experience. Neurol Sci. 2022, 43, 2845–2848. [Google Scholar] [CrossRef]
- Russo M, De Luca M, Gentile L, et al. NfL as a biomarker in ATTRv amyloidosis: potential and limitations. Neurol Sci. 2025, 46, 3263–3269. [Google Scholar] [CrossRef]
- Fridman V, Sillau S, Ritchie A, et al. Plasma neurofilament light chain concentrations are elevated in youth-onset type 2 diabetes and associated with neuropathy. J Peripher Nerv Syst. 2023, 28, 460–470. [Google Scholar] [CrossRef]
- Galosi E, Costanzo R, Forcina F, et al. Serum neurofilament light chain levels correlate with small fiber related parameters in patients with hereditary transthyretin amyloidosis with polyneuropathy (ATTRv-PN). Neurol Sci. 2024, 45, 5023–5032. [Google Scholar] [CrossRef] [PubMed]
- Leonardi L, Adam C, Beaudonnet G, et al. Skin amyloid deposits and nerve fiber loss as markers of neuropathy onset and progression in hereditary transthyretin amyloidosis. Eur J Neurol. 2022, 29, 1477–1487. [Google Scholar] [CrossRef]
- Ebenezer GJ, Liu Y, Judge DP, et al. Cutaneous nerve biomarkers in transthyretin familial amyloid polyneuropathy. Ann Neurol. 2017, 82, 44–56. [Google Scholar] [CrossRef]
- Chao CC, Hsueh HW, Kan HW, et al. Skin nerve pathology: Biomarkers of premanifest and manifest amyloid neuropathy. Ann Neurol. 2019, 85, 560–573. [Google Scholar] [CrossRef]
- Baka P, Steenken L, Escolano-Lozano F, et al. Studying serum neurofilament light chain levels as a potential new biomarker for small fiber neuropathy. Eur J Neurol. 2024, 31, e16192. [Google Scholar] [CrossRef]
- Coelho T, Conceição I, Waddington-Cruz M, et al. A natural history analysis of asymptomatic TTR gene carriers as they develop symptomatic transthyretin amyloidosis in the Transthyretin Amyloidosis Outcomes Survey (THAOS). Amyloid 2022, 29, 228–236. [Google Scholar] [CrossRef]
- Berends M, Brunger AF, Bijzet J, et al. Longitudinal analysis of serum neurofilament light chain levels as marker for neuronal damage in hereditary transthyretin amyloidosis. Amyloid 2024, 31, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Carroll AS, Razvi Y, O’Donnell L, et al. Serum neurofilament light chain in hereditary transthyretin amyloidosis: validation in real-life practice. Amyloid 2024, 31, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Yang Z, Wang KKW. Glial fibrillary acidic protein: From intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci. 2015, 38, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Plantone D, Luigetti M, Manco C, et al. Elevated serum concentrations of GFAP in hereditary transthyretin amyloidosis since pre-symptomatic stages. J Neurol. 2025, 272, 340. [Google Scholar] [CrossRef]
- Koike H, Nakamura T, Nishi R, et al. Cardiac and peripheral vasomotor autonomic functions in hereditary transthyretin amyloidosis with non-Val30Met mutation. Amyloid 2019, 26, 13–14. [Google Scholar] [CrossRef]
- Coutinho MCA, Cortez-Dias N, Cantinho G, et al. Reduced myocardial 123-iodine metaiodobenzylguanidine uptake: A prognostic marker in familial amyloid polyneuropathy. Circ Cardiovasc Imaging 2013, 6, 627–636. [Google Scholar] [CrossRef]
- Castro J, Miranda B, Castro I, et al. The diagnostic accuracy of sudoscan in transthyretin familial amyloid polyneuropathy. Clin Neurophysiol. 2016, 127, 2222–2227. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).