Submitted:
12 November 2025
Posted:
13 November 2025
You are already at the latest version
Abstract
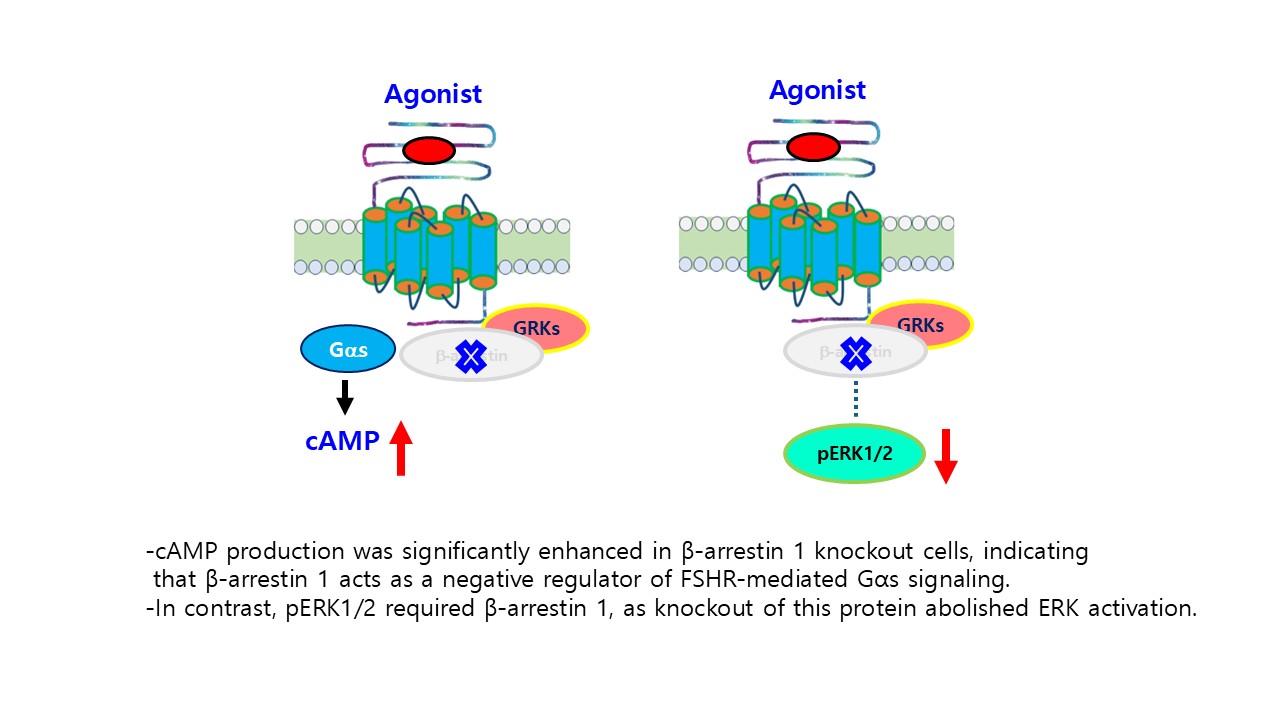
Gonadotropins, including luteinizing hormone (LH) and follicle-stimulating hormone (FSH), are essential regulators of reproductive function; however, the molecular characteristics and signaling properties of their recombinant forms in nonhuman primates remain incompletely defined. In this study, we performed a comparative sequence and functional analysis of the gonadotropin subunit cDNAs from Cynomolgus (Macaca fascicularis) and Rhesus monkeys (Macaca mulatta) and produced recombinant single-chain LHβ/α and FSHβ/α proteins for biochemical and signaling characterization. Sequence analysis revealed complete conservation of the α- and FSHβ subunit cDNAs between the two monkey species, whereas minor synonymous nucleotide variations were observed in the LHβ subunit. Recombinant FSHβ/α was expressed in CHO-K1 and CHO-S cells using dual epitope tagging, allowing for efficient detection and purification. Western blotting and enzymatic deglycosylation confirmed that both recombinant hormones were glycosylated, with N-linked glycosylation being the predominant modification. Functionally, recombinant monkey FSHβ/α stimulated dose-dependent cAMP accumulation in HEK293 cells expressing the human FSH receptor (hFSHR), demonstrating its full biological activity. cAMP production was significantly enhanced in β-arrestin 1 knockout cells, indicating that β-arrestin 1 acts as a negative regulator of FSH receptor (FSHR)-mediated Gαs signaling. In contrast, phosphorylated ERK1/2 (pERK1/2) requires β-arrestin 1, as knockout of this protein abolishes ERK activation. Pharmacological inhibition experiments further revealed that FSH-induced pERK1/2 activation largely depended on the cAMP/PKA pathway, whereas PKC inhibition had a minimal effect. Comparative analyses of cells expressing hFSHR or rat FSHR revealed distinct temporal patterns of pERK1/2 activation, reflecting species-specific signaling kinetics. Together, these results establish a robust system for the production of biologically active recombinant monkey gonadotropins and delineate the dual roles of β-arrestin 1 in modulating FSHR-mediated cAMP and ERK signaling pathways. This study provides new insights into the structure–function relationships of primate gonadotropins and β-arrestin–dependent GPCR regulation.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Total RNA Isolation and cDNA synthesis
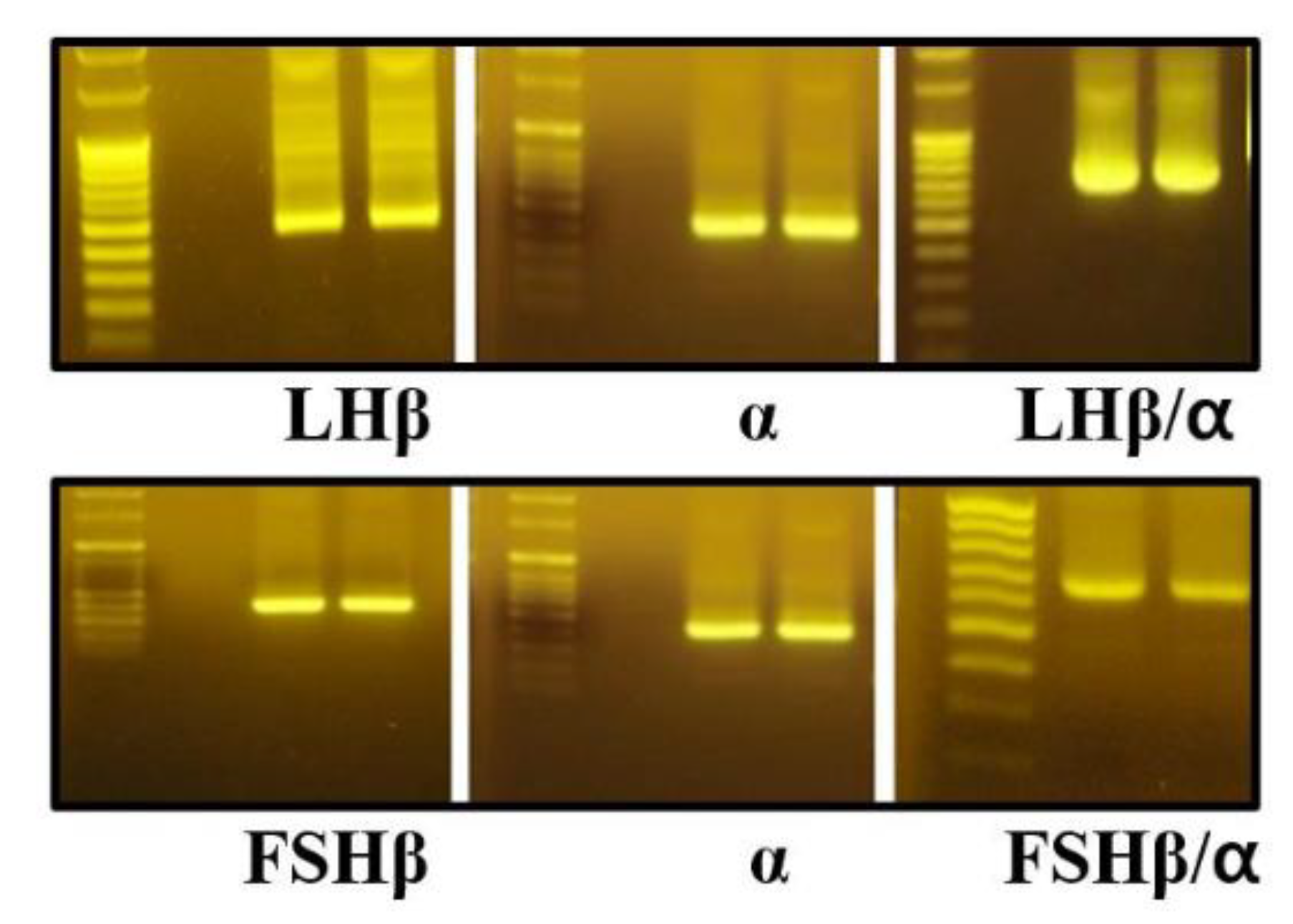
2.3. PCR Amplification of Monkey α-, LHβ- and FSHβ-Subunit cDNAs
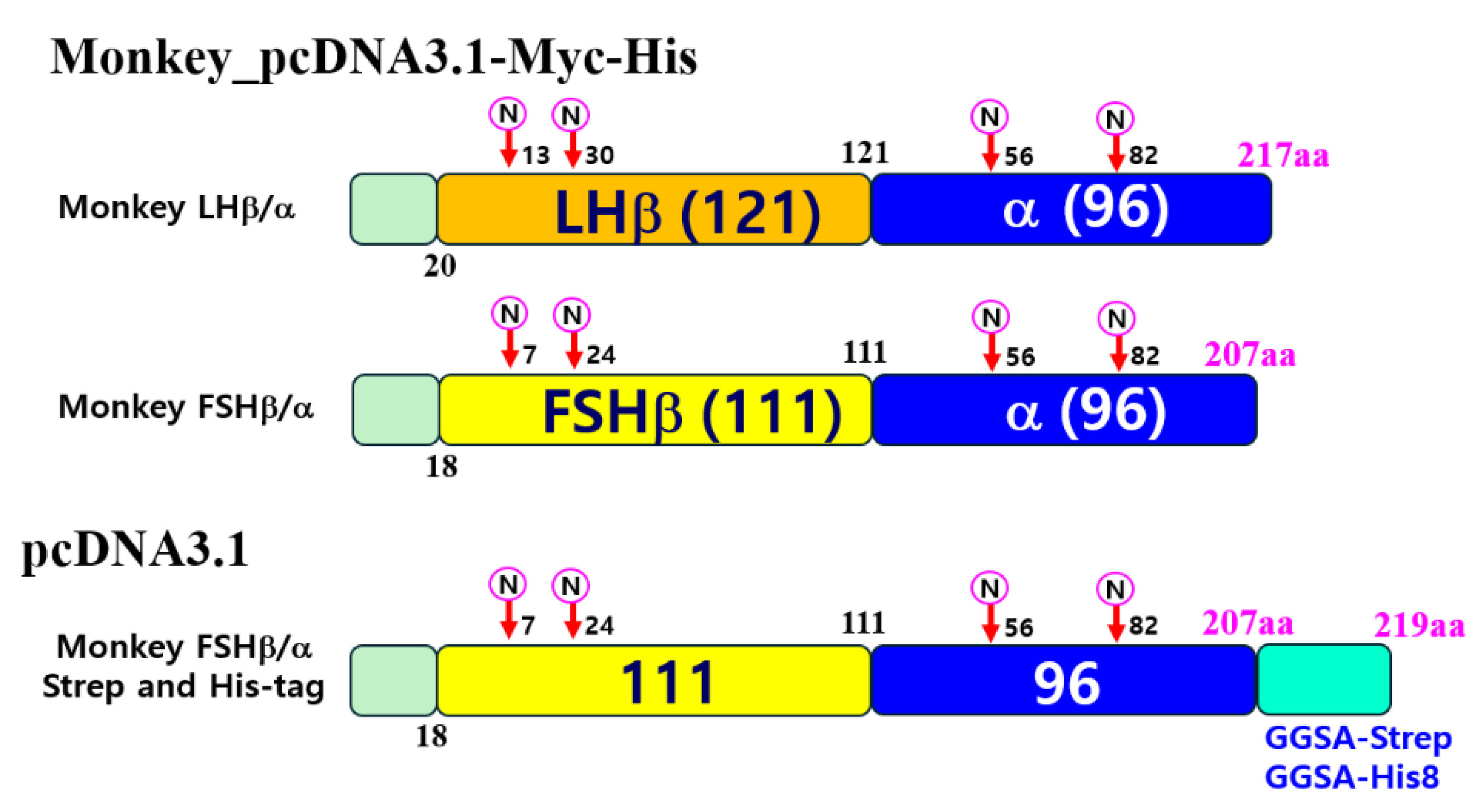
2.4. Construction of Single-Chain Monkey LHβ/α and FSHβ/α
2.5. Construction of Mammalian Expressing Vectors
2.6. Production of Rec-Monkey LHβ/α and FSH β/α into CHO-K1 and CHO Suspension Cells
2.7. Purification of Strep- and His-Tagged Rec-Monkey FSH Protein
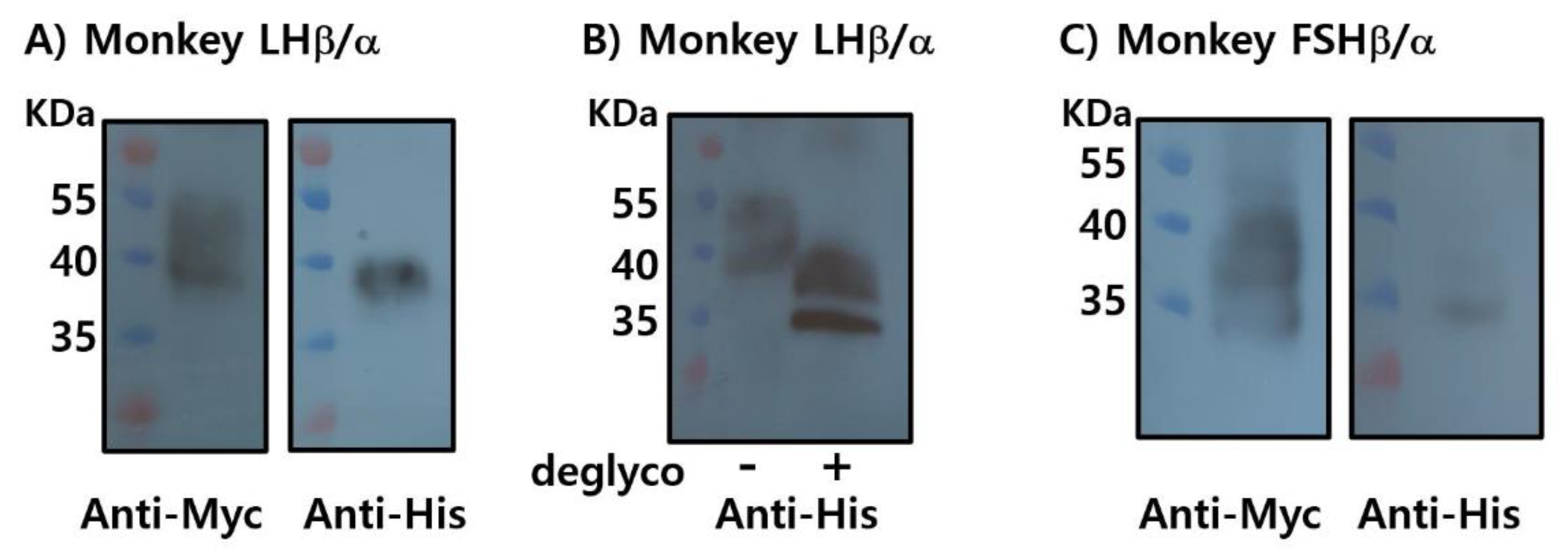
2.8. Western Blotting Analysis of Rec-Monkey LHβ/α and FSHβ/α
2.9. Enzymatic Deglycosylation of Rec-Monkey FSHβ/α
2.10. Transient Transfection with hLH/CGR, hFSHR, and rLH/CGR, and rFSHR cDNA Plasmids
2.11. Measurement of cAMP Accumulation in CHO-K1 Cells Expressing Gonadotropin Receptors
2.12. Construction of Beta-Arrestin 1 Knockout Cell Lines
2.13. Phospho-ERK1/2 Analysis
2.14. Data Analysis
3. Results
3.1. Comparative Sequence Analysis of Gonadotropin Subunit cDNAs Between Cynomolgus and Rhesus Monkeys
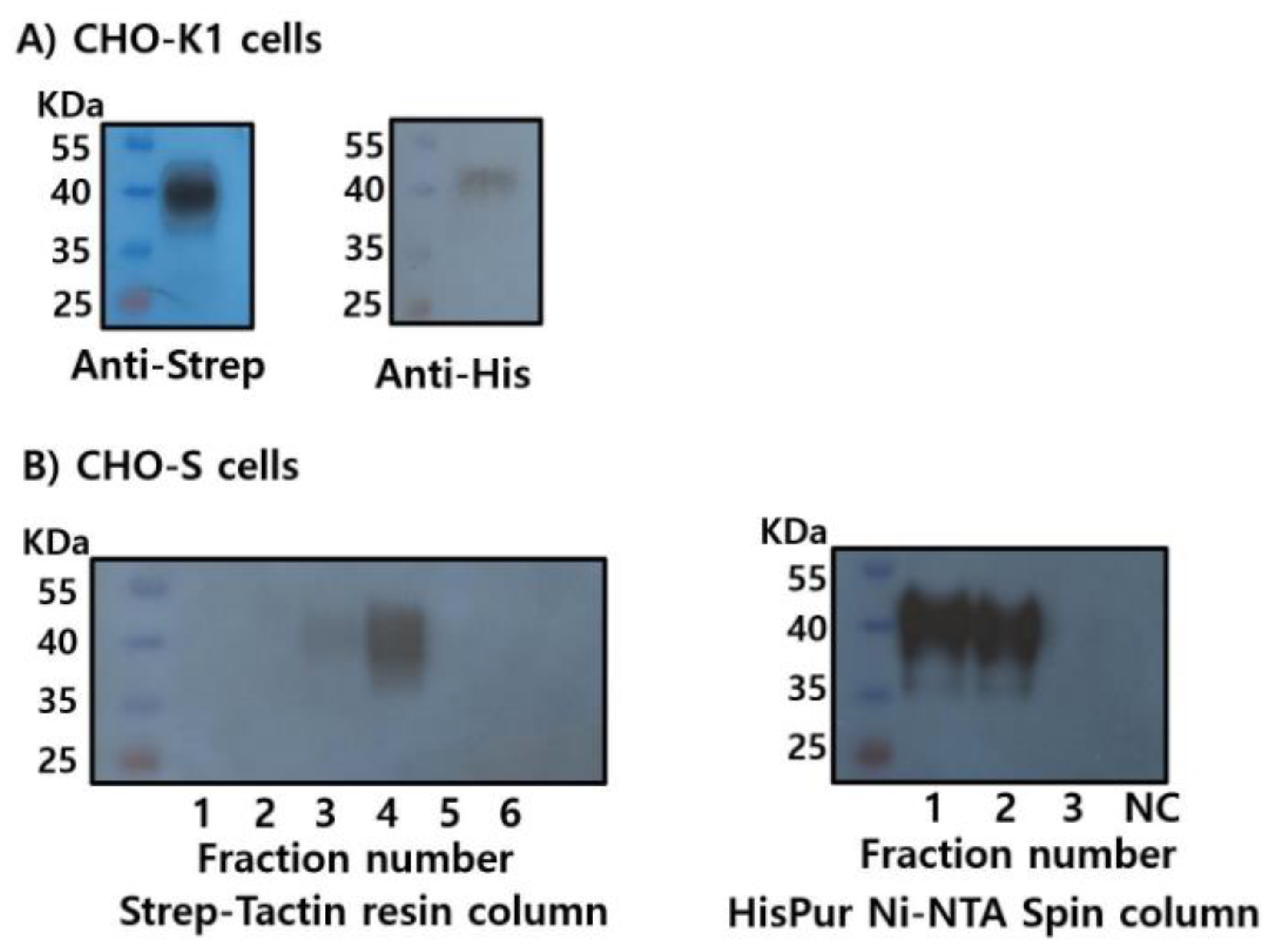
3.2. Production of Recombinant LHβ/α and FSHβ/α Proteins in CHO-K1 Cells
3.3. Optimization of Recombinant FSHβ/α Production and Purification Using Dual Epitope Tagging in CHO-K1 and CHO Suspension Cells
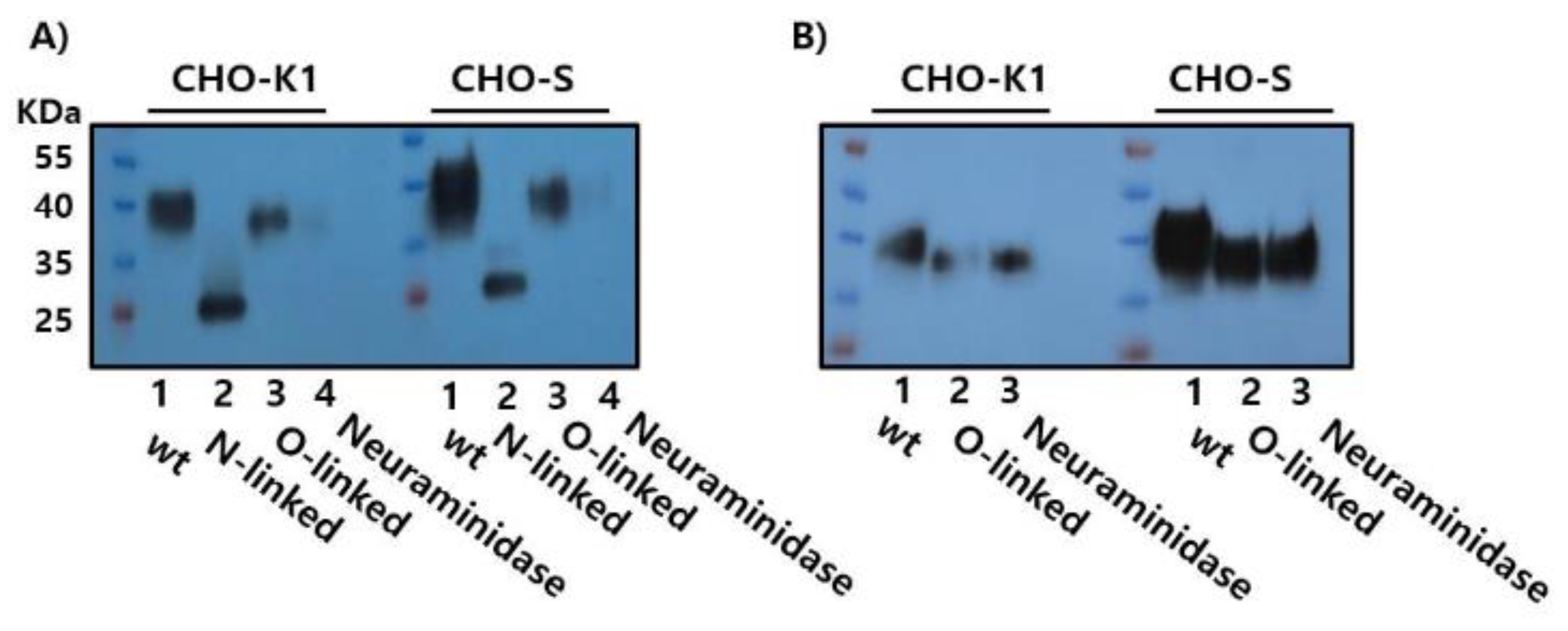
3.4. Enzymatic Deglycosylation Reveals N-Linked Glycosylation as the Predominant Modification in Recombinant Monkey FSHβ/α
3.5. Quantification of Recombinant Monkey LHβ/α and FSHβ/α Using Commercial ELISA Kits
3.6. cAMP Analysis
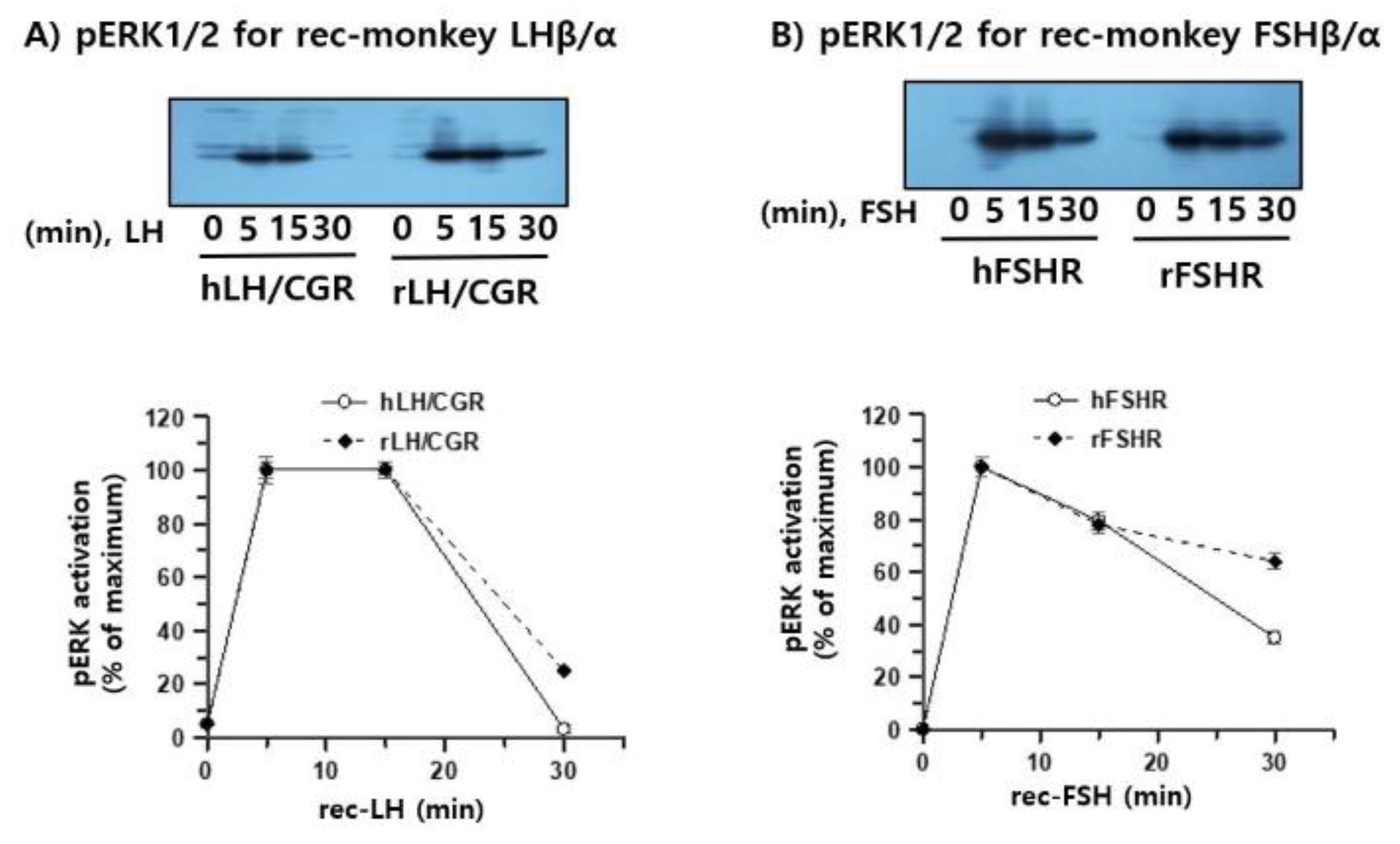
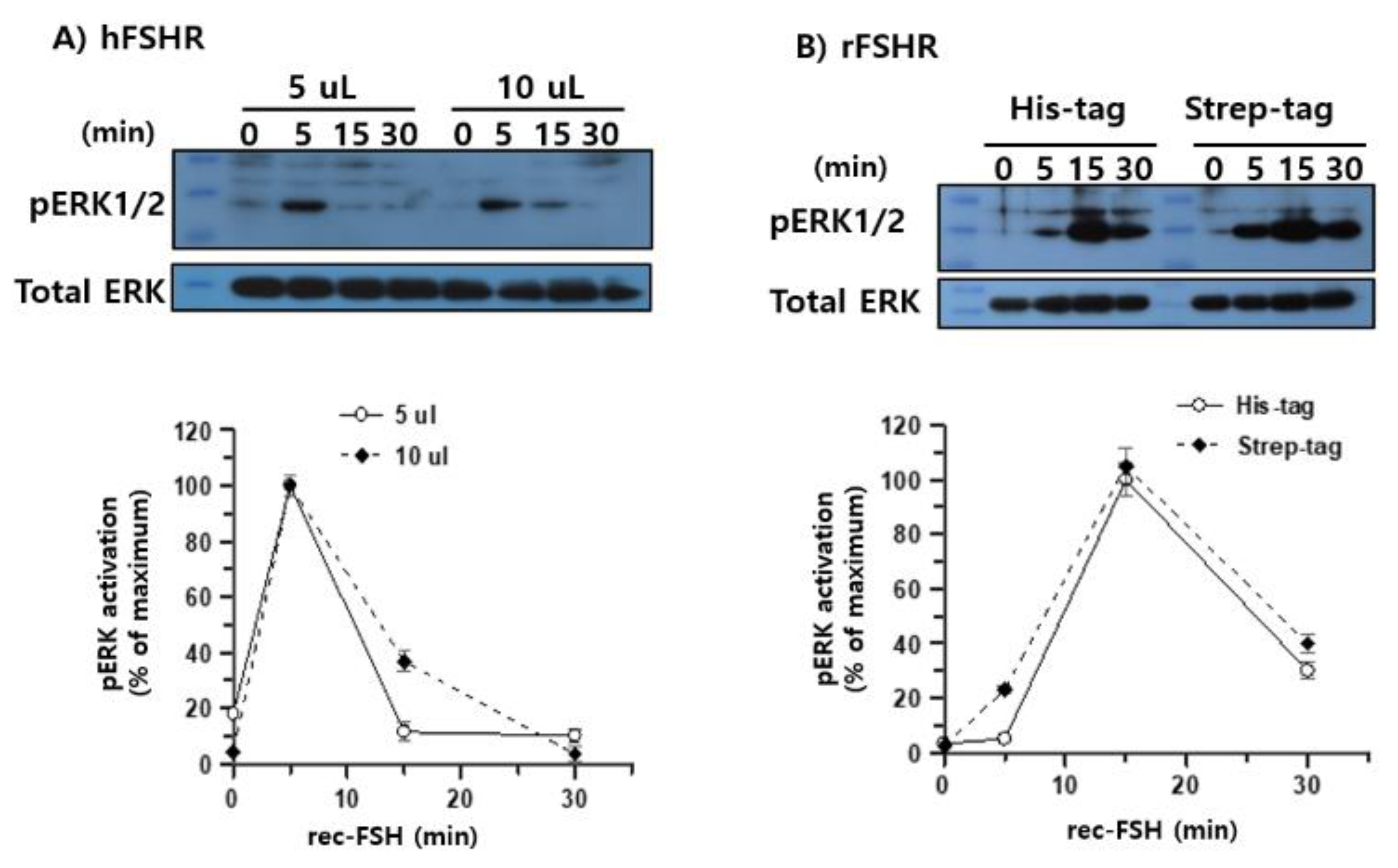
pERK1/2 Activation Through rLH/CGR, hLH/CGR, rFSHR, and hFSHR
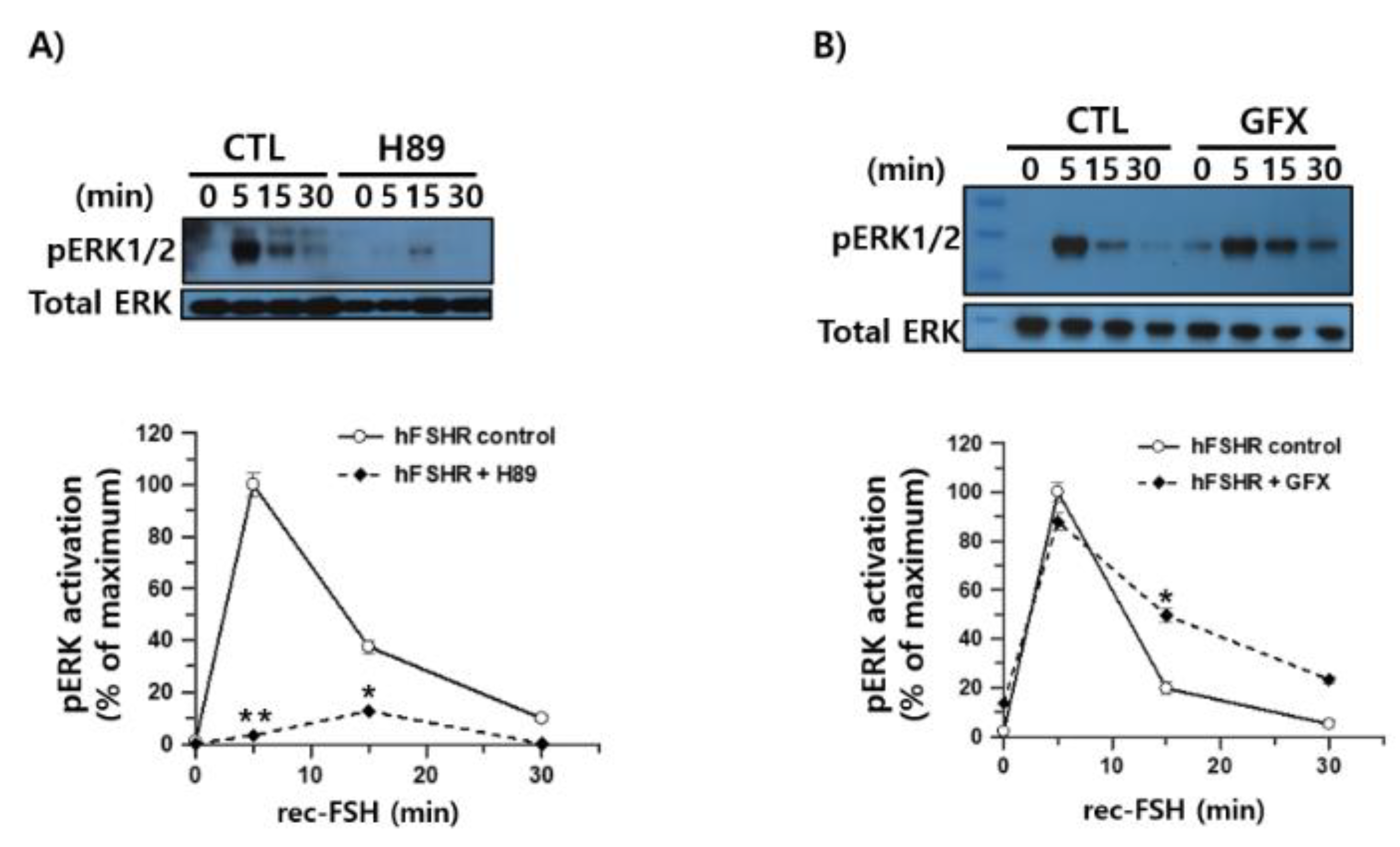
3.8. Inhibition of PKA and PKC Reveals Distinct Patterns of Rec-Monkey FSH-Induced pERK1/2 Activation
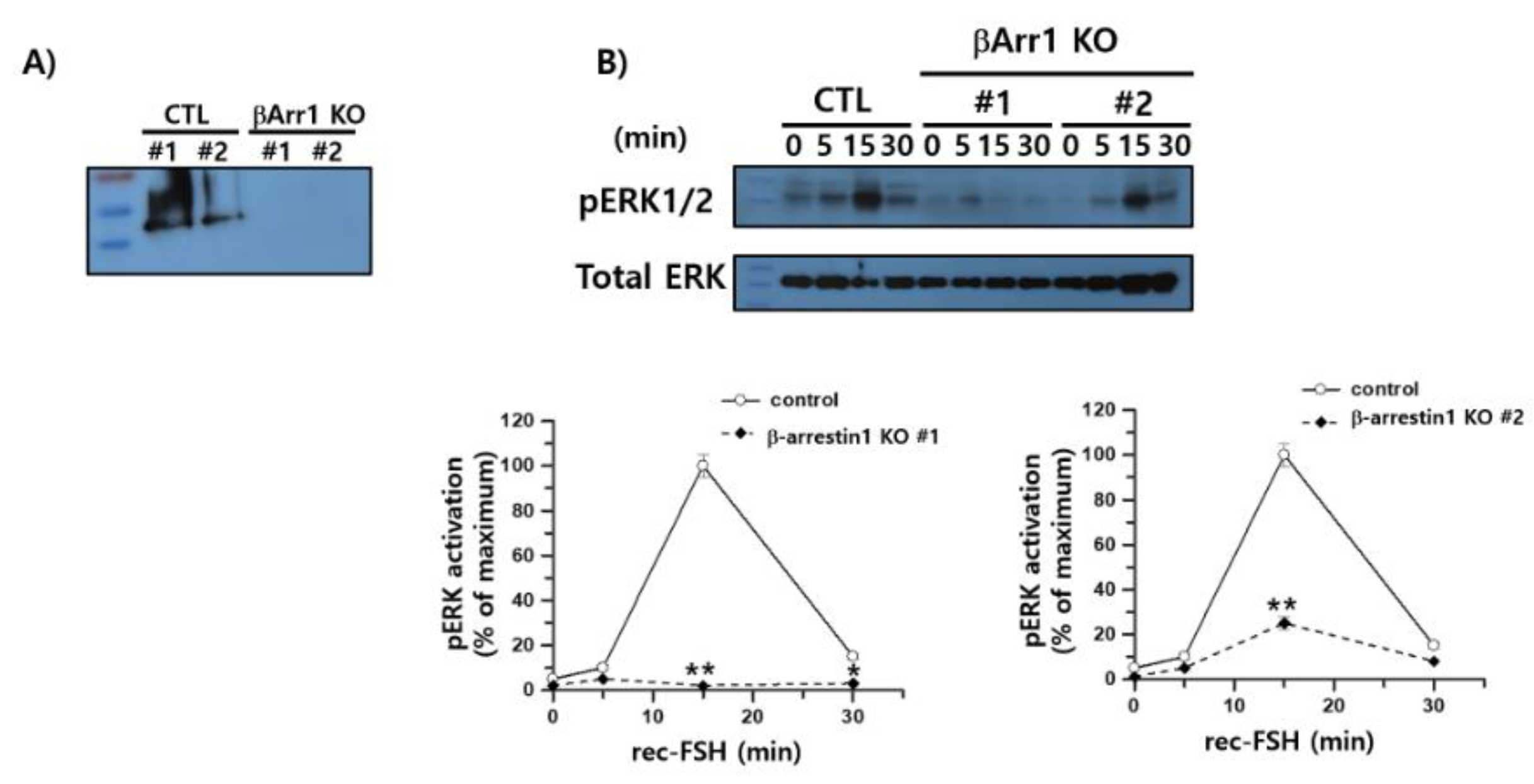
3.9. Inhibition of pERK1/2 Activation in the β-arrestin 1 Knockout Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Min, K.S.; Hiyama, T.; Seong, H.H.; Hattori, N.; Tanaka, S.; Shiota, K. Biological activities of tethered equine chorionic gonadotropin (eCG) and its deglycosylated mutants. J. Reprod. Dev. 2004, 50, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Valove, F.M.; Finch, C.; Anasti, J.N.; Froehlich, J.; Flack, M.R. Receptor binding and signal transduction are dissociable functions requires different sites on follicle-stimulating hormone. Endocrinology 1994, 135, 2657–2661. [Google Scholar] [CrossRef]
- Flack, M.R.; Froehlich, J.; Bennet, A.P.; Anasti, J.; Nisula, B.C. Site-directed mutagenesis defines the individual roles of the glycosylation sites on follicle-stimulating hormone. J. Biol. Chem. 1994, 269, 14015–14020. [Google Scholar] [CrossRef]
- Sugahara, T.; Sato, A.; Kudo, M.; Ben-Menahem, D.; Pixley, M.R.; Hsueh, A.J.W.; Boime, I. Expression of biological active fusion genes encoding the common subunit and the follicle-stimulating hormone β subunits. J. Biol. Chem. 1996, 271, 10445–10448. [Google Scholar] [CrossRef]
- Simoni, M.; Gromoll, J.; Nieschlag, E. The follicle-stimulating hormone receptor : biochemistry, molecular biology, physiology, and pathophysiology. Endocr. Rev. 1997, 18, 739–773. [Google Scholar]
- Hunzicker-Dunn, M.; Maizels, E.T. FSH signaling pathways in immature granulosa cells that regulate target gene expression branching out from protein kinase A. Cell Signal. 2006, 18, 1351–1359. [Google Scholar] [CrossRef]
- Duan, J.; Xu, P.; Cheng, X.; Mao, C.; Croll, T.; He, X.; Shi, J.; Luan, X.; Yin, W.; You, E.; et al. Structures of full-length glycoprotein hormone receptor signalling complexes. Nature 2021, 598, 688–692. [Google Scholar] [CrossRef]
- Duan, J.; Xu, P.; Zhang, H.; Luan, X.; Yang, J.; He, X.; Mao, C.; Shen, D.D.; Ji, Y.; Cheng, X.; et al. Mechanism of hormone and allosteric agonist mediated activation of follicle stimulating hormone receptor. Nat. Commun. 2023, 14, 519. [Google Scholar] [CrossRef]
- Lefkowitz, R.J.; Shenoy, S.K. Transduction of receptor signals by β-arrestins. Science 2005, 308, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Lefkowitz, R.J.; Rockman, H.; Shim, P.J.; Liu, S.; Ahn, S.; Pani, B.; Rajagopai, S.; Shenoy, S.K.; Bouvier, M.; Benovic, J.L.; et al. How carvedilol does not activate β2-adrenoreptors. Nat. Commun. 2023, 14, 7866. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Byambaragchaa, M.; Park, S.H.; Park, M.H.; Kang, M.H.; Min, K.S. The N-linked glycosylation Asn191 and Asn199 sites are controlled differently between PKA signal transduction and pERK1/2 activity in equine follicle-stimulating hormone receptor. Curr. Issues Mol. Biol. 2025, 47, 168. [Google Scholar] [CrossRef]
- Jean-Charles, P.Y.; Kaur, S.; Shenoy, S.K. GPCR signaling via b-arrestin-dependent mechanisms. J. Cardiovasc. Pharmacol. 2017, 70, 142–158. [Google Scholar] [CrossRef]
- Ulloa-Aguirre, A.; Reiter, E.; Crepieux, P. FSH receptor signaling: Complexity of interactions and signal diversity. Endocrinology 2018, 159, 3020–3035. [Google Scholar] [CrossRef]
- Reiter, E.; Lefkowitz, R.J. GRKs and β-arrestins: Roles in receptor silencing, trafficking and signaling. Trends Endocrinol. Metab. 2006, 17, 159–165. [Google Scholar] [CrossRef]
- Kara, E.; Crepieux, P.; Gauthier, C.; Martinat, N.; Piketty, V.; Guillou, F.; Reiter, E. A phosphorylation cluster of five serine and threonine residues in the C-terminus of the follicle-stimulating hormone receptor is important for desensitization but not for β-arrestin-mediated ERK activation. Mol. Endocrinol. 2006, 20, 3014–3026. [Google Scholar] [CrossRef]
- Reiter, E.; Ayoub, M.A.; Pellissier, L.P.; Landomiel, F.; Musnier, A.; Trefier, A.; et al. β-arrestin signalling and bias in hormone-responsive GPCRs. Mol. Cell Endocrinol. 2017, 449, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Ladomiel, F.; De Pascali, F.; Raynaud, P.; Jean-Alphonse, F.; Yvinec, R.; Pellissier, L.P.; Bozon, V.; Bruneau, G.; Crepieux, P.; Poupon, A.; et al. Front. Endocrinol. 2019, 10, 148.
- Brughi, V.; Paradis, J.S.; Officer, A.; Adame-Garcia, S.R.; Wu, X.; Matthees, E.S.F.; Barsi-Rhyne, B.; Ramms, D.J.; Clubb, L.; Acosta, M.; Tamayo, P.; Bouvier, M.; Inoue, A.; von Zastrow, M.; Hoffmann, C.; Gutkind, J.S. Gαs is dispensable for β-arrestin coupling but dictates GRK selectivity and is predominant for gene expression regulation by β2-adrenergic receptor. J. Biol. Chem. 2023, 299, 105293. [Google Scholar]
- Saneyoshi, T.; Min, K.S.; Ma, J.X.; Nambo, Y.; Hiyama, T.; Tanaka, S.; Shiota, K. Equine follicle-stimulating hormone: molecular cloning of beta subunit and biological role of the asparagine-linked oligosaccharide at asparagine56 of alpha subunit. Biol. Reprod. 2001, 65, 1686–1690. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Munkhuu, O.; Byambaragchaa, M.; Lee, B.I.; Kim, S.K.; Kang, M.H.; Kim, D.J.; Min, K.S. Site-specific roles of N-linked oligosaccharides in recombinant eel follicle-stimulating hormone for secretion and signal transduction. Gen. Comp. Endocrinol. 2019, 276, 37–44. [Google Scholar] [CrossRef]
- Lee, S.Y.; Byambaragchaa, M.; Choi, S.H.; Kang, H.J.; Kang, M.J.; Min, K.S. Roles of N-linked and O-linked glycosylation sites in the activity of equine chorionic gonadotropin in cells expressing rat luteinizing hormone/chorionic gonadotropin receptor and follicle-stimulating hormone receptor. BMC Biotechnol. 2021, 21, 52. [Google Scholar] [CrossRef]
- Schmidt, A.; Gromoll, J.; Weinbauer, G.F.; Galla, H.J.; Chappel, S.; Simoni, M. Cloning and expression of cynomolgus monkey (Macaca fascicularis) gonadotropins luteinizing hormone and follicle-stimulating hormone and identification of two polymorphic sites in the luteinizing hormone β subunit. Mole. Cell. Endocrinol. 1999, 156, 73–83. [Google Scholar] [CrossRef]
- Byambaragchaa, M.; Park, S.H.; Kim, S.G.; Shin, M.G.; Kim, S.K.; Park, M.H.; Kang, M.H.; Min, K.S. Stable production of a recombinant single-chain eel follicle-stimulating hormone analog in CHO DG44 cells. Int. J. Mol. Sci. 2024, 25, 7272. [Google Scholar] [CrossRef] [PubMed]
- Bishop, L.A.; Robertson, D.M.; Cahir, N.; Schofield, P.R. Specific roles for the asparagine-linked carbohydrate residues of recombinant human follicle stimulating hormone in receptor binding and signal transduction. Mol. Endocrinol 1994, 8, 722–731. [Google Scholar] [PubMed]
- Sugahara, T.; Grootenhuis, P.D.J.; Sato, A.; Kudo, M.; Ben-Menahem, D.; Pixley, M.R.; Hsueh, A.J.W.; Boime, I. Expression of biological active fusion genes encoding the common α subunit and either the CGβ or FSHβ subunits: role of a linker sequence. Mol. Cell. Endocrinol. 1996, 125, 71–77. [Google Scholar] [CrossRef]
- Byambaragchaa, M.; Kim, D.J.; Kang, M.H.; Min, K.S. Site specificity of eel luteinizing hormone N-linked oligosaccharides in signal transduction. Gen. Comp. Endocrinol. 2018, 268, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Byambaragchaa, M.; Park, S.H.; Park, M.H.; Kang, M.H.; Min, K.S. Enhanced production and functional characterization of recombinant equine chorionic gonadotropin (rec-eCG) in CHO-DG44 cells. Biomolecules 2025, 15, 289. [Google Scholar] [CrossRef]
- Losle, M.; Lin, C.W.; Beil-Wagner, J.; Aebi, M.; Buch, T. Comparison of pregnant mare serum gonadotropin products with surprising differences in protein content. Sci. Rep. 2025, 15, 6824. [Google Scholar] [CrossRef]
- Kohout, T.A.; Lin, F.T.; Perry, S.J.; Conner, D.A.; Lefkowitz, R.J. β-arrestin 1 and 2 differentially regulate heptahelical receptor signaling and trafficking. J. Biol. Chem. 2001, 98, 1601–1606. [Google Scholar] [CrossRef]
- Shenoy, S.K.; Barak, L.S.; Xiao, K.; Ahn, S.; Berthouze, M.; Shukla, A.K.; Luttrell, L.M.; Lefkowitz, R.J. Ubiquitination of β-arrestin links seven-transmembrane receptor endocytosis and ERK activation. J. Biol. Chem. 2007, 282, 29549–29562. [Google Scholar] [CrossRef]
- Piketty, V.; Kara, E.; Guillou, F.; Reiter, E.; Crepieux, P. Follicle-stimulating hormone (FSH) activates extracellular signal-regulated kinase phosphorylation independently of beta-arrestin- and dynamin-mediated FSH receptor internalization. Reprod. Biol. Endocrinol. 2006, 4, 33. [Google Scholar] [CrossRef]
- Ahn, S.; Shenoy, S.K.; Wei, H.; Lefkowitz, R.J. Differential kinetic and spatial patterns of β-arrestin and G protein-mediated ERK activation by the angiotensin II receptor. J. Biol. Chem. 2004, 279, 35518–35525. [Google Scholar] [CrossRef]
- Gesty-Palmer, D.; Chen, M.; Reiter, E.; Ahn, S.; Nelson, C.D.; Wang, S.; Eckhardt, A.E.; Cowan, C.L.; Spurney, R.F.; Luttrell, L.M.; et al. Distinct beta-arrestin-and G protein-dependent pathways for parathyroid hormone receptor-stimulated ERK1/2 activation. J. Biol. Chem. 2006, 281, 10856–10864. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Ahn, S.; Ren, X.R.; Whalen, E.J.; Reiter, E.; Wei, H.; Lefkowitz, R.J. Functional antagonism of different G protein-coupled receptor kinases for β-arrestin-mediated angiotensin II receptor signaling. Proc. Natl. Acad. Sci. USA 2005, 102, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, S.K.; Draka, M.T.; Nelson, C.D.; Houtz, D.A.; Xiao, K.; Madabushi, S.; Reiter, E.; Premont, R.T.; Lichtarge, O.; Lefkowitz, R.J. β-arrestin-dependent, G protein-independent ERK1/2 activation by the β2 adrenergic receptor. J. Biol. Chem. 2006, 281, 1261–1273. [Google Scholar] [CrossRef] [PubMed]
- Luttrell, L.M.; Wang, J.; Plouffe, B.; Smith, J.S.; Yamani, L.; Kaur, S.; Jean-Charles, P.Y.; Gauthier, C.; Lee, M.H.; Pani, B.; et al. Manifold roles of β-arrestin in GPCR signaling elucidated with siRNA and CRISPR/Cas9. Sci. Signal. 2018, 11, 549. [Google Scholar] [CrossRef]
- O’Hayre, M.; Eichel, K.; Avino, S.; Zhao, X.; Steffen, D.J.; Feng, X.; Kawakami, K.; Aoki, J.; Messer, K.; Sunahara, R.; Inoue, A.; von Zastrow, M.; Gutkind, J.S. Genetic evidence that β-arrestins are dispensable for the initiation of β2-adrenergic receptor signaling to ERK. Sci. Signal. 2018, 10, 484. [Google Scholar]
- Drube, J.; Haider, R.S.; Matthees, E.S.F.; Reichel, M.; Zeiner, J.; Fritzwanker, S.; Ziegler, C.; Barz, S.; Klement, L.; Filor, J.; et al. GPCR kinase knockout cells reveal the impact of individual GRKs on arrestin binding and GPCR regulation. Nat. Commun. 2022, 13, 540. [Google Scholar] [CrossRef]
- Moller, T.C.; Pedersen, M.F.; van Senten, J.R.; Seiersen, S.D.; Mathiesen, J.M.; Bouvier, M.; Brauner-Osborne, H. Dissecting the roles of GRK2 and GRK3 in mu-opioid receptor internalization and β-arrestin2 recruitment using CRISPR/Cas9-edited HEK293 cells. Sci. Rep. 2020, 10, 173955. [Google Scholar] [CrossRef]
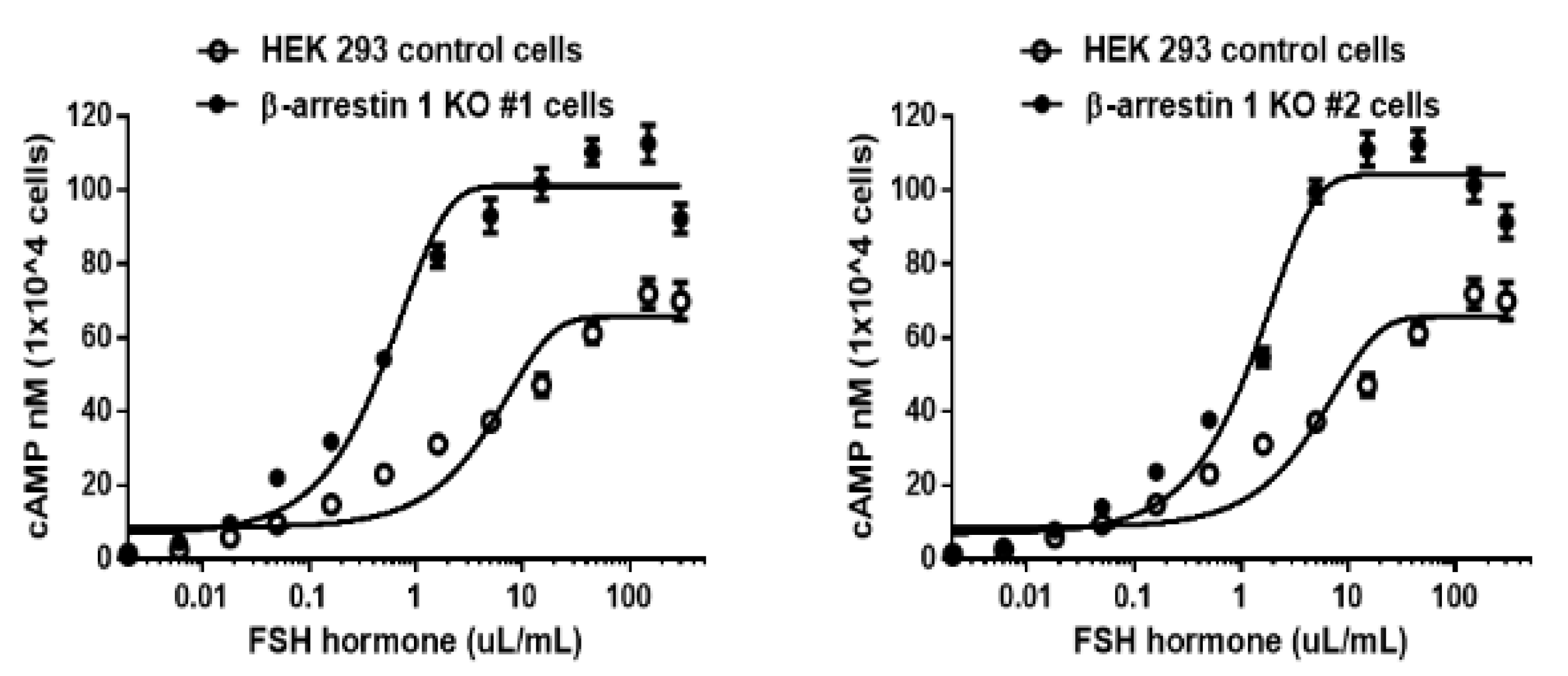
| 1. Cell lines | 2. cAMP Responses | ||
|
Basal a (nM/10⁴Cells) |
EC₅₀ (µl/mL) |
Rmaxb (nM/10⁴Cells) |
|
| HEK 293 | 8.6 ± 1.6 | 4.9 (1.0-fold) (3.5 to 8.0)c |
66.7 ± 2.4 (1.0-fold) |
| β-arrestin 1 KO 1 | 6.8 ± 1.6 | 0.5 (9.8-fold) (0.4 to 0.6) |
101.9 ± 1.7 (1.52-fold) |
| β-arrestin 1 KO 2 | 6.9 ± 1.7 | 1.2 (4.1-fold) (1.1 to 1.6) |
104.2 ± 1.9 (1.56-fold) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




